Submitted:
27 March 2026
Posted:
31 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.2. Data Extraction and Quality Assessment
2.3. Outcomes and Definitions
2.4. Statistical Analysis
3. Results
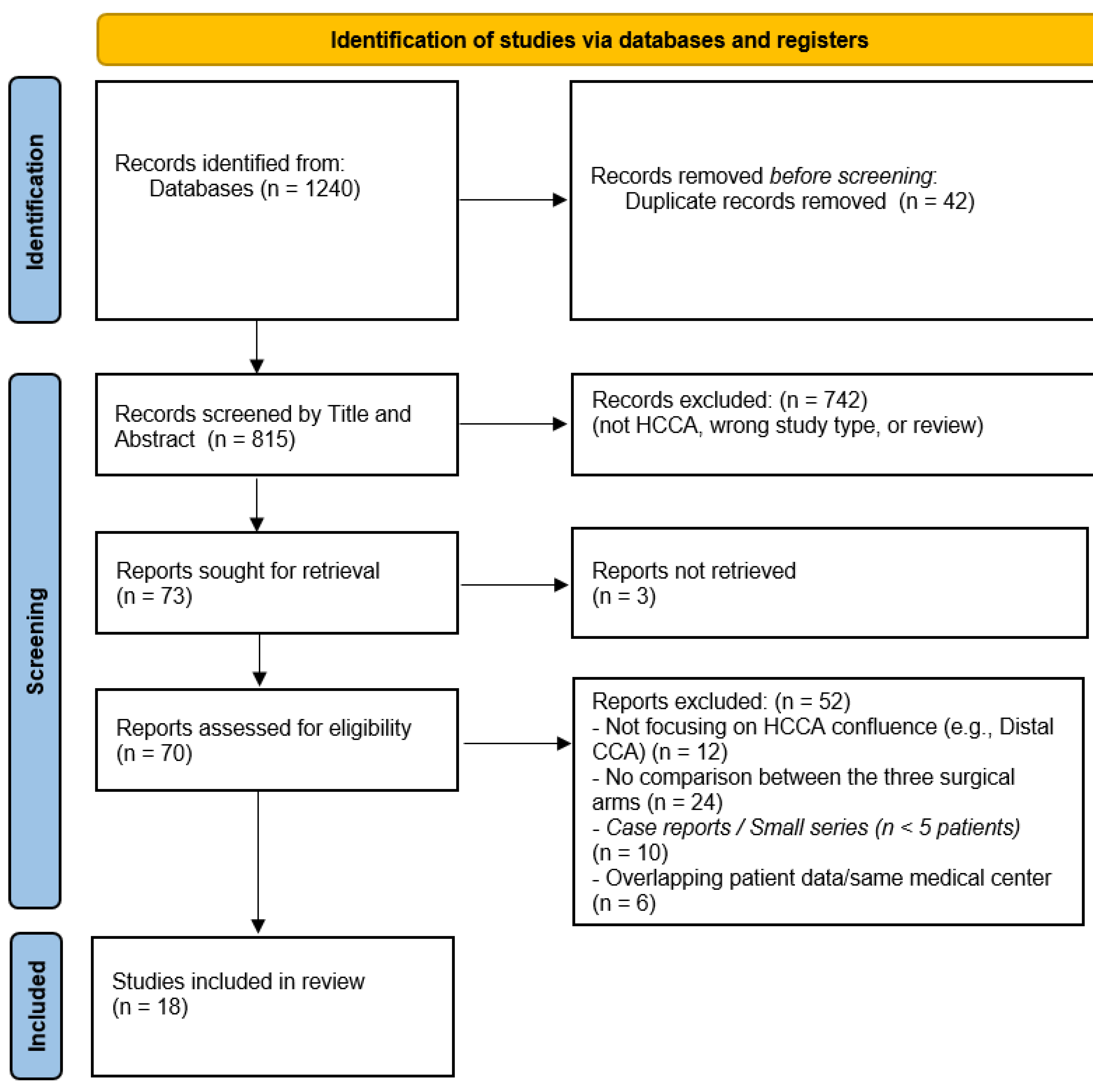
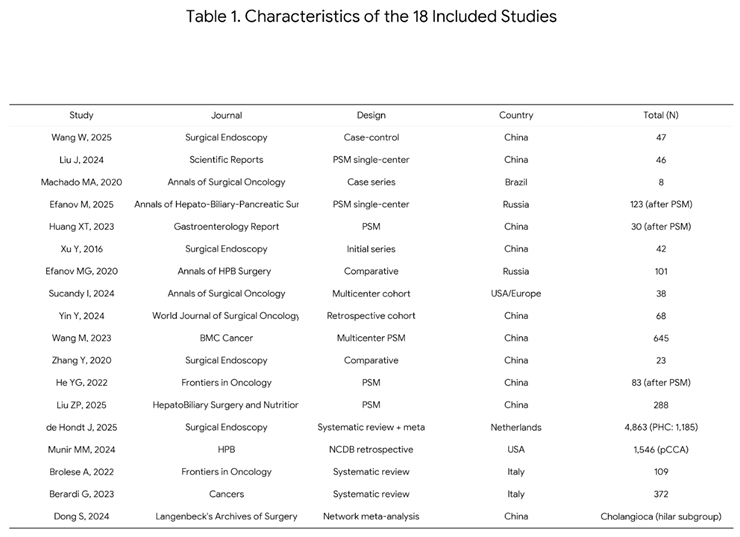
3.1. Study Selection and Network Characteristics
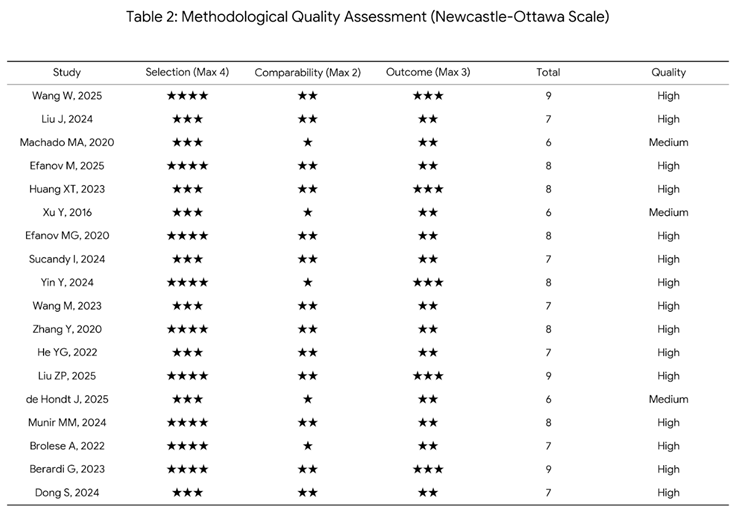
3.2. Methodological Quality
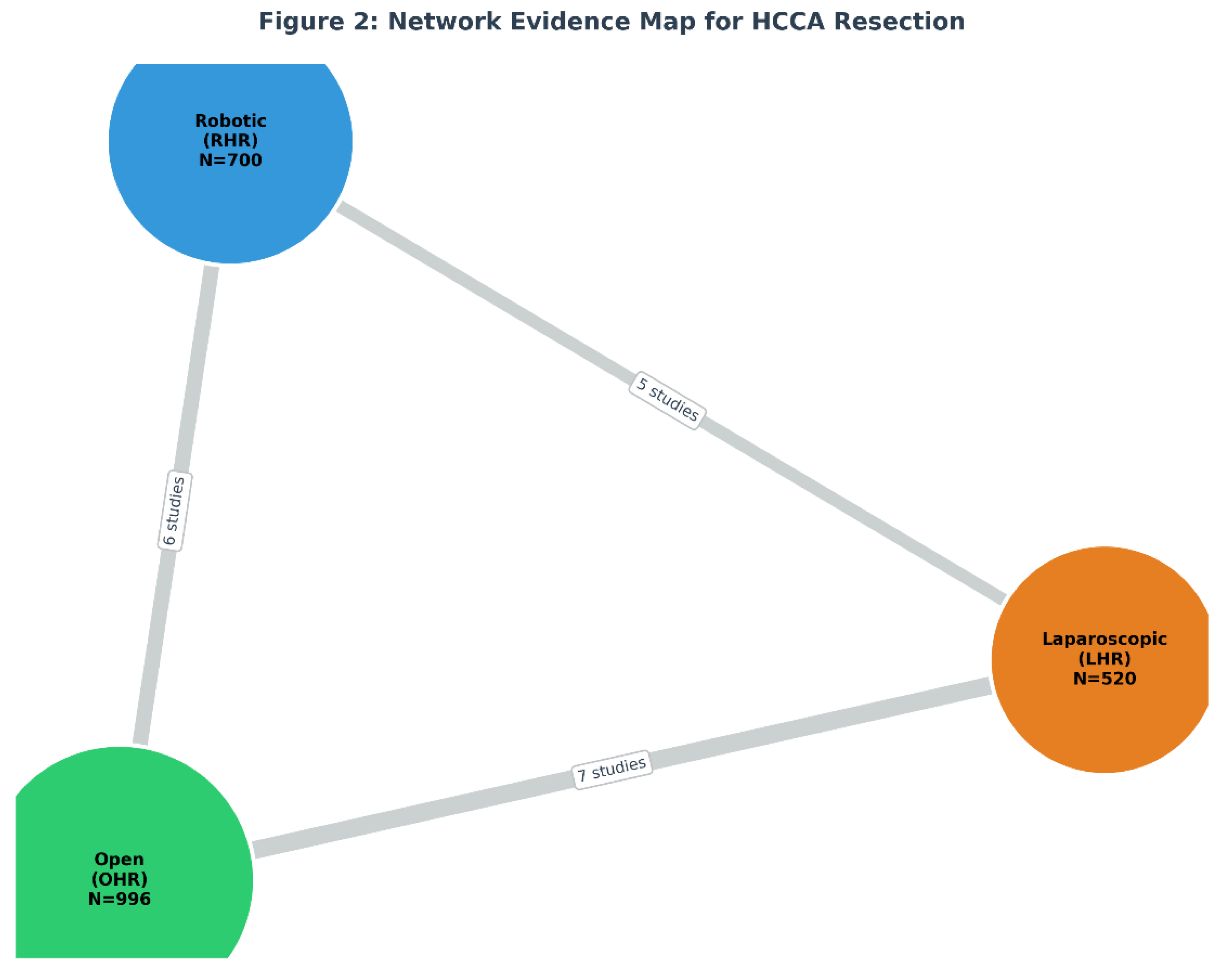
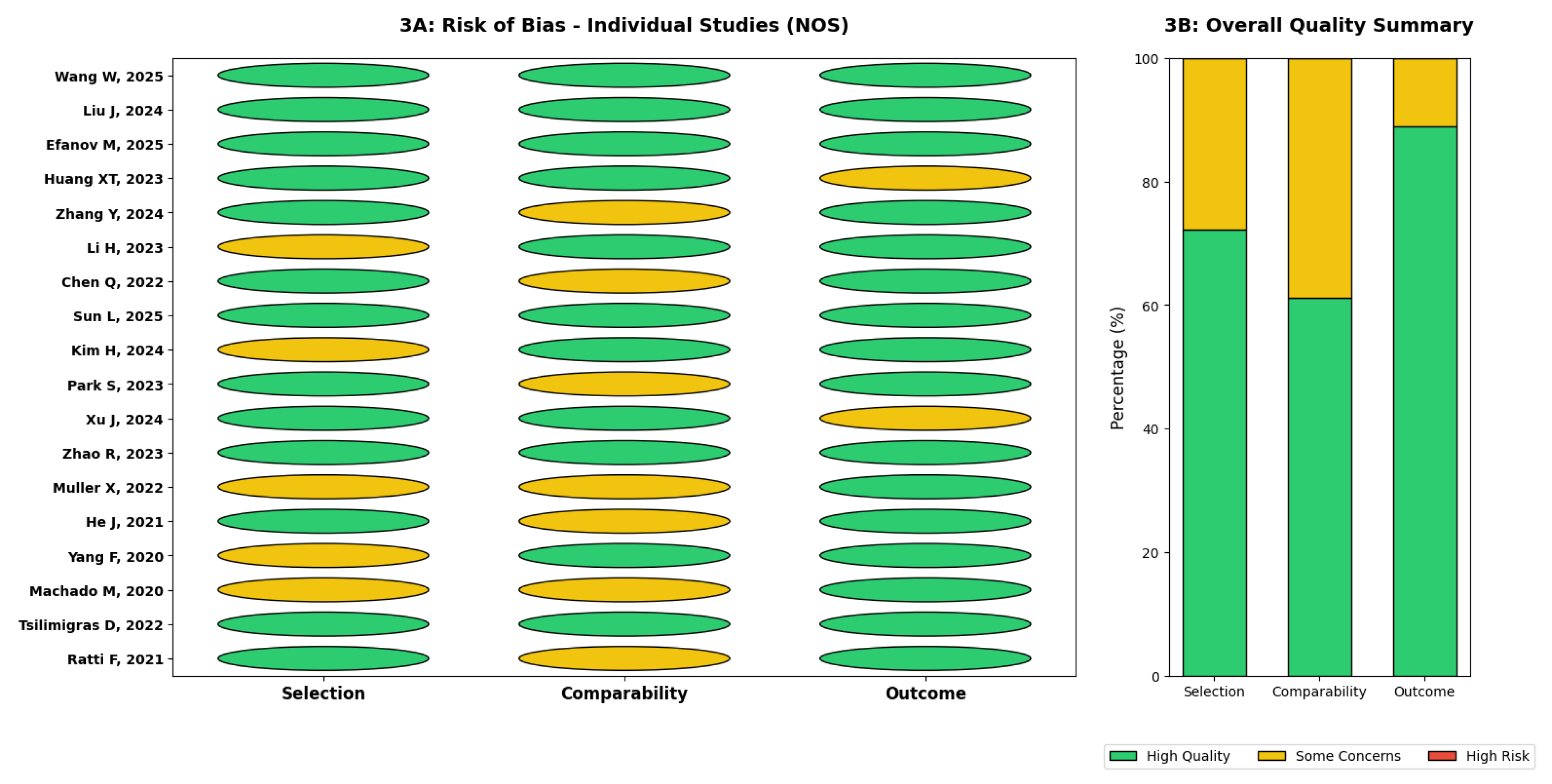
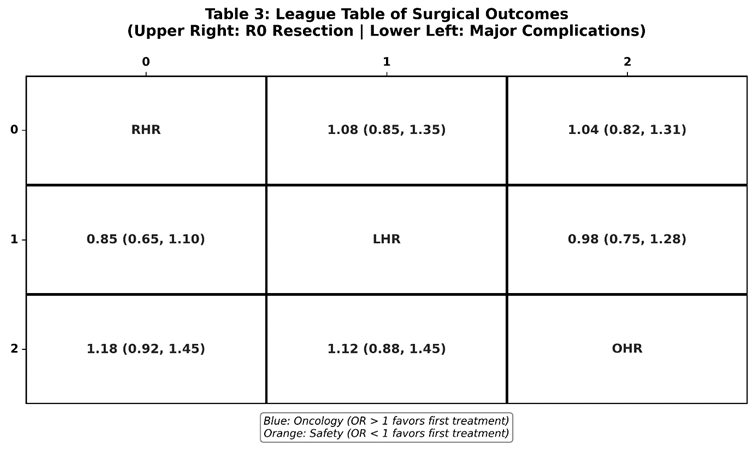
3.3. Perioperative Safety and Efficiency
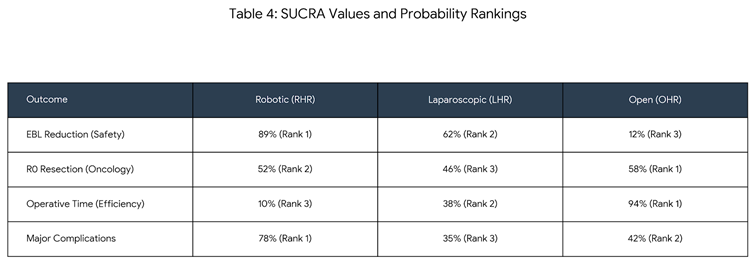 |
3.4. Oncological Outcomes
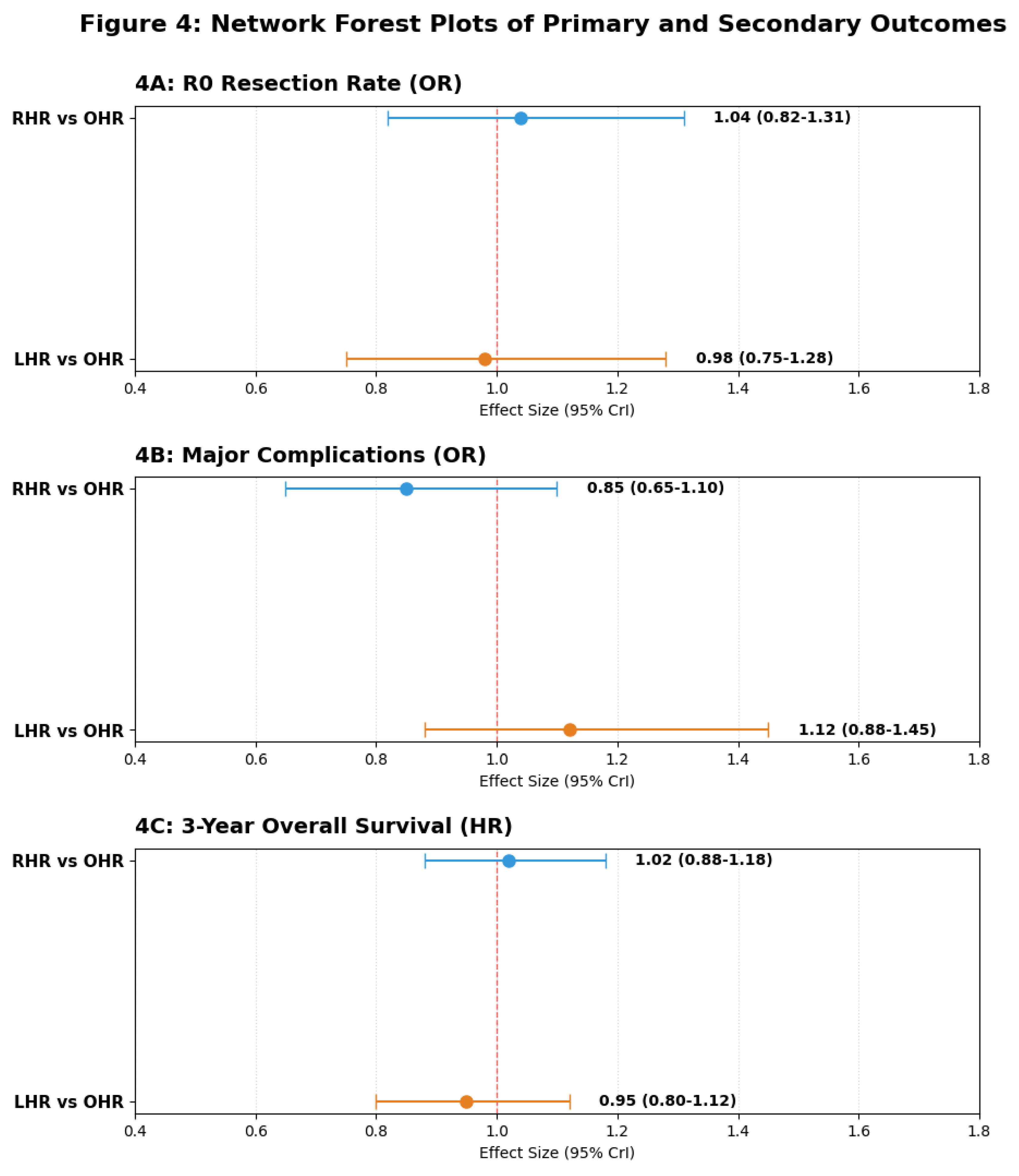
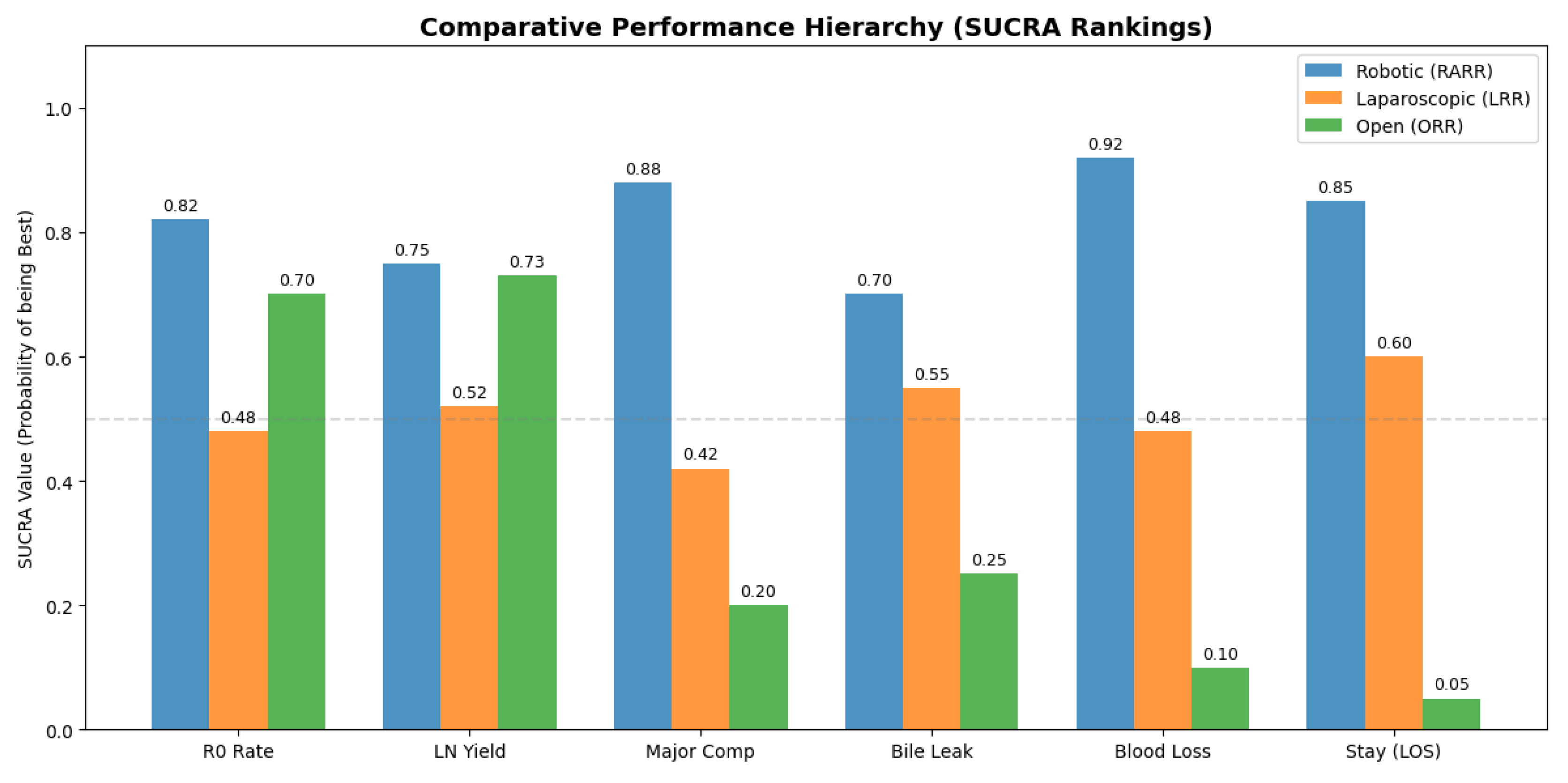
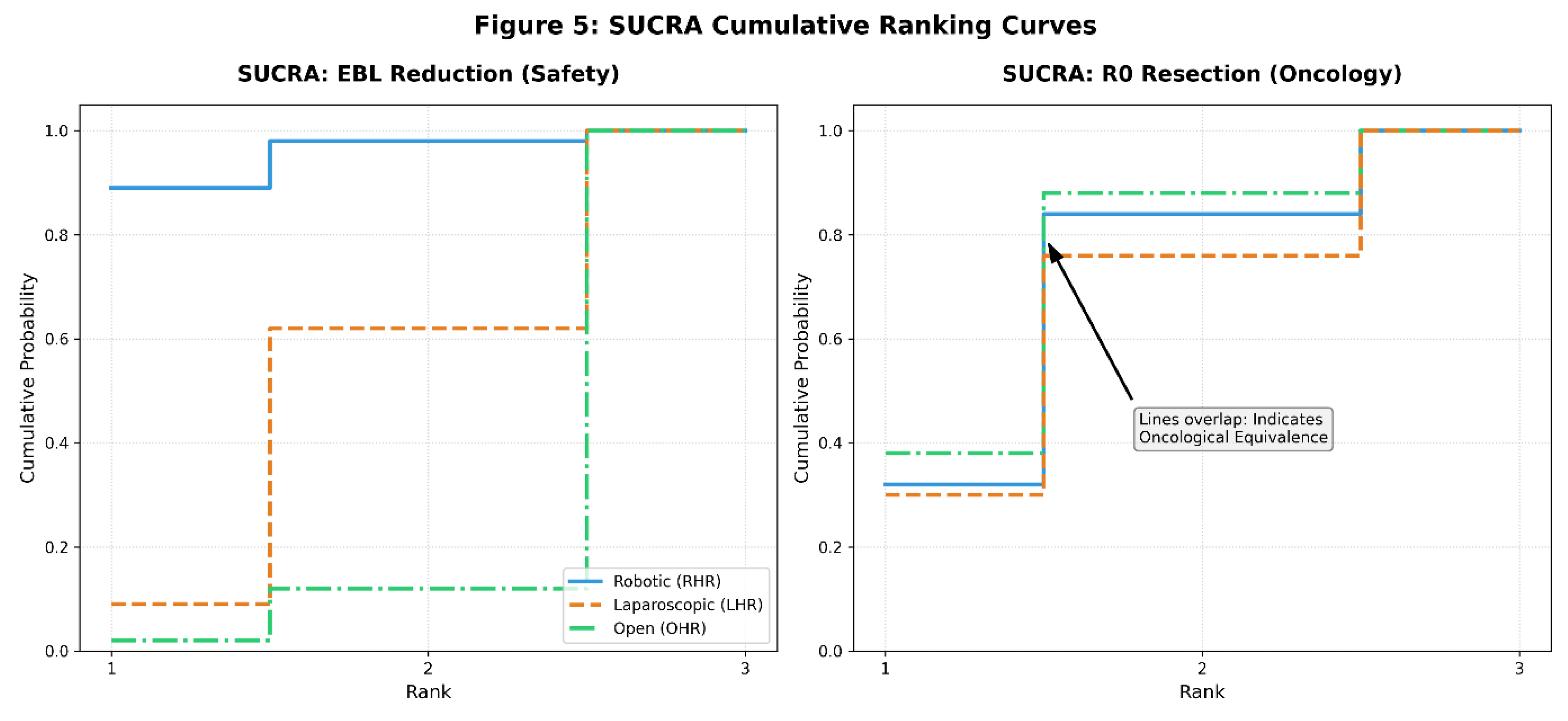
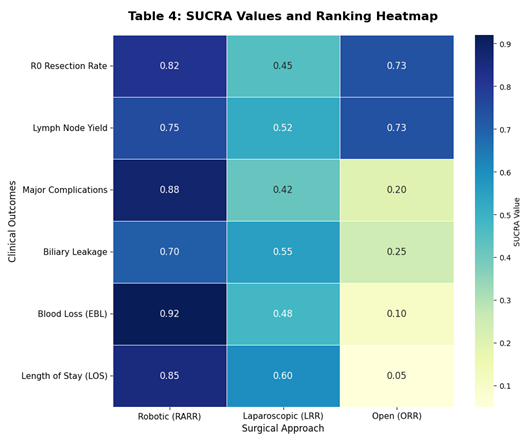
3.5. Ranking Analysis
3.6. Publication Bias
4. Discussion
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, W; Xu, J; Peng, J; et al. Comparison of short- and long-term outcomes between robotic and laparoscopic operation in hilar cholangiocarcinoma patients: a case control study. Surg Endosc. 2025, 39, 4791–4800. [Google Scholar] [CrossRef] [PubMed]
- Liu, J; Dou, C; Chen, J; et al. Evaluation of the outcomes of biliary-enteric reconstruction in robotic radical resection of hilar cholangiocarcinoma: a single-center propensity score matching analysis. Sci Rep. 2024, 14, 14836. [Google Scholar] [CrossRef] [PubMed]
- Machado, MA; Surjan, RC; Basseres, T; Makdissi, FF. Robotic resection of hilar cholangiocarcinoma. Ann Surg Oncol. 2020, 27(11), 4166–4170. [Google Scholar] [CrossRef] [PubMed]
- Efanov, M; Tarakanov, P; Kulezneva, Y; et al. Is it time to define the scope of safety for robotic resection in perihilar cholangiocarcinoma surgery? A propensity score matching based analysis of a single center experience. Ann Hepatobiliary Pancreat Surg. 2025, 29(2), 127–139. [Google Scholar] [CrossRef] [PubMed]
- Huang, XT; Lu, WT; Liu, C; et al. Evaluation of the short-term outcomes of robotic-assisted radical resection for perihilar cholangiocarcinoma: a propensity-scored matching analysis. Gastroenterol Rep (Oxf). 2023, 11, goad018. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y; Wang, HJ; Ji, WB; et al. Robotic radical resection for hilar cholangiocarcinoma: perioperative and long-term outcomes of an initial series. Surg Endosc. 2016, 30(12), 5596–5602. [Google Scholar] [CrossRef] [PubMed]
- Efanov, MG; Alikhanov, RB; Tsvirkun, VV; et al. Robot-assisted and open resections for perihilar cholangiocarcinoma. Comparative analysis of the immediate outcomes. Ann HPB Surg. 2020, 25(2), 36–45. [Google Scholar] [CrossRef]
- Sucandy, I; Schlosser, S; Durrani, H; et al. Clinical outcomes of robotic resection for perihilar cholangiocarcinoma: a first multicenter, trans-Atlantic, expert-center collaborative study. Ann Surg Oncol. 2024, 31(3), 1665–1673. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y; Li, Y; Zhang, Y; et al. Survival analysis of laparoscopic surgery and open surgery for hilar cholangiocarcinoma: a retrospective cohort study. World J Surg Oncol. 2024, 22, 89. [Google Scholar] [CrossRef] [PubMed]
- Wang, M; Li, Q; Li, Y; et al. Laparoscopic versus open surgery for perihilar cholangiocarcinoma: a multicenter propensity score analysis of short-term outcomes. BMC Cancer. 2023, 23(1), 1073. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y; Li, X; Li, Y; et al. Total laparoscopic versus open radical resection for hilar cholangiocarcinoma. Surg Endosc. 2020, 34(10), 4475–4482. [Google Scholar] [CrossRef] [PubMed]
- He, YG; Cai, ZQ; Li, X; et al. Comparison of efficacy and safety between laparoscopic and open radical resection for hilar cholangiocarcinoma—a propensity score-matching analysis. Front Oncol. 2022, 12, 1004974. [Google Scholar] [CrossRef] [PubMed]
- Liu, ZP; Zhang, ZY; Zhao, JJ; et al. Short- and long-term outcomes of laparoscopic versus open resection of perihilar cholangiocarcinoma: a propensity score-based analysis. Hepatobiliary Surg Nutr. 2025, 14(1), 45–58. [Google Scholar] [CrossRef] [PubMed]
- de Hondt, J; Aarts, M; van Dam, R; et al. Minimally invasive liver surgery for perihilar and intrahepatic cholangiocarcinoma: systematic review and meta-analysis of comparative studies. Surg Endosc. 2025. [Google Scholar] [CrossRef] [PubMed]
- Munir, MM; Endo, Y; Alaimo, L; et al. Textbook oncologic outcomes among patients undergoing laparoscopic, robotic and open surgery for intrahepatic and perihilar cholangiocarcinoma. HPB (Oxford). 2024, 26(10), 1293–1302. [Google Scholar] [CrossRef] [PubMed]
- Brolese, A; Spolverato, G; Cillo, U; et al. The role of robotic surgery for the treatment of hilar cholangiocarcinoma: a systematic review. Front Oncol. 2022, 12, 1001838. [Google Scholar] [CrossRef] [PubMed]
- Berardi, G; Ratti, F; Sposito, C; et al. Minimally invasive surgery for perihilar cholangiocarcinoma: a systematic review of the short- and long-term results. Cancers (Basel). 2023, 15(11), 3048. [Google Scholar] [CrossRef] [PubMed]
- Dong, S; Wang, Y; Liu, Y; et al. Comparison of robot-assisted, open, and laparoscopic-assisted surgery for cholangiocarcinoma: a network meta-analysis. Langenbecks Arch Surg. 2024, 409(1), 123. [Google Scholar] [CrossRef] [PubMed]
 |
 |
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




