Submitted:
20 March 2026
Posted:
23 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Sources of Toxic Elements and Routes of Exposure
3. Biomonitoring and Health Effects of Toxic Elements
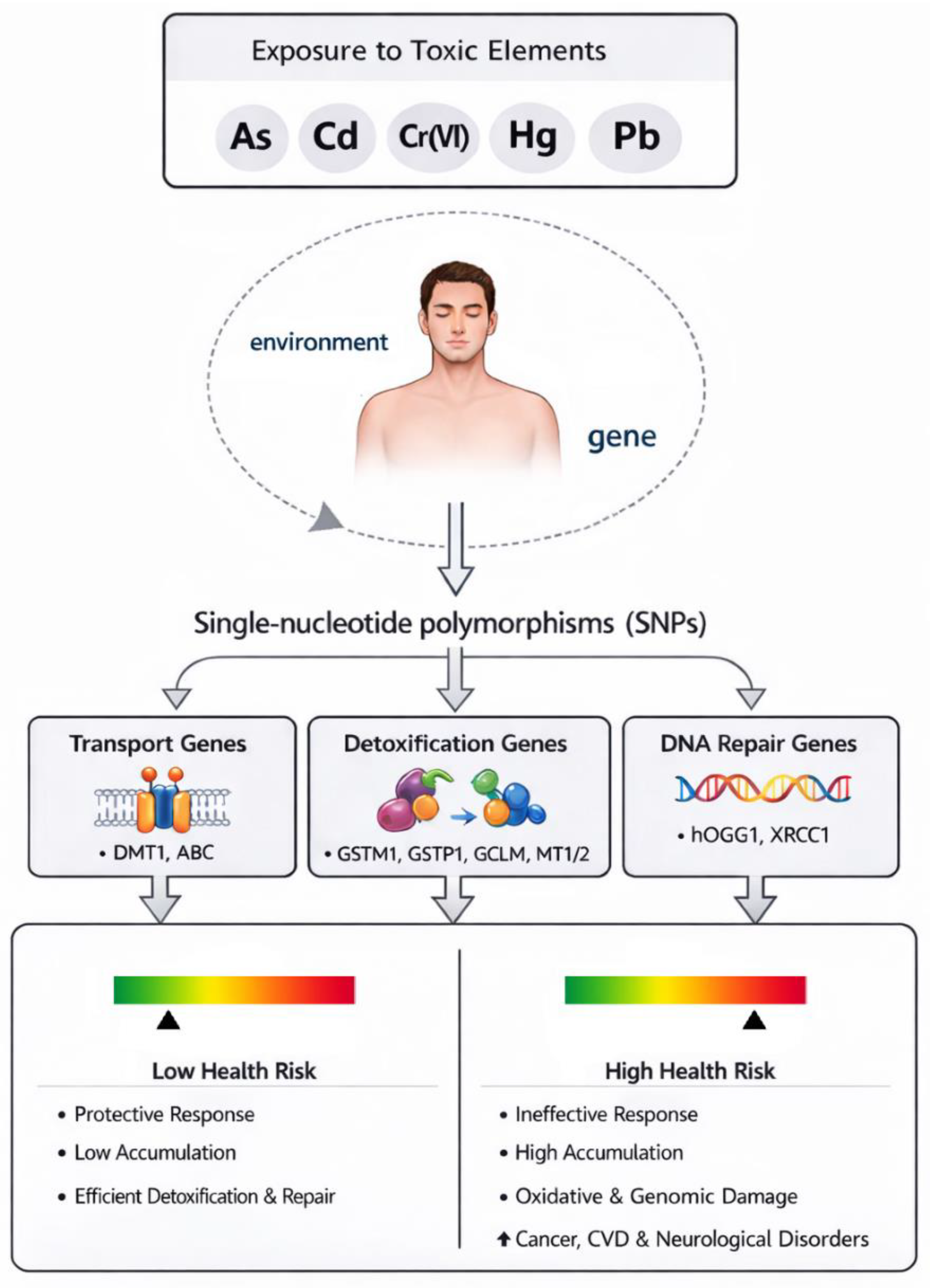
4. Gene-Environment Interaction: The Role of Single-Nucleotide Polymorphisms in Susceptibility to Toxic Element-Induced Effects
4.1. Transport
4.2. Detoxification
4.2.1. Glutathione-Related Genes
4.2.2. Metallothioneins
4.3. DNA Repair: Focus on the Base Excision Repair Pathway
|
Toxic element |
Pathway | Reference | Gene | SNP | Effect | ||
| As | Detoxification | [69] | GSTO1 | rs11191979 | impairment in body’s ability to methylate and detoxify arsenic efficiently, ↑ risk of skin lesions |
||
| rs2164624 |  |
||||||
| rs4925 | |||||||
| GSTO2 | rs156697 | ||||||
| rs2297235 | |||||||
| PNP | rs3790064 | ||||||
| [70] | GSTP1 | rs1695 AG + GG genotypes |
↓ efficiency in methylating inorganic As, ↑ urinary percentage of inorganic arsenic, ↑ toxicity |
 |
|||
| [72] | GSTT1 | + genotype | ↑ urinary As levels and ↑ carotid intima-media thickness |  |
|||
| [73] | GSTM1 | rs4025935 null genotype |
↓ As poisoning |  |
|||
| DNA repair | [87] | hOGG1 | rs1052133 Ser326Cys |
↑ urinary As levels, ↓ leukocyte telomere length, ↑ genomic instability |  |
||
| XRCC1 | rs25487 Arg399Gln |
||||||
| [88] | hOGG1 | rs1052133 | ↑ risk of urothelial carcinoma |  |
|||
| rs159153 | |||||||
| Cd | Detoxification | [74] | CAT | rs7943316 | altered expression of antioxidant enzymes, ↑ oxidative damage |  |
|
| GSTP1 | rs1695 | ||||||
| GSTM1 | null genotype | ||||||
| GSTT1 | null genotype | ||||||
| [79] | GSTT1 | null genotype | ↑ blood Cd levels |  |
|||
| [81] | MT1A | rs8044719 | ↑ tissue Cd retention, ↓ urinary Cd |  |
|||
| MT1B | rs1599823 | ||||||
| MT2A | rs28366003 | ||||||
| rs10636 | |||||||
| [82] | MT2A | rs28366003 GG genotype |
↑ blood Cd levels |  |
|||
| DNA repair | [9] | hOGG1 | rs1052133 | ↓ DNA repair capacity, ↑ genotoxic effects |  |
||
| XRCC1 | rs25487 | ||||||
| Cr(VI) | Detoxification | [76] | GSTP1 | rs1695 | ↑ risk of lung cancer |  |
|
| CAT | rs1001179 | ||||||
| DNA repair | [91] | XRCC1 | rs25487 Arg399Gln |
↑ chromosomal aberrations and susceptibility to chromosomal damage |
 |
||
| [92] | XRCC1 | rs25487 Arg399Gln |
↓ DNA damage |  |
|||
| [93] | XPD | rs13181 Lys751Gln |
↑ susceptibility to lung cancer |  |
|||
| XPC | rs2228000 Lys939Gln |
||||||
| [94] | XRCC3 | rs2295152 T allele |
↑ micronuclei frequency and genetic damage |  |
|||
| Hg | Transport | [68] | ABCC1 | rs11075290 C-allele |
↑ cord blood Hg levels and ↑ odds of small-for-gestational-age |
 |
|
| ABCB1 | rs2032582 GG genotype |
↑ cord blood Hg levels and ↓ mental development index |
 |
||||
| Detoxification | [75] | GCLM | rs41303970 TT genotype |
↓ Hg concentrations in blood and hair, ↑ efficient mercury elimination |
 |
||
| GSTM1 | null genotype | ↑ Hg accumulation |  |
||||
| [77] | GCLC | rs1555903 C allele |
↓ estimated glomerular filtration rate (eGFR) in non-exposed individuals and ↓ beta-2- microglobulin in exposed individuals, impairment of renal function |
 |
|||
| GSTA1 + GSS |
rs3957356 C allele + rs3761144 G allele |
↑ Hg retention |  |
||||
| GCLM | rs41303970 T allele | ↑ urinary Hg clearance, ↑ elimination |  |
||||
| [68] | GSTM1 | null genotype | ↑ Hg accumulation, ↑ oxidative stress, ↑ risk of preterm birth and ↓ weight in offspring |  |
|||
| GSTT1 | null genotype | ||||||
| GCLM | rs41303970 | ||||||
| GSTP1 | rs1695 G allele |
↓ mental development index in offspring |  |
||||
| GCLC | rs761142 TT genotype |
↓ psychomotor development index |  |
||||
| [84] | MT1M | rs2270837 AA genotype |
↓ urinary Hg levels, ↑ Hg retention |  |
|||
| MT2A | rs10636 CC genotype |
||||||
| MT1A | rs8052394 GA and GG genotypes |
↓ hair Hg levels |  |
||||
| MT1M | rs9936741 TT genotype |
||||||
| [85] | MT1M | rs2270836 | ↑ hair Hg levels |  |
|||
| rs9936741 | ↓ hair Hg levels |  |
|||||
| DNA repair | [90] | XRCC1 | rs25487 Arg399Gln |
↑ DNA damage and oxidative stress |  |
||
| hOGG1 | rs1052133 Ser326Cys |
||||||
| Pb | Transport | [66] | SLC11A2 | IVS4+44 C/A | ↑ risk of hypertension |  |
|
| [67] | SLC11A2 | rs224589 CA genotype |
↑ blood Pb concentration and ↑ hematological alterations |
 |
|||
| Detoxification | [78] | GCLC | rs17883901 | ↑ antioxidant response |  |
||
| GCLM | rs41303970 | ↓ blood and plasma Pb levels |
 |
||||
| [79] | GSTT1 | null genotype | ↑ blood Pb levels |  |
|||
| GSTP1 | rs1695 Ile/Val genotype |
↑ risk of Pb toxicity |  |
||||
| [82] | MT2A | rs28366003 GG genotype |
↑ blood Pb levels |  |
|||
| [83] | MT1A | rs11640851 | ↓ uric acid elimination, ↑ renal dysfunction |  |
|||
| rs8052394 | |||||||
| DNA repair | [89] | hOGG1 | rs1052133 | ↑ DNA instability and genotoxicity biomarkers |  |
||
| XPA | rs1800975 | ||||||
| XPC | rs2228000 | ||||||
5. Conclusions and Future Perspectives
| AD | Alzheimer’s disease |
| As | Arsenic |
| Cd | Cadmium |
| Cr | Chromium |
| Hg | Mercury |
| IARC | International Agency for Research and Cancer |
| MTs | Metallothioneins |
| Pb | Lead |
| PTEs | Potentially toxic elements |
| PVC | Polyvinyl Chloride |
| SNPs | Single-nucleotide polymorphisms |
| TEs | Toxic elements |
| WHO | World Health Organization |
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pourret, O.; Hursthouse, A. It’s Time to Replace the Term “Heavy Metals” with “Potentially Toxic Elements” When Reporting Environmental Research. Int. J. Environ. Res. Public Health 2019, 16, 4446. [Google Scholar] [CrossRef]
- Angon, P.B.; Islam, M.S.; Kc, S.; Das, A.; Anjum, N.; Poudel, A.; Suchi, S.A. Sources, Effects and Present Perspectives of Heavy Metals Contamination: Soil, Plants and Human Food Chain. Heliyon 2024, 10, e28357. [Google Scholar] [CrossRef] [PubMed]
- Järup, L. Hazards of Heavy Metal Contamination. Br. Med. Bull. 2003, 68, 167–182. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.F.; Zhao, Y.J.; Chen, C.; Zhang, F. Heavy Metals Toxicity: Mechanism, Health Effects, and Therapeutic Interventions. MedComm 2025, 6, e70241. [Google Scholar] [CrossRef]
- Zeise, L.; Bois, F.Y.; Chiu, W.A.; Hattis, D.; Rusyn, I.; Guyton, K.Z. Addressing Human Variability in Next-Generation Human Health Risk Assessments of Environmental Chemicals. Environ. Health Perspect. 2013, 121, 23–31. [Google Scholar] [CrossRef]
- Ottman, R. Gene-Environment Interaction: Definitions and Study Designs. Prev. Med. 1996, 25, 764–770. [Google Scholar] [CrossRef]
- Azizzadeh-Roodpish, S.; Garzon, M.H.; Mainali, S. Classifying Single Nucleotide Polymorphisms in Humans. Mol. Genet. Genomics 2021, 296, 1161–1173. [Google Scholar] [CrossRef]
- Joneidi, Z.; Mortazavi, Y.; Memari, F.; Roointan, A.; Chahardouli, B.; Rostami, S. The Impact of Genetic Variation on Metabolism of Heavy Metals: Genetic Predisposition? Biomed. Pharmacother. 2019, 113, 108642. [Google Scholar] [CrossRef]
- Pinto, T.G.; Dias, T.A.; Renno, A.C.M.; Dos Santos, J.N.; Cury, P.R.; Ribeiro, D.A. The Impact of Genetic Polymorphisms for Detecting Genotoxicity in Workers Occupationally Exposed to Metals: A Systematic Review. J. Appl. Toxicol. 2025, 45, 1070–1079. [Google Scholar] [CrossRef]
- Kumar, P. Genetic susceptibility to heavy metal toxicity. In Heavy Metal Toxicity and Neurodegeneration; Kumar, P., Gogia, N., Eds.; Elsevier: Amsterdam, The Netherlands, 2025; pp. 179–201. [Google Scholar]
- He, Z.L.; Yang, X.E.; Stoffella, P.J. Trace Elements in Agroecosystems and Impacts on the Environment. J. Trace Elem. Med. Biol. 2005, 19, 125–140. [Google Scholar] [CrossRef] [PubMed]
- Briffa, J.; Sinagra, E.; Blundell, R. Heavy Metal Pollution in the Environment and Their Toxicological Effects on Humans. Heliyon 2020, 6, e04691. [Google Scholar] [CrossRef]
- Garelick, H.; Jones, H.; Dybowska, A.; Valsami-Jones, E. Arsenic Pollution Sources. Rev. Environ. Contam. Toxicol. 2008, 197, 17–60. [Google Scholar] [CrossRef]
- Sattar, S.; Yahya, M.; Aslam, S.; Hussain, R.; Mukkarram Shah, S.M.; Rauf, Z.; Zamir, A.; Ullah, R.; Shahzad, A. Environmental Occurrence, Hazards, and Remediation Strategies for the Removal of Cadmium from the Polluted Environment. Results Eng. 2025, 25, 104322. [Google Scholar] [CrossRef]
- Ao, M.; Sun, S.; Deng, T.; Zhang, F.; Liu, T.; Tang, Y.; Li, J.; Wang, S.; Qiu, R. Natural Source of Cr(VI) in Soil: The Anoxic Oxidation of Cr(III) by Mn Oxides. J. Hazard. Mater. 2022, 433, 128805. [Google Scholar] [CrossRef]
- Ukhurebor, K.E.; Aigbe, U.O.; Onyancha, R.B.; Nwankwo, W.; Osibote, O.A.; Paumo, H.K.; Ama, O.M.; Adetunji, C.O.; Siloko, I.U. Effect of Hexavalent Chromium on the Environment and Removal Techniques: A Review. J. Environ. Manag. 2021, 280, 111809. [Google Scholar] [CrossRef]
- Cheema, A.I.; Liu, G.; Yousaf, B.; Abbas, Q.; Zhou, H. A Comprehensive Review of Biogeochemical Distribution and Fractionation of Lead Isotopes for Source Tracing in Distinct Interactive Environmental Compartments. Sci. Total Environ. 2020, 719, 135658. [Google Scholar] [CrossRef]
- Kim, M.K.; Zoh, K.D. Fate and Transport of Mercury in Environmental Media and Human Exposure. J. Prev. Med. Public Health 2012, 45, 335–343. [Google Scholar] [CrossRef]
- Baker, B.A.; Cassano, V.A.; Murray, C. ACOEM Task Force on Arsenic Exposure. Arsenic Exposure, Assessment, Toxicity, Diagnosis, and Management: Guidance for Occupational and Environmental Physicians. J. Occup. Environ. Med. 2018, 60, e634–e639. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Reynolds, M. Cadmium Exposure in Living Organisms: A Short Review. Sci. Total Environ. 2019, 678, 761–767. [Google Scholar] [CrossRef]
- Tumolo, M.; Ancona, V.; De Paola, D.; Losacco, D.; Campanale, C.; Massarelli, C.; Uricchio, V.F. Chromium Pollution in European Water, Sources, Health Risk, and Remediation Strategies: An Overview. Int. J. Environ. Res. Public Health 2020, 17, 5438. [Google Scholar] [CrossRef] [PubMed]
- Nag, R.; Cummins, E. Human Health Risk Assessment of Lead (Pb) through the Environmental-Food Pathway. Sci. Total Environ. 2022, 810, 151168. [Google Scholar] [CrossRef]
- Lanphear, B.; Navas-Acien, A.; Bellinger, D.C. Lead Poisoning. N. Engl. J. Med. 2024, 391, 1621–1631. [Google Scholar] [CrossRef]
- Clarkson, T.W.; Magos, L.; Myers, G.J. The Toxicology of Mercury—Current Exposures and Clinical Manifestations. N. Engl. J. Med. 2003, 349, 1731–1737. [Google Scholar] [CrossRef] [PubMed]
- Freire, B.M.; Gonzaga, R.G.; Pedron, T.; Monteiro, L.R.; Lange, C.N.; Pedreira Filho, W.D.R.; Batista, B.L. Occupational Exposure to Potentially Toxic Elements in the Foundry Industry: An Integrated Environmental and Biological Monitoring. Environ. Sci. Pollut. Res. Int. 2021, 28, 34630–34641. [Google Scholar] [CrossRef]
- Viegas, S.; Martins, C.; Bocca, B.; Bousoumah, R.; Duca, R.C.; Galea, K.S.; Godderis, L.; Iavicoli, I.; Janasik, B.; Jones, K.; Leese, E.; Leso, V.; Ndaw, S.; van Nieuwenhuyse, A.; Poels, K.; Porras, S.P.; Ruggieri, F.; Silva, M.J.; Verdonck, J.; Wasowicz, W. HBM4EU Chromates Study Team. HBM4EU Chromates Study: Determinants of Exposure to Hexavalent Chromium in Plating, Welding and Other Occupational Settings. Int. J. Environ. Res. Public Health 2022, 19, 3683. [Google Scholar] [CrossRef] [PubMed]
- Khoshakhlagh, A.H.; Ghobakhloo, S.; Peijnenburg, W.J.G.M.; Gruszecka-Kosowska, A.; Cicchella, D. To Breathe or Not to Breathe: Inhalational Exposure to Heavy Metals and Related Health Risk. Sci. Total Environ. 2024, 932, 172556. [Google Scholar] [CrossRef]
- Zumbado, M.; Luzardo, O.P.; Rodríguez-Hernández, Á.; Boada, L.D.; Henríquez-Hernández, L.A. Differential Exposure to 33 Toxic Elements through Cigarette Smoking, Based on the Type of Tobacco and Rolling Paper Used. Environ. Res. 2019, 169, 368–376. [Google Scholar] [CrossRef]
- da Silva, C.P.; de Almeida, T.E.; Zittel, R.; de Oliveira Stremel, T.R.; Domingues, C.E.; Kordiak, J.; de Campos, S.X. Translocation of Metal Ions from Soil to Tobacco Roots and Their Concentration in the Plant Parts. Environ. Monit. Assess. 2016, 188, 663. [Google Scholar] [CrossRef] [PubMed]
- Uwiringiyimana, E.; Gao, J.; Zhang, D.; Biswash, M.R.; Shi, Y.X. Bioaccumulation and Translocation of Hg and Cr by Tobacco in Sichuan Province, China: Understanding the Influence of Soil pH. Environ. Monit. Assess. 2023, 195, 1193. [Google Scholar] [CrossRef]
- Lopez-Galvez, N.; Mahabee-Gittens, E.M.; Quintana, P.J.E.; Merianos, A.L.; Dodder, N.G.; Hoh, E.; Stone, L.; Watanabe, K.; Matt, G.E. Environmental Tobacco Smoke Is a Major Contributor to Lead, Cadmium, and Arsenic in Settled House Dust. Chemosphere 2026, 394, 144820. [Google Scholar] [CrossRef]
- Patel, K.S.; Pandey, P.K.; Martín-Ramos, P.; Corns, W.T.; Varol, S.; Bhattacharya, P.; Zhu, Y. A Review on Arsenic in the Environment: Contamination, Mobility, Sources, and Exposure. RSC Adv. 2023, 13, 8803–8821. [Google Scholar] [CrossRef]
- Ouypornkochagorn, S.; Feldmann, J. Dermal Uptake of Arsenic through Human Skin Depends Strongly on Its Speciation. Environ. Sci. Technol. 2010, 44, 3972–3978. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Gao, P.; Li, M.Y.; Ma, J.Y.; Li, J.Y.; Yang, D.L.; Cui, D.L.; Xiang, P. Dermal Bioaccessibility and Cytotoxicity of Heavy Metals in Urban Soils from a Typical Plateau City: Implication for Human Health. Sci. Total Environ. 2022, 835, 155544. [Google Scholar] [CrossRef]
- Liu, C.; Liu, C.; Juhasz, A.L.; Dong, W.J.; Ma, L.Q. Human Health Risks of Chromium in Bright-Colored Leather Products: Speciation-Based Insights into Dermal Permeation. Environ. Int. 2025, 200, 109538. [Google Scholar] [CrossRef]
- Niemeier, R.T.; Maier, A.; Reichard, J.F. Rapid Review of Dermal Penetration and Absorption of Inorganic Lead Compounds for Occupational Risk Assessment. Ann. Work Expo. Health 2022, 66, 291–311. [Google Scholar] [CrossRef]
- Chan, T.Y. Inorganic Mercury Poisoning Associated with Skin-Lightening Cosmetic Products. Clin. Toxicol. 2011, 49, 886–891. [Google Scholar] [CrossRef]
- World Health Organization (WHO). 10 Chemicals of Public Health Concern. Available online: https://www.who.int/news-room/photo-story/detail/10-chemicals-of-public-health-concern (accessed on 19 February 2026).
- Gil, F.; Hernández, A.F. Toxicological Importance of Human Biomonitoring of Metallic and Metalloid Elements in Different Biological Samples. Food Chem. Toxicol. 2015, 80, 287–297. [Google Scholar] [CrossRef] [PubMed]
- Buekers, J.; Baken, K.; Govarts, E.; Martin, L.R.; Vogel, N.; Kolossa-Gehring, M.; Šlejkovec, Z.; Falnoga, I.; Horvat, M.; Lignell, S.; Lindroos, A.K.; Rambaud, L.; Riou, M.; Pedraza-Diaz, S.; Esteban-Lopez, M.; Castaño, A.; Den Hond, E.; Baeyens, W.; Santonen, T.; Schoeters, G. Human Urinary Arsenic Species, Associated Exposure Determinants and Potential Health Risks Assessed in the HBM4EU Aligned Studies. Int. J. Hyg. Environ. Health 2023, 248, 114115. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, F., Jr.; Tanus-Santos, J.E.; Gerlach, R.F.; Parsons, P.J. A Critical Review of Biomarkers Used for Monitoring Human Exposure to Lead: Advantages, Limitations, and Future Needs. Environ. Health Perspect. 2005, 113, 1669–1674. [Google Scholar] [CrossRef]
- Ndaw, S.; Leso, V.; Bousoumah, R.; Rémy, A.; Bocca, B.; Duca, R.C.; Godderis, L.; Hardy, E.; Janasik, B.; van Nieuwenhuyse, A.; Pinhal, H.; Poels, K.; Porras, S.P.; Ruggieri, F.; Santonen, T.; Santos, S.R.; Scheepers, P.T.J.; Silva, M.J.; Verdonck, J.; Viegas, S. HBM4EU Chromates Study Team. HBM4EU Chromates Study—Usefulness of Measurement of Blood Chromium Levels in the Assessment of Occupational Cr(VI) Exposure. Environ. Res. 2022, 214, 113758. [Google Scholar] [CrossRef]
- Adams, S.V.; Newcomb, P.A. Cadmium Blood and Urine Concentrations as Measures of Exposure: NHANES 1999–2010. J. Expo. Sci. Environ. Epidemiol. 2014, 24, 163–170. [Google Scholar] [CrossRef]
- So, S.C.A.; Tsoi, M.F.; Cheung, A.J.; Cheung, T.T.; Cheung, B.M.Y. Blood and Urine Inorganic and Organic Mercury Levels in the United States from 1999 to 2016. Am. J. Med. 2021, 134, e20–e30. [Google Scholar] [CrossRef]
- Lamas, G.A.; Bhatnagar, A.; Jones, M.R.; Mann, K.K.; Nasir, K.; Tellez-Plaza, M.; Ujueta, F.; Navas-Acien, A. American Heart Association Council on Epidemiology and Prevention; Council on Cardiovascular and Stroke Nursing; Council on Lifestyle and Cardiometabolic Health; Council on Peripheral Vascular Disease; Council on the Kidney in Cardiovascular Disease. Contaminant Metals as Cardiovascular Risk Factors: A Scientific Statement from the American Heart Association. J. Am. Heart Assoc. 2023, 12, e029852. [Google Scholar] [CrossRef]
- Cheema, S.; Hussain, S.I.; Faheem, M.S.B.; Jalal, A.A.; Rifai, M.; Dar, A.; Burhan, M.; Shahid, A.; Ali, M.S.; Anwar, A.; Khalid, M.; Samadi, S. Toxic Heavy Metal Exposure and Heart Health: A Systematic Review and Meta-Analysis of 324,331 Patients. BMC Cardiovasc. Disord. 2025, 25, 792. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, D.; Wu, X.; Tu, J.; Gong, C.; Li, Y.; Cui, W.; Chen, J.; Lu, S. Urinary metals as influencing factors of coronary heart disease among a population in Guangzhou, China. Ecotoxicol. Environ. Saf. 2022, 241, 113746. [Google Scholar] [CrossRef]
- Bashir, B.; Jarial, S.; Sood, S.; Vishwas, S. Heavy Metals and Alzheimer’s Disease: A Review. In Heavy Metal Toxicity and Neurodegeneration; Kumar, P., Gogia, N., Eds.; Academic Press, 2025; pp. 225–230. [Google Scholar] [CrossRef]
- Kooshki, A.; Farmani, R.; Mehrpour, O.; Naghizadeh, A.; Amirabadizadeh, A.; Kavoosi, S.; Vohra, V.; Nakhaee, S. Alzheimer’s Disease and Circulatory Imbalance of Toxic Heavy Metals: A Systematic Review and Meta-Analysis of Clinical Studies. Biol. Trace Elem. Res. 2025, 203, 1871–1885. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Chen, Y.; Fu, Z.; Wen, Y.; Zhao, W.; Li, J.; Wang, H.; Du, Y.; Deng, Y. Heavy Metal Exposure and Cognitive Impairment: An Umbrella Review of Meta-Analyses. J. Hazard. Mater. 2025, 498, 139881. [Google Scholar] [CrossRef]
- Wang, X.; Bakulski, K.M.; Walker, E.; Mukherjee, B.; Dodge, H.; Albin, R.L.; Paulson, H.L.; Park, S.K. Exposure to Lead and Incidence of Alzheimer’s Disease and All-Cause Dementia in the United States. Alzheimers Dement. 2026, 22, e71075. [Google Scholar] [CrossRef]
- Yang, Y.W.; Liou, S.H.; Hsueh, Y.M.; Lyu, W.S.; Liu, C.S.; Liu, H.J.; Chung, M.C.; Hung, P.H.; Chung, C.J. Risk of Alzheimer’s Disease with Metal Concentrations in Whole Blood and Urine: A Case-Control Study Using Propensity Score Matching. Toxicol. Appl. Pharmacol. 2018, 356, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Strumylaite, L.; Kregzdyte, R.; Kucikiene, O.; Baranauskiene, D.; Simakauskiene, V.; Naginiene, R.; Damuleviciene, G.; Lesauskaite, V.; Zemaitiene, R. Alzheimer’s Disease Association with Metals and Metalloids Concentration in Blood and Urine. Int. J. Environ. Res. Public Health 2022, 19, 7309. [Google Scholar] [CrossRef]
- Wise, J.P., Jr.; Young, J.L.; Cai, J.; Cai, L. Current Understanding of Hexavalent Chromium [Cr(VI)] Neurotoxicity and New Perspectives. Environ. Int. 2022, 158, 106877. [Google Scholar] [CrossRef]
- da Silva, A.S.; Santos, R.M.S.; De Marco, P.G.; Rezende, V.H.M.; Martins, T.C.; Silva, J.R.; Romano-Silva, M.A.; Miranda, D.M. Neurodevelopmental Outcomes Associated with Early-Life Exposure to Heavy Metals: A Systematic Review. Int. J. Environ. Res. Public Health 2025, 22, 1308. [Google Scholar] [CrossRef]
- Rahmani, A.; Khamutian, S.; Doosti-Irani, A.; Shokoohizadeh, M.J.; Shirmohammadi-Khorram, N.; Sahraeei, F.; Khodabakhshi, M.; Ahangaran, N. The Association of Arsenic Exposure with Mortality Due to Cancer, Diabetes, Alzheimer’s and Congenital Anomalies Using Poisson Regression. Sci. Rep. 2023, 13, 15456. [Google Scholar] [CrossRef]
- Lee, F.J.; Inchai, H.; Lin, J.T.; Koh, D.; Lin, R.T. Dose-Response Relationship of Cadmium and Pancreatic Cancer Risk: A Meta-Analysis. Occup. Environ. Med. 2025, 82, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Firmani, G.; Chiavarini, M.; Dolcini, J.; Quarta, S.; D’Errico, M.M.; Barbadoro, P. The Association Between Cadmium Exposure and Prostate Cancer: An Updated Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2024, 21, 1532. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, Y.; Li, T.; Zhuo, W.; Zhu, Y. Elevated Serum and Hair Levels of Cadmium as a Risk Factor for Liver Carcinoma: A Meta-Analysis. Nutr. Cancer 2023, 75, 1438–1447. [Google Scholar] [CrossRef]
- Lee, H.; Jo, Y.; Jung, M.; Lee, J.H.; Kim, T.H.; Lee, J.; Kim, D.J.; Rahmati, M.; Smith, L.; Pizzol, D.; Son, Y.; Park, J.; Ahn, S.H.; Yon, D.K.; Choi, D.W.; Kang, J. Heavy Metal Exposure and All Health Outcomes: An Umbrella Review of Meta-Analyses. J. Hazard. Mater. 2026, 503, 141141. [Google Scholar] [CrossRef]
- Abdelgawwad El-Sehrawy, A.A.M.; Hsu, C.Y.; Saleh, E.A.M.; Moharam, M.M.; Rekha, M.M.; Kundlas, M.; Premkumar, J.; Ray, S.; Hussein, A.R.; Kadhim, M.A. Associations between Cadmium and Lead Exposure and Thyroid Disorders: A Systematic Review and Meta-Analysis. J. Trace Elem. Med. Biol. 2025, 92, 127781. [Google Scholar] [CrossRef]
- Vagnoni, G.; Bortolotti, E.; Checchi, S.; Saieva, C.; Berti, G.; Doccioli, C.; Caini, S. Lead (Pb) in Biological Samples in Association with Cancer Risk and Mortality: A Systematic Literature Review. Cancer Epidemiol. 2024, 92, 102630. [Google Scholar] [CrossRef] [PubMed]
- Webster, A.M.; Pinion, D.; Pineda, E.; Aboueisha, H.; Hussein, M.H.; Fawzy, M.S.; Toraih, E.A.; Kandil, E. Elucidating the Link between Thyroid Cancer and Mercury Exposure: A Review and Meta-Analysis. Environ. Sci. Pollut. Res. 2024, 31, 12841–12855. [Google Scholar] [CrossRef]
- Deng, Y.; Wang, M.; Tian, T.; Lin, S.; Xu, P.; Zhou, L.; Dai, C.; Hao, Q.; Wu, Y.; Zhai, Z.; Zhu, Y.; Zhuang, G.; Dai, Z. The Effect of Hexavalent Chromium on the Incidence and Mortality of Human Cancers: A Meta-Analysis Based on Published Epidemiological Cohort Studies. Front. Oncol. 2019, 9, 24. [Google Scholar] [CrossRef]
- Virolainen, S.J.; VonHandorf, A.; Viel, K.C.M.F.; Weirauch, M.T.; Kottyan, L.C. Gene-Environment Interactions and Their Impact on Human Health. Genes Immun. 2023, 24, 1–11. [Google Scholar] [CrossRef]
- Kim, H.K.; Lee, H.; Kim, H.J. A Polymorphism in DMT1 Is Associated with Lead-Related Hypertensive Status. Mol. Cell. Toxicol. 2013, 9, 415–420. [Google Scholar] [CrossRef]
- Mani, M.S.; Dsouza, V.L.; Dsouza, H.S. Evaluation of Divalent Metal Transporter 1 (DMT1) (rs224589) Polymorphism on Blood Lead Levels of Occupationally Exposed Individuals. Toxicol. Lett. 2021, 353, 13–19. [Google Scholar] [CrossRef]
- Sadana, A.A.S.; Bakri, S.; Tokonami, S.; Nugraha, E.D.; Amqam, H.; Muniroh, M. Genetic Modulation of Mercury Exposure on Perinatal and Birth Outcomes: A Systematic Review and Meta-Analysis of Gene-Environment Interactions. J. Xenobiot. 2026, 16, 28. [Google Scholar] [CrossRef]
- Luo, L.; Li, Y.; Gao, Y.; Zhao, L.; Feng, H.; Wei, W.; Qiu, C.; He, Q.; Zhang, Y.; Fu, S.; Sun, D. Association between Arsenic Metabolism Gene Polymorphisms and Arsenic-Induced Skin Lesions in Individuals Exposed to High-Dose Inorganic Arsenic in Northwest China. Sci. Rep. 2018, 8, 413. [Google Scholar] [CrossRef]
- González-Martínez, F.; Sánchez-Rodas, D.; Cáceres, D.D.; Martínez, M.F.; Quiñones, L.A.; Johnson-Restrepo, B. Arsenic Exposure, Profiles of Urinary Arsenic Species, and Polymorphism Effects of Glutathione-S-Transferase and Metallothioneins. Chemosphere 2018, 212, 927–936. [Google Scholar] [CrossRef] [PubMed]
- Tamayo, L.I.; Tong, L.; Davydiuk, T.; Vander Griend, D.; Haque, S.E.; Islam, T.; Jasmine, F.; Kibriya, M.G.; Graziano, J.; Chen, L.; Le, X.C.; Ahsan, H.; Gamble, M.V.; Pierce, B.L. Genetic Variation in the FMO and GSTO Gene Clusters Impacts Arsenic Metabolism in Humans. PLoS Genet. 2025, 21, e1011826. [Google Scholar] [CrossRef] [PubMed]
- Stea, F.; Faita, F.; Borghini, A.; Faita, F.; Bianchi, F.; Bustaffa, E.; Minichilli, F.; Andreassi, M. G.; Sicari, R. Arsenic and Subclinical Vascular Damage in a Sample of Italian Young Adults: A Cross-Sectional Analysis. Environ. Sci. Pollut. Res. Int. 2016, 23, 20307–20314. [Google Scholar] [CrossRef] [PubMed]
- Jin, B.; Wan, S.; Boah, M.; Yang, J.; Ma, W.; Lv, M.; Li, H.; Wang, K. GSTM1 and GSTT1 Null Genotype Polymorphisms and Susceptibility to Arsenic Poisoning: A Meta-Analysis. Biol. Trace Elem. Res. 2021, 199, 2085–2095. [Google Scholar] [CrossRef]
- Khan, S.A.; Nurulain, S.M.; Qureshi, R.N.; Zafar, A.; Riaz, Z.; Shoukat, A.; Muneer, Z.; Bibi, N.; Raza, S.; Hussain, S.; Shah, S.T.A. Exposure to Heavy Metals, Antioxidant Status, and the Interaction of Single Nucleotide Polymorphisms in the Genes CAT rs7943316, GSTP1 rs1695, as well as GSTM1 and GSTT1 Genes, among Workers in Occupational Settings. Environ. Toxicol. Pharmacol. 2024, 108, 104452. [Google Scholar] [CrossRef]
- Barcelos, G.R.; Grotto, D.; de Marco, K.C.; Valentini, J.; Lengert, A.V.; de Oliveira, A.Á.; Garcia, S.C.; Braga, G.Ú.; Schläwicke Engström, K.; Cólus, I.M.; Broberg, K.; Barbosa, F., Jr. Polymorphisms in Glutathione-Related Genes Modify Mercury Concentrations and Antioxidant Status in Subjects Environmentally Exposed to Methylmercury. Sci. Total Environ. 2013, 463–464, 319–325. [Google Scholar] [CrossRef]
- Baszuk, P.; Janasik, B.; Pietrzak, S.; Marciniak, W.; Reszka, E.; Białkowska, K.; Jabłońska, E.; Muszyńska, M.; Lesicka, M.; Derkacz, R.; Grodzki, T.; Wójcik, J.; Wojtyś, M.; Dębniak, T.; Cybulski, C.; Gronwald, J.; Kubisa, B.; Wójcik, N.; Pieróg, J.; Gajić, D.; Lener, M.R. Lung Cancer Occurrence—Correlation with Serum Chromium Levels and Genotypes. Biol. Trace Elem. Res. 2021, 199, 1228–1236. [Google Scholar] [CrossRef]
- Medina Pérez, O.M.; Flórez-Vargas, O.; Rincón Cruz, G.; Rondón González, F.; Rocha Muñoz, L.; Sánchez Rodríguez, L.H. Glutathione-Related Genetic Polymorphisms Are Associated with Mercury Retention and Nephrotoxicity in Gold-Mining Settings of a Colombian Population. Sci. Rep. 2021, 11, 8716. [Google Scholar] [CrossRef] [PubMed]
- Gomes, W.R.; Devóz, P.P.; Luiz, B.L.C.; Grotto, D.; Batista, B.L.; Barbosa, F., Jr.; Barcelos, G.R.M. Polymorphisms of Genes Related to Metabolism of Lead (Pb) Are Associated with the Metal Body Burden and with Biomarkers of Oxidative Stress. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2018, 836 Pt B, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Yohannes, Y.B.; Nakayama, S.M.M.; Yabe, J.; Toyomaki, H.; Kataba, A.; Nakata, H.; Muzandu, K.; Ikenaka, Y.; Choongo, K.; Ishizuka, M. Glutathione S-Transferase Gene Polymorphisms in Association with Susceptibility to Lead Toxicity in Lead- and Cadmium-Exposed Children near an Abandoned Lead-Zinc Mining Area in Kabwe, Zambia. Environ. Sci. Pollut. Res. Int. 2022, 29, 6622–6632. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Roshani, D.; Gao, B.; Li, P.; Shang, N. Metallothionein: A Comprehensive Review of Its Classification, Structure, Biological Functions, and Applications. Antioxidants 2024, 13, 825. [Google Scholar] [CrossRef]
- Adams, S.V.; Barrick, B.; Christopher, E.P.; Shafer, M.M.; Makar, K.W.; Song, X.; Lampe, J.W.; Vilchis, H.; Ulery, A.; Newcomb, P.A. Genetic Variation in Metallothionein and Metal-Regulatory Transcription Factor 1 in Relation to Urinary Cadmium, Copper, and Zinc. Toxicol. Appl. Pharmacol. 2015, 289, 381–388. [Google Scholar] [CrossRef]
- Kayaaltı, Z.; Aliyev, V.; Söylemezoğlu, T. The Potential Effect of Metallothionein 2A -5A/G Single Nucleotide Polymorphism on Blood Cadmium, Lead, Zinc and Copper Levels. Toxicol. Appl. Pharmacol. 2011, 256, 1–7. [Google Scholar] [CrossRef]
- Yang, C.C.; Chen, H.I.; Chiu, Y.W.; Tsai, C.H.; Chuang, H.Y. Metallothionein 1A Polymorphisms May Influence Urine Uric Acid and N-Acetyl-Beta-D-Glucosaminidase (NAG) Excretion in Chronic Lead-Exposed Workers. Toxicology 2013, 306, 68–73. [Google Scholar] [CrossRef]
- Wang, Y.; Goodrich, J.M.; Gillespie, B.; Werner, R.; Basu, N.; Franzblau, A. An Investigation of Modifying Effects of Metallothionein Single-Nucleotide Polymorphisms on the Association between Mercury Exposure and Biomarker Levels. Environ. Health Perspect. 2012, 120, 530–534. [Google Scholar] [CrossRef]
- Chan, P.H.Y.; Chan, K.Y.Y.; Schooling, C.M.; Hui, L.L.; Chan, M.H.M.; Li, A.M.; Cheung, R.C.K.; Lam, H.S. Association between Genetic Variations in GSH-Related and MT Genes and Low-Dose Methylmercury Exposure in Children and Women of Childbearing Age: A Pilot Study. Environ. Res. 2020, 187, 109703. [Google Scholar] [CrossRef] [PubMed]
- Vodicka, P.; Stetina, R.; Polakova, V.; Tulupova, E.; Naccarati, A.; Vodickova, L.; Kumar, R.; Hanova, M.; Pardini, B.; Slyskova, J.; Musak, L.; De Palma, G.; Soucek, P.; Hemminki, K. Association of DNA Repair Polymorphisms with DNA Repair Functional Outcomes in Healthy Human Subjects. Carcinogenesis 2007, 28, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Borghini, A.; Faita, F.; Mercuri, A.; Minichilli, F.; Bustaffa, E.; Bianchi, F.; Andreassi, M. G. Arsenic Exposure, Genetic Susceptibility and Leukocyte Telomere Length in an Italian Young Adult Population. Mutagenesis 2016, 31, 539–546. [Google Scholar] [CrossRef]
- Huang, C. Y.; Pu, Y. S.; Shiue, H. S.; Chen, W. J.; Lin, Y. C.; Hsueh, Y. M. Polymorphisms of Human 8-Oxoguanine DNA Glycosylase 1 and 8-Hydroxydeoxyguanosine Increase Susceptibility to Arsenic Methylation Capacity-Related Urothelial Carcinoma. Arch. Toxicol. 2016, 90, 1917–1927. [Google Scholar] [CrossRef]
- Nunes, E.A.; Silva, H.C.D.; Duarte, N.A.A.; de Lima, L.E.; Maraslis, F.T.; Araújo, M.L.; Pedron, T.; Neves Lange, C.; Freire, B.M.; Matias, A.C.; Batista, B.L.; Barcelos, G.R.M. Impact of DNA Repair Polymorphisms on DNA Instability Biomarkers Induced by Lead (Pb) in Workers Exposed to the Metal. Chemosphere 2023, 334, 138897. [Google Scholar] [CrossRef]
- Andreoli, V.; Sprovieri, F. Genetic Aspects of Susceptibility to Mercury Toxicity: An Overview. Int. J. Environ. Res. Public Health 2017, 14, 93. [Google Scholar] [CrossRef]
- Halasova, E.; Matakova, T.; Musak, L.; Polakova, V.; Letkova, L.; Dobrota, D.; Vodicka, P. Evaluating Chromosomal Damage in Workers Exposed to Hexavalent Chromium and the Modulating Role of Polymorphisms of DNA Repair Genes. Int. Arch. Occup. Environ. Health 2012, 85, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, X.; Zhang, L.; Chen, Q.; Yang, Z.; Yu, J.; Fu, H.; Zhu, Y. XRCC1 Arg399Gln Was Associated with Repair Capacity for DNA Damage Induced by Occupational Chromium Exposure. BMC Res. Notes 2012, 5, 263. [Google Scholar] [CrossRef]
- Sarlinova, M.; Majerova, L.; Matakova, T.; Musak, L.; Slovakova, P.; Skereňová, M.; Kavcová, E.; Halašová, E. Polymorphisms of DNA Repair Genes and Lung Cancer in Chromium Exposure. Adv. Exp. Med. Biol. 2015, 833, 1–8. [Google Scholar] [CrossRef]
- Long, C.; Liu, J.; Hu, G.; Feng, H.; Zhou, D.; Wang, J.; Zhai, X.; Zhao, Z.; Yu, S.; Wang, T.; Jia, G. Modulation of Homologous Recombination Repair Gene Polymorphisms on Genetic Damage in Chromate Exposed Workers. Environ. Toxicol. Pharmacol. 2019, 66, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Fontanelli, L.; Castronovo, A.; Ferri, C.; Vozzi, F.; Recchia, F.A.; Borghini, A. iPSC-Derived Endothelial Cells as Experimental Models for Predictive and Personalized Strategies in Cardiovascular and Cerebrovascular Disease. Int. J. Mol. Sci. 2026, 27, 780. [Google Scholar] [CrossRef] [PubMed]
- Chandy, M.; Obal, D.; Wu, J.C. Elucidating Effects of Environmental Exposure Using Human-Induced Pluripotent Stem Cell Disease Modeling. EMBO Mol. Med. 2022, 14, e13260. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




