Submitted:
21 March 2026
Posted:
23 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Hedonic Capacity as an Independent Clinical Dimension
3. Functional Polarization of the Opioid System During Evolution
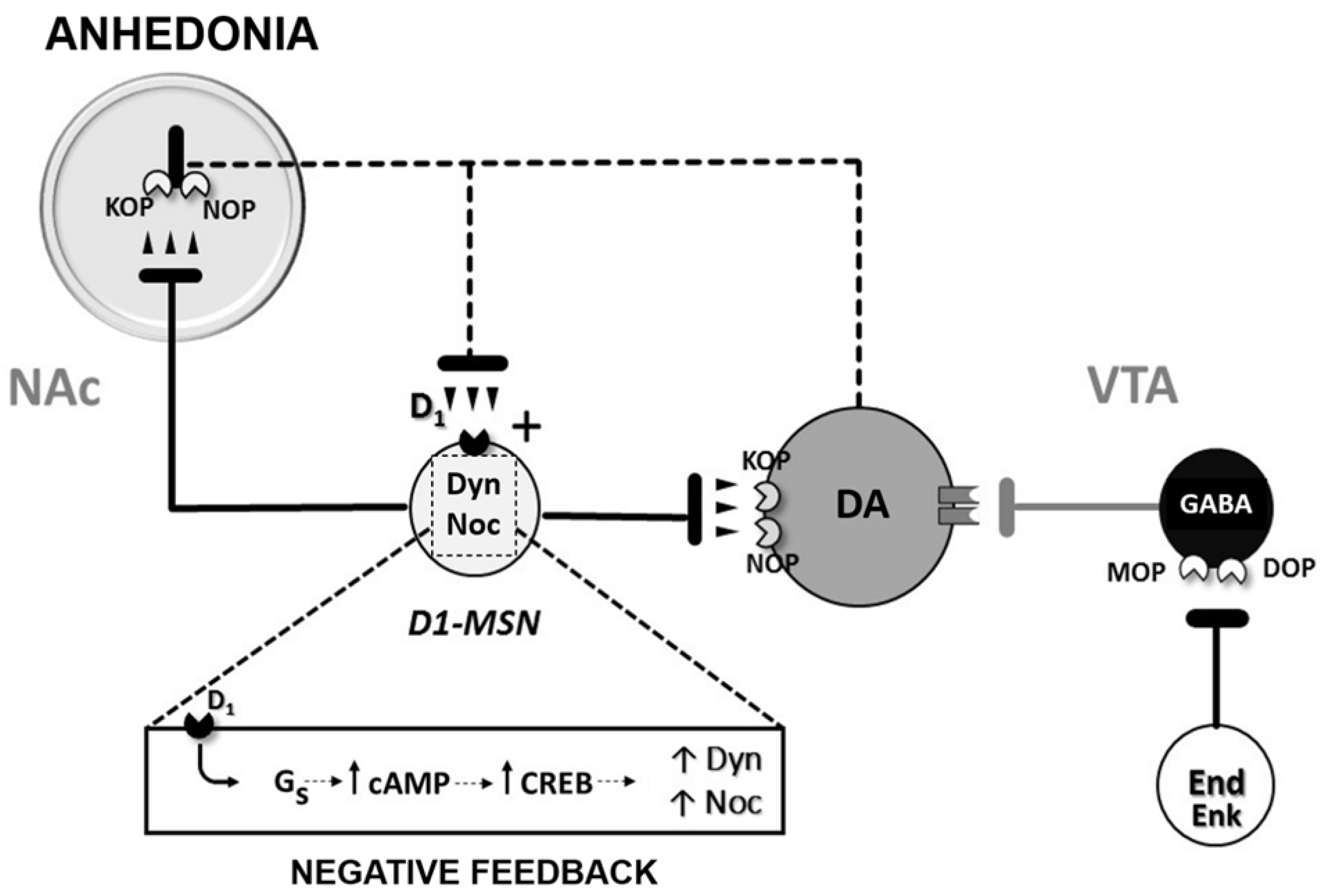
4. Dynorphin and Nociceptin Opioid Systems as Negative Regulators of Hedonic Homeostasis
5. Dynorphin and Nociceptin Opioid Systems and Stress
6. Dynorphin and Nociceptin Opioid Systems and Addiction
7. Clinical Trials of Kappa Opioid Receptor Antagonists
8. Clinical Trials of Nociceptin Opioid Receptor Antagonists
9. Novel Drugs Development Prospects
9.1. Ligand-Receptor Interactions
9.2. Receptor Profile
10. Conclusions
Conflicts of Interest
References
- Bassey, R.; Sun, X.; Jin, C.; Cui, C.; Chen, Z.; Dai, Q. Reward network mechanism in anhedonia and depression. PLOS One 2025, 20. [Google Scholar] [CrossRef]
- Wu, C.; Mu, Q.; Gao, W.; Lu, S. The characteristics of anhedonia in depression: a review from a clinically oriented perspective. Translational Psychiatry 2025, 15. [Google Scholar] [CrossRef]
- Lee, S.; Bae, S.-M. The Effect of Anhedonia on Suicidal Ideation: The Moderated Mediation Effect of Emotional Intelligence Through Loneliness. Psychiatry Investigation 2025, 22, 591–601. [Google Scholar] [CrossRef]
- Serretti, A. Anhedonia: Current and future treatments. Psychiatry and Clinical Neurosciences Reports 2025, 4. [Google Scholar] [CrossRef] [PubMed]
- Hoflich, A.; Michenthaler, P.; Kasper, S.; Lanzenberger, R. Circuit Mechanisms of Reward, Anhedonia, and Depression. Int J Neuropsychopharmacol 2019, 22, 105–118. [Google Scholar] [CrossRef]
- Wallace, C.W.; Holleran, K.M.; Slinkard, C.Y.; Centanni, S.W.; Lapish, C.C.; Jones, S.R. Kappa opioid receptors diminish spontaneous dopamine signals in awake mice through multiple mechanisms. Neuropharmacology 2025, 273. [Google Scholar] [CrossRef]
- Martinez Damonte, V.; Bailey, L.G.; Thakar, A.; Stralka, J.; Brown, T.E.; Kauer, J.A. Kappa Opioid Receptors Control a Stress-Sensitive Brain Circuit and Drive Cocaine Seeking. The Journal of Neuroscience 2025, 45. [Google Scholar] [CrossRef] [PubMed]
- Escobar, A.d.P.; Casanova, J.P.; Andrés, M.E.; Fuentealba, J.A. Crosstalk Between Kappa Opioid and Dopamine Systems in Compulsive Behaviors. Frontiers in Pharmacology 2020, 11. [Google Scholar] [CrossRef]
- Shamakina, I.Y.; Shagiakhmetov, F.S.; Anokhin, P.K.; Kohan, V.S.; Davidova, T.V. The Role of Nociceptin in Opioid Regulation of Brain Functions. Biochemistry (Moscow), Supplement Series B: Biomedical Chemistry 2021, 15, 171–183. [Google Scholar] [CrossRef]
- Dalefield, M.L.; Scouller, B.; Bibi, R.; Kivell, B.M. The Kappa Opioid Receptor: A Promising Therapeutic Target for Multiple Pathologies. Frontiers in Pharmacology 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Koob, G.F. Addiction is a Reward Deficit and Stress Surfeit Disorder. Frontiers in Psychiatry 2013, 4. [Google Scholar] [CrossRef]
- Jiang, Y.; Zou, D.; Li, Y.; Gu, S.; Dong, J.; Ma, X.; Xu, S.; Wang, F.; Huang, J.H. Monoamine Neurotransmitters Control Basic Emotions and Affect Major Depressive Disorders. Pharmaceuticals 2022, 15. [Google Scholar] [CrossRef] [PubMed]
- Martín-Hernández, D.; Pereira, M.P.; Tendilla-Beltrán, H.; Madrigal, J.L.M.; García-Bueno, B.; Leza, J.C.; Caso, J.R. Modulation of Monoaminergic Systems by Antidepressants in the Frontal Cortex of Rats After Chronic Mild Stress Exposure. Molecular Neurobiology 2019, 56, 7522–7533. [Google Scholar] [CrossRef] [PubMed]
- Dresp-Langley, B. From Reward to Anhedonia-Dopamine Function in the Global Mental Health Context. Biomedicines 2023, 11. [Google Scholar] [CrossRef]
- Lalanne, L.; Ayranci, G.; Kieffer, B.L.; Lutz, P.-E. The Kappa Opioid Receptor: From Addiction to Depression, and Back. Frontiers in Psychiatry 2014, 5. [Google Scholar] [CrossRef]
- Wong, S.; Le, G.H.; Vasudeva, S.; Teopiz, K.M.; Phan, L.; Meshkat, S.; Kwan, A.T.H.; Rhee, T.G.; Ho, R.; Choi, H.; et al. Preclinical and clinical efficacy of kappa opioid receptor antagonists for depression: A systematic review. Journal of Affective Disorders 2024, 362, 816–827. [Google Scholar] [CrossRef]
- Stevens, C.W. Bioinformatics and Evolution of Vertebrate Nociceptin and Opioid Receptors. In Nociceptin Opioid; Vitamins & Hormones, 2015; pp. 57–94. [Google Scholar]
- Katzman, M.; Sternat, T. Neurobiology of hedonic tone: the relationship between treatment-resistant depression, attention-deficit hyperactivity disorder, and substance abuse. Neuropsychiatric Disease and Treatment 2016, Volume 12, 2149–2164. [Google Scholar] [CrossRef] [PubMed]
- Pizzagalli, D.A. Toward a Better Understanding of the Mechanisms and Pathophysiology of Anhedonia: Are We Ready for Translation? American Journal of Psychiatry 2022, 179, 458–469. [Google Scholar] [CrossRef]
- Liang, S.; Wu, Y.; Hanxiaoran, L.; Greenshaw, A.J.; Li, T. Anhedonia in Depression and Schizophrenia: Brain Reward and Aversion Circuits. Neuropsychiatric Disease and Treatment 2022, Volume 18, 1385–1396. [Google Scholar] [CrossRef]
- Cooper, J.A.; Arulpragasam, A.R.; Treadway, M.T. Anhedonia in depression: biological mechanisms and computational models. Current Opinion in Behavioral Sciences 2018, 22, 128–135. [Google Scholar] [CrossRef]
- Janiri, L.; Giannantonio, M.D.; Martinotti, G.; Hatzigiakoumis, D.S. Anhedonia and Substance Dependence: Clinical Correlates and Treatment Options. Frontiers in Psychiatry 2011, 2. [Google Scholar] [CrossRef] [PubMed]
- Uher, R.; Perlis, R.H.; Henigsberg, N.; Zobel, A.; Rietschel, M.; Mors, O.; Hauser, J.; Dernovsek, M.Z.; Souery, D.; Bajs, M.; et al. Depression symptom dimensions as predictors of antidepressant treatment outcome: replicable evidence for interest-activity symptoms. Psychological Medicine 2011, 42, 967–980. [Google Scholar] [CrossRef]
- McMakin, D.L.; Olino, T.M.; Porta, G.; Dietz, L.J.; Emslie, G.; Clarke, G.; Wagner, K.D.; Asarnow, J.R.; Ryan, N.D.; Birmaher, B.; et al. Anhedonia Predicts Poorer Recovery Among Youth With Selective Serotonin Reuptake Inhibitor Treatment–Resistant Depression. Journal of the American Academy of Child & Adolescent Psychiatry 2012, 51, 404–411. [Google Scholar] [CrossRef]
- Dunlop, K.; Rizvi, S.J.; Kennedy, S.H.; Hassel, S.; Strother, S.C.; Harris, J.K.; Zamyadi, M.; Arnott, S.R.; Davis, A.D.; Mansouri, F.; et al. Clinical, behavioral, and neural measures of reward processing correlate with escitalopram response in depression: a Canadian Biomarker Integration Network in Depression (CAN-BIND-1) Report. Neuropsychopharmacology 2020, 45, 1390–1397. [Google Scholar] [CrossRef] [PubMed]
- Kiluk, B.D.; Yip, S.W.; DeVito, E.E.; Carroll, K.M.; Sofuoglu, M. Anhedonia as a Key Clinical Feature in the Maintenance and Treatment of Opioid Use Disorder. Clinical Psychological Science 2019, 7, 1190–1206. [Google Scholar] [CrossRef]
- Parvaz, M.A.; Gabbay, V.; Malaker, P.; Goldstein, R.Z. Objective and specific tracking of anhedonia via event-related potentials in individuals with cocaine use disorders. Drug and Alcohol Dependence 2016, 164, 158–165. [Google Scholar] [CrossRef]
- Vafaie, N.; Kober, H. Association of Drug Cues and Craving With Drug Use and Relapse. JAMA Psychiatry 2022, 79. [Google Scholar] [CrossRef]
- Sierra, M.; David, A.S. Depersonalization: A selective impairment of self-awareness. Consciousness and Cognition 2011, 20, 99–108. [Google Scholar] [CrossRef]
- Sass, L.; Feyaerts, J. Self-Disorder in Schizophrenia: A Revised View (2. Theoretical Revision – Hyperreflexivity). Schizophrenia Bulletin 2024, 50, 472–483. [Google Scholar] [CrossRef]
- Prasko, J.; Grambal, A.; Kasalova, P.; Kamaradova, D.; Ociskova, M.; Holubova, M.; Vrbova, K.; Sigmundova, Z.; Latalova, K.; Slepecky, M.; et al. Impact of dissociation on treatment of depressive and anxiety spectrum disorders with and without personality disorders. Neuropsychiatric Disease and Treatment 2016, Volume 12, 2659–2676. [Google Scholar] [CrossRef] [PubMed]
- Prasko, J.; Ociskova, M.; Latalova, K.; Kamaradova, D.; Grambal, A. Psychological factors and treatment effectiveness in resistant anxiety disorders in highly comorbid inpatients. Neuropsychiatric Disease and Treatment 2016, Volume 12, 1539–1551. [Google Scholar] [CrossRef]
- Prasko, J.; Vyskocilova, J.; Sipek, J. Cognitive behavioral therapy in pharmacoresistant obsessive–compulsive disorder. Neuropsychiatric Disease and Treatment 2016. [Google Scholar] [CrossRef]
- Stevens, C.W. The evolution of vertebrate opioid receptors. Frontiers in Bioscience 2009, Volume. [Google Scholar] [CrossRef] [PubMed]
- Dreborg, S.; Sundström, G.; Larsson, T.A.; Larhammar, D. Evolution of vertebrate opioid receptors. Proceedings of the National Academy of Sciences 2008, 105, 15487–15492. [Google Scholar] [CrossRef]
- Larhammar, D.; Dreborg, S.; Larsson, T.A.; Sundström, G. Early Duplications of Opioid Receptor and Peptide Genes in Vertebrate Evolution. Annals of the New York Academy of Sciences 2009, 1163, 451–453. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh Sahafi, O.; Sardari, M.; Alijanpour, S.; Rezayof, A. Shared Mechanisms of GABAergic and Opioidergic Transmission Regulate Corticolimbic Reward Systems and Cognitive Aspects of Motivational Behaviors. Brain Sciences 2023, 13. [Google Scholar] [CrossRef]
- Margolis, E.B.; Karkhanis, A.N. Dopaminergic cellular and circuit contributions to kappa opioid receptor mediated aversion. Neurochemistry International 2019, 129. [Google Scholar] [CrossRef] [PubMed]
- Edwards, N.J.; Tejeda, H.A.; Pignatelli, M.; Zhang, S.; McDevitt, R.A.; Wu, J.; Bass, C.E.; Bettler, B.; Morales, M.; Bonci, A. Circuit specificity in the inhibitory architecture of the VTA regulates cocaine-induced behavior. Nature Neuroscience 2017, 20, 438–448. [Google Scholar] [CrossRef]
- Norton, C.S.; Neal, C.R.; Kumar, S.; Akil, H.; Watson, S.J. Nociceptin/orphanin FQ and opioid receptor-like receptor mRNA expression in dopamine systems. Journal of Comparative Neurology 2002, 444, 358–368. [Google Scholar] [CrossRef]
- Khan, M.S.; Boileau, I.; Kolla, N.; Mizrahi, R. A systematic review of the role of the nociceptin receptor system in stress, cognition, and reward: relevance to schizophrenia. Translational Psychiatry 2018, 8. [Google Scholar] [CrossRef]
- Di Benedetto, M.; Cavina, C.; D’Addario, C.; Leoni, G.; Candeletti, S.; Cox, B.M.; Romualdi, P. Alterations of N/OFQ and NOP receptor gene expression in the substantia nigra and caudate putamen of MPP+ and 6-OHDA lesioned rats. Neuropharmacology 2009, 56, 761–767. [Google Scholar] [CrossRef]
- Gavioli, E.C.; Holanda, V.A.D.; Calo, G.; Ruzza, C. Nociceptin/orphanin FQ receptor system blockade as an innovative strategy for increasing resilience to stress. Peptides 2021, 141. [Google Scholar] [CrossRef]
- Olianas, M.C.; Dedoni, S.; Boi, M.; Onali, P. Activation of nociceptin/orphanin FQ-NOP receptor system inhibits tyrosine hydroxylase phosphorylation, dopamine synthesis, and dopamine D1 receptor signaling in rat nucleus accumbens and dorsal striatum. Journal of Neurochemistry 2008, 107, 544–556. [Google Scholar] [CrossRef] [PubMed]
- Marti, M.; Mela, F.; Veronesi, C.; Guerrini, R.; Salvadori, S.; Federici, M.; Mercuri, N.B.; Rizzi, A.; Franchi, G.; Beani, L.; et al. Blockade of Nociceptin/Orphanin FQ Receptor Signaling in Rat Substantia Nigra Pars Reticulata Stimulates Nigrostriatal Dopaminergic Transmission and Motor Behavior. The Journal of Neuroscience 2004, 24, 6659–6666. [Google Scholar] [CrossRef] [PubMed]
- Volta, M.; Viaro, R.; Trapella, C.; Marti, M.; Morari, M. Dopamine–nociceptin/orphanin FQ interactions in the substantia nigra reticulata of hemiparkinsonian rats: Involvement of D2/D3 receptors and impact on nigro-thalamic neurons and motor activity. Experimental Neurology 2011, 228, 126–137. [Google Scholar] [CrossRef]
- Soto, D.C.; Uribe-Salazar, J.M.; Kaya, G.; Valdarrago, R.; Sekar, A.; Haghani, N.K.; Hino, K.; La, G.; Mariano, N.A.F.; Ingamells, C.; et al. Human-specific gene expansions contribute to brain evolution. Cell 2025, 188, 5363–5383.e5322. [Google Scholar] [CrossRef] [PubMed]
- Venkatesh, A.; Quinn, N.; Upadhya, S.R.; De Kegel, B.; Bolado Carrancio, A.; Lefeivre, T.; Dennler, O.; Wynne, K.; von Kriegsheim, A.; Ryan, C.J. Proteomic compensation by paralogs preserves protein interaction networks after gene loss in cancer. Molecular Systems Biology 2025, 21, 1090–1118. [Google Scholar] [CrossRef]
- Ng, A.Y.E.; Chan, S.N.; Pek, J.W. Genetic compensation between ribosomal protein paralogs mediated by a cognate circular RNA. Cell Reports 2024, 43. [Google Scholar] [CrossRef]
- Xin, Y.; Zhang, Y. Paralog-based synthetic lethality: rationales and applications. Frontiers in Oncology 2023, 13. [Google Scholar] [CrossRef]
- Järving, R.; Järving, I.; Kurg, R.; Brash, A.R.; Samel, N. On the Evolutionary Origin of Cyclooxygenase (COX) Isozymes. Journal of Biological Chemistry 2004, 279, 13624–13633. [Google Scholar] [CrossRef]
- Yang-Feng, T.L.; Xue, F.Y.; Zhong, W.W.; Cotecchia, S.; Frielle, T.; Caron, M.G.; Lefkowitz, R.J.; Francke, U. Chromosomal organization of adrenergic receptor genes. Proceedings of the National Academy of Sciences 1990, 87, 1516–1520. [Google Scholar] [CrossRef] [PubMed]
- Robinson-Rechavi, M.; Carrigan, M.A.; Uryasev, O.; Davis, R.P.; Zhai, L.; Hurley, T.D.; Benner, S.A. The Natural History of Class I Primate Alcohol Dehydrogenases Includes Gene Duplication, Gene Loss, and Gene Conversion. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Shagiakhmetov, F.; Anokhin, P.; Shamakina, I.; Davydova, T. Comparative quantitative analysis of the brain opioid gene expression in rats with high and low voluntary alcohol consumption. ZHurnal «Patologicheskaia fiziologiia i eksperimental`naia terapiia» 2018, 53–57. [Google Scholar] [CrossRef]
- Shagiakhmetov, F.; Shamakina, I.; Davydova, T. Mechanisms underlying controlled alcohol consumption: nociceptine/kappa-opioid related gene expression in the rat brain. Nauchno-prakticheskii zhurnal «Patogenez» 2018, 112–114. [Google Scholar] [CrossRef]
- Maqueda, A.E.; Valle, M.; Addy, P.H.; Antonijoan, R.M.; Puntes, M.; Coimbra, J.; Ballester, M.R.; Garrido, M.; González, M.; Claramunt, J.; et al. Salvinorin-A Induces Intense Dissociative Effects, Blocking External Sensory Perception and Modulating Interoception and Sense of Body Ownership in Humans. International Journal of Neuropsychopharmacology 2015, 18. [Google Scholar] [CrossRef] [PubMed]
- MacLean, K.A.; Johnson, M.W.; Reissig, C.J.; Prisinzano, T.E.; Griffiths, R.R. Dose-related effects of salvinorin A in humans: dissociative, hallucinogenic, and memory effects. Psychopharmacology 2012, 226, 381–392. [Google Scholar] [CrossRef]
- Ranganathan, M.; Schnakenberg, A.; Skosnik, P.D.; Cohen, B.M.; Pittman, B.; Sewell, R.A.; D’Souza, D.C. Dose-Related Behavioral, Subjective, Endocrine, and Psychophysiological Effects of the κ Opioid Agonist Salvinorin A in Humans. Biological Psychiatry 2012, 72, 871–879. [Google Scholar] [CrossRef]
- Carlezon, W.A.; Chartoff, E.H. Intracranial self-stimulation (ICSS) in rodents to study the neurobiology of motivation. Nature Protocols 2007, 2, 2987–2995. [Google Scholar] [CrossRef] [PubMed]
- Carlezon, W.A.; Béguin, C.; DiNieri, J.A.; Baumann, M.H.; Richards, M.R.; Todtenkopf, M.S.; Rothman, R.B.; Ma, Z.; Lee, D.Y.W.; Cohen, B.M. Depressive-Like Effects of the κ-Opioid Receptor Agonist Salvinorin A on Behavior and Neurochemistry in Rats. The Journal of Pharmacology and Experimental Therapeutics 2006, 316, 440–447. [Google Scholar] [CrossRef]
- Carlezon, W.A.; Béguin, C.; Knoll, A.T.; Cohen, B.M. Kappa-opioid ligands in the study and treatment of mood disorders. Pharmacology & Therapeutics 2009, 123, 334–343. [Google Scholar] [CrossRef]
- Todtenkopf, M.S.; Marcus, J.F.; Portoghese, P.S.; Carlezon, W.A. Effects of κ-opioid receptor ligands on intracranial self-stimulation in rats. Psychopharmacology 2004, 172, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Wadenberg, M.L. A review of the properties of spiradoline: a potent and selective kappa-opioid receptor agonist. CNS Drug Rev 2003, 9, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Chappell, P.B.; Leckman, J.F.; Scahill, L.D.; Hardin, M.T.; Anderson, G.; Cohen, D.J. Neuroendocrine and behavioral effects of the selective kappa agonist spiradoline in Tourette’s syndrome: A pilot study. Psychiatry Research 1993, 47, 267–280. [Google Scholar] [CrossRef]
- Walsh, S.L.; Strain, E.C.; Abreu, M.E.; Bigelow, G.E. Enadoline, a selective kappa opioid agonist: comparison with butorphanol and hydromorphone in humans. Psychopharmacology (Berl) 2001, 157, 151–162. [Google Scholar] [CrossRef]
- Ur, E.; Wright, D.M.; Bouloux, P.M.G.; Grossman, A. The effects of spiradoline (U-62066E), a κ-opioid receptor agonist, on neuroendocrine function in man. British Journal of Pharmacology 2009, 120, 781–784. [Google Scholar] [CrossRef]
- White, K.L.; Roth, B.L. Psychotomimetic Effects of Kappa Opioid Receptor Agonists. Biological Psychiatry 2012, 72, 797–798. [Google Scholar] [CrossRef] [PubMed]
- Chefer, V.I.; Bäckman, C.M.; Gigante, E.D.; Shippenberg, T.S. Kappa Opioid Receptors on Dopaminergic Neurons Are Necessary for Kappa-Mediated Place Aversion. Neuropsychopharmacology 2013, 38, 2623–2631. [Google Scholar] [CrossRef]
- Wallace, C.W.; Holleran, K.M.; Slinkard, C.Y.; Centanni, S.W.; Jones, S.R. 2024. [CrossRef]
- Thompson, A.C.; Zapata, A.; Justice, J.B.; Vaughan, R.A.; Sharpe, L.G.; Shippenberg, T.S. κ-Opioid Receptor Activation Modifies Dopamine Uptake in the Nucleus Accumbens and Opposes the Effects of Cocaine. The Journal of Neuroscience 2000, 20, 9333–9340. [Google Scholar] [CrossRef]
- Kivell, B.; Uzelac, Z.; Sundaramurthy, S.; Rajamanickam, J.; Ewald, A.; Chefer, V.; Jaligam, V.; Bolan, E.; Simonson, B.; Annamalai, B.; et al. Salvinorin A regulates dopamine transporter function via a kappa opioid receptor and ERK1/2-dependent mechanism. Neuropharmacology 2014, 86, 228–240. [Google Scholar] [CrossRef]
- Tejeda, H.A.; Bonci, A. Dynorphin/kappa-opioid receptor control of dopamine dynamics: Implications for negative affective states and psychiatric disorders. Brain Research 2019, 1713, 91–101. [Google Scholar] [CrossRef]
- Trifilieff, P.; Martinez, D. Kappa-Opioid Receptor Signaling in the Striatum as a Potential Modulator of Dopamine Transmission in Cocaine Dependence. Frontiers in Psychiatry 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Korotkova, T.M.; Brown, R.E.; Sergeeva, O.A.; Ponomarenko, A.A.; Haas, H.L. Effects of arousal- and feeding-related neuropeptides on dopaminergic and GABAergic neurons in the ventral tegmental area of the rat. European Journal of Neuroscience 2006, 23, 2677–2685. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.J.; Lobo, M.K.; Spencer, S.; Kalivas, P.W. Cocaine-induced adaptations in D1 and D2 accumbens projection neurons (a dichotomy not necessarily synonymous with direct and indirect pathways). Current Opinion in Neurobiology 2013, 23, 546–552. [Google Scholar] [CrossRef]
- Margolis, E.B.; Lock, H.; Chefer, V.I.; Shippenberg, T.S.; Hjelmstad, G.O.; Fields, H.L. κ opioids selectively control dopaminergic neurons projecting to the prefrontal cortex. Proceedings of the National Academy of Sciences 2006, 103, 2938–2942. [Google Scholar] [CrossRef]
- Spanagel, R.; Herz, A.; Shippenberg, T.S. Opposing tonically active endogenous opioid systems modulate the mesolimbic dopaminergic pathway. Proceedings of the National Academy of Sciences 1992, 89, 2046–2050. [Google Scholar] [CrossRef]
- Devine, D.P.; Leone, P.; Pocock, D.; Wise, R.A. Differential involvement of ventral tegmental mu, delta and kappa opioid receptors in modulation of basal mesolimbic dopamine release: in vivo microdialysis studies. J Pharmacol Exp Ther 1993, 266, 1236–1246. [Google Scholar] [CrossRef]
- Ciccocioppo, R.; Borruto, A.M.; Domi, A.; Teshima, K.; Cannella, N.; Weiss, F. NOP-Related Mechanisms in Substance Use Disorders. In The Nociceptin/Orphanin FQ Peptide Receptor; Handbook of Experimental Pharmacology, 2019; pp. 187–212. [Google Scholar]
- Sakoori, K.; Murphy, N.P. Central administration of nociceptin/orphanin FQ blocks the acquisition of conditioned place preference to morphine and cocaine, but not conditioned place aversion to naloxone in mice. Psychopharmacology 2004, 172, 129–136. [Google Scholar] [CrossRef]
- Sakoori, K.; Murphy, N.P. Endogenous Nociceptin (Orphanin FQ) Suppresses Basal Hedonic State and Acute Reward Responses to Methamphetamine and Ethanol, but Facilitates Chronic Responses. Neuropsychopharmacology 2007, 33, 877–891. [Google Scholar] [CrossRef] [PubMed]
- Murphy, N.P. Nociceptin/orphanin FQ, hedonic state and the response to abused drugs. Nihon Shinkei Seishin Yakurigaku Zasshi 2004, 24, 295–298. [Google Scholar]
- Rutten, K.; De Vry, J.; Bruckmann, W.; Tzschentke, T.M. Effects of the NOP receptor agonist Ro65-6570 on the acquisition of opiate- and psychostimulant-induced conditioned place preference in rats. Eur J Pharmacol 2010, 645, 119–126. [Google Scholar] [CrossRef]
- Vazquez-DeRose, J.; Stauber, G.; Khroyan, T.V.; Xie, X.S.; Zaveri, N.T.; Toll, L. Retrodialysis of N/OFQ into the nucleus accumbens shell blocks cocaine-induced increases in extracellular dopamine and locomotor activity. European Journal of Pharmacology 2013, 699, 200–206. [Google Scholar] [CrossRef]
- Lutfy, K.; Zaveri, N.T. The Nociceptin Receptor as an Emerging Molecular Target for Cocaine Addiction. In The Molecular Basis of Drug Addiction; Progress in Molecular Biology and Translational Science, 2016; pp. 149–181. [Google Scholar]
- Zaveri, N.T. Nociceptin Opioid Receptor (NOP) as a Therapeutic Target: Progress in Translation from Preclinical Research to Clinical Utility. Journal of Medicinal Chemistry 2016, 59, 7011–7028. [Google Scholar] [CrossRef]
- Zaveri, N.T.; Marquez, P.V.; Meyer, M.E.; Polgar, W.E.; Hamid, A.; Lutfy, K. A Novel and Selective Nociceptin Receptor (NOP) Agonist (1-(1-((cis)-4-isopropylcyclohexyl)piperidin-4-yl)-1H-indol-2-yl)methanol (AT-312) Decreases Acquisition of Ethanol-Induced Conditioned Place Preference in Mice. Alcoholism: Clinical and Experimental Research 2018, 42, 461–471. [Google Scholar] [CrossRef]
- Devine, D.P.; Reinscheid, R.K.; Monsma, F.J.; Civelli, O.; Akil, H. The novel neuropeptide orphanin FQ fails to produce conditioned place preference or aversion. Brain Research 1996, 727, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Parker, K.E.; Pedersen, C.E.; Gomez, A.M.; Spangler, S.M.; Walicki, M.C.; Feng, S.Y.; Stewart, S.L.; Otis, J.M.; Al-Hasani, R.; McCall, J.G.; et al. A Paranigral VTA Nociceptin Circuit that Constrains Motivation for Reward. Cell 2019, 178, 653–671.e619. [Google Scholar] [CrossRef]
- Iturra-Mena, A.M.; Kangas, B.D.; Pizzagalli, D.A. Nociceptin Receptor Antagonism Modulates Electrophysiological Markers of Reward Learning. International Journal of Neuropsychopharmacology 2023, 26, 496–500. [Google Scholar] [CrossRef]
- Pizzagalli, D.A.; Treadway, M.T.; Kangas, B.D.; Romoli, B.; Breton, J.; Bruchas, M.R.; Graybiel, A.M.; Hueske, E.; Prakash, N.; Der-Avakian, A.; et al. 2025. [CrossRef] [PubMed]
- Charney, D.S.; Manji, H.K. Life Stress, Genes, and Depression: Multiple Pathways Lead to Increased Risk and New Opportunities for Intervention. Science’s STKE 2004 2004. [Google Scholar] [CrossRef] [PubMed]
- Bruchas, M.R.; Land, B.B.; Chavkin, C. The dynorphin/kappa opioid system as a modulator of stress-induced and pro-addictive behaviors. Brain Research 2010, 1314, 44–55. [Google Scholar] [CrossRef]
- Marchette, R.C.N.; Vendruscolo, L.F.; Koob, G.F. The Dynorphin/-Opioid Receptor System at the Interface of Hyperalgesia/Hyperkatifeia and Addiction. Current Addiction Reports 2025, 12. [Google Scholar] [CrossRef]
- Koob, G.F.; Volkow, N.D. Neurocircuitry of Addiction. Neuropsychopharmacology 2009, 35, 217–238. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, J.P.; Marton-Popovici, M.; Chavkin, C. κ Opioid Receptor Antagonism and Prodynorphin Gene Disruption Block Stress-Induced Behavioral Responses. The Journal of Neuroscience 2003, 23, 5674–5683. [Google Scholar] [CrossRef]
- Beardsley, P.M.; Howard, J.L.; Shelton, K.L.; Carroll, F.I. Differential effects of the novel kappa opioid receptor antagonist, JDTic, on reinstatement of cocaine-seeking induced by footshock stressors vs cocaine primes and its antidepressant-like effects in rats. Psychopharmacology 2005, 183, 118–126. [Google Scholar] [CrossRef]
- Redila, V.A.; Chavkin, C. Stress-induced reinstatement of cocaine seeking is mediated by the kappa opioid system. Psychopharmacology 2008, 200, 59–70. [Google Scholar] [CrossRef]
- Câmara, A.B.; Brandão, I.A. The neuroinflammatory effects of Nociceptin/Orphanin FQ receptor activation can be related to depressive-like behavior. Journal of Psychiatric Research 2025, 183, 174–188. [Google Scholar] [CrossRef]
- Holanda, V.A.D.; Oliveira, M.C.; Da Silva Junior, E.D.; Calo, G.; Ruzza, C.; Gavioli, E.C. Blockade of nociceptin/orphanin FQ signaling facilitates an active copying strategy due to acute and repeated stressful stimuli in mice. Neurobiology of Stress 2020, 13. [Google Scholar] [CrossRef]
- Vitale, G.; Ruggieri, V.; Filaferro, M.; Frigeri, C.; Alboni, S.; Tascedda, F.; Brunello, N.; Guerrini, R.; Cifani, C.; Massi, M. Chronic treatment with the selective NOP receptor antagonist [Nphe1,Arg14,Lys15]N/OFQ-NH2 (UFP-101) reverses the behavioural and biochemical effects of unpredictable chronic mild stress in rats. Psychopharmacology 2009, 207, 173–189. [Google Scholar] [CrossRef] [PubMed]
- Logrip, M.L.; Janak, P.H.; Ron, D. Blockade of ethanol reward by the kappa opioid receptor agonist U50,488H. Alcohol 2009, 43, 359–365. [Google Scholar] [CrossRef]
- Anderson, R.I.; Becker, H.C. Role of the Dynorphin/Kappa Opioid Receptor System in the Motivational Effects of Ethanol. Alcoholism: Clinical and Experimental Research 2017, 41, 1402–1418. [Google Scholar] [CrossRef]
- Wee, S.; Koob, G.F. The role of the dynorphin–κ opioid system in the reinforcing effects of drugs of abuse. Psychopharmacology 2010, 210, 121–135. [Google Scholar] [CrossRef]
- Glick, S.D.; Maisonneuve, I.M.; Raucci, J.; Sydney, A. Kappa opioid inhibition of morphine and cocaine self-administration in rats. Brain Research 1995, 681, 147–152. [Google Scholar] [CrossRef]
- Schenk, S.; Shippenberg, T.S.; Partridge, B. U69593, a kappa-opioid agonist, decreases cocaine self-administration and decreases cocaine-produced drug-seeking. Psychopharmacology 1999, 144, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Mello, N.K.; Negus, S.S. Effects of kappa opioid agonists on cocaine- and food-maintained responding by rhesus monkeys. J Pharmacol Exp Ther 1998, 286, 812–824. [Google Scholar] [CrossRef]
- Valdez, G.R.; Platt, D.M.; Rowlett, J.K.; Rüedi-Bettschen, D.; Spealman, R.D. κ Agonist-Induced Reinstatement of Cocaine Seeking in Squirrel Monkeys: A Role for Opioid and Stress-Related Mechanisms. The Journal of Pharmacology and Experimental Therapeutics 2007, 323, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Hölter, S.M.; Henniger, M.S.H.; Lipkowski, A.W.; Spanagel, R. Kappa-opioid receptors and relapse-like drinking in long-term ethanol-experienced rats. Psychopharmacology 2000, 153, 93–102. [Google Scholar] [CrossRef]
- Walker, B.M.; Zorrilla, E.P.; Koob, G.F. Systemic κ-opioid receptor antagonism by nor-binaltorphimine reduces dependence-induced excessive alcohol self-administration in rats. Addiction Biology 2011, 16, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Aujla, H.; Cannarsa, R.; Romualdi, P.; Ciccocioppo, R.; Martin-Fardon, R.; Weiss, F. Modification of anxiety-like behaviors by nociceptin/orphanin FQ (N/OFQ) and time-dependent changes in N/OFQ-NOP gene expression following ethanol withdrawal. Addiction Biology 2012, 18, 467–479. [Google Scholar] [CrossRef]
- Anokhin, P.K.; Shagiakhmetov, F.S.; Kokhan, V.S.; Tarabarko, I.E.; Proskuryakova, T.V.; Shokhonova, V.A.; Shamakina, I.Y. PREPRONOCICEPTIN AND NOCICEPTIN RECEPTOR mRNA EXPRESSION IN THE BRAIN OF RATS WITH PROGRESSIVE INCREASE IN VOLUNTARY ALCOHOL CONSUMPTION. Voprosy narkologii 2020, 22–35. [Google Scholar] [CrossRef]
- McIntyre, R.S.; Alsuwaidan, M.; Baune, B.T.; Berk, M.; Demyttenaere, K.; Goldberg, J.F.; Gorwood, P.; Ho, R.; Kasper, S.; Kennedy, S.H.; et al. Treatment-resistant depression: definition, prevalence, detection, management, and investigational interventions. World Psychiatry 2023, 22, 394–412. [Google Scholar] [CrossRef]
- Krystal, A.D.; Pizzagalli, D.A.; Smoski, M.; Mathew, S.J.; Nurnberger, J.; Lisanby, S.H.; Iosifescu, D.; Murrough, J.W.; Yang, H.; Weiner, R.D.; et al. A randomized proof-of-mechanism trial applying the ‘fast-fail’ approach to evaluating κ-opioid antagonism as a treatment for anhedonia. Nature Medicine 2020, 26, 760–768. [Google Scholar] [CrossRef]
- Schmidt, M.E.; Kezic, I.; Popova, V.; Melkote, R.; Van Der Ark, P.; Pemberton, D.J.; Mareels, G.; Canuso, C.M.; Fava, M.; Drevets, W.C. Efficacy and safety of aticaprant, a kappa receptor antagonist, adjunctive to oral SSRI/SNRI antidepressant in major depressive disorder: results of a phase 2 randomized, double-blind, placebo-controlled study. Neuropsychopharmacology 2024, 49, 1437–1447. [Google Scholar] [CrossRef] [PubMed]
- Hampsey, E.; Jelen, L.; Young, A.H. Aticaprant: (a κ-opioid receptor antagonist) for major depressive disorder. Expert Opinion on Emerging Drugs 2024, 29, 193–204. [Google Scholar] [CrossRef]
- Mathew, S.J.; Cutler, A.J.; Visitacion, N.C.; Gold, M.; Yuan, J.; Aurora, B. Navacaprant, a Novel and Highly Selective Kappa Opioid Receptor Antagonist, in Adults With Major Depressive Disorder. Journal of Clinical Psychopharmacology 2025, 45, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Gerra, G.; Fantoma, A.; Zaimovic, A. Naltrexone and buprenorphine combination in the treatment of opioid dependence. Journal of Psychopharmacology 2006, 20, 806–814. [Google Scholar] [CrossRef] [PubMed]
- Ling, W.; Hillhouse, M.P.; Saxon, A.J.; Mooney, L.J.; Thomas, C.M.; Ang, A.; Matthews, A.G.; Hasson, A.; Annon, J.; Sparenborg, S.; et al. Buprenorphine + naloxone plus naltrexone for the treatment of cocaine dependence: the Cocaine Use Reduction with Buprenorphine (CURB) study. Addiction 2016, 111, 1416–1427. [Google Scholar] [CrossRef]
- Fava, M.; Memisoglu, A.; Thase, M.E.; Bodkin, J.A.; Trivedi, M.H.; de Somer, M.; Du, Y.; Leigh-Pemberton, R.; DiPetrillo, L.; Silverman, B.; et al. Opioid Modulation With Buprenorphine/Samidorphan as Adjunctive Treatment for Inadequate Response to Antidepressants: A Randomized Double-Blind Placebo-Controlled Trial. American Journal of Psychiatry 2016, 173, 499–508. [Google Scholar] [CrossRef]
- Zajecka, J.M.; Stanford, A.D.; Memisoglu, A.; Martin, W.F.; Pathak, S. Buprenorphine/samidorphan combination for the adjunctive treatment of major depressive disorder: results of a phase III clinical trial (FORWARD-3). Neuropsychiatric Disease and Treatment 2019, Volume 15, 795–808. [Google Scholar] [CrossRef]
- Fava, M.; Thase, M.E.; Trivedi, M.H.; Ehrich, E.; Martin, W.F.; Memisoglu, A.; Nangia, N.; Stanford, A.D.; Yu, M.; Pathak, S. Opioid system modulation with buprenorphine/samidorphan combination for major depressive disorder: two randomized controlled studies. Molecular Psychiatry 2018, 25, 1580–1591. [Google Scholar] [CrossRef]
- Fava, M. How should we design future mechanistic and/or efficacy clinical trials? Neuropsychopharmacology 2023, 49, 197–204. [Google Scholar] [CrossRef]
- Wager, T.D.; Atlas, L.Y. The neuroscience of placebo effects: connecting context, learning and health. Nature Reviews Neuroscience 2015, 16, 403–418. [Google Scholar] [CrossRef]
- Merlo Pich, E. Placebo response in RCT for antidepressant may not always be the ‘villain’ to fight: are KOR antagonists possibly affecting the intrinsic placebo response? Neuropsychopharmacology 2024, 49, 1355–1356. [Google Scholar] [CrossRef]
- Wang, L.N.; Liu, L.F.; Zhang, J.X.; Zhao, G.F. Plasma levels of nociceptin/orphanin FQ in patients with bipolar disorders and health adults. Zhonghua Yi Xue Za Zhi 2009, 89, 916–918. [Google Scholar]
- Gu, H.; Hu, D.; Hong, X.R.; Mao, J.; Cui, Y.; Hui, N.; Sha, J.Y. Changes and significance of orphanin and serotonin in patients with postpartum depression. Zhonghua Fu Chan Ke Za Zhi 2003, 38, 727–728. [Google Scholar] [PubMed]
- Andero, R.; Brothers, S.P.; Jovanovic, T.; Chen, Y.T.; Salah-Uddin, H.; Cameron, M.; Bannister, T.D.; Almli, L.; Stevens, J.S.; Bradley, B.; et al. Amygdala-Dependent Fear Is Regulated by.
- Oprl1.
- in Mice and Humans with PTSD. Science Translational Medicine 2013, 5. [CrossRef]
- Levey, D.F.; Gelernter, J.; Polimanti, R.; Zhou, H.; Cheng, Z.; Aslan, M.; Quaden, R.; Concato, J.; Radhakrishnan, K.; Bryois, J.; et al. Reproducible Genetic Risk Loci for Anxiety: Results From ∼200,000 Participants in the Million Veteran Program. American Journal of Psychiatry 2020, 177, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Flanigan, M.; Tollefson, S.; Himes, M.L.; Jordan, R.; Roach, K.; Stoughton, C.; Lopresti, B.; Mason, N.S.; Ciccocioppo, R.; Narendran, R. Acute Elevations in Cortisol Increase the In Vivo Binding of [11C]NOP-1A to Nociceptin Receptors: A Novel Imaging Paradigm to Study the Interaction Between Stress- and Antistress-Regulating Neuropeptides. Biological Psychiatry 2020, 87, 570–576. [Google Scholar] [CrossRef]
- Narendran, R.; Tollefson, S.; Fasenmyer, K.; Paris, J.; Himes, M.L.; Lopresti, B.; Ciccocioppo, R.; Mason, N.S. Decreased Nociceptin Receptors Are Related to Resilience and Recovery in College Women Who Have Experienced Sexual Violence: Therapeutic Implications for Posttraumatic Stress Disorder. Biological Psychiatry 2019, 85, 1056–1064. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, F.; Rizzo, S.; Ruzza, C.; Calo, G. Detailed In Vitro Pharmacological Characterization of the Clinically Viable Nociceptin/Orphanin FQ Peptide Receptor Antagonist BTRX-246040. The Journal of Pharmacology and Experimental Therapeutics 2020, 373, 34–43. [Google Scholar] [CrossRef]
- Toledo, M.A.; Pedregal, C.; Lafuente, C.; Diaz, N.; Martinez-Grau, M.A.; Jiménez, A.; Benito, A.; Torrado, A.; Mateos, C.; Joshi, E.M.; et al. Discovery of a Novel Series of Orally Active Nociceptin/Orphanin FQ (NOP) Receptor Antagonists Based on a Dihydrospiro(piperidine-4,7′-thieno [2,3-c]pyran) Scaffold. Journal of Medicinal Chemistry 2014, 57, 3418–3429. [Google Scholar] [CrossRef]
- Post, A.; Smart, T.S.; Krikke-Workel, J.; Dawson, G.R.; Harmer, C.J.; Browning, M.; Jackson, K.; Kakar, R.; Mohs, R.; Statnick, M.; et al. Erratum: A Selective Nociceptin Receptor Antagonist to Treat Depression: Evidence from Preclinical and Clinical Studies. Neuropsychopharmacology 2016, 41, 2624–2624. [Google Scholar] [CrossRef]
- Post, A.; Smart, T.S.; Jackson, K.; Mann, J.; Mohs, R.; Rorick-Kehn, L.; Statnick, M.; Anton, R.; O’Malley, S.S.; Wong, C.J. Proof-of-Concept Study to Assess the Nociceptin Receptor Antagonist LY2940094 as a New Treatment for Alcohol Dependence. Alcoholism: Clinical and Experimental Research 2016, 40, 1935–1944. [Google Scholar] [CrossRef] [PubMed]
- Thomas, J.M.; Higgs, S.; Dourish, C.T. Test–retest reliability and effects of repeated testing and satiety on performance of an Emotional Test Battery. Journal of Clinical and Experimental Neuropsychology 2015, 38, 416–433. [Google Scholar] [CrossRef] [PubMed]
- Harmer, C.J.; Dawson, G.R.; Dourish, C.T.; Favaron, E.; Parsons, E.; Fiore, M.; Zucchetto, M.; Bifone, A.; Poggesi, I.; Fernandes, S.; et al. Combined NK1 antagonism and serotonin reuptake inhibition: effects on emotional processing in humans. Journal of Psychopharmacology 2013, 27, 435–443. [Google Scholar] [CrossRef]
- Pike, A.C.; Robinson, O.J. Reinforcement Learning in Patients With Mood and Anxiety Disorders vs Control Individuals. JAMA Psychiatry 2022, 79. [Google Scholar] [CrossRef]
- Seifert, R.; Wenzel-Seifert, K. Constitutive activity of G-protein-coupled receptors: cause of disease and common property of wild-type receptors. Naunyn-Schmiedeberg’s Archives of Pharmacology 2002, 366, 381–416. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Furness, S.G.B. The nature of efficacy at G protein-coupled receptors. Biochemical Pharmacology 2019, 170. [Google Scholar] [CrossRef]
- Meye, F.J.; Ramakers, G.M.J.; Adan, R.A.H. The vital role of constitutive GPCR activity in the mesolimbic dopamine system. Translational Psychiatry 2014, 4, e361–e361. [Google Scholar] [CrossRef]
- Michel, M.C.; Michel-Reher, M.B.; Hein, P. A Systematic Review of Inverse Agonism at Adrenoceptor Subtypes. Cells 2020, 9. [Google Scholar] [CrossRef]
- Carroll, I.; Thomas, J.B.; Dykstra, L.A.; Granger, A.L.; Allen, R.M.; Howard, J.L.; Pollard, G.T.; Aceto, M.D.; Harris, L.S. Pharmacological properties of JDTic: a novel κ-opioid receptor antagonist. European Journal of Pharmacology 2004, 501, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Bruchas, M.R.; Yang, T.; Schreiber, S.; DeFino, M.; Kwan, S.C.; Li, S.; Chavkin, C. Long-Acting κ Opioid Antagonists Disrupt Receptor Signaling And Produce Noncompetitive Effects By Activating C-Jun N-Terminal Kinase. Journal of Biological Chemistry 2007, 282, 29803–29811. [Google Scholar] [CrossRef]
- Endoh, T.; Matsuura, H.; Tanaka, C.; Nagase, H. Nor-binaltorphimine: a potent and selective kappa-opioid receptor antagonist with long-lasting activity in vivo. Arch Int Pharmacodyn Ther 1992, 316, 30–42. [Google Scholar]
- Casal-Dominguez, J.J.; Clark, M.; Traynor, J.R.; Husbands, S.M.; Bailey, S.J. In vivo and in vitro characterization of naltrindole-derived ligands at the κ-opioid receptor. Journal of Psychopharmacology 2012, 27, 192–202. [Google Scholar] [CrossRef]
- Black, S.L.; Jales, A.R.; Brandt, W.; Lewis, J.W.; Husbands, S.M. The Role of the Side Chain in Determining Relative δ- and κ-Affinity in C5‘-Substituted Analogues of Naltrindole. Journal of Medicinal Chemistry 2002, 46, 314–317. [Google Scholar] [CrossRef]
- Olmsted, S.L.; Takemori, A.E.; Portoghese, P.S. A remarkable change of opioid receptor selectivity on the attachment of a peptidomimetic.kappa. address element to the.delta. antagonist, natrindole: 5’-[(N2-alkylamidino)methyl]naltrindole derivatives as a novel class of.kappa. opioid receptor antagonists. Journal of Medicinal Chemistry 2002, 36, 179–180. [Google Scholar] [CrossRef]
- Jales, A.R.; Husbands, S.M.; Lewis, J.W. Selective κ-opioid antagonists related to naltrindole. effect of side-chain spacer in the 5′-amidinoalkyl series. Bioorganic & Medicinal Chemistry Letters 2000, 10, 2259–2261. [Google Scholar] [CrossRef]
- Bruchas, M.R.; Land, B.B.; Aita, M.; Xu, M.; Barot, S.K.; Li, S.; Chavkin, C. Stress-Induced p38 Mitogen-Activated Protein Kinase Activation Mediates κ-Opioid-Dependent Dysphoria. The Journal of Neuroscience 2007, 27, 11614–11623. [Google Scholar] [CrossRef]
- Melief, E.J.; Miyatake, M.; Bruchas, M.R.; Chavkin, C. Ligand-directed c-Jun N-terminal kinase activation disrupts opioid receptor signaling. Proceedings of the National Academy of Sciences 2010, 107, 11608–11613. [Google Scholar] [CrossRef] [PubMed]
- Schattauer, S.S.; Bedini, A.; Summers, F.; Reilly-Treat, A.; Andrews, M.M.; Land, B.B.; Chavkin, C. Reactive oxygen species (ROS) generation is stimulated by κ opioid receptor activation through phosphorylated c-Jun N-terminal kinase and inhibited by p38 mitogen-activated protein kinase (MAPK) activation. Journal of Biological Chemistry 2019, 294, 16884–16896. [Google Scholar] [CrossRef] [PubMed]
- Schattauer, S.S.; Land, B.B.; Reichard, K.L.; Abraham, A.D.; Burgeno, L.M.; Kuhar, J.R.; Phillips, P.E.M.; Ong, S.E.; Chavkin, C. Peroxiredoxin 6 mediates Gαi protein-coupled receptor inactivation by cJun kinase. Nature Communications 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Neiswanger, C.; Ruiz, M.V.; Kimball, K.; Lee, J.D.; Land, B.B.; Berndt, A.; Chavkin, C. G Protein Inactivation as a Mechanism for Addiction Treatment. Biological Psychiatry 2026, 99, 80–90. [Google Scholar] [CrossRef]
- Sadée, W.; Wang, Z. Agonist Induced Constitutive Receptor Activation as a Novel Regulatory Mechanism. In The Brain Immune Axis and Substance Abuse; Advances in Experimental Medicine and Biology, 1995; pp. 85–90. [Google Scholar]
- Liu, J.-G.; Prather, P.L. Chronic Exposure to μ-Opioid Agonists Produces Constitutive Activation of μ-Opioid Receptors in Direct Proportion to the Efficacy of the Agonist Used for Pretreatment. Molecular Pharmacology 2001, 60, 53–62. [Google Scholar] [CrossRef]
- Polter, A.M.; Barcomb, K.; Chen, R.W.; Dingess, P.M.; Graziane, N.M.; Brown, T.E.; Kauer, J.A. Constitutive activation of kappa opioid receptors at ventral tegmental area inhibitory synapses following acute stress. eLife 2017, 6. [Google Scholar] [CrossRef]
- Graziane, Nicholas M.; Polter, Abigail M.; Briand, Lisa A.; Pierce, R.C.; Kauer, Julie A. Kappa Opioid Receptors Regulate Stress-Induced Cocaine Seeking and Synaptic Plasticity. Neuron 2013, 77, 942–954. [Google Scholar] [CrossRef] [PubMed]
- Grosse Hartlage, M.A.; Theisen, M.M.; Monteiro de Oliveira, N.P.; Van Aken, H.; Fobker, M.; Weber, T.P. ??-Opioid Receptor Antagonism Improves Recovery from Myocardial Stunning in Chronically Instrumented Dogs. Anesthesia & Analgesia 2006, 103, 822–832. [Google Scholar] [CrossRef] [PubMed]
- Chavkin, C.; Martinez, D. Kappa Antagonist JDTic in Phase 1 Clinical Trial. Neuropsychopharmacology 2015, 40, 2057–2058. [Google Scholar] [CrossRef]
- Tyson, A.S.; Khan, S.; Motiwala, Z.; Han, G.W.; Zhang, Z.; Ranjbar, M.; Styrpejko, D.; Ramos-Gonzalez, N.; Woo, S.; Villers, K.; et al. Molecular mechanisms of inverse agonism via κ-opioid receptor–G protein complexes. Nature Chemical Biology 2025, 21, 1046–1057. [Google Scholar] [CrossRef]
- An, X.; Bai, Q.; Bing, Z.; Zhou, S.; Shi, D.; Liu, H.; Yao, X. How Does Agonist and Antagonist Binding Lead to Different Conformational Ensemble Equilibria of the κ-Opioid Receptor: Insight from Long-Time Gaussian Accelerated Molecular Dynamics Simulation. ACS Chemical Neuroscience 2018, 10, 1575–1584. [Google Scholar] [CrossRef]
- Xu, Y.; Ye, H. Progress in understanding the mechanisms of resistance to BCL-2 inhibitors. Experimental Hematology & Oncology 2022, 11. [Google Scholar] [CrossRef]
- Murai, J.; Huang, S.-y.N.; Das, B.B.; Renaud, A.; Zhang, Y.; Doroshow, J.H.; Ji, J.; Takeda, S.; Pommier, Y. Trapping of PARP1 and PARP2 by Clinical PARP Inhibitors. Cancer Research 2012, 72, 5588–5599. [Google Scholar] [CrossRef] [PubMed]
- Eslambol Nassaj, A.; Nekouei, A.H.; Fereidooni, R.; Kamyabi, H.; Pardakhty, A.; Shahravan, A. Comparative Efficacy of Analgesics for Pain Relief in Patients with Symptomatic Irreversible Pulpitis Prior to Emergency Endodontic Treatment: A Randomized Controlled Trial. Iran Endod J 2023, 18, 194–201. [Google Scholar] [CrossRef]
- Zaveri, N.T.; Journigan, V.B.; Polgar, W.E. Discovery of the first small-molecule opioid pan antagonist with nanomolar affinity at mu, delta, kappa, and nociceptin opioid receptors. ACS Chem Neurosci 2015, 6, 646–657. [Google Scholar] [CrossRef] [PubMed]
- Krupitsky, E.; Zvartau, E.; Blokhina, E.; Verbitskaya, E.; Wahlgren, V.; Tsoy-Podosenin, M.; Bushara, N.; Burakov, A.; Masalov, D.; Romanova, T.; et al. Anhedonia, depression, anxiety, and craving in opiate dependent patients stabilized on oral naltrexone or an extended release naltrexone implant. The American Journal of Drug and Alcohol Abuse 2016, 42, 614–620. [Google Scholar] [CrossRef] [PubMed]
| 1 | Johnson & Johnson. (2025, 6 Mar). Johnson & Johnson statement on Ventura program [Press release]. https://www.jnj.com/media-center/press-releases/johnson-johnson-statement-on-ventura-program [Accessed 22.07.2025] |
| 2 | Fierce Biotech (2025, 7 Mar). J&J fails an Ace Ventura, stopping phase 3 depression program over insufficient efficacy. Fierce Biotech. https://www.fiercebiotech.com/biotech/jj-fails-ace-ventura-stopping-phase-3-depression-program-over-insufficient-efficacy [Accessed 22.07.2025] |
| 3 | Fierce Biotech (2025, 2 Jan). Neumora stumbles at start of phase 3 depression readout run, sending stock down 80%. Fierce Biotech. https://www.fiercebiotech.com/biotech/neumora-stumbles-start-phase-3-depression-readout-run-sending-stock-down-80 [Accessed 22.07.2025] |
| 4 | |
| 5 | FDA Advisors Overwhelmingly Reject Alkermes Depression Drug. BioSpace, 2 Nov. 2018 [cited 2025 Jan 10]. Available from: www.biospace.com/article/fda-advisors-overwhelmingly-reject-alkermes-depression-drug
|
| 6 | Johnson & Johnson. (2024). Johnson & Johnson statement on Ventura program. [cited 2025 Jan 10]. Available from: https://www.jnj.com/media-center/press-releases/johnson-johnson-statement-on-ventura-program
|
| 7 | Neumora Therapeutics. (2025). Neumora Therapeutics reports data from KOASTAL-1 study of navacaprant. [cited 2026 Jan 10]. Available from:https://ir.neumoratx.com/news-releases/news-release-details/neumora-therapeutics-reports-data-koastal-1-study-navacaprant
|
| 8 | BioCentury. (2019, 11 Jan). BlackThorn deprioritizing MDD compound after Phase IIa miss. https://www.biocentury.com/article/300266/blackthorn-deprioritizing-mdd-compound-after-phase-iia-miss [Accessed 22.07.2025] |
| 9 | Emax [agonist efficacy] –– the magnitude of the maximum activation of the secondary messenger system under the action of an agonist at its maximum concentration. Previously, the term intrinsic (agonistic) activity was used in the literature. |
| 10 | Neumora Therapeutics. Phase 2 results of NMRA-140 in major depressive disorder [poster presentation]. ACNP Annual Meeting; December 2023. https://neumoratx.com/wp-content/uploads/2024/08/ACNP23_Ph2_Poster_Final.pdf [Accessed 22.07.2025] |
| 11 | Shagiakhmetov F, Mukhametshina E, Samsonov M. Novel triple opioid receptor antagonist ondelopran (LY2196044) for the treatment of alcohol dependence. Phase III study: female subjects subset efficacy and safety analysis [poster]. Presented at: European Psychiatric Association Congress (EPA); 2019; Nice. Available from: https://doi.org/10.13140/RG.2.2.35480.96001. |
| Study | Indication | Study Design | Number of Participants (N) | Groups and daily doses | Efficacy Results |
| Aticaprant Phase 2a FAST-MAS Proof-of-Mechanism Study [114] |
Anhedonia (MDD/anxiety disorders) | RCT, double-blind, 8 weeks | 89 | 10 mg | Aticaprant significantly increased fMRI ventral striatum activation during reward anticipation (primary outcome) compared with placebo. |
| Aticaprant Phase 2 [115] |
Major depressive disorder | RCT, double-blind, 6 weeks, placebo lead-in | 166 (fITT); 121 (eITT) |
10 mg (adjunctive to SSRI/SNRI) |
Improvement in MADRS total score for aticaprant was significant versus placebo (eITT: −2.1 [−1.09], 1-sided p = 0.044; effect size (ES) 0.23; fITT −3.1 [2.21], 1-sided p = 0.002; ES 0.36). The between-group difference was larger among participants with SHAPS score greater/equal to versus less than baseline median SHAPS. Responders: 36.4% (JNJ) vs 24.0% (PBO) fITT, p<0.05; QIDS: p=0.029; CGI-I: p=0.046 |
| Aticaprant Phase 3 VENTURA Program (2025) [J&J Press Release]6 |
MDD with moderate-to-severe anhedonia | RCT, double-blind, 42 days | Not disclosed | 10 mg (adjunctive to SSRI/SNRI) |
Insufficient efficacy; primary endpoint (change in MADRS) not achieved; Safety confirmed; secondary efficacy not disclosed |
| Navacaprant Phase 2a [117] |
MDD with anhedonia and anxiety | RCT, double-blind, 8 weeks | 204 | 80 mg | Primary endpoint (change in HAMD-17) not achieved; but was statistically significant in moderate-to-severe subgroup (n=100) at weeks 4 and 8 as well as SHAPS score at week 8 |
| Navacaprant Phase 3 KOASTAL-1 [Neumora Therapeutics press release]7 |
Moderate-to-severe MDD | RCT, double-blind, 6 weeks | 383 | 80 mg | Primary endpoint not achieved; change in SHAPS and MADRS was identical for both groups; change in SHAPS was statistically significant in favor of navacaprant in female subset of patients https://ir.neumoratx.com/news-releases/news-release-details/neumora-therapeutics-reports-data-koastal-1-study-navacaprant |
| ALKS-5461 Phase 2 [120] |
MDD with inadequate antidepressant response | RCT, double-blind, two-stage sequential parallel comparison design, 4 weeks | 142 | 2 mg/2 mg 8 mg/8 mg Placebo (adjunctive to antidepressant) |
Greater improvement over placebo was observed in the 2 mg /2 mg group across all three depression outcome measures (HAM-D; MADRS; CGI-S). There was also evidence of improvement in the 8 mg /8 mg dosage group, although it did not achieve statistical significance. HAM-D response rates: 60% (2 mg /2 mg dose) vs 20% (PBO); |
| ALKS-5461 Phase 3 FORWARD-3 [121] |
MDD with inadequate antidepressant response | Double-blind, placebo run-in, 10 weeks | 295 |
2 mg/2 mg Placebo (adjunctive to antidepressant) |
Change in MADRS-10 did not achieve statistical significance. Response rate 16.9% (ALKS-5461) vs 14.4% (placebo), non-significant; Remission 14.1% vs 12.3%, non-significant. |
| ALKS-5461 Phase 3 FORWARD-4 FORWARD-5 [122] |
MDD with inadequate antidepressant response | Two phase 3 RCT, double-blind, studies that utilized the same sequential parallel-comparison design, 2 stages, 11 weeks | Not formally reported | 2 mg/2 mg 1 mg/1 mg 0.5 mg/0.5 mg Placebo (adjunctive to antidepressant) |
FORWARD-5 achieved the primary endpoint (change from baseline through end of treatment (EOT) in MADRS-6 and -10) in 2 mg/2 mg group. FORWARD-4 did not achieve the primary endpoint (change from baseline in MADRS-10 at week 5 versus placebo, P = 0.109). The pooled analysis of the two studies demonstrated consistently greater reduction in MADRS-10 scores from baseline for 2 mg/2 mg group at multiple timepoints including primary endpoint. |
| Study | Indication | Study Design | Number of Participants (N) | Groups and daily doses | Efficacy Results |
| BTRX-246040 Phase 2 [134] |
Major Depressive Disorder | RCT, double-blind, 8 weeks | 136 | 40 mg | The primary endpoint (probability of BTRX-246040 being better than placebo⩾88% on the GRID-HAMD-17 depression scale) in the full analysis set was not met (the actual value was 82,9%), but was met in per protocol population (88,6%). When the analysis in the full analysis set was extended to include a follow-up visit (weeks 9-10), the probability of BTRX-246040 being better than placebo reached 97,4%. The probability of BTRX-246040 being better than placebo in improving the ability to recognize positive emotional expressions was 92.4% |
| BTRX-246040 Phase 2 [135] |
Alcohol dependence | RCT, double-blind, 8 weeks | 88 | 40 mg |
NDD did not differ versus placebo; HDD showed improvement versus placebo (-24.5 vs. -15.7%; 93% better than placebo); mean percentage of abstinent days was higher compared to placebo (9.1 vs. 1.9%; 91% better than placebo) |
| BTRX-246040 Phase 2 [91] |
Major Depressive Disorder | RCT, double-blind, 8 weeks | 102 | 40-80 mg | BTRX-246040 did not significantly separate from placebo on the primary MADRS or on secondary endpoints (DARS, SHAPS). EEfRT (t) parametr showed significant group×time interaction (b=0.84, p=0.015); was significantly increasing for the BRTX 246060 group (b = 0.88, p <0.001). |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




