Submitted:
20 March 2026
Posted:
23 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
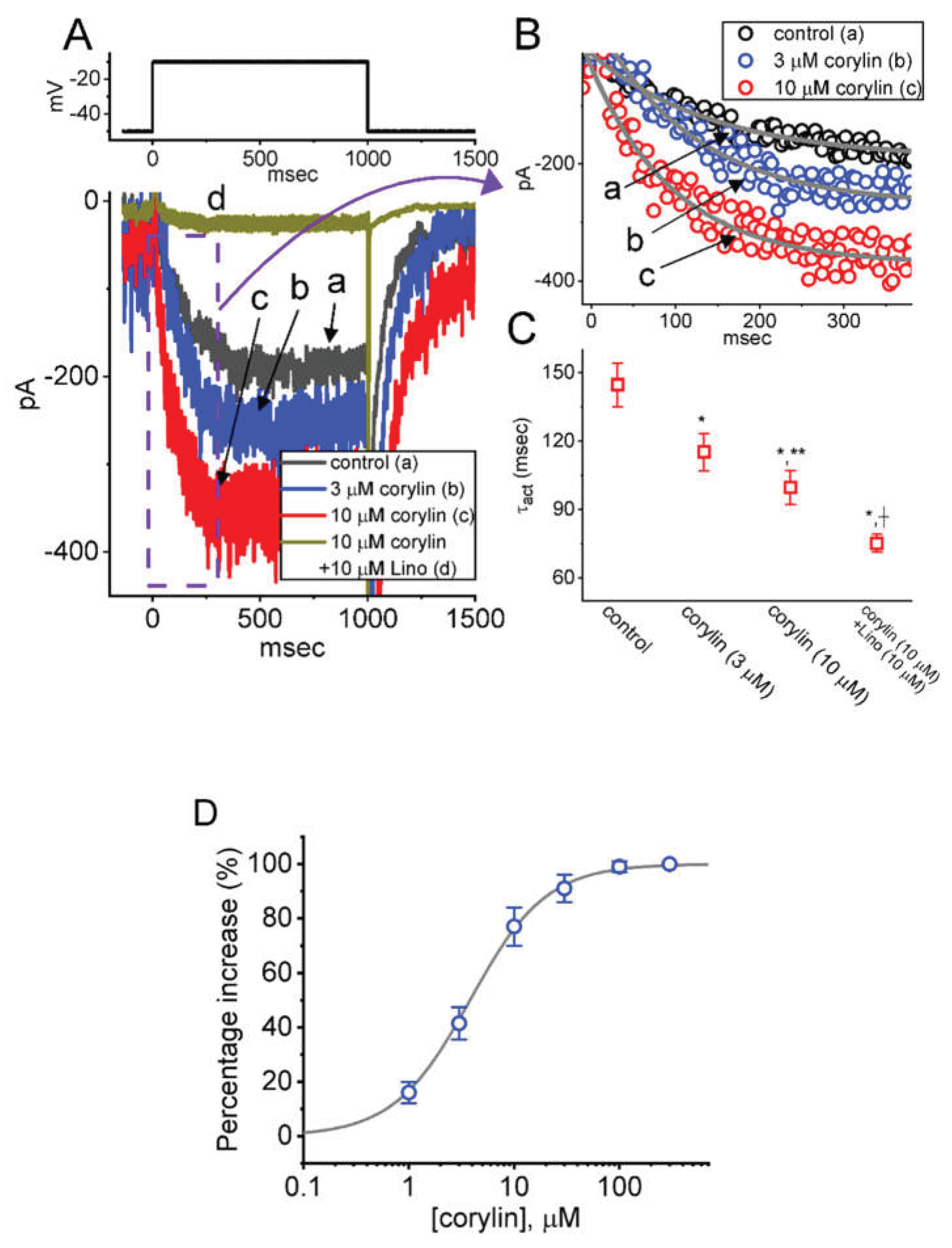
2.1. Stimulatory Effect of Corylin on M-Type K+ Current (IK(M)) Recorded from Pituitary GH3 Cells
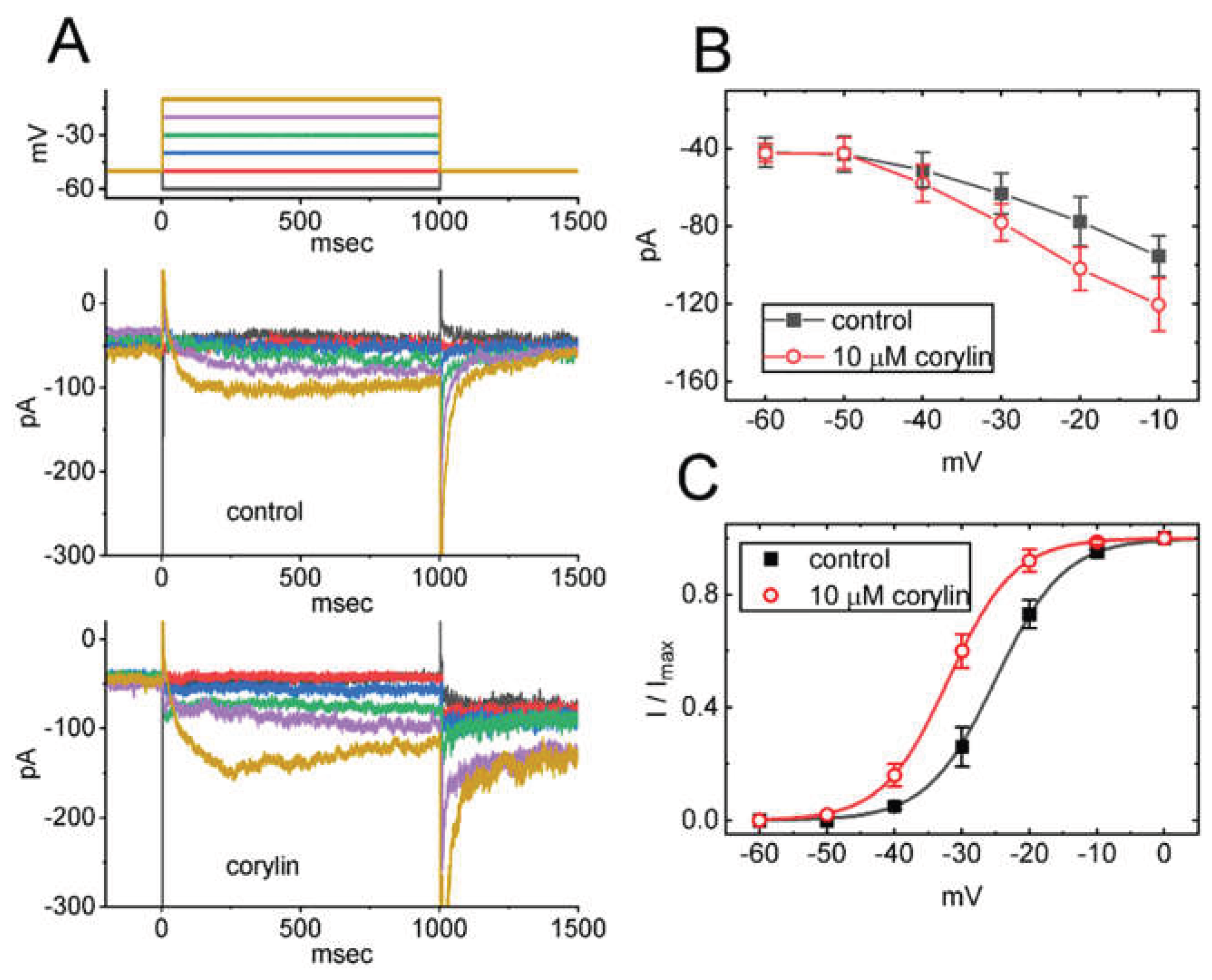
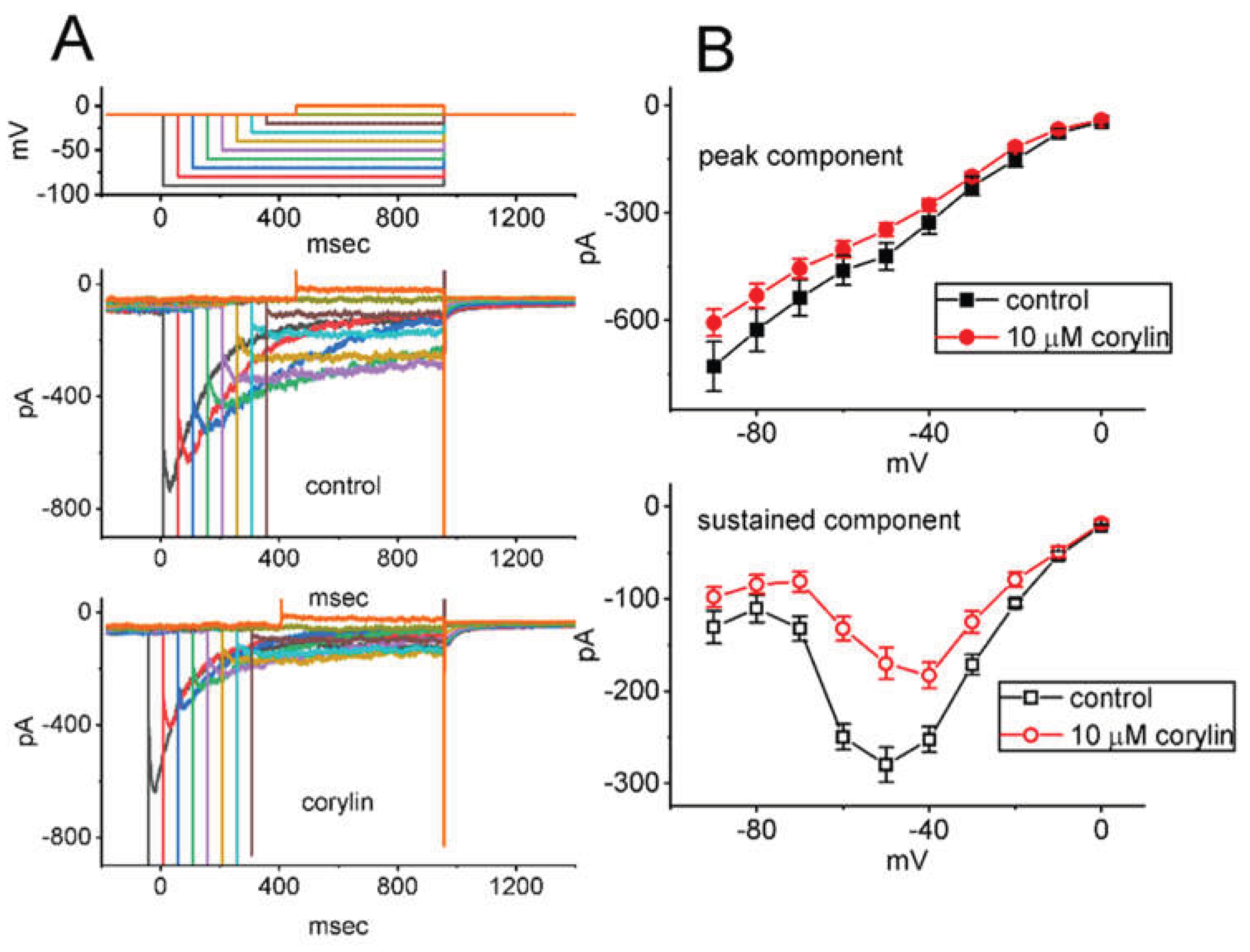
2.2. Effect of Corylin on the Steady-State Current Versus Voltage (I-V) Relation and Activation Curve of IK(M)
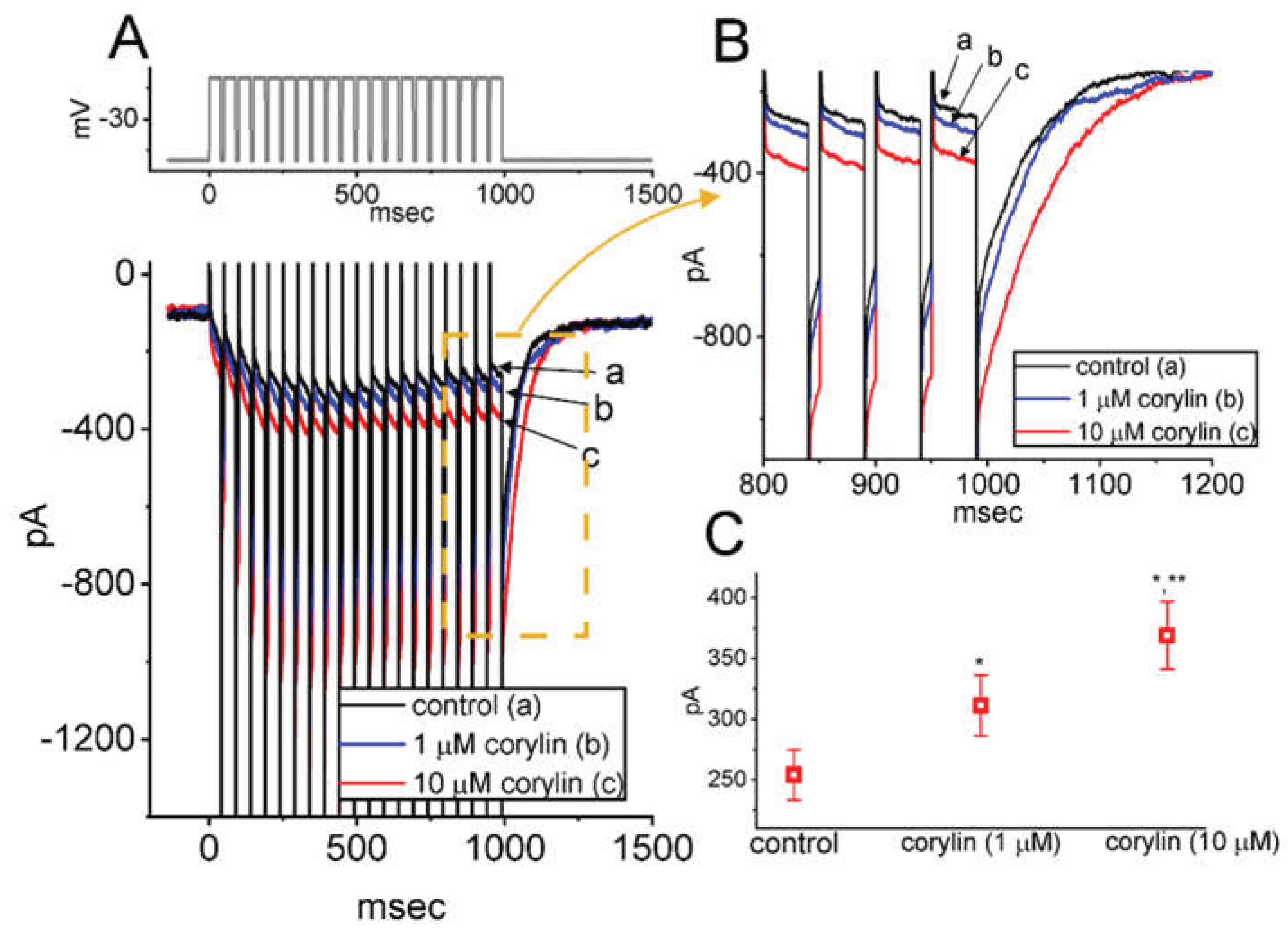
2.3. Corylin Effect on IK(M) Amplitude Evoked During a Train of Depolarizing Command Voltages in GH3 Cells
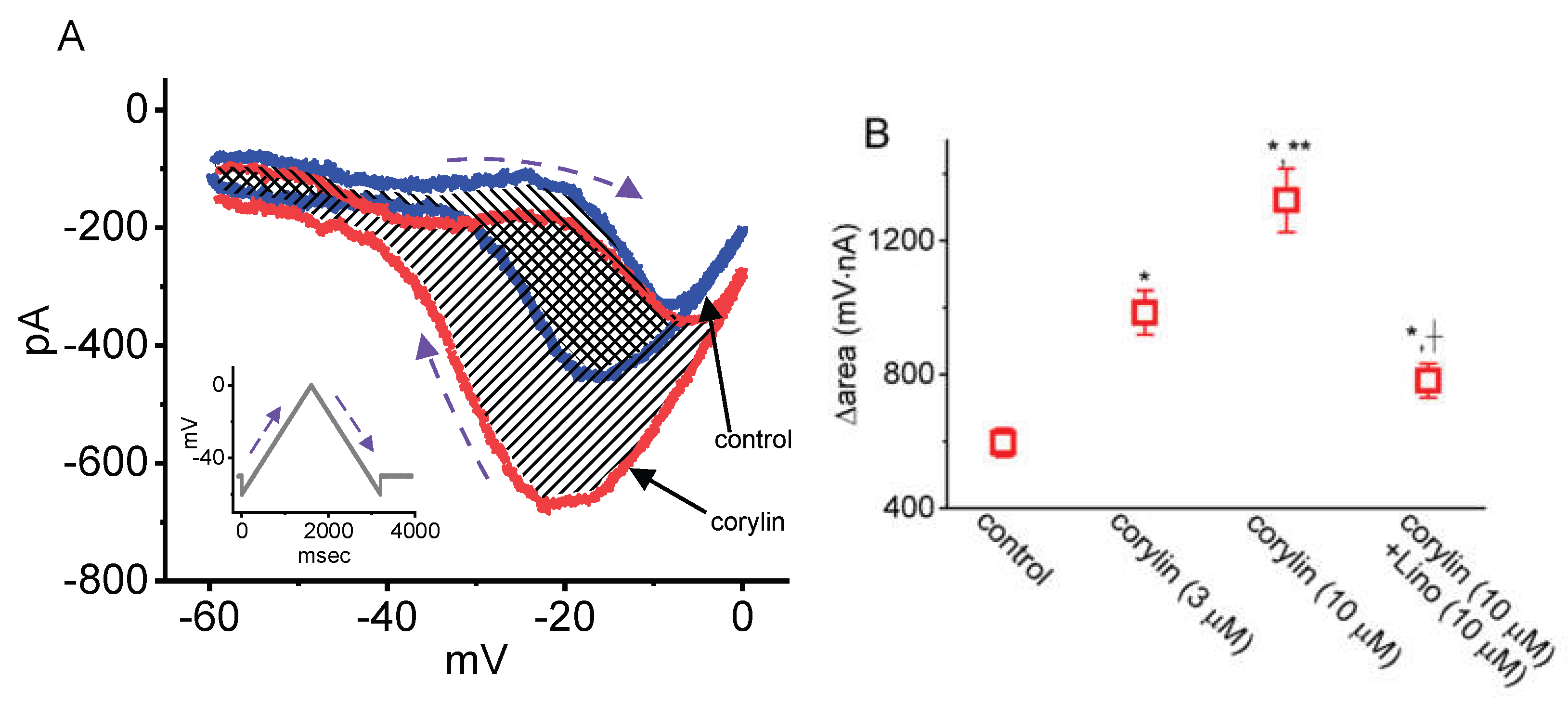
2.4. Augmentation of the Strength in Voltage-Dependent Hysteresis (Hys(V)) of IK(M) Caused by Corylin
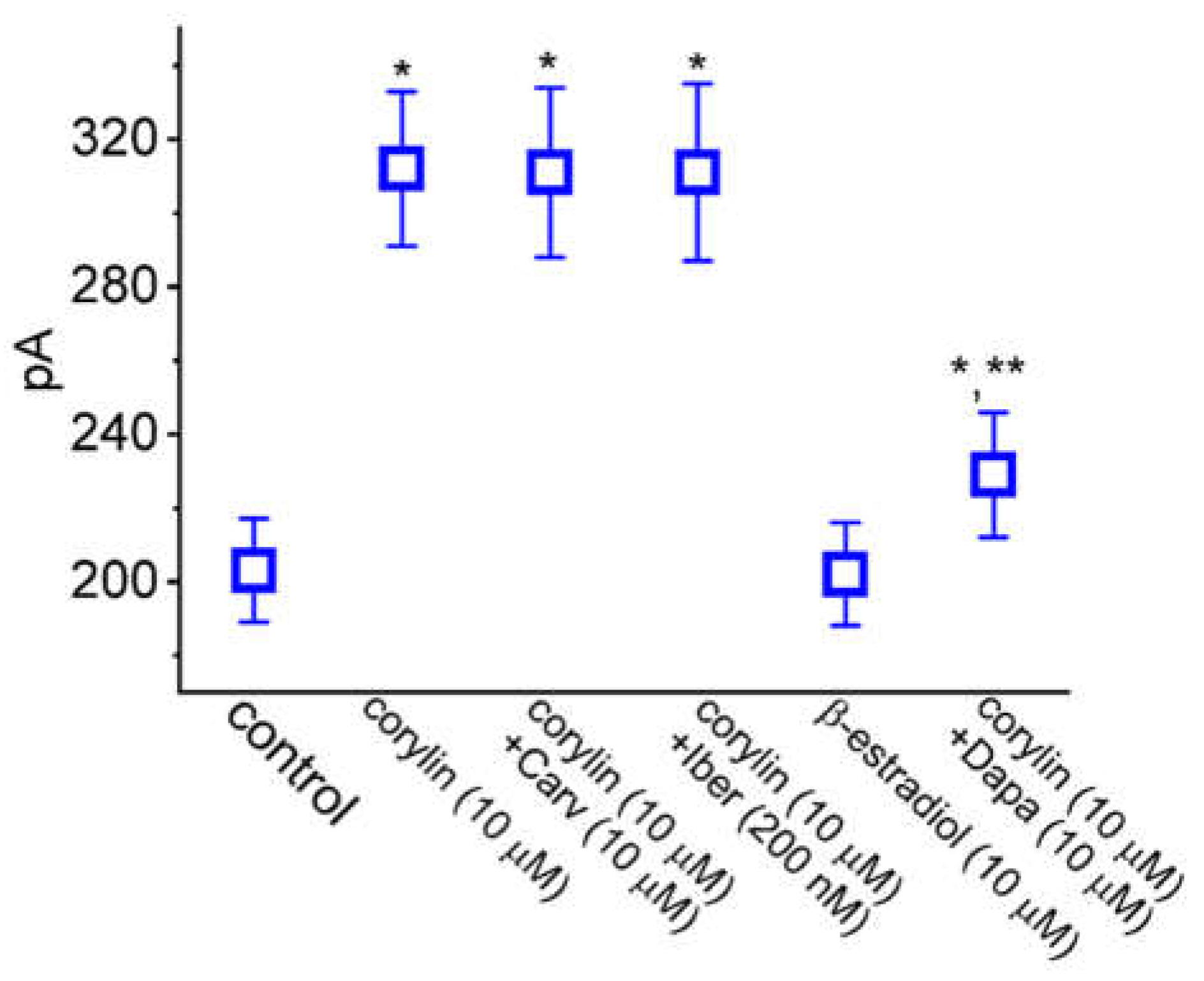
2.5. Comparison Among Effect of Corylin, Corylin Plus Carvedilol (Carv), Corylin Plus Iberiotoxin (Iber), 17β-Estradiol, and Corylin Plus Dapagliflozin (Dapa) on IK(M) Amplitude Observed in GH3 Cells
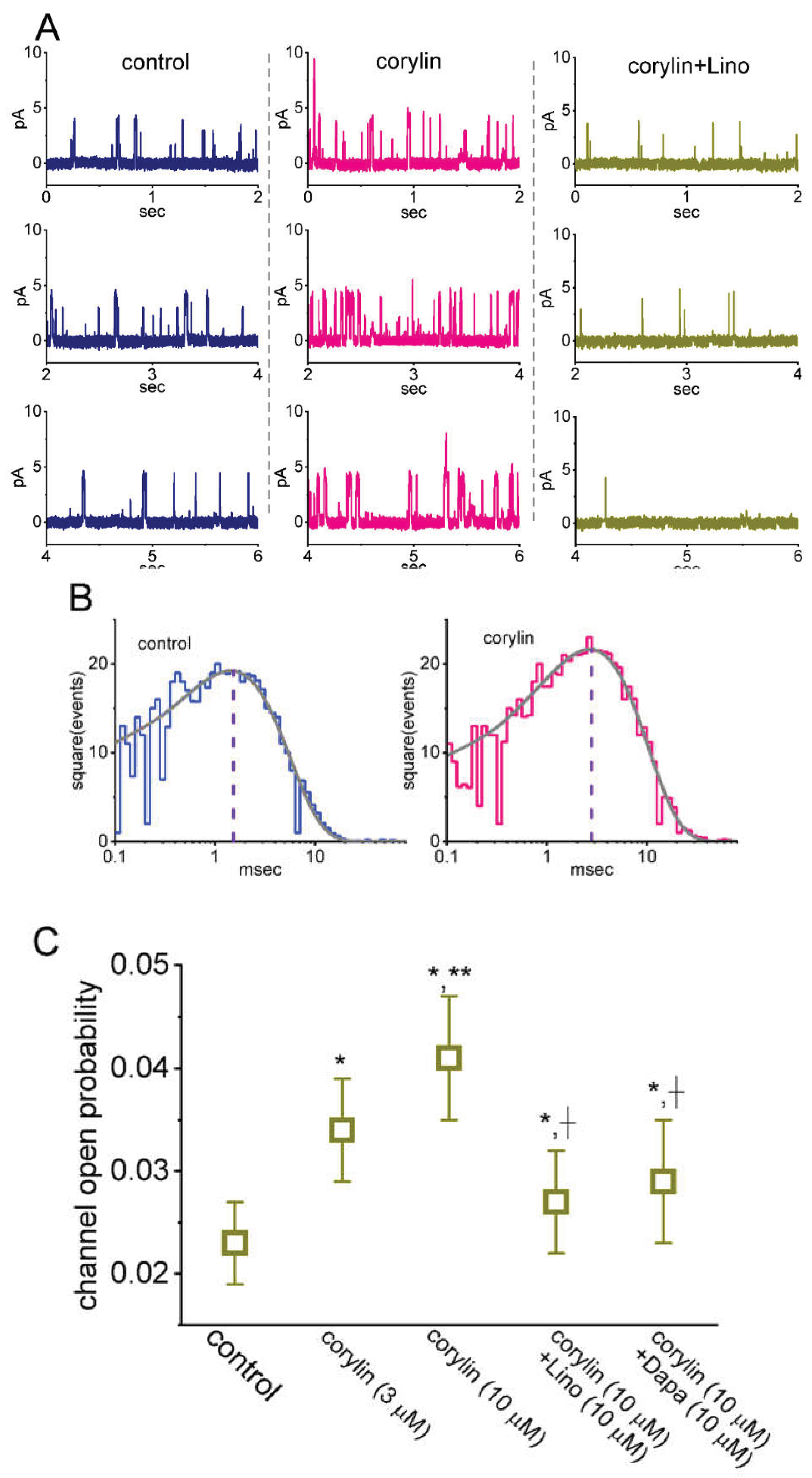
2.6. Effect of Corylin on M-type K+ (KM) Channels Recorded from GH3 Cells
2.7. Effect of Corylin on erg-Mediated K+ Current (IK(erg)) Recorded from GH3 Cells
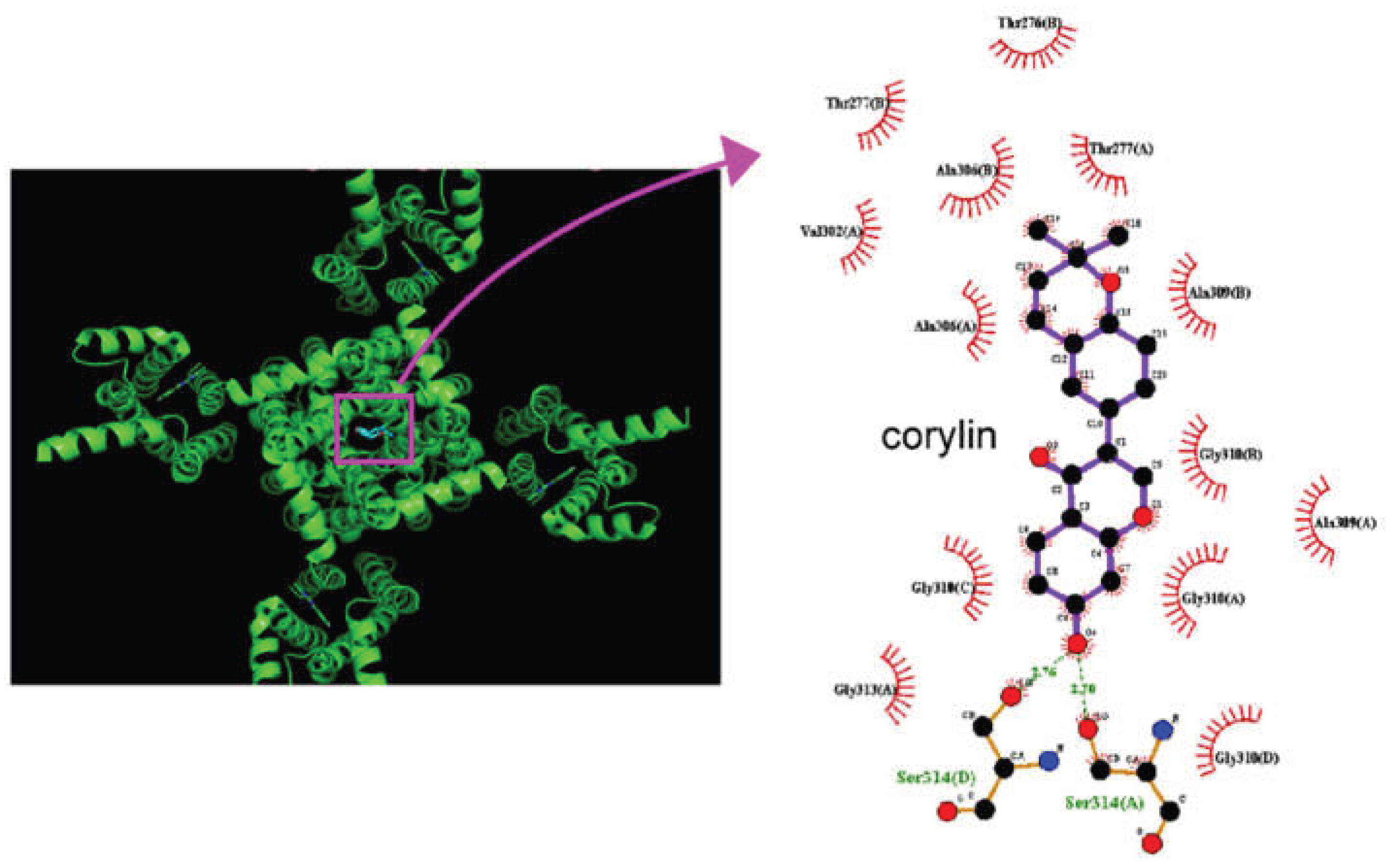
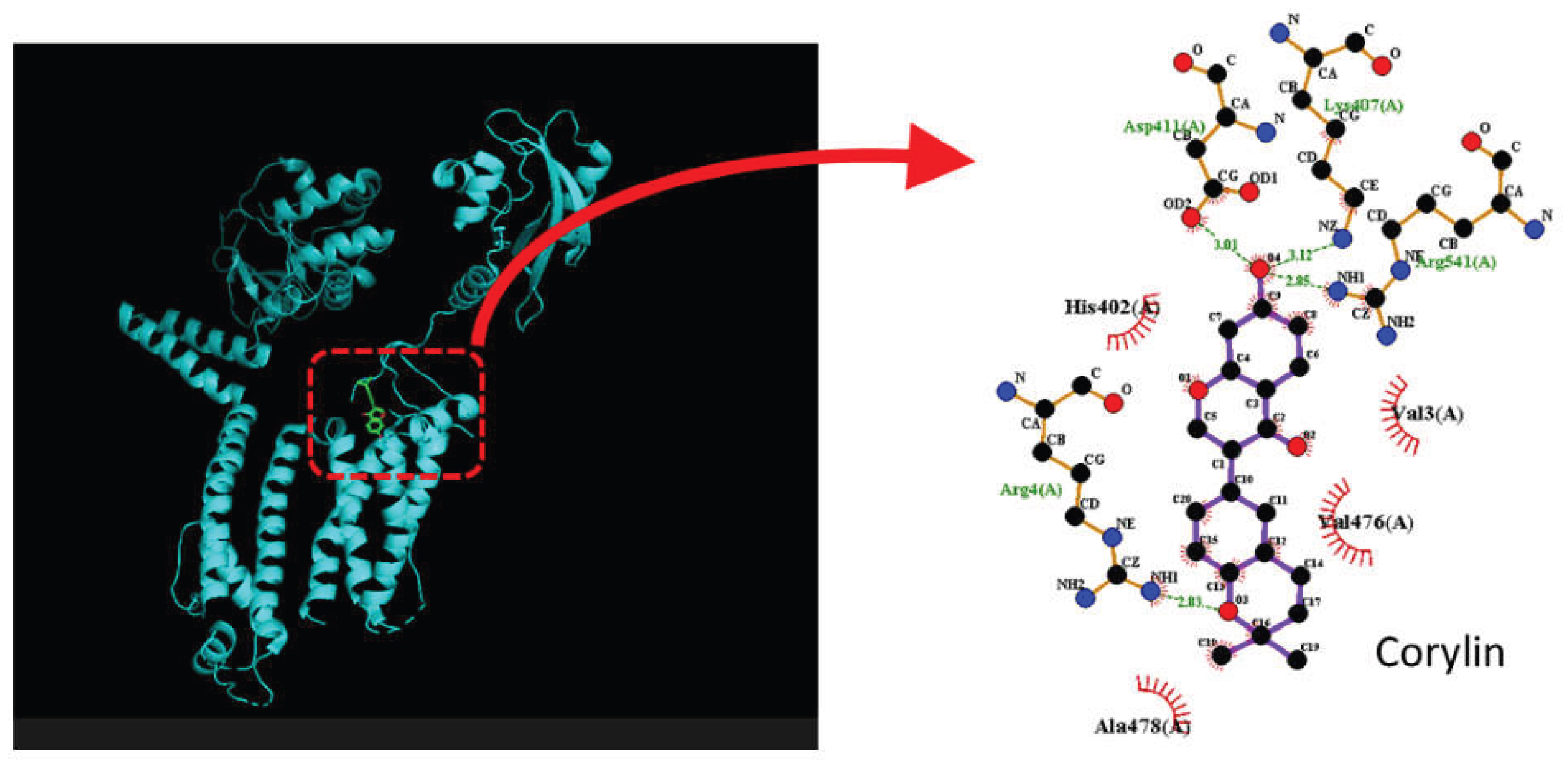
2.8. Docking Results of the Molecular Interactions Between Corylin and the KCNQ2 or KCNH2 Channel
3. Discussion
4. Materials and Methods
4.1. Chemicals, Drugs, Reagents and Solutions
4.2. Cell Preparations
4.3. Electrophysiological Measurements
4.4. Data Analyses
4.5. Single-Channel Analysis of the KM Channel
4.6. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AP | action potential |
| Carv | carvedilol |
| Corylin | 3-(2,2-dimethylchromen-6-yl)-7-hydroxychromen-4-one |
| Dapa | dapagliflozin |
| EC50 | concentration required for 50% stimulation |
| Erg | ether-à-go-go-related gene |
| HERG | human ether-à-go-go-related gene |
| Hys(V) | voltage-dependent hysteresis; |
| I-V | current versus voltage |
| IK(erg) | erg-mediated K+ current |
| IK(M) | M-type K+ current |
| KM channel | M-type K+ (KCNQ/K7) channel |
| Lino | linopirdine |
| SEM | standard error of the mean |
| TTX | tetrodotoxin |
| τact | activation time constant |
| Vramp | ramp voltage |
References
- Khushboo, P.S.; Jadhav, V.M.; Kadam, V.J.; Sathe, N.S. Psoralea corylifolia Linn.-”Kushtanashini”. Pharmacogn. Rev. 2010, 4, 69–76. [Google Scholar] [CrossRef]
- Lee, S.W.; Yun, B.R.; Kim, M.H.; Park, C.S.; Lee, W.S.; Oh, H.M.; Rho, M.C. Phenolic compounds isolated from Psoralea corylifolia inhibit IL-6-induced STAT3 activation. Planta Med. 2012, 78, 903–906. [Google Scholar] [CrossRef]
- Chopra, B.; Dhingra, A.K.; Dhar, K.L. Psoralea corylifolia L. (Buguchi) - folklore to modern evidence: review. Fitoterapia 2013, 90, 44–56. [Google Scholar] [CrossRef]
- Kim, K.A.; Shim, S.H.; Ahn, H.R.; Jung, S.H. Protective effects of the compounds isolated from the seed of Psoralea corylifolia on oxidative stress-induced retinal damage. Toxicol. Appl. Pharmacol. 2013, 269, 109–120. [Google Scholar] [CrossRef]
- Wang, T.X.; Yin, Z.H.; Zhang, W.; Peng, T.; Kang, W.Y. Chemical constituents from Psoralea corylifolia and their antioxidant alpha-glucosidase inhibitory and antimicrobial activities. Zhongguo Zhong Yao Za Zhi 2013, 38, 2328–2333, Chinese. [Google Scholar]
- Alam, F.; Khan, G.N.; Asad, M.H.H.B. Psoralea corylifolia L: Ethnobotanical, biological, and chemical aspects: A review. Phytother. Res. 2018, 32, 597–615. [Google Scholar] [CrossRef] [PubMed]
- Hung, Y.L.; Fang, S.H.; Wang, S.C.; Cheng, W.C.; Liu, P.L.; Su, C.C.; Chen, C.S.; Huang, M.Y.; Hua, K.F.; Shen, K.H.; Wang, Y.T.; Suzuki, K.; Li, C.Y. Corylin protects LPS-induced sepsis and attenuates LPS-induced inflammatory response. Sci. Rep. 2017, 7, 46299. [Google Scholar] [CrossRef]
- Chen, C.C.; Chen, C.Y.; Ueng, S.H.; Hsueh, C.; Yeh, C.T.; Ho, J.Y.; Chou, L.F.; Wang, T.H. Corylin increases the sensitivity of hepatocellular carcinoma cells to chemotherapy through long noncoding RNA RAD51-AS1-mediated inhibition of DNA repair. Cell Death Dis. 2018, 9, 543. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Chen, C.C.; Shieh, T.M.; Hsueh, C.; Wang, S.H.; Leu, Y.L.; Lian, J.H.; Wang, T.H. Corylin Suppresses Hepatocellular Carcinoma Progression via the Inhibition of Epithelial-Mesenchymal Transition, Mediated by Long Noncoding RNA GAS5. Int. J. Mol. Sci. 2018, 19, 380. [Google Scholar] [CrossRef]
- Chen, C.C.; Li, H.Y.; Leu, Y.L.; Chen, Y.J.; Wang, C.J.; Wang, S.H. Corylin Inhibits Vascular Cell Inflammation, Proliferation and Migration and Reduces Atherosclerosis in ApoE-Deficient Mice. Antioxidants (Basel) 2020, 9, 275. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Kuo, C.H.; Leu, Y.L.; Wang, S.H. Corylin reduces obesity and insulin resistance and promotes adipose tissue browning through SIRT-1 and β3-AR activation. Pharmacol. Res. 2021, 164, 105291. [Google Scholar] [CrossRef]
- Chen, I.C.; Wang, S.C.; Chen, Y.T.; Tseng, H.H.; Liu, P.L.; Lin, T.C.; Wu, H.E.; Chen, Y.R.; Tseng, Y.H.; Hsu, J.H.; Dai, Z.K.; Suen, J.L.; Li, C.Y. Corylin Ameliorates LPS-Induced Acute Lung Injury via Suppressing the MAPKs and IL-6/STAT3 Signaling Pathways. Pharmaceuticals (Basel) 2021, 14, 1046. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.Y.; Tu, C.E.; Wang, S.C.; Hung, Y.L.; Su, C.C.; Fang, S.H.; Chen, C.S.; Liu, P.L.; Cheng, W.C.; Huang, Y.W.; Li, C.Y. Corylin inhibits LPS-induced inflammatory response and attenuates the activation of NLRP3 inflammasome in microglia. BMC Complement. Altern. Med. 2018, 18, 221. [Google Scholar] [CrossRef]
- Qin, Z.; Li, S.; Yao, Z.; Hong, X.; Xu, J.; Lin, P.; Zhao, G.; Gonzalez, F.J.; Yao, X. Metabolic profiling of corylin in vivo and in vitro. J. Pharm. Biomed. Anal. 2018, 155, 157–168. [Google Scholar] [CrossRef]
- Che, L.; Yang, H.; Wang, D.; Liu, S. Corylin sensitizes breast cancer cells to overcome tamoxifen resistance by regulating OAS1/miR-22-3p/SIRT1 axis. Acta Biochim. Pol. 2021, 68, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wang, L.; Zhang, R. Corylin suppresses metastasis of breast cancer cells by modulating miR-34c/LINC00963 target. Libyan J. Med. 2021, 16, 1883224. [Google Scholar] [CrossRef]
- Zhao, Z.; Liu, T.; Zhu, S.; Yang, Y.; Wang, Z.; Ma, H.; Wang, X.; Liu, T.; Guo, P.; Pi, J.; Qi, D.; Tian, B.; Liu, Z.; Li, N. Development and evaluation studies of Corylin loaded nanostructured lipid carriers gel for topical treatment of UV-induced skin aging. Exp. Gerontol. 2021, 153, 111499. [Google Scholar] [CrossRef]
- Yu, A.X.; Xu, M.L.; Yao, P.; Kwan, K.K.; Liu, Y.X.; Duan, R.; Dong, T.T.; Ko, R.K.; Tsim, K.W. Corylin, a flavonoid derived from Psoralea Fructus, induces osteoblastic differentiation via estrogen and Wnt/β-catenin signaling pathways. FASEB J. 2020, 34, 4311–4328. [Google Scholar] [CrossRef] [PubMed]
- Yu, A.X.; Xiao, J.; Zhao, S.Z.; Kong, X.P.; Kwan, K.K.; Zheng, B.Z.; Wu, K.Q.; Dong, T.T.; Tsim, K.W. Biological Evaluation and Transcriptomic Analysis of Corylin as an Inhibitor of Osteoclast Differentiation. Int. J. Mol. Sci. 2021, 22, 3540. [Google Scholar] [CrossRef]
- Chang, Z.Y.; Liu, H.M.; Leu, Y.L.; Hsu, C.H.; Lee, T.Y. Modulation of Gut Microbiota Combined with Upregulation of Intestinal Tight Junction Explains Anti-Inflammatory Effect of Corylin on Colitis-Associated Cancer in Mice. Int. J. Mol. Sci. 2022, 23, 2667. [Google Scholar] [CrossRef]
- Lee, Y.; Jun, H.S.; Oh, Y.S. Protective effect of Psoralea corylifolia L. seed extract against palmitate-induced neuronal apoptosis in PC12 cells. Evid. Based Complement. Alternat. Med. 2016 2016, 5410419. [Google Scholar] [CrossRef]
- Kim, Y.J.; Lim, H.S.; Lee, J.; Jeong, S.J. Quantitative Analysis of Psoralea corylifolia Linne and its Neuroprotective and Anti-Neuroinflammatory Effects in HT22 Hippocampal Cells and BV-2 Microglia. Molecules 2016, 21, 1076. [Google Scholar] [CrossRef]
- Wu, S.N.; Chiang, H.T.; Shen, A.Y.; Lo, Y.K. Differential effects of quercetin, a natural polyphenolic flavonoid, on L-type calcium current in pituitary tumor (GH3) cells and neuronal NG108-15 cells. J. Cell. Physiol. 2003, 195, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Fusi, F.; Spiga, O.; Trezza, A.; Sgaragli, G.; Saponara, S. The surge of flavonoids as novel, fine regulators of cardiovascular Cav channels. Eur. J. Pharmacol. 2017, 796, 158–174. [Google Scholar] [CrossRef] [PubMed]
- Redford, K.E.; Abbott, G.W. KCNQ Potassium Channels as Targets of Botanical Folk Medicines. Annu. Rev. Pharmacol. Toxicol. 2022, 62, 447–464. [Google Scholar] [CrossRef]
- Liu, Y.C.; So, E.C.; Wu, S.N. Cannabidiol Modulates M-Type K+ and Hyperpolarization-Activated Cation Currents. Biomedicines 2023, 11, 2651. [Google Scholar] [CrossRef] [PubMed]
- Abbott, G.W.; Redford, K.E.; Yoshimura, R.F.; Manville, R.W.; Moreira, L.; Tran, K.; Arena, G.; Kookootsedes, A.; Lasky, E.; Gunnison, E. KCNQ and KCNE Isoform-Dependent Pharmacology Rationalizes Native American Dual Use of Specific Plants as Both Analgesics and Gastrointestinal Therapeutics. Front. Physiol. 2021, 12, 777057. [Google Scholar] [CrossRef]
- Brown, D.A.; Adams, P.R. Muscarinic suppression of a novel voltage-sensitive K+ current in a vertebrate neurone. Nature 1980, 283, 673–676. [Google Scholar] [CrossRef] [PubMed]
- Springer, K.; Soh, H.; Paz Zavala, R.; Varghese, N.; Lutz, C.; Zuberi, A.R.; Jackson, A.C.; Tzingounis, A.V. Mislocalization of KCNQ2 Channels as a Pathogenic Mechanism in KCNQ2 Developmental and Epileptic Encephalopathy. J. Neurosci. 2026, 46, e0947252025. [Google Scholar] [CrossRef]
- Wu, S.N.; Huang, C.W. Editorial to the special issue “Electrophysiology”. Int. J. Mol. Sci. 2021, 22, 2956. [Google Scholar] [CrossRef]
- Selyanko, A.A.; Hadley, J.K.; Wood, I.C.; Abogadie, F.C.; Delmas, P.; Buckley, N.J.; London, B.; Brown, D.A. Two types of K+ channel subunit, Erg1 and KCNQ2/3, contribute to the M-like current in a mammalian neuronal cell. J. Neurosci. 1999, 19, 7742–7756. [Google Scholar] [CrossRef]
- Chang, W.T.; Liu, P.Y.; Gao, Z.H.; Lee, S.W.; Lee, W.K.; Wu, S.N. Evidence for the Effectiveness of Remdesivir (GS-5734), a Nucleoside-Analog Antiviral Drug in the Inhibition of IK(M) or IK(DR) and in the Stimulation of IMEP. Front. Pharmacol. 2020, 11, 1091. [Google Scholar] [CrossRef]
- Lu, T.L.; Gao, Z.H.; Li, S.W.; Wu, S.N. High Efficacy by GAL-021: A Known Intravenous Peripheral Chemoreceptor Modulator that Suppresses BKCa-Channel Activity and Inhibits IK(M) or Ih. Biomolecules 2020, 10, 188. [Google Scholar] [CrossRef]
- Lu, T.L.; Liutkevičienė, R.; Rovite, V.; Gao, Z.H.; Wu, S.N. Evaluation of Small-Molecule Candidates as Modulators of M-Type K+ Currents: Impacts on Current Amplitude, Gating, and Voltage-Dependent Hysteresis. Int. J. Mol. Sci. 2025, 26, 1504. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, D.; Darwish, Y.; Fu, X.; Trussell, L.O.; Huang, H. KCNQ channels enable reliable presynaptic spiking and synaptic transmission at high frequency. J. Neurosci. 2022, 42, 3305–3315. [Google Scholar] [CrossRef] [PubMed]
- Costi, S.; Han, M.H.; Murrough, J.W. The potential of KCNQ potassium channel openers as novel antidepressants. CNS Drugs 2022, 36, 207–216. [Google Scholar] [CrossRef]
- Li, S.B.; Damonte, V.M.; Chen, C.; Wang, G.X.; Kebschull, J.M.; Yamaguchi, H; Bian, W.J.; Purmann, C.; Pattni, R.; Urban, A.E.; Mourrain, P.; Kauer, J.A.; Scherrer, G.; de Lecea, L. Hyperexcitable arousal circuits drive sleep instability during aging. Science 2022, 375, eabh3021. [Google Scholar] [CrossRef]
- Sankaranarayanan, S.; Simasko, S.M. Characterization of an M-like current modulated by thyrotropin-releasing hormone in normal rat lactotrophs. J. Neurosci. 1996, 16, 1668–1678. [Google Scholar] [CrossRef]
- So, E.C.; Liu, P.Y; Wu, S. N, Effectiveness in the inhibition of dapagliflozin and canagliflozin on M-type K+ current and α-methylglucoside-induced current in pituitary tumor (GH3) and pheochromocytoma PC12 cells. Eur. J. Pharmacol. 2020, 879, 173141. [Google Scholar] [CrossRef] [PubMed]
- Lo, Y.C.; Lin, C.L.; Fang, W.Y.; Lőrinczi, B.; Szatmári, I.; Chang, W.H.; Fülöp, F.; Wu, S.N. Effective activation by kynurenic acid and its aminoalkylated derivatives on M-type K+ current. Int. J. Mol. Sci. 2021, 22 1300. [Google Scholar] [CrossRef]
- Passmore, G.M.; Selyanko, A.A.; Mistry, M.; Al-Qatari, M.; Marsh, S.J.; Matthews, E.A.; Dickenson, A.H.; Brown, T.A.; Burbidge, S.A.; Main, M.; Brown, D.A. KCNQ/M currents in sensory neurons: significance for pain therapy. J. Neurosci 2003, 23, 7227–7236. [Google Scholar] [CrossRef]
- Qiu, Z.C.; Tang, X.Y.; Wu, Q.C.; Tang, Z.L.; Wong, M.S.; Chen, J.X.; Yao, X.S.; Dai, Y. A new strategy for discovering effective substances and mechanisms of traditional Chinese medicine based on standardized drug containing plasma and the absorbed ingredients composition, a case study of Xian-Ling-Gu-Bao capsules. J. Ethnopharmacol. 2021, 279, 114396. [Google Scholar] [CrossRef]
- Zárate, S.; Seilicovich, A. Estrogen receptors and signaling pathways in lactotropes and somatotropes. Neuroendocrinology 2010, 92, 215–223. [Google Scholar] [CrossRef]
- Zhang, R.; Kang, X.; Wang, Y.; Wang, F.; Yu, P.; Shen, J.; Fu, L. Effects of carvedilol on ventricular remodeling and the expression of β3-adrenergic receptor in a diabetic rat model subjected myocardial infarction. Int. J. Cardiol. 2016, 222, 178–184. [Google Scholar] [CrossRef]
- Meera, P.; Wallner, M.; Toro, L. A neuronal beta subunit (KCNMB4) makes the large conductance, voltage- and Ca2+-activated K+ channel resistant to charybdotoxin and iberiotoxin. Proc. Natl. Acad. Sci. U S A 2000, 97, 5562–5567. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.N.; Yang, W.H.; Yeh, C.C.; Huang, H.C. The inhibition by di(2-ethylhexyl)-phthalate of erg-mediated K+ current in pituitary tumor (GH3) cells. Arch. Toxicol. 2012, 86, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.H.; Tseng, W.C.; Leu, Y.L.; Chen, C.Y.; Lee, W.C.; Chi, Y.C.; Cheng, S.F.; Lai, C.Y.; Kuo, C.H.; Yang, S.L.; Yang, S.H.; Shen, J.J.; Feng, C.H.; Wu, C.C.; Hwang, T.L.; Wang, C.J.; Wang, S.H.; Chen, C.C. The flavonoid corylin exhibits lifespan extension properties in mouse. Nat. Commun. 2022, 13, 1238. [Google Scholar] [CrossRef]
- Wu, C.L.; Chuang, C.W.; Cho, H.Y.; Chuang, T.H.; Wu, S.N. The Evidence for Effective Inhibition of INa Produced by Mirogabalin ((1R,5S,6S)-6-(aminomethyl)-3-ethyl-bicyclo [3.2.0] hept-3-ene-6-acetic acid), a Known Blocker of CaV Channels. Int. J. Mol. Sci. 2022, 23, 3845. [Google Scholar] [CrossRef] [PubMed]
- Meng, G.; Shi, Y.; Wang, Y.; Shi, Y. Modulation of SpiroOMeTAD Hole-Transport Layers for Carbon-Based Perovskite Solar Cells. Small 2026, 22, e12519. [Google Scholar] [CrossRef]
- Zhang, T.; Zhong, S.; Meng, Y.; Deng, W.; Hou, L.; Wang, Y.; Xing, X.; Guan, T.; Zhang, J.; Li, T. Quantitative structure-activity relationship for estrogenic flavonoids from Psoralea corylifolia. J. Pharm. Biomed. Anal. 2018, 161, 129–135. [Google Scholar] [CrossRef]
- Men, W.J.; Cheng, L.Y.; Chen, M.Y.; Zhang, X.Y.; Zhang, Y.; Zhou, K. Study on pharmacokinetics of eight active compounds from Bufei-Huoxue Capsule based on UHPLC-MS/MS. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2021, 1184, 122974. [Google Scholar] [CrossRef]
- Lin, Z.R.; Yin, H.; Wan, T.F.; Ruan, Z.; Mao, J.Y.; Pan, Z.Y.; Huang, Y.; Zeng, S.Y.; Ran, L.; Liu, J.H.; Xiao, X.H.; Xie, H.; Hu, Y. Corylin ameliorates diabetic osteoporosis by inhibiting inflammation via targeting RAG1. Phytomedicine 2026, 150, 157671. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Gamper, N.; Shapiro, M.S. Single-channel analysis of KCNQ K+ channels reveals the mechanism of augmentation by a cysteine-modifying reagent. J. Neurosci. 2004, 24, 5079–5090. [Google Scholar] [CrossRef] [PubMed]
- Selyanko, A.A.; Brown, D.A. Regulation of M-type potassium channels in mammalian sympathetic neurons: action of intracellular calcium on single channel currents. Neuropharmacology 1996, 35, 933–947. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.N.; Wang, Y.J.; Gao, Z.H.; Liutkevičienė, R.; Rovite, V. Recent Advances in Ionic Mechanisms in Pituitary Cells: Implications for Electrophysiological and Electropharmacological Research. J. Clin. Med. 2025, 14, 3117. [Google Scholar] [CrossRef]
- Lin, C.Y.; Gao, Z.H.; Cheung, C.W.; So, E.C.; Wu, S.N. Modulation of Voltage-Gated Na+ Channel Currents by Small Molecules: Effects on Amplitude and Gating During High-Frequency Stimulation. Sci. Pharm. 2025, 93, 33. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



