Submitted:
26 March 2026
Posted:
27 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Sarcopenia and Cachexia in Heart Failure
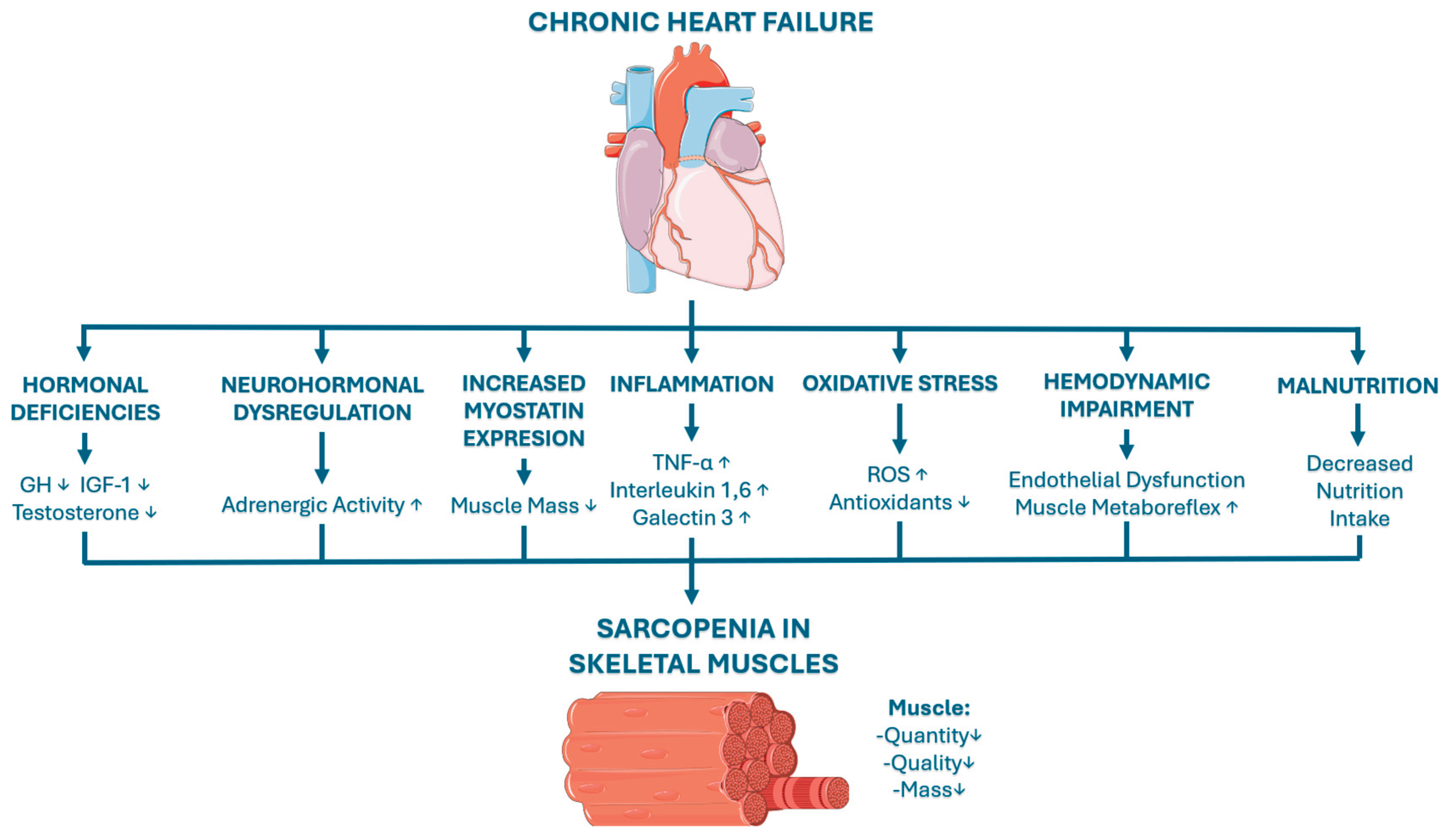
3. Pathophysiology of Sarcopenia in Chronic Heart Failure
3.1. Alterations in Skeletal Muscle Tissue
3.2. Main Factors in the Pathophysiology of Sarcopenia in CHF
3.2.1. Hormonal Dysregulation
3.2.2. Neurohormonal Dysfunction
3.2.3. Increased Myostatin Expression
3.2.4. Chronic Inflammation
3.2.5. Oxidative Stress and ROS
3.2.6. Disturbed Hemodynamics and Endothelial Dysfunction
3.2.7. Malnutrition and Anorexia
4. Nutritional and Pharmacological Interventions Targeting Sarcopenia in Chronic Heart Failure
4.1. Nutritional Interventions
4.2. Pharmacological INTERVENTIONS
5. Physical Training
6. Conclusions and Further Directions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| HF | Heart Failure |
| CHF | Chronic Heart Failure |
| NYHA | New York Heart Association |
| 6MWT | Six-Minute Walk Test |
| pVO₂ | Peak Oxygen Uptake |
| SPPB | Short Physical Performance Battery |
| IGF-1 | Insulin-like Growth Factor 1 |
| TNF-α | Tumor Necrosis Factor Alpha |
| ROS | Reactive Oxygen Species |
| GNRI | Geriatric Nutritional Risk Index |
| CONUT | Controlling Nutritional Status Score |
| ACEI | Angiotensin-Converting Enzyme Inhibitor |
| MRA | Mineralocorticoid Receptor Antagonists |
| SARM | Selective Androgen Receptor Modulators |
| TRIM63 | Tripartite Motif-Containing 63 |
| MuRF-1 | Muscle RING Finger 1 |
| NT-proBNP | N-terminal prohormone of brain natriuretic peptide |
| LVEF | Left Ventricular Ejection Fraction |
| ET | Exercise Tolerance |
| QOL | Quality Of Life |
| HFpEF | Heart Failure Preserved Ejection Fraction |
| CS | Citrate Synthase |
References
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; Cleland, J.G.F.; Coats, A.J.S.; Crespo-Leiro, M.G.; Farmakis, D.; Gilard, M.; Heymans, S.; Jaarsma, T.; Jankowska, E.A.; Lainscak, M.; Lam, C.S.P.; Lyon, A.R.; McMurray, J.J.V.; Mebazaa, A.; Mindham, R.; Muneretto, C.; Francesco Piepoli, M.; Price, S.; Rosano, G.M.C.; Ruschitzka, F.; Kathrine Skibelund, A. 2021 ESC Guidelines for the Diagnosis and Treatment of Acute and Chronic Heart Failure. European Heart Journal 2021, 42, 3599–3726. [Google Scholar] [CrossRef]
- Hogg, K.; Swedberg, K.; McMurray, J. Heart Failure with Preserved Left Ventricular Systolic Function: Epidemiology, Clinical Characteristics, and Prognosis. Journal of the American College of Cardiology 2004, 43, 317–327. [Google Scholar] [CrossRef]
- Packer, M. The Neurohormonal Hypothesis: A Theory to Explain the Mechanism of Disease Progression in Heart Failure. Journal of the American College of Cardiology 1992, 20, 248–254. [Google Scholar] [CrossRef]
- Lena, A.; Hadzibegović, S.; von Haehling, S.; Springer, J.; Coats, A.J.S.; Anker, M.S. Sarcopenia and Cachexia in Chronic Diseases: From Mechanisms to Treatment. Polish Archives of Internal Medicine 2021, 131. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; Schneider, S.M.; Sieber, C.C.; Topinkova, E.; Vandewoude, M.; Visser, M.; Zamboni, M. Sarcopenia: Revised European Consensus on Definition and Diagnosis. Age and Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Yuan, S.; Larsson, S.C. Epidemiology of Sarcopenia: Prevalence, Risk Factors, and Consequences. Metabolism 2023, 144, 155533. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, J.; Ni, W.; Yuan, X.; Zhang, H.; Zhang, L.; Zhang, M.; Zhang, Y. Sarcopenia in Heart Failure: A Systematic Review and Meta-Analysis. ESC Heart Failure 2021, 8, 1007–1017. [Google Scholar] [CrossRef] [PubMed]
- Lopez, P.D.; Nepal, P.; Akinlonu, A.; Ogunbayo, G.O.; Mishra, T.; Rangaswami, J.; Ahmed, H.M. Low Skeletal Muscle Mass Independently Predicts Mortality in Patients with Chronic Heart Failure after an Acute Hospitalization. Cardiology 2019, 142, 28–36. [Google Scholar] [CrossRef]
- von Haehling, S.; Garfias Macedo, T.; Valentova, M.; Ebner, N.; Bekfani, T.; Sandek, A.; Doehner, W.; Anker, S.D. Muscle Wasting as an Independent Predictor of Survival in Patients with Chronic Heart Failure. Journal of Cachexia, Sarcopenia and Muscle 2020, 11, 1242–1249. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, L.; Ferrucci, L.; Cherubini, A.; Maggio, M.; Bandinelli, S.; Savino, E.; Brombo, G.; Ceda, G.P.; Guralnik, J.M.; Landi, F. The Predictive Value of the EWGSOP Definition of Sarcopenia: Results from the InCHIANTI Study. The Journals of Gerontology: Series A, Biological Sciences and Medical Sciences 2016, 71, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.; Thunestvedt, M.; Lamberts, M.; Hovland, A.; Atar, D.; Omland, T. Heart Failure—Chronic Heart Failure, Pathophysiology and Mechanisms, Heart Failure with Reduced Ejection Fraction (HFrEF). European Heart Journal 2024, 45, ehae666.843. [Google Scholar] [CrossRef]
- de Jorge-Huerta, L.; Marco-Alacid, C.; Grande, C.; Velardo Andrés, C. A Narrative Review of the Diagnosis and Treatment of Sarcopenia and Malnutrition in Patients with Heart Failure. Nutrients 2024, 16, 2717. [Google Scholar] [CrossRef]
- Emami, A.; Saitoh, M.; Valentova, M.; Sandek, A.; Evertz, R.; Ebner, N.; Bekfani, T.; Valentova, M.; Doehner, W.; Anker, S.D.; von Haehling, S. Comparison of Sarcopenia and Cachexia in Men with Chronic Heart Failure: Results from the Studies Investigating Co-Morbidities Aggravating Heart Failure (SICA-HF). European Journal of Heart Failure 2018, 20, 1580–1587. [Google Scholar] [CrossRef] [PubMed]
- von Haehling, S. The Wasting Continuum in Heart Failure: From Sarcopenia to Cachexia. Proceedings of the Nutrition Society 2015, 74, 367–377. [Google Scholar] [CrossRef]
- Valentova, M.; Anker, S.D.; von Haehling, S. Cardiac Cachexia Revisited: The Role of Wasting in Heart Failure. Heart Failure Clinics 2020, 16, 61–69. [Google Scholar] [CrossRef]
- Anker, S.D.; Ponikowski, P.; Varney, S.; Chua, T.P.; Clark, A.L.; Webb-Peploe, K.M.; Harrington, D.; Kox, W.J.; Poole-Wilson, P.A.; Coats, A.J.S. Wasting as an Independent Risk Factor for Mortality in Chronic Heart Failure. The Lancet 1997, 349, 1050–1053. [Google Scholar] [CrossRef]
- Georgiadou, P.; Adamopoulos, S. Skeletal Muscle Abnormalities in Chronic Heart Failure. Current Heart Failure Reports 2012, 9, 128–132. [Google Scholar] [CrossRef]
- Drexler, H.; Riede, U.; Münzel, T.; König, H. Alterations of Skeletal Muscle in Chronic Heart Failure. Circulation 1992, 85, 1751–1759. [Google Scholar] [CrossRef] [PubMed]
- Zamboni, M.; Rossi, A.P.; Corzato, F.; Bambace, C.; Mazzali, G.; Fantin, F. Sarcopenia, Cachexia and Congestive Heart Failure in the Elderly. Current Pharmaceutical Design 2013, 19, 3656–3664. [Google Scholar] [CrossRef]
- Filippatos, G.S.; Kanatselos, C.; Manolatos, D.D.; Kouris, N.T.; Koniavitou, K.; Baltopoulos, P.; Lekakis, J.; Kremastinos, D.T. Studies on Apoptosis and Fibrosis in Skeletal Musculature: A Comparison of Heart Failure Patients with and without Cardiac Cachexia. International Journal of Cardiology 2003, 90, 107–113. [Google Scholar] [CrossRef]
- Lin, J.; Lopez, E.F.; Jin, Y.; Van Remmen, H.; Bauch, T.; Han, H.C.; Lindsey, M.L. Age-Related Cardiac Muscle Sarcopenia: Combining Experimental and Mathematical Modeling to Identify Mechanisms. Biophysical Journal 2008, 95, 5459–5467. [Google Scholar] [CrossRef] [PubMed]
- Cittadini, A.; Salzano, A.; Iacoviello, M.; Sirico, D.; D’Assante, R.; Giallauria, F.; Bossone, E.; Agostoni, P.; Passantino, A.; Perrone-Filardi, P. Multiple Hormonal and Metabolic Deficiency Syndrome Predicts Outcome in Heart Failure: The T.O.S.CA. Registry. European Journal of Preventive Cardiology 2021, 28, 1691–1700. [Google Scholar] [CrossRef]
- Hambrecht, R.; Schulze, P.C.; Gielen, S.; Linke, A.; Möbius-Winkler, S.; Erbs, S.; Kratzsch, J.; Schuler, G. Reduction of Insulin-Like Growth Factor-I Expression in the Skeletal Muscle of Noncachectic Patients with Chronic Heart Failure. Journal of the American College of Cardiology 2002, 39, 1175–1181. [Google Scholar] [CrossRef]
- Niebauer, J.; Pflaum, C.D.; Clark, A.L.; Strasburger, C.J.; Hooper, J.; Poole-Wilson, P.A.; Coats, A.J.S. Deficient Insulin-Like Growth Factor I in Chronic Heart Failure Predicts Altered Body Composition, Anabolic Deficiency, Cytokine and Neurohormonal Activation. Journal of the American College of Cardiology 1998, 32, 393–397. [Google Scholar] [CrossRef]
- Jankowska, E.A.; Biel, B.; Majda, J.; Banasiak, W.; Poole-Wilson, P.A.; Ponikowski, P. Anabolic Deficiency in Men with Chronic Heart Failure: Prevalence and Detrimental Impact on Survival. Circulation 2006, 114, 1829–1837. [Google Scholar] [CrossRef]
- Josiak, K.; Jankowska, E.A.; Piepoli, M.F.; Banasiak, W.; Ponikowski, P. Skeletal Myopathy in Patients with Chronic Heart Failure: Significance of Anabolic-Androgenic Hormones. Journal of Cachexia, Sarcopenia and Muscle 2014, 5, 287–296. [Google Scholar] [CrossRef]
- da Fonseca, G.W.P.; Dos Santos, M.R.; de Souza, F.R.; Takayama, L.; Rodrigues Pereira, R.M.; Alves, M.J.N.N.; Bacal, F.; Brum, P.C. Sympatho-Vagal Imbalance Is Associated with Sarcopenia in Male Patients with Heart Failure. Arquivos Brasileiros de Cardiologia 2019, 112, 739–746. [Google Scholar] [CrossRef]
- Gruson, D.; Ahn, S.A.; Ketelslegers, J.M.; Rousseau, M.F. Increased Plasma Myostatin in Heart Failure. European Journal of Heart Failure 2011, 13, 734–736. [Google Scholar] [CrossRef]
- Heineke, J.; Auger-Messier, M.; Xu, J.; Sargent, M.; York, A.; Welle, S.; Molkentin, J.D. Genetic Deletion of Myostatin from the Heart Prevents Skeletal Muscle Atrophy in Heart Failure. Circulation 2010, 121, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Breitbart, A.; Auger-Messier, M.; Molkentin, J.D.; Heineke, J. Myostatin from the Heart: Local and Systemic Actions in Cardiac Failure and Muscle Wasting. American Journal of Physiology – Heart and Circulatory Physiology 2011, 300, H1973–H1982. [Google Scholar] [CrossRef] [PubMed]
- Dick, S.A.; Epelman, S. Chronic Heart Failure and Inflammation. Circulation Research 2016, 119, 159–176. [Google Scholar] [CrossRef]
- Schaap, L.A.; Pluijm, S.M.F.; Deeg, D.J.H.; Visser, M. Higher Inflammatory Marker Levels in Older Persons: Associations with 5-Year Change in Muscle Mass and Muscle Strength. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 2009, 64, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Fulle, S.; Protasi, F.; Di Tano, G.; Pietrangelo, T.; Beltramin, A.; Boncompagni, S.; Vecchiet, J.; Fanò, G. The Contribution of Reactive Oxygen Species to Sarcopenia and Muscle Ageing. Experimental Gerontology 2004, 39, 17–24. [Google Scholar] [CrossRef]
- van der Pol, A.; van Gilst, W.H.; Voors, A.A.; van der Meer, P. Treating Oxidative Stress in Heart Failure: Past, Present and Future. European Journal of Heart Failure 2019, 21, 425–435. [Google Scholar] [CrossRef]
- Alem, M.M. Endothelial Dysfunction in Chronic Heart Failure: Assessment, Findings, Significance, and Potential Therapeutic Targets. International Journal of Molecular Sciences 2019, 20, 3198. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.F.; Barrett-O’Keefe, Z.; Nelson, A.D.; Garten, R.S.; Ryan, J.J.; Nativi-Nicolau, J.; Richardson, R.S.; Wray, D.W. Impaired Skeletal Muscle Vasodilation during Exercise in Heart Failure with Preserved Ejection Fraction. International Journal of Cardiology 2016, 211, 14–21. [Google Scholar] [CrossRef]
- Piepoli, M.F.; Crisafulli, A. Pathophysiology of Human Heart Failure: Importance of Skeletal Muscle Myopathy and Reflexes. Experimental Physiology 2014, 99, 609–615. [Google Scholar] [CrossRef]
- Houssiere, A.; Gujic, M.; Deboeck, G.; Ciarka, A.; Naeije, R.; Van De Borne, P. Increased Metaboreflex Activity Is Related to Exercise Intolerance in Heart Transplant Patients. American Journal of Physiology – Heart and Circulatory Physiology 2007, 293, H3699–H3706. [Google Scholar] [CrossRef]
- Sze, S.; Pellicori, P.; Kazmi, S.; Rigby, A.; Cleland, J.G.F. Prevalence and Prognostic Significance of Malnutrition Using 3 Scoring Systems among Outpatients with Heart Failure: A Comparison with Body Mass Index. JACC: Heart Failure 2018, 6, 476–486. [Google Scholar] [CrossRef]
- Poehlman, E.T.; Scheffers, J.; Gottlieb, S.S.; Fisher, M.L.; Vaitekevicius, P. Increased Resting Metabolic Rate in Patients with Congestive Heart Failure. Annals of Internal Medicine 1994, 121, 860–862. [Google Scholar] [CrossRef] [PubMed]
- İlhan, B.; Bahat, G.; Erdoğan, T.; Kiliç, C.; Karan, M.A. Anorexia Is Independently Associated with Decreased Muscle Mass and Strength in Community-Dwelling Older Adults. The Journal of Nutrition, Health & Aging 2019, 23, 202–206. [Google Scholar] [CrossRef]
- Sandek, A.; Doehner, W.; Anker, S.D.; von Haehling, S. Nutrition in Heart Failure: An Update. Current Opinion in Clinical Nutrition and Metabolic Care 2009, 12, 384–391. [Google Scholar] [CrossRef]
- Hussain, Z.; Swindle, J.; Hauptman, P.J. Digoxin Use and Digoxin Toxicity in the Post-DIG Trial Era. Journal of Cardiac Failure 2006, 12, 343–346. [Google Scholar] [CrossRef]
- Najm, A.; Niculescu, A.G.; Grumezescu, A.M.; Beuran, M. Emerging Therapeutic Strategies in Sarcopenia: An Updated Review on Pathogenesis and Treatment Advances. International Journal of Molecular Sciences 2024, 25, 4300. [Google Scholar] [CrossRef]
- Naseeb, M.A.; Volpe, S.L. Protein and Exercise in the Prevention of Sarcopenia and Aging. Nutrition Research 2017, 40, 1–20. [Google Scholar] [CrossRef]
- Aquilani, R.; Opasich, C.; Gualco, A.; Verri, M.; Boschi, F.; Febo, O.; Pasini, E. Adequate Energy-Protein Intake Is Not Enough to Improve Nutritional and Metabolic Status in Muscle-Depleted Patients with Chronic Heart Failure. European Journal of Heart Failure 2008, 10, 1127–1135. [Google Scholar] [CrossRef] [PubMed]
- Rozentryt, P.; von Haehling, S.; Lainscak, M.; Springer, J.; Taegtmeyer, H.; Anker, S.D. The Effects of a High-Caloric Protein-Rich Oral Nutritional Supplement in Patients with Chronic Heart Failure and Cachexia on Quality of Life, Body Composition, and Inflammation Markers: A Randomized, Double-Blind Pilot Study. Journal of Cachexia, Sarcopenia and Muscle 2010, 1, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.R.; Tan, Z.J.; Zhang, Q.; Gui, Q.F.; Yang, Y.M. The Effectiveness of Leucine on Muscle Protein Synthesis, Lean Body Mass and Leg Lean Mass Accretion in Older People: A Systematic Review and Meta-Analysis. British Journal of Nutrition 2015, 113, 25–34. [Google Scholar] [CrossRef]
- Chen, L.K.; Arai, H.; Assantachai, P.; Akishita, M.; Chew, S.T.H.; Dumlao, L.C.; Duque, G.; Woo, J.; Won, C.W.; Yamada, M.; Yoshida, H.; Yu, S.C.Y.; Zeng, P.; Zhang, T.; Zeng, Y. Roles of Nutrition in Muscle Health of Community-Dwelling Older Adults: Evidence-Based Expert Consensus from the Asian Working Group for Sarcopenia. Journal of Cachexia, Sarcopenia and Muscle 2022, 13, 1653–1672. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, A.M. Safety and Efficacy of High-Protein Diets for Weight Loss. Proceedings of the Nutrition Society 2012, 71, 339–349. [Google Scholar] [CrossRef]
- Ata, A.M.; Kara, M.; Ekiz, T.; Özçakar, L. Reassessing Sarcopenia in Hypertension: STAR and ACE Inhibitors Excel. International Journal of Clinical Practice 2021, 75, e13800. [Google Scholar] [CrossRef]
- Springer, J.; von Haehling, S. ACE Inhibitors and Sarcopenia: Covering All the BASEs? Drugs & Aging 2013, 30, 321–323. [Google Scholar]
- Júnior, M.; de Oliveira, M.R.; de Almeida, A.A.; de Souza, J.D.; de Oliveira, L.F.; de Oliveira, R.J. Associated Sarcopenia and Use of Diuretics. Clinical Interventions in Aging 2022, 17, 35–44. [Google Scholar]
- Zhou, L.S.; Xu, L.J.; Wang, X.Q.; Huang, Y.H.; Xiao, Q. Effect of Angiotensin-Converting Enzyme Inhibitors on Physical Function in Elderly Subjects: A Systematic Review and Meta-Analysis. Drugs & Aging 2015, 32, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Burton, L.A.; McMurdo, M.E.T.; Struthers, A.D. Mineralocorticoid Antagonism: A Novel Way to Treat Sarcopenia and Physical Impairment in Older People? Clinical Endocrinology 2011, 75, 725–729. [Google Scholar] [CrossRef]
- Farquharson, C.A.J.; Struthers, A.D. Spironolactone Increases Nitric Oxide Bioactivity, Improves Endothelial Vasodilator Dysfunction, and Suppresses Vascular Angiotensin I/Angiotensin II Conversion in Patients with Chronic Heart Failure. Circulation 2000, 101, 594–597. [Google Scholar] [CrossRef]
- Bendtzen, K.; Hansen, P.R.; Rieneck, K.; Nielsen, C.H.; Kharazmi, A.; Diamant, M. Spironolactone Inhibits Production of Proinflammatory Cytokines, Including Tumour Necrosis Factor-α and Interferon-γ, and Has Potential in the Treatment of Arthritis. Clinical and Experimental Immunology 2003, 134, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E. Pharmacologic Options for the Treatment of Sarcopenia. Calcified Tissue International 2016, 98, 319–333. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Lu, X.; Qian, Z.; Xu, W.; Zhou, X. New Insights into the Pathogenesis and Treatment of Sarcopenia in Chronic Heart Failure. Theranostics 2019, 9, 4019–4030. [Google Scholar] [CrossRef]
- Saitoh, M.; Ebner, N.; von Haehling, S.; Anker, S.D.; Springer, J. Therapeutic Considerations of Sarcopenia in Heart Failure Patients. Expert Review of Cardiovascular Therapy 2018, 16, 133–142. [Google Scholar] [CrossRef]
- Caminiti, G.; Volterrani, M.; Iellamo, F.; Marazzi, G.; Massaro, R.; Miceli, M.; Mammi, C.; Piepoli, M.; Rosano, G.M.C. Effect of Long-Acting Testosterone Treatment on Functional Exercise Capacity, Skeletal Muscle Performance, Insulin Resistance, and Baroreflex Sensitivity in Elderly Patients with Chronic Heart Failure: A Double-Blind, Placebo-Controlled, Randomized Study. Journal of the American College of Cardiology 2009, 54, 919–927. [Google Scholar] [CrossRef] [PubMed]
- Iellamo, F.; Volterrani, M.; Caminiti, G.; Marazzi, G.; Massaro, R.; Miceli, M.; Mammi, C.; Piepoli, M.; Rosano, G.M.C. Testosterone Therapy in Women with Chronic Heart Failure: A Pilot Double-Blind, Randomized, Placebo-Controlled Study. Journal of the American College of Cardiology 2010, 56, 1310–1316. [Google Scholar] [CrossRef]
- Solomon, Z.J.; Mirabal, J.R.; Mazur, D.J.; Kohn, T.P.; Lipshultz, L.I.; Pastuszak, A.W. Selective Androgen Receptor Modulators: Current Knowledge and Clinical Applications. Sexual Medicine Reviews 2019, 7, 84–94. [Google Scholar] [CrossRef] [PubMed]
- Dobs, A.S.; Boccia, R.V.; Croot, C.C.; Prokocimer, P.; Molnar, I.; Sinha, R.; Hancock, M.L.; Johnston, M.A.; Steiner, M.S. Effects of Enobosarm on Muscle Wasting and Physical Function in Patients with Cancer: A Double-Blind, Randomised Controlled Phase 2 Trial. The Lancet Oncology 2013, 14, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Papanicolaou, D.A.; Ather, S.N.; Zhu, H.; Zhou, Y.; Aroniadis, O.C.; Tsigos, C.; Barkan, A.L.; Bhasin, S. A Phase IIA Randomized, Placebo-Controlled Clinical Trial to Study the Efficacy and Safety of the Selective Androgen Receptor Modulator (SARM) MK-0773 in Female Participants with Sarcopenia. The Journal of Nutrition, Health & Aging 2013, 17, 533–543. [Google Scholar] [CrossRef]
- Nagaya, N.; Moriya, J.; Yasumura, Y.; Uematsu, M.; Ono, F.; Shimizu, W.; Ueno, K.; Kitakaze, M.; Miyatake, K.; Kangawa, K. Effects of Ghrelin Administration on Left Ventricular Function, Exercise Capacity, and Muscle Wasting in Patients with Chronic Heart Failure. Circulation 2004, 110, 3674–3679. [Google Scholar] [CrossRef]
- Becker, C.; Lord, S.R.; Studenski, S.A.; Smith, J.L.; Holcomb, R.; McDermott, M.M.; Gill, T.M.; Fielding, R.A.; Magaziner, J.; Newman, A.B. Myostatin Antibody (LY2495655) in Older Weak Fallers: A Proof-of-Concept, Randomised, Phase 2 Trial. The Lancet Diabetes & Endocrinology 2015, 3, 948–957. [Google Scholar] [CrossRef]
- Rooks, D.; Praestgaard, J.; Hariry, S.; Laurent, D.; Petricoul, O.; Perry, R.; Lach-Trifilieff, E.; Roubenoff, R. Treatment of Sarcopenia with Bimagrumab: Results from a Phase II, Randomized, Controlled, Proof-of-Concept Study. Journal of the American Geriatrics Society 2017, 65, 1988–1995. [Google Scholar] [CrossRef]
- Yamakage, H.; Tanaka, M.; Inoue, T.; Odori, S.; Kusakabe, T.; Satoh-Asahara, N. Effects of Dapagliflozin on the Serum Levels of Fibroblast Growth Factor 21 and Myokines and Muscle Mass in Japanese Patients with Type 2 Diabetes: A Randomized, Controlled Trial. Journal of Diabetes Investigation 2020, 11, 653–661. [Google Scholar] [CrossRef]
- Hu, Y.; Li, Y.; Ma, H.; Zhang, X.; Liu, Y.; Wang, J.; Chen, L. Prevalence and Prognostic Differences between Sarcopenia and Sarcopenic Obesity in Heart Failure. The American Journal of Cardiology 2025, 257, 127–130. [Google Scholar] [CrossRef]
- Baik, J.; Lee, Y.S. Emerging Role of Myostatin Inhibitors in the Management of Glucagon-Like Peptide-1-Associated Sarcopenia and Metabolic Disorders. Journal of Bone Metabolism 2025, 32, 263–275. [Google Scholar] [CrossRef]
- von Haehling, S.; Ebner, N.; Dos Santos, M.R.; Springer, J.; Anker, S.D. Muscle Wasting and Cachexia in Heart Failure: Mechanisms and Therapies. Nature Reviews Cardiology 2017, 14, 323–341. [Google Scholar] [CrossRef] [PubMed]
- Sato, R.; Vatic, M.; da Fonseca, G.W.P.; Anker, S.D.; von Haehling, S. Biological Basis and Treatment of Frailty and Sarcopenia. Cardiovascular Research 2024, 120, 982–998. [Google Scholar] [CrossRef]
- Cho, J.; Choi, Y.; Sajgalik, P.; Kim, D.; Park, J.; Lee, S.; Lee, J.; Park, S. Exercise as a Therapeutic Strategy for Sarcopenia in Heart Failure: Insights into Underlying Mechanisms. Cells 2020, 9, 2284. [Google Scholar] [CrossRef]
- Gielen, S.; Sandri, M.; Kozarez, I.; Kratzsch, J.; Teupser, D.; Thiery, J.; Erbs, S.; Mangner, N.; Lenk, K.; Hambrecht, R.; Schuler, G. Heart Failure Exercise Training Attenuates MuRF-1 Expression in the Skeletal Muscle of Patients with Chronic Heart Failure Independent of Age: The Randomized Leipzig Exercise Intervention in Chronic Heart Failure and Aging Catabolism Study. Circulation 2012, 125, 2716–2727. [Google Scholar] [CrossRef]
- Lenk, K.; Erbs, S.; Höllriegel, R.; Beck, E.; Linke, A.; Gielen, S.; Hambrecht, R.; Schuler, G. Exercise Training Leads to a Reduction of Elevated Myostatin Levels in Patients with Chronic Heart Failure. European Journal of Preventive Cardiology 2012, 19, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Lelyavina, T.A.; Galenko, V.L.; Ivanova, O.A.; Shlyakhto, E.V.; Mareev, V.Y.; Mareev, Y.V.; Malovichko, S.I.; Belenkov, Y.N. Clinical Response to Personalized Exercise Therapy in Heart Failure Patients with Reduced Ejection Fraction Is Accompanied by Skeletal Muscle Histological Alterations. International Journal of Molecular Sciences 2019, 20, 5514. [Google Scholar] [CrossRef]
- O’Connor, C.M.; Whellan, D.J.; Lee, K.L.; Keteyian, S.J.; Cooper, L.S.; Ellis, S.J.; Leifer, E.S.; Kraus, W.E.; Kitzman, D.W.; Blumenthal, J.A.; Rendall, D.S.; Houston-Miller, N.; Fleg, J.L.; Schulman, K.A.; Piña, I.L. Efficacy and Safety of Exercise Training in Patients with Chronic Heart Failure: HF-ACTION Randomized Controlled Trial. JAMA 2009, 301, 1439–1450. [Google Scholar] [CrossRef]
- Williams, A.D.; Carey, M.F.; Selig, S.; Hayes, A.; Krum, H.; Patterson, J.; Geerling, R.H.; Hare, D.L. Circuit Resistance Training in Chronic Heart Failure Improves Skeletal Muscle Mitochondrial ATP Production Rate—A Randomized Controlled Trial. Journal of Cardiac Failure 2007, 13, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Pu, C.T.; Johnson, M.T.; Forman, D.E.; et al. Randomized Trial of Progressive Resistance Training to Counteract the Myopathy of Chronic Heart Failure. J. Appl. Physiol. 2001, 90, 2341–2347. [Google Scholar] [CrossRef]
- Lu, L.; Mao, L.; Feng, Y.; Ainsworth, B.E.; Liu, Y.; Chen, N. Effects of Different Exercise Training Modes on Muscle Strength and Physical Performance in Older People with Sarcopenia: A Systematic Review and Meta-Analysis. BMC Geriatr. 2021, 21, 708. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov. Whey Protein Supplementation During Cardiac Rehabilitation in Heart Failure (PROT-HF). Preprint Posted Online. 2017. Available online: https://clinicaltrials.gov/ct2/show/NCT03004833.
- ClinicalTrials.gov. A SkeleTal Muscle Recovery Intervention with Dietary Protein in Heart Failure (ASTRID-HF). Preprint Posted Online. 2022. Available online: https://clinicaltrials.gov/ct2/show/NCT05434976.
- ClinicalTrials.gov. STRIVE Trial: Whey Protein Supplementation and Resistance Training in HFpEF. Preprint Posted Online. 2024. Available online: https://clinicaltrials.gov/ct2/show/NCT06301560.
- Curcio, F.; Testa, G.; Liguori, I.; Papalia, R.; Della-Morte, D.; Gargiulo, G.; Cacciatore, F.; Bonaduce, D.; Abete, P. Sarcopenia and Heart Failure. Nutrients 2020, 12, 211. [Google Scholar] [CrossRef] [PubMed]
| Therapeutic approach | Targeted mechanism | Evidence and clinical impact |
Limitations and considerations |
|---|---|---|---|
| Resistance and aerobic exercise training | Stimulation of muscle hypertrophy; enhancement of mitochondrial function and skeletal muscle perfusion | Improves muscle strength, peak oxygen uptake (VO₂peak), and quality of life | Limited feasibility in patients with advanced heart failure or severe frailty |
| Nutritional support (adequate protein and caloric intake) | Correction of malnutrition; promotion of muscle protein synthesis | Associated with improvements in muscle mass and functional outcomes | Evidence remains heterogeneous; individualized nutritional assessment is required |
| Micronutrient supplementation (e.g., iron, vitamin D) | Correction of specific deficiencies affecting skeletal muscle metabolism | Iron repletion improves exercise capacity; vitamin D supplementation may support muscle function | Clinical benefit depends on the presence and severity of baseline deficiency |
| Anabolic hormone–based therapies (testosterone, growth hormone, insulin-like growth factor 1) | Restoration of anabolic–catabolic balance; stimulation of muscle protein synthesis | May improve muscle mass, strength, and exercise capacity in carefully selected patients | Safety concerns; limited efficacy in unselected populations; lack of robust outcome data |
| Anti-inflammatory strategies | Reduction of cytokine-mediated catabolic processes | Potential to attenuate muscle wasting | Clinical trials have not demonstrated consistent clinical benefit |
| Myostatin inhibition | Suppression of negative regulation of skeletal muscle growth | Demonstrates favorable effects on muscle mass in experimental models | Limited clinical evidence; long-term safety remains uncertain |
| Appetite stimulation and management of anorexia | Enhancement of caloric intake | May support nutritional status and preservation of muscle mass | Limited heart failure–specific evidence |
| Multidisciplinary rehabilitation programs | Integrated management of physical, nutritional, and psychosocial factors | Improves functional capacity and quality of life | Resource-intensive; availability varies across healthcare settings |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




