Submitted:
24 March 2026
Posted:
25 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
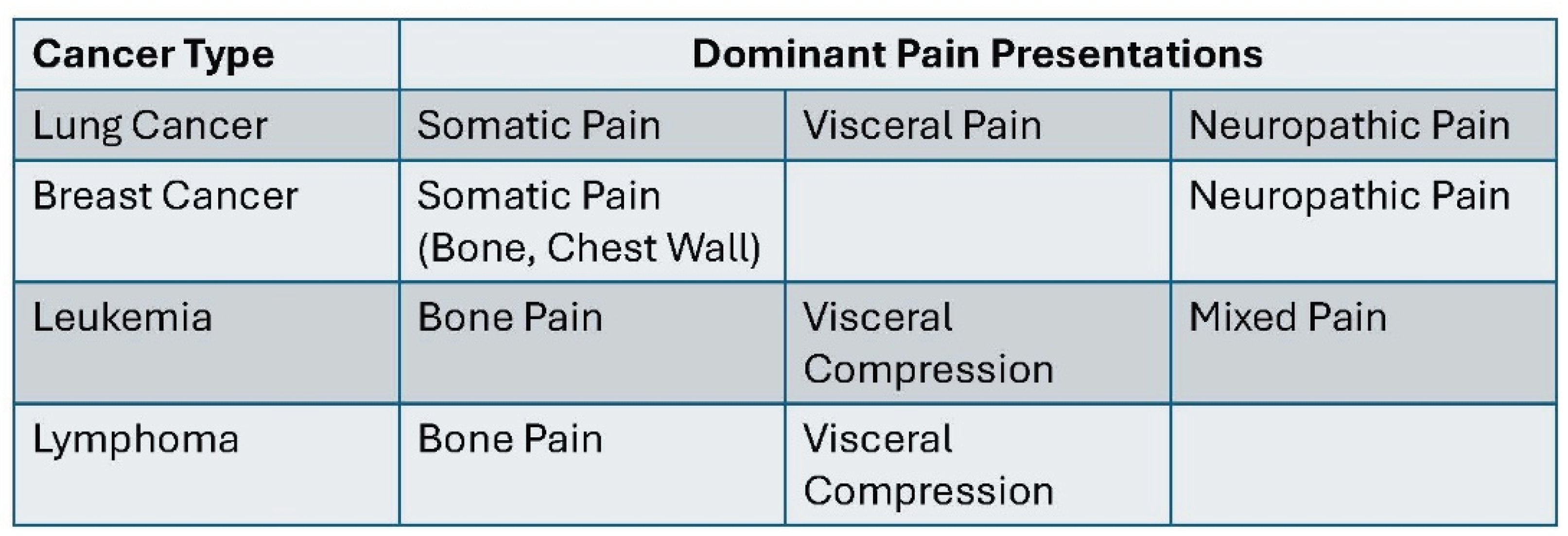
2. Common Cancers Associated with Pain
2.1. Lung Cancer
2.2. Breast Cancer
2.3. Leukemia and Lymphoma
3. Pain Presentations
3.1. Neuropathic Pain
3.2. Visceral Pain
3.3. Somatic Pain
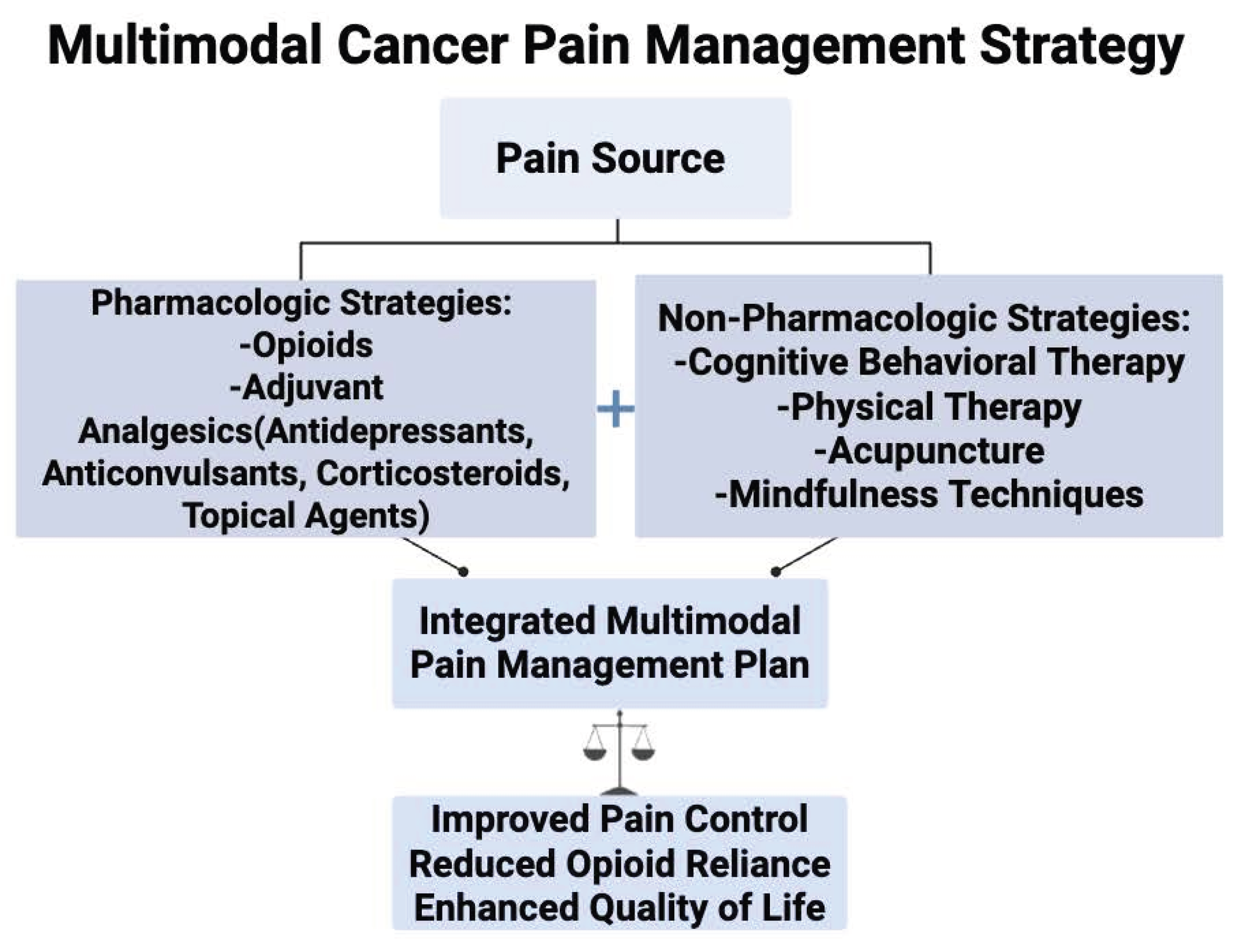
4. Pain Therapies
4.1. Non-Pharmacological Approaches
4.2. Pharmacological Approaches
5. Adjuvant Analgesics and Multimodal Strategies in Cancer Pain Management
5.1. Anticonvulsants
5.2. Antidepressants
5.3. Corticosteroids
5.4. Topical Agents
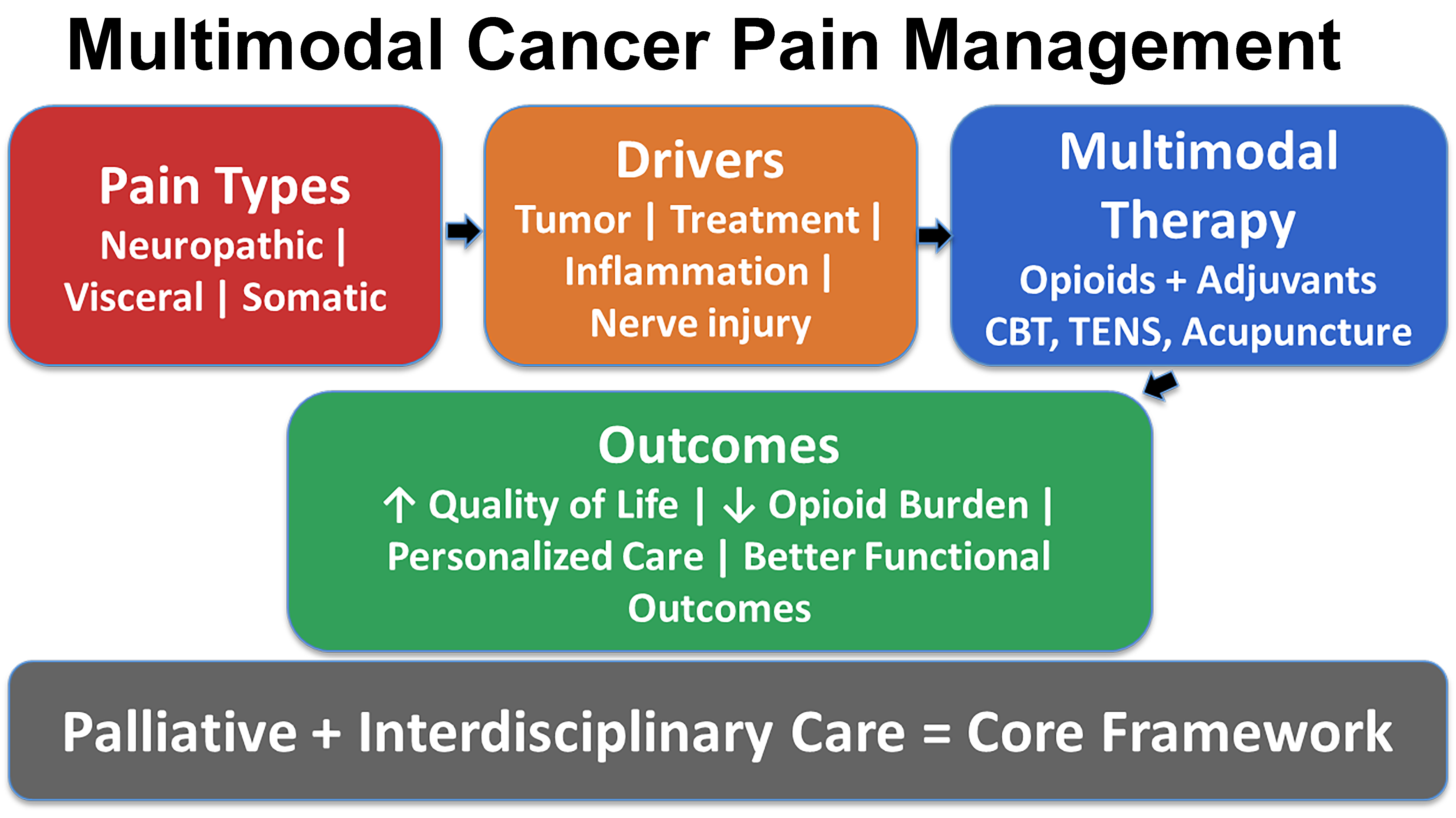
6. Multimodal Pain Management
6.1. Palliative Care and Interdisciplinary Approaches to Cancer Pain
7. Controversies and Evolving Paradigms in Cancer Pain Management
8. Emerging Therapies and Future Directions in the Field
8.1. Cannabinoids
8.2. Gene-Targeted Therapies
8.3. Novel Non-Opioid Analgesics and Molecular Targets
8.4. Integrative and Technological Approaches to Cancer Pain Care
9. Current Research Gaps
10. Conclusions
Funding
Conflicts of Interest statement
References
- Colosia, A., et al., The Burden of Metastatic Cancer-Induced Bone Pain: A Narrative Review. J Pain Res, 2022. 15: p. 3399-3412. [CrossRef]
- Kaye, A.D., et al., Prescription Opioid Abuse in Chronic Pain: An Updated Review of Opioid Abuse Predictors and Strategies to Curb Opioid Abuse: Part 1. Pain Physician, 2017. 20(2S): p. S93-S109.
- Gallaway, M.S., et al., Pain Among Cancer Survivors. Prev Chronic Dis, 2020. 17: p. E54. [CrossRef]
- Marchevsky, A.M., et al., The differential diagnosis between pleural sarcomatoid mesothelioma and spindle cell/pleomorphic (sarcomatoid) carcinomas of the lung: evidence-based guidelines from the International Mesothelioma Panel and the MESOPATH National Reference Center. Hum Pathol, 2017. 67: p. 160-168. [CrossRef]
- Potter, J. and I.J. Higginson, Pain experienced by lung cancer patients: a review of prevalence, causes and pathophysiology. Lung Cancer, 2004. 43(3): p. 247-57. [CrossRef]
- Hamood, R., et al., Chronic pain and other symptoms among breast cancer survivors: prevalence, predictors, and effects on quality of life. Breast Cancer Res Treat, 2018. 167(1): p. 157-169. [CrossRef]
- Smith, W.C., et al., A retrospective cohort study of post mastectomy pain syndrome. Pain, 1999. 83(1): p. 91-5.
- Parkes, A., et al., Prognostic Factors in Patients with Metastatic Breast Cancer with Bone-Only Metastases. Oncologist, 2018. 23(11): p. 1282-1288. [CrossRef]
- Niscola, P., et al., Pain syndromes in haematological malignancies: an overview. Hematol J, 2004. 5(4): p. 293-303. [CrossRef]
- Niscola, P., Effective pain management in hematological malignancies. Expert Rev Hematol, 2009. 2(3): p. 219-22. [CrossRef]
- Niscola, P., et al., Pain in blood cancers. Indian J Palliat Care, 2011. 17(3): p. 175-83. [CrossRef]
- Maman, E., et al., Acute lymphoblastic leukemia in children: correlation of musculoskeletal manifestations and immunophenotypes. J Child Orthop, 2007. 1(1): p. 63-8. [CrossRef]
- Lewis, W.D., S. Lilly, and K.L. Jones, Lymphoma: Diagnosis and Treatment. Am Fam Physician, 2020. 101(1): p. 34-41.
- Finnerup, N.B., R. Kuner, and T.S. Jensen, Neuropathic Pain: From Mechanisms to Treatment. Physiol Rev, 2021. 101(1): p. 259-301.
- Campbell, J.N. and R.A. Meyer, Mechanisms of neuropathic pain. Neuron, 2006. 52(1): p. 77-92.
- Borsook, D., Neurological diseases and pain. Brain, 2012. 135(Pt 2): p. 320-44. [CrossRef]
- Finnerup, N.B., et al., Neuropathic pain: an updated grading system for research and clinical practice. Pain, 2016. 157(8): p. 1599-1606. [CrossRef]
- Colloca, L., et al., Neuropathic pain. Nat Rev Dis Primers, 2017. 3: p. 17002.
- Bennett, M.I., et al., Prevalence and aetiology of neuropathic pain in cancer patients: a systematic review. Pain, 2012. 153(2): p. 359-365. [CrossRef]
- Edwards, H.L., M.R. Mulvey, and M.I. Bennett, Cancer-Related Neuropathic Pain. Cancers (Basel), 2019. 11(3).
- Delanian, S., J.L. Lefaix, and P.F. Pradat, Radiation-induced neuropathy in cancer survivors. Radiother Oncol, 2012. 105(3): p. 273-82. [CrossRef]
- Fathers, E., et al., Radiation-induced brachial plexopathy in women treated for carcinoma of the breast. Clin Rehabil, 2002. 16(2): p. 160-5. [CrossRef]
- Sikandar, S. and A.H. Dickenson, Visceral pain: the ins and outs, the ups and downs. Curr Opin Support Palliat Care, 2012. 6(1): p. 17-26.
- AC., C. and F. KM., Types of Pain. 6th edition ed. Holland-Frei Cancer Medicine, ed. K. DW, et al. 2003, Hamilton (ON): BC Decker.
- Raffaeli, W. and E. Arnaudo, Pain as a disease: an overview. J Pain Res, 2017. 10: p. 2003-2008. [CrossRef]
- Loeser, J.D. and R. Melzack, Pain: an overview. Lancet, 1999. 353(9164): p. 1607-9.
- Basbaum, A.I., et al., Cellular and molecular mechanisms of pain. Cell, 2009. 139(2): p. 267-84.
- Venkatachalam, K. and C. Montell, TRP channels. Annu Rev Biochem, 2007. 76: p. 387-417.
- Dahlhamer, J., et al., Prevalence of Chronic Pain and High-Impact Chronic Pain Among Adults—United States, 2016. MMWR Morb Mortal Wkly Rep, 2018. 67(36): p. 1001-1006. [CrossRef]
- van den Beuken-van Everdingen, M.H., et al., Prevalence of pain in patients with cancer: a systematic review of the past 40 years. Ann Oncol, 2007. 18(9): p. 1437-49. [CrossRef]
- Singh, P. and A. Chaturvedi, Complementary and alternative medicine in cancer pain management: a systematic review. Indian J Palliat Care, 2015. 21(1): p. 105-15. [CrossRef]
- Keefe, F.J., A.P. Abernethy, and L. C Campbell, Psychological approaches to understanding and treating disease-related pain. Annu Rev Psychol, 2005. 56: p. 601-30. [CrossRef]
- Zeltzer, L. and S. LeBaron, Hypnosis and nonhypnotic techniques for reduction of pain and anxiety during painful procedures in children and adolescents with cancer. J Pediatr, 1982. 101(6): p. 1032-5. [CrossRef]
- Spiegel, D. and J.R. Bloom, Group therapy and hypnosis reduce metastatic breast carcinoma pain. Psychosom Med, 1983. 45(4): p. 333-9. [CrossRef]
- Syrjala, K.L., et al., Relaxation and imagery and cognitive-behavioral training reduce pain during cancer treatment: a controlled clinical trial. Pain, 1995. 63(2): p. 189-198. [CrossRef]
- Vickers, A.J., et al., Acupuncture for Chronic Pain: Update of an Individual Patient Data Meta-Analysis. J Pain, 2018. 19(5): p. 455-474. [CrossRef]
- AC, C. and F. KM., Therapeutic Approaches to Cancer Pain., in Holland-Frei Cancer Medicine. 2003, BC Decker: Hamilton (ON).
- Klepstad, P., et al., Influence from genetic variability on opioid use for cancer pain: a European genetic association study of 2294 cancer pain patients. Pain, 2011. 152(5): p. 1139-1145. [CrossRef]
- Bennett, M., J.A. Paice, and M. Wallace, Pain and Opioids in Cancer Care: Benefits, Risks, and Alternatives. Am Soc Clin Oncol Educ Book, 2017. 37: p. 705-713.
- Boland, J.W. and A.G. Pockley, Clinically relevant concentrations of opioids for in vitro studies. J Opioid Manag, 2016. 12(5): p. 313-321.
- Boland, J.W., Effect of Opioids on Survival in Patients with Cancer. Cancers (Basel), 2022. 14(22). [CrossRef]
- Bedson, J., et al., Risk of adverse events in patients prescribed long-term opioids: A cohort study in the UK Clinical Practice Research Datalink. Eur J Pain, 2019. 23(5): p. 908-922. [CrossRef]
- Azizoddin, D.R., et al., Cancer pain self-management in the context of a national opioid epidemic: Experiences of patients with advanced cancer using opioids. Cancer, 2021. 127(17): p. 3239-3245. [CrossRef]
- Kwon, J.H., Overcoming barriers in cancer pain management. J Clin Oncol, 2014. 32(16): p. 1727-33. [CrossRef]
- Ezenwa, M.O. and M.F. Fleming, Racial Disparities in Pain Management in Primary Care. J Health Dispar Res Pract, 2012. 5(3): p. 12-26.
- Mitra, R. and S. Jones, Adjuvant analgesics in cancer pain: a review. Am J Hosp Palliat Care, 2012. 29(1): p. 70-9. [CrossRef]
- Lussier, D., A.G. Huskey, and R.K. Portenoy, Adjuvant analgesics in cancer pain management. Oncologist, 2004. 9(5): p. 571-91. [CrossRef]
- Lamont, L.A., W.J. Tranquilli, and K.A. Mathews, Adjunctive analgesic therapy. Vet Clin North Am Small Anim Pract, 2000. 30(4): p. 805-13, vii. [CrossRef]
- Khan, M.I., D. Walsh, and N. Brito-Dellan, Opioid and adjuvant analgesics: compared and contrasted. Am J Hosp Palliat Care, 2011. 28(5): p. 378-83. [CrossRef]
- Organization, W.H., WHO Guidelines for the pharmacological and radiotherapeutic management of cancer pain in adults and adolescents. 2019. p. 138.
- Jensen, T.S., Anticonvulsants in neuropathic pain: rationale and clinical evidence. Eur J Pain, 2002. 6 Suppl A: p. 61-8. [CrossRef]
- Mezzanotte, J.N., et al., Updates in the Treatment of Chemotherapy-Induced Peripheral Neuropathy. Curr Treat Options Oncol, 2022. 23(1): p. 29-42. [CrossRef]
- Tremont-Lukats, I.W., C. Megeff, and M.M. Backonja, Anticonvulsants for neuropathic pain syndromes: mechanisms of action and place in therapy. Drugs, 2000. 60(5): p. 1029-52.
- Patel, R. and A.H. Dickenson, Mechanisms of the gabapentinoids and α 2 δ-1 calcium channel subunit in neuropathic pain. Pharmacol Res Perspect, 2016. 4(2): p. e00205. [CrossRef]
- Sindrup, S.H., et al., Antidepressants in the treatment of neuropathic pain. Basic Clin Pharmacol Toxicol, 2005. 96(6): p. 399-409. [CrossRef]
- Vyvey, M., Steroids as pain relief adjuvants. Can Fam Physician, 2010. 56(12): p. 1295-7, e415.
- Riechelmann, R.P., et al., Symptom and medication profiles among cancer patients attending a palliative care clinic. Support Care Cancer, 2007. 15(12): p. 1407-12. [CrossRef]
- Ayyar, V.S. and W.J. Jusko, Transitioning from Basic toward Systems Pharmacodynamic Models: Lessons from Corticosteroids. Pharmacol Rev, 2020. 72(2): p. 414-438. [CrossRef]
- Haywood, A., et al., Corticosteroids for the management of cancer-related pain in adults. Cochrane Database Syst Rev, 2015. 2015(4): p. CD010756. [CrossRef]
- Yennurajalingam, S., et al., Impact of a palliative care consultation team on cancer-related symptoms in advanced cancer patients referred to an outpatient supportive care clinic. J Pain Symptom Manage, 2011. 41(1): p. 49-56. [CrossRef]
- Ness, T.J., L. Jones, and H. Smith, Use of compounded topical analgesics--results of an Internet survey. Reg Anesth Pain Med, 2002. 27(3): p. 309-12.
- Stanos, S.P. and K.E. Galluzzi, Topical therapies in the management of chronic pain. Postgrad Med, 2013. 125(4 Suppl 1): p. 25-33. [CrossRef]
- Choi, E., et al., Topical agents: a thoughtful choice for multimodal analgesia. Korean J Anesthesiol, 2020. 73(5): p. 384-393. [CrossRef]
- Sawynok, J., Topical analgesics for neuropathic pain: preclinical exploration, clinical validation, future development. Eur J Pain, 2014. 18(4): p. 465-81. [CrossRef]
- Anand, P. and K. Bley, Topical capsaicin for pain management: therapeutic potential and mechanisms of action of the new high-concentration capsaicin 8% patch. Br J Anaesth, 2011. 107(4): p. 490-502. [CrossRef]
- Raffa, R.B., Pharmacology of oral combination analgesics: rational therapy for pain. J Clin Pharm Ther, 2001. 26(4): p. 257-64. [CrossRef]
- Vijan, S., S. Manaker, and A. Qaseem, Noninvasive Treatments for Acute, Subacute, and Chronic Low Back Pain. Ann Intern Med, 2017. 167(11): p. 835-836. [CrossRef]
- Johnson, M.I., et al., Efficacy and safety of transcutaneous electrical nerve stimulation (TENS) for acute and chronic pain in adults: a systematic review and meta-analysis of 381 studies (the meta-TENS study). BMJ Open, 2022. 12(2): p. e051073. [CrossRef]
- Field, T., Massage therapy research review. Complement Ther Clin Pract, 2014. 20(4): p. 224-9. [CrossRef]
- Ehde, D.M., T.M. Dillworth, and J.A. Turner, Cognitive-behavioral therapy for individuals with chronic pain: efficacy, innovations, and directions for research. Am Psychol, 2014. 69(2): p. 153-66. [CrossRef]
- Shi, Y., et al., Brain Network to Placebo and Nocebo Responses in Acute Experimental Lower Back Pain: A Multivariate Granger Causality Analysis of fMRI Data. Front Behav Neurosci, 2021. 15: p. 696577. [CrossRef]
- Temel, J.S., et al., Early Palliative Care for Patients with Metastatic Non–Small-Cell Lung Cancer. New England Journal of Medicine, 2010. 363(8): p. 733-742. [CrossRef]
- Bakitas, M., et al., Effects of a Palliative Care Intervention on Clinical Outcomes in Patients With Advanced Cancer: The Project ENABLE II Randomized Controlled Trial. JAMA, 2009. 302(7): p. 741-749.
- Paice, J.A., Cancer pain management and the opioid crisis in America: How to preserve hard-earned gains in improving the quality of cancer pain management. Cancer, 2018. 124(12): p. 2491-2497. [CrossRef]
- Bucklar, N., et al., Early integration or last consultation: in-house palliative care involvement for hospitalized patients in tertiary medicine—a retrospective analysis. Supportive Care in Cancer, 2025. 33(4): p. 251.
- Schaefer, K.G., et al., Raising the bar for the care of seriously ill patients: results of a national survey to define essential palliative care competencies for medical students and residents. Acad Med, 2014. 89(7): p. 1024-31.
- Reed, M.K., et al., Sorting through life: evaluating patient-important measures of success in a medication for opioid use disorder (MOUD) treatment program. Substance Abuse Treatment, Prevention, and Policy, 2023. 18(1): p. 4. [CrossRef]
- Trang, T., et al., Pain and Poppies: The Good, the Bad, and the Ugly of Opioid Analgesics. J Neurosci, 2015. 35(41): p. 13879-88. [CrossRef]
- Harsanyi, H., C. Cuthbert, and F. Schulte, The Stigma Surrounding Opioid Use as a Barrier to Cancer-Pain Management: An Overview of Experiences with Fear, Shame, and Poorly Controlled Pain in the Context of Advanced Cancer. Curr Oncol, 2023. 30(6): p. 5835-5848. [CrossRef]
- Bulls, H.W., et al., Framework for opioid stigma in cancer pain. Pain, 2022. 163(2): p. e182-e189. [CrossRef]
- Carnago, L., M.R. Knisely, and J. Malek, Ethical considerations and decision making in opioid prescribing for chronic pain: A case study in rheumatology practice. J Am Assoc Nurse Pract, 2024. 36(5): p. 300-306. [CrossRef]
- Kotalik, J., Controlling pain and reducing misuse of opioids: ethical considerations. Can Fam Physician, 2012. 58(4): p. 381-5, e190-5.
- Smeltzer, K.E., G.J. Desai, and B. Johnson, Ethical considerations in prescribing or withholding opioids for chronic pain. Osteopathic Family Physician, 2017. 9: p. 26-29.
- Salins, N., et al., Definition of Terms Used in Limitation of Treatment and Providing Palliative Care at the End of Life: The Indian Council of Medical Research Commission Report. Indian J Crit Care Med, 2018. 22(4): p. 249-262. [CrossRef]
- Bhyan, P., et al., Palliative Sedation in Patients With Terminal Illness, in StatPearls. 2025, StatPearls Publishing Copyright © 2025, StatPearls Publishing LLC.: Treasure Island (FL).
- Vargas, A.J., et al., Exploring Racial Disparities in Chronic Pain Management. J Pain Res, 2025. 18: p. 2901-2908. [CrossRef]
- Enzinger, A.C., et al., Racial and Ethnic Disparities in Opioid Access and Urine Drug Screening Among Older Patients With Poor-Prognosis Cancer Near the End of Life. Journal of Clinical Oncology, 2023. 41(14): p. 2511-2522. [CrossRef]
- Baryakova, T.H., et al., Overcoming barriers to patient adherence: the case for developing innovative drug delivery systems. Nat Rev Drug Discov, 2023. 22(5): p. 387-409. [CrossRef]
- Krukowska, K., et al., The effectiveness of acupuncture in cancer pain treatment. Frontiers in Oncology, 2024. Volume 14-2024. [CrossRef]
- Yang, J., et al., Acupuncture for chronic cancer-related pain: a systematic review and network meta-analysis protocol. BMJ Open, 2020. 10(10): p. e039087. [CrossRef]
- Vučković, S., et al., Cannabinoids and Pain: New Insights From Old Molecules. Front Pharmacol, 2018. 9: p. 1259. [CrossRef]
- Boland, E.G., et al., Cannabinoids for adult cancer-related pain: systematic review and meta-analysis. BMJ Supportive & Palliative Care, 2020. 10(1): p. 14. [CrossRef]
- Qie, B., et al., Gene therapy for genetic diseases: challenges and future directions. MedComm (2020), 2025. 6(2): p. e70091. [CrossRef]
- Meng, H., et al., Cannabis and cannabinoids in cancer pain management. Curr Opin Support Palliat Care, 2020. 14(2): p. 87-93. [CrossRef]
- Gamage, T.F. and A.H. Lichtman, The endocannabinoid system: role in energy regulation. Pediatr Blood Cancer, 2012. 58(1): p. 144-8. [CrossRef]
- Handy, C.R., C. Krudy, and N. Boulis, Gene therapy: a potential approach for cancer pain. Pain Res Treat, 2011. 2011: p. 987597. [CrossRef]
- Kaye, A.D., et al., Update on the pharmacogenomics of pain management. Pharmgenomics Pers Med, 2019. 12: p. 125-143. [CrossRef]
- Raguram, A., S. Banskota, and D.R. Liu, Therapeutic in vivo delivery of gene editing agents. Cell, 2022. 185(15): p. 2806-2827. [CrossRef]
- Agulló, L., et al., Pharmacogenetic Guided Opioid Therapy Improves Chronic Pain Outcomes and Comorbid Mental Health: A Randomized, Double-Blind, Controlled Study. Int J Mol Sci, 2023. 24(13). [CrossRef]
- Pulskamp, T.G., L.M. Johnson, and D.J. Berlau, Novel non-opioid analgesics in pain management. Pain Manag, 2024. 14(12): p. 641-651. [CrossRef]
- Kaneko, Y. and A. Szallasi, Transient receptor potential (TRP) channels: a clinical perspective. Br J Pharmacol, 2014. 171(10): p. 2474-507. [CrossRef]
- Basch, E., A.B. Leahy, and A.C. Dueck, Benefits of Digital Symptom Monitoring With Patient-Reported Outcomes During Adjuvant Cancer Treatment. J Clin Oncol, 2021. 39(7): p. 701-703. [CrossRef]
- Blake, H., W.J. Chaplin, and A. Gupta, The effectiveness of digital interventions for self-management of chronic pain in employment settings: a systematic review. Br Med Bull, 2024. 151(1): p. 36-48. [CrossRef]
- Orujlu, S., et al., Barriers to cancer pain management from the perspective of patients: A qualitative study. Nurs Open, 2022. 9(1): p. 541-549. [CrossRef]
- Cluxton, C., The Challenge of Cancer Pain Assessment. Ulster Med J, 2019. 88(1): p. 43-46.
- Hjermstad, M.J., et al., Pain assessment tools in palliative care: an urgent need for consensus. Palliat Med, 2008. 22(8): p. 895-903. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




