Submitted:
30 March 2026
Posted:
31 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Pathophysiology and Therapeutic Challenges of Alzheimer’s Disease
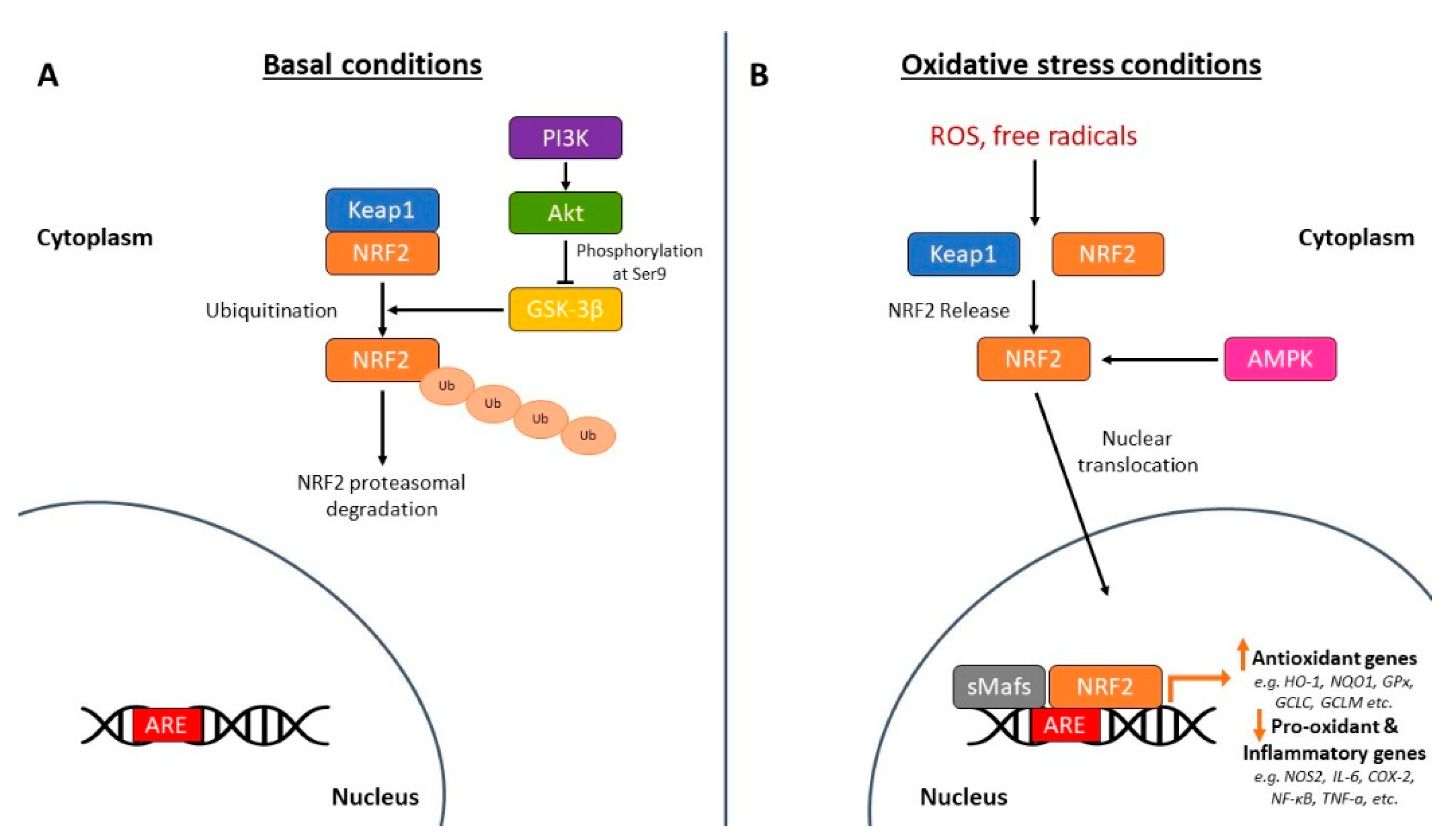
1.2. The Nrf2 Pathway in Oxidative Stress and Neuroprotection
1.3. Dimethyl Fumarate: From Traditional Medicine to Neurotherapeutics
1.4. Rationale and Objectives of the Present Systematic Review
- Activation of the Nrf2/ARE signaling pathway,
- Modulation of oxidative stress and antioxidant responses,
- Effects on amyloid-β and tau pathology,
- Anti-inflammatory and neuroprotective mechanisms, and
- Functional outcomes, including learning and memory performance.
2. Methodology
2.1. Search Strategy
2.2. Study Inclusion and Exclusion Criteria
- Review articles or meta-analyses.
- Articles that were not primary research reports.
- Studies that did not specifically address the selected keywords.
- Studies not related to the treatment of Alzheimer’s disease specifically.
- Studies in which DMF was not used as a therapeutic agent.
- Articles for which full-text access was not available.
2.3. Study Quality Assessment
3. Results
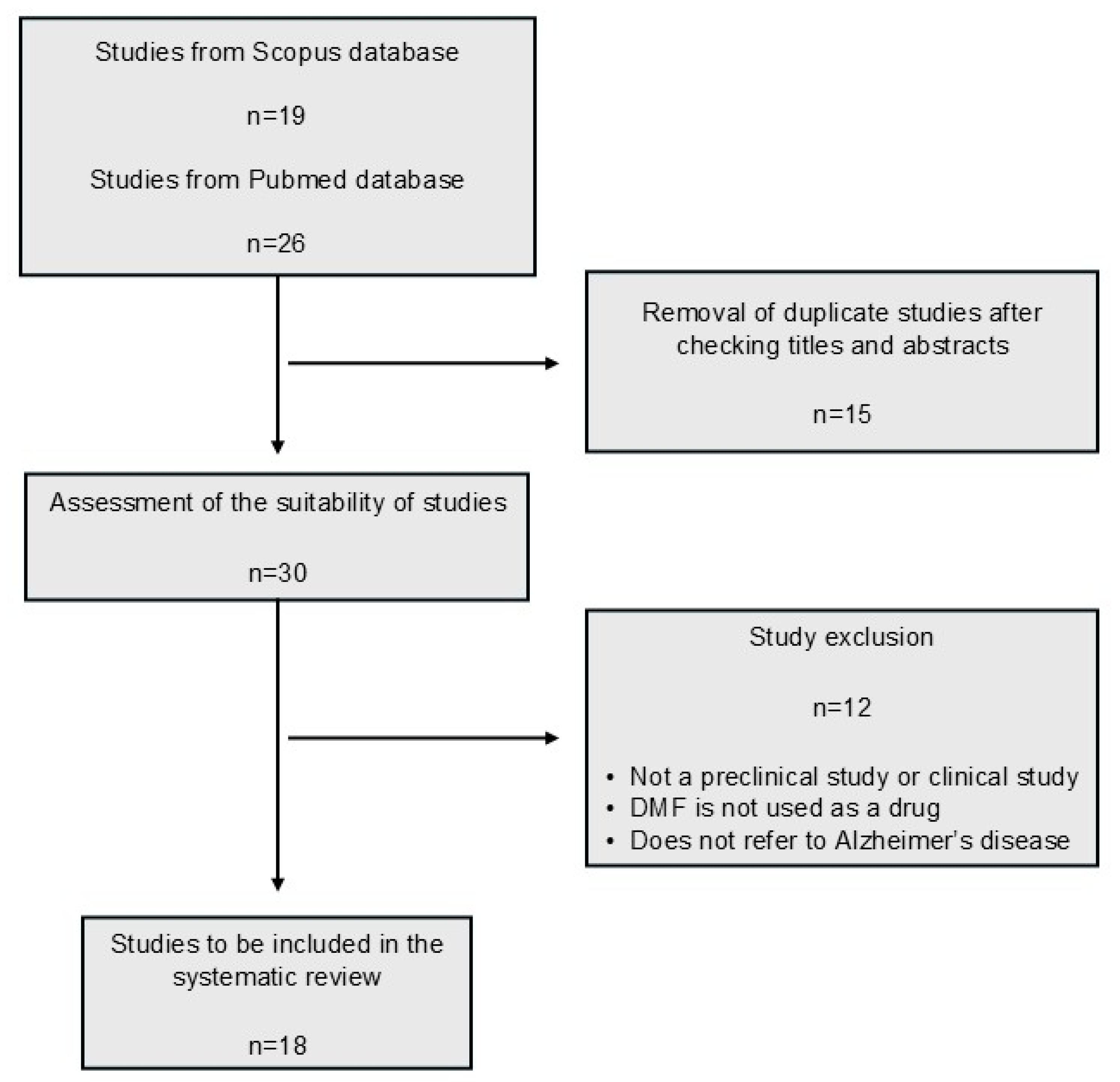
3.1. Selection of Studies
3.2. Study Quality
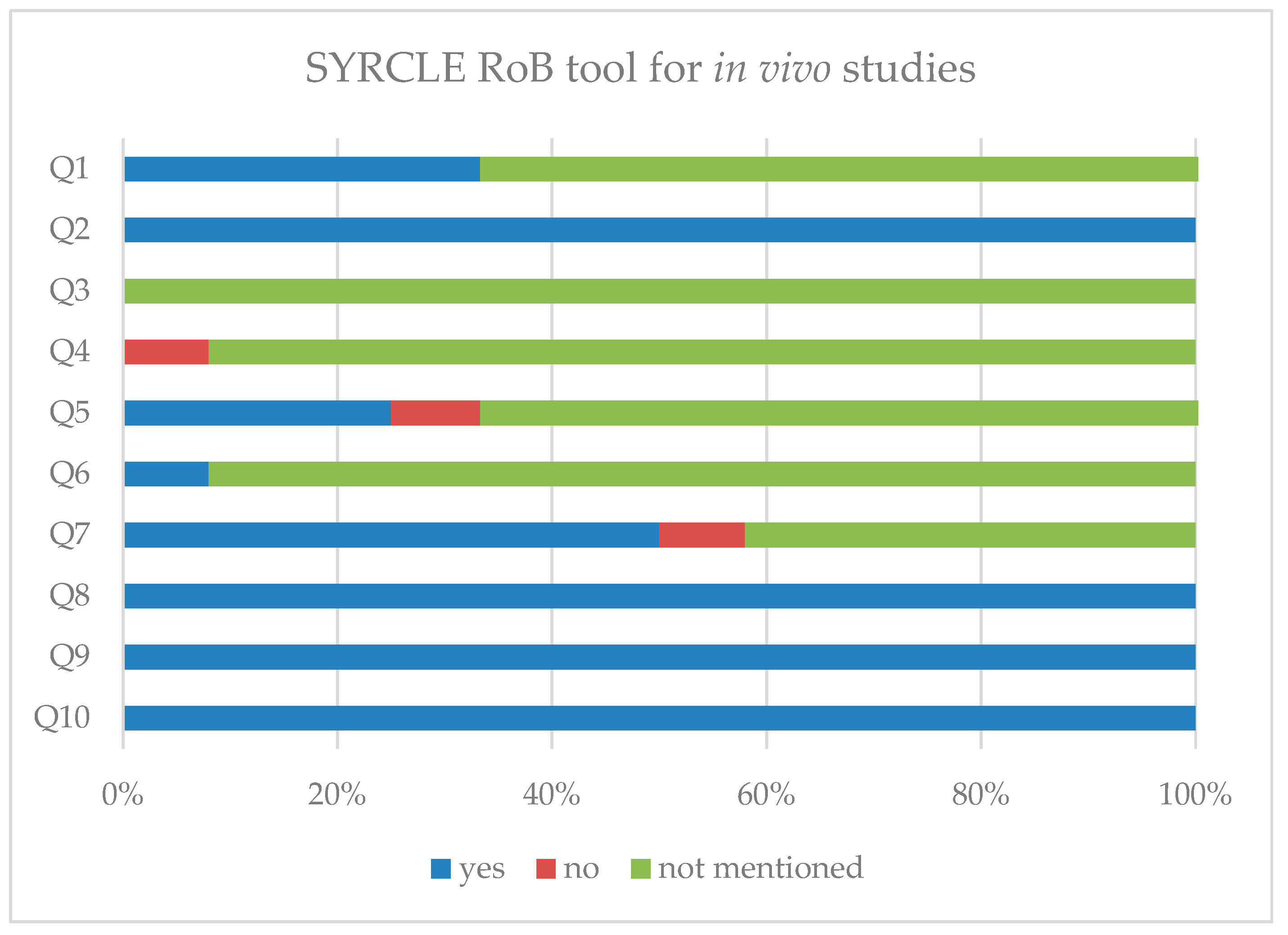
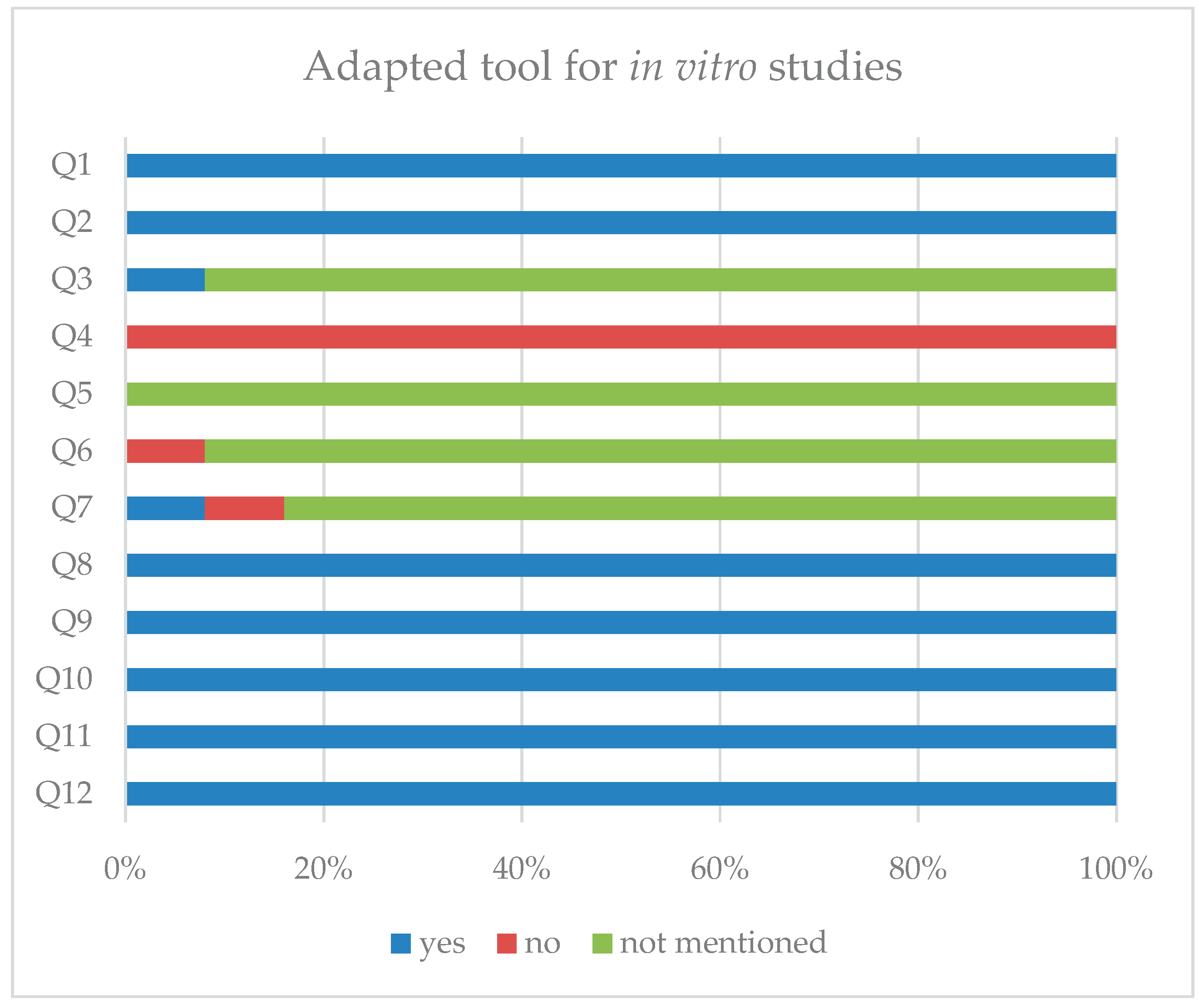
3.3. Analysis of Included Studies
3.3.1. In Vitro Studies
3.3.2. Mixed Methodology Studies
3.3.3. In Vivo Studies
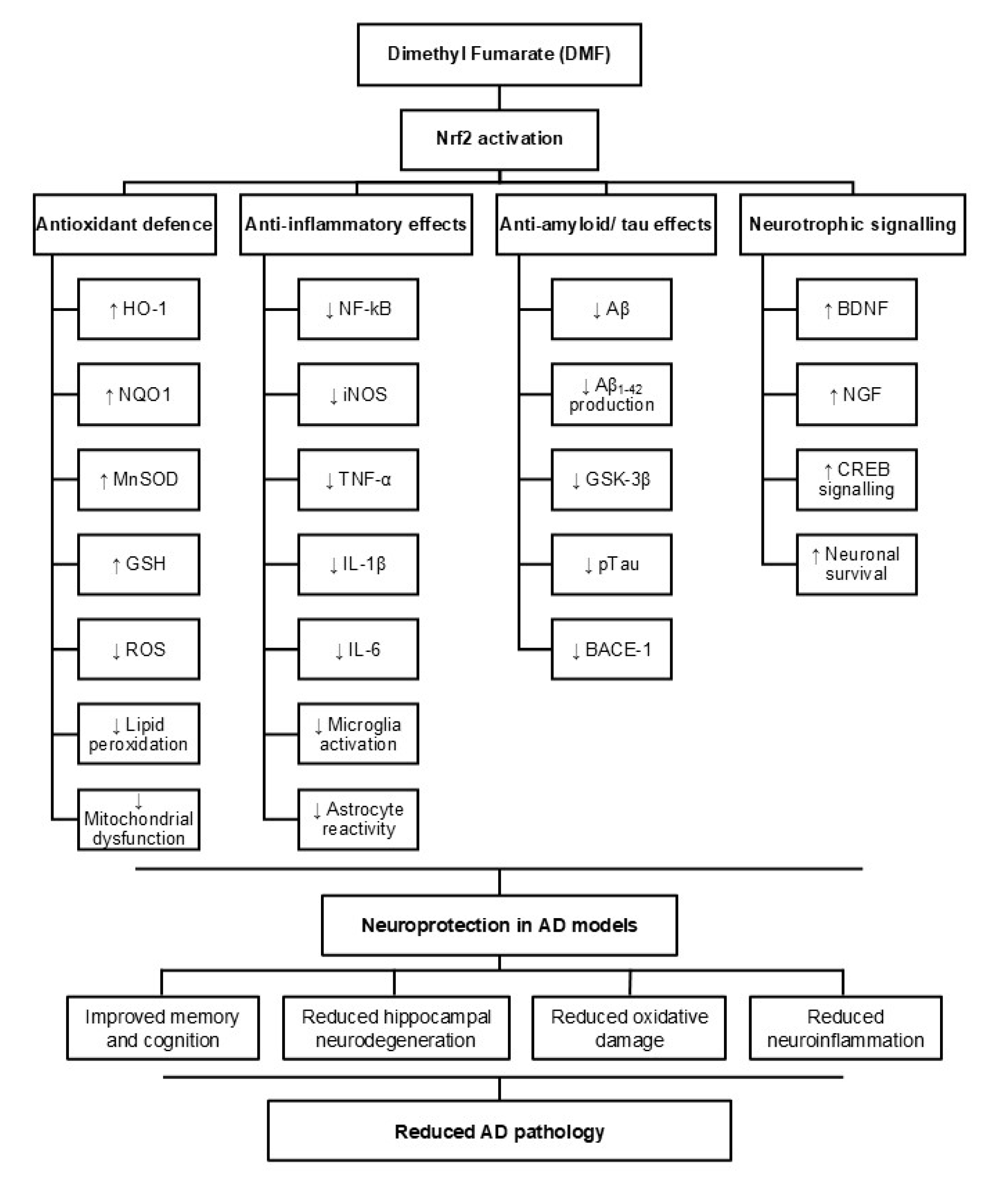
4. Discussion
5. Future Directions and Research Perspectives
6. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviation | Full Term / Description |
| Aβ AD AMPK |
Amyloid-β Alzheimer’s disease Adenosine monophosphate–activated protein kinase |
| APP | Amyloid precursor protein |
| ARE | Antioxidant response elements |
| BACE1 | β-Secretase 1 |
| BDNF | Brain-derived neurotrophic factor |
| COX-2 | Cyclooxygenase-2 |
| CREB Cyt c |
cAMP response element-binding protein Cytochrome c |
| DG | Dentate gyrus |
| D-Gal | D-galactose |
| DMF | Dimethyl fumarate |
| FDA | Food and Drug Administration |
| GCLM | Glutamate–cysteine ligase modifier subunit |
| GFAP | Glial fibrillary acidic protein |
| GPX4 | Glutathione peroxidase 4 |
| GSH | Glutathione |
| GSK-3β | Glycogen synthase kinase-3 beta |
| Hmox1 / HO-1 | Haem oxygenase-1 |
| IBA1 IFN-γ IL |
Ionized calcium-binding adapter molecule 1 Interferon-γ Interleukin |
| iNOS | Inducible nitric oxide synthase |
| LDH | Lactate dehydrogenase |
| LPS | Lipopolysaccharide |
| MAPK | Mitogen-activated protein kinase |
| MDA | Malondialdehyde |
| MG | Microglia |
| MG-H1 | Methylglyoxal-derived hydroimidazolone 1 |
| MGO | Methylglyoxal |
| MHCII | Major histocompatibility complex class II |
| MMF | Monomethyl fumarate |
| MnSOD / SOD2 | Manganese superoxide dismutase |
| MSC | Mesenchymal stem cells |
| NEH | Nrf2–ECH homology domains |
| NFAT1 | Nuclear factor of activated T cells 1 |
| NF-kB | Nuclear factor kappa B |
| NFTs | Neurofibrillary tangles |
| NGF | Nerve growth factor |
| NOS-2 | Nitric oxide synthase 2 |
| NQO1 | NAD(P)H quinone dehydrogenase 1 |
| Nrf2 | Nuclear factor erythroid 2–related factor 2 |
| NT3 | 3-Nitrotyrosine |
| OPTN | Optineurin |
| OVX | Ovariectomy |
| PI3K | Phosphoinositide 3-kinase |
| PKB / AKT PP2A |
Protein kinase B Protein phosphatase 2A |
| PP2B | Protein phosphatase 2B (Calcineurin) |
| ROMO1 | Reactive oxygen species modulator 1 |
| ROS | Reactive oxygen species |
| SIRT-1 | Sirtuin 1 |
| sMAFs SOCS3 |
Small musculoaponeurotic fibrosarcoma proteins Suppressor of cytokine signalling 3 |
| STAT3 | Signal transducer and activator of transcription 3 |
| STZ | Streptozotocin |
| TNF-α | Tumour necrosis factor alpha |
References
- Twarowski, B.; Herbet, M. Inflammatory Processes in Alzheimer’s Disease—Pathomechanism, Diagnosis and Treatment: A Review. Int. J. Mol. Sci. 2023, 24, 6518. [CrossRef]
- Breijyeh, Z.; Karaman, R. Comprehensive Review on Alzheimer’s Disease: Causes and Treatment. Molecules 2020, 25, 5789. [CrossRef]
- Lane, C.A.; Hardy, J.; Schott, J.M. Alzheimer’s Disease. Eur. J. Neurol. 2018, 25, 59–70. [CrossRef]
- World Health Organization WHO - Dementia Available online: https://www.who.int/news-room/fact-sheets/detail/dementia (accessed on 4 March 2026).
- Eratne, D.; Loi, S.M.; Farrand, S.; Kelso, W.; Velakoulis, D.; Looi, J.C. Alzheimer’s Disease: Clinical Update on Epidemiology, Pathophysiology and Diagnosis. Australasian Psychiatry 2018, 26, 347–357. [CrossRef]
- Zhang, X.-X.; Tian, Y.; Wang, Z.-T.; Ma, Y.-H.; Tan, L.; Yu, J.-T. The Epidemiology of Alzheimer’s Disease Modifiable Risk Factors and Prevention. J. Prev. Alzheimers Dis. 2021, 8, 313–321. [CrossRef]
- Aggarwal, N.T.; Mielke, M.M. Sex Differences in Alzheimer’s Disease. Neurol. Clin. 2023, 41, 343–358. [CrossRef]
- Beata, B.-K.; Wojciech, J.; Johannes, K.; Piotr, L.; Barbara, M. Alzheimer’s Disease—Biochemical and Psychological Background for Diagnosis and Treatment. Int. J. Mol. Sci. 2023, 24, 1059. [CrossRef]
- Rostagno, A.A. Pathogenesis of Alzheimer’s Disease. Int. J. Mol. Sci. 2022, 24, 107. [CrossRef]
- Aranda-Abreu, G.E.; Rojas-Durán, F.; Hernández-Aguilar, M.E.; Herrera-Covarrubias, D.; Tlapa-Monge, L.R.; Mestizo-Gutiérrez, S.L. Alzheimer’s Disease as a Disorder of Neuroimmune Dysregulation. Neurol. Int. 2026, 18, 37. [CrossRef]
- Lushchak, V.I. Free Radicals, Reactive Oxygen Species, Oxidative Stress and Its Classification. Chem. Biol. Interact. 2014, 224, 164–175. [CrossRef]
- Cheignon, C.; Tomas, M.; Bonnefont-Rousselot, D.; Faller, P.; Hureau, C.; Collin, F. Oxidative Stress and the Amyloid Beta Peptide in Alzheimer’s Disease. Redox Biol. 2018, 14, 450–464. [CrossRef]
- Ionescu-Tucker, A.; Cotman, C.W. Emerging Roles of Oxidative Stress in Brain Aging and Alzheimer’s Disease. Neurobiol. Aging 2021, 107, 86–95. [CrossRef]
- Tönnies, E.; Trushina, E. Oxidative Stress, Synaptic Dysfunction, and Alzheimer’s Disease. Journal of Alzheimer’s Disease 2017, 57, 1105–1121. [CrossRef]
- Sun, B.-L.; Li, W.-W.; Zhu, C.; Jin, W.-S.; Zeng, F.; Liu, Y.-H.; Bu, X.-L.; Zhu, J.; Yao, X.-Q.; Wang, Y.-J. Clinical Research on Alzheimer’s Disease: Progress and Perspectives. Neurosci. Bull. 2018, 34, 1111–1118. [CrossRef]
- Briggs, R.; Kennelly, S.P.; O’Neill, D. Drug Treatments in Alzheimer’s Disease. Clinical Medicine 2016, 16, 247–253. [CrossRef]
- Madav, Y.; Wairkar, S.; Prabhakar, B. Recent Therapeutic Strategies Targeting Beta Amyloid and Tauopathies in Alzheimer’s Disease. Brain Res. Bull. 2019, 146, 171–184. [CrossRef]
- Ozben, T.; Ozben, S. Neuro-Inflammation and Anti-Inflammatory Treatment Options for Alzheimer’s Disease. Clin. Biochem. 2019, 72, 87–89. [CrossRef]
- van Dyck, C.H. Anti-Amyloid-β Monoclonal Antibodies for Alzheimer’s Disease: Pitfalls and Promise. Biol. Psychiatry 2018, 83, 311–319. [CrossRef]
- Kim, B.-H.; Kim, S.; Nam, Y.; Park, Y.H.; Shin, S.M.; Moon, M. Second-Generation Anti-Amyloid Monoclonal Antibodies for Alzheimer’s Disease: Current Landscape and Future Perspectives. Transl. Neurodegener. 2025, 14, 6. [CrossRef]
- van Dyck, C.H.; Swanson, C.J.; Aisen, P.; Bateman, R.J.; Chen, C.; Gee, M.; Kanekiyo, M.; Li, D.; Reyderman, L.; Cohen, S.; et al. Lecanemab in Early Alzheimer’s Disease. New England Journal of Medicine 2023, 388, 9–21. [CrossRef]
- Sims, J.R.; Zimmer, J.A.; Evans, C.D.; Lu, M.; Ardayfio, P.; Sparks, J.; Wessels, A.M.; Shcherbinin, S.; Wang, H.; Monkul Nery, E.S.; et al. Donanemab in Early Symptomatic Alzheimer Disease. JAMA 2023, 330, 512. [CrossRef]
- Higgins, M.; Wasef, V.; Kwakowsky, A. FDA-Approved Passive Immunization Treatments Against Aβ in Alzheimer’s Disease: Where Are We Now? Int. J. Mol. Sci. 2026, 27, 883. [CrossRef]
- Yang, H.-M. Recent Advances in Antibody Therapy for Alzheimer’s Disease: Focus on Bispecific Antibodies. Int. J. Mol. Sci. 2025, 26, 6271. [CrossRef]
- Yi, L.X.; Tan, E.K.; Zhou, Z.D. Passive Immunotherapy for Alzheimer’s Disease: Challenges & Future Directions. J. Transl. Med. 2024, 22, 430. [CrossRef]
- Budd Haeberlein, S.; Aisen, P.S.; Barkhof, F.; Chalkias, S.; Chen, T.; Cohen, S.; Dent, G.; Hansson, O.; Harrison, K.; von Hehn, C.; et al. Two Randomized Phase 3 Studies of Aducanumab in Early Alzheimer’s Disease. J. Prev. Alzheimers Dis. 2022, 9, 197–210. [CrossRef]
- Barakos, J.; Purcell, D.; Suhy, J.; Chalkias, S.; Burkett, P.; Grassi, C.M.; Castrillo-Viguera, C.; Rubino, I.; Vijverberg, E. Detection and Management of Amyloid-Related Imaging Abnormalities in Patients with Alzheimer’s Disease Treated with Anti-Amyloid Beta Therapy. J. Prev. Alzheimers Dis. 2022, 9, 211–220. [CrossRef]
- Hampel, H.; Elhage, A.; Cho, M.; Apostolova, L.G.; Nicoll, J.A.R.; Atri, A. Amyloid-Related Imaging Abnormalities (ARIA): Radiological, Biological and Clinical Characteristics. Brain 2023, 146, 4414–4424. [CrossRef]
- Bejanin, A.; Schonhaut, D.R.; La Joie, R.; Kramer, J.H.; Baker, S.L.; Sosa, N.; Ayakta, N.; Cantwell, A.; Janabi, M.; Lauriola, M.; et al. Tau Pathology and Neurodegeneration Contribute to Cognitive Impairment in Alzheimer’s Disease. Brain 2017, 140, 3286–3300. [CrossRef]
- Rawal, S.; Wildsmith, K.R.; Aluri, J.; Yagi, T.; Chang, M.-K.; Niu, H.; Zhou, J.; Horie, K.; Takahashi, E.; Boyd, P.; et al. An Anti-Tau Therapeutic Antibody Etalanetug (E2814). Alzheimer Dis. Assoc. Disord. 2025, 39, 151–157. [CrossRef]
- Horie, K.; Salvadó, G.; Barthélemy, N.R.; Janelidze, S.; Li, Y.; He, Y.; Saef, B.; Chen, C.D.; Jiang, H.; Strandberg, O.; et al. CSF MTBR-Tau243 Is a Specific Biomarker of Tau Tangle Pathology in Alzheimer’s Disease. Nat. Med. 2023, 29, 1954–1963. [CrossRef]
- Galpern, W.R.; Triana-Baltzer, G.; Li, L.; Van Kolen, K.; Timmers, M.; Haeverans, K.; Janssens, L.; Kolb, H.; Nandy, P.; Aida, K.; et al. Phase 1 Studies of the Anti-Tau Monoclonal Antibody JNJ-63733657 in Healthy Participants and Participants with Alzheimer’s Disease. J. Prev. Alzheimers Dis. 2024, 11, 1592–1603. [CrossRef]
- Mullard, A. Anti-Tau Antibody Stumbles in Phase II Alzheimer Trial. Nat. Rev. Drug Discov. 2024, 23, 883–883. [CrossRef]
- Sidiropoulou, G.A.; Metaxas, A.; Kourti, M. Natural Antioxidants That Act against Alzheimer’s Disease through Modulation of the NRF2 Pathway: A Focus on Their Molecular Mechanisms of Action. Front. Endocrinol. (Lausanne). 2023, 14. [CrossRef]
- Lu, J.; Zhao, Y.; Liu, M.; Lu, J.; Guan, S. Toward Improved Human Health: Nrf2 Plays a Critical Role in Regulating Ferroptosis. Food Funct. 2021, 12, 9583–9606. [CrossRef]
- George, M.; Tharakan, M.; Culberson, J.; Reddy, A.P.; Reddy, P.H. Role of Nrf2 in Aging, Alzheimer’s and Other Neurodegenerative Diseases. Ageing Res. Rev. 2022, 82, 101756. [CrossRef]
- Hasan, S.K.; Jayakumar, S.; Espina Barroso, E.; Jha, A.; Catalano, G.; Sandur, S.K.; Noguera, N.I. Molecular Targets of Oxidative Stress: Focus on Nuclear Factor Erythroid 2–Related Factor 2 Function in Leukemia and Other Cancers. Cells 2025, 14, 713. [CrossRef]
- He, F.; Ru, X.; Wen, T. NRF2, a Transcription Factor for Stress Response and Beyond. Int. J. Mol. Sci. 2020, 21, 4777. [CrossRef]
- Baird, L.; Yamamoto, M. The Molecular Mechanisms Regulating the KEAP1-NRF2 Pathway. Mol. Cell. Biol. 2020, 40. [CrossRef]
- Camiña, N.; Penning, T.M. Genetic and Epigenetic Regulation of the NRF2-KEAP1 Pathway in Human Lung Cancer. Br. J. Cancer 2022, 126, 1244–1252. [CrossRef]
- Khalil, A.A.K.; Woo, M.-S.; Kang, D.-M.; Ahn, M.-J.; Kim, J.-A.; Yang, H.; Kim, J.-H. Activation of Nuclear Factor Erythroid 2-Related Factor-2 by Oxylipin from Mangifera Indica Leaves. Antioxidants 2024, 13, 1119. [CrossRef]
- Gumeni, S.; Papanagnou, E.-D.; Manola, M.S.; Trougakos, I.P. Nrf2 Activation Induces Mitophagy and Reverses Parkin/Pink1 Knock down-Mediated Neuronal and Muscle Degeneration Phenotypes. Cell Death Dis. 2021, 12, 671. [CrossRef]
- Kasai, S.; Shimizu, S.; Tatara, Y.; Mimura, J.; Itoh, K. Regulation of Nrf2 by Mitochondrial Reactive Oxygen Species in Physiology and Pathology. Biomolecules 2020, 10, 320. [CrossRef]
- Aguiar, R. Fumaria Officinalis L. Active Compounds and Biological Activities: A Review. Int. J. Herb. Med. 2023, 11, 144–151. [CrossRef]
- Majkutewicz, I. Dimethyl Fumarate: A Review of Preclinical Efficacy in Models of Neurodegenerative Diseases. Eur. J. Pharmacol. 2022, 926, 175025. [CrossRef]
- Raafat, K.M.; El-Zahaby, S.A. Niosomes of Active Fumaria Officinalis Phytochemicals: Antidiabetic, Antineuropathic, Anti-Inflammatory, and Possible Mechanisms of Action. Chin. Med. 2020, 15, 40. [CrossRef]
- Prokopenko, Y.; Surzhykov, I.; Golovchenko, O.; Mishchenko, V.; Georgiyants, V. Fumaria Officinalis: Phytochemical Complexity and Its Medicinal Significance. Fitoterapia 2025, 186, 106780. [CrossRef]
- Corazza, M.; Odorici, G.; Conti, A.; Di Lernia, V.; Motolese, A.; Bardazzi, F.; Di Nuzzo, S.; Monti, A.; Arginelli, F.; Filippi, F.; et al. Dimethyl Fumarate Treatment for Psoriasis in a Real-life Setting: A Multicentric Retrospective Study. Dermatol. Ther. 2021, 34. [CrossRef]
- Xu, Z.; Zhang, F.; Sun, F.; Gu, K.; Dong, S.; He, D. Dimethyl Fumarate for Multiple Sclerosis. Cochrane Database of Systematic Reviews 2015, 2015. [CrossRef]
- Scuderi, S.A.; Ardizzone, A.; Paterniti, I.; Esposito, E.; Campolo, M. Antioxidant and Anti-Inflammatory Effect of Nrf2 Inducer Dimethyl Fumarate in Neurodegenerative Diseases. Antioxidants 2020, 9, 630. [CrossRef]
- Brück, J.; Dringen, R.; Amasuno, A.; Pau-Charles, I.; Ghoreschi, K. A Review of the Mechanisms of Action of Dimethylfumarate in the Treatment of Psoriasis. Exp. Dermatol. 2018, 27, 611–624. [CrossRef]
- Brennan, M.S.; Matos, M.F.; Li, B.; Hronowski, X.; Gao, B.; Juhasz, P.; Rhodes, K.J.; Scannevin, R.H. Dimethyl Fumarate and Monoethyl Fumarate Exhibit Differential Effects on KEAP1, NRF2 Activation, and Glutathione Depletion In Vitro. PLoS One 2015, 10, e0120254. [CrossRef]
- Diebold, M.; Sievers, C.; Bantug, G.; Sanderson, N.; Kappos, L.; Kuhle, J.; Lindberg, R.L.P.; Derfuss, T. Dimethyl Fumarate Influences Innate and Adaptive Immunity in Multiple Sclerosis. J. Autoimmun. 2018, 86, 39–50. [CrossRef]
- Wang, C.; Guo, H.; Wang, L.; Wang, Q.; Liu, T.; Huang, J.; Wei, Y.; Wang, C. Dimethyl Fumarate Alleviates Inflammation during High Altitude Hypoxia Induced Acute Lung Injury by Upregulating Nrf2/SLC7A11 Pathway in Ferroptosis. Clin. Proteomics 2025, 22, 42. [CrossRef]
- Sharkus, R.; Thakkar, R.; Kolson, D.L.; Constantinescu, C.S. Dimethyl Fumarate as Potential Treatment for Alzheimer’s Disease: Rationale and Clinical Trial Design. Biomedicines 2023, 11, 1387. [CrossRef]
- Sun, X.; Suo, X.; Xia, X.; Yu, C.; Dou, Y. Dimethyl Fumarate Is a Potential Therapeutic Option for Alzheimer’s Disease. Journal of Alzheimer’s Disease 2022, 85, 443–456. [CrossRef]
- Hooijmans, C.R.; Rovers, M.M.; de Vries, R.B.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s Risk of Bias Tool for Animal Studies. BMC Med. Res. Methodol. 2014, 14, 43. [CrossRef]
- Sheth, V.H.; Shah, N.P.; Jain, R.; Bhanushali, N.; Bhatnagar, V. Development and Validation of a Risk-of-Bias Tool for Assessing in Vitro Studies Conducted in Dentistry: The QUIN. J. Prosthet. Dent. 2024, 131, 1038–1042. [CrossRef]
- Roth, N.; Zilliacus, J.; Beronius, A. Development of the SciRAP Approach for Evaluating the Reliability and Relevance of in Vitro Toxicity Data. Frontiers in Toxicology 2021, 3. [CrossRef]
- Rajput, M.S.; Nirmal, N.P.; Rathore, D.; Dahima, R. Dimethyl Fumarate Mitigates Tauopathy in Aβ-Induced Neuroblastoma SH-SY5Y Cells. Neurochem. Res. 2020, 45, 2641–2652. [CrossRef]
- Rajput, M.S.; Nirmal, N.P.; Rathore, D.; Dahima, R. Dimethyl Fumarate Exerts Neuroprotection by Modulating Calcineurin/NFAT1 and NFκB Dependent BACE1 Activity in Aβ1-42 Treated Neuroblastoma SH-SY5Y Cells. Brain Res. Bull. 2020, 165, 97–107. [CrossRef]
- Campolo, M.; Casili, G.; Lanza, M.; Filippone, A.; Paterniti, I.; Cuzzocrea, S.; Esposito, E. Multiple Mechanisms of Dimethyl Fumarate in Amyloid Β-induced Neurotoxicity in Human Neuronal Cells. J. Cell. Mol. Med. 2018, 22, 1081–1094. [CrossRef]
- Silva, A.; Pereira, M.; Carrascal, M.A.; Brites, G.; Neves, B.; Moreira, P.; Resende, R.; Silva, M.M.; Santos, A.E.; Pereira, C.; et al. Calcium Modulation, Anti-Oxidant and Anti-Inflammatory Effect of Skin Allergens Targeting the Nrf2 Signaling Pathway in Alzheimer’s Disease Cellular Models. Int. J. Mol. Sci. 2020, 21, 7791. [CrossRef]
- Lanza, M.; Basilotta, R.; Cuzzocrea, S.; Bulzomì, M.; Oddo, S.; Casili, G.; Esposito, E. An RNAi-Mediated Reduction in Transcription Factor Nrf-2 Blocks the Positive Effects of Dimethyl Fumarate on Metabolic Stress in Alzheimer’s Disease. Int. J. Mol. Sci. 2023, 24, 11303. [CrossRef]
- Koike, S.; Tsurudome, S.; Okano, S.; Kishida, A.; Ogasawara, Y. Dimethyl Fumarate Reduces Methylglyoxal-Derived Carbonyl Stress Through Nrf2/GSH Activation in SH-SY5Y Cells. Neurochem. Res. 2025, 50, 28. [CrossRef]
- Wang, T.; Sobue, A.; Watanabe, S.; Komine, O.; Saido, T.C.; Saito, T.; Yamanaka, K. Dimethyl Fumarate Improves Cognitive Impairment and Neuroinflammation in Mice with Alzheimer’s Disease. J. Neuroinflammation 2024, 21, 55. [CrossRef]
- Babaei, H.; Kheirollah, A.; Ranjbaran, M.; Cheraghzadeh, M.; Sarkaki, A.; Adelipour, M. Preconditioning Adipose-Derived Mesenchymal Stem Cells with Dimethyl Fumarate Promotes Their Therapeutic Efficacy in the Brain Tissues of Rats with Alzheimer’s Disease. Biochem. Biophys. Res. Commun. 2023, 672, 120–127. [CrossRef]
- Paraiso, H.C.; Kuo, P.-C.; Curfman, E.T.; Moon, H.J.; Sweazey, R.D.; Yen, J.-H.; Chang, F.-L.; Yu, I.-C. Dimethyl Fumarate Attenuates Reactive Microglia and Long-Term Memory Deficits Following Systemic Immune Challenge. J. Neuroinflammation 2018, 15, 100. [CrossRef]
- Huang, Y.; Han, M.; Fu, Y.; Wang, G.; Kong, L.; Mo, J.; Cao, D.; Chu, Z.; Li, W. HY-021068 Improves Neuronal Ferroptosis by Activating Nrf2 Signaling in APP/PS1 Mice and Aβ1-42-Induced HT22 Cells. Eur. J. Pharmacol. 2025, 1008, 178349. [CrossRef]
- Majkutewicz, I.; Kurowska, E.; Podlacha, M.; Myślińska, D.; Grembecka, B.; Ruciński, J.; Plucińska, K.; Jerzemowska, G.; Wrona, D. Dimethyl Fumarate Attenuates Intracerebroventricular Streptozotocin-Induced Spatial Memory Impairment and Hippocampal Neurodegeneration in Rats. Behavioural Brain Research 2016, 308, 24–37. [CrossRef]
- Majkutewicz, I.; Kurowska, E.; Podlacha, M.; Myślińska, D.; Grembecka, B.; Ruciński, J.; Pierzynowska, K.; Wrona, D. Age-Dependent Effects of Dimethyl Fumarate on Cognitive and Neuropathological Features in the Streptozotocin-Induced Rat Model of Alzheimer’s Disease. Brain Res. 2018, 1686, 19–33. [CrossRef]
- Abd El-Fatah, I.M.; Abdelrazek, H.M.A.; Ibrahim, S.M.; Abdallah, D.M.; El-Abhar, H.S. Dimethyl Fumarate Abridged Tauo-/Amyloidopathy in a D-Galactose/Ovariectomy-Induced Alzheimer’s-like Disease: Modulation of AMPK/SIRT-1, AKT/CREB/BDNF, AKT/GSK-3β, Adiponectin/Adipo1R, and NF-ΚB/IL-1β/ROS Trajectories. Neurochem. Int. 2021, 148, 105082. [CrossRef]
- Rojo, A.I.; Pajares, M.; García-Yagüe, A.J.; Buendia, I.; Van Leuven, F.; Yamamoto, M.; López, M.G.; Cuadrado, A. Deficiency in the Transcription Factor NRF2 Worsens Inflammatory Parameters in a Mouse Model with Combined Tauopathy and Amyloidopathy. Redox Biol. 2018, 18, 173–180. [CrossRef]
- Wrona, D.; Majkutewicz, I.; Świątek, G.; Dunacka, J.; Grembecka, B.; Glac, W. Dimethyl Fumarate as the Peripheral Blood Inflammatory Mediators Inhibitor in Prevention of Streptozotocin-Induced Neuroinflammation in Aged Rats. J. Inflamm. Res. 2022, Volume 15, 33–52. [CrossRef]
- Piekarczyk, N.; Berezka, P.; Majkutewicz, I.; Myślińska, D.; Kaczor, J.J. Combined Vitamin D3 and Dimethyl Fumarate Treatment Alleviates Cognitive Dysfunction, Oxidative Stress, and Inflammation in a Rat Model of Sporadic Alzheimer’s Disease. Free Radic. Biol. Med. 2025, 241, 748–759. [CrossRef]
- Möhle, L.; Brackhan, M.; Bascuñana, P.; Pahnke, J. Dimethyl Fumarate Does Not Mitigate Cognitive Decline and β-Amyloidosis in Female APPPS1 Mice. Brain Res. 2021, 1768, 147579. [CrossRef]
- Akhtar, A.; Gupta, S.M.; Dwivedi, S.; Kumar, D.; Shaikh, Mohd.F.; Negi, A. Preclinical Models for Alzheimer’s Disease: Past, Present, and Future Approaches. ACS Omega 2022, 7, 47504–47517. [CrossRef]
- Hui, C.K.; Dedkova, E.N.; Montgomery, C.; Cortopassi, G. Dimethyl Fumarate Dose-Dependently Increases Mitochondrial Gene Expression and Function in Muscle and Brain of Friedreich’s Ataxia Model Mice. Hum. Mol. Genet. 2021, 29, 3954–3965. [CrossRef]
- Passeri, E.; Elkhoury, K.; Morsink, M.; Broersen, K.; Linder, M.; Tamayol, A.; Malaplate, C.; Yen, F.T.; Arab-Tehrany, E. Alzheimer’s Disease: Treatment Strategies and Their Limitations. Int. J. Mol. Sci. 2022, 23, 13954. [CrossRef]
- Metaxas, A.; Anzalone, M.; Vaitheeswaran, R.; Petersen, S.; Landau, A.M.; Finsen, B. Neuroinflammation and Amyloid-Beta 40 Are Associated with Reduced Serotonin Transporter (SERT) Activity in a Transgenic Model of Familial Alzheimer’s Disease. Alzheimers Res. Ther. 2019, 11, 38. [CrossRef]
- Li, X.; Sun, Y.; Tjahjono, A.W.; Wei, Y.; Li, X.; Zheng, Q.; Qi, W.; Liang, F. Acupuncture Attenuates Myocardial Ischemia/Reperfusion Injury-Induced Ferroptosis via the Nrf2/HO-1 Pathway. Chin. Med. 2025, 20, 61. [CrossRef]
- Ma, H.; Dong, Y.; Chu, Y.; Guo, Y.; Li, L. The Mechanisms of Ferroptosis and Its Role in Alzheimer’s Disease. Front. Mol. Biosci. 2022, 9, 965064. [CrossRef]
| Number | Question |
|---|---|
| Q1 | Was the allocation sequence adequately generated and applied? (Selection bias) |
| Q2 | Were the groups similar at baseline, or were they adjusted for confounders in the analysis? (Selection bias) |
| Q3 | Was the allocation adequately concealed? (Selection bias) |
| Q4 | Were the animals housed randomly during the experiment? (Performance bias) |
| Q5 | Were the caregivers and/or investigators blinded from knowledge of which intervention each animal received during the experiment? (Performance bias) |
| Q6 | Were animals selected at random for outcome assessment? (Detection bias) |
| Q7 | Was the outcome assessor blinded? (Detection bias) |
| Q8 | Were incomplete outcome data adequately addressed? (Attrition bias) |
| Q9 | Are reports of the study free of selective outcome reporting? (Reporting bias) |
| Q10 | Was the study apparently free of other problems that could result in a high risk of bias? (Other bias) |
| Number | Question |
|---|---|
| Q1 | Was the experimental objective or hypothesis clearly stated? (Reporting bias) |
| Q2 | Was the cell type or cell line clearly described and appropriate for the research question? (Other bias) |
| Q3 | Was cell line authentication or contamination testing (e.g., mycoplasma testing) reported? (Other bias) |
| Q4 | Was the number of biological replicates justified or explained? (Other bias) |
| Q5 | Were samples (e.g., wells, cultures, plates) randomly allocated to experimental groups? (Selection bias) |
| Q6 | Were investigators blinded to treatment groups during the experiment or data collection? (Performance bias) |
| Q7 | Was outcome assessment performed blinded to group allocation? (Detection bias) |
| Q8 | Were culture conditions (media, incubation, temperature, passage number, treatment timing) standardized and described? (Performance bias) |
| Q9 | Were experiments replicated independently (biological replicates or separate experiments)? (Other bias) |
| Q10 | Were outcome measurement methods clearly described and validated (e.g., assay type, antibodies, imaging protocol)? (Detection bias) |
| Q11 | Were appropriate statistical analyses reported and clearly described? (Reporting bias) |
| Q12 | Were all measured outcomes reported without evidence of selective reporting? (Reporting bias) |
| Preclinical study | Experimental model* | DMF dose | Dose of other interventions | Main results |
|---|---|---|---|---|
| In vitro studies | ||||
| Rajput et al. (2020a) | Human neuroblastoma SH-SY5Y cells | 30 μM DMF | 1 μM oligomer or Aβ1-42 |
|
| Rajput et al. (2020b) | Human neuroblastoma SH-SY5Y cells | 10 and 30 μM DMF | 1 μM oligomer or Aβ1-42 |
|
| Campolo et al. (2018) | In vitro: Human neuroblastoma SH-SY5Y cells Ex vivo: Organotypic hippocampal slices |
30 μM DMF | 1 μM Aβ1-42 | In vitro:
|
| Silva et al. (2020) | Mouse neuroblastoma N2a cells Mouse microglial BV-2 cells |
N2a: 14μM DMF BV-2: 30μM DMF |
50 ng/mL LPS in BV-2 |
N2a:
|
| Lanza et al. (2023) | Human neuroblastoma SH-SY5Y cells | 0.1, 1 and 10 mM DMF | 17.5 mM glucose |
|
| Koike et al. (2025) | Human neuroblastoma SH-SY5Y cells | 10, 20 and 30 μM DMF | 800 μM MGO |
|
| Mixed methodology studies | ||||
| Wang et al. (2024) | In vitro: • T98G astrocyte cells • A1 astrocyte cells In vivo: • AppNL-G-F/NL-G-F (App-KI) and WT mice 6m, 11m |
In vitro: • T98G: 0 and 35 μM DMF • A1: 35 μM DMF In vivo: • 300 mg/kg DMF |
N/S | In vitro:
|
| Sun et al. (2022) | In vitro: • Primary mouse hippocampal neurons (embryonic, Nrf2+/+ and Nrf2-/-, females) In vivo: • C57BL/6J mice (male) 8w |
In vitro: • 0, 20, 40, 60, 80 and 100 μM DMF In vivo: • 10 mg/mL DMF |
2 μg/μL Aβ1-42 1μg/μL IBO |
In vitro:
|
| Babaei et al. (2023) | In vitro: • Rat mesenchymal stem cells (MSCs, male) In vivo: • Wistar rats (male) adult |
In vitro: • 5, 10, 15, 20, 30, 40 and 50 μM DMF In vivo: • 20 μM DMF |
In vivo: • 1μg/μL Aβ1–42 |
In vitro:
|
| Paraiso et al. (2018) | In vitro: • Microglial cells (MG) In vivo: • C57BL/6, Cx3cr1gfp/+, and Nrf2−/− mice (female) 12-16w |
In vitro: • 0, 10 and 100 μM DMF In vivo: • 45 mg/kg DMF |
In vitro: 100 ng/ml LPS In vivo: Saline or 1 mg/kg LPS |
In vitro:
|
| Huang et al. (2025) | In vitro: • Mouse hippocampal HT22 cells In vivo: • Transgenic APP/PS1 and WT mice (male) 6m |
In vitro: • 30 μM DMF In vivo: • 50 mg/kg DMF |
In vitro: 40 μM HY, 5 μM Aβ1–42 In vivo: 5, 10 and 20 mg/kg HY |
In vitro:
|
| In vivo studies | ||||
| Majkutewicz et al. (2016) | Wistar Rats (male) 4m | 0.4% DMF | 3 mg/kg STZ |
|
| Majkutewicz et al. (2018) | Wistar Rats (male) 22m | 0.4% DMF | 3 mg/kg STZ |
|
| Abd El-Fatah et al. (2021) | Wistar Rats (female) 18m | 45 mg/kg DMF | 150 mg/kg/day D-Gal |
|
| Rojo et al. (2018) | NRF2-KO and NRF2-WT mice with combined amyloidosis and tauopathy 6m, 9m, 11m | 100 mg/kg DMF | N/S |
|
| Wrona et al. (2022) | Wistar Rats (male) 4m and 22m | 0.4% DMF | 3 mg/kg STZ |
|
| Piekarczyk et al. (2025) | Wistar Rats (male) 4m | 50 mg/kg DMF | 3 mg/kg STZ 2000 IU/kg Vit D3 |
|
| Möhle et al. (2021) | Transgenic APPPS1-21 mice (female) 40d, 60d | 75 mg/kg DMF | N/S |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




