Submitted:
01 April 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
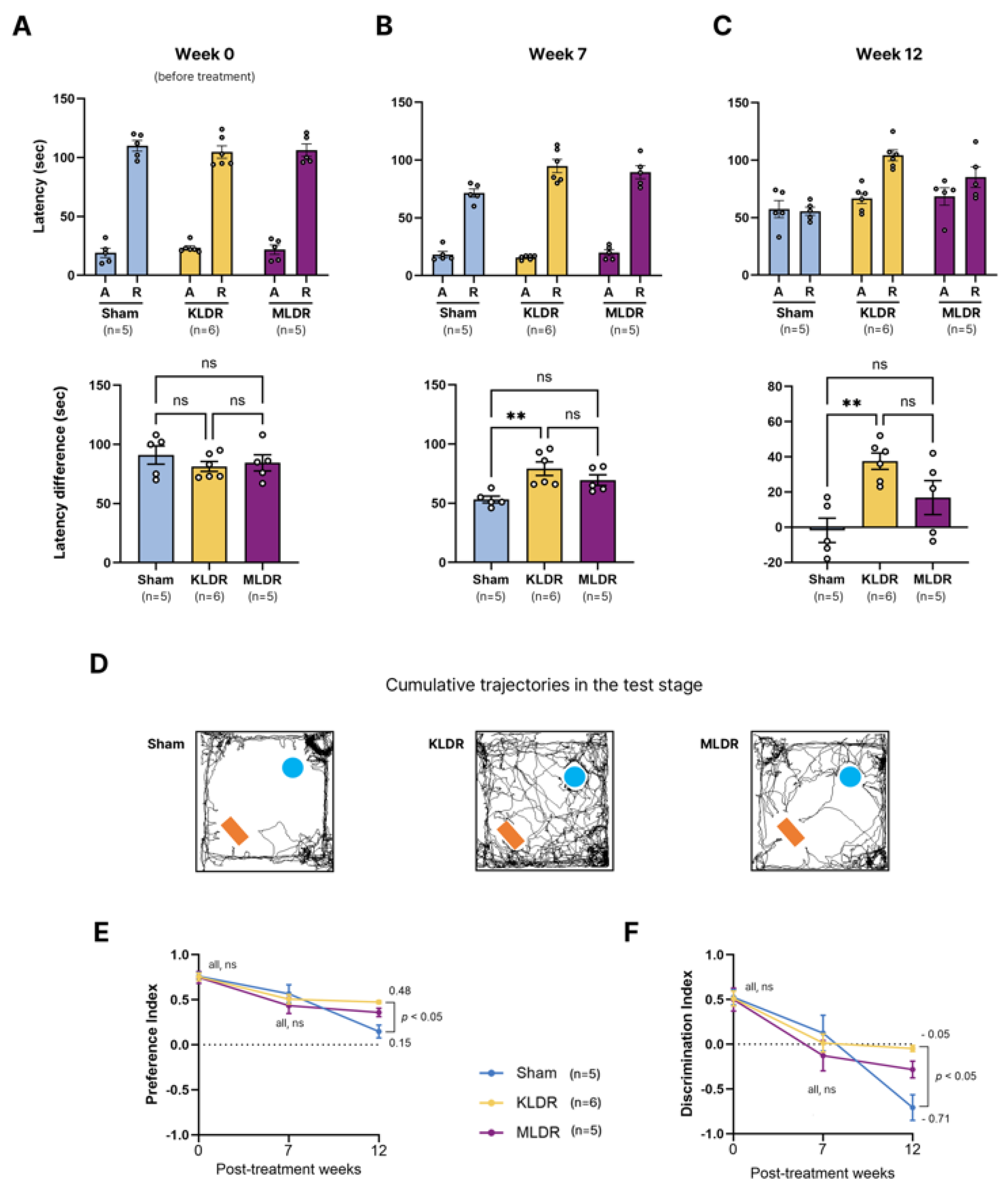
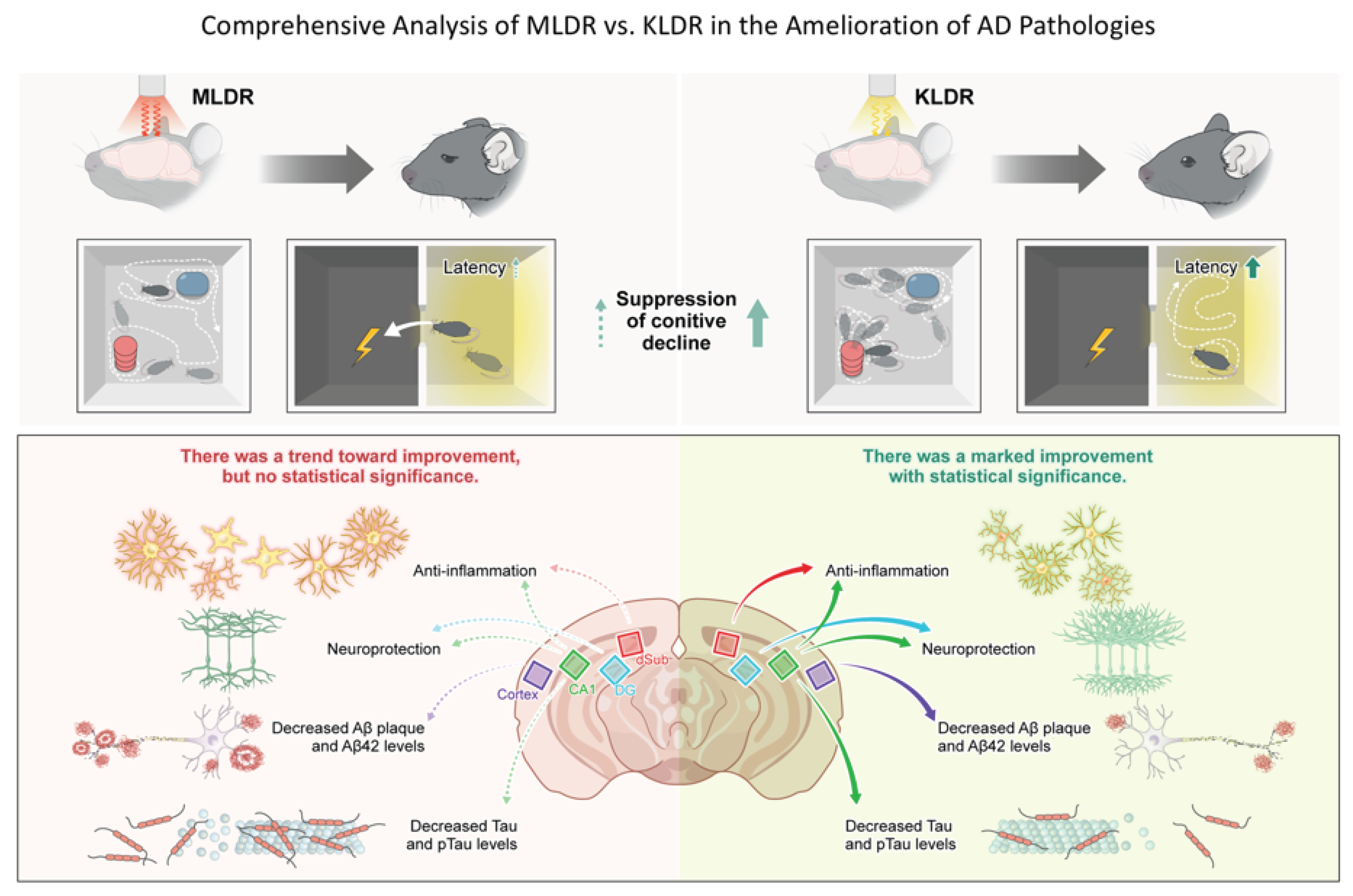
2.1. A Marked Inhibition of Cognitive Decline at Kilovoltage, but not Megavoltage, Energy
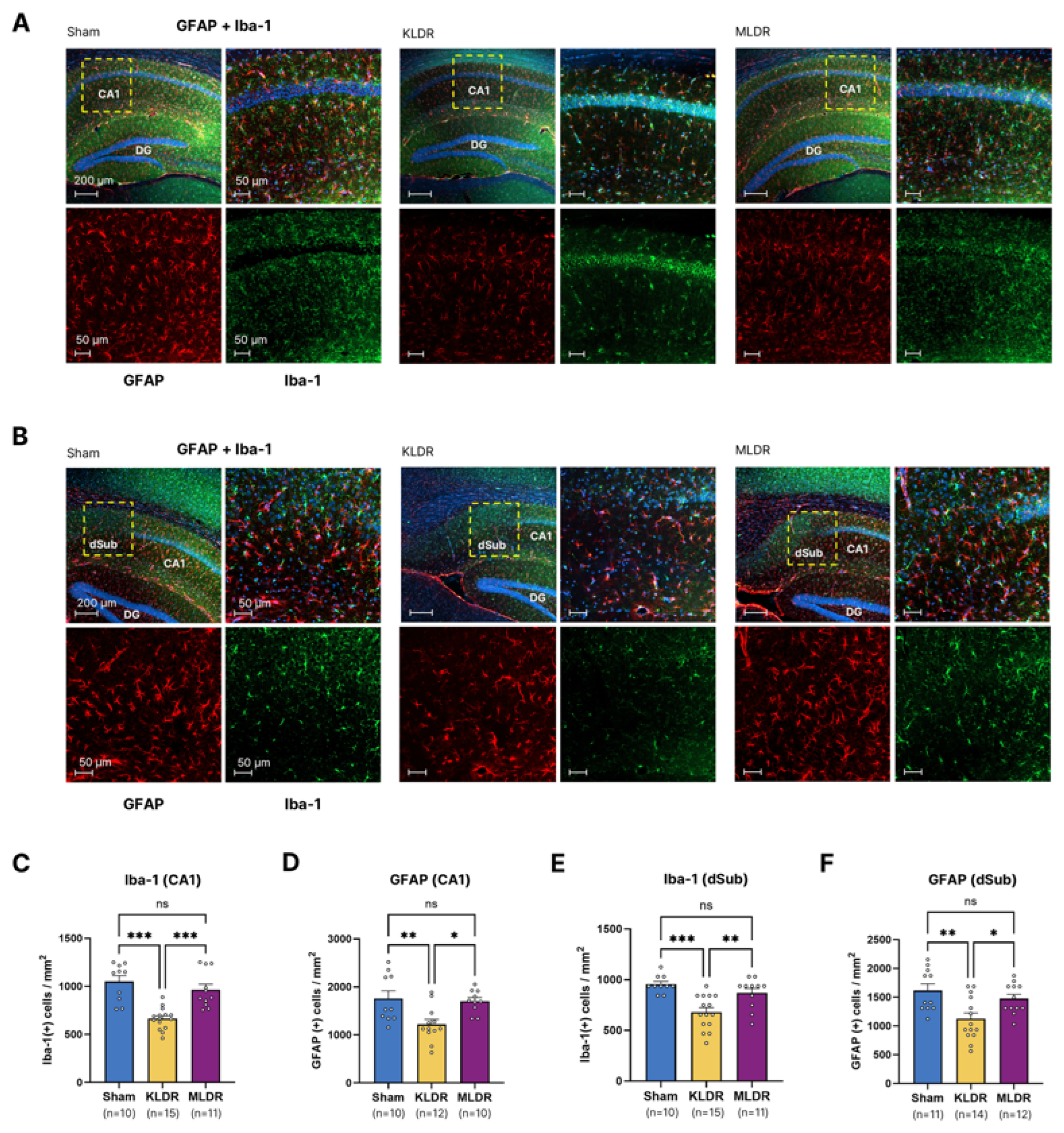
2.2. A Marked Reduction in Inflammatory Marker Levels at Kilovoltage, but not Megavoltage, Energy
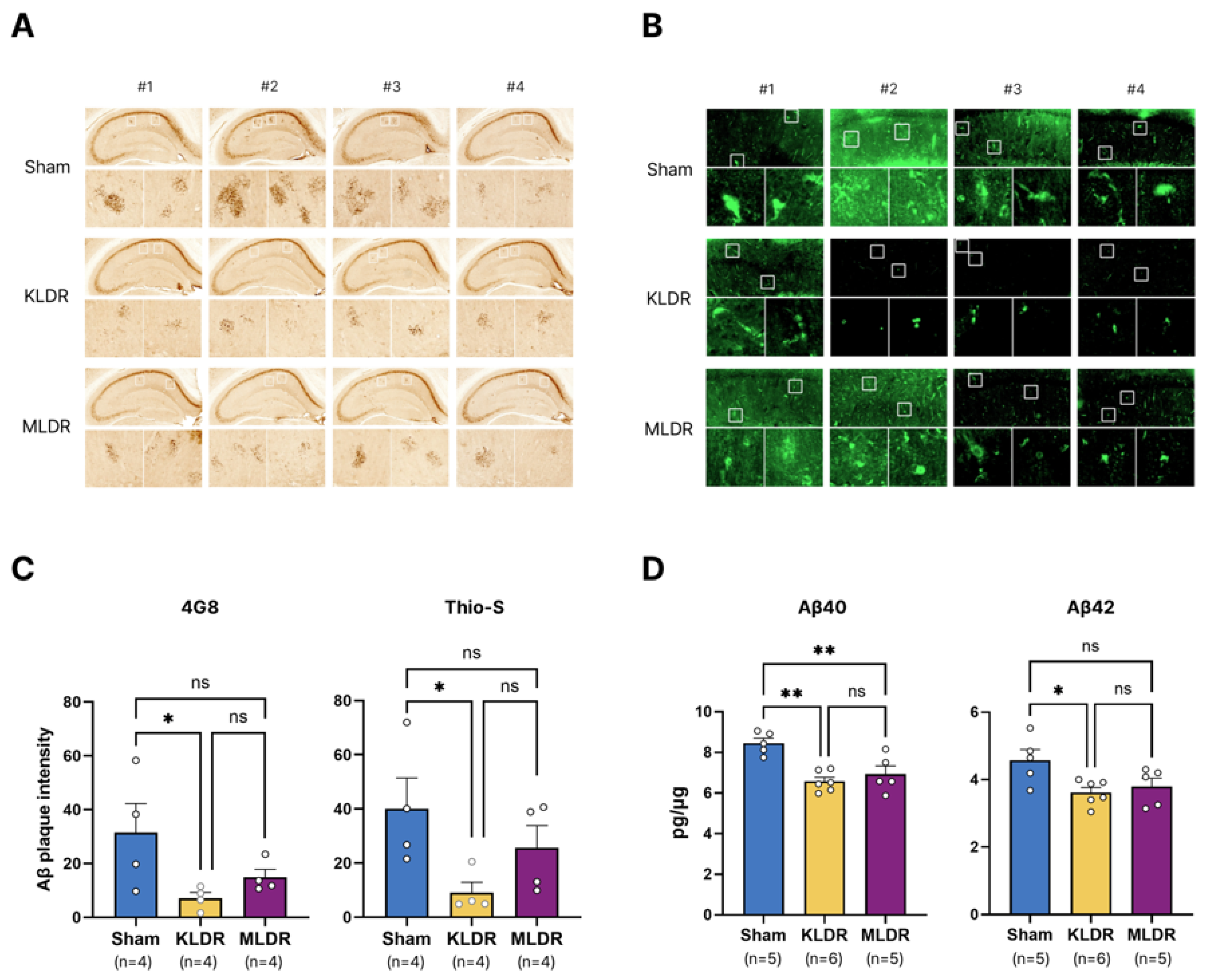
2.3. A Marked Reduction in Aβ Deposition at Kilovoltage, but not Megavoltage, Energy
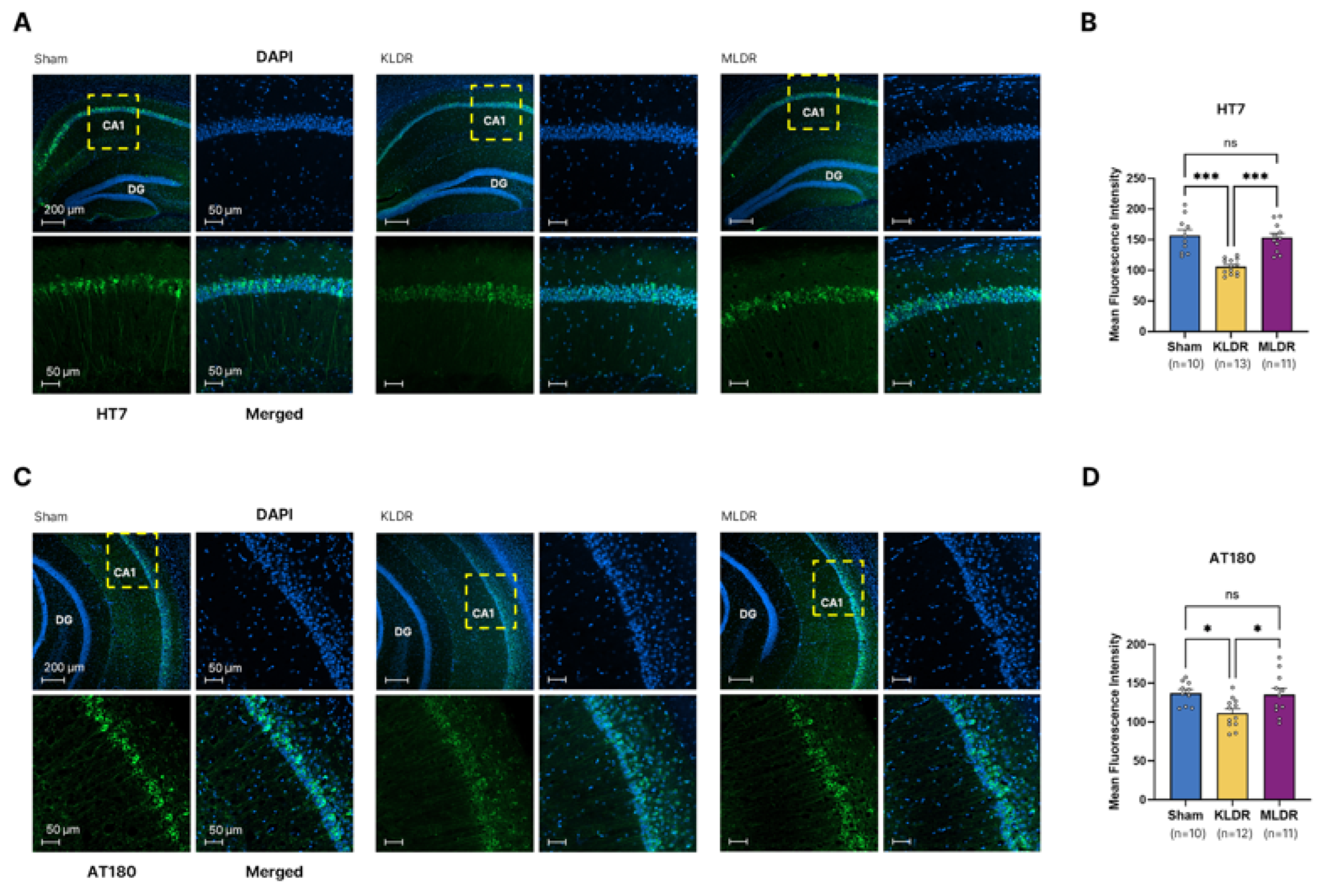
2.4. A Marked Reduction in Tau Deposition at Kilovoltage, but not Megavoltage, Energy
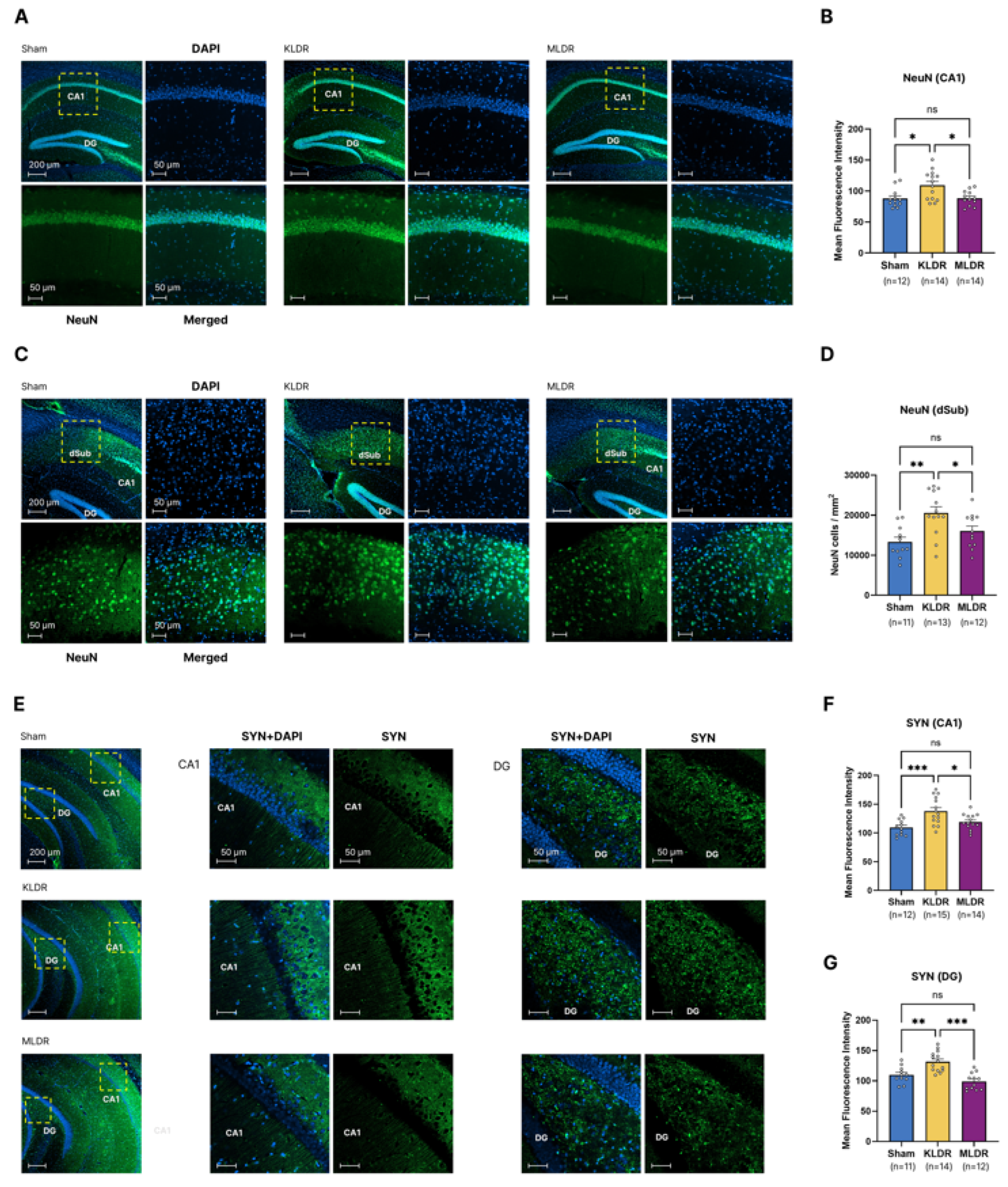
2.5. Significant Inhibition of Neuronal and Synaptic Degeneration at Kilovoltage, but not megavoltage, Energy
2.6. KLDR Shows Multimodal Actions, Improving Typical AD Pathological Features
3. Discussion
4. Materials and Methods
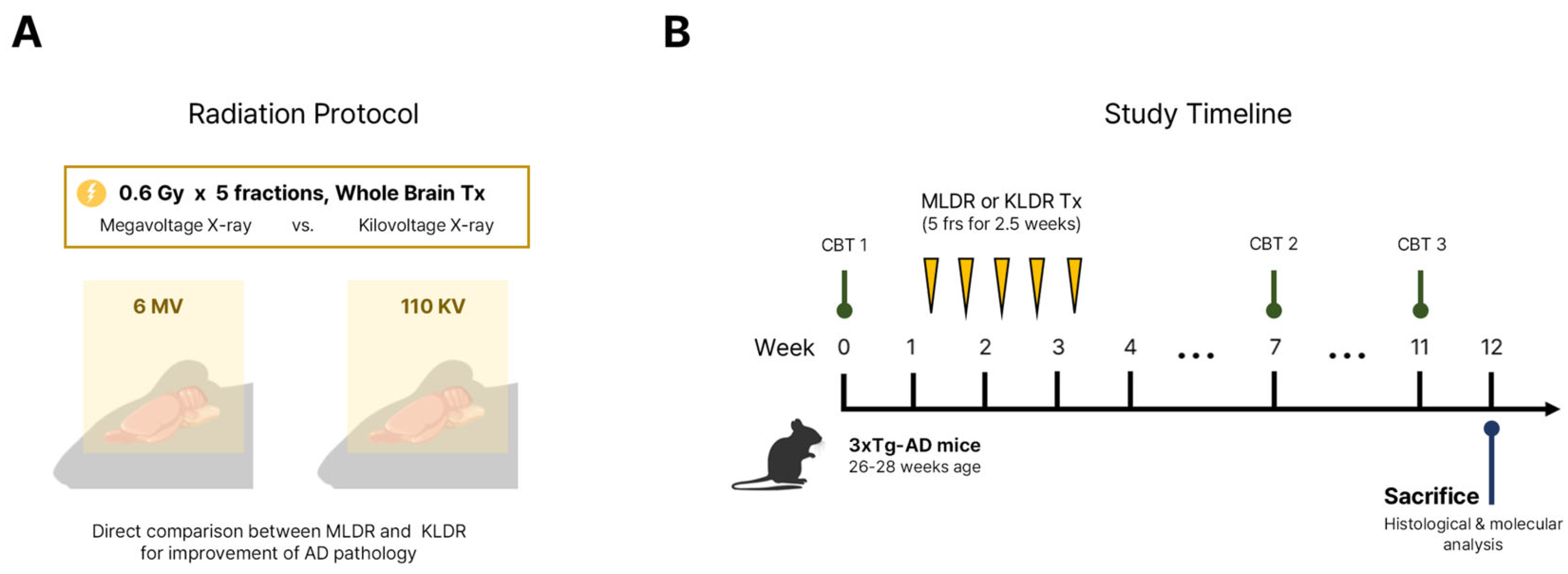
4.1. Animals and Group Assignment
4.2. Cranial LDR Procedure
4.3. Cognitive Behavioral Tests
4.3.1. Passive Avoidance (PA) Test
4.3.2. Novel Object Recognition (NOR) Test
4.4. Brain Tissue Preparation
4.5. Histological Analyses
4.5.1. Immunofluorescence Analysis
4.5.2. Immunohistochemical Analysis
4.5.3. Thioflavin S Staining
4.6. Quantitative Molecular Analyses
4.6.1. Quantitative Real-Time Reverse-Transcription PCR (qRT-PCR)
4.6.2. Enzyme-Linked Immunosorbent Assay (ELISA)
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data availability statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s disease |
| KLDR | kilovoltage low-dose radiation |
| MLDR | megavoltage low-dose radiation |
| LDR | low-dose radiation |
| LDRT | low-dose radiation therapy |
| PCR | polymerase chain reaction |
| ELISA | enzyme-linked immunosorbent assay |
| GFAP | glial fibrillary acidic protein |
| IL | interleukin |
| TNF | tumor necrosis factor |
| LPS | lipopolysaccharide |
References
- Vaiserman, A.; Cuttler, J. M.; Socol, Y. Low-dose ionizing radiation as a hormetin: experimental observations and therapeutic perspective for age-related disorders. Biogerontology 2021, 22((2)), 145–164. [Google Scholar] [CrossRef]
- Baldwin, J.; Grantham, V. Radiation Hormesis: Historical and Current Perspectives. Journal of nuclear medicine technology 2015, 43((4)), 242–6. [Google Scholar] [CrossRef]
- Bevelacqua, J. J.; Mortazavi, S. M. J. Alzheimer ‘s Disease: Possible Mechanisms Behind Neurohormesis Induced by Exposure to Low Doses of Ionizing Radiation. Journal of biomedical physics & engineering 2018, 8((2)), 153–156. [Google Scholar]
- Paithankar, J. G.; Gupta, S. C.; Sharma, A. Therapeutic potential of low dose ionizing radiation against cancer, dementia, and diabetes: evidences from epidemiological, clinical, and preclinical studies. Molecular biology reports 2023, 50((3)), 2823–2834. [Google Scholar] [CrossRef]
- Yin, E.; Nelson, D. O.; Coleman, M. A.; Peterson, L. E.; Wyrobek, A. J. Gene expression changes in mouse brain after exposure to low-dose ionizing radiation. International journal of radiation biology 2003, 79((10)), 759–75. [Google Scholar] [CrossRef]
- Lowe, X. R.; Bhattacharya, S.; Marchetti, F.; Wyrobek, A. J. Early brain response to low-dose radiation exposure involves molecular networks and pathways associated with cognitive functions, advanced aging and Alzheimer’s disease. Radiation research 2009, 171((1)), 53–65. [Google Scholar] [CrossRef] [PubMed]
- Hautmann, M. G.; Rechner, P.; Neumaier, U.; Süß, C.; Dietl, B.; Putz, F. J.; Behr, M.; Kölbl, O.; Steger, F. Radiotherapy for osteoarthritis-an analysis of 295 joints treated with a linear accelerator. Strahlentherapie und Onkologie: Organ der Deutschen Rontgengesellschaft. 2020, 196((8)), 715–724. [Google Scholar] [CrossRef]
- Kim, S.; Chung, H.; Ngoc Mai, H.; Nam, Y.; Shin, S. J.; Park, Y. H.; Chung, M. J.; Lee, J. K.; Rhee, H. Y.; Jahng, G. H.; Kim, Y.; Lim, Y. J.; Kong, M.; Moon, M.; Chung, W. K. Low-Dose Ionizing Radiation Modulates Microglia Phenotypes in the Models of Alzheimer’s Disease. International journal of molecular sciences 2020, 21((12)). [Google Scholar] [CrossRef] [PubMed]
- Boyd, A.; Byrne, S.; Middleton, R. J.; Banati, R. B.; Liu, G. J. Control of Neuroinflammation through Radiation-Induced Microglial Changes. Cells 2021, 10((9)). [Google Scholar] [CrossRef] [PubMed]
- Gao, C.; Jiang, J.; Tan, Y.; Chen, S. Microglia in neurodegenerative diseases: mechanism and potential therapeutic targets. Signal transduction and targeted therapy 2023, 8((1)), 359. [Google Scholar] [CrossRef]
- Kim, S.; Nam, Y.; Kim, C.; Lee, H.; Hong, S.; Kim, H. S.; Shin, S. J.; Park, Y. H.; Mai, H. N.; Oh, S. M.; Kim, K. S.; Yoo, D. H.; Chung, W. K.; Chung, H.; Moon, M. Neuroprotective and Anti-Inflammatory Effects of Low-Moderate Dose Ionizing Radiation in Models of Alzheimer’s Disease. International journal of molecular sciences 2020, 21((10)). [Google Scholar] [CrossRef]
- Cuttler, J. M.; Moore, E. R.; Hosfeld, V. D.; Nadolski, D. L. Treatment of Alzheimer Disease With CT Scans: A Case Report. Dose-response: a publication of International Hormesis Society 2016, 14((2)), 1559325816640073. [Google Scholar] [CrossRef]
- Cuttler, J. M.; Abdellah, E.; Goldberg, Y.; Al-Shamaa, S.; Symons, S. P.; Black, S. E.; Freedman, M. Low Doses of Ionizing Radiation as a Treatment for Alzheimer’s Disease: A Pilot Study. Journal of Alzheimer’s disease: JAD 2021, 80((3)), 1119–1128. [Google Scholar] [CrossRef]
- Rogers, C. L.; Lageman, S. K.; Fontanesi, J.; Wilson, G. D.; Boling, P. A.; Bansal, S.; Karis, J. P.; Sabbagh, M.; Mehta, M. P.; Harris, T. J. Low-Dose Whole Brain Radiation Therapy for Alzheimer’s Dementia: Results From a Pilot Trial in Humans. International journal of radiation oncology, biology, physics 2023, 117((1)), 87–95. [Google Scholar] [CrossRef]
- Ceyzériat, K.; Zilli, T.; Fall, A. B.; Millet, P.; Koutsouvelis, N.; Dipasquale, G.; Frisoni, G. B.; Tournier, B. B.; Garibotto, V. Treatment by low-dose brain radiation therapy improves memory performances without changes of the amyloid load in the TgF344-AD rat model. Neurobiology of aging 2021, 103, 117–127. [Google Scholar] [CrossRef]
- Ceyzériat, K.; Tournier, B. B.; Millet, P.; Dipasquale, G.; Koutsouvelis, N.; Frisoni, G. B.; Garibotto, V.; Zilli, T. Low-Dose Radiation Therapy Reduces Amyloid Load in Young 3xTg-AD Mice. Journal of Alzheimer’s disease: JAD 2022, 86((2)), 641–653. [Google Scholar] [CrossRef]
- Linnerbauer, M.; Wheeler, M. A.; Quintana, F. J. Astrocyte Crosstalk in CNS Inflammation. Neuron 2020, 108((4)), 608–622. [Google Scholar] [CrossRef]
- Leng, F.; Edison, P. Neuroinflammation and microglial activation in Alzheimer disease: where do we go from here? Nature reviews. Neurology 2021, 17((3)), 157–172. [Google Scholar] [PubMed]
- Hur, J. Y.; Frost, G. R.; Wu, X.; Crump, C.; Pan, S. J.; Wong, E.; Barros, M.; Li, T.; Nie, P.; Zhai, Y.; Wang, J. C.; Tcw, J.; Guo, L.; McKenzie, A.; Ming, C.; Zhou, X.; Wang, M.; Sagi, Y.; Renton, A. E.; Esposito, B. T.; Kim, Y.; Sadleir, K. R.; Trinh, I.; Rissman, R. A.; Vassar, R.; Zhang, B.; Johnson, D. S.; Masliah, E.; Greengard, P.; Goate, A.; Li, Y. M. The innate immunity protein IFITM3 modulates γ-secretase in Alzheimer’s disease. Nature 2020, 586((7831)), 735–740. [Google Scholar] [CrossRef] [PubMed]
- Hur, J. Y. Innate Immunity Protein IFITM3 in Alzheimer’s Disease. DNA and cell biology 2021, 40((11)), 1351–1355. [Google Scholar] [CrossRef] [PubMed]
- Friedlová, N.; Zavadil Kokáš, F.; Hupp, T. R.; Vojtěšek, B.; Nekulová, M. IFITM protein regulation and functions: Far beyond the fight against viruses. Frontiers in immunology 2022, 13, 1042368. [Google Scholar] [CrossRef]
- Son, Y.; Lee, C. G.; Kim, J. S.; Lee, H. J. Low-dose-rate ionizing radiation affects innate immunity protein IFITM3 in a mouse model of Alzheimer’s disease. International journal of radiation biology 2023, 99((11)), 1649–1659. [Google Scholar] [CrossRef]
- Ismail, R.; Parbo, P.; Madsen, L. S.; Hansen, A. K.; Hansen, K. V.; Schaldemose, J. L.; Kjeldsen, P. L.; Stokholm, M. G.; Gottrup, H.; Eskildsen, S. F.; Brooks, D. J. The relationships between neuroinflammation, beta-amyloid and tau deposition in Alzheimer’s disease: a longitudinal PET study. J Neuroinflammation 2020, 17((1)), 151. [Google Scholar] [CrossRef]
- Wu, H. Y.; Kuo, P. C.; Wang, Y. T.; Lin, H. T.; Roe, A. D.; Wang, B. Y.; Han, C. L.; Hyman, B. T.; Chen, Y. J.; Tai, H. C. β-Amyloid Induces Pathology-Related Patterns of Tau Hyperphosphorylation at Synaptic Terminals. Journal of neuropathology and experimental neurology 2018, 77((9)), 814–826. [Google Scholar] [CrossRef] [PubMed]
- Duyckaerts, C.; Delatour, B.; Potier, M. C. Classification and basic pathology of Alzheimer disease. Acta neuropathologica 2009, 118((1)), 5–36. [Google Scholar] [CrossRef] [PubMed]
- Kent, S. A.; Spires-Jones, T. L.; Durrant, C. S. The physiological roles of tau and Aβ: implications for Alzheimer’s disease pathology and therapeutics. Acta neuropathologica 2020, 140((4)), 417–447. [Google Scholar] [CrossRef] [PubMed]
- Hampel, H.; Hardy, J.; Blennow, K.; Chen, C.; Perry, G.; Kim, S. H.; Villemagne, V. L.; Aisen, P.; Vendruscolo, M.; Iwatsubo, T.; Masters, C. L.; Cho, M.; Lannfelt, L.; Cummings, J. L.; Vergallo, A. The Amyloid-β Pathway in Alzheimer’s Disease. Molecular psychiatry 2021, 26((10)), 5481–5503. [Google Scholar] [CrossRef]
- LaFerla, F. M.; Green, K. N.; Oddo, S. Intracellular amyloid-beta in Alzheimer’s disease. Nature reviews. Neuroscience 2007, 8((7)), 499–509. [Google Scholar] [CrossRef]
- Ji, L.; Zhao, X.; Lu, W.; Zhang, Q.; Hua, Z. Intracellular Aβ and its Pathological Role in Alzheimer’s Disease: Lessons from Cellular to Animal Models. Current Alzheimer research 2016, 13((6)), 621–30. [Google Scholar] [CrossRef]
- Belley, M. D.; Ashcraft, K. A.; Lee, C. T.; Cornwall-Brady, M. R.; Chen, J. J.; Gunasingha, R.; Burkhart, M.; Dewhirst, M.; Yoshizumi, T. T.; Down, J. D. Microdosimetric and Biological Effects of Photon Irradiation at Different Energies in Bone Marrow. Radiation research 2015, 184((4)), 378–91. [Google Scholar] [CrossRef]
- Bell, B. I.; Vercellino, J.; Brodin, N. P.; Velten, C.; Nanduri, L. S. Y.; Nagesh, P. K. B.; Tanaka, K. E.; Fang, Y.; Wang, Y.; Macedo, R.; English, J.; Schumacher, M. M.; Duddempudi, P. K.; Asp, P.; Koba, W.; Shajahan, S.; Liu, L.; Tomé, W. A.; Yang, W. L.; Kolesnick, R.; Guha, C. Orthovoltage X-Rays Exhibit Increased Efficacy Compared with γ-Rays in Preclinical Irradiation. Cancer research 2022, 82((15)), 2678–2691. [Google Scholar] [CrossRef] [PubMed]
- Amols, H. I.; Lagueux, B.; Cagna, D. Radiobiological effectiveness (RBE) of megavoltage X-ray and electron beams in radiotherapy. Radiation research 1986, 105((1)), 58–67. [Google Scholar] [CrossRef]
- Pattanayak, A.; Firdous, S. M. The 3xTg-AD Mouse Model: A Comprehensive Tool for Understanding Alzheimer’s Disease. Cellular and molecular neurobiology 2026, 46((1)), 41. [Google Scholar] [CrossRef]
- Drummond, E.; Wisniewski, T. Alzheimer’s disease: experimental models and reality. Acta neuropathologica 2017, 133((2)), 155–175. [Google Scholar] [CrossRef] [PubMed]
- Javonillo, D. I.; Tran, K. M.; Phan, J.; Hingco, E.; Kramár, E. A.; da Cunha, C.; Forner, S.; Kawauchi, S.; Milinkeviciute, G.; Gomez-Arboledas, A.; Neumann, J.; Banh, C. E.; Huynh, M.; Matheos, D. P.; Rezaie, N.; Alcantara, J. A.; Mortazavi, A.; Wood, M. A.; Tenner, A. J.; MacGregor, G. R.; Green, K. N.; LaFerla, F. M. Systematic Phenotyping and Characterization of the 3xTg-AD Mouse Model of Alzheimer’s Disease. Front Neurosci 2021, 15, 785276. [Google Scholar] [CrossRef] [PubMed]
- Kamatham, P. T.; Shukla, R.; Khatri, D. K.; Vora, L. K. Pathogenesis, diagnostics, and therapeutics for Alzheimer’s disease: Breaking the memory barrier. Ageing research reviews 2024, 101, 102481. [Google Scholar] [CrossRef]
- DeTure, M. A.; Dickson, D. W. The neuropathological diagnosis of Alzheimer’s disease. Molecular neurodegeneration 2019, 14((1)), 32. [Google Scholar] [CrossRef]
- Ceyzériat, K.; Zilli, T.; Millet, P.; Koutsouvelis, N.; Dipasquale, G.; Fossey, C.; Cailly, T.; Fabis, F.; Frisoni, G. B.; Garibotto, V.; Tournier, B. B. Low-dose brain irradiation normalizes TSPO and CLUSTERIN levels and promotes the non-amyloidogenic pathway in pre-symptomatic TgF344-AD rats. Journal of neuroinflammation 2022, 19((1)), 311. [Google Scholar] [CrossRef]
- Khan, A.; Sati, J.; Kamal, R.; Dhawan, D.K.; Chadha, V.D. Amelioration of cognitive and biochemical impairment in Aβ-based rodent model of Alzheimer’s disease following fractionated X-irradiation. Radiation and environmental biophysics 2022, 61((2)), 205–219. [Google Scholar] [CrossRef]
- Marples, B.; McGee, M.; Callan, S.; Bowen, S. E.; Thibodeau, B. J.; Michael, D. B.; Wilson, G. D.; Maddens, M. E.; Fontanesi, J.; Martinez, A. A. Cranial irradiation significantly reduces beta amyloid plaques in the brain and improves cognition in a murine model of Alzheimer’s Disease (AD). Radiotherapy and oncology: journal of the European Society for Therapeutic Radiology and Oncology 2016, 118((1)), 43–51. [Google Scholar] [CrossRef]
- Wilson, G. D.; Rogers, C. L.; Mehta, M. P.; Marples, B.; Michael, D. B.; Welsh, J. S.; Martinez, A. A.; Fontanesi, J. The Rationale for Radiation Therapy in Alzheimer’s Disease. Radiation research 2023, 199((5)), 506–516. [Google Scholar] [CrossRef]
- Yang, E. J.; Kim, H.; Choi, Y.; Kim, H. J.; Kim, J. H.; Yoon, J.; Seo, Y. S.; Kim, H. S. Modulation of Neuroinflammation by Low-Dose Radiation Therapy in an Animal Model of Alzheimer’s Disease. International journal of radiation oncology, biology, physics 2021, 111((3)), 658–670. [Google Scholar] [CrossRef]
- Wilson, G. D.; Wilson, T. G.; Hanna, A.; Fontanesi, G.; Kulchycki, J.; Buelow, K.; Pruetz, B. L.; Michael, D. B.; Chinnaiyan, P.; Maddens, M. E.; Martinez, A. A.; Fontanesi, J. Low Dose Brain Irradiation Reduces Amyloid-β and Tau in 3xTg-AD Mice. Journal of Alzheimer’s disease: JAD 2020, 75((1)), 15–21. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Lee, J.; Moon, H.; Kim, C.; Yoo, M. Y.; Park, W. Y.; Kim, W. D.; Seo, Y. S. The effects of low-dose radiation therapy in patients with mild-to-moderate Alzheimer’s dementia: an interim analysis of a pilot study. Radiation oncology journal 2023, 41((2)), 89–97. [Google Scholar] [CrossRef] [PubMed]
- Koneru, B. N.; Sick, J.; Shaikh, H. A.; Spengler, H.; Small, W., Jr.; Shaffer, R. Low-Dose Radiation Therapy for Osteoarthritis: A Retrospective Single-Institution Analysis of 69 Patients and 168 Joints. International journal of radiation oncology, biology, physics 2025, 123((2)), 352–360. [Google Scholar] [CrossRef]
- Faiz, M.; Khan, J.P.G. The Physics of Radiation Therapy, 5th ed.; 2014; pp. 546–547. [Google Scholar]
- Lillie, E. M.; Urban, J. E.; Lynch, S. K.; Weaver, A. A.; Stitzel, J. D. Evaluation of Skull Cortical Thickness Changes With Age and Sex From Computed Tomography Scans. Journal of bone and mineral research: the official journal of the American Society for Bone and Mineral Research 2016, 31((2)), 299–307. [Google Scholar] [CrossRef]
- Kurkinen, M. Lecanemab (Leqembi) is not the right drug for patients with Alzheimer’s disease. Advances in clinical and experimental medicine: official organ Wroclaw Medical University 2023, 32((9)), 943–947. [Google Scholar] [CrossRef]
- Hall, E. J. Radiation dose-rate: a factor of importance in radiobiology and radiotherapy. The British journal of radiology 1972, 45((530)), 81–97. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



