Submitted:
24 March 2026
Posted:
25 March 2026
You are already at the latest version
Abstract
Background/Objectives: Radial extracorporeal shock wave therapy (rESWT) is used to treat neuromuscular disorders such as spasticity, but the mechanisms by which rESWT modulates muscle tone remain incompletely understood. One proposed mechanism involves mechanical perturbation of the neuromuscular junction (NMJ), particularly destabilization of acetylcholine receptor clusters in the postsynaptic membrane. Because rapsyn knockout mice are not viable, Caenorhabditis elegans (C. elegans) provides an alternative model through its rapsyn homolog RPY-1. This study examined whether loss of RPY-1 alters locomotor responses of C. elegans to radial extracorporeal shock wave (rESW) exposure. Methods: Wild-type worms and rpy-1 knockout worms (rpy-1-KOs) were exposed to defined numbers of rESWs. Locomotor behavior was quantified using automated tracking of parameters describing speed, trajectory and body-wave dynamics. Results: rESW exposure produced pronounced alterations in locomotor behavior across all parameters analyzed. After normalization to genotype-specific baseline values (because baseline locomotion differed between genotypes), wild-type worms and rpy-1-KOs responded similarly to moderate exposure levels. In contrast, higher exposure levels produced stronger locomotor impairment in rpy-1-KOs than in wild-type worms. Locomotor impairment was most pronounced immediately after exposure but improved during the subsequent recovery period of three hours. Conclusions: rESWs induced strong but largely reversible locomotor alterations in C. elegans during the first hours after exposure. The stronger impairment observed in rpy-1-KOs at higher exposure levels suggests that absence of the rapsyn homolog increased the vulnerability of the neuromuscular system of C. elegans to stronger mechanical perturbation induced by rESWs.
Keywords:
1. Introduction
2. Materials and Methods
3. Results
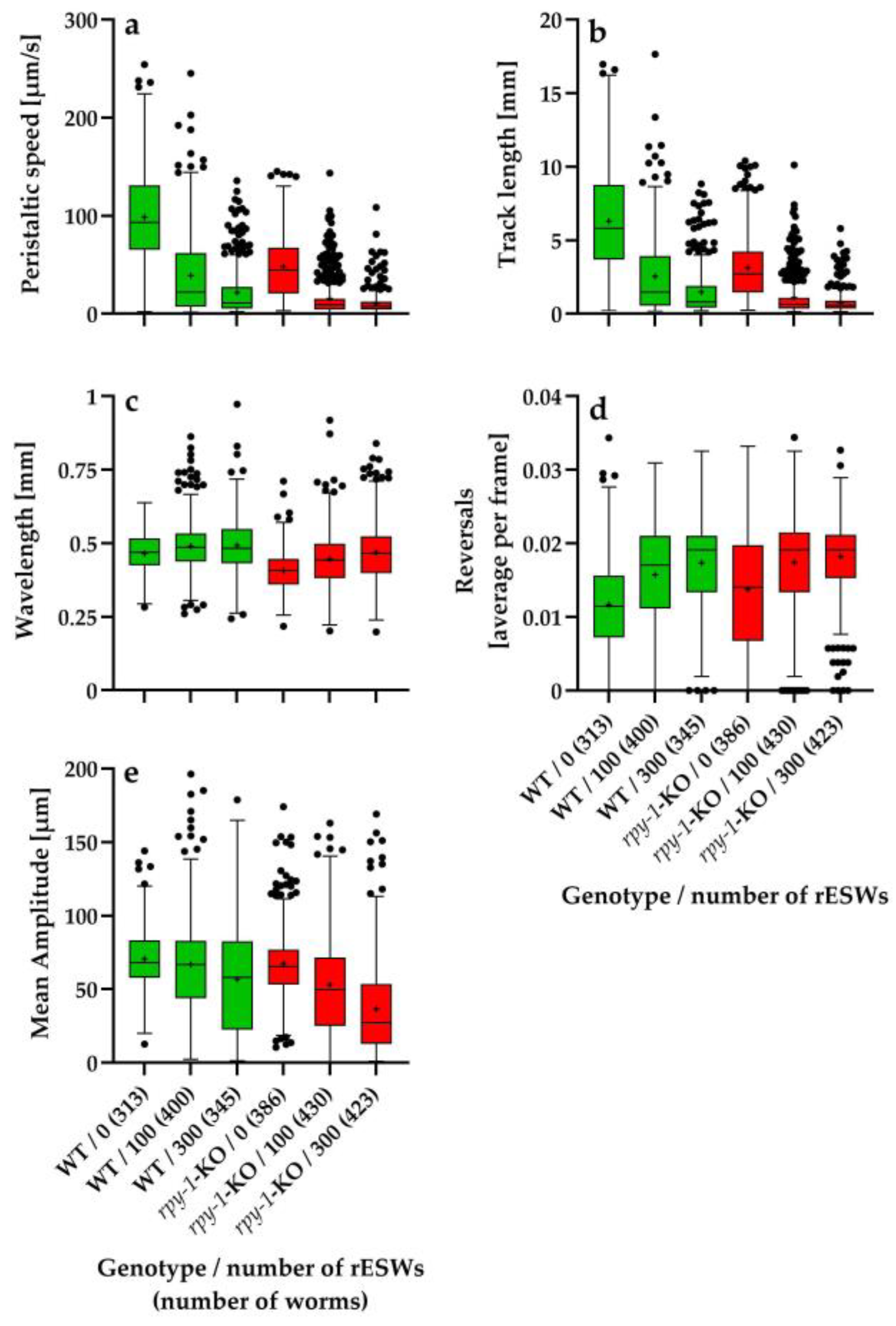
3.1. Absolute Values, and Baseline Locomotor Differences Between Wild-Type Worms and Rpy-1-KOs
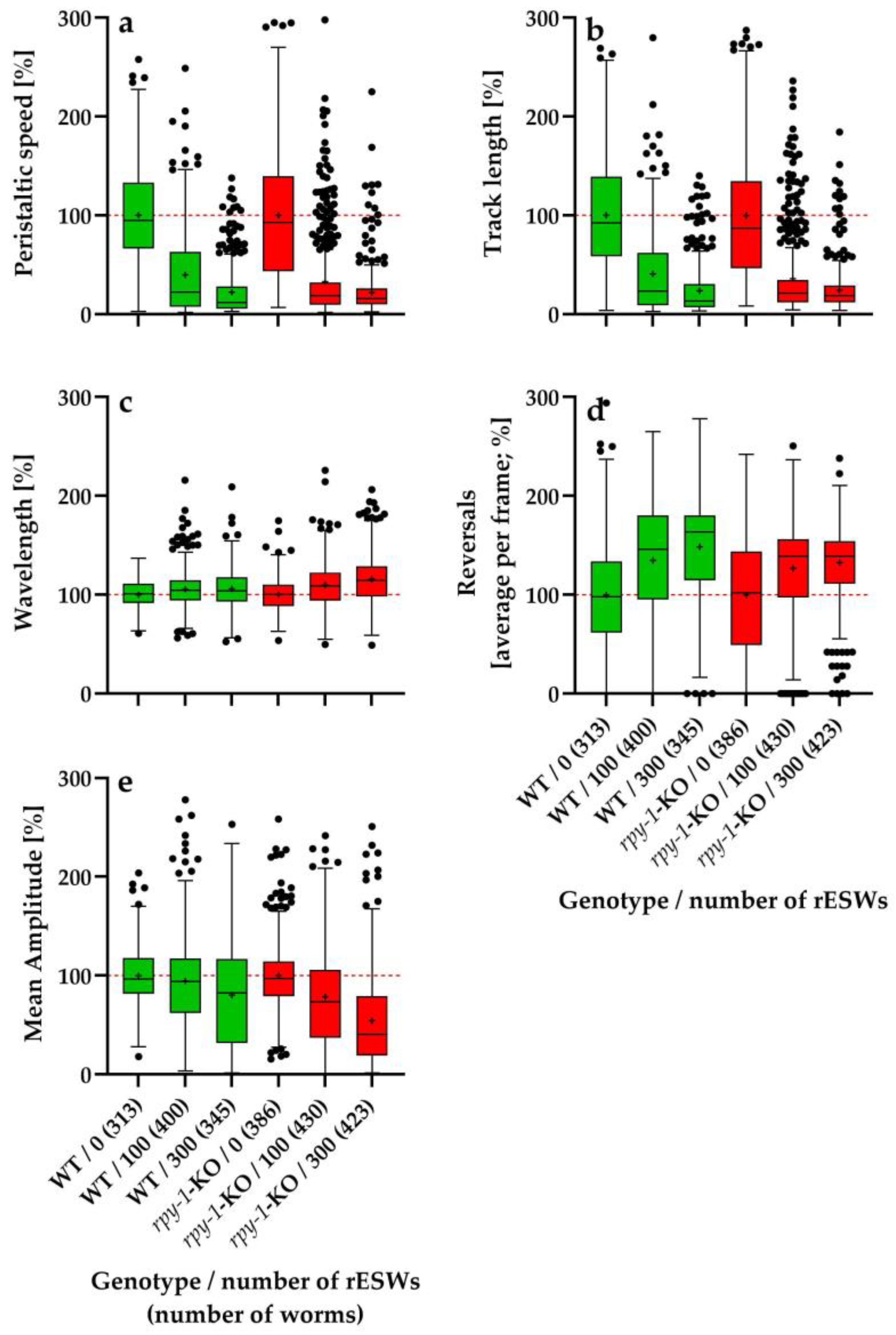
3.1. Dose-Dependent Effects of rESW Exposure on Relative Locomotor Behavior
3.1.1. Relative Peristaltic Speed
3.1.2. Relative Track Length
3.1.3. Relative Wavelength
3.1.4. Relative Reversal Frequency
3.1.5. Relative Mean Body-Wave Amplitude
3.1.6. Overall Pattern in the First Experimental Series
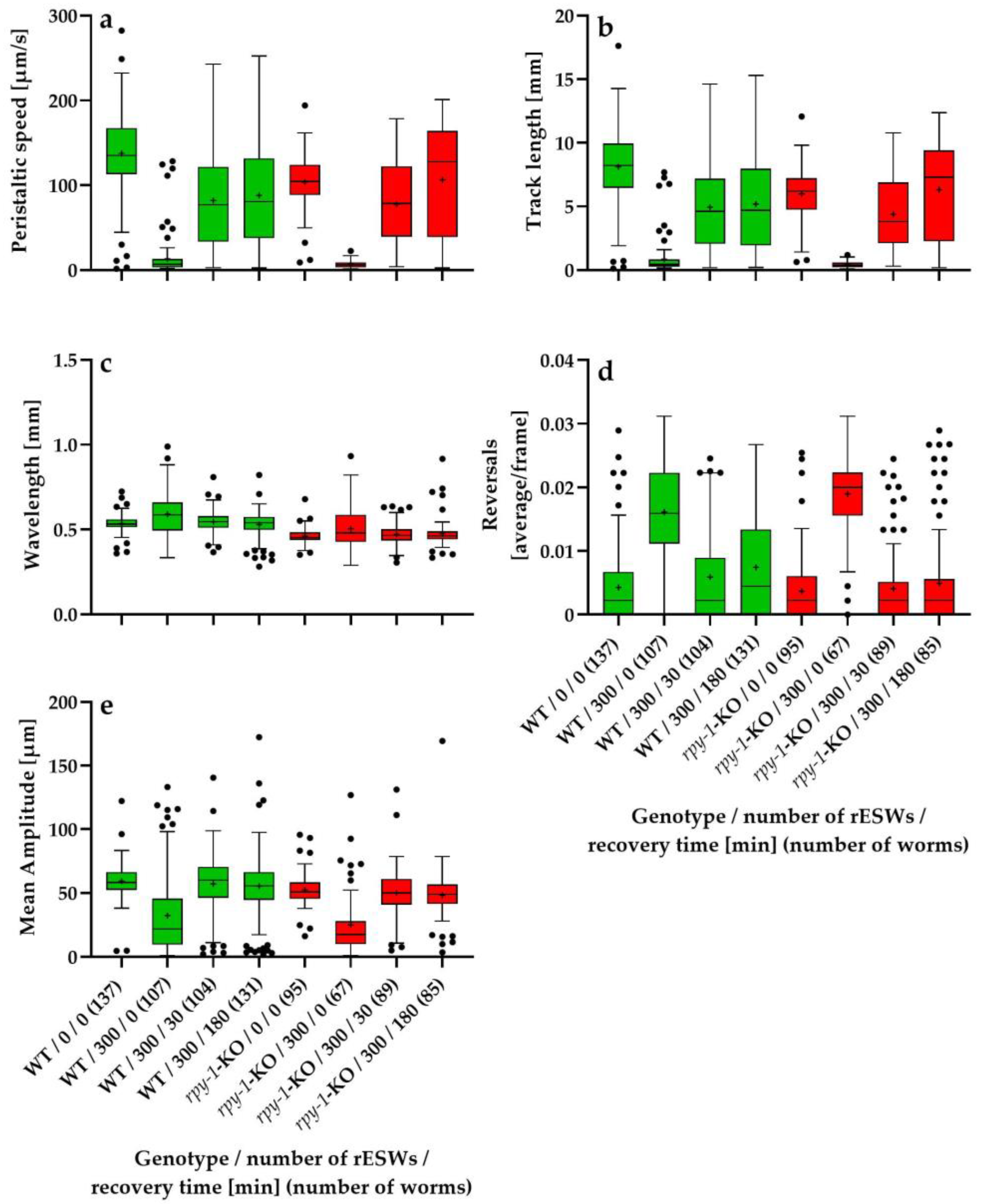
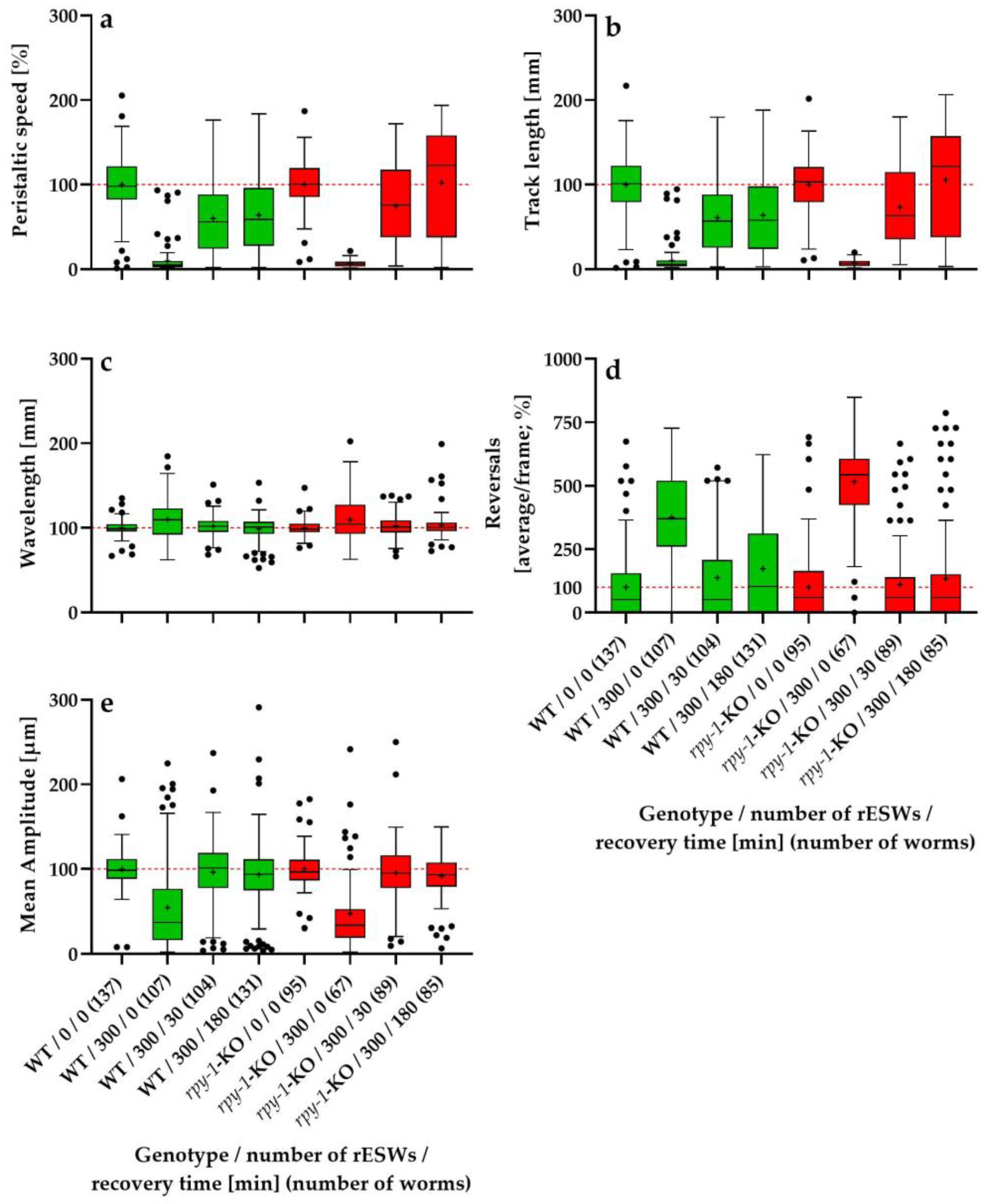
3.2. Recovery Dynamics of Relative Locomotor Behavior Within the First 180 Minutes After Exposure to 300 rESWs
3.2.1. Relative Peristaltic Speed
3.2.2. Relative Track Length
3.2.3. Relative Wavelength
3.2.4. Relative Reversal Frequency
3.2.5. Relative Mean Body-Wave Amplitude
3.2.6. Overall Pattern in the First Experimental Series
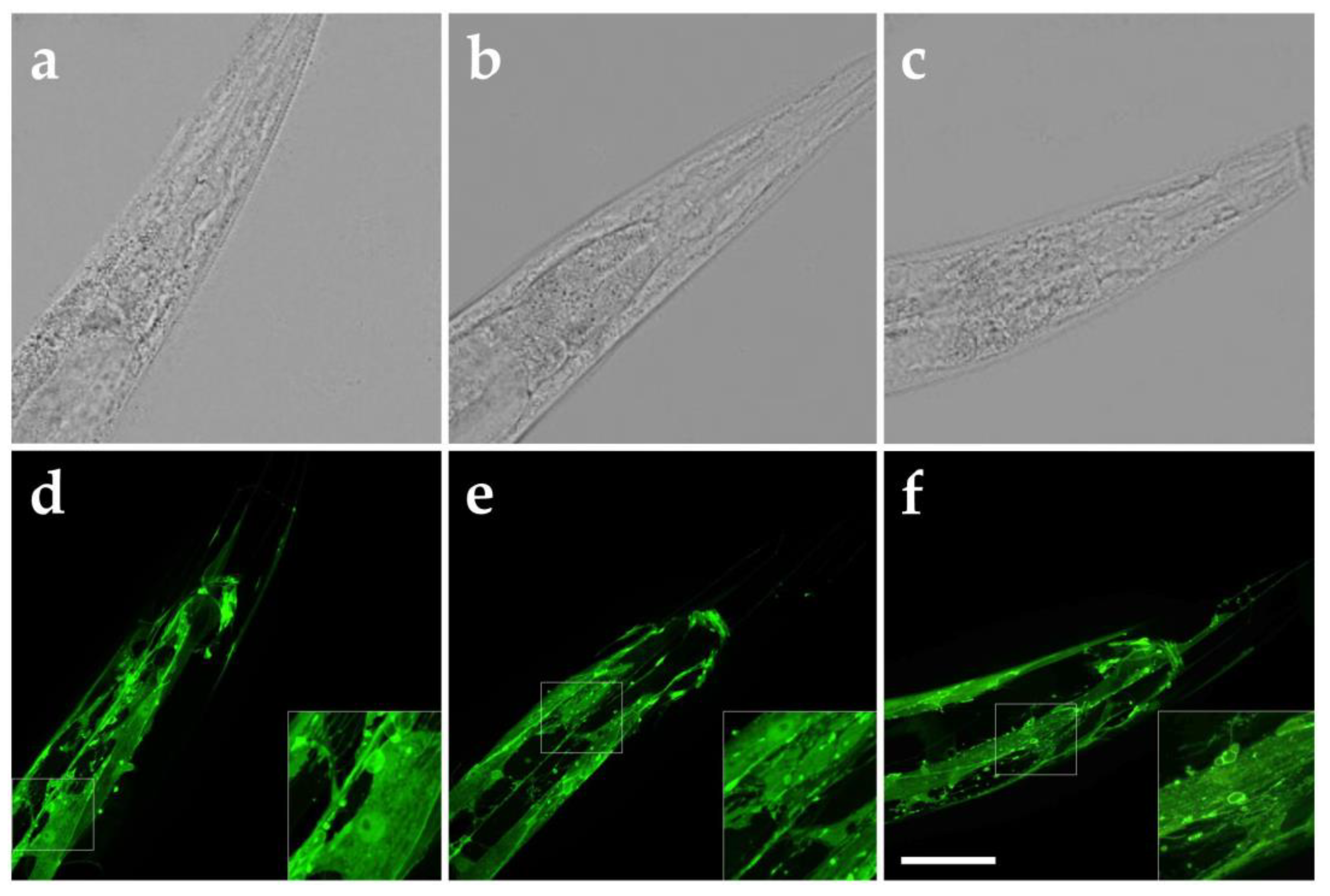
3.3. Localization of RPY-1 Following rESW Exposure
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| rESWT | Radial extracorporeal shock wave therapy |
| rESWs | Radial extracorporeal shock waves |
| NMJ | Neuromuscular junction |
| AChR | Acetylcholine receptor |
| C. elegans | Caenorhabditis elegans |
| GFP | green fluorescent protein |
References
- Schmitz, C.; Császár, N.B.; Milz, S.; Schieker, M.; Maffulli, N.; Rompe, J.D.; Furia, J.P. Efficacy and safety of extracorporeal shock wave therapy for orthopedic conditions: A systematic review on studies listed in the PEDro database. Br. Med. Bull. 2015, 116, 115–138. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Du, L.; Shan, L.; Dong, H.; Feng, J.; Kiessling, M.C.; Angstman, N.B.; Schmitz, C.; Jia, F. A prospective case-control study of radial extracorporeal shock wave therapy for spastic plantar flexor muscles in very young children with cerebral palsy. Medicine (Baltimore) 2016, 95, e3649. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Yan, Z.; Zhang, Y.; Duan, H.; Lu, P.; Liu, N.; Liu, F.; Li, Z. Therapeutic effect of slow stretching training combined with extracorporeal shock wave on biceps brachii spasticity in stroke patients. J. Jilin Univ. 2018, 44, 374–378. [Google Scholar] [CrossRef]
- Vidal, X.; Martí-Fàbregas, J.; Canet, O.; Roqué, M.; Morral, A.; Tur, M.; Schmitz, C.; Sitjà-Rabert, M. Efficacy of radial extracorporeal shock wave therapy compared with botulinum toxin type A injection in treatment of lower extremity spasticity in subjects with cerebral palsy: A randomized, controlled, cross-over study. J. Rehabil. Med. 2020, 52, jrm00076. [Google Scholar] [CrossRef]
- Császár, N.B.; Angstman, N.B.; Milz, S.; Sprecher, C.M.; Kobel, P.; Farhat, M.; Furia, J.P.; Schmitz, C. Radial shock wave devices generate cavitation. PLoS ONE 2015, 10, e0140541. [Google Scholar] [CrossRef]
- Wuerfel, T.; Schmitz, C.; Jokinen, L.L.J. The effects of the exposure of musculoskeletal tissue to extracorporeal shock waves. Biomedicines 2022, 10, 1084. [Google Scholar] [CrossRef]
- Hochstrasser, T.; Kaub, L.; Maier, L.; Angstman, N.B.; Kenmoku, T.; Nussbaum-Krammer, C.; Schmitz, C. Behavioral changes in Caenorhabditis elegans after exposure to radial extracorporeal shock waves. J. Clin. Med. 2025, 14, 7206. [Google Scholar] [CrossRef]
- Dymarek, R.; Ptaszkowski, K.; Ptaszkowska, L.; Kowal, M.; Sopel, M.; Taradaj, J.; Rosińczuk, J. Shock waves as a treatment modality for spasticity reduction and recovery improvement in post-stroke adults—Current evidence and qualitative systematic review. Clin. Interv. Aging 2020, 15, 9–28. [Google Scholar] [CrossRef]
- Yang, E.; Lew, H.L.; Özçakar, L.; Wu, C.H. Recent advances in the treatment of spasticity: Extracorporeal shock wave therapy. J. Clin. Med. 2021, 10, 4723. [Google Scholar] [CrossRef]
- Peng, H.H.; Sung, M.J.; Lee, Y.H.; Huang, S.W.; Lin, L.C. Effects of extracorporeal shock wave therapy on motor function in patients with cerebral palsy: A systematic review and meta-analysis. Disabil. Rehabil. 2025, 47, 6526–6535. [Google Scholar] [CrossRef] [PubMed]
- Kenmoku, T.; Ochiai, N.; Ohtori, S.; Saisu, T.; Sasho, T.; Nakagawa, K.; Iwakura, N.; Miyagi, M.; Ishikawa, T.; Tatsuoka, H.; Inoue, G.; Nakamura, J.; Kishida, S.; Saito, A.; Takahashi, K. Degeneration and recovery of the neuromuscular junction after application of extracorporeal shock wave therapy. J. Orthop. Res. 2012, 30, 1660–1665. [Google Scholar] [CrossRef]
- Kenmoku, T.; Nemoto, N.; Iwakura, N.; Ochiai, N.; Uchida, K.; Saisu, T.; Ohtori, S.; Nakagawa, K.; Sasho, T.; Takaso, M. Extracorporeal shock wave treatment can selectively destroy end plates in neuromuscular junctions. Muscle Nerve 2018, 57, 466–472. [Google Scholar] [CrossRef]
- Kenmoku, T.; Iwakura, N.; Ochiai, N.; Saisu, T.; Ohtori, S.; Takahashi, K.; Nakazawa, T.; Fukuda, M.; Takaso, M. Influence of different energy patterns on efficacy of radial shock wave therapy. J. Orthop. Sci. 2021, 26, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, G.; Benfenati, F.; Poulain, B.; Rossetto, O.; Polverino de Laureto, P.; DasGupta, B.R.; Montecucco, C. Tetanus and botulinum-B neurotoxins block neurotransmitter release by proteolytic cleavage of synaptobrevin. Nature 1992, 359, 832–835. [Google Scholar] [CrossRef] [PubMed]
- Blasi, J.; Chapman, E.R.; Link, E.; Binz, T.; Yamasaki, S.; De Camilli, P.; Südhof, T.C.; Niemann, H.; Jahn, R. Botulinum neurotoxin A selectively cleaves the synaptic protein SNAP-25. Nature 1993, 365, 160–163. [Google Scholar] [CrossRef]
- Schiavo, G.; Matteoli, M.; Montecucco, C. Neurotoxins affecting neuroexocytosis. Physiol. Rev. 2000, 80, 717–766. [Google Scholar] [CrossRef]
- Gautam, M.; Noakes, P.G.; Mudd, J.; Nichol, M.; Chu, G.C.; Sanes, J.R.; Merlie, J.P. Failure of postsynaptic specialization to develop at neuromuscular junctions of rapsyn-deficient mice. Nature 1995, 377, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Apel, E.D.; Glass, D.J.; Moscoso, L.M.; Yancopoulos, G.D.; Sanes, J.R. Rapsyn is required for MuSK signaling and recruits synaptic components to a MuSK-containing scaffold. Neuron 1997, 18, 623–635. [Google Scholar] [CrossRef]
- Sanes, J.R.; Lichtman, J.W. Induction, assembly, maturation and maintenance of a postsynaptic apparatus. Nat. Rev. Neurosci. 2001, 2, 791–805. [Google Scholar] [CrossRef]
- Maselli, R.; Dris, H.; Schnier, J.; Cockrell, J.; Wollmann, R. Congenital myasthenic syndrome caused by two non-N88K rapsyn mutations. Clin. Genet. 2007, 72, 63–65. [Google Scholar] [CrossRef]
- Bargmann, C.I. Neurobiology of the Caenorhabditis elegans genome. Science 1998, 282, 2028–2033. [Google Scholar] [CrossRef]
- Richmond, J.E.; Jorgensen, E.M. One GABA and two acetylcholine receptors function at the C. elegans neuromuscular junction. Nat. Neurosci. 1999, 2, 791–797. [Google Scholar] [CrossRef] [PubMed]
- Rand, J.B. Acetylcholine. WormBook 2007, 1–21. [Google Scholar] [CrossRef]
- Nam, S.; Min, K.; Hwang, H.; Lee, H.O.; Lee, J.H.; Yoon, J.; Lee, H.; Park, S.; Lee, J. Control of rapsyn stability by the CUL-3-containing E3 ligase complex. J. Biol. Chem. 2009, 284, 8195–8206. [Google Scholar] [CrossRef] [PubMed]
- Angstman, N.B.; Kiessling, M.C.; Frank, H.G.; Schmitz, C. High interindividual variability in dose-dependent reduction in speed of movement after exposing C. elegans to shock waves. Front. Behav. Neurosci. 2015, 9, 12. [Google Scholar] [CrossRef]
- Tittelmeier, J.; Seeber, H.; Grasse, D.; Barapatre, N.; Schmitz, C.; Krentel, D.; Grobert, S.; Nussbaum-Krammer, C. Explosive shock wave exposure leads to age-accelerated motor and sensory decline in C. elegans. bioRxiv 2025, 2025.12.19, 695190. [Google Scholar] [CrossRef]
- C. elegans Deletion Mutant Consortium. Large-scale screening for targeted knockouts in the Caenorhabditis elegans genome. G3 (Bethesda) 2012, 2, 1415–1425. [Google Scholar] [CrossRef]
- Stiernagle, T. Maintenance of C. elegans. WormBook 2006, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Roussel, N.; Sprenger, J.; Tappan, S.J.; Glaser, J.R. Robust tracking and quantification of C. elegans body shape and locomotion through coiling, entanglement, and omega bends. Worm 2015, 3, e982437. [Google Scholar] [CrossRef]
- Weeks, J.C.; Roberts, W.M.; Leasure, C.; Suzuki, B.M.; Robinson, K.J.; Currey, H.; Wangchuk, P.; Eichenberger, R.M.; Saxton, A.D.; Bird, T.D.; Kraemer, B.C.; Loukas, A.; Hawdon, J.M.; Caffrey, C.R.; Liachko, N.F. Sertraline, paroxetine, and chlorpromazine are rapidly acting anthelmintic drugs capable of clinical repurposing. Sci. Rep. 2018, 8, 975. [Google Scholar] [CrossRef]
- Thapa, P.; Olek, K.; Kowalska, A.; Serwa, R.A.; Pokrzywa, W. SAM, SAH and C. elegans longevity: Insights from a partial AHCY deficiency model. NPJ Aging 2023, 9, 27. [Google Scholar] [CrossRef]
- Kutzner, C.E.; Bauer, K.C.; Lackmann, J.W.; Acton, R.J.; Sarkar, A.; Pokrzywa, W.; Hoppe, T. Optogenetic induction of mechanical muscle stress identifies myosin regulatory ubiquitin ligase NHL-1 in C. elegans. Nat. Commun. 2024, 15, 6879. [Google Scholar] [CrossRef]
- Scheirer, C.J.; Ray, W.S.; Hare, N. The analysis of ranked data derived from completely randomized factorial designs. Biometrics 1976, 32, 429–434. [Google Scholar] [CrossRef]
- Holm, S. A simple sequentially rejective multiple test procedure. Scand. J. Stat. 1979, 6, 65–70. [Google Scholar]
- Liao, X.; Wang, Y.; Lai, X.; Wang, S. The role of Rapsyn in neuromuscular junction and congenital myasthenic syndrome. Biomol. Biomed. 2023, 23, 772–784. [Google Scholar] [CrossRef]
- Burden, S.J.; Huijbers, M.G.; Remedio, L. Fundamental molecules and mechanisms for forming and maintaining neuromuscular synapses. Int. J. Mol. Sci. 2018, 19, 490. [Google Scholar] [CrossRef] [PubMed]
- Millar, N.S.; Harkness, P.C. Assembly and trafficking of nicotinic acetylcholine receptors (Review). Mol. Membr. Biol. 2008, 25, 279–292. [Google Scholar] [CrossRef]
- Zong, Y.; Jin, R. Structural mechanisms of the agrin-LRP4-MuSK signaling pathway in neuromuscular junction differentiation. Cell. Mol. Life Sci. 2013, 70, 3077–3088. [Google Scholar] [CrossRef] [PubMed]
- Willmann, R.; Fuhrer, C. Neuromuscular synaptogenesis: Clustering of acetylcholine receptors revisited. Cell. Mol. Life Sci. 2002, 59, 1296–1316. [Google Scholar] [CrossRef]
- Morgan, J.P.M.; Hamm, M.; Schmitz, C.; Brem, M.H. Return to play after treating acute muscle injuries in elite football players with radial extracorporeal shock wave therapy. J. Orthop. Surg. Res. 2021, 16, 708. [Google Scholar] [CrossRef]
- Crupnik, J.; Silveti, S.; Wajnstein, N.; Rolon, A.; Wuerfel, T.; Stiller, P.; Morral, A.; Furia, J.P.; Maffulli, N.; Schmitz, C. Radial ESWT combined with a specific rehabilitation program (rESWT+RP) is more effective than sham rESWT+RP for acute hamstring muscle complex injury type 3b: A randomized, controlled trial. Br. Med. Bull. 2025, 155, ldaf009. [Google Scholar] [CrossRef] [PubMed]
- Balch, M.H.H.; Harris, H.; Chugh, D.; Gnyawali, S.; Rink, C.; Nimjee, S.M.; Arnold, W.D. Ischemic stroke-induced polyaxonal innervation at the neuromuscular junction is attenuated by robot-assisted mechanical therapy. Exp. Neurol. 2021, 343, 113767. [Google Scholar] [CrossRef] [PubMed]
| PS | TL | WL | R | MA | |
| Normality testing (Shapiro-Wilk test) | |||||
| Group | |||||
| WT / 0 | <0.001 | <0.001 | 0.003 | <0.001 | <0.001 |
| WT / 100 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| WT / 300 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| rpy-1-KO / 0 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| rpy-1-KO / 100 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| rpy-1-KO / 300 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| Non-parametric two-factor analysis (Scheirer-Ray-Hare test) | |||||
| Genotype | 0.554 | 0.127 | <0.001 | <0.001 | <0.001 |
| Exposure | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| Genotype x Exposure | 0.001 | 0.001 | <0.001 | 0.005 | <0.001 |
| Post-hoc pairwise comparisons (Dunn-type comparisons; Mann-Whitney tests with Holm correction | |||||
| -Within wildtype | |||||
| WT / 0 vs WT / 100 | <0.001 | <0.001 | 0.003 | <0.001 | 0.026 |
| WT / 0 vs WT / 300 | <0.001 | <0.001 | 0.003 | <0.001 | <0.001 |
| WT / 100 vs WT / 300 | <0.001 | <0.001 | 0.836 | 0.001 | <0.001 |
| -Withing rpy-1-KO | |||||
| rpy-1-KO / 0 vs rpy-1-KO / 100 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| rpy-1-KO / 0 vs rpy-1-KO / 300 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| rpy-1-KO / 100 vs rpy-1-KO / 300 | 0.004 | 0.004 | 0.003 | 0.097 | <0.001 |
| -Between genotypes (same exposure) | |||||
| WT / 0 vs rpy-1-KO / 0 | 0.779 | 0.618 | 0.548 | 0.740 | 0.458 |
| WT / 100 vs rpy-1-KO / 100 | 0.779 | 0.865 | 0.006 | 0.128 | <0.001 |
| WT / 300 vs rpy-1-KO / 300 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| PS | TL | WL | R | MA | |
| Normality testing (Shapiro-Wilk test) | |||||
| Group | |||||
| WT / 0 / 0 | 0.059 | 0.129 | <0.001 | <0.001 | <0.001 |
| WT / 300 / 0 | <0.001 | <0.001 | 0.099 | 0.041 | <0.001 |
| WT / 300 / 30 | <0.001 | <0.001 | 0.003 | <0.001 | <0.001 |
| WT / 300 / 180 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| rpy-1-KO / 0 / 0 | 0.028 | 0.175 | <0.001 | <0.001 | <0.001 |
| rpy-1-KO / 300 / 0 | <0.001 | 0.001 | 0.002 | 0.001 | <0.001 |
| rpy-1-KO / 300 / 30 | 0.007 | 0.001 | 0.007 | <0.001 | <0.001 |
| rpy-1-KO / 300 / 180 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| Non-parametric two-factor analysis (Scheirer-Ray-Hare test) | |||||
| Genotype | <0.001 | <0.001 | 0.727 | 0.531 | 0.305 |
| Exposure | <0.001 | <0.001 | 0.005 | <0.001 | <0.001 |
| Recovery | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| Genotype x Exposure | 0.008 | 0.010 | 0.725 | 0.621 | 0.767 |
| Genotype x Recovery | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| Exposure x Recovery | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| Genotype x Exposure x Recovery | <0.001 | <0.001 | 0.465 | <0.001 | 0.681 |
| Post-hoc pairwise comparisons (Dunn-type comparisons; Mann-Whitney tests with Holm correction | |||||
| -Baseline comparison | |||||
| WT / 0 / 0 vs rpy-1-KO / 0 / 0 | 0.869 | 0.813 | 0.401 | 0.939 | 0.649 |
| -Immediate effects after exposure (0 minutes) | |||||
| WT / 0 / 0 vs WT / 300 / 0 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| rpy-1-KO / 0 / 0 vs rpy-1-KO / 300 / 0 | <0.001 | <0.001 | 0.043 | <0.001 | <0.001 |
| WT / 300 / 0 vs rpy-1-KO / 300 / 0 | 0.318 | 0.249 | 0.683 | <0.001 | 0.757 |
| -Effects at 30 minutes after exposure | |||||
| WT / 300 / 0 vs WT / 300 / 30 | <0.001 | <0.001 | 0.028 | <0.001 | <0.001 |
| rpy-1-KO / 300 / 0 vs rpy-1-KO / 300 / 30 | <0.001 | <0.001 | 0.757 | <0.001 | <0.001 |
| WT / 300 / 30 vs rpy-1-KO / 300 / 30 | 0.015 | 0.049 | 0.952 | 0.216 | 0.809 |
| -Effects at 180 minutes after exposure | |||||
| WT / 300 / 0 vs WT / 300 / 180 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| rpy-1-KO / 300 / 0 vs rpy-1-KO / 300 / 180 | <0.001 | <0.001 | 0.757 | <0.001 | <0.001 |
| WT / 300 / 180 vs rpy-1-KO / 300 / 180 | <0.001 | <0.001 | 0.952 | 0.031 | 0.892 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




