Submitted:
24 March 2026
Posted:
25 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
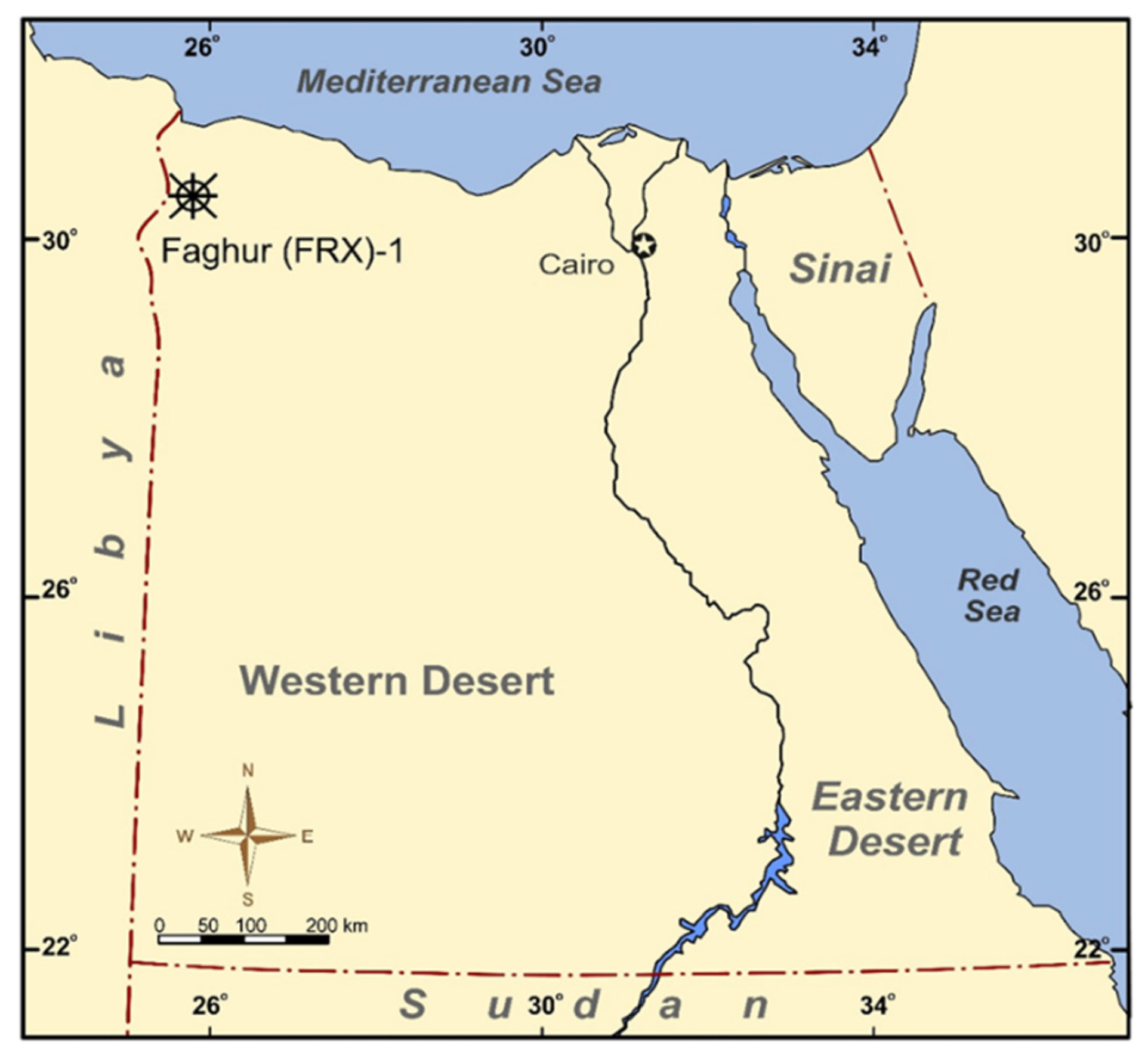
2. Geological Settings
3. Material and Methods
3.1. Organic Petrography and Statistical Analysis
3.2. Palynofacies and Biostratigraphic Analyses
3.3. Supervised Machine Learning and Maceral Quantification
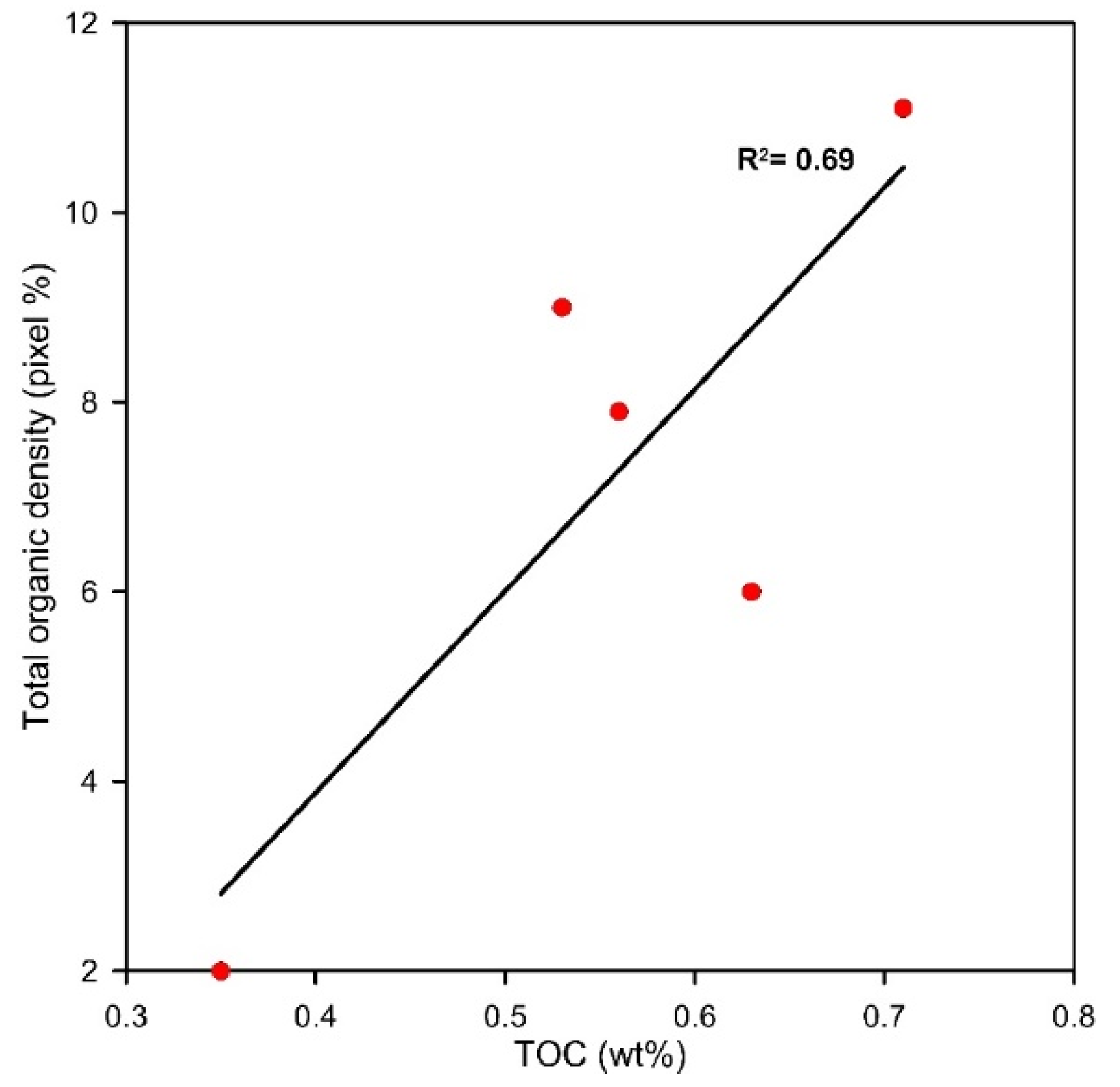
3.4. Total Organic Carbon (TOC)
4. Results
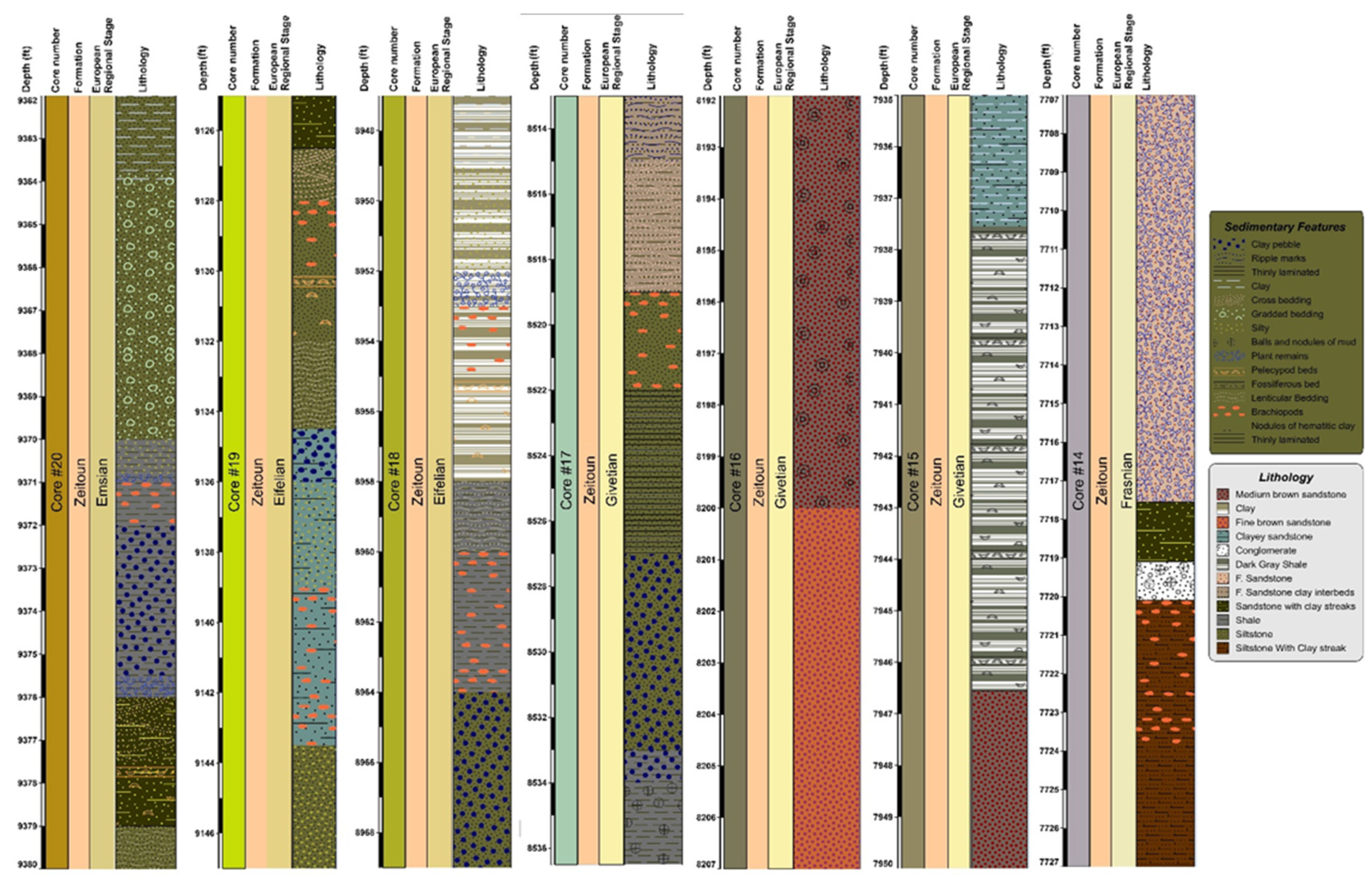
4.1. Cores Lithological Variations
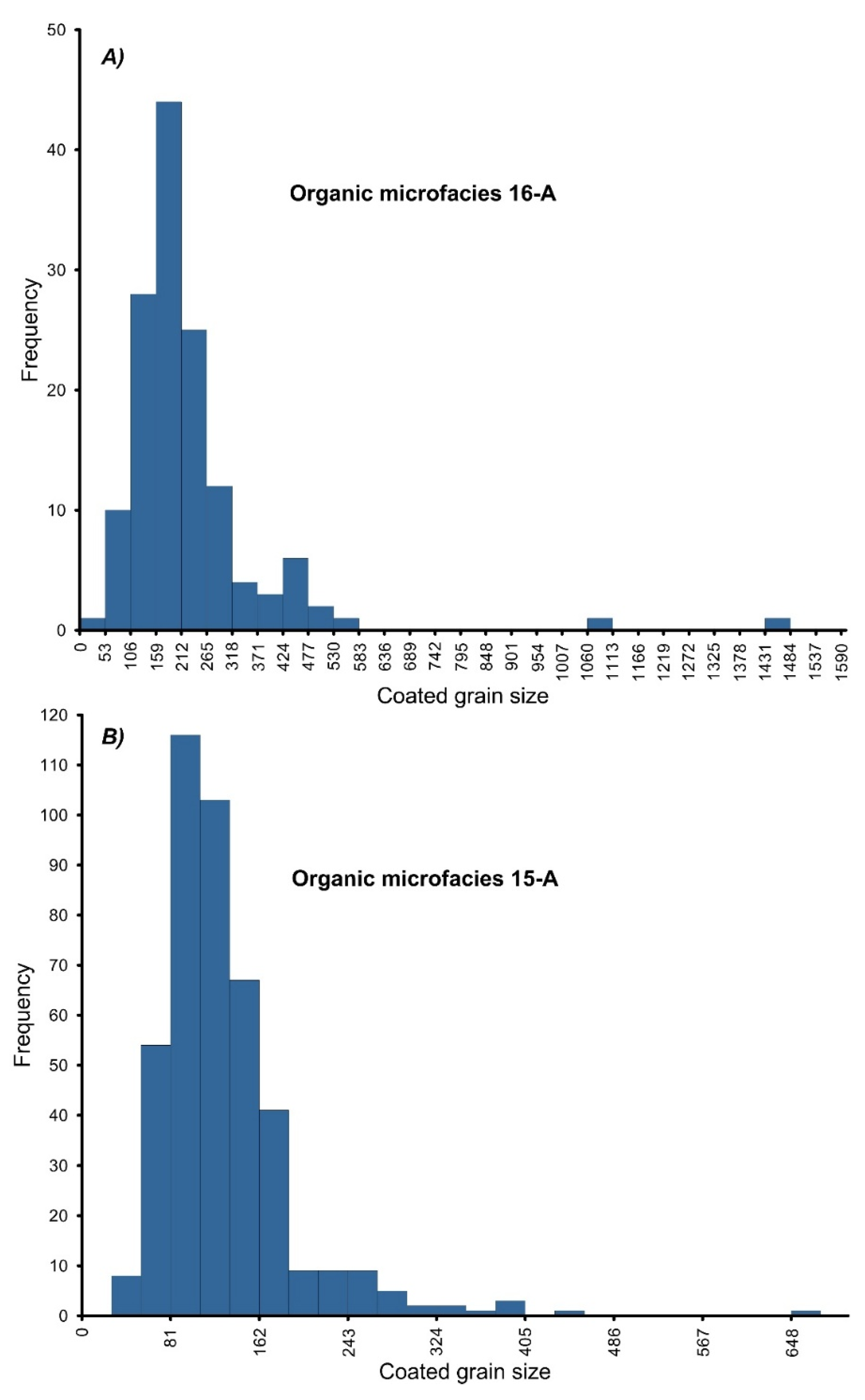
4.2. Organic Density
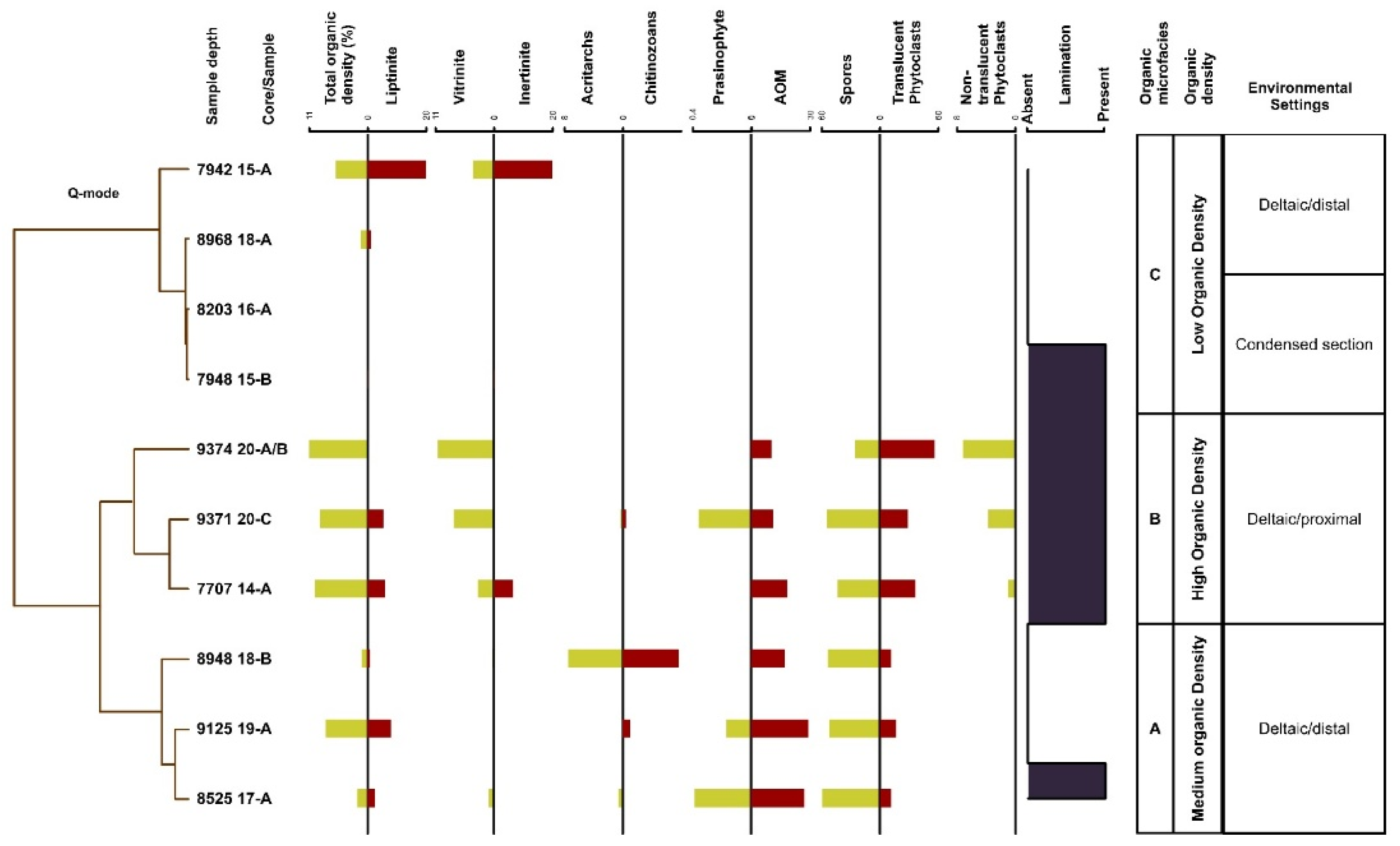
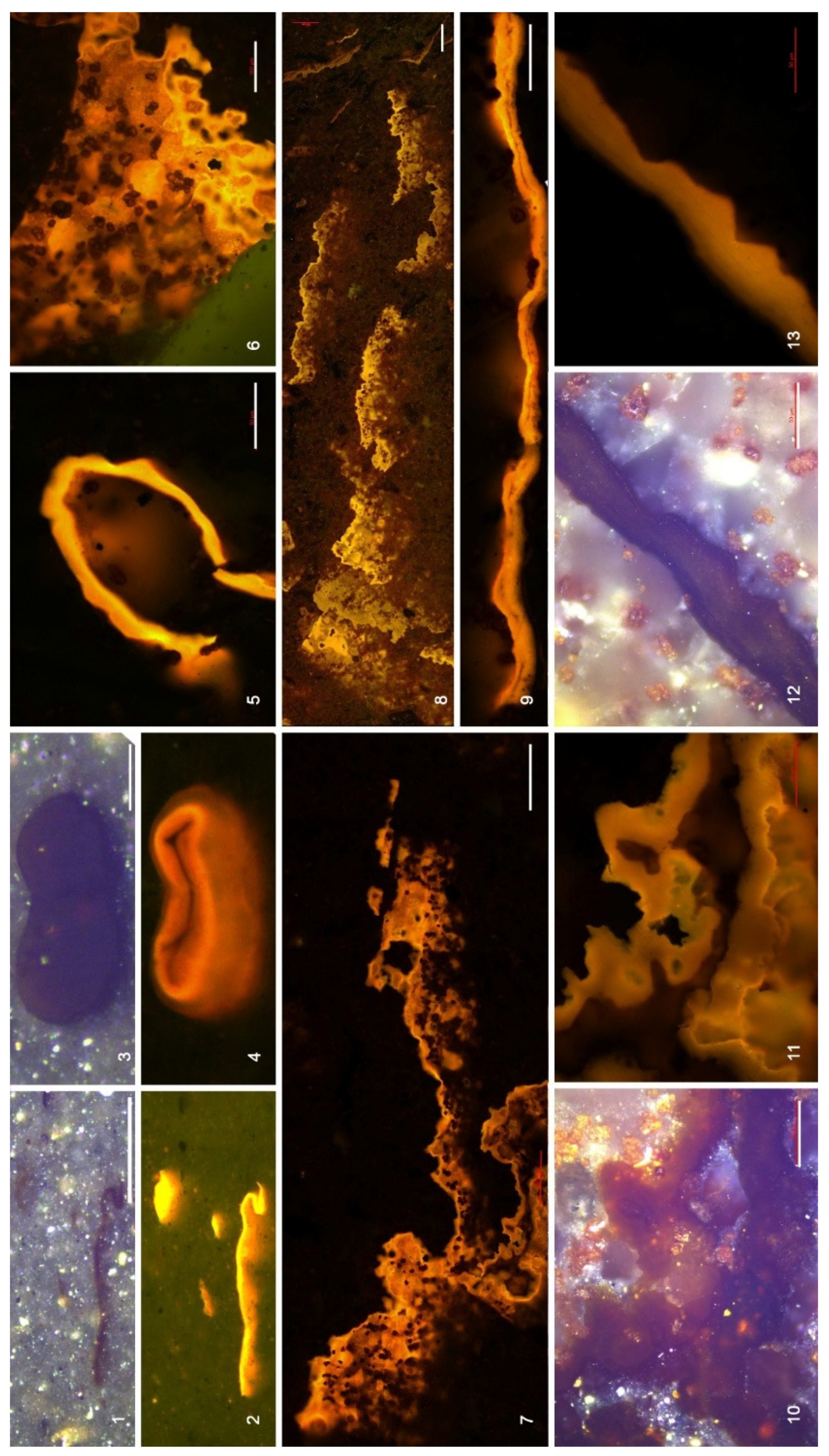
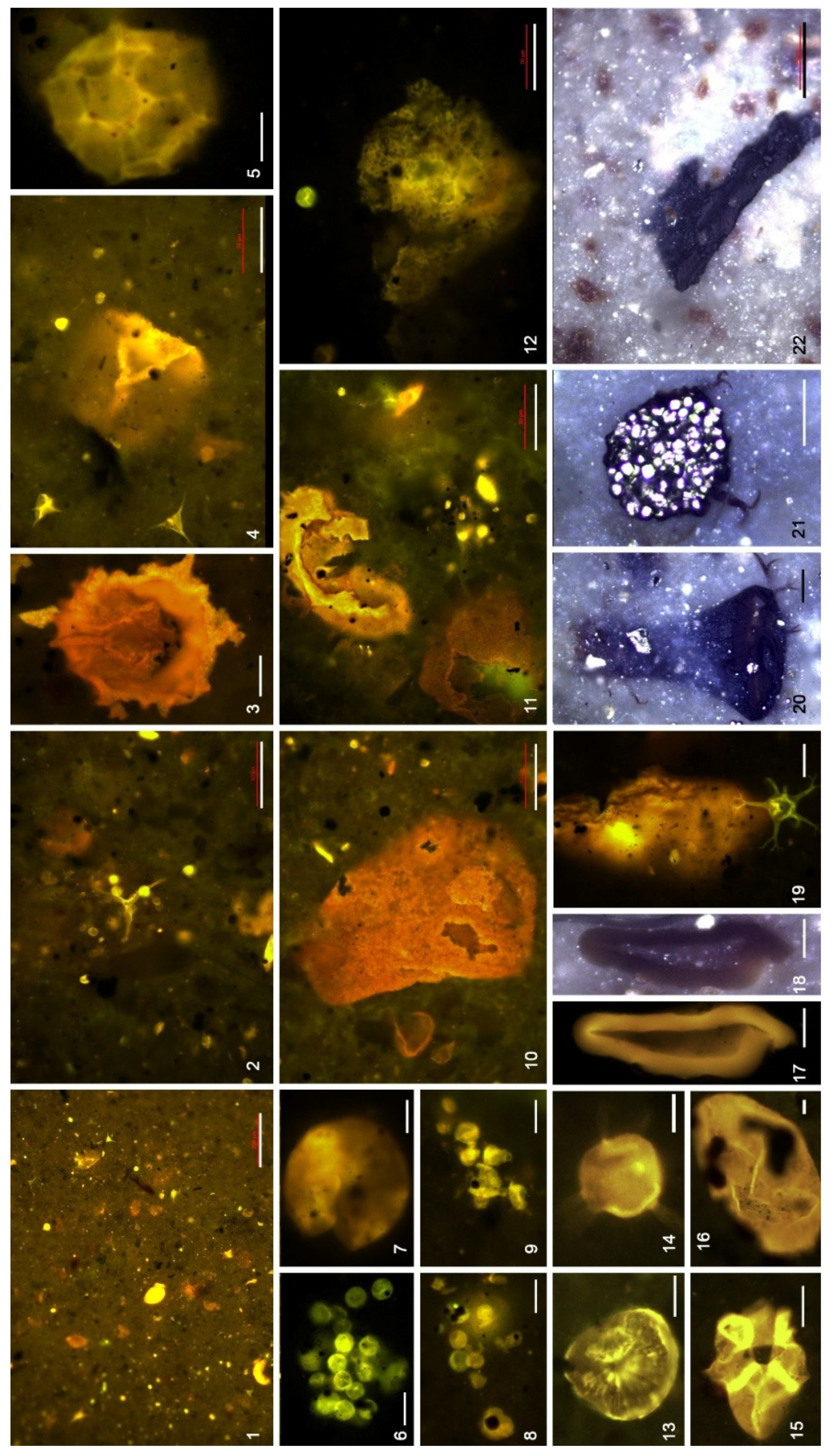
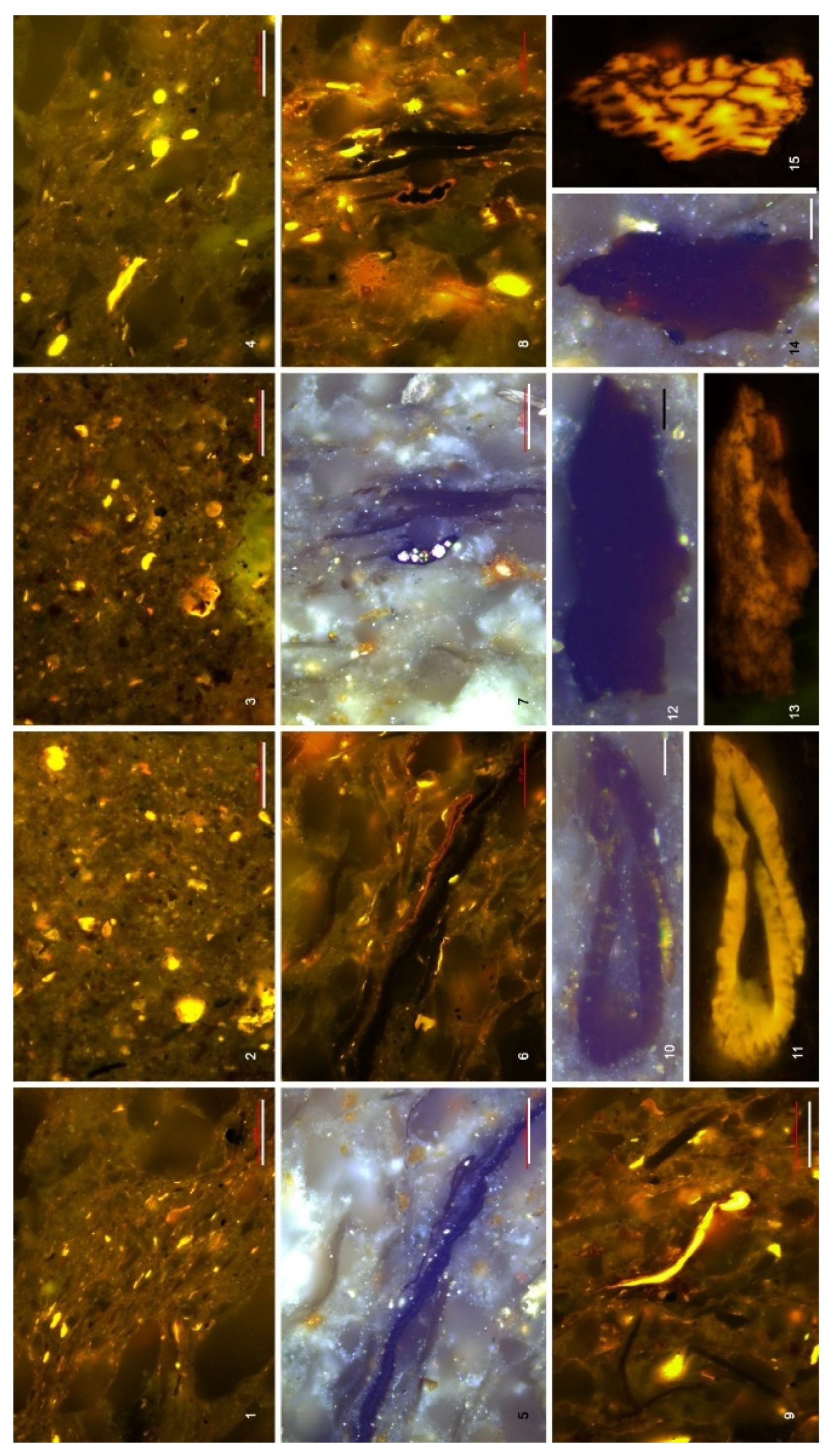
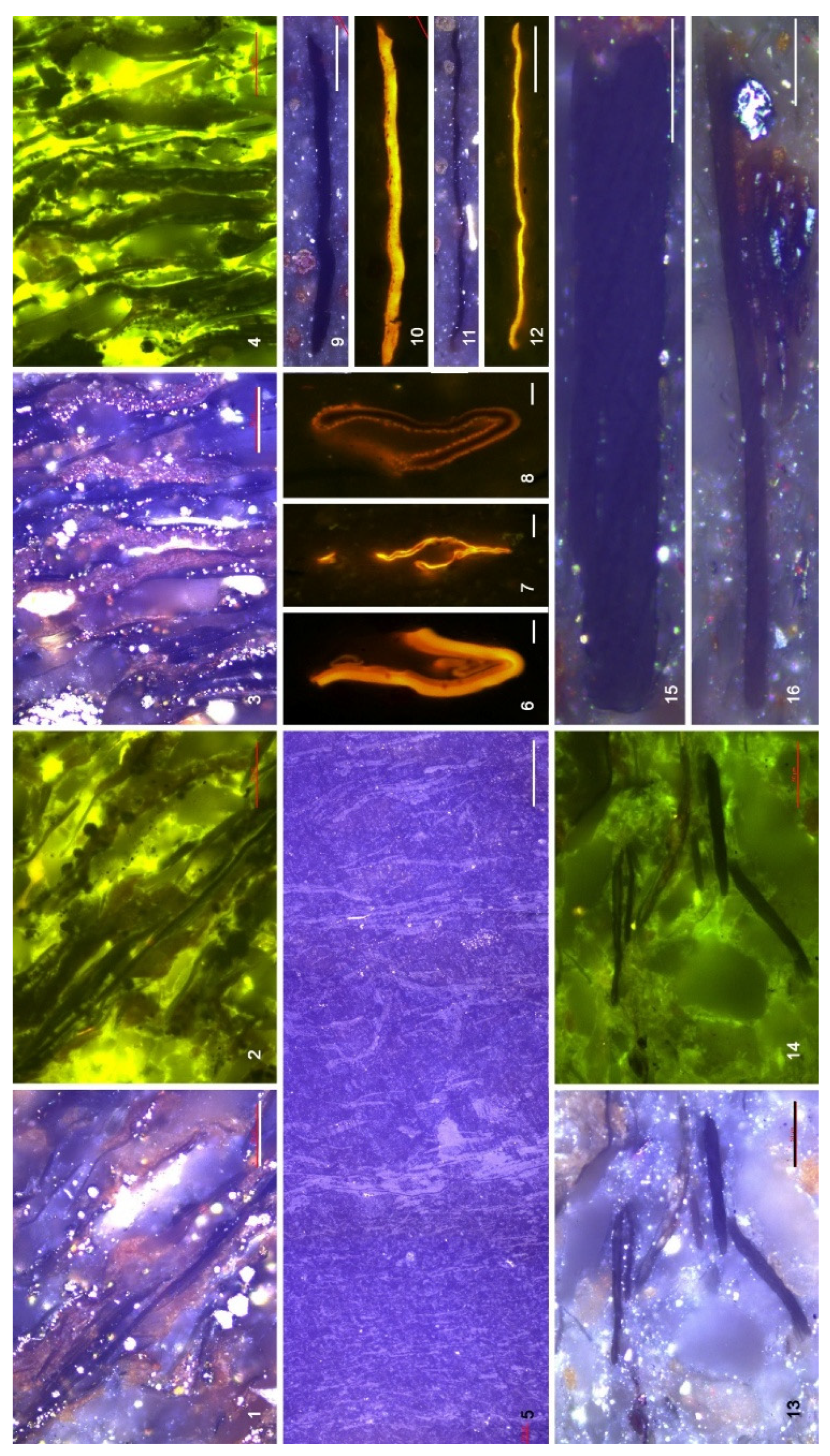
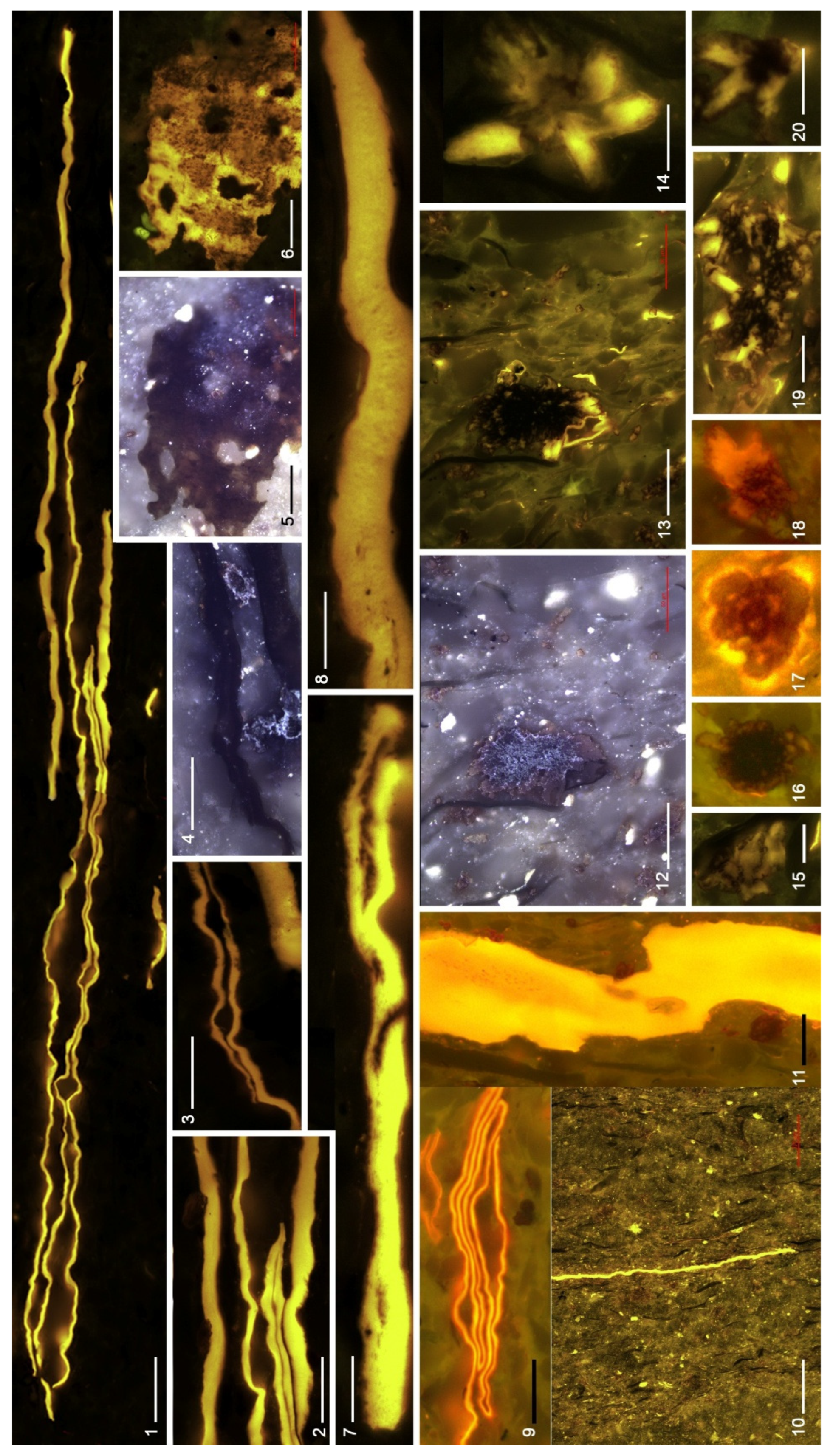
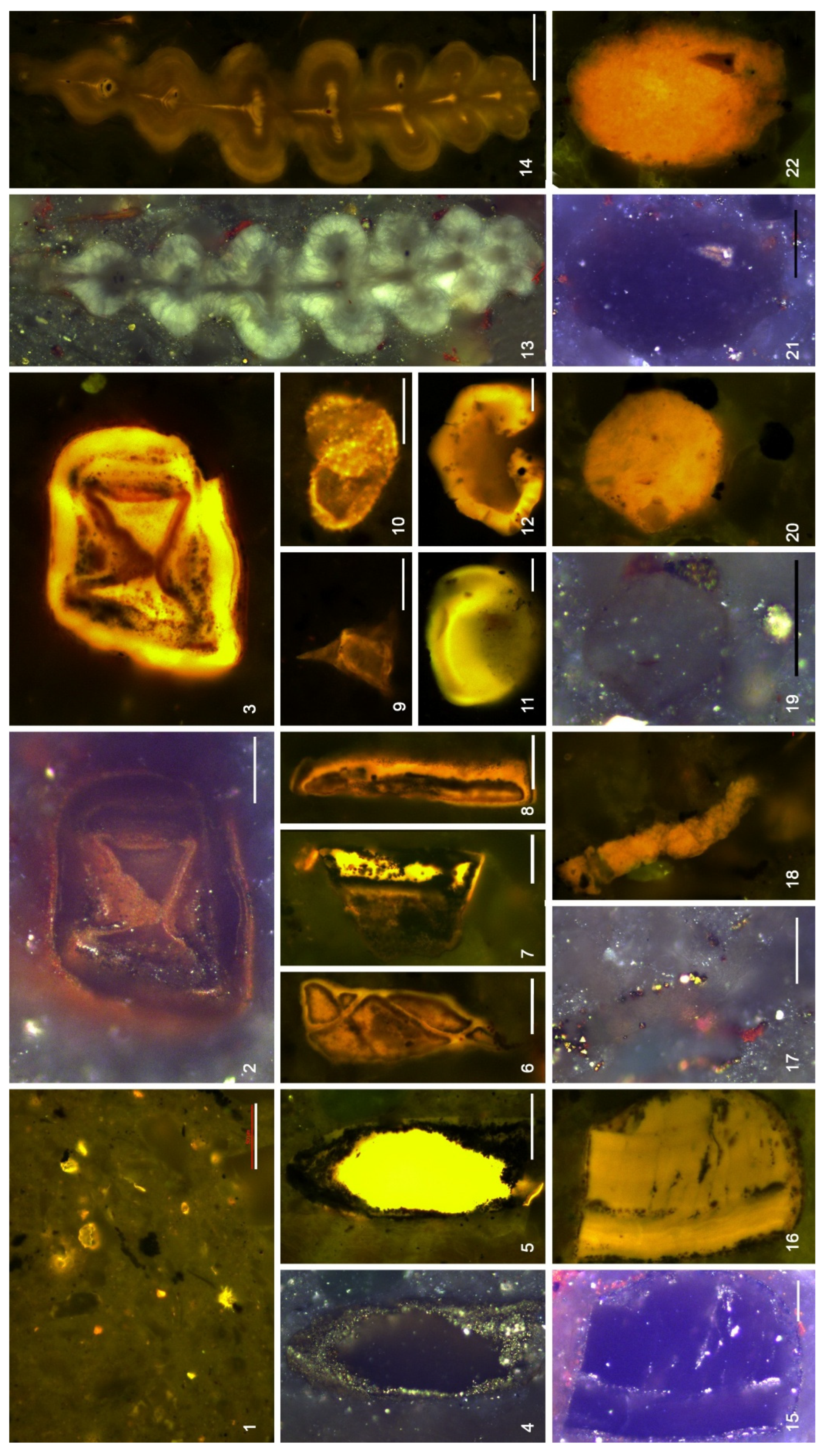
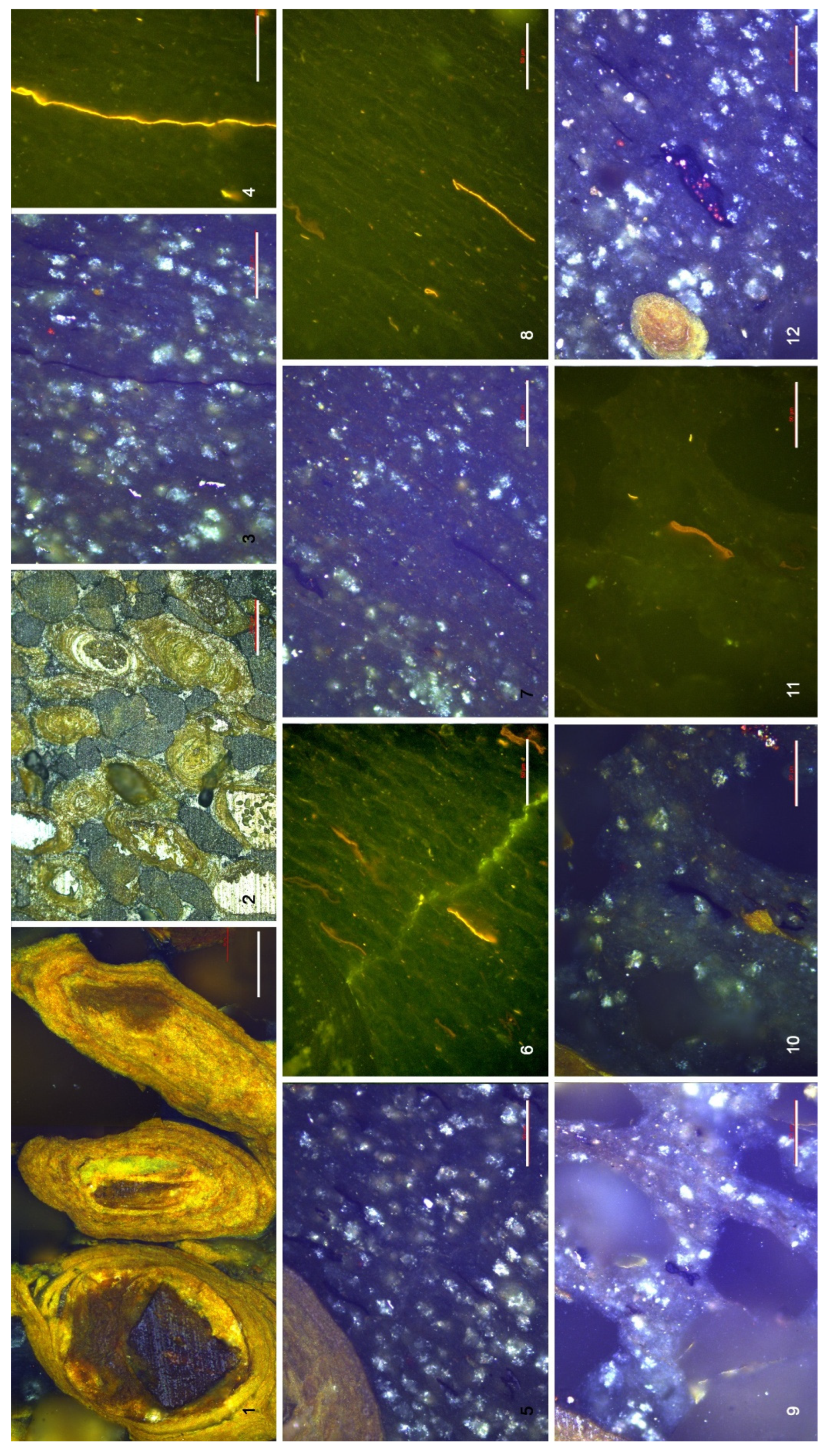
4.3. Organic Microfacies
5. Discussion
5.1. Paleoenvironmental Analysis of Organic Microfacies
5.1.1. Organic Microfacies A
5.1.2. Organic Microfacies B
5.1.3. Organic Microfacies C
5.2. Evolution of Organic Facies Through the Devonian and Their Significance
6. Conclusions
Author Contributions
Funding
Acknowledgments
References
- Markit, IHS. Petroleum exploration and production database; IRS Energy, Inc.: Houston, Texas, 2018; (database available from IHS Energy). [Google Scholar]
- EGPC (EGYPTIAN GENERAL PETROLEUM CORPORATION). Western Desert, oil and gas fields; EGPC: Cairo, Egypt, 1992; 431p. [Google Scholar]
- Makled, W.A.; Mostafa, T.F.; Mousa, D.A.; Abdou, A.A. Source rock evaluation and sequence stratigraphic model based on the palynofacies and geochemical analysis of the subsurface Devonian rocks in the Western Desert, Egypt. Marine and Petroleum Geology 2018, 89, 560–584. [Google Scholar] [CrossRef]
- Makled, W.A.; Hints, O.; Hosny, A.M.; Shahat, W.I.; Gentzis, T. Exotic Devonian palynomorphs from the Sifa-1X well in the Western Desert, Egypt. Palynology 2021a, 45(2), 363–380. [Google Scholar] [CrossRef]
- Tyson, R.V. The “productivity versus preservation” controversy: Cause, flaws, and resolution. In Deposition of Organic-Carbon-Rich Sediments: Models, Mechanisms, and Consequences; Harris, N., Ed.; SEPM Special Publication 82; 2005; pp. 17–33. [Google Scholar]
- Stasiuk, L.D.; Osadetz, K.G.; Goodarzi, F.; Gentzis, T. Organic microfacies and basinal tectonic control on source rock accumulation; a microscopic approach with examples from an intracratonic and extensional basin. International Journal of Coal Geology 1991, 19(1-4), 457–481. [Google Scholar] [CrossRef]
- Al-Juboury, A.I.; Al-Auqadi, R.S.; Alarifi, N.; Shingaly, W.S.; Vincent, S.J.; Howard, J.P.; Vautravers, B.P.H. Petrography and geochemistry of the Late Devonian Pirispiki Formation in northern Iraq: Implications for Reservoir Characteristics and reconstruction of depositional environments. Journal of African Earth Science 2024, 214, 105240. [Google Scholar] [CrossRef]
- Al-Auqadi, R.S.; Al-Juboury, A.I.; Alkhafaji, M.W.; Alarifi, N.; Makled, W.A.; Rowe, H.; Zanoni, G.; Dettman, D.L. Depositional environments and thermal maturity of the hydrocarbon source rocks in the Devonian–Early carboniferous Ora Formation from palynological organic petrographic investigations in northern and western Iraq. Journal of African Earth Science 2024a, 219, 105400. [Google Scholar] [CrossRef]
- Al-Auqadi, R.S.; Alkhafaji, M.W.; Al-Juboury, A.I.; Zumberge, A.; Alarifi, N.; Jarvie, D.; Zanoni, G.; Rowe, H. Organic geochemistry of the middle Paleozoic Ora Formation in Iraq: Implications for source rock assessment and hydrocarbon potentiality. Heliyon 2024b, 10, e29782. [Google Scholar] [CrossRef]
- Makled, W.A.; Gentzis, T.; Hosny, A.M.; Mousa, D.A.; Lotfy, M.M.; Abd El Ghany, A.A.; Shahat, W.I. Depositional dynamics of the Devonian rocks and their influence on the distribution patterns of liptinite in the Sifa-1X well, Western Desert, Egypt: implications for hydrocarbon generation. Marine and Petroleum Geology 2021b, 126, 104935. [Google Scholar] [CrossRef]
- Klitzsch, E. The Paleozoic. In The Geology of Egypt; Said, R., Ed.; Balkema: Rotterdam, 1990; pp. 393–406. [Google Scholar]
- Wanas, H.A. The Lower Paleozoic rock units in Egypt: an overview. Geoscience Frontiers 2011, 2(4), 491–507. [Google Scholar] [CrossRef]
- Keeley, M.L. Phanerozoic evolution of the basins of Northern Egypt and adjacent areas. Geol. Rundsch 1994, 83, 728–742. [Google Scholar] [CrossRef]
- Abd El Gawad, E.A.; Ghanem, M.F.; Makled, W.A.; Mousa, D.A.; Lotfy, M.M.; Temraz, M.G.; Shehata, A.M. Source rock evaluation of subsurface Devonian–Carboniferous succession based on palyno-organic facies analysis in Faghur Basin, north Western Desert of Egypt: a division of the north Africa Paleozoic basins. Arabian Journal of Geosciences 2019, 12, 655. [Google Scholar] [CrossRef]
- Ghori, K.A.R. Petroleum geochemical aspects of Cyrenaica, NE Libya. Third symposium on the geology of Libya; Salem, M.J., Busrewil, M.T., Ben Ashour, A.M., Eds.; Elsevier: Amsterdam, 1990; Volume 7, pp. 2743–2756. [Google Scholar]
- El-Arnauti, A.; Shelmani, M. Stratigraphic and structural setting. In: Thusu, B., Owens, B. (Eds.), The Palynostratigraphy of North-East Libya. Journal of Micropalaeontology, 1985, 4, 1–10.
- ASTM. D2797; Standard Practice for Preparing Coal Samples for Microscopical Analysis by Reflected Light; ASTM International: West Conshohocken, PA, USA, 2015. [Google Scholar]
- Taylor, G.H.; Teichmüller, Davis A.; Diessel, C.F.K.; Littke, R.; Robert, P. Organic Petrology; Gerbrüder Borntraeger: Berlin, 1998; p. 704. [Google Scholar]
- ASTM. D2799-23; Standard Test Method for Microscopical Determination of the Maceral Composition of Coal. ASTM International: West Conshohocken, PA, USA, 2023.
- ASTM (2023b). D7708-23a; Standard Test Method for Microscopical Determination of the Reflectance of Vitrinite Dispersed in Sedimentary Rocks. ASTM International: West Conshohocken, PA, USA. West Conshohocken, PA, USA.
- International Committee for Coal and Organic Petrology (ICCP). The new vitrinite classification (ICCP System 1994). Fuel 1998, 77(5), 349–358. [Google Scholar] [CrossRef]
- International Committee for Coal and Organic Petrology (ICCP). The new inertinite classification (ICCP System 1994). Fuel 2001, 80(4), 459–471. [Google Scholar] [CrossRef]
- Pickel, W.; Kus, J.; Flores, D.; Kalaitzidis, S.; Christanis, K.; Cardott, B. J.; Misz-Kennan, M.; Rodrigues, S.; Hentschel, A.; Hamor-Vido, M.; ICCP. Classification of liptinite—ICCP system 1994. International Journal of Coal Geology 2017, 169, 40–61. [Google Scholar] [CrossRef]
- Ward, J. H., Jr. Hierarchical Grouping to Optimize an Objective Function. Journal of the American Statistical Association 1963, 58(301), 236–244. [Google Scholar] [CrossRef]
- Makled, W.A.; Baioumi, A.H.; Saleh, R.A. Palynostratigraphical studies on some subsurface middle Albian–early Cenomanian sediments from North Western Desert, Egypt. Egyptian Journal of Petroleum 2013, 22(4), 501–515. [Google Scholar] [CrossRef]
- Makled, W.A.; Mostafa, T.F.; Maky, A. Mechanism of Late Campanian–Early Maastrichtian oil shale deposition and its sequence stratigraphy implications inferred from the palynological and geochemical analysis. Egyptian Journal of Petroleum 2014, 23(4). [Google Scholar] [CrossRef]
- Riding, J.B. A guide to preparation protocols in palynology. Palynology 2021, 45(sup1), 1–110. [Google Scholar] [CrossRef]
- Mostafa, T.F. Palynostratigraphic Study of Devonian Rocks of the Western Desert, Egypt. Unpublished PhD thesis, Ain Shams University, Faculty of Science, Geology Department, 1997. [Google Scholar]
- El Shamma, A.A.; Mostafa, T.F.; Abdel Malik, W.M. Devonian spores from subsurface rocks in the Western Desert, Egypt. In Proceedings of the 14th Petroleum Conference, EGPC, Cairo, 1998; pp. 1451–1465. [Google Scholar]
- Tyson, R.V. Sedimentary organic matter: organic facies and palynofacies; Springer Science and Business Media, 1995. [Google Scholar]
- Wang, H.; Lei, M.; Chen, Y.; Li, M.; Zou, L. Intelligent identification of maceral components of coal based on image segmentation and classification. Applied Sciences 2019, 9(16), 3245. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Cardona, A. Fiji: an open-source platform for biological-image analysis. Nature methods 2012, 9(7), 676–682. [Google Scholar] [CrossRef]
- Skiba, M. The influence of the discrepancies in the observers’ decisions on the process of identification of maceral groups using artificial neural networks. Journal of Sustainable Mining 2016, 15(4), 151–155. [Google Scholar] [CrossRef]
- Rubo, R.A.; de Carvalho Carneiro, C.; Michelon, M.F.; dos Santos Gioria, R. Digital petrography: Mineralogy and porosity identification using machine learning algorithms in petrographic thin section images. Journal of Petroleum Science and Engineering 2019, 183, 106382. [Google Scholar] [CrossRef]
- Witten, I.H.; Frank, E.; Hall, M.A.; Pal, C.J.; Data, M. Practical machine learning tools and techniques. In Data mining; Elsevier: Amsterdam, The Netherlands, 2005; Volume 2, 4, pp. 403–413. [Google Scholar]
- Wendt, J. The first tunicate with a calcareous exoskeleton (Upper Triassic, northern Italy). Palaeontology 2018, 61(4), 575–595. [Google Scholar] [CrossRef]
- Bankole, S.; Buckman, J.; Stow, D. Unusual Components Within a Fine-Grained Contourite Deposit: Significance for Interpretation of Provenance and the Contourite Budget. Minerals 2020, 10(6), 488. [Google Scholar] [CrossRef]
- Goodarzi, F. Chitinous fragments in coal. Fuel 1984, 63(11), 1504–1507. [Google Scholar] [CrossRef]
- Eisenack, A. Chitinozoen, Hystrichospharen und andere Mikrofossilien aus dem Beyrichia-Kalk. Senckenbergiana lethaea 1995, 36, 157- 188, pl. 1-5. [Google Scholar]
- Taugourdeau, P. Debris microscopiques d’eurypterides du Paleozoique saharien. Revue de Micropaleontologie 1967, 10 pl. 1(2), 119–127. [Google Scholar]
- Miller, M.A. Chapter 13E. Invertebrate cuticular fragments. In Palynology: principles and applications; Jansonius, J., McGregor, D.C., Eds.; American Association of Stratigraphic Palynologists Foundation, 1966; Volume 1, pp. 381–382. [Google Scholar]
- Braun, A. Occurrence, investigation methods and significance of animal cuticle in Devonian and Carboniferous coal-bearing sedimentary rocks. Palaeontographica Abteilung 1997, A 245, 83–156. [Google Scholar] [CrossRef]
- Makled, A.W.; Al-Auqadi, R.S.; Al-Juboury, A.I.; El Garhy, M.M.; Alarifi, N.; Omar, N.; Mahmoud, A. Eurypterid setae and cuticle fragments from the Ora Fomation (Upper Devonian) of Iraq. Palynology 2024. [Google Scholar] [CrossRef]
- Mikrotax. Ascidian spicules [Nannotax3 website]. Woodside Energy and Shell USA, 2024. Available online: https://www.mikrotax.org/Nannotax3/index.php?taxon=Ascidian%20spiculesandmodule=non_cocco.
- Flügel, E.A. Microfacies of carbonate rocks: analysis, interpretation and application; Springer: Berlin, 2010; Volume 976, p. 2004. [Google Scholar]
- Katz, B.J. Controlling factors on source rock development—a review of productivity, preservation, and sedimentation rate. In Deposition of Organic-Carbon-Rich Sediments: Models, Mechanisms, and Consequences; Harris, N., Ed.; SEPM Special Publication 82; 2005; pp. 7–16. [Google Scholar]
- Röhl, H.-J.; Schmid-Röhl, A. Lower Toarcian (Upper Liassic) Black Shales of the Central European Epicontinental Basin: A Sequence Stratigraphic Case Study from the SW German Posidonia Shale. In Deposition of Organic-Carbon-Rich Sediments: Models, Mechanisms, and Consequences, SEPM Special Publication; Harris, N., Ed.; SEPM Special Publication 82; 2005; pp. 165–189. [Google Scholar]
- Abou El-Anwar, E.; Salman, S.; Makled, W.; Mousa, D.; Gentzis, T.; Shazly, T.F. Depositional mechanism of Duwi Formation organic-rich rocks in anoxic Campanian-early Maastrichtian condensed sections in the Qusseir–Safaga region in Eastern Desert of Egypt and their economic importance. Marine and Petroleum Geology 2024, 163, 106759. [Google Scholar]
- Van Wagoner, J.C.; Mitchum, R.M.; Campion, K.M.; Rahmanian, V.D. Siliciclastic sequence stratigraphy in well logs, cores, and outcrops: concepts for high-resolution correlation of time and facies. In American Association of Petroleum Geologists; 1990; Volume 7. [Google Scholar] [CrossRef]
- Gao, P.; Khong, H.Y.; Wibowo, A.; Zhen, Y.; Peng, C.; Miao, W. Chemical compositions and nutritional profiles of two edible tunicate species (Halocynthia roretzi and Halocynthia aurantium). Heliyon 2024, 10(12). [Google Scholar] [CrossRef]
- Millar, R.H. The biology of ascidians. In Advances in Marine Biology; Academic Press, 1971; Volume 9, pp. 1–100. [Google Scholar]
- O’Brien, G.W.; Milnes, A.R.; Veeh, H.H.; Heggie, D.T.; Riggs, S.R.; Cullen, D.J.; Marshall, J.F.; Cook, P.J. Sedimentation dynamics and redox iron-cycling: controlling factors for the apatite-glauconite association on the East Australian continental margin. In Phosphorite Research and Development; Northolt, A.J.G., Jarvis, I., Eds.; Geological Society of London Special Publication, 1990; vol. 52, pp. 61–86. [Google Scholar]
- Scholle, P.A.; Ulmer-Scholle, D.S. A color guide to the petrography of carbonate rocks: grains, textures, porosity, diagenesis. AAPG Memoir 2003, 77, 77. [Google Scholar]
- Tang, D.; Xie, B.; Shi, X.; Zhou, X. Low level of phosphorous concentration in terminal Paleoproterozoic shallow seawater: Evidence from Chuanlinggou ironstone on North China Platform. Precambrian Research 2022, 370, 106554. [Google Scholar] [CrossRef]
- Rudmin, M.; Mazurov, A.; Banerjee, S. Origin of ooidal ironstones in relation to warming events: Cretaceous-Eocene Bakchar deposit, south-east Western Siberia. Marine and Petroleum Geology 2019, 100, 309–325. [Google Scholar] [CrossRef]
- Abram, M.B.; Holz, M. Early to Middle Devonian ironstone and phosphorite in the northwestern Gondwana Parnaíba Basin, Brazil: A record of an epeiric margin paleoceanographic changes. Sedimentary Geology 2020, 402, 105646. [Google Scholar] [CrossRef]
- Rudmin, M.; Banerjee, S.; Maximov, P.; Novoselov, A.; Trubin, Y.; Smirnov, P.; Mazurov, A. Origin of ooids, peloids and micro-oncoids of marine ironstone deposits in Western Siberia (Russia). Journal of Asian Earth Sciences 2022, 237, 105361. [Google Scholar] [CrossRef]
- Vodrážková, S.; Kumpan, T.; Vodrážka, R.; Frýda, J.; Čopjaková, R.; Koubová, M.; Holá, M. Ferruginous coated grains of microbial origin from the Lower Devonian (Pragian) of the Prague Basin (Czech Republic)–Petrological and geochemical perspective. Sedimentary Geology 2022, 438, 106194. [Google Scholar]
- Zhou, M.F.; Lyu, Y.J.; Liu, Z.R.; Liu, P.P.; Meng, L.; Qiu, W.J.; Zhao, W.W. Devonian stratiform ironstone deposits in South China formed in a shallow marine environment of a passive continental margin. Journal of Asian Earth Sciences 2024, 262, 105997. [Google Scholar] [CrossRef]
- Young, T.P. Eustatically controlled ooidal ironstone deposition: facies relationships of the Ordovician open-shelf ironstones of Western Europe. In Geological Society, London, Special Publications; 1989; Volume 46, 1, pp. 51–63. [Google Scholar]
- Peryt, T.M. Phanerozoic oncoids—an overview. Facies 1981, 4, 197–213. [Google Scholar] [CrossRef]
- Jones, B.; Renaut, R.W. Formation of silica oncoids around geysers and hot springs at El Tatio, Chile. Sedimentology 1997, 44, 287–304. [Google Scholar] [CrossRef]
- Riding, R. Microbial carbonates: the geological record of calcified bacterial–algal mats and biofilms. Sedimentology 2000, 47, 179–214. [Google Scholar] [CrossRef]
- Védrine, S.; Strasser, A.; Hug, W. Oncoid growth and distribution controlled by sea level fluctuations and climate (Late Oxfordian, Swiss Jura Mountains). Facies 2007, 53, 535–552. [Google Scholar] [CrossRef]
- Bádenas, B.; Aurell, M. Facies models of a shallow-water carbonate ramp based on distribution of non-skeletal grains (Kimmeridgian, Spain). Facies 2010, 56, 89–110. [Google Scholar] [CrossRef]
- Zaton, M.; Kremer, B.; Marynowski, L.; Wilson, M.A.; Krawczynski, W. Middle Jurassic (Bathonian) encrusted oncoids from the Polish Jura, southern Poland. Facies 2012, 58, 57–77. [Google Scholar] [CrossRef]
- Sequero, C.; Aurell, M.; Bádenas, B. Sedimentary evolution of a shallow carbonate ramp (Kimmeridgian, NE Spain): unravelling controlling factors for facies heterogeneities at reservoir scale. Marine and Petroleum Geology 2019, 109, 145–174. [Google Scholar] [CrossRef]
- Sequero, C.; Aurell, M.; Bádenas, B. Oncoid distribution in the shallow domains of a Kimmeridgian carbonate ramp (Late Jurassic, NE Spain). Sedimentary Geology 2020, 398, 105585. [Google Scholar] [CrossRef]
- Stasiuk, L.D.; Fowler, M.G. Organic facies in Devonian and Mississippian strata of Western Canada Sedimentary Basin: relation to kerogen type, paleoenvironment, and paleogeography. Bulletin of Canadian Petroleum Geology 2014, 52(3), 234–255. [Google Scholar] [CrossRef]
- Stasiuk, L.D. Microscopic Studies of Sedimentary Organic Matter: Key to Understanding Organic-Rich Strata, with Paleozoic Examples from Western Canada Basin. Geoscience Canada 1999, 26(4), 149–172. [Google Scholar]
- Loutit, T.S.; Hardenbol, J.; Vail, P.R.; Baum, P. Condensed sections: the key to age determination and correlation of continental margin sequences. In Sea-level Changes — An Integrated Approach. Society of Economic Paleontologists and Mineralogists; Wilgus, C.K., Hastings, B.S., Posamentier, H., Van Wagoner, J., Ross, C.A., Kendall, C.G.S.C., Eds.; 1988; Volume Special Publication 42, pp. 183–213. [Google Scholar]
- Föllmi, K.B. Sedimentary condensation. Earth-Science Reviews 2016, 152, 143–180. [Google Scholar] [CrossRef]
- Hallett, D.; Clark-Lowes, D. Petroleum geology of Libya; Elsevier, 2017; p. 391. [Google Scholar]
- Rubino, J.L.; Blanpied, C. Sedimentology and sequence stratigraphy of the Devonian to lowermost Carboniferous succession on the Gargaf Uplift (Murzuq Basin, Libya). In Geological Exploration in Murzuq Basin; Elsevier Science BV, 2000; pp. 321–348. [Google Scholar]
- Guerrak, S. Time and space distribution of Palaeozoic oolitic ironstones in the Tindouf Basin, Algerian Sahara. In Geological Society, London; Special Publications, 1989; Volume 46, 1, pp. 197–212. [Google Scholar]







| Stratified cross-validation (10 folds) | 19-A sample | 18-B sample | 14-A sample |
|---|---|---|---|
| Correctly Classified Instances | 140699 (99.9%) | 97117(99.5%) | 173181 (100%) |
| Incorrectly Classified Instances | 4 (0.0028%) | 527 (0.54%) | 15(0.01%) |
| Kappa statistic | 0.9985 | 0.99 | 0.9997 |
| Mean absolute error | 0.0003 | 0.02 | 0.003 |
| Root mean squared error | 0.0064 | 0.07 | 0.02 |
| Relative absolute error | 2.6172% | 4.98% | 1.7864% |
| Root relative squared error | 7.9321% | 15.1% | 6.05% |
| Total Number of Instances | 140703 | 97644 | 173196 |
| Organic microfacies | Depth (ft) | Total organic density | Liptinite | Vitrinite | Inertinite | Acritarchs | Chitinozoans | Scolecodonts | Prasinophyte | AOM | Spores | Translucent Phytoclasts | Non-translucent Phytoclasts | Lamination | Organic microfacies |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 14-A | 7707 | 10.0 | 6.0 | 3.0 | 1.0 | 0.0 | 0.0 | 0.0 | 0.0 | 18.8 | 43.8 | 36.4 | 1.0 | 1 | B |
| 15-A | 7942 | 6.0 | 4.4 | 0.9 | 0.7 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0 | C |
| 15-B | 7948 | 0.1 | 0.1 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 1 | C |
| 16-A | 8203 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0 | C |
| 17-A | 8525 | 2.0 | 1.5 | 0.5 | 0.0 | 0.7 | 0.0 | 0.0 | 0.4 | 27.5 | 59.6 | 11.9 | 0.0 | 1 | A |
| 18-B | 8948 | 1.1 | 0.8 | 0.3 | 0.0 | 7.6 | 9.6 | 0.1 | 0.0 | 17.5 | 53.7 | 11.6 | 0.0 | 0 | A |
| 18-A | 8968 | 1.3 | 1.2 | 0.1 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0 | C |
| 19-A | 9125 | 7.9 | 7.9 | 0.0 | 0.0 | 0.0 | 1.2 | 0.5 | 0.2 | 29.4 | 52.2 | 16.6 | 0.0 | 0 | A |
| 20-C | 9371 | 9.0 | 3.4 | 5.6 | 0.0 | 0.4 | 0.5 | 0.2 | 0.4 | 11.4 | 54.7 | 28.8 | 3.8 | 1 | B |
| 20-A/B | 9374 | 11.1 | 0.0 | 11.1 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 10.6 | 25.8 | 56.4 | 7.2 | 1 | B |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




