Submitted:
24 March 2026
Posted:
26 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
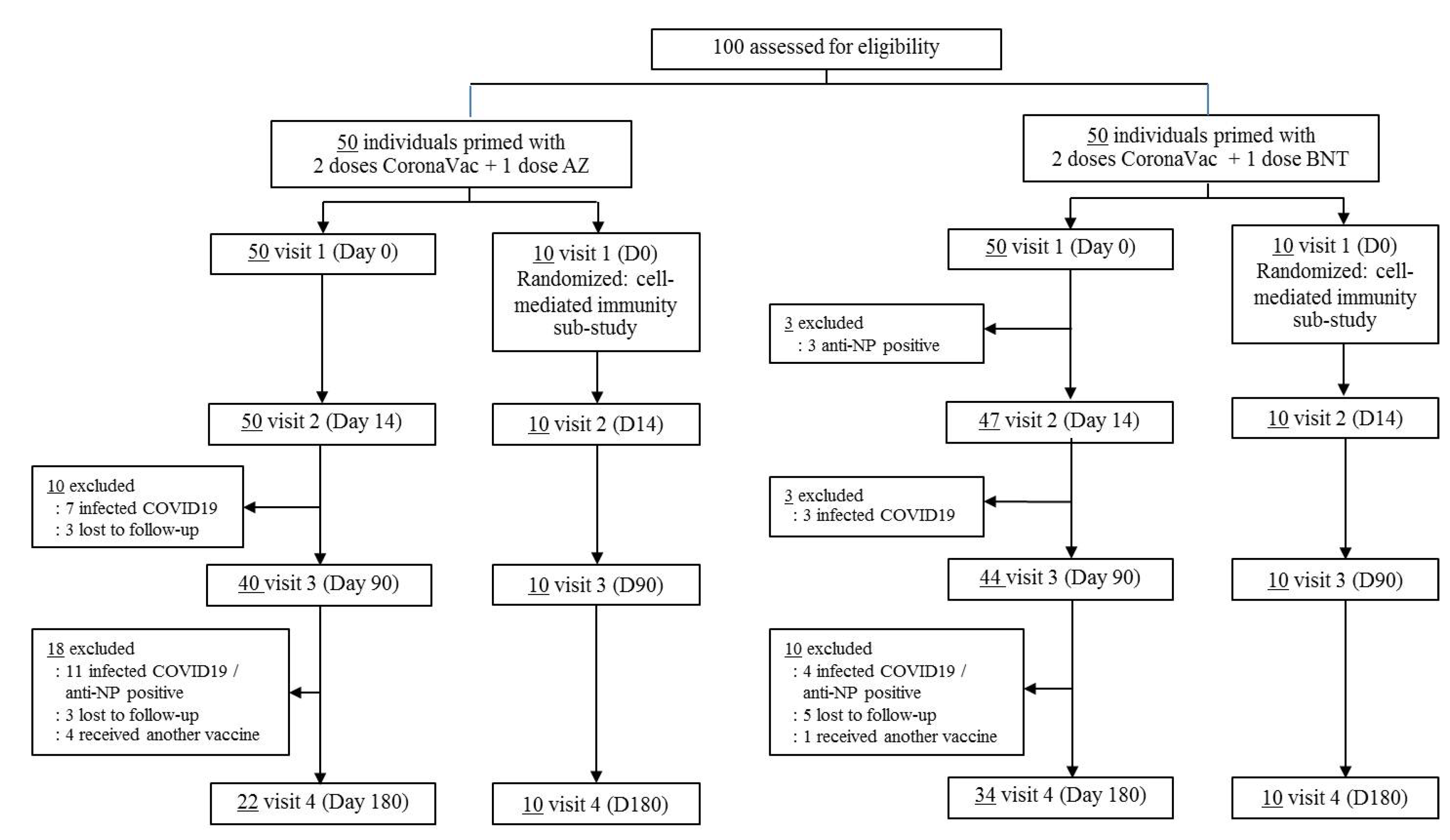
2.1. Ethics Approval, Study Design, and Participants
2.2. Study Procedures
2.3. Immunogenicity Outcomes
2.3.1. Quantitative Receptor Binding Domain IgG (anti-RBD IgG) and Anti-Neucleocapsid Protein (anti-Np)
2.3.2. Surrogate Virus Neutralization Test (sVNT)
2.3.3. Interferon-Gamma (IFN-γ) Release Assay (IGRA) to Evaluate T Cell Responses
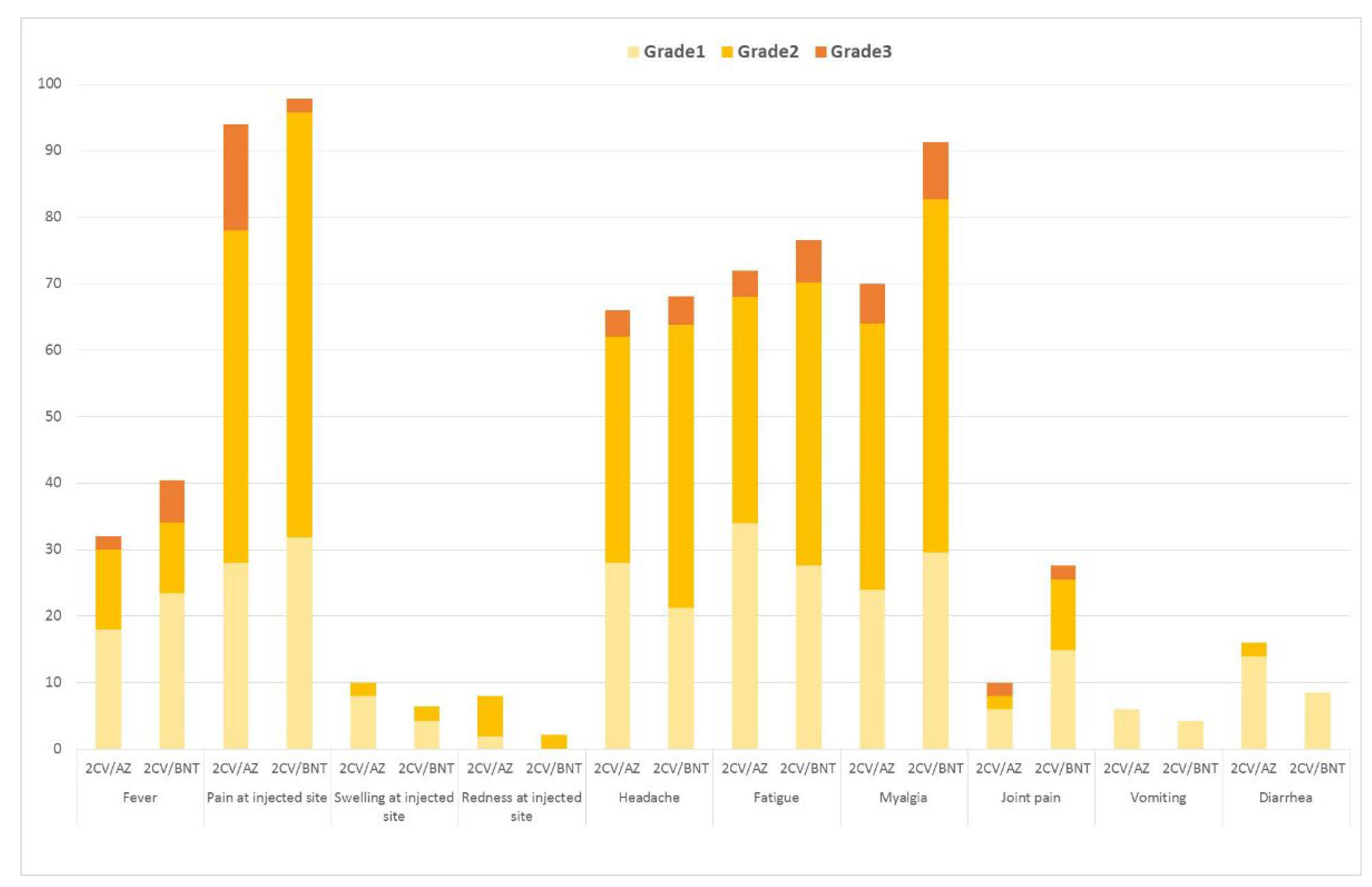
2.4. Reactogenicity
2.5. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Reactogenicity
3.3. Immunogenicity
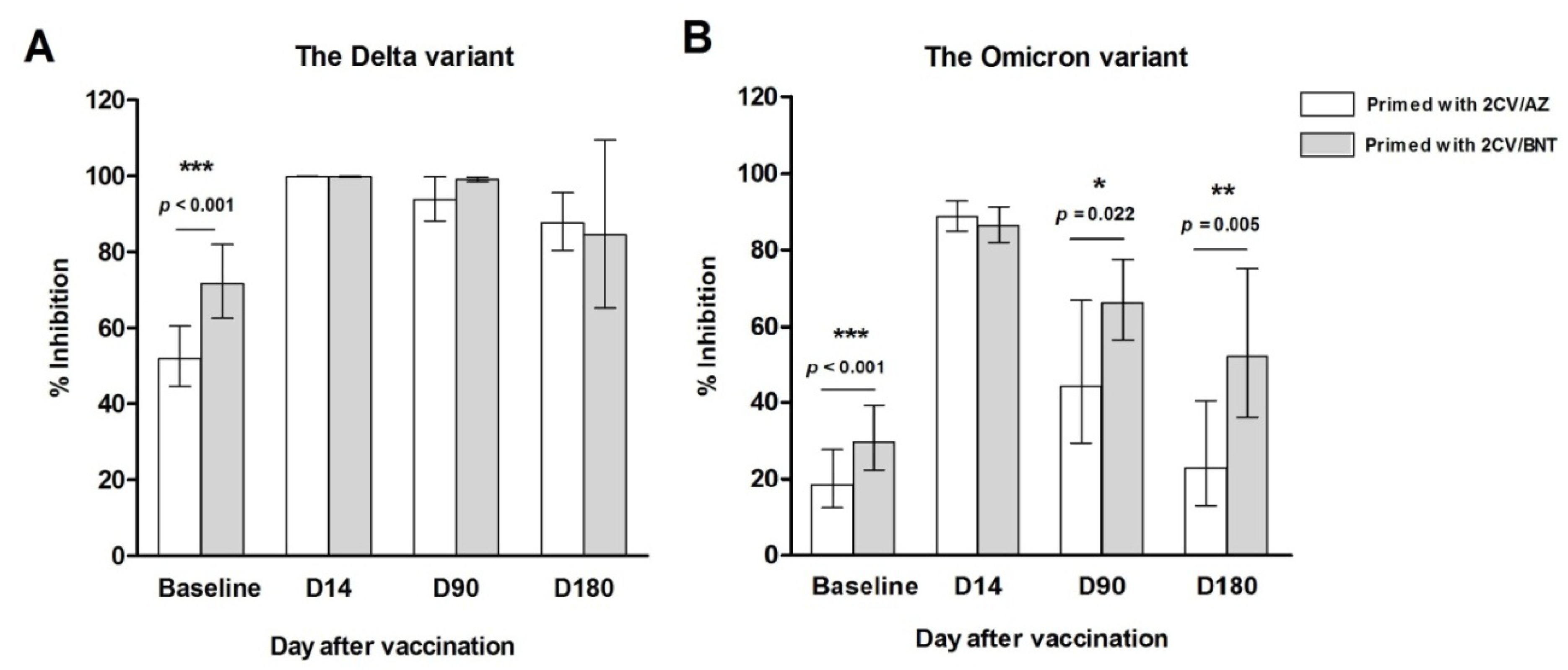
3.3.1. SARS-CoV-2 Neutralizing Antibody by Surrogate Virus Neutralization Test (sVNT)
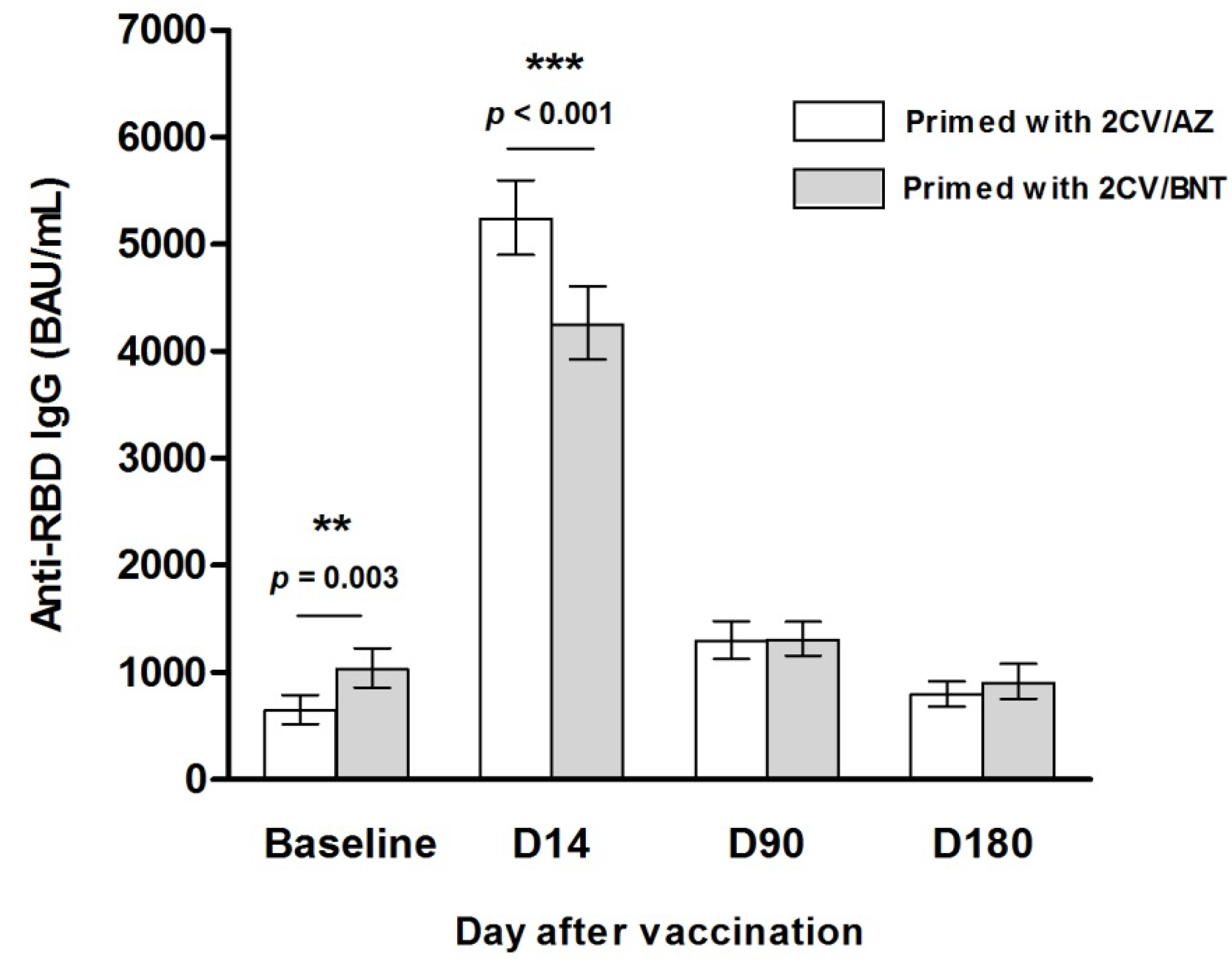
3.3.2. Quantitative IgG Against Receptor-Binding Domain (anti-RBD IgG)
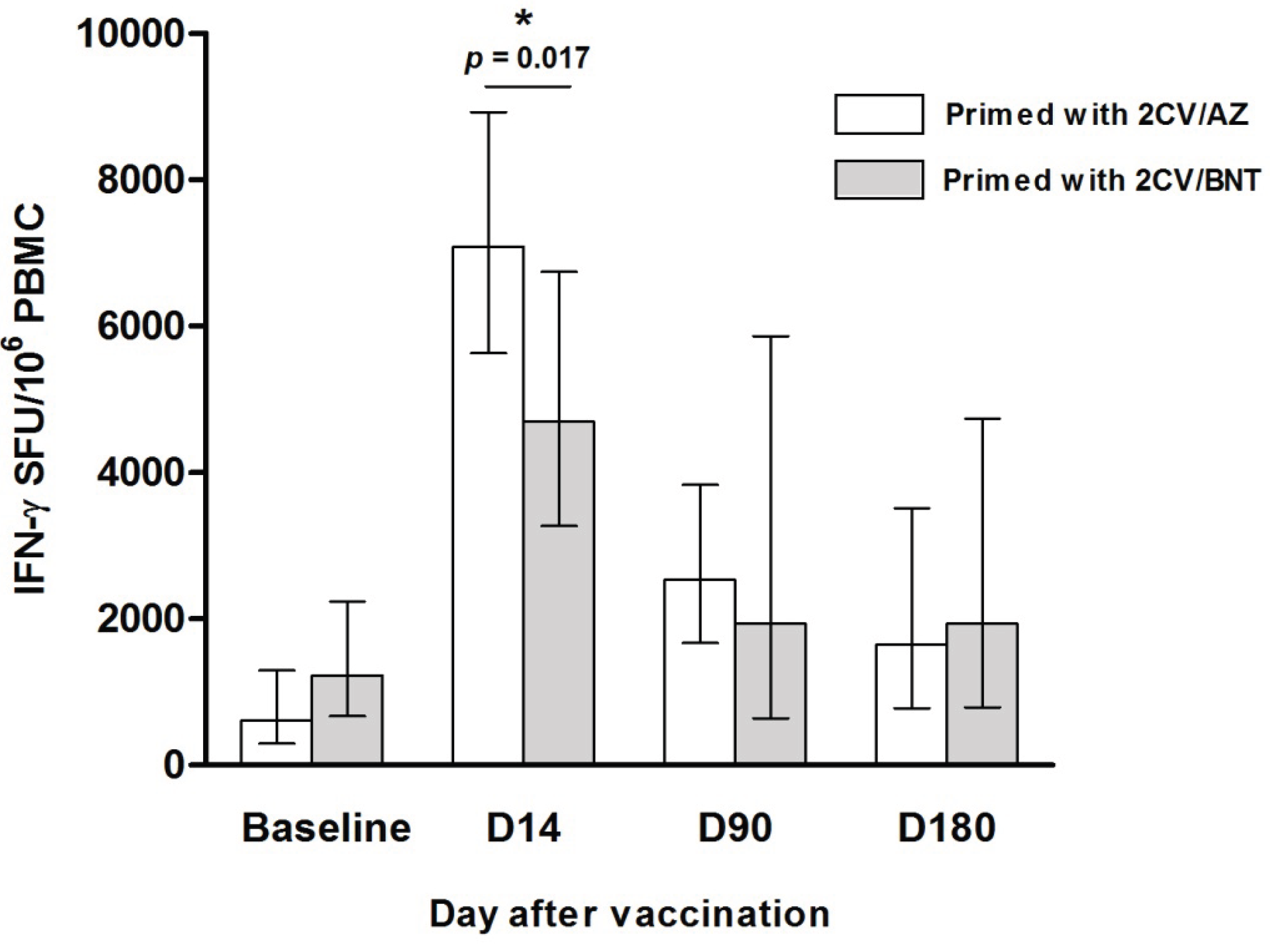
3.4. Cell-Mediated Immune Response by IGRA Assay
4. Discussion
5. Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A novel coronavirus from patients with pneumonia in China. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Novel Coronavirus—Thailand (ex-China). Available online: https://www.who.int/emergencies/disease-outbreak-news/item/2020-DON234 (accessed on 24 March 2022).
- WHO. COVID-19Weekly Epidemiological Update Edition 68; WHO: Geneva, Switzerland, 2021; Available online: https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---30-november-2021 (accessed on 7 December 2021).
- Yamasoba, D.; Kimura, I.; Nasser, H.; Morioka, Y.; Nao, N.; Ito, J.; Uriu, K.; Tsuda, M.; Zahradnik, J.; Shirakawa, K.; et al. Virological characteristics of the SARS-CoV-2 Omicron BA.2 spike. Cell 2022, 185. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Draft Landscape of COVID-19 Candidate Vaccines. Available online: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines (accessed on 20 October 2020).
- Zhang, Y.; Zeng, G.; Pan, H.; Li, C.; Hu, Y.; Chu, K.; et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18-59 years: a randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infect Dis. 2021, 21(2), 181–92. [Google Scholar] [CrossRef] [PubMed]
- Jara, A.; Undurraga, E.A.; Gonzalez, C.; Paredes, F.; Fontecilla, T.; Jara, G.; Pizarro, A.; Acevedo, J.; Leo, K.; Leon, F.; et al. Effectiveness of an Inactivated SARS-CoV-2 Vaccine in Chile. N. Engl. J. Med. 2021, 385, 875–884. [Google Scholar] [CrossRef]
- WHO. Interim Statement on COVID-19 Vaccine Booster Doses 2021. 10 August 2021. Available online: https://www.who.int/news/item/10-08-2021-interim-statement-on-covid-19-vaccine-booster-doses (accessed on 29 August 2021).
- Jantarabenjakul, W.; Chantasrisawad, N.; Puthanakit, T.; Wacharapluesadee, S.; Hirankarn, N.; Ruenjaiman, V.; Paitoonpong, L.; Suwanpimolkul, G.; Torvorapanit, P.; Pradit, R.; et al. Short-term immune response after inactivated SARS-CoV-2 (CoronaVac(R), Sinovac) and AZD1222nCoV-19 (Vaxzevria(R), Oxford-AstraZeneca) vaccinations in health care workers. Asian Pac. J. Allergy Immunol. 2021, 40, 269–277. [Google Scholar]
- Feikin; Higdon, M.M.; Abu-Raddad, L.J.; Andrews, N.; Araos, R.; Goldberg, Y.; Groome, M.J.; Huppert, A.; O'Brien, K.L.; Smith, P.G.; Wilder-Smith, A.; Zeger, S.; Deloria Knoll, M.; Patel, M.K. Duration of effectiveness of vaccines against SARS-CoV-2 infection and COVID-19 disease: results of a systematic review and meta-regression. Lancet 2022, 399(10328), 924–944. [Google Scholar] [CrossRef]
- Liu, Y.; Rocklöv, J. The effective reproductive number of the Omicron variant of SARS-CoV-2 is several times relative to Delta. J. Travel. Med. 2022, 29(3), taac037. [Google Scholar] [CrossRef]
- Andrews, N.; Stowe, J.; Kirsebom, F.; et al. Covid-19 Vaccine Effectiveness against the Omicron (B.1.1.529) Variant. N. Engl. J. Med. 2022, 386, 1532–1546. [Google Scholar] [CrossRef]
- Pulliam, J.R.C.; et al. Increased risk of SARS-CoV-2 reinfection associated with emergence of Omicron in South Africa. Science 2022, 376(6593), eabn4947. [Google Scholar] [CrossRef]
- Costa Clemens, S.A.; Weckx, L.; Clemens, R.; Almeida Mendes, A.V.; Ramos Souza, A.; Silveira, M.B.V.; et al. Heterologous versus homologous COVID-19 booster vaccination in previous recipients of two doses of CoronaVac COVID-19 vaccine in Brazil (RHH-001): a phase 4, non-inferiority, single blind, randomised study. Lancet 2022, 399(10324), 521–529. [Google Scholar] [CrossRef]
- Niyomnaitham, S.; Chokephaibulkit, K.; Pheerapanyawaranun, C.; Toh, Z.Q.; Licciardi, P.V.; Satayasanskul, A.; Jansarikit, L.; Assantachai, P. Immunogenicity of BNT162b2 as a first booster after a ChAdOx1 primary series in a Thai geriatric population living with frailty. J. Nutr. Health. Aging. 2024, 28(8), 100315. [Google Scholar] [CrossRef] [PubMed]
- Wanlapakorn, N.; Suntronwong, N.; Kanokudom, S.; Assawakosri, S.; Nilyanimit, P.; Yorsaeng, R.; Chansaenroj, J.; Poovorawan, Y. Immunogenicity of the BNT162b2 COVID-19 vaccine as a third dose (booster) following two doses of different primary series regimens in Thailand. Pathog. Glob. Health 2022, 116(7), 395–397. [Google Scholar] [CrossRef] [PubMed]
- Barros-Martins, J.; Hammerschmidt, S.I.; Cossmann, A.; Odak, I.; Stankov, M.V.; Morillas Ramos, G.; Dopfer-Jablonka, A.; Heidemann, A.; Ritter, C.; Friedrichsen, M.; et al. Immune responses against SARS-CoV-2 variants after heterologous and homologous AZD1222nCoV-19/BNT162b2 vaccination. Nat. Med. 2021, 27, 1525–1529. [Google Scholar] [CrossRef] [PubMed]
- Munro, A.P.S.; Janani, L.; Cornelius, V.; Aley, P.K.; Babbage, G.; Baxter, D.; Bula, M.; Cathie, K.; Chatterjee, K.; Dodd, K.; et al. Safety and immunogenicity of seven covid-19 vaccines as a third dose (booster) following two doses of AZD1222ncov-19 or bnt162b2 in the uk (cov-boost): A blinded, multicentre, randomised, controlled, phase 2 trial. Lancet 2021, 398, 2258–2276. [Google Scholar] [CrossRef]
- Mbaeyi, S.; Oliver, S.E.; Collins, J.P.; Godfrey, M.; Goswami, N.D.; Hadler, S.C.; et al. The Advisory Committee on Immunization Practices' Interim Recommendations for Additional Primary and Booster Doses of COVID-19 Vaccines - United States, 2021. MMWR. Morb. Mortal. Wkly. Rep. 2021, 70(44), 1545–1552. [Google Scholar] [CrossRef]
- Rosenblum, H.G.; Wallace, M.; Godfrey, M.; Roper, L.E.; Hall, E.; Fleming-Dutra, K.E.; et al. Interim Recommendations from the Advisory Committee on Immunization Practices for the Use of Bivalent Booster Doses of COVID-19 Vaccines - United States, October 2022. MMWR. Morb. Mortal. Wkly. Rep. 2022, 71(45), 1436–1441. [Google Scholar] [CrossRef]
- Tangsathapornpong, A.; Nanthapisal, S.; Pontan, K.; Bunjoungmanee, P.; Neamkul, Y.; Boonyarangkul, A.; Wanpen, S.; Fukpho, W.; Jitpokasem, S.; Tharabenjasin, P.; Jaru-Ampornpan, P. Immunogenicity and safety of the third booster dose with mRNA-1273 COVID-19 vaccine after receiving two doses of inactivated or viral vector COVID-19 vaccine. Vaccines (Basel). 2023, 11(3), 553. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services; Department of Health and Human Services; Food and Drug Administration; CBER. Guidance for Industry Toxicity Grading Scale for Healthy Adult and Adolescent Volunteers Enrolled in Preventive Vaccine Clinical Trials September 2007. Available online: https://www.fda.gov/media/73679/download (accessed on 30 November 2021).
- Zou, Y.; Huang, D.; Jiang, Q.; Guo, Y.; Chen, C. The vaccine efficacy against the SARS-CoV-2 Omicron: A systemic review and meta-analysis. Front. Public. Health. 2022, 10, 940956. [Google Scholar] [CrossRef]
- Garcia-Beltran, W.F.; St Denis, K.J.; Hoelzemer, A.; Lam, E.C.; Nitido, A.D.; Sheehan, M.L.; et al. mRNA-based COVID-19 vaccine boosters induce neutralizing immunity against SARS-CoV-2 Omicron variant. Cell 2022, 185(3), 457–66.e4. [Google Scholar] [CrossRef]
- Wang, L.; Kainulainen, M.H.; Jiang, N.; Di, H.; Bonenfant, G.; Mills, L.; Currier, M.; et al. Differential neutralization and inhibition of SARS-CoV-2 variants by antibodies elicited by COVID-19 mRNA vaccines. Nat. Commun. 2022, 13(1), 4350. [Google Scholar] [CrossRef] [PubMed]
- Young-Xu, Y.; Zwain, G.M.; Izurieta, H.S.; Korves, C.; Powell, E.I.; Smith, J.; et al. Effectiveness of mRNA COVID-19 vaccines against Omicron and Delta variants in a matched test-negative case-control study among US veterans. BMJ Open. 2022, 12(8), e063935. [Google Scholar] [CrossRef] [PubMed]
- Thompson, M.G.; Natarajan, K.; Irving, S.A.; Rowley, E.A.; Griggs, E.P.; Gaglani, M.; et al. Effectiveness of a Third Dose of mRNA Vaccines Against COVID-19-Associated Emergency Department and Urgent Care Encounters and Hospitalizations Among Adults During Periods of Delta and Omicron Variant Predominance - VISION Network, 10 States, August 2021-January 2022. MMWR. Morb. Mortal. Wkly. Rep. 2022, 71(4), 139–45. [Google Scholar] [PubMed]
- Samieefar, N.; Rashedi, R.; Akhlaghdoust, M.; Mashhadi, M.; Darzi, P.; Rezaei, N. Delta Variant: The New Challenge of COVID-19 Pandemic, an Overview of Epidemiological, Clinical, and Immune Characteristics. Acta. Biomed. 2022, 93(1), e2022179. [Google Scholar]
- Chen, L.; He, Y.; Liu, H.; Shang, Y.; Guo, G. Potential immune evasion of the severe acute respiratory syndrome coronavirus 2 Omicron variants. Front. Immunol. 2024, 15, 1339660. [Google Scholar] [CrossRef]
- Tseng, H.F.; Ackerson, B.K.; Bruxvoort, K.J.; Sy, L.S.; Tubert, J.E.; Lee, G.S.; et al. Effectiveness of mRNA-1273 vaccination against SARS-CoV-2 omicron subvariants BA.1, BA.2, BA.2.12.1, BA.4, and BA.5. Nat. Commun. 2023, 14(1), 189. [Google Scholar] [CrossRef]
- Munro, A.P.S.; Janani, L.; Cornelius, V.; et al. Safety and immunogenicity of seven COVID-19 vaccines as a third dose (booster) following two doses of ChAdOx1 nCoV-19 or BNT162b2 in the UK (COV-BOOST): a blinded, multicentre, randomised, controlled, phase 2 trial. Lancet 2021, 398(10318), 2258–76. [Google Scholar] [CrossRef]
- Pozzetto, B.; Legros, V.; Djebali, S.; et al. Immunogenicity and efficacy of heterologous ChAdOx1–BNT162b2 vaccination. Nature 2021, 600(7890), 701–6. [Google Scholar] [CrossRef]
- Ndzouboukou, J.B.; Kamara, A.A.; Ullah, N.; Lei, Q.; Fan, X.L. A Meta-analysis on the immunogenicity of homologous versus heterologous immunization regimens against SARS-CoV-2 Beta, Delta, and Omicron BA.1 VoCs in healthy adults. J. Microbiol. Biotechnol. 2025, 35, e2411059. [Google Scholar] [CrossRef]
- Atmar, R.L.; Lyke, K.E.; Deming, M.E.; Jackson, L.A.; Branche, A.R.; El Sahly, H.M.; Rostad, C.A.; Martin, J.M.; Johnston, C.; Rupp, R.E.; et al. Homologous and heterologous Covid-19 booster vaccinations. N. Engl. J. Med. 2022. [Google Scholar] [CrossRef]
- Nikolova, M.; Todorova, Y.; Emilova, R.; Trifonova, I.; Gladnishka, T.; Petrova-Yancheva, N.; et al. Induction of humoral and cellular immune responses to COVID-19 mRNA and vector vaccines: A prospective cohort study in Bulgarian healthcare workers. Med. Virol. 2022, 94(5), 2008–2018. [Google Scholar] [CrossRef]
- Zhang, Z.; Mateus, J.; Coelho, C.H.; Dan, J.M.; Moderbacher, C.R.; Gálvez, R.I.; et al. Humoral and cellular immune memory to four COVID-19 vaccines. Cell 2022, 185(14), 2434–2451.e17. [Google Scholar] [CrossRef]
- Isogawa, M.; Onodera, T.; Ainai, A.; Kotaki, R.; Kanno, T.; Saito, S.; et al. Prolonged effects of adenoviral vector priming on T-cell cytokine production in heterologous adenoviral vector/mRNA COVID-19 vaccination regimens. Sci. Rep. 2025, 15(1), 18684. [Google Scholar] [CrossRef]
- Chalkias, S.; Harper, C.; Vrbicky, K.; Walsh, S.R.; Essink, B.; Brosz, A.; et al. Safety and immunogenicity of a 100 µg mRNA-1273 vaccine booster in adults. N. Engl. J. Med. 2022, 386(11), 1088–91. [Google Scholar]
| Baseline characteristics | Total (n = 97) |
Post 2CV/AZ (n = 47) |
Post 2CV/BNT (n = 50) |
P-value |
|---|---|---|---|---|
| Age median y (IQR) | 39 (31-44) | 40 (31-45) | 37.5 (29-43) | 0.267 |
| Female, n (%) | 79 (81.44) | 44 (88.00) | 35 (74.47) | 0.118 |
| BMI, median (IQR) - < 25 kg/m2, n (%) - ≥ 25 kg/m2, n (%) |
23.23 (20.20-27.48) 61 (64.21) 34 (35.79) |
24.03 (20.57-28.37) 27 (55.10) 22 (44.90) |
22.53 (20.06-25.69) 34 (73.91) 12 (26.09) |
0.169 |
| Comorbidities, n (%) - Allergic rhinitis - Diabetes mellitus - Obesity - CVS / hypertension - Thyroid - Chronic lung disease |
25 (26.32) 10 (10.53) 5 (5.26) 4 (4.21) 3 (3.16) 2 (2.06) 1 (1.05) |
15 (30.61) 4 (8.16) 3 (6.12) 4 (8.16) 2 (4.08) 1 (2.00) 1 (2.04) |
10 (21.74) 6 (13.04) 2 (4.35) 0 (0.00) 1 (2.17) 1 (2.13) 0 (0.00) |
0.360 1.000 1.000 0.118 1.000 0.516 1.000 |
| Interval between 3rd dose of AZD1222 or BNT162b2 to mRNA1273 booster (day); median (IQR) | 164 (153-185) | 184 (149-190) | 162 (158-164) | 0.005** |
| sVNT to Delta variant (%inhibition), GM (95% CI) | 60.71 (54.66-67.43) |
51.93 (44.63-60.43) |
71.68 (62.58-81.98) |
< 0.001*** |
| sVNT to Omicron variant (%inhibition), GM (95% CI) | 24.55 (19.46-30.95) |
18.57 (12.45-27.69) |
29.56 (22.35-39.10) |
< 0.001*** |
| Anti-S-RBD IgG (BAU/ml), GM (95%CI) |
804.89 (696.38-930.31) |
640.70 (518.95-791.02) |
1025.99 (857.18-1228.04) |
0.003** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




