Submitted:
13 March 2026
Posted:
20 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods: Ethics Approval and Participant Selection for Clinical Irritation and Sensitization Trials
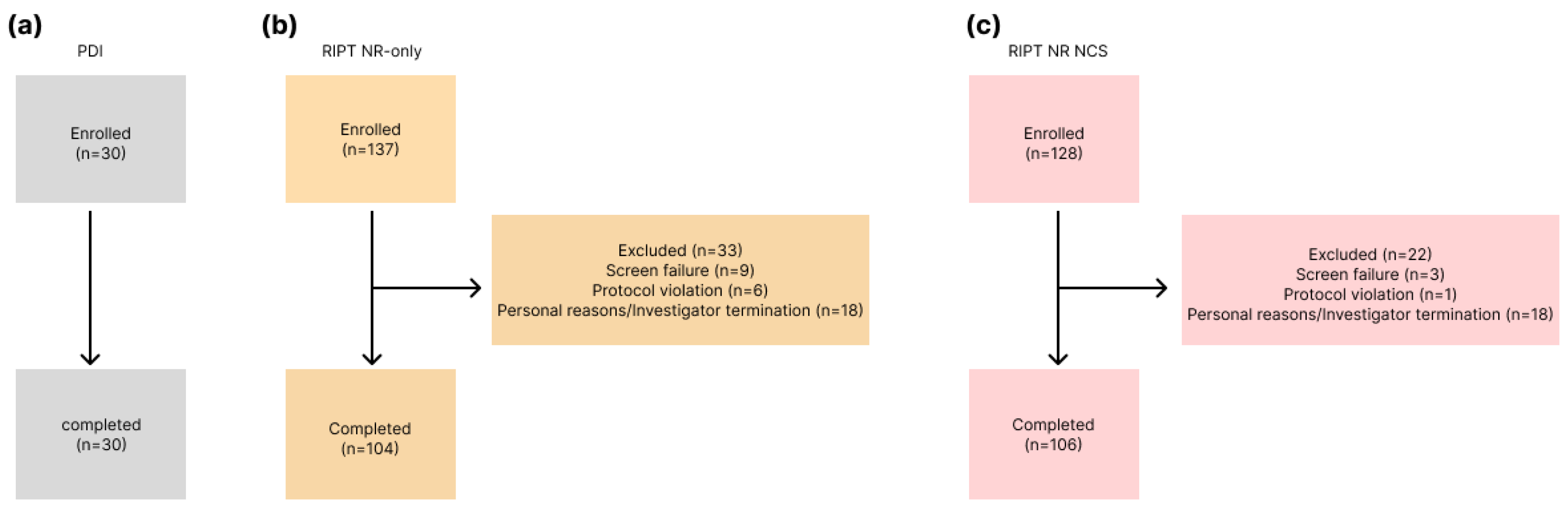
2.2.1. Primary Dermal Irritation (PDI)
2.2.2. Repeated Insult Patch Test (RIPT)
2.3. Methods: Clinical Acute Efficacy of NR
2.3.1. Ethical Approval and Trial Participants
2.3.2. Skin Moisturizing
2.3.3. Skin Protecting
2.3.4. Skin Smoothing
2.4. Statistics
2.5. AI Use
3. Results
3.1. Irritation
3.2. Efficacy
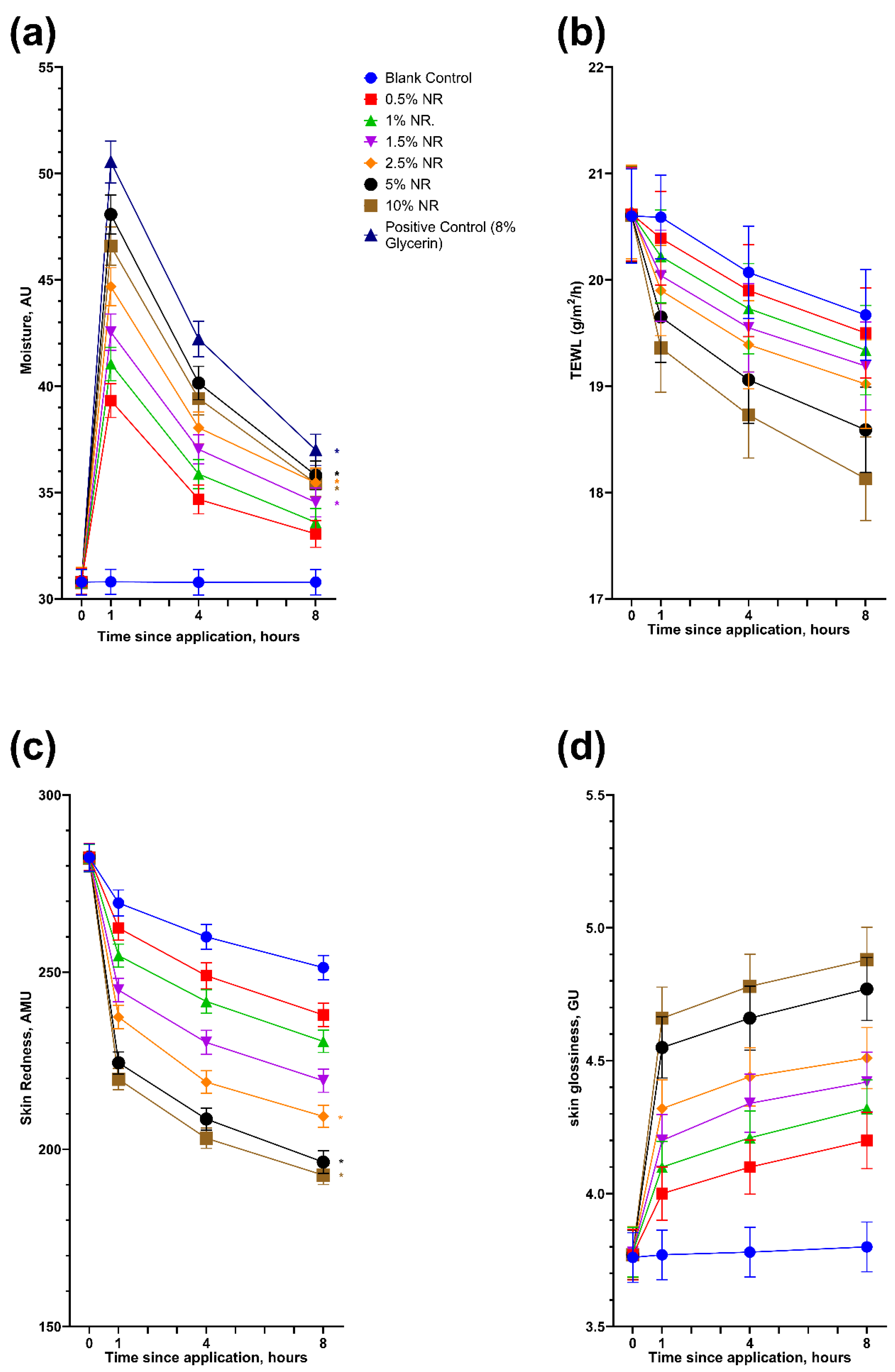
3.2.1. Skin Moisturizing
3.2.2. Participant Reported Outcomes Related to Skin Moisturizing
3.2.3. Skin Protecting
3.2.4. Participant Reported Outcomes Related to Skin Protection
3.2.5. Skin Smoothing
3.2.6. Participant Reported Outcomes Related to Skin Smoothing
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 3M | Medical tape brand |
| ADP-ribose | Adenosine diphosphate-ribose |
| AI | Artificial Intelligence |
| ANOVA | Analysis of variance |
| ATP | Adenosine triphosphate |
| AU | Arbitrary Units |
| AUC | Area Under the Curve |
| CD38 | NADase |
| CFR | Code of Federal Regulations |
| CI | Confidence Interval |
| DNA | Deoxyribonucleic Acid |
| GCP | Good Clinical Practice |
| GU | Glossymeter Units |
| ICH | International Council for Harmonization |
| IRB (HSS/OHRP) | Institutional Review Board (Health and Human Services/Office for Human Research Protections) |
| LED | Light Emitting Diode |
| N | Number of participants |
| NA | Nicotinic acid |
| NAD+ | Nicotinamide adenine dinucleotide |
| NADH | Reduced nicotinamide adenine dinucleotide |
| NAM | Nicotinamide (niacinamide) |
| NAMPT | Nicotinamide phosphoribosyl transferase |
| NCS | NanoCloud™ sachet |
| NMN | Nicotinamide mononucleotide |
| NR | Nicotinamide (niacinamide) riboside |
| PARP | Poly (ADP-Ribose) Polymerase |
| PDI | Primary Dermal Irritation |
| PRO | Participant Reported Outcome |
| RH | Relative Humidity |
| RIPT | Repeat Irritation Patch Test |
| SD | Standard Deviation |
| SEM | Standard Error of the Mean |
| TEWL | Transepidermal water loss |
| Type IV hypertension | Isolated systolic hypertension |
| UV | Ultra Violet |
References
- Massudi, H.; Grant, R.; Braidy, N.; Guest, J.; Farnsworth, B.; Guillemin, G.J. Age-Associated Changes In Oxidative Stress and NAD+ Metabolism In Human Tissue. Plos One 2012, 7, e42357. [Google Scholar] [CrossRef]
- Kang, S.; Park, J.; Cho, E.; Kim, D.; Ye, S.; Jeong, E.T.; Jun, S.-H.; Kang, N.-G. Distinctive Gene Expression Profiles and Biological Responses of Skin Fibroblasts to Nicotinamide Mononucleotide: Implications for Longevity Effects on Skin. Biomedicines 2025, 13, 2395. [Google Scholar] [CrossRef]
- Tan, C.L.; Chin, T.; Tan, C.Y.R.; Rovito, H.A.; Quek, L.S.; Oblong, J.E.; Bellanger, S. Nicotinamide Metabolism Modulates the Proliferation/Differentiation Balance and Senescence of Human Primary Keratinocytes. J Invest Dermatol 2019, 139, 1638–1647.e3. [Google Scholar] [CrossRef]
- Cantó, C.; Menzies, K.J.; Auwerx, J. NAD+ Metabolism and the Control of Energy Homeostasis: A Balancing Act between Mitochondria and the Nucleus. Cell Metab 2015, 22, 31–53. [Google Scholar] [CrossRef]
- Papaccio, F.; D′Arino, A.; Caputo, S.; Bellei, B. Focus on the Contribution of Oxidative Stress in Skin Aging. Antioxidants 2022, 11, 1121. [Google Scholar] [CrossRef] [PubMed]
- Velarde, M.C.; Flynn, J.M.; Day, N.U.; Melov, S.; Campisi, J. Mitochondrial Oxidative Stress Caused by Sod2 Deficiency Promotes Cellular Senescence and Aging Phenotypes in the Skin. Aging (Albany NY) 2012, 4, 3–12. [Google Scholar] [CrossRef]
- Verdin, E. NAD+ in Aging, Metabolism, and Neurodegeneration. Science 2015, 350, 1208–1213. [Google Scholar] [CrossRef]
- Amjad, S.; Nisar, S.; Bhat, A.A.; Shah, A.R.; Frenneaux, M.P.; Fakhro, K.; Haris, M.; Reddy, R.; Patay, Z.; Baur, J.; et al. Role of NAD+ in Regulating Cellular and Metabolic Signaling Pathways. Mol Metab 2021, 49, 101195. [Google Scholar] [CrossRef] [PubMed]
- Boo, Y.C. Mechanistic Basis and Clinical Evidence for the Applications of Nicotinamide (Niacinamide) to Control Skin Aging and Pigmentation. Antioxidants 2021, 10, 1315. [Google Scholar] [CrossRef] [PubMed]
- Bhasin, S.; Seals, D.; Migaud, M.; Musi, N.; Baur, J.A. Nicotinamide Adenine Dinucleotide in Aging Biology: Potential Applications and Many Unknowns. Endocr. Rev. 2023. [Google Scholar] [CrossRef]
- Iqbal, T.; Nakagawa, T. The Therapeutic Perspective of NAD+ Precursors in Age-Related Diseases. Biochem. Biophys. Res. Commun. 2024, 149590. [Google Scholar] [CrossRef] [PubMed]
- Fang, E.F.; Lautrup, S.; Hou, Y.; Demarest, T.G.; Croteau, D.L.; Mattson, M.P.; Bohr, V.A. NAD+ in Aging: Molecular Mechanisms and Translational Implications. Trends Mol Med 2017, 23, 899–916. [Google Scholar] [CrossRef] [PubMed]
- Covarrubias, A.J.; Perrone, R.; Grozio, A.; Verdin, E. NAD+ Metabolism and Its Roles in Cellular Processes during Ageing. Nat Rev Mol Cell Bio 2021, 22, 119–141. [Google Scholar] [CrossRef]
- Camacho-Pereira, J.; Tarragó, M.G.; Chini, C.C.S.; Nin, V.; Escande, C.; Warner, G.M.; Puranik, A.S.; Schoon, R.A.; Reid, J.M.; Galina, A.; et al. CD38 Dictates Age-Related NAD Decline and Mitochondrial Dysfunction through an SIRT3-Dependent Mechanism. Cell Metab 2016, 23, 1127–1139. [Google Scholar] [CrossRef]
- Chini, C.; Hogan, K.A.; Warner, G.M.; Tarragó, M.G.; Peclat, T.R.; Tchkonia, T.; Kirkland, J.L.; Chini, E. The NADase CD38 Is Induced by Factors Secreted from Senescent Cells Providing a Potential Link between Senescence and Age-Related Cellular NAD+ Decline. Biochem Bioph Res Co 2019, 513, 486–493. [Google Scholar] [CrossRef]
- Bissett, D.L.; Oblong, J.E.; Berge, C.A. Niacinamide: A B Vitamin That Improves Aging Facial Skin Appearance. Dermatol Surg 2005, 31, 860–866. [Google Scholar] [CrossRef] [PubMed]
- Strømland, Ø.; Diab, J.; Ferrario, E.; Sverkeli, L.J.; Ziegler, M. The Balance between NAD+ Biosynthesis and Consumption in Ageing. Mech Ageing Dev 2021, 199, 111569. [Google Scholar] [CrossRef]
- Katayoshi, T.; Nakajo, T.; Tsuji-Naito, K. Restoring NAD+ by NAMPT Is Essential for the SIRT1/P53-Mediated Survival of UVA- and UVB-Irradiated Epidermal Keratinocytes. J Photochem Photobiology B Biology 2021, 221, 112238. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Wang, D.D.; Qiu, Y.; Airhart, S.; Liu, Y.; Stempien-Otero, A.; O’Brien, K.D.; Tian, R. Boosting NAD Level Suppresses Inflammatory Activation of PBMC in Heart Failure. J. Clin. Investig. 2020, 130, 6054–6063. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Singh, K.; Lin, A.; Meadows, A.M.; Wu, K.; Shing, V.; Bley, M.; Hassanzadeh, S.; Huffstutler, R.D.; Schmidt, M.S.; et al. Boosting NAD+ Blunts Toll-like Receptor-4 Induced Type-I Interferon in Control and Systemic Lupus Erythematosus Monocytes. J. Clin. Investig. 2022, 132, e139828. [Google Scholar] [CrossRef]
- Martens, C.R.; Denman, B.A.; Mazzo, M.R.; Armstrong, M.L.; Reisdorph, N.; McQueen, M.B.; Chonchol, M.; Seals, D.R. Chronic Nicotinamide Riboside Supplementation Is Well-Tolerated and Elevates NAD+ in Healthy Middle-Aged and Older Adults. Nat. Commun. 2018, 9, 1286. [Google Scholar] [CrossRef]
- Presterud, R.; Deng, W.H.; Wennerström, A.B.; Burgers, T.; Gajera, B.; Mattsson, K.; Solberg, A.; Fang, E.F.; Nieminen, A.I.; Stray-Pedersen, A.; et al. Long-Term Nicotinamide Riboside Use Improves Coordination and Eye Movements in Ataxia Telangiectasia. Mov. Disord. 2024, 39, 360–369. [Google Scholar] [CrossRef]
- Vallbona-Garcia, A.; Tribble, J.R.; Gustavsson, S.T.; Benedikter, B.J.; Lindsey, P.J.; Webers, C.A.B.; Smeets, H.J.M.; Jóhannesson, G.; Gorgels, T.G.; Williams, P.A. Short-Term High-Dose Nicotinamide Treatment across Glaucoma Subtypes Reveals Increased mtDNA Content and Minimal Metabolomic Change in Blood. Exp. Eye Res. 2025, 263, 110807. [Google Scholar] [CrossRef]
- Wu, C.; Kupferschmid, A.C.; Chen, L.; McManus, A.J.; Kivisäkk, P.; Galler, J.A.; Schwab, N.A.; DesRuisseaux, L.A.; Williams, V.J.; Gerber, J.; et al. Cognitive and Alzheimer’s Disease Biomarker Effects of Oral Nicotinamide Riboside (NR) Supplementation in Older Adults with Subjective Cognitive Decline and Mild Cognitive Impairment. Alzheimer’s Dement.: Transl. Res. Clin. Interv. 2025, 11. [Google Scholar] [CrossRef]
- Ahmadi, A.; Valencia, A.P.; Begue, G.; Norman, J.E.; Fan, S.; Durbin-Johnson, B.P.; Jenner, B.N.; Campbell, M.D.; Reyes, G.; Kapahi, P.; et al. A Pilot Trial of Nicotinamide Riboside and Coenzyme Q10 on Inflammation and Oxidative Stress in Chronic Kidney Disease. JAMA 2025, 267, 1844–1844. [Google Scholar] [CrossRef]
- Berven, H.; Kverneng, S.; Sheard, E.; Søgnen, M.; Geijerstam, S.A.A.; Haugarvoll, K.; Skeie, G.-O.; Dölle, C.; Tzoulis, C. NR-SAFE: A Randomized, Double-Blind Safety Trial of High Dose Nicotinamide Riboside in Parkinson’s Disease. Nat. Commun. 2023, 14, 7793. [Google Scholar] [CrossRef] [PubMed]
- Akasaka, H.; Nakagami, H.; Sugimoto, K.; Yasunobe, Y.; Minami, T.; Fujimoto, T.; Yamamoto, K.; Hara, C.; Shiraki, A.; Nishida, K.; et al. Effects of Nicotinamide Mononucleotide on Older Patients with Diabetes and Impaired Physical Performance: A Prospective, Placebo-controlled, Double-blind Study. Geriatr. Gerontol. Int. 2023, 23, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Morifuji, M.; Higashi, S.; Ebihara, S.; Nagata, M. Ingestion of β-Nicotinamide Mononucleotide Increased Blood NAD Levels, Maintained Walking Speed, and Improved Sleep Quality in Older Adults in a Double-Blind Randomized, Placebo-Controlled Study. GeroScience 2024, 46, 4671–4688. [Google Scholar] [CrossRef]
- Wagner, R.N.; Hofbauer, J.P.; Wally, V.; Kofler, B.; Schmuth, M.; Rosa, L.D.; Luca, M.D.; Bauer, J.W. Epigenetic and Metabolic Regulation of Epidermal Homeostasis. Exp. Dermatol 2021, 30, 1009–1022. [Google Scholar] [CrossRef] [PubMed]
- Moriwaki, S.; Takahashi, Y. Photoaging and DNA Repair. J. Dermatol. Sci. 2008, 50, 169–176. [Google Scholar] [CrossRef]
- Hussein, R.S.; Dayel, S.B.; Abahussein, O.; El-Sherbiny, A.A. Influences on Skin and Intrinsic Aging: Biological, Environmental, and Therapeutic Insights. J. Cosmet. Dermatol. 2025, 24, e16688. [Google Scholar] [CrossRef]
- Furman, D.; Auwerx, J.; Bulteau, A.-L.; Church, G.; Couturaud, V.; Crabbe, L.; Davies, K.J.A.; Decottignies, A.; Gladyshev, V.N.; Kennedy, B.K.; et al. Skin Health and Biological Aging. Nat. Aging 2025, 5, 1195–1206. [Google Scholar] [CrossRef]
- Nguyen, D.D.N.; Vu, D.M.; Vo, N.; Tran, N.H.B.; Ho, D.T.K.; Nguyen, T.; Nguyen, T.A.; Nguyen, H.; Tu, L.N. Skin Rejuvenation and Photoaging Protection Using Adipose-derived Stem Cell Extracellular Vesicles Loaded with Exogenous Cargos. Ski. Res. Technol. 2024, 30, e13599. [Google Scholar] [CrossRef]
- SHALITA, A.R.; SMITH, J.G.; PARISH, L.C.; SOFMAN, M.S.; CHALKER, D.K. TOPICAL NICOTINAMIDE COMPARED WITH CLINDAMYCIN GEL IN THE TREATMENT OF INELAMMATORY ACNE VULGARIS. Int. J. Dermatol. 1995, 34, 434–437. [Google Scholar] [CrossRef]
- Marques, C.; Hadjab, F.; Porcello, A.; Lourenço, K.; Scaletta, C.; Abdel-Sayed, P.; Hirt-Burri, N.; Applegate, L.A.; Laurent, A. Mechanistic Insights into the Multiple Functions of Niacinamide: Therapeutic Implications and Cosmeceutical Applications in Functional Skincare Products. Antioxidants 2024, 13, 425. [Google Scholar] [CrossRef] [PubMed]
- Yiasemides, E.; Sivapirabu, G.; Halliday, G.M.; Park, J.; Damian, D.L. Oral Nicotinamide Protects against Ultraviolet Radiation-Induced Immunosuppression in Humans. Carcinogenesis 2009, 30, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Conze, D.; Brenner, C.; Kruger, C.L. Safety and Metabolism of Long-Term Administration of NIAGEN (Nicotinamide Riboside Chloride) in a Randomized, Double-Blind, Placebo-Controlled Clinical Trial of Healthy Overweight Adults. Sci. Rep. 2019, 9, 9772. [Google Scholar] [CrossRef] [PubMed]
- Trammell, S.A.J.; Schmidt, M.S.; Weidemann, B.J.; Redpath, P.; Jaksch, F.; Dellinger, R.W.; Li, Z.; Abel, E.D.; Migaud, M.E.; Brenner, C. Nicotinamide Riboside Is Uniquely and Orally Bioavailable in Mice and Humans. Nat. Commun. 2016, 7, 12948. [Google Scholar] [CrossRef]
- Conze, D.; Brenner, C.; Kruger, C.L. Safety and Metabolism of Long-Term Administration of NIAGEN (Nicotinamide Riboside Chloride) in a Randomized, Double-Blind, Placebo-Controlled Clinical Trial of Healthy Overweight Adults. Sci. Rep. 2019, 9, 9772. [Google Scholar] [CrossRef]
- Elhassan, Y.S.; Kluckova, K.; Fletcher, R.S.; Schmidt, M.S.; Garten, A.; Doig, C.L.; Cartwright, D.M.; Oakey, L.; Burley, C.V.; Jenkinson, N.; et al. Nicotinamide Riboside Augments the Aged Human Skeletal Muscle NAD+ Metabolome and Induces Transcriptomic and Anti-Inflammatory Signatures. Cell Rep. 2019, 28, 1717–1728.e6. [Google Scholar] [CrossRef]
- Airhart, S.E.; Shireman, L.M.; Risler, L.J.; Anderson, G.D.; Gowda, G.A.N.; Raftery, D.; Tian, R.; Shen, D.D.; O’Brien, K.D. An Open-Label, Non-Randomized Study of the Pharmacokinetics of the Nutritional Supplement Nicotinamide Riboside (NR) and Its Effects on Blood NAD+ Levels in Healthy Volunteers. PLoS ONE 2017, 12, e0186459. [Google Scholar] [CrossRef]
- Airhart, S.E.; Shireman, L.M.; Risler, L.J.; Anderson, G.D.; Gowda, G.A.N.; Raftery, D.; Tian, R.; Shen, D.D.; O’Brien, K.D. An Open-Label, Non-Randomized Study of the Pharmacokinetics of the Nutritional Supplement Nicotinamide Riboside (NR) and Its Effects on Blood NAD+ Levels in Healthy Volunteers. PLoS ONE 2017, 12, e0186459. [Google Scholar] [CrossRef]
- Remie, C.M.; Roumans, K.H.; Moonen, M.P.; Connell, N.J.; Havekes, B.; Mevenkamp, J.; Lindeboom, L.; de Wit, V.H.; de Weijer, T. van; Aarts, S.A.; et al. Nicotinamide Riboside Supplementation Alters Body Composition and Skeletal Muscle Acetylcarnitine Concentrations in Healthy Obese Humans. Am. J. Clin. Nutr. 2020, 112, 413–426. [Google Scholar] [CrossRef] [PubMed]
- Veenhuis, S.J.G.; van Os, N.J.H.; Janssen, A.J.W.M.; van Gerven, M.H.J.C.; Coene, K.L.M.; Engelke, Udo.F.H.; Wevers, R.A.; Tinnevelt, G.H.; Heine, R. ter; de Warrenburg, B.P.C. van; et al. Nicotinamide Riboside Improves Ataxia Scores and Immunoglobulin Levels in Ataxia Telangiectasia. Mov. Disord. 2021, 36, 2951–2957. [Google Scholar] [CrossRef] [PubMed]
- Brakedal, B.; Dölle, C.; Riemer, F.; Ma, Y.; Nido, G.S.; Skeie, G.O.; Craven, A.R.; Schwarzlmüller, T.; Brekke, N.; Diab, J.; et al. The NADPARK Study: A Randomized Phase I Trial of Nicotinamide Riboside Supplementation in Parkinson’s Disease. Cell Metab. 2022, 34, 396–407.e6. [Google Scholar] [CrossRef]
- Lapatto, H.A.K.; Kuusela, M.; Heikkinen, A.; Muniandy, M.; der Kolk, B.W. van; Gopalakrishnan, S.; Pöllänen, N.; Sandvik, M.; Schmidt, M.S.; Heinonen, S.; et al. Nicotinamide Riboside Improves Muscle Mitochondrial Biogenesis, Satellite Cell Differentiation, and Gut Microbiota in a Twin Study. Sci. Adv. 2023, 9, eadd5163. [Google Scholar] [CrossRef]
- McDermott, M.M.; Martens, C.R.; Domanchuk, K.J.; Zhang, D.; Peek, C.B.; Criqui, M.H.; Ferrucci, L.; Greenland, P.; Guralnik, J.M.; Ho, K.J.; et al. Nicotinamide Riboside for Peripheral Artery Disease: The NICE Randomized Clinical Trial. Nat. Commun. 2024, 15, 5046. [Google Scholar] [CrossRef]
- Berven, H.; Svensen, M.; Eikeland, H.; Tvedten, N.; Sheard, E.V.; Geijerstam, S.A.A.; Søgnen, M.; McCann, A.; Arnsten, L.; Årseth, O.; et al. The NAD-Brain Pharmacokinetic Study of NAD Augmentation in Blood and Brain Using Oral Precursor Supplementation. iScience 2026, 114764. [Google Scholar] [CrossRef]
- Shoji, M.; Kato, H.; Koshizaka, M.; Kaneko, H.; Baba, Y.; Ishikawa, T.; Teramoto, N.; Kinoshita, D.; Yamaguchi, A.; Maeda, Y.; et al. Nicotinamide Riboside Supplementation Benefits in Patients With Werner Syndrome: A Double-Blind Randomized Crossover Placebo-Controlled Trial. Aging Cell 2025, 24, e70093. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-Y.; Reynolds, W.C.; Abril, I.; McManus, A.J.; Brenner, C.; González-Irizarry, G.; Gutiérrez-Martínez, L.; Sun, O.; Rosand, J.; Tanzi, R.E.; et al. Effects of Nicotinamide Riboside on NAD+ Levels, Cognition, and Symptom Recovery in Long-COVID: A Randomized Controlled Trial. eClinicalMedicine 2025, 89, 103633. [Google Scholar] [CrossRef]
- Boshi Resources. Available online: https://boshibeauty.com/resources/ (accessed on 7 March 2026).
- Jr., M.D.J.F.F. Fisher’s Contact Dermatitis, 6th ed.; Rietschel, R.L., Ed.; Pmph Usa: Philadelphia, 2008; ISBN 978-1550093780. [Google Scholar]
- Dollerup, O.L.; Christensen, B.; Svart, M.; Schmidt, M.S.; Sulek, K.; Ringgaard, S.; Stødkilde-Jørgensen, H.; Møller, N.; Brenner, C.; Treebak, J.T.; et al. A Randomized Placebo-Controlled Clinical Trial of Nicotinamide Riboside in Obese Men: Safety, Insulin-Sensitivity, and Lipid-Mobilizing Effects. Am. J. Clin. Nutr. 2018, 108, 343–353. [Google Scholar] [CrossRef]
- Wang, D.D.; Airhart, S.E.; Zhou, B.; Shireman, L.M.; Jiang, S.; Rodriguez, C.M.; Kirkpatrick, J.N.; Shen, D.D.; Tian, R.; O’Brien, K.D. Safety and Tolerability of Nicotinamide Riboside in Heart Failure With Reduced Ejection Fraction. JACC: Basic Transl. Sci. 2022, 7, 1183–1196. [Google Scholar] [CrossRef]
- Bieganowski, P.; Brenner, C. Discoveries of Nicotinamide Riboside as a Nutrient and Conserved NRK Genes Establish a Preiss-Handler Independent Route to NAD+ in Fungi and Humans. Cell 2004, 117, 495–502. [Google Scholar] [CrossRef]
- Council for Responsible Nutrition (CRN);© Copyright 2025 Council for Responsible Nutrition; Published by; Washington, D.C.
- Panel, C.I.R.E. Final Report of the Safety Assessment of Niacinamide and Niacin1. Int J Toxicol 2005, 24, 31–Jan. [Google Scholar] [CrossRef]
- Bissett, D.L.; Miyamoto, K.; Sun, P.; Li, J.; Berge, C.A. Topical Niacinamide Reduces Yellowing, Wrinkling, Red Blotchiness, and Hyperpigmented Spots in Aging Facial Skin1. Int. J. Cosmet. Sci. 2004, 26, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, L.; Huang, J.; Zhao, L. Skin-Condition-Improving Effects of Nicotinamide Riboside Contained Supplement: A Randomized Clinical Trial in Naturally Aged Asian Women. Curr. Dev. Nutr. 2024, 8, 102227. [Google Scholar] [CrossRef]
- Shoji, M.; Kato, H.; Koshizaka, M.; Kaneko, H.; Baba, Y.; Ishikawa, T.; Teramoto, N.; Kinoshita, D.; Yamaguchi, A.; Maeda, Y.; et al. Nicotinamide Riboside Supplementation Benefits in Patients With Werner Syndrome: A Double-Blind Randomized Crossover Placebo-Controlled Trial. Aging Cell 2025, 24, e70093. [Google Scholar] [CrossRef]
- Yoshino, J.; Baur, J.A.; Imai, S. NAD+ Intermediates: The Biology and Therapeutic Potential of NMN and NR. Cell Metab 2017, 27, 513–528. [Google Scholar] [CrossRef] [PubMed]
- Amini-Nik, S.; Abdullahi, A.; Vinaik, R.; Yao, R.J.R.; Yu, N.; Datu, A.; Belo, C.; Jeschke, M.G. Aging Impairs the Cellular Interplay between Myeloid Cells and Mesenchymal Cells during Skin Healing in Mice. Aging Dis. 2022, 13, 540–551. [Google Scholar] [CrossRef]
- Taburet, A.M.; Singlas, E.; Glass, R.C.; Thomas, F.; Leutenegger, E. Pharmacokinetic Comparison of Oral and Local Action Transcutaneous Flurbiprofen in Healthy Volunteers. J. Clin. Pharm. Ther. 1995, 20, 101–107. [Google Scholar] [CrossRef]
- Baker, P.; Huang, C.; Radi, R.; Moll, S.B.; Jules, E.; Arbiser, J.L. Skin Barrier Function: The Interplay of Physical, Chemical, and Immunologic Properties. Cells 2023, 12, 2745. [Google Scholar] [CrossRef]
- Martin, R.J.; Denyer, S.P.; Hadgraft, J. Skin Metabolism of Topically Applied Compounds. Int. J. Pharm. 1987, 39, 23–32. [Google Scholar] [CrossRef]
- Chen, A.C.; Martin, A.J.; Choy, B.; Pablo, F.-P.; Dalziell, R.A.; McKenzie, C.A.; Scolyer, R.A.; Dhillon, H.M.; Vardy, J.L.; Kricker, A.; et al. A Phase 3 Randomized Trial of Nicotinamide for Skin-Cancer Chemoprevention. N. Engl. J. Med. 2015, 373, 1618–1626. [Google Scholar] [CrossRef] [PubMed]
- Gudmundsrud, R.; Skjånes, T.H.; Gilmour, B.C.; Caponio, D.; Lautrup, S.; Fang, E.F. Crosstalk among DNA Damage, Mitochondrial Dysfunction, Impaired Mitophagy, Stem Cell Attrition, and Senescence in the Accelerated Ageing Disorder Werner Syndrome. Cytogenet Genome Res 2021, 161, 297–304. [Google Scholar] [CrossRef]
- Clarys, P.; Clijsen, R.; Taeymans, J.; Barel, A.O. Hydration Measurements of the Stratum Corneum: Comparison between the Capacitance Method (Digital Version of the Corneometer CM 825®) and the Impedance Method (Skicon-200EX®). Ski. Res. Technol. 2012, 18, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Christman, J.C.; Fix, D.K.; Lucus, S.C.; Watson, D.; Desmier, E.; Wilkerson, R.J.J.; Fixler, C. Two Randomized, Controlled, Comparative Studies of the Stratum Corneum Integrity Benefits of Two Cosmetic Niacinamide/Glycerin Body Moisturizers vs. Conventional Body Moisturizers. J. drugs Dermatol.: JDD 2012, 11, 22–29. [Google Scholar] [PubMed]
- Fluhr, J.W.; Wiora, G.; Nikolaeva, D.G.; Miséry, L.; Darlenski, R. In Vivo Transepidermal Water Loss: Validation of a New Multi-sensor Open Chamber Water Evaporation System Tewameter TM Hex. Ski. Res. Technol. 2023, 29, e13307. [Google Scholar] [CrossRef] [PubMed]
- Anqi, S.; Xiukun, S.; Ai’e, X. Quantitative Evaluation of Sensitive Skin by ANTERA 3D® Combined with GPSkin Barrier®. Ski. Res. Technol. 2022, 28, 840–845. [Google Scholar] [CrossRef]
- Matias, A.R.; Ferreira, M.; Costa, P.; Neto, P. Skin Colour, Skin Redness and Melanin Biometric Measurements: Comparison Study between Antera® 3D, Mexameter® and Colorimeter®. Ski. Res. Technol. 2015, 21, 346–362. [Google Scholar] [CrossRef]
- Kollias, N.; Gillies, R.; Muccini, J.A.; Uyeyama, R.K.; Phillips, S.B.; Drake, L.A. A Single Parameter, Oxygenated Hemoglobin, Can Be Used to Quantify Experimental Irritant-Induced Inflammation. J. Investig. Dermatol. 1995, 104, 421–424. [Google Scholar] [CrossRef]
- Nisbet, S.J.; Targett, D.; Rawlings, A.V.; Qian, K.; Wang, X.; Lin, C.B.; Thompson, M.A.; Bulsara, P.A.; Moore, D.J. Clinical and in Vitro Evaluation of New Anti-redness Cosmetic Products in Subjects with Winter Xerosis and Sensitive Skin. Int. J. Cosmet. Sci. 2019, 41, 534–547. [Google Scholar] [CrossRef] [PubMed]
- Moy, A.J.; Tunnell, J.W.; Author 1, A.B.; Author 2, C.D.; Imaging in Dermatology. Title of the article. In Abbreviated Journal Name Year, Volume; page range, 2016; pp. 203–215. [Google Scholar] [CrossRef]
|
Test Material# |
Test Material |
24 hr |
72 hr |
Outcomes |
| 1 | 100 mg NR + Water | 3.30% | 3.30% | negligible dermal irritation potential |
| 2 | 50 mg NR + Water | 0.00% | 3.33% | negligible dermal irritation potential |
| 3 | 50 mg NR + Glycerin + Water | 3.30% | 0.00% | negligible dermal irritation potential |
| 4 | NR (50 mg) NCS + Water | 10.00% | 6.67% | minimal dermal irritation potential |
| 5 | NR (50 mg) NCS + Glycerin + Water | 0.00% | 0.00% | no dermal irritation |
| 6 | Glycerin + Water | 0.00% | 0.00% | no dermal irritation |
| 7 | Plain NCS + Water | 3.30% | 3.30% | negligible dermal irritation potential |
| 8 | Plain NCS + Glycerin + Water | 6.67% | 3.33% | negligible dermal irritation potential |
| Induction reading | Challenge reading | Outcomes | ||||||||||||
| 50 mg of NR applied with water | ||||||||||||||
| Grade | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 24 hr | 48 hr | 72 hr | 96 hr | Did not induce dermal sensitization in participants |
| 0 | 119 | 113 | 112 | 110 | 110 | 110 | 111 | 110 | 110 | 106 | 104 | |||
| ± | 1 | 1 | 1 | 1 | 1 | |||||||||
| 1 | 1 | |||||||||||||
| Total | 120 | 114 | 113 | 111 | 111 | 111 | 111 | 110 | 110 | 106 | 104 | |||
| NR in NCS applied with 50/50 glycerin-water | ||||||||||||||
| Grade | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 24 hr | 48 hr | 72 hr | 96 hr | Did not induce dermal sensitization in participants |
| 0 | 116 | 114 | 112 | 113 | 113 | 112 | 111 | 111 | 110 | 109 | 14 | 104 | 9 | |
| ± | ||||||||||||||
| 1 | 1 | |||||||||||||
| - | 92 | 97 | ||||||||||||
| Total | 116 | 114 | 113 | 113 | 113 | 112 | 111 | 111 | 110 | 109 | 106 | 106 | 106 | |
| NR, % | Time (hr) | Plumpness | Smoothness | Moisture | Softness |
| 0.50% | 1 | 112.8 | 116.2 | 106.4 | 112.8 |
| 4 | 222.6 | 225.8 | 216.2 | 222.6 | |
| 8 | 332.2 | 335.8 | 325.8 | 325.8 | |
| 1.00% | 1 | 93.5 | 103.3 | 93.5 | 103.3 |
| 4 | 203.4 | 212.7 | 203.4 | 212.7 | |
| 8 | 312.6 | 322.6 | 312.6 | 322.6 | |
| 1.50% | 1 | 80.7 | 83.9 | 83.9 | 83.9 |
| 4 | 193.5 | 203.4 | 203.4 | 193.5 | |
| 8 | 306.4 | 306.4 | 306.4 | 312.6 | |
| 2.50% | 1 | 74.1 | 74.1 | 67.7 | 77.5 |
| 4 | 177.5 | 187.1 | 187.1 | 183.9 | |
| 8 | 293.5 | 297.1 | 287.1 | 297.1 | |
| 5.00% | 1 | 70.6 | 67.7 | 67.7 | 77.5 |
| 4 | 167.7 | 177.5 | 177.5 | 183.9 | |
| 8 | 273.9 | 283.9 | 277.5 | 283.9 | |
| 10.00% | 1 | 67.7 | 74.1 | 67.7 | 77.5 |
| 4 | 177.5 | 187.1 | 177.5 | 183.9 | |
| 8 | 287.1 | 297.1 | 287.1 | 297.1 |
| NR, % | Time (hr) | Moisture Locking | Redness |
| 0.50% | 1 | 135.4 | 138.6 |
| 4 | 238.5 | 242.0 | |
| 8 | 348.4 | 348.4 | |
| 1.00% | 1 | 122.6 | 125.8 |
| 4 | 229.0 | 229.0 | |
| 8 | 329.0 | 332.2 | |
| 1.50% | 1 | 112.8 | 116.2 |
| 4 | 212.7 | 219.4 | |
| 8 | 322.6 | 319.4 | |
| 2.50% | 1 | 93.6 | 93.6 |
| 4 | 203.2 | 200.0 | |
| 8 | 309.6 | 309.6 | |
| 5.00% | 1 | 71.1 | 67.8 |
| 4 | 177.6 | 180.6 | |
| 8 | 286.8 | 286.8 | |
| 10.00% | 1 | 48.5 | 48.5 |
| 4 | 154.9 | 154.9 | |
| 8 | 264.5 | 258.1 |
| NR, % | Time (hr) | Relieves skin sensitivity after use | Improves skin Redness after use | Reported smoothing skin after use | After use, the effect of maintaining skin condition | After use, it can enhance the overall skin barrier |
| 0.50% | 1 | 87.1% | 87.1% | 87.1% | 87.1% | 87.1% |
| 4 | 87.1% | 87.1% | 83.9% | 87.1% | 87.1% | |
| 8 | 83.9% | 87.1% | 83.9% | 87.1% | 87.1% | |
| 1.00% | 1 | 87.1% | 87.1% | 90.3% | 90.3% | 90.3% |
| 4 | 87.1% | 87.1% | 90.3% | 87.1% | 90.3% | |
| 8 | 87.1% | 87.1% | 87.1% | 87.1% | 90.3% | |
| 1.50% | 1 | 90.3% | 90.3% | 90.3% | 90.3% | 93.5% |
| 4 | 90.3% | 90.3% | 90.3% | 90.3% | 90.3% | |
| 8 | 87.1% | 90.3% | 90.3% | 90.3% | 90.3% | |
| 2.50% | 1 | 93.5% | 93.5% | 90.3% | 93.5% | 93.5% |
| 4 | 93.5% | 90.3% | 90.3% | 93.5% | 93.5% | |
| 8 | 90.3% | 90.3% | 90.3% | 93.5% | 93.5% | |
| 5.00% | 1 | 96.8% | 93.5% | 96.8% | 93.5% | 93.5% |
| 4 | 93.5% | 93.5% | 96.8% | 93.5% | 93.5% | |
| 8 | 93.5% | 93.5% | 93.5% | 93.5% | 93.5% | |
| 10.00% | 1 | 96.8% | 96.8% | 96.8% | 96.8% | 96.8% |
| 4 | 96.8% | 93.5% | 96.8% | 96.8% | 96.8% | |
| 8 | 96.8% | 93.5% | 96.8% | 96.8% | 93.5% |
| NR, % | Timepoint(hr) | Brightness | Moisture | Oiliness | Heaviness | Stickiness | Slipperiness |
| 0.50% | 1 | 122.6 | 119.4 | 109.6 | 771.8 | 122.6 | 119.4 |
| 4 | 229.0 | 225.8 | 219.4 | 664.5 | 229.0 | 225.8 | |
| 8 | 342.0 | 335.4 | 329.0 | 561.3 | 339.0 | 339.0 | |
| 1.00% | 1 | 106.4 | 96.9 | 103.3 | 778.2 | 116.1 | 109.7 |
| 4 | 212.7 | 206.4 | 203.4 | 674.9 | 219.4 | 219.7 | |
| 8 | 316.2 | 312.6 | 316.2 | 571.6 | 329.0 | 325.8 | |
| 1.50% | 1 | 93.5 | 87.1 | 93.5 | 786.3 | 116.1 | 106.4 |
| 4 | 203.4 | 197.0 | 203.4 | 683.9 | 219.4 | 212.7 | |
| 8 | 309.7 | 306.4 | 312.6 | 578.0 | 322.6 | 322.6 | |
| 2.50% | 1 | 87.1 | 80.7 | 90.3 | 800.0 | 109.7 | 100.0 |
| 4 | 197.0 | 193.6 | 200.0 | 693.6 | 212.7 | 212.7 | |
| 8 | 306.4 | 306.4 | 312.6 | 590.4 | 319.4 | 319.4 | |
| 5.00% | 1 | 80.7 | 77.5 | 87.1 | 812.1 | 80.7 | 87.1 |
| 4 | 187.1 | 193.5 | 193.5 | 706.4 | 203.4 | 206.4 | |
| 8 | 306.4 | 306.4 | 312.6 | 603.2 | 306.4 | 316.2 | |
| 10.00% | 1 | 64.5 | 67.8 | 74.2 | 829.8 | 61.2 | 64.5 |
| 4 | 180.6 | 174.2 | 183.8 | 723.3 | 174.2 | 177.6 | |
| 8 | 290.4 | 283.8 | 296.8 | 616.1 | 287.4 | 296.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




