Submitted:
20 March 2026
Posted:
23 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
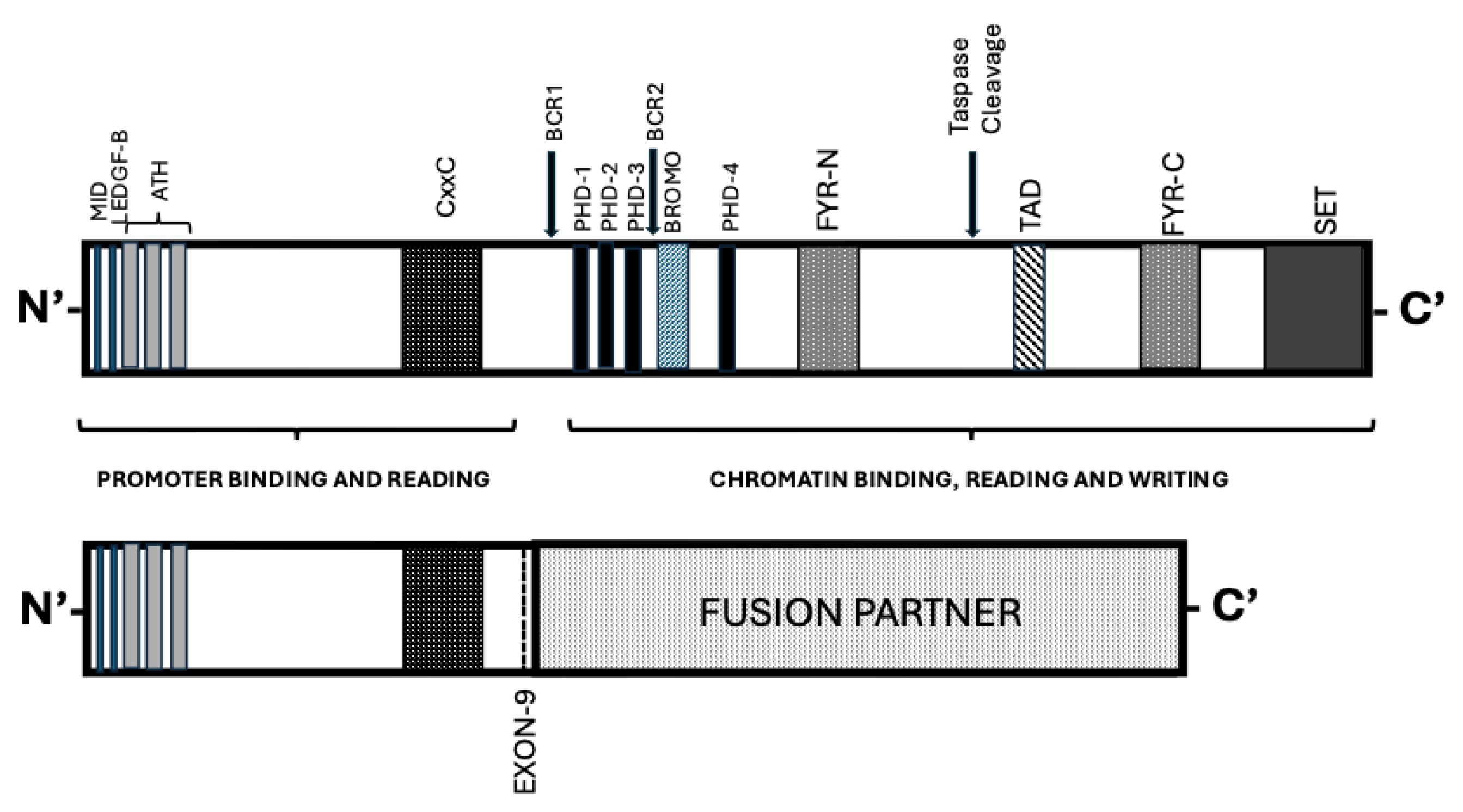
2. KMT2A Gene and Protein Structure
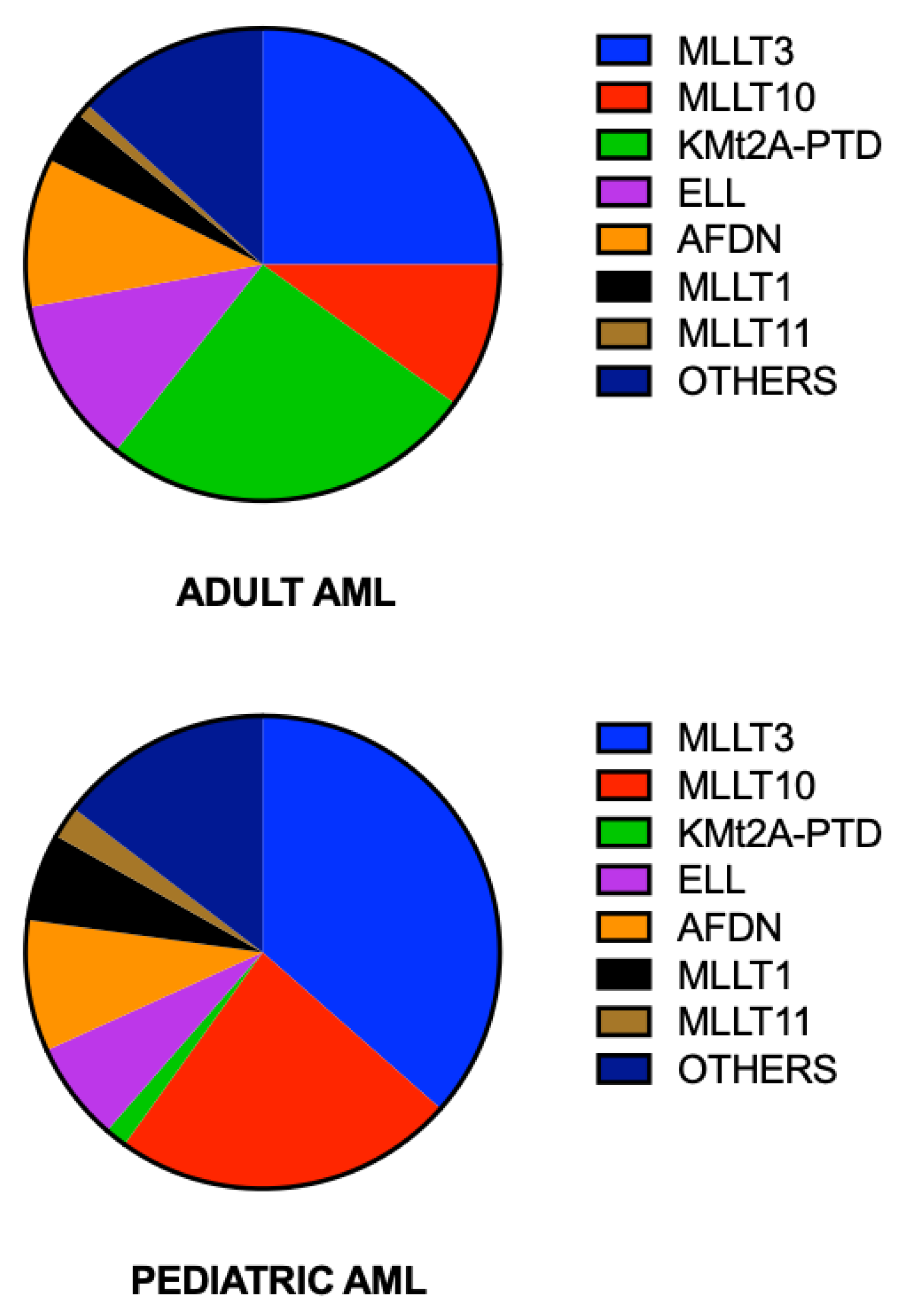
3. AMLs with KMT2A Rearrangements
4. Standard Treatment of AML Patients with KMT2A Rearrangements
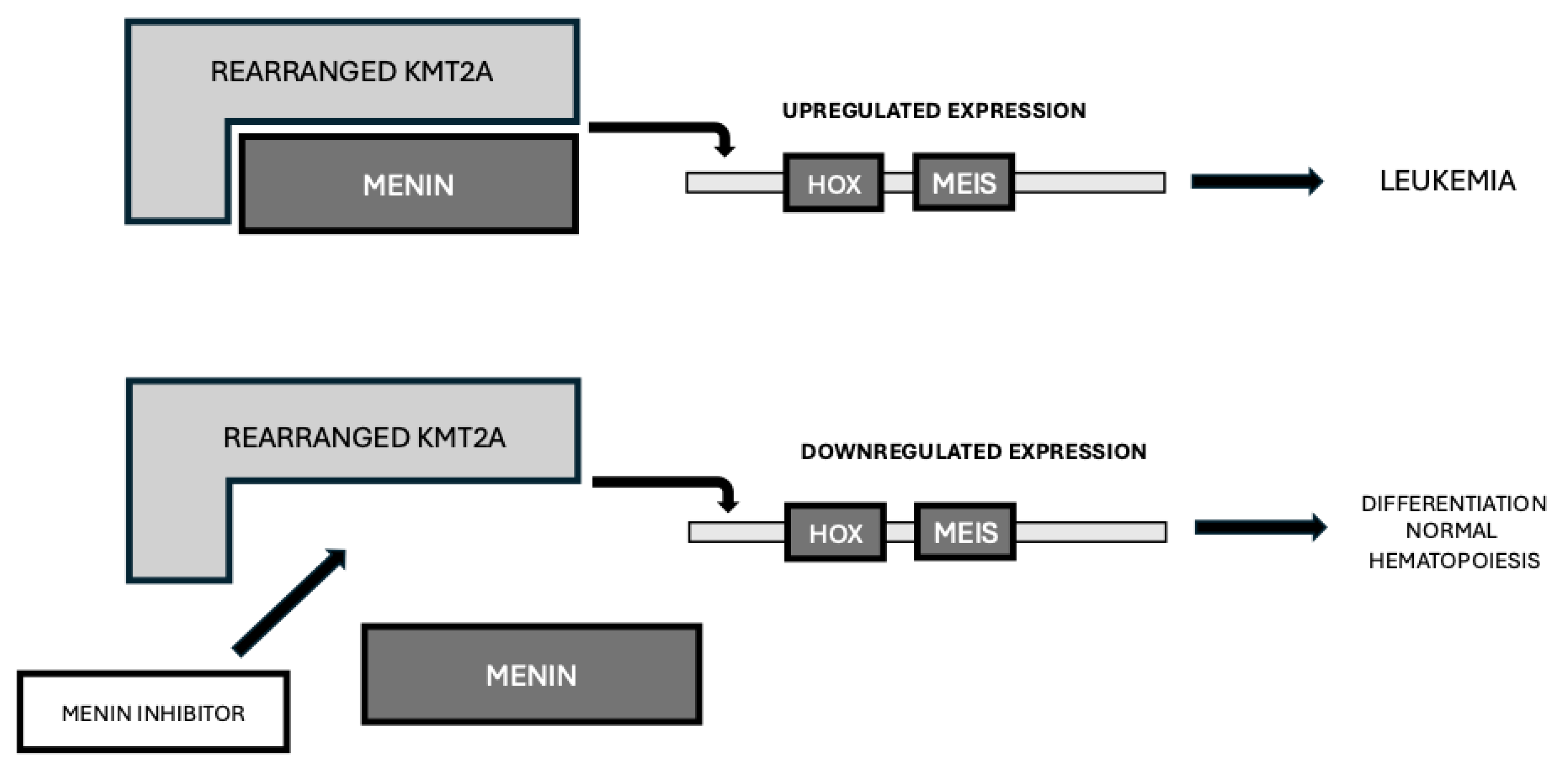
5. Target Therapy of KMT2A-r AMLs
5.1. Monotherapy Studies with Menin Inhibitors
5.1.1. Monotherapy Studies with Revumenib
5.1.2. Monotherapy Studies with Enzomenib
5.1.3. Monotherapy Studies with Ziftomenib
5.1.4. Monotherapy Studies with Bleximenib
5.2. Menin Inhibitors in Combination with Chemotherapy
5.2.1. Revumenib in Combination with Chemotherapy
5.2.2. Ziftomenib in Association with Chemotherapy
5.2.3. Bleximenib in Association with Chemotherapy
5.3. Menin Inhibitors in Combination with Venetoclax
5.3.1. Revumenib in Association with Venetoclax
5.3.2. Zeftomenib in Association with Venetoclax
5.3.3. Bleximenib in Association with Venetoclax
5.3.4. Enzomenib in Association with Venetoclax
5.4. Menin Inhibitors in Combination with FLT3 Inhibitors
5.5. Menin Inhibitors as Post-Transplant Maintenance Therapy in KMT2A-r AML
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Bawwab, A.; Nurse, D.P.; El-Asmar, J.; Abu-Farshak, Y.; Albliwi, M.; Hanna, J.; Rauf, A.; Batah, H.; Abuamsha, H.; Zabor, E.C.; et al. Comprehensive analysis of KMT2A alterations in acute myeloid leukemia and their impact on survival response rates: A single center experience. Blood (suppl.1), 4249–4250. [CrossRef]
- Van, H.T.; Xie, G.; Dong, P.; Liu, Z.; Ge, K. KMT2 family of H3K4 methyltransferases: Enzymatic activity-pendent and -independent functions. J Mol Biol 2024, 146, 168453. [Google Scholar] [CrossRef]
- Ogino, J.; Dou, Y. Histone methyltransferase KMT2A: Developmental regulation to oncogenic transformation. J Biol Chem 2024, 300, 107791. [Google Scholar] [CrossRef]
- Meyer, C.; Larghero, P.; Lopes, B.A.; Marschalek, R. The KMT2A/MLL consensus gene: A comprehensive update for research and diagnostic implications. Leukemia 2024, 38, 1403–1406. [Google Scholar] [CrossRef]
- Zehtabchech, S.; Samarkhazan, H.S.; Asan, M.; Zabihi, M.; Parkhiden, S.; Mohammadi, M.H. Insights into KMT2A rearrangements in acute myeloid leukemia: From molecular characteristics to targeted therapies. 2025, 13, 73. [Google Scholar]
- Guarnera, L.; D’Addona, M.; Bravo-Perez, C.; Visconte, V. KMT2A rearrangements in leukemias: Molecular aspects and therapeutic perspectives. Int J Mol Sci 2024, 25, 9023. [Google Scholar] [CrossRef] [PubMed]
- Ernst, P.; Kyel, P.; Yokoyama, A. KMT2a-rearrangement leukemia: From mechanism to drug development. Exp Hematol 2025, 151, 105247. [Google Scholar] [CrossRef] [PubMed]
- Seto, A.; Downs, G.; King, O.; Salehi-Rad, S.; Baptista, A.; Chin, K.; Grenier, S.; Nwachukuwu, B.; Tierens, A.; Minden, M.D.; et al. Genomic characterization of partial tandem duplication involving the KMT2A gene in adult acute myeloid leukemia. Cancers 2024, 26, 1693. [Google Scholar] [CrossRef]
- Wenge, D.; Wessels, M.; Schweber, S.; Boileau, M.; Marinaccio, C.; Bourgeois, W.; Hatton, C.; Godfrey, L.; Shan, Y.; Akele, T.; et al. The KMT2A-PTD oncoprotein depends on ENL but not menin to drive AML gene expression. Blood 2025, 146, 321–322. [Google Scholar] [CrossRef]
- Huang, W.; Zhang, F.; Zhang, Z.; Wang, Q.; Chen, S. Dysregulation of HOXA genes in acute myeloid leukemia and targeted therapy. Npj Precision Oncol 2025, 9, 396. [Google Scholar] [CrossRef]
- Wong, S.; Goode, D.L.; Ieasaki, M.; Wei, M.; Kuo, H.P.; Zhu, L.; Schneidawind, D.; Duque-Afonso, J.; Weng, Z.; Cleary, M. The H3K4-methyl epigenome regulates leukemia stem cell oncogenic potential. Cancer Cell 2015, 28, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Izumi, S.; Ohtani, K.; Matsumoto, M.; Shibata, S.; Rahmutulla, B.; Fukuyo, M.; Nishimoto, M.; Miyagama, H.; Sakaida, E.; Yokote, K.; et al. Regulation of H3K4me3 breadth and MYC expression by the SETD1B catalytic domain in MLL-rearranged leukemia. Leukemia 2025, 39, 1627–1639. [Google Scholar] [CrossRef] [PubMed]
- Bolouri, H.; Farrar, J.E.; Triche, T.; Ries, R.; Lim, E.; Alonzo, T.A.; Ma, Y.; Moore, R.; Mungall, A.J.; Marra, M.; et al. The molecular landscape of pediatric acute myeloid leukemia reveals recurrent structural alterations and age-specific mutational interactions. Nat Med 2018, 24, 103–112. [Google Scholar] [CrossRef]
- Yuen, K.Y.; Liu, Y.; Zhou, Y.Z.; Wang, Y.; Zhou, D.H.; Fang, J.P.; Xu, L.H. Mutational landscape and clinical outcome of pediatric acute myeloid leukemia with 11q23/KMT2A rearrangements. Cancer Medicine 2023, 12, 1418–1430. [Google Scholar] [CrossRef]
- Dong, Y.; Zhao, X.; Feng, X.; Zhou, Y.; Yan, X.; Zhang, Y.; Bu, J.; Zhan, D.; Hayashi, Y.; Zhang, Y.; et al. SETD2 mutations confer chemoresistance in acute myeloid leukemia partly through altered cell cycle checkpoints. Leukemia 2019, 33, 2585–2589. [Google Scholar] [CrossRef]
- Van Weelderen, R.; Harrison, C.; Klein, K.; Jiang, Y.; Abrahmsson, J.; Alonzo, T.; Apleno, R.; Arad-Cohen, N.; Bert-Delabesse, E.; Buldini, B.; et al. Optimized cytogenetic risk group stratification of KMT2A-rearranged pediatric acute myeloid leukemia. Blood Adv 2024, 8, 3200–3213. [Google Scholar] [CrossRef]
- Van Weldeeren, R; Klein, K.; Harrison, C.J.; Jiang, Y.; Abrahamsson, J.; Arad-Cohen, N.; Bart-Delabesse, E.; Buldini, B.; De Moerloose, B.; Dworzak, M.N.; et al. Measurable residual disease and fusion partner independently predict survival and relapse risk in childhood KMT2A-rearranged acute myeloid leukemia: A study by the international Berlin Frankfurt-Munster study group. J Clin Oncol 2023, 41, 2963–2974. [Google Scholar] [CrossRef] [PubMed]
- Shoji, K.; Yoshida, K.; Iyoda, S.; Ishikawa, M.; Tanaka, M.; Nobe, M.; Saito, N.; Shino, Y.; Nannva, Y.; Yamato, G.; et al. Age-specific mutation profiles and their prognostic implications in pediatric KMT2A-rearranged acute myeloid leukemia. Haematologica 2026. [Google Scholar] [CrossRef]
- Iyoda, S.; Yoshida, K.; Shoji, K.; Ito, N.; Tanaka, M.; Nannya, Y.; Yamato, G.; Tsujimoto, S.; Shiba, N.; Hayashi, Y.; et al. KRAS G12 mutations as adverse prognostic factors in KMT2A-rearranged acute myeloid leukemia. Leukemia 2024, 38, 1609–1612. [Google Scholar] [CrossRef]
- Hernandez-Sancho, A.; Gonzalez, T.; Sobas, M.; Starng, E.; Castellani, G.; Abadigar, M.; Valk, P.; Vivvavertde Ramiro, A.; Benner, A.; Metzler, K.; et al. Rearrangements involving 11q23.3/KMT2A in adult AML: Mutational landscape and prognostic implications—A HARMONY study. Leukemia 2024, 38, 1929–1937. [Google Scholar] [CrossRef]
- Batayneh, O.; Moein, M.; Naji, N.S.; Patel, A.; Mandawa, A.; Goodman, A.; Ross, J.; Ho, C.; Marcus, C.; Zhou, Z.; et al. Characterizing KMT2A rearrangement in acute myeloid leukemia: A comprehensive genomic study. Cancers 2026, 18, 161. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Yuan, X.; Lai, X.; Liu, L.; Shi, J.; Zhang, W.; Yu, J.; Zhao, Y.; Sun, J.; Ye, X.; et al. The mutated of KRAS gene determine the fate of KMT2A-rearranged AML patients. Blood 2024, 144, 843–844. [Google Scholar] [CrossRef]
- Bill, M.; Mrozek, K.; Kohlschmidt, J.; Eisfeld, A.K.; Walker, C.J.; Nicolet, D.; Papaioannu, D.; Bachly, J.S.; Orwick, S.; Carroll, A.J.; et al. Mutational landscape and clinical outcome of patients with de novo acute myeloid leukemia and rearrangements involving 11q23/KMT2A. Proc Natl Acad Sci USA 2020, 117, 26340–26346. [Google Scholar] [CrossRef]
- Othman, J.; Potter, N.; Freeman, S.D.; McCarthy, N.; Jovanovic, J.; Runglall, M.; Cunham, J.; Thomas, I.; Johnson, S.; Gilkes, A.; et al. Induction chemotherapy and molecular MRD influence outcomes in KMT2A-rearranged AML. Blood 2025, 146, 1862–1867. [Google Scholar] [CrossRef]
- DiNardo, C.D.; Jen, W.Y.; Takahashi, K.; Kadia, T.; Loghavi, S.; Daver, N.G.; Xiao, L.; Reville, P.K.; Issa, G.C.; Short, N.J.; et al. Long term results of venetoclax combined with FLAG+IDA induction and consolidation for newly diagnosed and relapsed or refractory acute myeloid leukemia. Leukemia 2025, 39, 854–863. [Google Scholar] [CrossRef]
- Zheng, Y.; Li, J.; Li, H.; Xheng, M.; Zhang, B.; Mai, H.; Liu, S.; Li, C.; Yang, L.; Yu, L.; et al. FLAG-IDA as frontline induction improves outcomes in pediatric patients with core-binding factor or KMT2A-rearranged acute myeloid leukemia: A multicenter cohort study in southern China. Blood 2025, 146, 5152. [Google Scholar] [CrossRef]
- Loo, S.; Potter, N.; Ivey, A.; O’Nions, J.; Moon, R.; Fong, C.Y.; Anstee, N.; Tiong, I.S.; Chua, C.C.; Othman, J.; et al. Pre-transplant measurable residual disease (MRD) detection of KMT2A-rearranged acute myeloid leukemia is strongly associated with inferior post-transplant outcome. Blood 2023, 142, 1577–1579. [Google Scholar] [CrossRef]
- Wang, L.; Chen, Y.; Zang, M.; Zhou, J.; Xiao, M.; Fu, H.; Mo, X.; Wang, F.; Han, W.; Zhang, Y.; et al. Molecular measurable residual disease before transplantation independently predicts survival and relapse risk in adult lysine methyltransferase 2a-rearranged acute myeloid leukemia. Cancer 2025, e35717. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Huang, H.; Zhang, Y.; Xia, Y.; Huang, J.; Jiang, C.; Wang, L.; Lu, H.; Pan, Z.; Wang, G.; et al. Oucomes of acute myeloid leukemia with kMT2A (MLL) rearrangement: A multicenter study of TROPHY group. Blood Cancer J 2025, 15, 84. [Google Scholar] [CrossRef]
- McMahon, C.; Nicolet, D.; Mims, A.; Foucar, C.; Madanat, Y.; Ragon, B.; Sahasrabudhe, K.; Chan, O.; Zeidner, J.; Hochman, M.; et al. Defining the clinical characteristics and outcomes of adult patients (pts) with 11q23/KMT2A-rearranged acute myeloid leukemia (AML): And analysis from the MARROW consortium. Blood 146, 292–293. [CrossRef]
- Singh, A.; Bosma, G.; Zhang, J.; Abbott, D.; Amaya, M.; Kent, A.; Angelos, M.; Schwartz, M.; Gutman, J.A.; Pollyea, D.A.; et al. Outcomes for adults with KMTA-r acute myeloid leukemia treated with venetoclax plus azacitidine (Ven/Aza) or intensive chemotherapy (IC): A single-center retrospective study. J Clin Oncol 2025, 43, e18537. [Google Scholar] [CrossRef]
- Khaire, N.; Bouligny, I.; Issa, G.; Daver, N.; DiNardo, C.D.; Jabbour, E.; Short, N.; Jen, W.Y.; Tang, G.; Loghavi, S.; et al. Oucomes in patients with newly diagnosed acute myeloid leukemia with KMT2A rearrangement treated with cladribine, idarubicin, cytarabine (CLIA) with or without venetoclax. Blood 2025, 146, 3459–3460. [Google Scholar] [CrossRef]
- Bataller, A.; Goulart, H.; Issa, G.C.; DiNardo, C.D.; Daver, N.; Kadia, T.; Bazinet, A.; Bouligny, I.M.; Senapati, J.; Haddad, F.G.; et al. Characteristics and outcomes of newly diagnosed acute myeloid leukemia with KMT2A rearrangements. Leukemia 2025, 39, 1640–1649. [Google Scholar] [CrossRef]
- Zeidner, J.F.; Jain, J.; Welkie, R.L.; Huang, Y.; Swords, R.T.; Traer, E.; Stein, E.M.; Lin, T.L.; Madanat, Y.F.; Collins, R.H.; et al. Clinical outcomes of older patients with NPM1-mutated or KMT2A-rearranged AML before menin inhbitors: A Beat AML report. Blood Adv 2026. [Google Scholar] [CrossRef]
- Chen, L.; Zhu, Y.; Zhao, L.; Li, J.; An, R.; Liu, Z.; Xu, X.; Zhang, S. prognostic implications of KMT2A rearrangements and partial tandem duplications in AML: Outcomes and the role of allogeneic hematopoietic cell transplantation. Blood 2024, 144, 4291–4292. [Google Scholar] [CrossRef]
- Alrakali, O.; Zomrokji, Z.; Al Ali, N.; Xie, Z.; Chan, O.; Kuykendall, A.; Yun, S.; Shalis, R.; Reynolds, S.; Walker, A.; et al. Setting the stage in KMT2A rearranged acute myelogenous leukemia in the era of menin inhibitors. Blood 2025, 146, 3393–3394. [Google Scholar] [CrossRef]
- Shen, H.; Chen, J.; Gong, X.; Zhou, C.; Lin, D.; Liu, K.; Gong, B.; Zhang, G.; Li, Y.; Liu, Y.; et al. Impact of fusion partners and transplantation benefit in intensively treated KMT2A-rearranged acute myeloid leukemia. Cancers 2026, 18, 401. [Google Scholar] [CrossRef]
- Liu, J.; Zhao, X.S.; Chang, Y.J.; Qin, Y.Z.; Jiang, Q.; Jiang, H.; Zhang, Y.H.; Xu, L.P.; Wang, Y.; Lv, M.; et al. Monitoring the KMT2A gene post-chemotherapy independently predicts the relapse and survival risk after allogeneic hematopoietic stem cell transplantation. Br J Haematol 2025, 206, 1418–1429. [Google Scholar] [CrossRef] [PubMed]
- Kannan, N.; Achrekar, A.; Terse, V.; Gawde, V.; Joshi, S.; Bhanshe, P.; Chaudhary, S.; Salunke, P.; Ghogale, S.; Deshpande, N.; et al. Acute myeloid leukemia (AML) harbouring KMT2A-PTD should be considered as myelodysplasia-related abnormality?
- Kunadt, D.; Zukunft, S.; Metzeler, K.H.; Rollig, C.; Schliemann, C.; Braess, J.; Muller-Tidow, C.; Kraemer, A.; Scholl, S.; Hilgendorf, I.; et al. Impact of KMT2A-PTD mutational subgroups on outcome of AML patients after therapy and allogeneic hematopoietic stem cell transplantation. Blood 2023, 142, 2236–2238. [Google Scholar] [CrossRef]
- Abdelmessieh, P.; Shah, B.; Shestovska, Y.; Stack, A.; Khanak, R.; Styler, M.; Slamm, S.; Stapor, D.C.; Vartanov, A.; Yanovsky, A.; et al. Efficacy of allogeneic stem cell transplant in KMT2A partial tandem duplication (KMT”A-PTD) myeloid neoplasms. Transplant Cell Therapy 2026, 32 suppl) S110. [Google Scholar]
- Candoni, A.; Coppola, G. A 2024 update on menin inhibitors. A new class of target agents against KMT2A-rearranged and NPM1-mutatted acute myeloid leukemia. Hematol Rep 2024, 16, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Issa, G.C.; Aldoss, I.; Thirman, M.J.; DiPersio, J.; Arellano, M.; Blachy, J.S.; Mannis, G.N.; Perl, A.; Dickens, D.S.; McMahon, C.; et al. Menin inhibition with revumenib for KMT2A-rearranged relapsed or refractory acute leukemia (AUGMENT-101). J Clin Oncol 2025, 43, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Aldoss, I.; Issa, G.C.; Blachy, J.S.; Thirman, M.; Mannis, G.; Arellano, M.; DiPersio, J.; Traer, E.; Zwaan, C.; Shukla, N.; et al. Revumenib for patients with relapsed or refractory (R/R) KMT2Ar acute leukemia: Outcomes by leukemia type in the phase 2 AUGMENT-101 study. Blood 2025, 146, 1001–1002. [Google Scholar] [CrossRef]
- Loghavi, S.; Farhat, A.; Jamison, T.J.; El Hajjar, G.; Bataller, A.; Wang, S.A.; Tang, G.; Quesada, A.E.; Bazinet, A.; Cuglievan, B.; et al. Immunophenotypic changes following menin inhibition in acute myeloid leukemia. Leukemia 2026. [Google Scholar] [CrossRef] [PubMed]
- Perner, F.; Stein, E.M.; Wenge, D.V.; Singh, S.; Kim, J.; Apazidis, A.; Rahnamoun, H.; Anand, D.; Marinaccio, C.; Hatton, C.; et al. MEN1 mutations mediate clinical resistance to menin inhibition. Nature 2023, 615, 913–919. [Google Scholar] [CrossRef]
- Bourgeois, W.; Cutler, J.; Rice, H.; Regalado, B.; Wenge, D.; Perner, F.; Doench, J.; McGeehan, J.; Armstrong, S. Discerning the landscape of menin inhibitor resistance. Blood 2024, 144, 724–725. [Google Scholar] [CrossRef]
- D’Souza, J.; Leung, C.J.; Ballapuram, A.C.; Lin, A.S.; Batingana, A.R.; Lamble, A.J.; Ries, R.E.; Morales, C.E.; Cottonham, C.; Gona, P.; et al. TP53 Inactivation Confers Resistance to the Menin Inhibitor Revumenib in Acute Myeloid Leukemia. Blood 2025, 146, 3470. [Google Scholar] [CrossRef]
- Soto-Feliciano, Y.; Sanchez-Rivera, F.; Perner, F.; Barrows, D.; Kastenhuber, E.; Ho, Y.J.; Carroll, T.; Xiong, Y.; Anand, D.; Soshnev, A.; et al. A molecular switch between mammalian MLL complexes dictates response in menin-MLL inhibition. Cancer Discov 202313, 146–169. [CrossRef]
- Perner, F.; Rahnamoun, H.; Wenge, D.V.; Xiong, Y.; Apazidis, A.; Anand, D.; Hatton, C.; Wen, Y.; Gu, S.; Liu, X.S.; et al. S125: Non-Genetic Resistance to Menin Inhibition in AML Is Reversible by Perturbation of KAT6A. Hemasphere 2023, 7, e6233123. [Google Scholar] [CrossRef]
- Gordon, S.; Perner, F.; MacPherson, L.; Fennell, K.; Wenge, D.; Bourgeois, W.; Klaus, T.; Plenge, T.; Murat, A.; Petrovic, J.; et al. Catalytic inhibition of KAT6/KAT7 enhances the efficacy and overcomes primary and acquired resistance to menin inhibitors in MLL leukemia. Cancer Discov 2025, 15, 2117–2138. [Google Scholar] [CrossRef]
- Daver, N.; Zeidner, J.; Watts, J.; Yuda, J.; Levis, M.; Montesinos, P.; Papayannidis, C.; Fukushima, K.; Shima, T.; Raffoux, E.; et al. Monotherapy update from Phase 1 portion in Phase1/2 trial of the menin-MLL inhibitor enzomenib (DSP-5336) in patients with relapsed or refractory acute leukemia. Blood 2025, 146, 763. [Google Scholar] [CrossRef]
- Wang, E.S.; Issa, G.C.; Erba, H.P.; Altman, J.K.; Montesinos, P.; DeBotton, S.; Walter, R.B.; Pettit, K.; Savona, M.R.; Shah, M.V.; et al. Ziftomenib in relapsed or refractory acute myeloid leukaemia (KOMET-001): A multicentre, open-label, multi-cohort, phase 1 trial. Lancet Oncol 2024, 25, 1310–1324. [Google Scholar] [CrossRef]
- Jabbour, E.; Searle, E.; Abdul-Hay, M.; Abedin, S.; Aldoss, I.; Alfonso Piérola, A.; Alonso-Dominguez, J.M.; Chevallier, P.; Cost, C.; Daskalakis, N.; et al. A First-in-Human Phase 1 Study of the Menin-KMT2A (MLL1) Inhibitor JNJ-75276617 in Adult Patients with Relapsed/Refractory Acute Leukemia Harboring KMT2A or NPM1 Alterations. Blood 2023, 142, 57. [Google Scholar] [CrossRef]
- Searle, E.; Recher, C.; Abdul-Hay, M.; Abedin, S.; Aldoss, I.; Pierole, A.; Aolnso-Dominguiez, J.; Chevallier, P.; Cost, C.; Daskalakis, N.; et al. Bleximenib dose optimization and determination of RP2D from a phase 1 stud in relapsed/refractor acute leukemia patients with KMt2A and NPM1 alterations. Blood 2024, 144 (Suppl. S1), 212. [Google Scholar] [CrossRef]
- Shukla, N.; Guest, E.; Tasian, S.K.; Breese, E.; Schafer, J.; DiPersio, G.; Issa, G.C.; Silverman, B.; Stieglitz, B.; Pollard, J. Safety and activity of revumenib in combination with fludarabine/cytarabine (FLA) in patients with relapsed/refractory acute leukemias. HemaSphere 2024, 8, e104. [Google Scholar]
- Aldoss, I.; Shultz, C.; Hunter, B.; Swoboda, D.; Montesinos, P.; Assouline, S.; Fleming, S.; Grove, C.; Schuh, A.; Taussig, D. Phase 1 study of revumenib in combination with intensive chemotherapy (IC) in patients (Pts) with newly diagnosed (ND) Acute Myeloid Leukemia (AML) harboring genetic alterations in KMT2A, NPM1, or NUP98: SNDX-5613-0708. Blood 2025, 146, 3425. [Google Scholar] [CrossRef]
- Zeidan, A.M.; Wang, E.; Fathi, A.; Roboz, G.; Madanat, Y.; Strickland, S.; Balasubramianan, S.; Mangan, J.; Pratz, K.; Advani, A.; et al. Ziftomenib combined with intensive induction chemotherapy (7+3) in newly diagnosed NPM1-m or KMT2A-r acute myeloid leukemia (AML): Updated phase 1°/b results from KOMET-007. Clin Lymphoma Myeloma Leukemia 2025, 25, S452–S453. [Google Scholar] [CrossRef]
- Döhner, H.; Schuh, A.; Recher, C.; O’Nions, J.; Aldoss, I.; Alfonso-Pierola, A.; Allred, A.; Alonso-Dominguez, J.M.; Barreyro, L.; Bories, P.; et al. Bleximenib in combination with intensive chemotherapy: A phase 1b study in newly diagnosed Acute Myeloid Leukemia with KMT2A or NPM1 alterations. Blood 2025, 146, 5199. [Google Scholar] [CrossRef]
- Raaijmakers, M.; Döhner, H.; Breems, D.; Byrd, J.; Döhner, K.; Esteve, J.; Gjertsen, B.; Gradowska, P.; Huls, G.A.; Kaare, A.; et al. Bleximenib or placebo in combination with standard induction and consolidation therapy followed by maintenance for the treatment of patients with newly diagnosed KMT2A-rearranged or NPM1-mutant Acute Myeloid Leukemia eligible for intensive chemotherapy: A double-blind Phase 3 study (HOVON 181 AML / AMLSG 37-25). Blood 2025, 146, 1654. [Google Scholar]
- Issa, G.C.; Cuglievan, B.; Daver, N.; DiNardo, C.D.; Farhat, A.; Short, N.J.; McCall, D.; Pike, A.; Tan, S.; Kammerer, B.; et al. PhaseI/II Study of the All-Oral Combination of Revumenib (SNDX-5613) with Decitabine/Cedazuridine (ASTX727) and Venetoclax (SAVE) in R/R AML. Blood 2024, 144, 216. [Google Scholar] [CrossRef]
- Jen, W.-Y.; DiNardo, C.; Short, N.; Farhat, A.; El Hajjar, G.; Zhang, B.; Duose, D.; Daver, N.; Kadia, T.; Cuglievan, B.; et al. Phase II study of the all-oral combination of revumenib (SNDX-5613) with decitabine/cedazuridine (ASTX727) and venetoclax (SAVE) in newly diagnosed AML. Blood 2025, 146, 47. [Google Scholar] [CrossRef]
- Zeidner, J.F.; Lin, T.L.; Welkie, R.L.; Curran, E.; Koenig, K.; Stock, W.; Madanat, Y.F.; Swords, R.; Baer, M.R.; Blum, W.; et al. Azacitidine, Venetoclax, and Revumenib for Newly Diagnosed NPM1 -Mutated or KMT2A -Rearranged AML. J. Clin. Oncol. 2025, 43, 2606–2615. [Google Scholar] [CrossRef]
- Issa, G.; Fathi, A.; Zeidan, A.; Erba, H.; Roboz, G.; Altman, J.; Pratz, K.; Juckett, M.; Lin, T.; Balasubramanian, S.K.; et al. Ziftomenib in combination with venetoclax and azacitidine in relapsed/refractory NPM1-m or KMT2A-r acute myeloid leukemia: Updated phase 1a/b safety and clinical activity results from KOMET-007. Blood 2025, 146, 764. [Google Scholar] [CrossRef]
- Roboz, G.; Wang, E.; Fathi, A.; Erba, H.; Pratz, K.; Guru Murthy, G.S.; Alsfeld, L.; Blachly, J.; Naqvi, K.; Issa, G.; et al. Ziftomenib in combination with venetoclax and azacitidine in newly diagnosed NPM1-m acute myeloid leukemia: Phase 1b results from KOMET-007. Blood 2025, 146, 766. [Google Scholar] [CrossRef]
- O’Nions, J.; Wei, A.; Esteve, J.; Aldoss, I.; Alfonso-Pierola, A.; Allred, A.; Alonso-Dominguez, J.M.; Barreyro, L.; Bories, P.; Daskalakis, N.; et al. Phase 1b study of bleximenib in combination with venetoclax in Acute Myeloid Leukemia with KMT2A or NPM1 alterations. Blood 2025, 146, 5200. [Google Scholar] [CrossRef]
- Wei, A.H.; Reyner, J.E.; Garciaz, S.; Aldoss, I.; Piérola, A.A.; Allred, A.; Alonso Dominguez, J.M.; Barreyro, L.; Bories, P.; Daskalakis, N.; et al. Recommended phase 2 dose (RP2D) determination of bleximenib in combination with venetoclax (VEN+AZA): Phase Ib study in newly diagnosed (ND) and relapsed/refractory (R/R) acute myeloid leukemia (AML) with KMT2A or NPM1 alterations. Clin Lymphoma Myeloma Leukemia 2025(25), S423–S424. [CrossRef]
- Watts, J.; Borate, U.; Levis, M; Abaza, Y.; Wang, E.; Zeidner, J.; Cai, H.; Xu, B.; Robson, P.; Watanabe, A.; et al. Preliminary data from the ongoing phase 1 study of the menin-MLL inhibitor enzomenib (DSP-5336) in combination with venetoclax and azacitidine in patients with relapsed or refractory acute myeloid leukemia. Blood 2025, 146, 765. [Google Scholar] [CrossRef]
- Borate, U.; Jain, J.; Huang, Y.; Dvorak-Kornaus, K.; Perl, A.; Zeidner, J.; Mims, A. Preliminary results of a phase 1 study of the safety and tolerability of the combination of revumenib (REV) with gilteritinib (GILT) in relapsed/refractory (R/R) acute myeloid leukemia. Blood 2025, 146, 3427–3428. [Google Scholar] [CrossRef]
- Zucenka, A.; Issa, G.C.; Arellano, M.; Khazal, S.; Khera, N.; Stock, W.; Cuglievan, B.; Gu, Y.; Nguyen, H.V.; Smith, A.R.; et al. Revumenib maintenance therapy following revumenib-induced remission and transplant. Blood 2023, 142, 4950–4952. [Google Scholar] [CrossRef]
- Cuglievan, B.; Okeleji, O.; Catueno, S.; McCall, D.; Garcia, M.; Gibson, A.; Nunez, C.; Roth, M.; Farhat, A.; El Hajjar, G.; et al. Post-transplant maintenance with revumenib in children in children with HOX-driven AML. Blood 2025, 146, 3461–3462. [Google Scholar] [CrossRef]
- Okoniewski, M.; Lugo, J.G.; Lin, T.L. Moving away from standard induction in newly diagnosed acute myeloid leukemia. Am Soc Clin Oncol Educ Book 2025, 45, e473646. [Google Scholar] [CrossRef] [PubMed]
- Author 1, A.B.; Author 2, C. Title of Unpublished Work. Abbreviated Journal Name year, phrase indicating stage of publication. submitted; accepted. in press. [Google Scholar]
- Author 1, A.B.; Author 2, C. Personal communication, 2012.
- Author 1, A.B.; Author 2, C.D.; Author 3, E.F. Title of Presentation. In Proceedings of the Name of the Conference, Location of Conference, Country, Date of Conference (Day Month Year).
- Author 1, A.B. Title of Thesis. Level of Thesis, Degree-Granting University, Location of University, Date of Completion.
- Title of Site. Available online: URL (accessed on Day Month Year).
| Trial Name NCT Identifier Phase |
Patient Number and Disease Status |
Therapeutic Regimen | Efficacy | Toxicity |
|---|---|---|---|---|
| AUGMENT-101 NCT04065399 Phase I/II |
Adult and pediatric patients with R/R NPM1m (116) and KMT2A-r (84) AML | Revumenib (single arm) | KMT2A-r ORR 63.2% CRc 22.8% DOR 4.3 mo OS 8.0 mo NPM1m ORR 46.9% CRc 23.4% DOR 4.4 mo OS 4.0 mo |
KMT2A-r DS 27% QTc prol 29% NPM1m DS 19% QTc prol 43% |
| KOMET-001 NCT04067336 Phase I/II |
Phase Ib: patients with R/R NPM1m or KMT2A-r AML Phase II: patients with R/R NPM1m 200 mg 20 pts 600 mg 130 pts |
Ziftomenib (single arm) | KMT2A-r (18 pts) ORR 17% CRc 11% DOR 2.1 mo NPM1m (112 pts) ORR 33% CRc 25% DOR 3.7 mo OS 6.1 mo |
All patients DS 14% QTc prol NR |
| DSP-5336-101 NCT04988555 Phase I/II |
Phase I: patients with R/R Acute Leukemia Phase II: patients with R/R NPM1m or KMT2A-r |
Enzomenib (single arm) | KMT2A-r (300mg 11 pts) ORR 72.7% CRc 45.5% DOR 4.3 NR OS 11.4 mo NPM1m (200-300 mg 17 pts) ORR 58.8% CRc 47% DOR 5.9-6.7 mo OS 8.5 mo |
All patients (116) DS 12.9% QTc prol 4.3% |
| cAMELot-1 NCT04811560 Phase I/II |
Phase Ib: patients with R/R NPM1m or KMT2A-r or NUP-98-r AML Phase II: patients with R/R NPM1m or R/R KMT2A-r AML |
Bleximenib (single arm) | KMT2A-r (90-100 mg 9 pts) CRc 33.3% NPM1m (90-100 mg 12 pts) CRc 33.3% All patients (146) DOR 6 mo |
All patients DS 19.4% QTc prol NR |
| Trial Name NCT Identifier Phase |
Patient Number and Disease Status |
Therapeutic Regimen | Efficacy | Toxicity |
|---|---|---|---|---|
| SAVE NCT005360160 Phase I |
Adult patients with R/R NPM1m, KMT2A-r, NUP-98-r AML (26) | Revumenib Venetoclax Decitabine/ Cedazuridine |
ORR 88% CRc 43.9% |
DS 4% QTc prol 8% |
| SAVE NCT005360160 Phase II |
Adult patients with ND NPM1m, KMT2A-r, NUP-98-r AML (17) | Revumenib Venetoclax Decitabine/ Cedazuridine |
ORR 94% CR 88% 6-mo EFS 59% Median OS NR |
DS 12% QTc prol 18% |
| BEAT AML Master Trial | Adult patients with ND NPM1m (34 patients) or KMT2A-r (9 patients) | Revumenib Venetoclax Azacitidine |
KMT2A-r (9 pts) ORR 100% CRc 88.9% CR 78% OS 18 mo NPM1m (34 pts) ORR 85.3% CRc 79.4% CR 65% OS 15 mo |
All patients DS 19% QTc prol 44% |
| KOMET-007 NCT05735184 Phase Ia/Ib |
Patients with R/R NPM1m or KMT2A-r | Ziftomenib Azacitidine Venetoclax |
KMT2A-r (29 pts) ORR 33% CRc 22% OS 21 wks NPM1m (51 pts) ORR 65% CRc 49% OS NR |
All patients DS 12% QTc prol 0% |
| ALE1002 NCT05453903 Phase Ib |
Patients with R/R NPM1m (10) or KMT2A-r (3) | Bleximenib Venetoclax |
ORR 69.2% CRc 38.5% |
All patients DS 0/13 QTc prol 1/13 |
| ALE1002 NCT05453903 Phase Ib |
Patients with NPM1m (68) or KMT2A-r (52) R/R (86) or ND (34) |
Bleximenib Venetoclax Azacitidine |
R/R (50/100mg) ORR 76/79% CRc 32/54% ND (50/100ng) ORR 72/92% CRc 62/85% |
All patients DS 4% QTc prol 0% |
| Phase I | Patients with R/R NPM1m or KMT2A-r (18) |
Enzomenib Venetoclax Azacitidine |
ORR 83% CRc 56% |
All patients DS 0% QTc prol 1/18 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




