Submitted:
20 March 2026
Posted:
23 March 2026
You are already at the latest version
Abstract
Keywords:
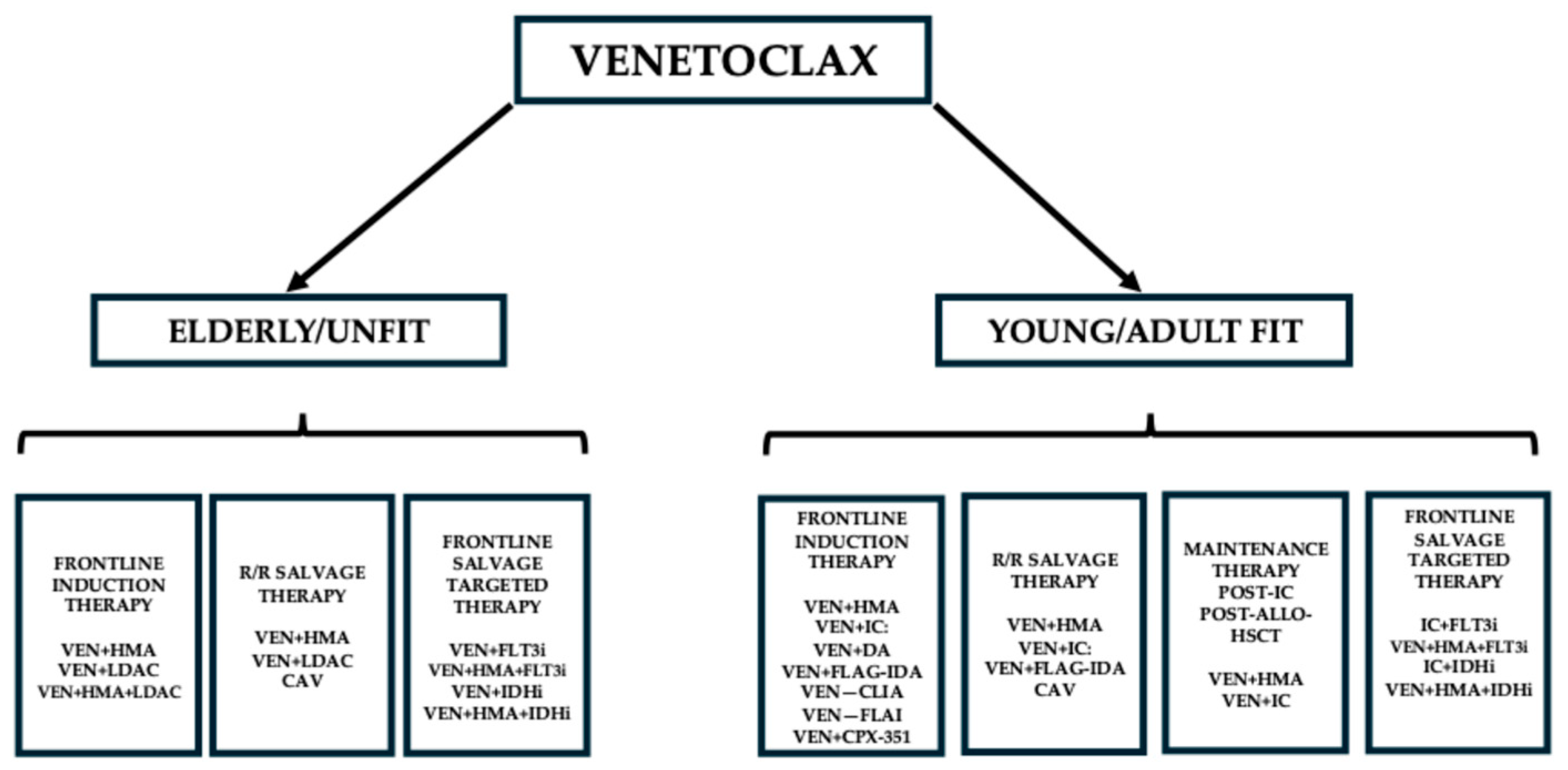
1. Introduction
2. Effects of Venetoclax on Newly Diagnosed and Relapsed/Refractory AML Patients Alone or in Association with a Hypomethylating Agent
2.1. Responses in AML Molecular Subtypes
2.2. Responses in a Real-World Setting
2.3. Genetic Risk Stratification in AML Patients Treated with Venetoclax and Hypomethylating Agents
2.4. The Sensitivity of Leukemic Blasts to VEN+HMAs Is Correlated with Cell Differentiation
2.5. Ventoclax and Hypomethylating Agents in Relapsed/Refractory AML Patients
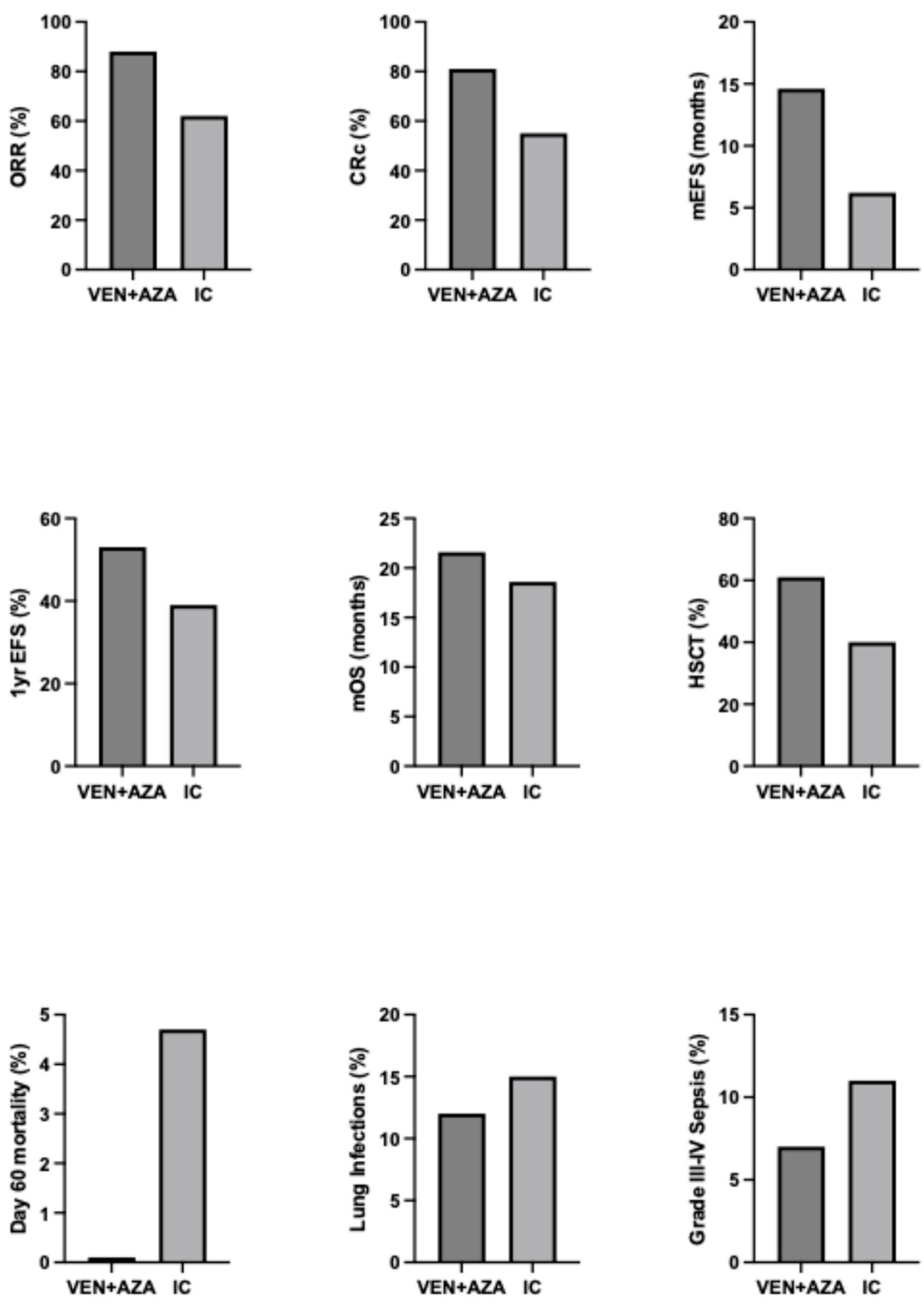
2.6. Venetoclax Plus HMAs in Adult AML Patients Versus Induction Chemotherapy
2.7. Venetoclax and Low-Dose Citarabine (VEN+LDAC)
2.8. Venetoclax in Association with Intensive Chemotherapy
2.10. Venetoclax-Based Treatments as Maintenance Therapy in AML
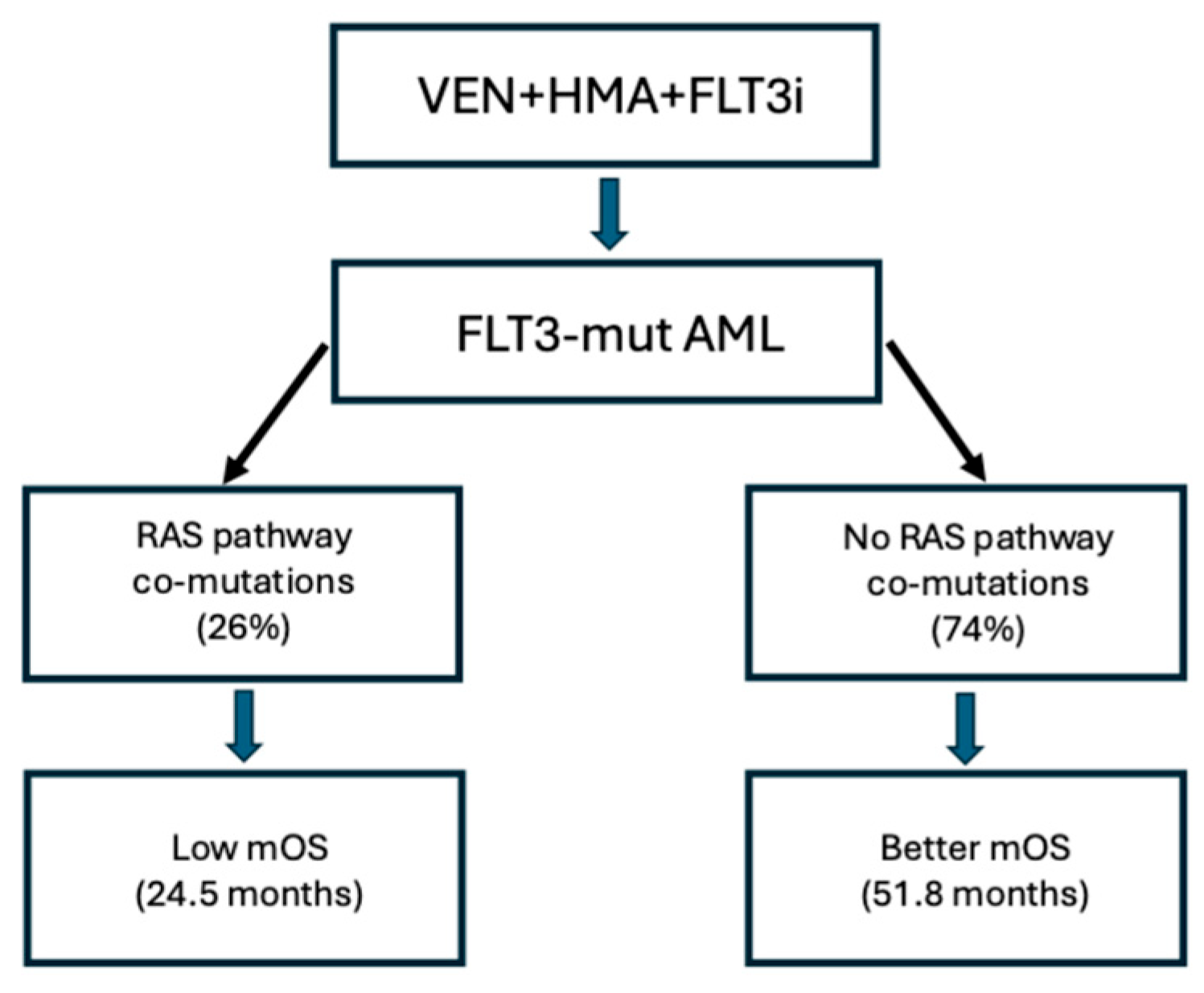
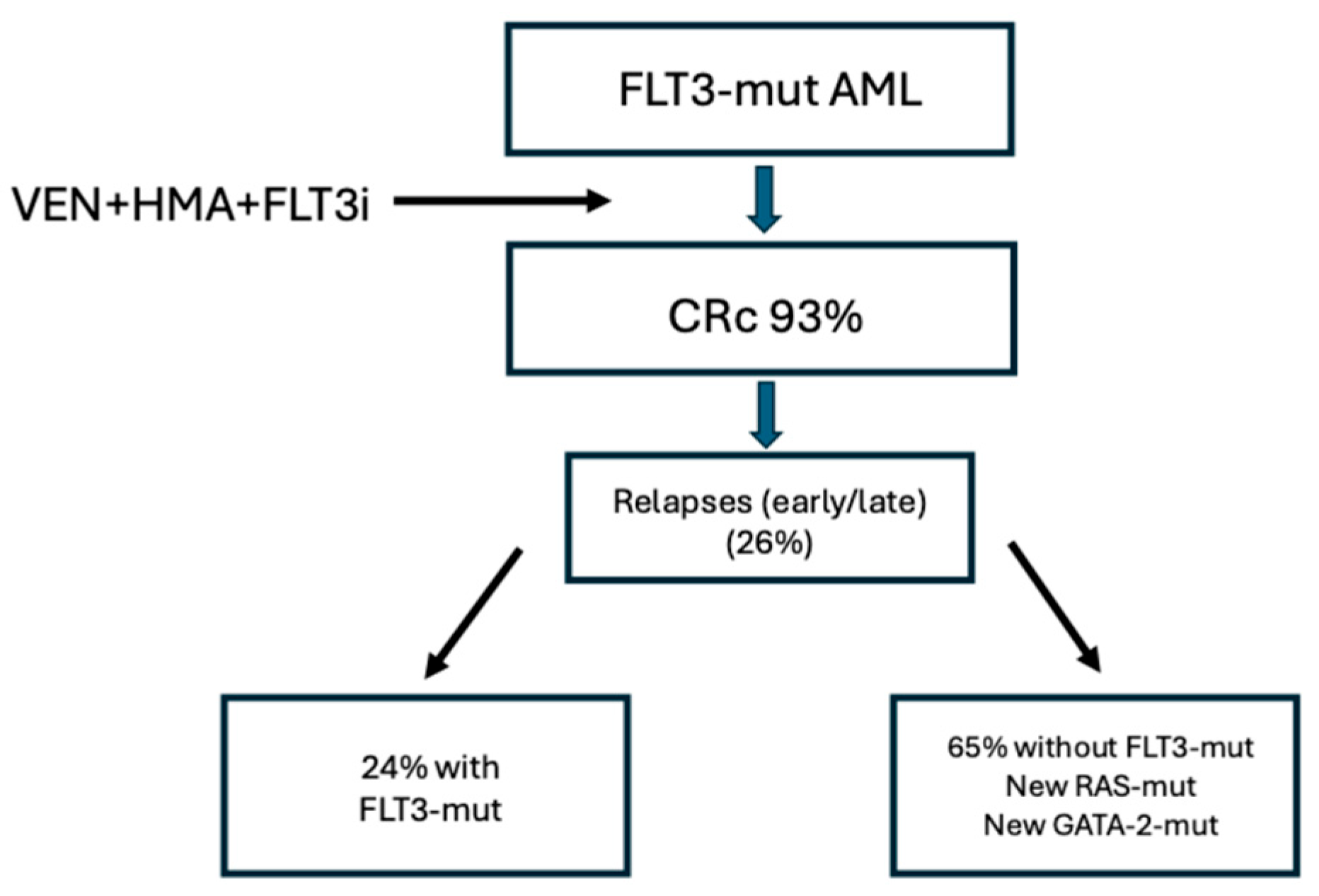
2.11. Treatment of FLT3-Mutant AMLs with Combinations of Venetoclax and FLT3 Inhibitors
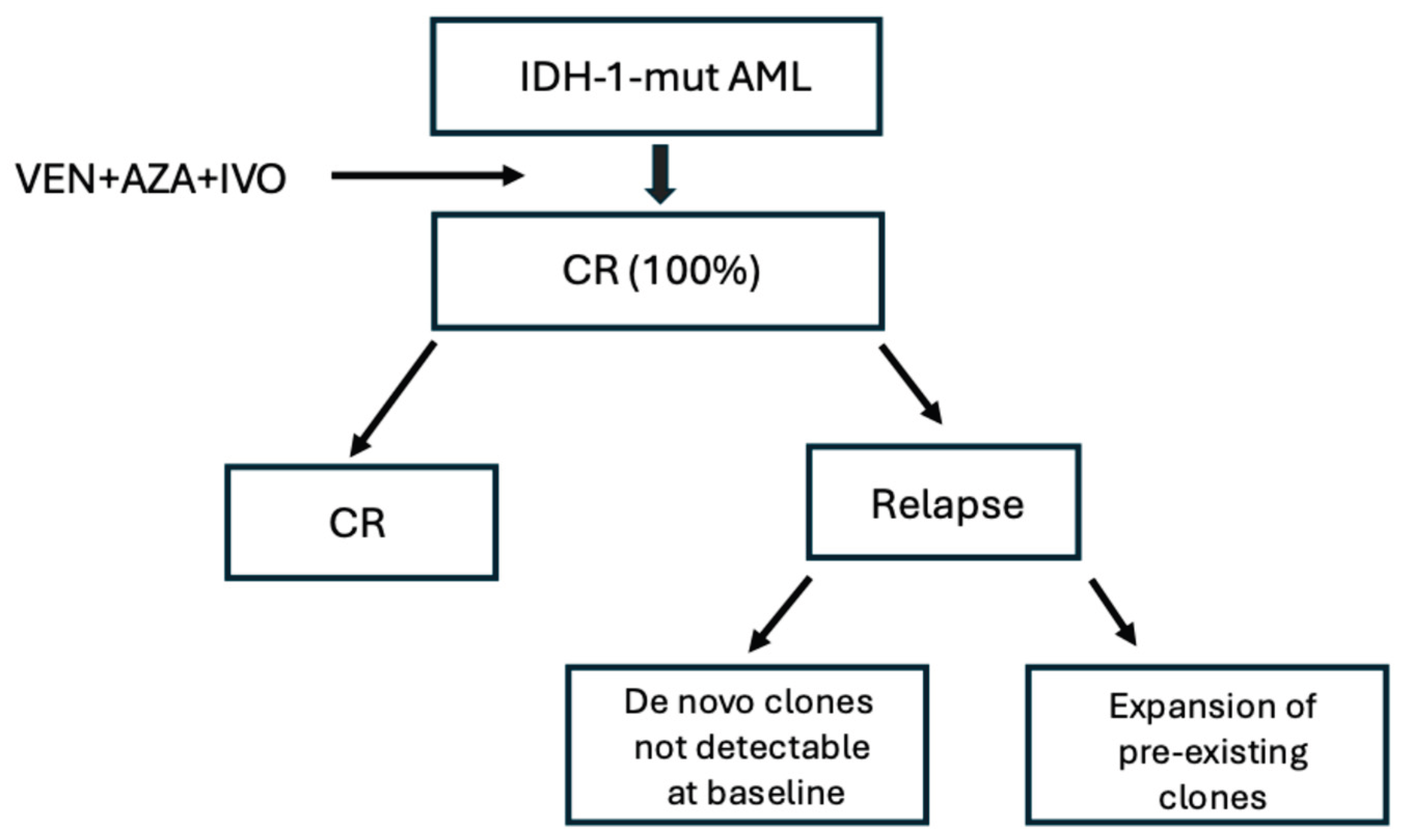
2.12. Treatment of IDH-Mutant AML with Venetoclax-Based Regimens and IDH Inhibitors
2.13. Targeting CD123 with Anti-CD123 Agents in Combination with Venetoclax
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goulart, H.; Kantarjian, H.; Pemmaraju, N.; Daver, N.; DiNardo, C.D.; Rausch, C.R.; Ravandi, F.; Kadia, T.M. Ventoclax-based combination regimens in acute myeloid leukemia. Blood Cancer Discovery 2025, 6, 23–37. [Google Scholar] [CrossRef] [PubMed]
- Wei, A.H.; Loo, S.; Daver, N. How I treat patients with AML using azacitidine and venetoclax. Blood 2025, 145, 1237–1250. [Google Scholar] [CrossRef] [PubMed]
- Dinardo, C.D.; Pratz, K.; Pullarkat, V.; Jonas, B.A.; Arellano, M.; Becker, P.S.; Frankfurt, O.; Konopleva, M.; Wei, A.H.; Kantarjian, H.M.; et al. Venetoclax combined with decitabine or azacitidine in treatment-naive, elderly patients with acute myeloid leukemia. Blood 2019, 133, 7–17. [Google Scholar] [CrossRef] [PubMed]
- DiNardo, C.D.; Jonas, B.A.; Pullarkat, V.; Thirman, M.J.; Garcia, J.S.; Wei, A.H.; Konopleva, M.; Döhner, H.; Letai, A.; Fenaux, P.; et al. Azacitidine and Venetoclax in Previously Untreated Acute Myeloid Leukemia. N Engl J Med 2020, 383, 617–629. [Google Scholar] [CrossRef]
- Pratz, K.W.; Jonas, B.A.; Pullarkat, V.; Thirman, M.J.; Garcia, J.S.; Dohner, H.; Récher, C.; Fiedler, W.; Yamamoto, K.; Wang, J.; et al. Long-term follow-up of VIALE-A: venetoclax and azacitidine in chemotherapy-ineligible untreated acute myeloid leukemia. Am J Hematol 2024, 99, 615–624. [Google Scholar] [CrossRef]
- Pratz, K.W.; DiNardo, C.D.; Arellano, M.; Thirman, M.J.; Pullarkat, V.; Becker, P.S.; Smith, B.D.; Zhang, M.; Miu, K.; Potluri, J.; et al. Long-term outcomes of stem cell transplant in older patients with acute myeloid leukemia treated with venetoclax-based therapies. Am J Hematol 2026, 0, 1–4. [Google Scholar] [CrossRef]
- Pullarkat, V.; Pratz, K.W.; Dohner, H.; Recher, C.; Thirman, M.J.; DiNardo, C.D.; Fenaux, P.; Schuh, A.; Wei, A.H.; Pigneux, A.; et al. Venetoclax and azacitidine in untreated patients with therapy-related acute myeloid leukemia, antecedent myelodysplastic syndromes in chronic myelomonocytic leukemia. Blood Cancer J 2025, 15, 49. [Google Scholar] [CrossRef]
- Venditti, A.; Hou, J.Z.; Fenaux, P.; Jonas, B.A.; Vhrovac, R.; Montesinos, P.; Garcia, J.S.; Rizzieri, D.; Thirman, M.J.; Zhang, M.; et al. Outcomes of patients treated with venetoclax plus azacitidine alone stratified by advanced age and acute myeloid leukemia composite model. Leukemia 2026, in press. [Google Scholar] [CrossRef]
- Efficace, F.; Kicinski, M.; Coens, C.; Suciu, S.; van der Velden, W.; Noppeney, R.; Chantepie, S.; Griskevicius, L.; Neubauer, A.; Audisio, E.; et al. Decitabine il older patients with AML: quality of life results of the EORTC-GIMEMA-GMDS-SG randomized phase 3 trial. Blood 2024, 144, 541–548. [Google Scholar] [CrossRef]
- Tarantini, F.; Contento, C.; Vigna, E.; Carluccio, V.; Greco, G.; Pisciolla, C.; Ciuffreda, L.; Rossi, G.; Urbano, M.A.; D’Ambrosio, A.; et al. Hypomethylating agents alone or in combination with venetoclax in very elderly acute myeloid leukemia patients: less treatment, better care? Ann Hematol 2026, 105, 1. [Google Scholar] [CrossRef]
- Konopleva, M.; Thirman, M.J.; Pratz, K.W.; Garcia, J.S.; Recher, C.; Pullarkat, V.; Kantarjian, H.M.; DiNardo, C.D.; Dail, M.; Duan, Y.; et al. Impact of FLT3 Mutation on Outcomes after Venetoclax and Azacitidine for Patients with Treatment-Naïve Acute Myeloid Leukemia. Clin Cancer Res 2022, 28, 2744–2752. [Google Scholar] [CrossRef]
- Weng, G.; Huang, J.; An, N.; Zhang, Y.; Yu, G.; Sun, Z.; Lin, D.; Deng, L.; Liang, X.; Xiao, J.; et al. Clinical and genetic characteristics predict outcomes of acute myeloid leukemia patients with FLT3 mutations receiving venetoclax-based therapy. Cancer Medicine 2024, 13, e6885. [Google Scholar] [CrossRef]
- Ivey, A.; Hills, R.K.; Simpson, M. A, Assessment of minimal residual disease in standard-risk AML. N Engl J Med 2016, 374, 422–433. [Google Scholar] [CrossRef]
- Othman, J.; Tiong, I.S.; O’Nions, J.; Dennis, M.; Mokretar, K.; Ivey, A.; Austin, M.J.; Latif, A.-L.; Amer, M.; Chan, W.Y.; et al. Molecular MRD is strongly prognostic in patients with NPM1-mutated AML receiving venetoclax-based nonintensive therapy. Blood 2024, 143, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Chua, C.C.; Hammond, D.; Kent, A.; Tiong, I.S.; Konopleva, M.Y.; Pollyea, D.A.; DiNardo, C.D.; Wei, A.H. Treatment-free remission after ceasing venetoclax-based therapy in patients with acute myeloid leukemia. Blood Adv 2022, 6, 3879–3883. [Google Scholar] [CrossRef]
- Garciaz, S.; Bertoli, S.; A Sallman, D.; Decroocq, J.; Dumas, P.-Y.; Belhabri, A.; Orvain, C.; Requena, G.A.; Simand, C.; Laribi, K.; et al. Acute Myeloid Leukemia Patients Who Stopped Venetoclax or/and Azacytidine for Other Reasons Than Progression Have a Prolonged Treatment Free Remission and Overall Survival. a Filo Study. Blood 2023, 142, 161–163. [Google Scholar] [CrossRef]
- Molica, M.; Simio, C.; De Fazio, L.; Alati, C.; Martino, M.; Rossi, M. Harnessing venetoclax in NPM1-mutatted AML: a path to sustained remission and beyond. Cancers 2025, 17, 3733. [Google Scholar] [CrossRef]
- Hoff, F.; Li, A.; Huang, Y.; Wellde, R.; Swords, R.; Traer, E.; Stein, E.; Lin, T.; Baer, M.; Duong, V.; et al. RAS mutations negate the favorable impact of NPM1 in older patients with newly diagnosed acute myeloid leukemia treated with Ven/HMA. Blood 2025 146, 995. [CrossRef]
- Kim, K.; Maiti, A.; Kadia, T.; Ravandi, F.; Daver, N.; Pemmaraju, N.; Borthakur, G.; Bose, P.; Issa, G.C.; Short, N.J.; et al. Outcome s of TP53-mutant acute myeloid leuekmia with venetoclax and decitabine. Blood 2020, 136, 33–36. [Google Scholar] [CrossRef]
- Kim, K.; Maiti, A.; Loghavi, S.; Pourebrahim, R.; Kadia, T.M.; Rausch, C.; Furudate, K.; Daver, N.; Alvarado, Y.; Ohanina, M; et al. Outcomes of tP53-mutant acute myeloid leukemia with decitabine and venetoclax. Cancer 2020, 135, 791–803. [Google Scholar]
- DiNardo, C.D.; Tiong, I.S.; Quaglieri, A.; MacRaild, S.; Loghavi, S.; Brown, F.C.; Thijssen, R.; Pomilio, G.; Ivey, A.; Salmon, J.M.; et al. Molecular patterns of response and treatment failure after frontline venetoclax combinations in ilder patients with AML. Blood 2020, 135, 791–803. [Google Scholar] [CrossRef] [PubMed]
- Pollyea, D.A.; Pratz, K.W.; Wie, A.H.; Pullarkat, V.; Jonas, B.A.; Recher, C.; Babu, S.; Schuh, A.C.; Dail, M.; Sun, Y.; et al. Outcomes in patients with poor-risk cytogenetics with or without TP53 mutations treated with venetoclax and azacitidine. Clin Cancer Res 2022, 28, 5272–5279. [Google Scholar] [CrossRef] [PubMed]
- Badar, T.; Nanaa, A.; Atallah, E.; Shallis, R.; Guillherme, S.; Goldberg, A.D.; Saliba, A.N.; Patel, A.; Bewersdorf, J.; Duvall, A.; et al. Comparing venetoclax in combination with hypomethylating agent-based therapies for treatment naïve TP53-mutated acute myeloid leukemia: results from the Consortium on myeloid malignancies and neoplastic diseases (COMMAND). Blood Cancer J 2024, 14, 32. [Google Scholar] [CrossRef]
- Mueller, D.; Santhosh, S.; Singhal, A.; Chitty, D.W.; Gladstone, D.; Goldberg, B.; Jou, E.; Kolitz, J.; Boisclair, S. TP53-mutated AML and treatment outcomes in the era of hypomethylating agent/venetoclax therapy. Blood 2025, 146, 1705. [Google Scholar] [CrossRef]
- Goldfinger, M.; Mantzaris, I.; Shastri, A.; Saunthararajah, Y.; Sica, R.A.; Klombum, N.; Shah, N.; Levitz, D.; Rockwell, B.; Shapiro, L.C.; et al. A weekly low-dose regimen of decitabine and venetoclax is efficacious and less myelotoxic in a racially diverse cohort. Blood 2024, 144, 2360–2363. [Google Scholar] [CrossRef]
- Rockwell, B.; Mantzaris, I.; Shastri, A.; Jonas, B.A.; Saunthararajah, Y.; Levitz, D.; Verceles, J.; Cooper, D.; Munioz, A.; Dhaeman, A.; Pradhan, K.; et al. Dosing decitabine and venetoclax for terminal differentiation to improve outcomes in TP53 mutant MDS and AML. Blood 2025, 146, 6514. [Google Scholar] [CrossRef]
- Yang, X.; Li, C.; Zheng, H.; Huang, X. The venetoclax combined with metronomic low-dose decitabine and interferon for preventing post-transplant relapse in high-risk MDS and AML patients. Blood 2025, 146, 7793–7794. [Google Scholar] [CrossRef]
- Vanttinen, I.; Ruokoranta, T.; Saad, J.; Kytola, S.; Hellesoy, M.; Gullaksen, S.E.; Ettala, P.S.; Pyorala, M.; Rimpilainen, J.; Siitonen, T.; et al. Targeting venetoclax resistance in TP53-mutated acute myeloid leukemia. Blood 2023, 142, 2927–2928. [Google Scholar] [CrossRef]
- Pollyea, D.A.; DiNardo, C.D.; Arellano, M.L.; Pigneux, A.; Fiedler, W.; Konopleva, M.; Rizzieri, D.A.; Smith, D.; Shinagawa, A.; Lemoli, R.M.; et al. Impact of venetoclax and azacitidine in treatment-naïve patients with acute myeloid leukemia and iDH1/2 mutations. Clin Cancer Res 2022, 28, 2753–2761. [Google Scholar] [CrossRef]
- Hammond, D.; Loghavi, S.; Wang, S.; Konopleva, M.; Kadia, T.; Daver, N.; Ohanian, M.; Issa, G.; Alvarado, Y.; Short, N.J.; et al. Response patterns and impact of MRD in patients with IDH1/2-mutated AML treated with venetoclax and hypomethylating agents. Blood Cancer J 2023, 13, 148. [Google Scholar] [CrossRef]
- Wolach, O.; Levi, I.; Nacmias, B.; Tavor, S.; Amitai, I.; Ofran, Y.; Ganzel, C.; Zuckerman, T.; Okasha, D.; Helmann, I.; et al. Trial eligibility, treatment patterns, and outcome for venetoclax-based therapy in AML: a prospective cohort study. Blood Adv 2025, 9, 1544–1554. [Google Scholar] [CrossRef]
- Othman, J.; Lam, H.P.; Leong, S.; Basheer, F.; Abdallah, I.; Fleming, K.; Mehta, P.; Yassin, H.; Laurie, J.; Austin, M.; et al. Real-world outcomes of newly diagnosed AML treated with venetoclax and azacitidine or low-dose cytarabine in the UK NHS. Blood Adv 2024, 1, 100017. [Google Scholar] [CrossRef]
- Brandwein, J.; Page, D.; Liew, E.; Hnatiuk, M.; Bolster, L.; Hamilton, M.; Sawler, D.; Wang, P. A real-world evaluation of frontline treatment for acute myeloid leukemia with azacitidine plus venetoclax. Clin Lymphoma Myeloma Leukemia 2025, 7, e435–e442. [Google Scholar] [CrossRef]
- Solana-Altabella, A.; Rodrigiuez-Veiga, R.; Martinez-Cuadròn, D.; Montesinos, P. A systematic review of venetoclax for the treatment of unfit AML patients in real-world: is all that glitters gold? Ann Hematol 2025, 104, 913–935. [Google Scholar] [CrossRef] [PubMed]
- Dohner, H; Pratz, KW; DiNardo, CD; Jonas, BA; Lullarkat, VA; Thirman, MJ; Recher, C; Schuh, AC; Babu, S; Dail, M; et al. ELN risk is not predictive of outcomes for treatment-naïve patients with acute myeloid leukemia treated with venetoclax and azacitidine. Blood 2022, 140, 1441–1444. [Google Scholar] [CrossRef]
- Dohner, H; Pratz, KW; DiNardo, CD; Wei, AH; Jonas, BA; Pullkart, VA; Thiman, MJ; Recher, C; Schuh, A; Babu, S. Genetic risk stratification and outcomes among treatment-naïve patients with AML treated with venetoclax and azacitidine. Blood 2024, 144, 2211–2222. [Google Scholar] [CrossRef]
- Bataller, A; Bazinet, A; DiNardo, CD; Maiti, A; Borthakur, G; Daver, NG; Short, NJ; Jabbour, EJ; Issa, GC; Pemmaraju, N; et al. Prognostic risk signature in patients with acute myeloid leukemia treated with hypomethylating agents and venetoclax. Blood Adv 2024, 8, 927–936. [Google Scholar] [CrossRef]
- Dohner, H; DiNardo, CD; Appelbaum, F; Craddock, C; Dombret, H; Ebert, B; Fenaux, P; Godley, LA; Hanerjian, RP; Larson, RA; et al. Genetic risk classification for adults with AML receiving less-intensive therapies: the 2024 ELN recommendations. Blood 2024, 144, 2169–2173. [Google Scholar] [CrossRef] [PubMed]
- Ciolli, G.; Piccinni, M.; Mannelli, F.; Gianfaldoni, G.; Scappini, B.; Fasano, L.; Crupi, F.; Quinti, E.; Pasquini, A.; Caroprese, J.; et al. Prognostic stratification in venetoclax-based acute myeloid leukemia treatments: the molecular prognostic signature tested in a real-world setting. Haematologica 2025, 110, 239–243. [Google Scholar] [CrossRef]
- Hoff, FW; Blum, WG; Huang, Y; Walkie, R; Swords, R; Fraer, E; Stein, E; Lin, T; Archer, KJ; Patel, P; et al. Beat-AML 2024 ELN-refined risk stratification for adults with newly diagnosed AML given lower-intensity therapy. Blood Adv 2024, 8, 5297–5303. [Google Scholar] [CrossRef] [PubMed]
- Lachowiez, C.A.; Ravikumar, V.I.; Othman, J.; O’Nions, J.; Peters, D.T.; McMahon, C.; Swords, R.; Cook, R.; Saultz, J.N.; Tyner, J.W.; et al. Refined ELN 2024 risk stratification improves survival prognostication following venetoclax-based therapy in AML. Blood 2024, 144, 2788–2792. [Google Scholar] [CrossRef]
- Hoff, F.W.; Yocum, A.O.; Borate, U.; Mims, A.S.; Byrd, J.C.; Madanat, Y.F. Beat AML genetic risk stratification model in an older VEN/HMA-treated AML patient cohort. Blood Neoplasia 2025, in press. [Google Scholar] [CrossRef] [PubMed]
- Gangat, N.; Karrar, O.; Iftikkar, M.; Macullough, K.; Johnson, M.; Abdelmagid, M.; Abdallah, M.; Al-Kali, A.; Alkateeb, H.B.; Begna, K.H.; et al. Venetoclax and hypomethylating agent combination therapy in newly diagnosed acute myeloid leukemia: genotype signatures for response and survival among 301 consecutive patients. Am J Hematol 2024, 99, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Gangat, N.; Elbeih, A.; Ghosoun, N.; McCullough, K.; Aperne, F.; Johnson, I.M.; Abdelmagid, M.; Al-Kali, A.; Alkhateeb, H.B.; Begna, K.H.; et al. Mayo genetic risk models for newly diagnosed acute myeloid leukemia treated with venetoclax + hypomethylating agent. Am J Hematol 2025, 100, 260–271. [Google Scholar] [CrossRef]
- Lachowiez, C.; Kaempf, A.; Ohtman, J.; Yun, S.; Heiblig, M.; Heuser, M.; Shahswar, R.; Patel, A.; Madero-Marroquin, R.; Cannova, J.; et al. Prognostic risk integration for survival modeling (PRISM) in newly diagnosed acute myeloid leukemia treated with venetoclax: a multinational retrospective cohort study. Blood 2025, 146, 453. [Google Scholar] [CrossRef]
- Mothy, R.; Diebold, K.; Mudd, T.; Hardee, M.; Jarodiya, J.; Parks, K.; Bachiashvili, K.; Espinoza-Gutarra, M.; Rangaraju, S.; Vachhani, P.; et al. Refined genetic risk model with complex karyotype predicts outcomes of nearly diagnosed acuite myeloid leukemia treated with decitabine plus venetoclax. Blood 2025, 146, 5272–5273. [Google Scholar]
- Pei, S.; Pollyea, D.A.; Gustafson, A.; Stevens, B.M.; Minhajuddin, M.; Fu, R.; Riemondy, K.A.; Gillen, A.E.; Sheridan, R.M.; Kim, J.; Costello, J.C.; et al. Monocytic subclones confer resistance to venetoclax-based therapy in patients with acute myeloid leukemia. Cancer Discov 2020, 10, 536–551. [Google Scholar] [CrossRef]
- Kuusanmald, H.; Leppa, A.M.; Polonen, P.; Kontro, M.; Dufva, O.; Deb, D.; Yadav, B.; Bruck, O.; Kumar, A.; Everaus, E.; et al. Phenotype-based drug screening reveals association between venetoclax response and differentiation stage in acute myeloid leukemia. Haematologica 2020, 105, 708–720. [Google Scholar] [CrossRef]
- Kuusanmald, H.; Dufva, O.; Vaha-Koskela, M.; Leppa, A.M.; Huuhtanen, J.; Vanttinen, I.; Nygren, P.; Klievink, J.; Bouhlal, J.; Polonen, P.; et al. Erythroid/megakaryocytic differentiation confers BCL-XL dependency and venetoclax resistance in acute myeloid leukemia. Blood 2023, 141, 1610–1616. [Google Scholar] [CrossRef]
- Waclawiczek, A.; Leppa, A.M.; Renders, S.; Stumpf, K.; Reyneri, C.; Betz, B.; Janssen, M.; Sahswar, R.; Donato, E.; Karpova, D.; et al. Combinatorial BCL2 family expression in acute myeloid leukemia stem cells predicts clinical response to azacitidine/venetoclax. Cancer Discov 2023, 13, 1408–1427. [Google Scholar] [CrossRef]
- Zhao, L.P.; Dumas-Rivero, T.; Barette, L.; Aguinaga, L.; Cheffai, A.; Chauvel, C.; Dal Bello, R.; Raffoux, E.; Clappier, E.; Duchmann, M.; et al. Prognostic significance of monocytic-like phenotype in patients with AML treated with venetoclax and azacytidine. Blood Adv 2025, 9, 3556–3565. [Google Scholar] [CrossRef]
- Lachowiez, C.A.; Heiblig, M.; Requena, G.A.; Tavernier- Tardy, E.; Dai, F.; Ashango, A.B.; Peters, D.T.; Fang, J.; Kaempf, A.; Long, N.; et al. Genetic and phenotypic correlates of clinical outcomes with venetoclax in acute myeloid leukemia: the GEN-PHEN-VEN study. Blood Cancer Discover 2025, 6, 437–449. [Google Scholar] [CrossRef]
- Zeng, A.; Bansalk, S.; Jin, L.; Mitchell, A.; Chen, W.C.; Abbas, H.A.; Chan-Seng-Yue, M.; Voisin, V.; van Galen, P.; Tierens, A.; et al. A cellular hierarchy framework for understanding heterogeneity and predicting drug response in acute myeloid leukemia. Nat Med 2022, 28, 1212–1223. [Google Scholar] [CrossRef]
- Tan, K.W.; Pei, S.; Wang, M.; Dai, L.; Zhang, H.; Chen, C.Y.; Huang, Z.Q.; Dai, H.P.; Wang, Y.; Miao, M.; et al. Comparing the efficacy and safety of VAC (Venetoclax, Azacitidine, Chidamide) and VA regimens as induction therapy for newly diagnosed patients with immunophenotypically mature monocytic AML. Blood 2024, 144, 1507–1508. [Google Scholar] [CrossRef]
- Cai, Q.; Xiao, J.; Weng, C.; Chen, H. Efficacy and safety of venetoclax plus azacitidine based regimens in the treatment of relapsed or refractory acute myeloid leukemia: a systematic review and meta-analysis. Ann Hematol 2025, 104, 4931–4948. [Google Scholar] [CrossRef]
- Stahl, M.; Menghrajani, K.; Derkach, A.; Chan, A.; Xiao, W.; Glass, J.L.; King, A.C.; Daniyan, A.; Famulare, C.; Cuello, B.M.; et al. Venetoclax therapy for relapsed and treatment refractory AML: clinical outcomes and molecular predictors. Blood 2020, 136, 47–48. [Google Scholar] [CrossRef]
- Jimenez-Chillon, C.; Ohtman, J.; Taussig, D.; Jimenez-Vicente, C.; Martinez-Toca, A.; Tiong, I.S.; Jain, M.; Aries, J.; Cakmak, S.; Knapper, S.; et al. Ventoclax-based low intensity therapy in molecular failure of NPM1-mutated AML. Blood Adv 2024, 8, 343–352. [Google Scholar] [CrossRef]
- Higué, J.; Orvain, C.; Dumas, P.Y.; Peterlin, P.; Hospital, M.A.; Barrière, S.; Couturier, A.; Carre, M.; Chantzi, A.; Tavernier, E.; et al. Venetoclax and azacitidine for molecular relapse after intensive chemotherapy in NPM1 or CBF AML: a FILO study. Blood Cancer J 2025, 15, 141. [Google Scholar] [CrossRef]
- Unglaub, J.M.; Schlenk, R.F.; Middeke, J.M.; Krause, S.W.; Kraus, S.; Einsele, H.; Kramer, M.; Zukunft, S.; Kauer, J.; Renders, S.; et al. Venetoclax-based salvage therapy as a bridge to transplant is feasible and effective in patients with relapsed/refractory AML. Blood Adv 2025, 9, 375–384. [Google Scholar] [CrossRef]
- Kruger, K.; Wichmann, M.; Gabdoulline, R.; Braitsch, K.; Modermann, F.; Fiedler, W.; Trummer, A.; Krauter, J.; Kaun, S.; Roterman, S.; Vob, A.; et al. Predictors of response and outcome in patients with refractory/relapsed acute myeloid leukemia receiving venetoclax with non-intensive chemotherapy. Hemasphere 2023, 7(S3), 873–874. [Google Scholar] [CrossRef]
- Shahswar, R.; Gabdoulline, R.; Krueger, K.; Wichmann, M.; Gotze, K.S.; Braitsch, K.; Meggendorfer, M.; Schmalbrock, L.; Bullinger, L.; Modelmann, F.; et al. A novel prognostic risk model for patients with refractory/relapsed acute myeloid leukemia receiving venetoclax plus hypomethylating agents. Leukemia 2025, 39, 614–622. [Google Scholar] [CrossRef]
- Cao, H.Y.; Liu, F.; Tan, K.W.; Wang, Z.H.; Huang, S.M.; Wan, C.L.; Huang, Y.H.; Liu, M.J.; Yao, Z.; Li, Z.; et al. Venetoclax, cladribine plus low-dose cytarabine for relapsed/refractory acute myeloid leukemia: a multicenter, randomized phase II study. Blood 2015, 146, 5202. [Google Scholar] [CrossRef]
- Jin, H.; Zhang, Y.; Yu, S.; Du, X.; Xu, N.; Shao, R.; Lin, D.; Chen, Y.; Xiao, J.; Sun, Z.; et al. Venetoclax combined with azacitidine and homoarringtonine in relapsed/refractory AML: a multicenter, phase 2 trial. J Hematol Oncol 2023, 16, 42. [Google Scholar] [CrossRef]
- Yu, G.; Zhang, Y.; Yu, S.; Yin, Z.; Weng, G.; Xu, N.; Du, X.; Lin, D.; Xiao, J.; Sun, Z.; et al. Homoharringtonine added to venetoclax and azacitidine improves outcome and mitigates genetic impact in relapsed/refractory AML: a multicenter cohort study. Clin Cancer Res 2025, 31, 87–97. [Google Scholar] [CrossRef]
- Yu, G.; Chen, Y.; Yin, Z.; Zhang, Y.; Zhao, W.; Weng, G.; Du, X.; Nie, D.; Guo, Z.; Sun, Z.; et al. Poor response of venetoclax/azacitidine in monocytic-like acute myeloid leukemia might be associated with genetic patterns and could be improved by addition of Homoharringtonine. Blood 2024, 144, 1516. [Google Scholar] [CrossRef]
- DiNardo, C.; Jen, W.Y.; Montalban-Bravo, G.; Wang, X.; Loghavi, S.; Lavu, S.; Short, N.J.; Chien, K.; Issa, G.C.; Pemmaraju, N.; et al. Omecetaxine and venetoclax in relapsed/refractory acute myeloid leukemia or myelodysplastic syndrome with mutant RUNX1. Blood Neoplasia 2025, 2, 100145. [Google Scholar] [CrossRef]
- Murthy, G.S.; Saliba, A.N.; Szabo, A.; Harrington, A.; Abedin, S.; Carlson, K.; Michaelis, L.; Runaas, L.; Baim, A.; Hinman, A.; et al. A phase I study of pevonedistat, azacitidine, and venetoclax in patients with relapsed/refractory acute myeloid leukemia. Haematologica 2024, 109, 2864–2872. [Google Scholar] [CrossRef]
- Shahzad, M.; Mumtaz, A.; Basharat, A.; Irfan, S.; Saquid, M. H.; Altaf, F.; Faisal, S.; Zhara, F.T.; Zeeshan, M.; Akram, N.; et al. OPutcomes with venetoclax and hypomethylating agents in relapsed acute myeloid leukemia after allogeneic hematopoietic cell transplantation: a systematic review and meta-analysis. Transplant Cellular Therapy 2025, 31, S128–S129. [Google Scholar] [CrossRef]
- Moukalled, N.; Gallimard, J.E.; Bazarbachi, A.; Houhou, M.; Blaise, D.; Franke, G.N.; Salmenniemi, U.; Holderried, T.; Versluis, J.; et al. Venetoclax in combination with hypomethylating agents shows promising activity in acute myeloid leukemia with late relapse post allogeneic stem cell transplant: from the EBMT acute leukemia working party. Blood 2025, 146, 6079–6080. [Google Scholar] [CrossRef]
- Peters, D.T.; Chang, C.; Snow, A.; Buhlinger, K.M.; Patel, B.; Dennison, T.; Dawson, S.; Gallagher, S.; Tolley, R.; et al. Azacitidine and venetoclax with or without lymphocyte infusion in relapsed AML post-HSCT: a single-institution retrospective study. Blood 2024, 144, 1481. [Google Scholar] [CrossRef]
- Chiusolo, P.; Malagola, M.; Giglio, F.; Frioni, F.; Secreto, C.; Lessi, F.; D’Amore, F.; Monella, I.M.; Abbenante, M.; Marconi, G.; et al. Venetoclax plus hypomethylating agents as first salvage therapy for myeloid malignancies relapsing after allogeneic HSCT. Bone Marrow Transplant 2026, 61, 101–103. [Google Scholar] [CrossRef]
- Xie, J.; Bao, X.; Xue, S.L.; Shen, M.; Cen, J.; Yao, L.; Pan, J.; Zhu, M.; Liu, D.; Hu, X.; et al. Venetoclax with decitabine as frontline treatment in younger adults with newly diagnosed ELN adverse-risk AML. Blood 2023, 142, 1323–1327. [Google Scholar] [CrossRef]
- Cherry, E.M.; Abbott, D.; Amaya, M.; McMahon, C.; Schwartz, M.; Rosser, J.; Sato, A.; Schowinsky, J.; Inguva, A.; Minhajuddin, M.; et al. Venetoclax and azacitidine compared with induction chemotherapy for newly diagnosed patients with acute myeloid leukemia. Blood Adv 2021, 5, 5565–5573. [Google Scholar] [CrossRef]
- Maiti, A.; Qiao, W.; Sasaki, K.; Ravandi, F.; Kadia, T.M.; Jabbour, E.J.; Daver, N.G.; Borthakur, G.; Garia-Manero, G.; Pierce, S.A.; et al. Venetocalx with decitabine vs intensive chemotherapy in acute myeloid leukemia: a propensity score matched analysis stratified by risk of treatment-related mortality. Am J Hematol 2021, 96, 282–291. [Google Scholar] [CrossRef]
- Shimada, T.; Takahata, A.; Tanaka, K.; Toyota, S. Efficacy and safety of venetoclax-based regimens versus intensive chemotherapy in older adults with newly diagnosed acute myeloid leukemia: a single-center retrospective study. Int J Hematol 2025, in press. [Google Scholar] [CrossRef]
- Lu, J.; Xue, S.; Wang, Y.; He, X.; Hu, X.; Miao, M.; Zhang, Y.; Tanf, Z.; Xie, J.; Yang, X.; et al. Venetoclax and decitabine vs intensive chemotherapy as induction for young patients with newly diagnosed AML. Blood 145, 2645–2655. [CrossRef]
- Fathi, A.; Perl, A.; Fell, G.; Jonas, B.; Ragon, B.; Mims, A.; Borate, U.; Mannis, G.; Quillen, K.; Stahl, M.; et al. Results from paradigm – a phase 2 randomized multi-center study comparing azacitidine and venetoclax to conventional induction chemotherapy for newly diagnosed fit adults with acute myeloid leukemia. Blood 2025, 146, 6–7. [Google Scholar] [CrossRef]
- Fang, Q.; Li, F.; Liao, A.; Yin, O.; Yang, L.; Xu, L.; Ma, J.; Xu, J.; Zhang, H.; Wang, J.; et al. This is a title in sentence case: venetoclax-based regimens versus intensive chemotherapy in fit older adults with newly diagnosed acute myeloid leukemia (AML): a multicenter, perspective, randomized phase II trial. Blood 2025, 146, 650–651. [Google Scholar] [CrossRef]
- Wan, C.L.; Liu, Y.C.; Liu, F.T.; Huang, Y.H.; Cao, H.Y.; Huang, S.M.; Tan, K.W.; Ge, S.S.; Wang, M.; Liu, M.J.; et al. Venetoclax with hypomethylating agents versus intensive chemotherapy in newly diagnosed acute myeloid leukemia with myelodysplasia related changes: a propensity score-matched analysis based on International Consensus Classification. Blood Cancer J 2014, 14, 144. [Google Scholar] [CrossRef]
- Bewersdorf, J.P.; Shimonoy, S.; Shallie, R.M.; Liu, Y.; Berton, G.; Shaefer, E.J.; Zeidan, A.M.; Goldberg, A.D.; Stein, E.M.; Marcucci, G.; et al. Intensive induction chemotherapy vs hypomethylating agents in combination with venetoclax in NPM1-mutant AML. Blood Adv 2024, 8, 4845–4856. [Google Scholar] [CrossRef]
- Duan, W.; Chen, Q.; Yu, W.; Jia, J.; Wang, J.; Zhu, X.; Zhao, T.; Jiang, Q.; Ruan, G.; Zhao, X.; et al. The well tolerated VA-based regimen bringing to high-quality of life did not absolutely inferior to intensive chemotherapy in AML patients with NPM1 mutation. Blood 2024, 144, 5952–5953. [Google Scholar] [CrossRef]
- Zale, A.D.; Kaduluri, V.P.S.; Webster, J.A.; Levis, M.J.; Gojo, I.; DeZern, A.E.; Ghiaur, G.; Gondek, L.P.; Dalton, W.B.; Karantanos, T.; et al. Intensive chemotherapy vs venetoclax/hypomethylating agents for patients aged 60 to 75 years with favorable, NPM1-mutated AML. Blood Neoplasia 2025, 2, 100133. [Google Scholar] [CrossRef]
- Kretschmer, L.; Ruhnke, L; Schliemann, C.; Francesky, L.; Steffen, B.; Kaufmann, M.; Burchert, A.; Schmid, C.; Hanoun, M.; Sauer, T.; et al. VINCENT: a randomized-controlled trial evaluating venetoclax plus azacitidine versus intensive chemotherapy in patients with newly diagnosed, NPM1-mutated AML. Ann Hematol 2025, 104, 3647–3654. [Google Scholar] [CrossRef]
- Miyashita, A.; Basendwah, A.M.; Fero, H.; Alabass, F. CPX-351 versus venetoclax plus hypomethylating agents for newly diagnosed acute myeloid leukemia: a systematic review and meta-analysis. Leuk Res 2026, 162, 108179. [Google Scholar] [CrossRef]
- Fathima, S.; Rokach, L.; Ghosoun, N.; Razasoltani, M.; Alsugair, A.; McCullough, K.; Abdelmagid, M.; Al-Kali, A.; Alkhateeb, H.; Begna, K.; et al. CPX-351 versus venetoclax plus hypomethylating agents in newly diagnosed acute myeloid leukemia: a comparative analysis of outcomes in 600 patients. Blood 2025, 146, 1602–1603. [Google Scholar] [CrossRef]
- Boussi, L.; Bewersdorf, J.P.; Liu, Y.; Shallis, R.M.; Agulme, L.; Bystrom, R.; Zucenka, A.; Garciaz, S.; DeAngelo, D.; Stone, R.M.; et al. Hypomethylating agents plus venetoclax versus intensive chemotherapy in acute myelogenous leukemia with chromosome 5 and 7 abnormalities. Haematologica 2026, in press. [Google Scholar] [CrossRef]
- Aguirre, L.E.; Bewersdorf, J.P.; Liu, Y.; Shallis, R.M.; Boussi, L.; Zucenka, A.; Garciaz, S.; Bystrom, R.P.; DeAngelo, D.J.; Stone, R.M.; et al. Comparative effectiveness of HMA plus venetoclax versus intensive chemotherapy in AML with very-high-risk cytogenetics. Blood Neoplasia 2026, in press. [Google Scholar] [CrossRef]
- Bouligny, I.; Murray, G.; Ho, T.; Gor, J.; Zacholski, K.; Wages, N.; Grant, S.; Maher, K. First line venetoclax and a hypomethylating agent or conventional chemotherapy in older adults with IDH-mutated AML. Blood 2023, 142, 1488–1489. [Google Scholar] [CrossRef]
- Bewersdorf, J.P.; Shimony, S.; Shallis, R.; Liu, Y.; Berton, G.; Schaefer, E.; Zeidan, A.; Goldberg, A.; Stein, E.; Marcucci, G.; et al. Combination therapy with hypomethylating agents and venetoclax versus intensive induction chemotherapy in IDH1- or IDH2-mutant newly diagnosed acute myeloid leukemia- A multicenter cohort study. Am J Hematol 2024, 99, 1640–1643. [Google Scholar] [CrossRef]
- Khan, F.; Ajenaghughrure, G.; Qadri, H.; Zamani, T.; Alsidawi, S. AML 851: real-world comparison of hypomethylating agent-venetoclax and intensive chemotherapy in elderly acute myeloid leukemia: survival and safety insights from a global matched cohort. Clin Lymphoma Myeloma Leukemia 2025(25), S464.
- Chen, X.; Yan, Y.; Cheng, Q.; Tang, Y.; Xiao, H.; Li, Y.; Wang, E.; Zhang, J.; Wang, U.; Zhang, Q.; et al. Genotype-guided comparison of VEN/HMA versus intensive chemotherapy in newly diagnosed intermediate-risk AML: a multicenter real-world study. Blood Cancer J 2025, 15, 153. [Google Scholar] [CrossRef] [PubMed]
- Matthews, A.; Perl, A.; Luger, S.; Gill, S.; Lai, C.; Porter, D.L.; Skull, S.; Bruno, X.; Carroll, M.; Freier, C.; et al. Real-world effectiveness of intensive chemotherapy with 7&3 versus venetoclax and hypomethylating agent in acute myeloid leukemia. Am J Hematol 2023, 98, 1254–1264. [Google Scholar]
- Moghaddam, A.S.; Ruby, H.; Syed, A.; Fanizza, J.; Nasrollahi, E.; Papp, S.R.; Rajagopalan, V.; Atrash, A. Real-world outcomes of venetoclax-based therapy vs intensive chemotherapy in older adults with newly diagnosed acute myeloid leukemia. Blood 2025(146), 6996.
- Wei, A.H.; Strickland, S.A.; Hou, J.Z.; Fiedler, W.; Lin, T.L.; Walter, R.B.; Enjeti, A.; Tiong, I.S.; Savona, M.; Lee, S.; et al. Venetoclax combined with low-dose cytarabine for previously untreated patients with acute myeloid leukemia: results from a phase Ib/II study. J Clin Oncol 2019, 37, 1277–1284. [Google Scholar] [CrossRef]
- Wei, A.H.; Montesinos, P.; Ivanov, V.; DiNardo, C.D.; Novak, J.; Laribi, K.; Kim, I.; Stevens, D.A.; Fiedler, W.; Pagoni, M.; et al. Venetoclax plus LDAC for newly diagnosed AML ineligible for intensive chemotherapy: A phase 3 randomized placebo-controlled trial. Blood 2020, 135, 2137–2145. [Google Scholar] [CrossRef]
- Wei, A.H.; Panayotidis, P.; Montesinos, P.; Laribi, K.; Ivanov, V.; Kim, I.; Novak, I.; Champion, R.; Fiedler, W.; Pagoni, M.; et al. Long-term follow-up of VIALE-C in patients with untreated AML ineligible for intensive chemotherapy. Blood 2022, 140, 2754–2756. [Google Scholar] [CrossRef]
- Wang, K.; Pokima, N.; Keesari, P.R.; El-Gharib, K.; Widjaja, M; El-Sayegh. The efficacy of LDAC+Venetoclax and CPX-351 over traditional regimen in older patients with AML not eligible for intensive therapy: a meta-analysis and systematic review. Blood 2024, 144, 6073–6074. [Google Scholar] [CrossRef]
- Wei, A.H.; Panayotidis, P.; Montesinos, P.; Ivanov, V.; DiNardo, C.D.; Novak, J.; Laribi, K.; Kim, I.; Stevens, D.A.; Fiedler, W.; Pagoni, M.; et al. Risk stratification at low-dose cytarabine and venetoclax in patients with AML ineligible for intensive chemotherapy. Blood Adv 2026. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, L.; Lv, Z.; Mao, L.; Hu, C.; Wang, J.; Zhou, Y.; Jin, J.; Meng, H.; You, L. Venetoclax plus azacitidine and LDAC induced high response rates in acute myelkoid leukemia in routine clinical practice. Br J Haematol 2023, 201, 995–999. [Google Scholar] [CrossRef]
- Han, X.; Yi, H.; Yan, H.; Zhang, Y.; Luo, Y.; Xiao, Y.; Zhang, M.; Fang, L.; Chen, G.; Peng, Z.; et al. Venetoclax, Azacitidine, combined with low-dose cytarabine in older newly diagnosed acute myeloid leukemia: the result of a prospective, multicenter, open, randomized controlled trials. Blood 2024, 144, 2902–2903. [Google Scholar] [CrossRef]
- Chen, X.T.; Li, Q.; Zhao, Y.Q.; Fan, S.J. Safety and efficacy of venetoclax-azacitidine combined with low-dose idarubicin and cytarabine regimens for the treatment of newly diagnosed acute myeloid leukemia patients compared to the standard chemotherapy: a propensity score-matched real-world single-center experience. Ann Hematol 2025, 104, 6197–6204. [Google Scholar]
- Wang, H.; Mao, L.; Yang, M.; Qian, P.; Lu, H.; Tong, H.; Xie, W.; Zhou, D.; Huang, X.; Wang, Y.; et al. Venetoclax plus 3+7 daunorubicin and cytarabine chemotherapy as first-line treatment for adults with acute myeloid leukemia: a multicentre, single-arm, phase 2 trial. Lancet Hematol 2022, 9, e415–e424. [Google Scholar] [CrossRef]
- Wang, R.; Zhang, Y.; Chang, J.; Wang, H.; Lou, Y.; Yang, M.; Xu, G.; Tong, H.; Xie, W.; Zhou, D.; et al. Venetoclax plus daunorubicin and cytarabine in newly diagnosed acute myeloid leukemia patients: a propensity score-matched analysis. Hematol Oncol 2024, 42, e3260. [Google Scholar] [CrossRef] [PubMed]
- Mantzaris, I.; Goldfinger, M.; Uniel, M.; Shastri, A.; Gritsman, K.; Komblum, N.; Shapiro, L.; Sica, R.A.; Munoz, A.; Chambers, N.; et al. Venetoclax plus daunorubicin and cytarabine for newly diagnosed acute myeloid leukemia: results of a phase 1b study. Blood 2025, 145, 1870–1875. [Google Scholar] [CrossRef]
- Liu, Y.; Bai, G.; Zhao, X.; Suo, X.; Bai, Y.; Zhao, W.; Peng, H.; Zhang, F.; Wang, D.; Zhao, L.; et al. Venetoclax combined with intensive chemotherapy as an induction chemotherapy in newly diagnosed FLT3-ITD-positive AML. Blood 2024, 144, 1485–1486. [Google Scholar] [CrossRef]
- Chua, C.C.; Loo, S.; Fong, V.Y.; Ting, S.B.; Tiong, I.S.; Fleming, S.; Anatee, N.; Ivey, A.; Ashby, M.; The, T.C.; et al. Final analysis of the phase 1b chemotherapy and venetoclax in elderly acute myeloid leukemia trial (CAVEAT). Blood Adv 2025, 9, 1827–1835. [Google Scholar] [CrossRef]
- DiNardo, C.D.; Lachowiez, C.A.; Takahashi, K.; Loghavi, S.; Xiao, L.; Kadia, T.; Daver, N.; Adeoti, M.; Short, N.J.; Sasaki, K.; et al. Venetoclax Combined with FLAG-IDA Induction and Consolidation in Newly Diagnosed and Relapsed or Refractory Acute Myeloid Leukemia. J Clin Oncol 2021, 39, 2768–2778. [Google Scholar] [CrossRef] [PubMed]
- DiNardo, C.D.; Wen, W. Y.; Takahashi, K.; Kadia, T.; Loghavi, S.; Daver, N.; Xiao, L.; Reville, P.K.; Issa, G.C.; Short, N.J.; et al. Long term results of venetoclax combined with FLAG-IDA induction and consolidation for newly diagnosed and relapsed or refractory acute myeloid leukemia. Leukemia 2025, 39, 854–863. [Google Scholar] [CrossRef]
- Al-Shaibani, E.; Davidson, M.B.; Bankar, A.; Chan, S.M.; Sibai, H.; Maze, D.C.; Schimmer, A.D.; Minden, M.D.; Yee, K.; Gupta, V.; et al. FLAG-IDA in high-risk newly diagnosed and relapsed or refractory acute myeloid leukemia: the Pricess Margaret Cancer Center experience. Blood 2024, 144, 4273–4274. [Google Scholar] [CrossRef]
- Jen, W.Y.; Takahashi, K.; Loghavi, S.; Kadia, T.M.; Daver, N.G.; Xiao, L.; Short, N.J.; Borthakur, G.; Issa, G.; Pemmaraju, N.; et al. FLAG-IDA + venetoclax in newly diagnosed (ND) or relapsed/refractory (RR) AML. J Clin Oncol 2024. [Google Scholar] [CrossRef]
- Wille, K.; Dumke, M.; Wilsdorf, N.; Sadjadian, P.; Schnedider, A.; Jender-Bartling, S.; Kolatzki, V.; Horstmann, A.; Meixner, R.; Jimenez-Munoz, M.; et al. Venetoclax combined with FLAG-IDA in refractory or relapsed acute myeloid leukemia. Eur J Haematol 2025, 114, 17–25. [Google Scholar] [CrossRef]
- Shahswar, R.; Beutel, G.; Gabdoulline, R.; Schwarzer, A.; Kloos, A.; Koenecke, C.; Stadler, M.; Gohring, G.; Behrens, Y.L.; Li, Z.; et al. Fludarabine, cytarabine, and idarubicin with or without venetoclax in patients with relapsed/refractory acute myeloid leukemia. Haematologica 2024, 109, 72–80. [Google Scholar] [CrossRef]
- Raj, R.; Catueno, S.; Gibson, A.; McCall, D.; Garcia, M.B.; Nunez, C.; Roth, M.; Sasaki, K.; Tewari, P.; Issa, G.C.; et al. FLAG-IDA plus venetoclax for children, adolescents, and young adults with newly diagnosed AML. Blood 2025, 146, 2608–2611. [Google Scholar] [CrossRef]
- Fedorov, K.; Kennedy, K.; Mittlebeeler, E.; Profitt, S.; Yalvington, B.; Brooks, C.; Kishtagari, A.; Mohan, S.; Savona, M.; Ball, S. High remission rates with FLAG-IDA-venetoclax in acute myeloid leukemia: “Less is more” on days of venetoclax in the intensive regimen. Blood 2025, 146, 1666–1667. [Google Scholar] [CrossRef]
- Bashey, A.; Zhang, X.; Morris, L.E.; Holland, E.K.; Brachier-Rodriguez, L.; Solomon, S.R.; Solh, M.M. Comparison of FLAG-idarubicin to FLAG-idarubicin plus venetoclax for treatment of newly diagnosed AML in patients eligible for intensive induction. Analysis of 200 consecutive patients treated as a single center. Blood 2024, 144, 1522–1523. [Google Scholar] [CrossRef]
- Schonrock, M.; Sonnemann, P.; Michalowski, P.; Heuser, M.; Thol, F.; Ayuk, F.A.; Wolschke, C.; Klyucnikov, E.; Bokemeyer, C.; Fiedler, W.; et al. Dose-reduced FLA-IDA in combination with venetoclax is an effective and safe salvage therapy in relapsed and refractory acute myeloid leukemia (R/R AML). Cancers 2024, 16, 3872. [Google Scholar] [CrossRef]
- Marconi, G.; Piciocchi, A.; Audisio, E.; Papayannidis, C.; Cerrano, M.; Minotti, C.; Paoloni, F.; Guolo, F.; Bocchia, M.; Rondoni, M.; et al. Safety run-in and part 1 of GIMEMA AML1718: venetoclax combined with FLAI as induction treatment in non-low-risk AML. Blood Adv 2025, 9, 2542–2550. [Google Scholar] [CrossRef] [PubMed]
- Ruhnke, L.; Schlieman, C.; Mikesch, J.H.; Stelljes, M.; Fransecky, L.; Steffen, B.; Kaufmann, M.; Burchert, A.; Rank, A.; Hanoun, M.; et al. Venetoclax plus high-dose cytarabine and mitoxantrone as salvage treatment for relapsed or refractory acute myeloid leukemia (RELAX): a multicentre, single-arm, phase 1-2 trial. Lancet Hemtol 2026, 13, e157–e168. [Google Scholar] [CrossRef] [PubMed]
- Kadia, T.M.; Reville, P.K.; Borthakur, G.; Yilmaz, M.; Komblau, S.; Alvarado, Y.; Dinardo, C.D.; Daver, N.; Jain, N.; Pemmaraju, N.; et al. Venetoclax plus intensive chemotherapy with cladribine, idarubicin, and cytarabine in patients with newly diagnosed acute myeloid leukemia or high-risk myelodysplastic syndrome: a cohort from a single-centre, single-arm, phase 1 trial. Lancet Hematol 2021, 8, e552–e561. [Google Scholar] [CrossRef] [PubMed]
- Bambace, N.; Eilo, E.; Al Sagheer, T.; Andersen, A.; El Chaer, F.; Feinstein, L.; Corrales, E.G.; Perez, A.P.; Morales, A.R.; Shwin, M.; et al. Clia plus venetoclax for AML: feasibility and efficacy to a hybrid community setting. Blood 2025, 146, 6962. [Google Scholar] [CrossRef]
- Lee, B.J.; Griffin, S.; Doh, J.; Quach, A.; Sun, Y.; Chamnusiriwath, P.; Ciurea, S.O.; Ramos-Perez, J.; Bellman, P.; Jeyakumar, P.M.; et al. Venetoclax combined with cladribine, idarubicin, cytarabine (CLIA-VEN) results in higher remission rates over conventional 7+3 chemotherapy without increased toxicity in newly diagnosed acute myeloid leukemia. Brit J Haematol 2026, in press. [Google Scholar] [CrossRef]
- Jen, W.Y.; Croden, J.; Almanza-Huante, E.; DiNardo, C.; Chien, K.; Hammond, D.; Qiao, W.; Alvarado, Y.; Masanova, L.; Quesada, A.E.; et al. A phase 2 study of CPX-351 in combination with venetoclax in patients with newly diagnosed high-risk acute myeloid leukemia. HemaSphere 2025, 9, e70214. [Google Scholar] [CrossRef]
- Croden, J.; Jen, W.Y.; Marvin-Peek, J.; Xiao, L.; Bouligny, I.M.; Loghavi, S.; Borthakur, G.; Daver, N.; Abbas, H.A.; Tkahashi, K.; et al. Outcomes of adult patients with newly diagnosed IDH-mutated AML treated with intensive chemotherapy and venetoclax. Leukemia 2025, 39, 2538–2541. [Google Scholar] [CrossRef] [PubMed]
- Wei, A.H.; Dohner, H.; Pocock, C.; Pocok, C.; Montesinos, P.; Afamyes, B.; Dombret, H.; Ravadi, F.; Sayar, H.; Jang, J.H.; et al. Oral azacitidine maintenance therapy for acute myeloid leukemia in first remission. New England J Med 2020, 383, 2526–2537. [Google Scholar] [CrossRef]
- Dohner, H.; Wei, A.H.; Roboz, G.J.; Montesinos, P.; Thol, F.R.; Ravandi, F.; Dombret, H.; Parkka, K.; Sandhu, I.; Skikne, B.; et al. Prognostic impact of NPM1 and FLT3 mutations in patients with AML in first remission treated with oral azicitidine. Blood 2022, 140, 1674–1685. [Google Scholar] [CrossRef]
- Wei, A.H.; Dohner, H.; Sayar, H.; Ravandi, F.; Montesinos, P.; Dombret, H.; Sellesleg, D.; Parkka, K.; Jang, J.H.; Sikne, B.; et al. Long-term survival with oral azacitidine after chemotherapy: updated results from the randomized, placebo-controlled, phase 3 QUAZAR AML-001 trial. Am J Hematol 2023, 98, E84–E87. [Google Scholar] [CrossRef]
- Oran, B.; de Lima, M.; Garcia-Manero, G.; Thall, P.F.; Lion, R.; Popat, U.; Alousi, A.M.; Hosing, C.; Giralt, S.; Rondon, G.; et al. A phase 3 randomized study of 5-azacitidine maintenance vs observation after transplant in high-risk AML and MDS patients. Blood Adv 2020, 4, 5580–5588. [Google Scholar] [CrossRef]
- Bazinet, A.; Kantarjian, H.; Bataller, A.; Pemmaraju, N.; Borthakur, G.; Chien, K.; Alvarado, Y.; Bose, P.; Jabbour, E.; Yilmaz, M.; et al. reduced-dose azacitidine plus venetoclax as maintenance therapy in acute myeloid leukemia following intensive or low-intensity induction: results of a single-centre phase 2 trial. Lancet Hematol 2024, 11, e287–e298. [Google Scholar] [CrossRef]
- Medawar, G.; Graf, K.; Anoma, J.S.; Guo, Q.J.; Hockman, A.; McCraney, S.; Weiss, J.; Lin, C.; Sastow, D.; Tremblay, D.; et al. Maintenance with hypomethylating agent+ venetoclax after intensive induction chemotherapy in patients with newly diagnosed acute myeloid leukemia. Blood 2025, 146, 6960–6961. [Google Scholar] [CrossRef]
- Kent, A.; Schwartz, M.; McMahon, C.; Amaya, M.; Smith, C.A.; Tobin, J.; Marciano, K.; Rezac, R.; Bosma, G.; Pollyea, D.A.; et al. Venetoclax is safe and tolerable as post-transplant maintenance therapy for AML patients at high risk for relapse. Bone Marrow Transplant 2023, 58, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Oran, B.; Thall, P.; Alatrash, G.; Im, J.; Ramdial, J.; Bassett, R.; Popat, U.; Kebriaei, P.; Smallbone, P.; Jewell, J.; et al. Phase II rtial of venetoclax in combination with azacitidine as maintenance therapy for high-risk acute leukemia following allogeneic stem cell transplantation. Blood 2025, 146, 2511. [Google Scholar] [CrossRef]
- Garcia, J.S.; Kim, H.T.; Murdock, M.; Ansuinelli, M.; Brock, J.; Cutler, C.S.; Gooptu, M.; Ho, V.T.; Koreth, J.; Nikiforow, S.; et al. Prophylactic maintenance with venetoclax/azacitidine after reduced-intensity conditioning allogeneic transplant for high-risk MDS and AML. Blood Adv 2024, 8, 978–987. [Google Scholar] [CrossRef]
- Diebold, K.; Mudd, T.; Hardee, M.; Thigpey, H.; Espinoza-Gutarra, M.; Di Stasi, A.; Salzman, D.; Mothy, R.; Bhatia, R.; Jamy, O. Improved outcomes with low-dose hypomethylating agent plus venetoclax as post-transplant maintenance in patients undergoing reduced intensity conditioning allogeneic hematopoietic stem cell transplantation for myeloid malignancies. Blood 2025, 146, 4261–4262. [Google Scholar] [CrossRef]
- Wang, E.S.; Montesinos, P.; Minden, M.D.; Lee, J.H.; Heuser, M.; Naoe, T.; Chou, V.C.; Laribi, K.; Esteve, J.; Altman, J.K.; et al. Phase 3 trial of gilteritinib plus axacitidine vs azacitidine for newly diagnosed FLT3mut+ AML ineligible for intensive chemotherapy. Blood 2022, 140, 1845–1857. [Google Scholar] [CrossRef]
- Mai, R.S.; Zhang, Q.; DeFilippis, R.A.; Cavazos, A.; Kuruvilla, V.M.; Raman, J.; Mody, V.; Choo, E.F.; Dail, M.; et al. Venetoclax combines synergistically with FLT3 inhibition to effectively target leukemic cells in FLT3-ITD+ acute myeloid leukemia. Haematologica 2021, 106, 1034–1046. [Google Scholar]
- Janseen, M.; Schmidt, C.; Bruch, P.M.; Blank, M.F.; Rohde, C.; Wadawiczek, A.; Heid, D.; Renders, S.; Gollner, S.; Vierbaum, L.; et al. Venetoclax synergizes with gilteritinib in FLT3 wild-type high-risk acute myeloid leukemia by suppressing MCL-1. Blood 2002, 140, 2594–2600. [Google Scholar] [CrossRef]
- Daver, N.; Perl, A.E.; Maly, J.; Levis, M.; Ritchie, E.; Litzow, M.; McCloskey, J.; Smith, C.C.; Schiller, G.; Bradley, T.; et al. Venetoclax plus gilteritinib for FLT3-mutated relapsed/refractory acute myeloid leukemiua. J Clin Oncol 2022, 40, 4048–4059. [Google Scholar] [CrossRef] [PubMed]
- Maiti, A.; DiNardo, C.D.; Daver, N.G.; Rausch, C.R.; Ravandi, F.; Kadia, T.M.; Pemmaraju, N.; Borthakur, G.; Bose, P.; Issa, G.C.; et al. Triplet therapy with venetoclax, FLT3 inhibitor and decitabine for FLT3-mutated acute myeloid leukemia. Blood Cancer J 2021, 11, 25. [Google Scholar] [CrossRef]
- Yilmaz, M.; Kantarjian, H.; Short, N.J.; Reville, P.; Konopleva, M.; Kadia, T.; DiNardo, C.; Borthakur, G.; Pemmaraju, N.; Maiti, A.; et al. Hypomethylating agent and venetoclax with FLT3 inhibitor triplet therapy in older/unfit patients with FLT3 mutated AML. Blood Cancer J 2022, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Short, N.J.; Nguyen, D.; Ravandi, F. Treatment of older adults with FLT3-mutated AML: emerging paradigma and the role of frontline FLRT3 inhibitors. Blood Cancer J 2023, 13, 142. [Google Scholar] [CrossRef] [PubMed]
- Short, N.J.; Daver, N.; DiNardo, C.D.; Kadia, T.; Nasar, L.; Macaron, W.; Yilmaz, M.; Borthakur, G.; Montalban-Bravo, G.; Garcia-Manero, G.; et al. Azacitidine, venetoclax, and gilteritinib in newly diagnosed and relapsed or refractory FLT3-mutated AML. J Clin Oncol 2024, 42, 1499–1508. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, R.S.; Daver, N.; Karrar, O.; DiNardo, C.D.; Kadia, T.; Yilmaz, M.; Borthakur, G.; Issa, G.; Jabbour, E.; Huang, X.; et al. Long-term follow-up of azacitidine, venetoclax, and gilteritinib in patients with newly diagnosed FLT3-mutated acute myeloid leukemia. Blood 2025, 146, 45–46. [Google Scholar] [CrossRef]
- Altman, J.; Perl, A.; Wang, E.; Borate, U.; Traer, E.; Dugan, J.; Kasner, M.; Larson, R.; Deutsch, Y.; Shastri, A.; et al. Venetoclax (VEN) and azacitidine (AZA) with gilteritinib (GILT) in patients with newly diagnosed (ND) FLT3mut+ acute myeloid leukemia (AML) ineligible for intensive chemotherapy (chemo): interim results from the phase 1-2 VICEROY study. Blood 2025, 146, 654–655. [Google Scholar] [CrossRef]
- Arora, S.; Kantarjian, H.; Maroun, M.; Borthakur, G.; Garcia-Manero, G.; Montaban-Bravo, G.; Issa, G.; Chien, K.; Maiti, A.; Ohanina, M.; et al. A phase II study of azacitidine, venetoclax, and gilteritinib for newly diagnosed adverse risk FLT3-wild type acute myeloid leukemia. Blood 2025, 146, 5226–5227. [Google Scholar] [CrossRef]
- Jiang, E.; Liang, C.; Yang, D.; He, Y.; Pang, A.; Zhang, R.; Han, M.; Feng, S. Efficacy of the triplet tehrapy with venetoclax, azacitidine and gilteritinib for the treatment of FLT3-ITD-mutated AML patients with relapsed/refractory disease or persistent minimal residual disease bridging to allogeneic hematopoietic stem cell transplantation. Blood 2025, 146, 2519–2520. [Google Scholar]
- Short, N.J.; Loghavi, S.; Yilmaz, M.; Karrar, O.; Kim, K.; DiNardo, C.D.; Kadia, T.M.; Maround, M.; Borthakur, G.; Issa, G.C.; et al. Long-term outcomes in FLT3-mutated acute myeloid leukemia after frontline hypomethylating agent, venetoclax and a FLT3 inhibitor. Haematologica 2026. [Google Scholar] [CrossRef]
- Arora, S.; Loghavi, S.; Daver, N.; Ravandi, F.; DiNardo, C.D.; Kadia, T.M.; Borthakur, G.; Jabbour, E.; Yilmaz, M.; Issa, G.C.; et al. Cytomolecular mechanisms of relapse and post-relapse outcomes after frontline FLT3 inhibitor-based therapy in FLT3-mutated AML. Am J Hematol 2025, 100, 2155–2159. [Google Scholar] [CrossRef]
- Yilmaz, M.; Muftuoglu, M.; Short, N.; Loghavi, S.; Kadia, T.; DiNardo, C.D.; Borthakur, G.; Pemmaraju, N.; Alvarado, Y.; Maiti, A.; et al. AML-804: triplet therapy with decitabine, venetoclax, and quizartinib for FLT3-ITD mutated AML: a phase 1-2 study. Clin Lymphoma Myeloma Leukemia 2025, 25(supll. 1), S460. [Google Scholar] [CrossRef]
- Chua, C.; Hsu, B.; Enjeti, A.K.; Bajel, A.; Marlton, P.; Fleming, S.; Hiwase, D.; Ma, C.K.K.; Browett, P.J.; Perera, T.; et al. A phase II randomized trial comparing low-dose cytarabine and venetoclax +/- midostaurin in non-adverse cytogenetic risk acute myeloid leukemia: the ALLG AMLM25 intervene trial. Blood 2025, 146, 217–219. [Google Scholar] [CrossRef]
- Montesinos, P.; Marchione, D.; Recher, C.; Heuser, M; Vives, S.; Zarzycka, E.; Wang, J.; Riva, M.; Calado, R.; Schuh, A.; et al. Long-term results from the AGILE study of azacitidine plus ivosidenib vs placebo in newly diagnosed IDH1-mutated AML. Blood Adv 2025, 9, 5177–5185. [Google Scholar] [CrossRef]
- DiNardo, C.D.; Marvin-Peek, J.; Loghavi, S.; Takahashi, K.; Issa, G.C.; Jen, W.Y.; Daver, N.G.; Reville, P.K.; Short, N.J.; Sasaki, K.; et al. Outcomes of frontline triplet regimens with a hypomethylating agent, venetoclax, and isocitrate dehydrogenase inhibitor for intensive chemotherapy-ineligible patients with isocitrate dehydrogenase-mutated AML. J Clin Oncol 2025, 43, 2692–2699. [Google Scholar] [CrossRef]
- Marvin-Peek, J.; DiNardo, C.D.; DiNardo, C.D. Reply to: late non-relapse mortality: a hidden risk in triplet therapy for IDH-mutated AML? J Clin Oncol 2026, 44, 68–70. [Google Scholar] [CrossRef]
- Marvin-Peek, J.; Maiti, A.; Alvarado, Y.; Daver, N.; Sasaki, K.; Borthakur, G.; Short, N.; Chien, K.; Hadda, F.; Hammond, D.E.; et al. AML-150: a phase 1b/2 trial of an all-oral triplet regimen for IDH-mutated myeloid malignancies: decitabine or cedazuridine and venetoclax in combination with ivosidenib or enasidenib. Clin Lymphoma Myeloma Leukemia 2025(25), S404–S405.
- Wang, F.; Morita, K.; DiNardo, C.D.; Furudate, K.; Tanaka, T.; Yan, Y.; Patel, K.P.; MacBeth, K.; Wu, B.; Liu, G.; et al. Leukemia stemness and co-occurring mutations drive resistance to IDH inhibitorsa in acute myeloid leukemia. Nat Commun 2021, 12, 2607. [Google Scholar] [CrossRef]
- Lachowiez, C.A.; Loghavi, S.; Zeng, Z.; Tanaka, T.; Kim, Y.J.; Uryu, H.; Turkalji, S.; Jakobsen, N.A.; Luskin, M.R.; Duose, D.Y.; et al. A phase Ib/II study of ivosidenib with venetoclax±azacitidine in IDH1-mutated myeloid malignancies. Blood Cancer Discov 2023, 4, 276–293. [Google Scholar] [CrossRef]
- Sirenko, M.; Lee, S.; Sun, Z.; Cheligne, R.; Loghavi, S.; Asimomitis, G.; Brierley, C.; Bernard, E.; Cai, S.; Myers, R.M.; et al. Deconvoluting clonal and cellular architecture in IDH-mutant acute myeloid leukemia. Cell Stem Cell 2025, 32, 1102–1121. [Google Scholar] [CrossRef]
- Turkalji, S.; Radtke, F.A.; Stoilova, B.; Mecklenbrauck, R.; Groom, A.J.; Jakobsen, N.A.; Lachowiez, C.A.; Mertzner, M.; Usukhbayar, B.; Salazar, M.A.; et al. Rapid clonal selection within early hematopoietic cell compartment presages the outcome of ivosidenib combination therapy. Blood 2026, 147, 613–635. [Google Scholar] [CrossRef] [PubMed]
- Testa, U.; Riccioni, R.; Diverio, D.; Rossini, A.; Lo-Coco, F.; Peschle, C. Interleukin-3 receptor in acute leukemia. Leukemia 2004, 18, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Pelosi, E.; Castelli, G.; Testa, U. CD123 a therapeutic target for acute myeloid leukemia and blastic plasmocytoid dendritic neoplasm. Int J Mol Sci 2023, 24, 2718. [Google Scholar] [CrossRef]
- Testa, U.; Riccioni, R.; Coccia, E.; Stellacci, E.; Samoggia, P.; Latagliata, R.; Mariani, G.; Rossini, A.; Battistini, A.; Lo-Coco, F.; et al. Elevated expression of IL-3Ralpha in acute myelogenous leukemia is associated with enhanced blast proliferation, increased cellularity and poor prognosis. Blood 2002, 100, 2980–2988. [Google Scholar] [CrossRef] [PubMed]
- Riccioni, R.; Diverio, D.; Riti, V.; Buffolino, S.; Mariani, G.; Boe, A.; Cedrone, M.; Ototne, T.; Foà, R.; Testa, U. Interleukin (IL)-3/granulocyte macrophage-colony stimulating factor /IL-5 receptor alpha and beta chains are preferentially expressed in acute myeloid leukemias with mutated FMS-related tyrosine 3 kinase receptor. Brit J Haematol 2009, 144, 376–387. [Google Scholar] [CrossRef]
- Riccioni, R.; Pelosi, E.; Riti, V.; Castelli, G.; Lo-Coco, F.; Testa, U. Immunophenotypic features of acute myeloid leukemia patients exhibiting high Flt3 expression not associated with mutations. Brit J Haematol 2011, 153, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Perriello, V.M.; Gionfriddo, I.; Rossi, R.; Milano, F.; Mezzasoma, F.; Marra, A.; Spinelli, O.; Rambaldi, A.; Annibali, O.; Avvisati, G.; et al. CD123 is consistently expressed on NPM1-mutated AML cells. Cancers 2021, 13, 496. [Google Scholar] [CrossRef] [PubMed]
- Skwarska, A.; Kuruvilla, V.M.; Zhang, Q.; Chaudry, S.; Daver, N.; Watkins, K.; Sloss, C.M.; Zweilder-McKay, P.; Sampath, D.; Konopleva, M. Venetoclax synergizes with IMGN632, a novel CD123 targeting antibody conjugated to a DNA alkylating payload, by suppressing DNA damage response and potentiating apoptosis in acute myeloid leukemia in vitro models. Blood 2023, 142, 4155–4156. [Google Scholar] [CrossRef]
- Tiu, B.C.; Su, C.J.; St Martin, E.; Oak, J.S.; Zhang, T.Y.; Mannis, G.N. Prognostic significance of CD123 expression in acute myeloid leukemia treated with venetoclax and hypomethylating agents. Blood 2024, 144, 6104–6105. [Google Scholar] [CrossRef]
- Daver, N.; Montesinos, P.; Altman, J.K.; Wang, E.S.; Martinelli, G.; Roboz, G.J.; Begana, K.; Vyas, P.; Lunghi, M.; Platzbecker, U.; et al. Pivekimab surinine (PVEK, IMGN632), a CD123-targeting antibody-drug conjugate, in combination with azacitidineand venetoclax in patients with newly diagnosed acute myeloid leukemia. Blood 2023, 142, 2906–2911. [Google Scholar] [CrossRef]
- Daver, N.; Advani, A.; De La Fuente Burguera, A.; Montesinos, P.; Garciaz, S.; Giannini, M.B.; Wang, E.; Roboz, G.; Curti, A.; Lunghi, M.; et al. Efficacy and safety of pevikimab sunirine in combination with venetoclax plus azacitidine in unfit patients with newly diagnosed acute myeloid leukemia. Blood 2025, 146, 651. [Google Scholar] [CrossRef]
- Pemmaraju, N.; Mazzarella, L.; Marconi, G.; Sallman, D.; Ulrickson, M.; Walter, R.; Maisano, V.; Fishman, B.; Du, Y.; et al. Efficacy and safety of pivekimab sunirine in blastic plasmocytoid dendritic cell neoplasm in patients with prior or concomitant hematologic malignancy; results from the cadenza study. Blood 2025, 146, 5195. [Google Scholar] [CrossRef]
- Lane, A.; Luskin, M.; Keating, J.; Leboeuf, N.; Garcia, J.; Shimony, S.; Leonard, R.; Goodpaster, A.; Nguyen, T.; Galinsky, I.; et al. Tagroxofusp, azacitidine and venetoclax (TAG-AZA-VEN) triplet therapy shows efficacy, tolerability, and transplant potential in patients with blastic plasmocytoid dendritic cell neoplasm (BPDCN): results of a phase 2 trial. Blood 2025, 146, 653–654. [Google Scholar] [CrossRef]
- Watts, J.; Borthakur, G.; Lin, T.; Gabrail, N.; Patel, K.; Madanat, Y.; Zeidner, J.; Khaskely, N.; Bonham, L.; Nelson, M.; et al. A dose optimization phase 1b/2 study evaluating mipletamig (formerly APVO436), a novel bispecific CD123 x CD3 ADAPTIRO’ molecule in combination therapy for the treatment of frontline acute myeloid leukemia (AML) in patients unfit for intensive chemotherapy. Blood 2025, 146, 5198–5199. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



