Submitted:
25 March 2026
Posted:
26 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
General Objective
Specific Objectives
Methods
Study Design and Setting
Participants
Data Collection and Variables
Behavioral Phenotyping (EFCA)
Anthropometric Outcomes
Statistical Analysis
Within-Medication Change and Effect Size (Cohen’s dz)
Weight Loss Models and Association with Behavioral Change
3. Results
Cohort Overview and Analytic Approach
Weight Loss Benchmarking Across Pharmacotherapies
Cardiometabolic Response Among Responders
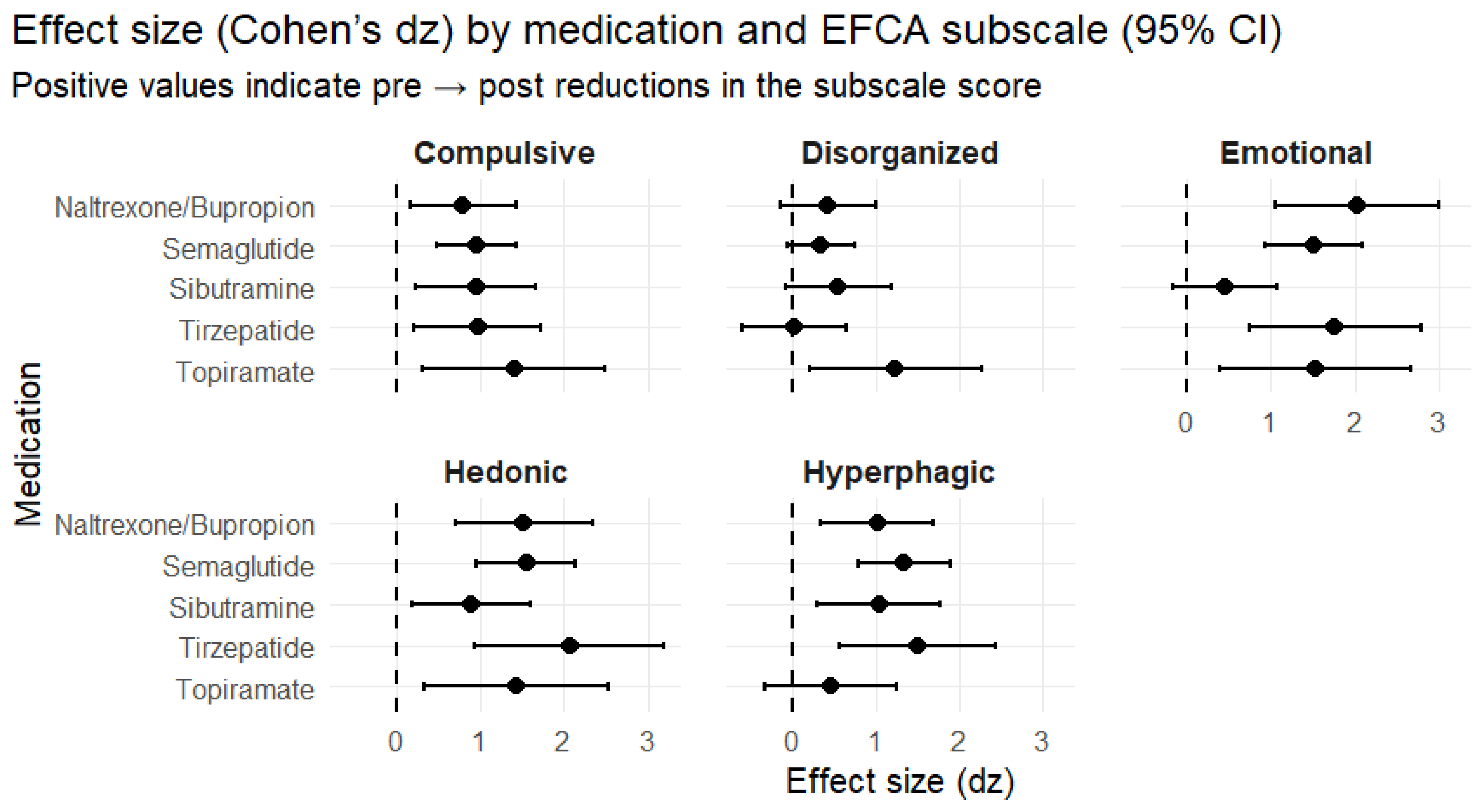
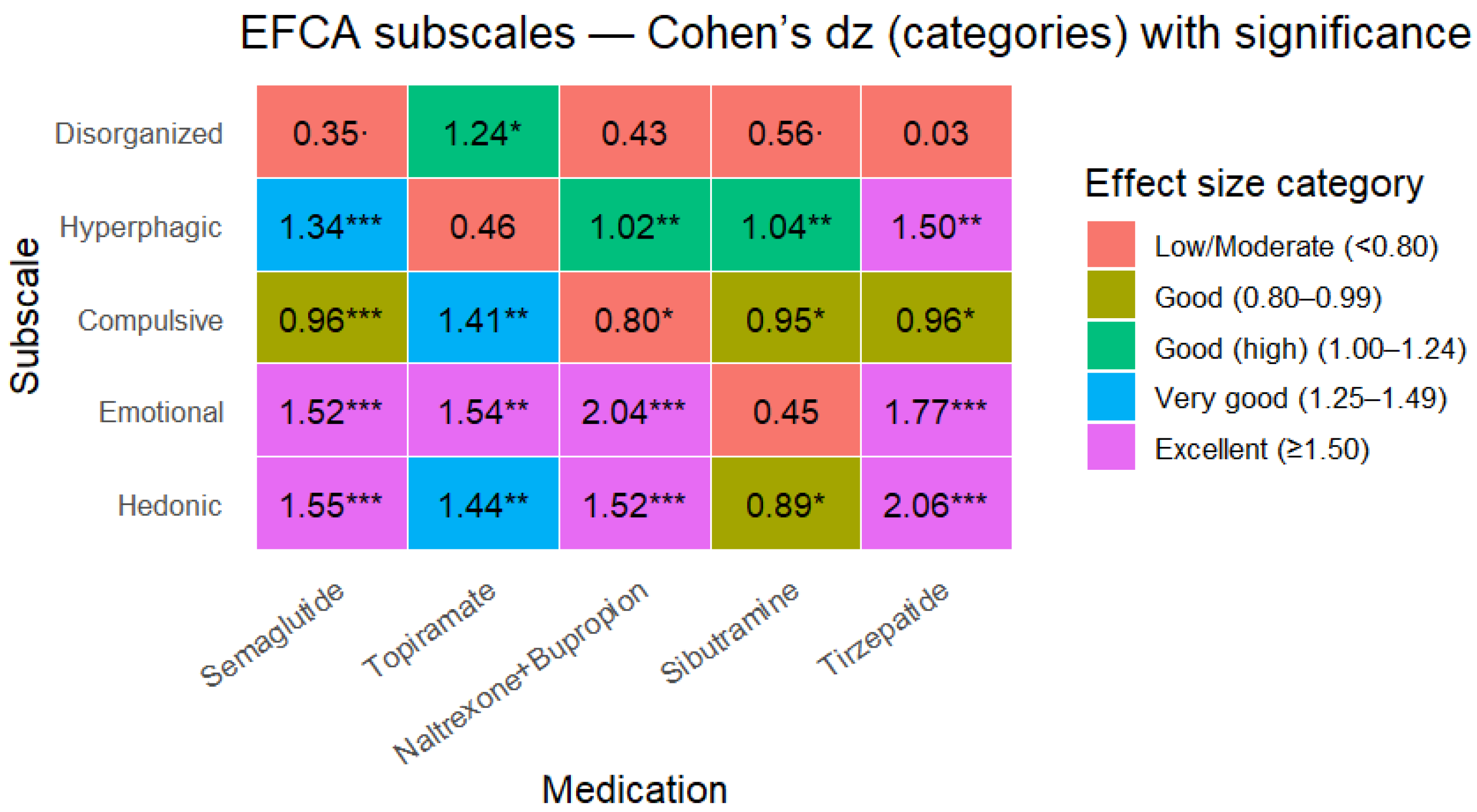
EFCA Subscale Changes Show Distinct “Behavioral Pharmacodynamics”
- Emotional eating: naltrexone/bupropion dz 2.04 (1.06-3.01), p<0.001; tirzepatide dz 1.77 (0.74-2.80), p<0.001; semaglutide dz 1.52 (0.94-2.10), p<0.001; topiramate dz 1.54 (0.40-2.68), p=0.0066.
- Hedonic eating: tirzepatide dz 2.06 (0.93-3.20), p<0.001; semaglutide dz 1.55 (0.96-2.13), p<0.001; naltrexone/bupropion dz 1.52 (0.70-2.34), p<0.001; topiramate dz 1.44 (0.34-2.53), p=0.009.
- Hyperphagic pattern: tirzepatide dz 1.50 (0.57-2.43), p=0.001; semaglutide dz 1.34 (0.80-1.89), p<0.001; sibutramine dz 1.04 (0.29-1.78), p=0.006; naltrexone/bupropion dz 1.02 (0.34-1.70), p=0.003.
- Compulsive pattern: topiramate dz 1.41 (0.32-2.49), p=0.0099; tirzepatide dz 0.96 (0.20-1.73), p=0.014; semaglutide dz 0.96 (0.48-1.44), p<0.001; sibutramine dz 0.95 (0.22-1.67), p=0.011.
- Disorganized eating: effects were inconsistent; topiramate showed a large dz (1.24) but with wide CI, while tirzepatide was essentially neutral (dz 0.03; p=0.93).
4. Discussion
5. Conclusions
Abbreviations
| EFCA | Eating Behavior Phenotypical Scale |
| AOM | Anti-Obesity Medication |
References
- Endocrinology, T.L.D. Redefining obesity: advancing care for better lives. Lancet Diabetes Endocrinol.;PubMed 2025, 13, 75. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.A.; Christiansen, P.; Halford, J.C. Pharmaceutical approaches to weight management: behavioural mechanisms of action. Curr Opin Physiol. 2019, 12, 26–32. [Google Scholar] [CrossRef]
- Apovian, C.M.; Aronne, L.J.; Bessesen, D.H.; McDonnell, M.E.; Murad, M.H.; Pagotto, U.; et al. Pharmacological management of obesity: an endocrine Society clinical practice guideline. J Clin Endocrinol Metab.;PubMed 2015, 100, 342–62. [Google Scholar] [CrossRef] [PubMed]
- Diretriz Brasileira de Tratamento Farmacológico da Obesidade - ABESO 2026 [Internet]. 4 Jan 2026. Available online: https://abeso.org.br/wp-content/uploads/2025/12/Diretriz-Brasileira-de-Tratamento-Farmacologico-da-Obesidade-ABESO-2026.pdf.
- Anger, V.E.; Formoso, J.; Katz, M.T. Scale of Eating Behavior Phenotypes (EFCA), confirmatory factor analysis and psychometric properties]. Nutr Hosp.;PubMed 2022, 39, 405–10. [Google Scholar] [CrossRef] [PubMed]
- Pineda-Wieselberg, R.J.; Soares, A.H.; Napoli, T.F.; Anger, V.E.; Formoso, J.; Sarto, M.L.L.; et al. Validation for Brazilian Portuguese of the Eating Behavior Phenotypes Scale (EFCA): confirmatory factor analysis and psychometric properties. Arch Endocrinol Metab. 2025, 69, e240404. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bouhlal, S.; McBride, C.M.; Trivedi, N.S.; Agurs-Collins, T.; Persky, S. Identifying eating behavior phenotypes and their correlates: a novel direction toward improving weight management interventions. Appetite 2017, 111, 142–50. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Macht, M. How emotions affect eating: A five-way model. Appetite 2008, 50, 1–11. [Google Scholar] [CrossRef]
- Acosta, A.; Camilleri, M.; Abu Dayyeh, B.; Calderon, G.; Gonzalez, D.; McRae, A.; et al. Selection of Antiobesity Medications Based on Phenotypes Enhances Weight Loss: A Pragmatic Trial in an Obesity Clinic. Obes Silver Spring Md. 2021, 29, 662–71. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kushner, R.F.; Calanna, S.; Davies, M.; Dicker, D.; Garvey, W.T.; Goldman, B.; et al. Semaglutide 2.4 mg for the Treatment of Obesity: Key Elements of the STEP Trials 1 to 5. Obes Silver Spring Md. 2020, 28, 1050–61. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Knop, F.K.; Aroda, V.R.; do Vale, R.D.; Holst-Hansen, T.; Laursen, P.N.; Rosenstock, J.; et al. Oral semaglutide 50 mg taken once per day in adults with overweight or obesity (OASIS 1): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Lond Engl.;PubMed 2023, 402, 705–19. [Google Scholar] [CrossRef] [PubMed]
- Wharton, S.; Lingvay, I.; Bogdanski, P.; Duque do Vale, R.; Jacob, S.; Karlsson, T.; et al. Oral Semaglutide at a Dose of 25 mg in Adults with Overweight or Obesity. N Engl J Med.;PubMed 2025, 393, 1077–87. [Google Scholar] [CrossRef] [PubMed]
- Greenway, F.L.; Fujioka, K.; Plodkowski, R.A.; Mudaliar, S.; Guttadauria, M.; Erickson, J.; et al. Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. The Lancet.;PubMed 2010, 376, 595–605. [Google Scholar] [CrossRef] [PubMed]
- Apovian, C.M.; Aronne, L.; Rubino, D.; Still, C.; Wyatt, H.; Burns, C.; et al. A randomized, phase 3 trial of naltrexone SR/bupropion SR on weight and obesity-related risk factors (COR-II). Obes Silver Spring Md. 2013, 21, 935–43. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wadden, T.A.; Foreyt, J.P.; Foster, G.D.; Hill, J.O.; Klein, S.; O’Neil, P.M.; et al. Weight Loss With Naltrexone SR/Bupropion SR Combination Therapy as an adjunct to Behavior Modification: The COR-BMOD Trial. Obes Silver Spring Md 2011, 19, 110–20. [Google Scholar] [CrossRef] [PubMed Central]
- James, W.P.T.; Astrup, A.; Finer, N.; Hilsted, J.; Kopelman, P.; Rössner, S.; et al. Effect of sibutramine on weight maintenance after weight loss: a randomised trial. The Lancet.;PubMed 2000, 356, 2119–25. [Google Scholar] [CrossRef] [PubMed]
- James, W.P.T.; Caterson, I.D.; Coutinho, W.; Finer, N.; Gaal, L.F.V.; Maggioni, A.P.; et al. Effect of Sibutramine on Cardiovascular Outcomes in Overweight and Obese Subjects. N Engl J Med. 2010, 363, 905–17. [Google Scholar] [CrossRef]
- Kazerooni, R.; Lim, J. Topiramate-Associated Weight Loss in a Veteran Population. Mil Med.;PubMed 2016, 181, 283–6. [Google Scholar] [CrossRef] [PubMed]
- Jastreboff, A.M.; le Roux, C.W.; Stefanski, A.; Aronne, L.J.; Halpern, B.; Wharton, S.; et al. Tirzepatide for Obesity Treatment and Diabetes Prevention. N Engl J Med.;PubMed 2025, 392, 958–71. [Google Scholar] [CrossRef] [PubMed]
- Ard, J.; Lee, C.J.; Gudzune, K.; Addison, B.; Lingvay, I.; Cao, D.; et al. Weight reduction over time in tirzepatide-treated participants by early weight loss response: Post hoc analysis in SURMOUNT-1. Diabetes Obes Metab. 2025, 27, 5064–71. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Amy Bartee, T.T. Early Weight Loss and Cardiometabolic Parameters in Tirzepatide-Treated Participants in SURMOUNT 1-2 [Apresentação Oral]. Apresentação Oral presented at: Obesity Week. 2024 Nov 3.
- Morton, G.J.; Meek, T.H.; Schwartz, M.W. Neurobiology of food intake in health and disease. Nat Rev Neurosci. 2014, 15, 367–78. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gudzune, K.A.; Kushner, R.F. Medications for Obesity: A Review. JAMA 2024, 332, 571–84. [Google Scholar] [CrossRef] [PubMed]
- Heck, A.M.; Yanovski, J.A.; Calis, K.A. Orlistat, a New Lipase Inhibitor for the Management of Obesity. Pharmacotherapy 2000, 20, 270–9. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wharton, S.; Lau, D.C.W.; Vallis, M.; Sharma, A.M.; Biertho, L.; Campbell-Scherer, D.; et al. Obesity in adults: a clinical practice guideline. CMAJ.;PubMed 2020, 192, E875–91. [Google Scholar] [CrossRef] [PubMed]
- Breen, C.; O’Connell, J.; Geoghegan, J.; O’Shea, D.; Birney, S.; Tully, L.; et al. Obesity in Adults: A 2022 Adapted Clinical Practice Guideline for Ireland. Obes Facts 2022, 15, 736–52. [Google Scholar] [CrossRef]
- Gasoyan, H.; Butsch, W.S.; Casacchia, N.J.; Schulte, R.; Criswell, V.; Fox, J.; et al. Reasons for Discontinuation of Obesity Pharmacotherapy With Semaglutide or Tirzepatide in Clinical Practice. Obesity 2025, 33, 2296–303. [Google Scholar] [CrossRef]
- Eli Lilly and Company. A Phase 3b, Randomized, Double-Blind, Placebo-Controlled Study to Evaluate the Efficacy and Safety of Orforglipron Once Daily Versus Placebo for Maintenance of Body Weight Reduction in Participants Who Have Obesity or Overweight With Weight-Related Comorbidities [Clinical trial registration] [Internet] Report No.: NCT06584916. clinicaltrials.gov. 2025 Dec [cited 2026 Mar 10. Available online: https://clinicaltrials.gov/study/NCT06584916.
- Smith, S.M.; Meyer, M.; Trinkley, K.E. Phentermine/topiramate for the treatment of obesity. Ann Pharmacother. 2013, 47, 340–9. [Google Scholar] [CrossRef] [PubMed]
- Cercato, C.; Stumpf, M.A.M.; da Cunha Freire, G.N.; Kawahara, E.Z.; Fernandes, A.E.; de Melo, M.E.; et al. Combination of sibutramine and topiramate for the treatment of obesity: the SIBAMATE retrospective cohort study. Diabetol Metab Syndr. 2025, 17, 289. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
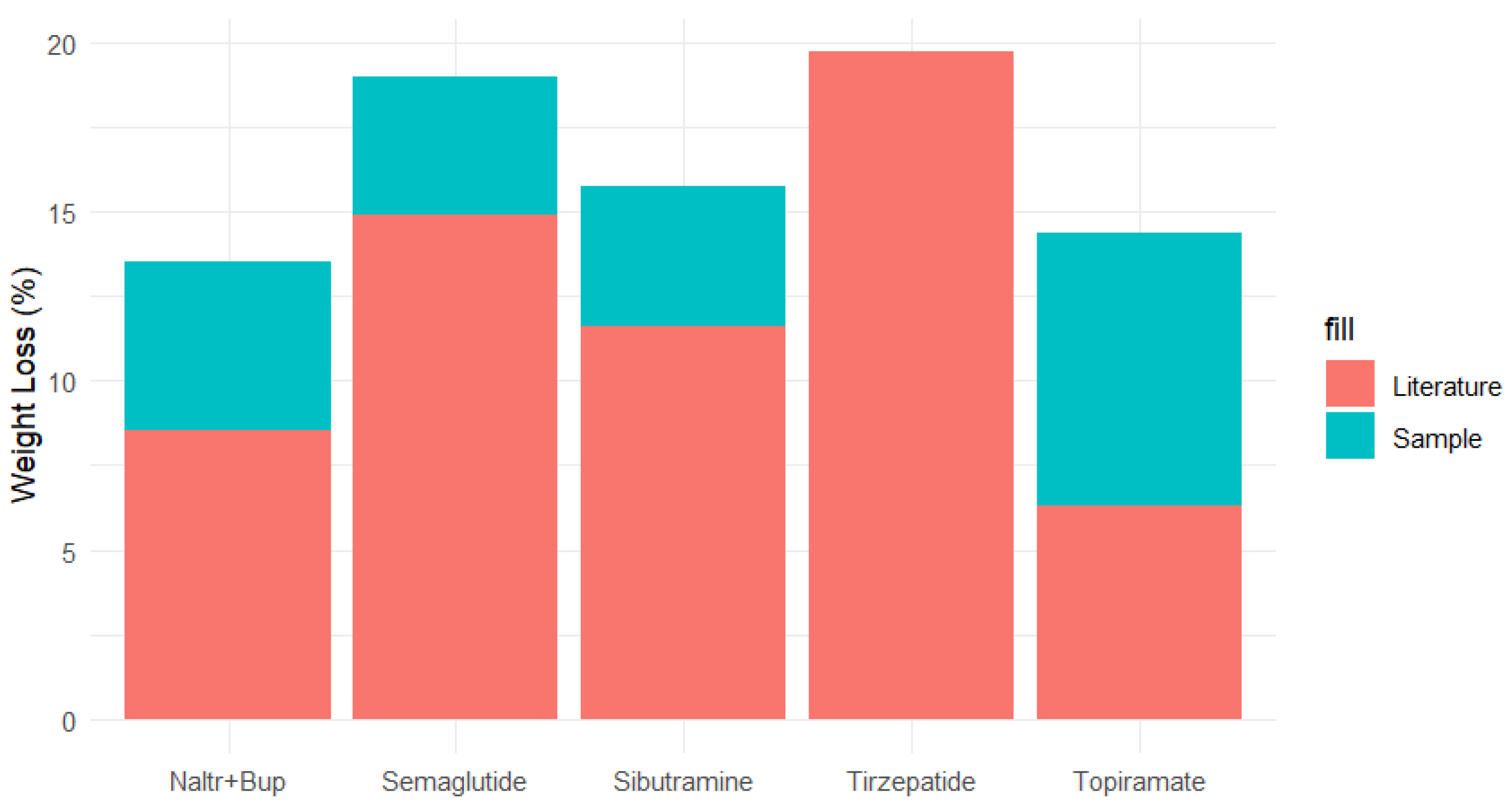
| AOM | Participants (n, %) |
| Semaglutide | 25 (37.8%) |
| Naltrexone/Bupropion | 13 (19.7%) |
| Sibutramine | 11 (16.7%) |
| Tirzepatide | 10 (15.1%) |
| Topiramate | 7 (10.6%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




