Submitted:
18 March 2026
Posted:
23 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Ultrasound Examination
2.3. Transient Elastography and CAP Measurement
2.4. Definition of Concordance and Discordance
2.5. Clinical and Biochemical Variables
2.6. Statistical Analysis
3. Results
4. Discussions
5. Conclusions
Consent for publication
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Fatiha Nassir, Jamal A. Ibdah - Pathogenesis and Prevention of Hepatic Steatosis Gastroenterol Hepatol (N Y). 2015 Mar; 11(3): 167–175.
- Streba LAM, Vere CC, Rogoveanu I, Streba CT. Nonalcoholic fatty liver disease, metabolic risk factors, and hepatocellular carcinoma: An open question. World J Gastroenterol 2015; 21(14): 4103-4110 [PMID: 25892859 DOI: 10.3748/wjg. v21.i14.4103].
- Fatiha Nassir - NAFLD: Mechanisms, Treatments, and Biomarkers Biomolecules. 2022 Jun 13;12(6):824. doi: 10.3390/biom12060824.
- Dina G. Tiniakos,1 Miriam B. Vos,2 and Elizabeth M. Brunt3 - Nonalcoholic Fatty Liver Disease: Pathology and Pathogenesis. Annual Review of Pathology: Mechanisms of Disease Volume 5, 2010 Tiniakos, pp 145-171.
- Elizabeth M. Brunt. Nonalcoholic Fatty Liver Disease: Pros and Cons of Histologic Systems of Evaluation Int. J. Mol. Sci. 2016, 17(1), 97; https://doi.org/10.3390/ijms17010097.
- Marchesini G, Brizi M, Morselli-Labate AM, Bianchi G, Bugianesi E, McCullough AJ, Forlani G, Melchionda N. Association of nonalcoholic fatty liver disease with insulin resistance. Am J Med. 1999; 107:450-455.
- Marchesini G, Brizi M, Bianchi G, Tomassetti S, Bugianesi E, Lenzi M, McCullough AJ, Natale S, Forlani G, Melchionda N. Nonalcoholic fatty liver disease: a feature of the metabolic syndrome. Diabetes. 2001; 50:1844-1850.
- Hsiao PJ, Kuo KK, Shin SJ, Yang YH, Lin WY, Yang JF, Chiu CC, Chuang WL, Tsai TR, Yu ML. Significant correlations between severe fatty liver and risk factors for metabolic syndrome. J Gastroenterol Hepatol. 2007; 22:2118-2123.
- Jimba S, Nakagami T, Takahashi M, Wakamatsu T, Hirota Y, Iwamoto Y, Wasada T. Prevalence of non-alcoholic fatty liver disease and its association with impaired glucose metabolism in Japanese adults. Diabet Med. 2005; 22:1141-1145.
- A Lum Han - Validation of fatty liver index as a marker for metabolic dysfunction-associated fatty liver disease Diabetology & Metabolic Syndrome (2022) 14:44 https://doi.org/10.1186/s13098-022-00811-2.
- Ah Reum Khang, Hye Won Lee, Dongwon Yi, Yang Ho Kang, Seok Man Son- The fatty liver index, a simple and useful predictor of metabolic syndrome: analysis of the Korea National Health and Nutrition Examination Survey 2010–2011 - Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy 2019:12 181–190.
- Kavya Koneru 1, Varsha Bhatt 1, Arjun Kakrani 1, Manaswini Edara 1, Vuluvala T Reddy 1, Pranav Gopal Jawade 1- A study of non-alcoholic fatty liver disease-liver fat score in overweight and obese individuals J Family Med Prim Care. 2022 Aug;11(8):4368-4374. doi: 10.4103/jfmpc.jfmpc_58_22. Epub 2022 Aug 30.
- Amir M. Pirmoazen, Aman Khurana, Ahmed El Kaffas, Aya Kamaya Quantitative ultrasound approaches for diagnosis and monitoring hepatic steatosis in nonalcoholic fatty liver disease Theranostics, 2020; 10(9): 4277-4289. doi:10.7150/thno.40249 1335–48.
- Sasso, M; Beaugrand, M; de Ledinghen, V; Douvin, C; Marcellin, P; Poupon, R; et al. Controlled Attenuation Parameter (CAP): A Novel VCTETM Guided Ultrasonic Attenuation Measurement for the Evaluation of Hepatic Steatosis: Preliminary Study and Validation in a Cohort of Patients with Chronic Liver Disease from Various Causes. Ultrasound Med Biol. 2010, 36, 1825–35. [Google Scholar] [CrossRef] [PubMed]
- Karlas, T; Petroff, D; Garnov, N; Böhm, S; Tenckhoff, H; Wittekind, C; et al. Non-invasive assessment of hepatic steatosis in patients with NAFLD using controlled attenuation parameter and 1H-MR spectroscopy. PLoS One 2014, 9, e91987. [Google Scholar] [CrossRef]
- Chan, W-K; Mustapha, NRN; Mahadeva, S. Controlled attenuation parameter for the detection and quantification of hepatic steatosis in nonalcoholic fatty liver disease. J Gastroenterol Hepatol. 2014, 29, 1470–6. [Google Scholar] [CrossRef]
- de Lédinghen, V; Wong, GL-H; Vergniol, J; Chan, HL-Y; Hiriart, J-B; Chan, AW-H; et al. Controlled attenuation parameter for the diagnosis of steatosis in non-alcoholic fatty liver disease. J Gastroenterol Hepatol. 2016, 31, 848–55. [Google Scholar] [CrossRef]
- Lee, HW; Park, SY; Kim, SU; Jang, JY; Park, H; Kim, JK; et al. Discrimination of Nonalcoholic Steatohepatitis Using Transient Elastography in Patients with Nonalcoholic Fatty Liver Disease. PLoS One 2016, 11, e0157358. [Google Scholar] [CrossRef]
- Chan, W-K; Nik Mustapha, NR; Wong, GL-H; Wong, VW-S; Mahadeva, S. Controlled attenuation parameter using the FibroScan® XL probe for quantification of hepatic steatosis for non-alcoholic fatty liver disease in an Asian population. United Eur Gastroenterol J. 2017, 5, 76–85. [Google Scholar] [CrossRef]
- Park, CC; Nguyen, P; Hernandez, C; Bettencourt, R; Ramirez, K; Fortney, L; et al. Magnetic Resonance Elastography vs Transient Elastography in Detection of Fibrosis and Noninvasive Measurement of Steatosis in Patients with Biopsy-Proven Nonalcoholic Fatty Liver Disease. Gastroenterology 2017, 152, 598–607.e2. [Google Scholar] [CrossRef]
- Chan, W-K; Nik Mustapha, NR; Mahadeva, S; Wong, VW-S; Cheng, JY-K; Wong, GL-H. Can the same controlled attenuation parameter cut-offs be used for M and XL probes for diagnosing hepatic steatosis? J Gastroenterol Hepatol. 2018, 33, 1787–94. [Google Scholar] [CrossRef]
- Runge, JH; Smits, LP; Verheij, J; Depla, A; Kuiken, SD; Baak, BC; et al. MR Spectroscopy–derived Proton Density Fat Fraction Is Superior to Controlled Attenuation Parameter for Detecting and Grading Hepatic Steatosis. Radiology 2017, 286, 547–56. [Google Scholar] [CrossRef]
- Imajo, K; Kessoku, T; Honda, Y; Tomeno, W; Ogawa, Y; Mawatari, H; et al. Magnetic Resonance Imaging More Accurately Classifies Steatosis and Fibrosis in Patients with Nonalcoholic Fatty Liver Disease Than Transient Elastography. Gastroenterology 2016, 150, 626–637.e7. [Google Scholar] [CrossRef]
- Caussy, C; Alquiraish, MH; Nguyen, P; Hernandez, C; Cepin, S; Fortney, LE; et al. Optimal threshold of controlled attenuation parameter with MRI-PDFF as the gold standard for the detection of hepatic steatosis. Hepatology 2018, 67, 1348–59. [Google Scholar] [CrossRef]
- Sasso, M; Audière, S; Kemgang, A; Gaouar, F; Corpechot, C; Chazouillères, O; et al. Liver Steatosis Assessed by Controlled Attenuation Parameter (CAP) Measured with the XL Probe of the FibroScan: A Pilot Study Assessing Diagnostic Accuracy. Ultrasound Med Biol. 2016, 42, 92–103. [Google Scholar] [CrossRef]
- Sterling, RK; Lissen, E; Clumeck, N. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 2006, 43(6), 1317–1325. [Google Scholar] [CrossRef] [PubMed]
- Hernaez, R; Lazo, M; Bonekamp, S; Kamel, I; Brancati, FL; Guallar, E; et al. Diagnostic accuracy and reliability of ultrasonography for the detection of fatty liver: A meta-analysis. Hepatology 2011, 54, 1082–90. [Google Scholar] [CrossRef] [PubMed]
- Palmentieri, B; de Sio, I; La Mura, V; Masarone, M; Vecchione, R; Bruno, S; et al. The role of bright liver echo pattern on ultrasound B-mode examination in the diagnosis of liver steatosis. Dig Liver Dis. 2006, 38, 485–9. [Google Scholar] [CrossRef] [PubMed]
- Saadeh, S; Younossi, ZM; Remer, EM; Gramlich, T; Ong, JP; Hurley, M; et al. The utility of radiological imaging in nonalcoholic fatty liver disease. Gastroenterology 2002, 123, 745–50. [Google Scholar] [CrossRef]
- Strauss, S; Gavish, E; Gottlieb, P; Katsnelson, L. Interobserver and intraobserver variability in the sonographic assessment of fatty liver. AJR Am J Roentgenol. 2007, 189, W320–323. [Google Scholar] [CrossRef]
- Hepburn, MJ; Vos, JA; Fillman, EP; Lawitz, EJ. The accuracy of the report of hepatic steatosis on ultrasonography in patients infected with hepatitis C in a clinical setting: A retrospective observational study. BMC Gastroenterol 2005, 5, 14. [Google Scholar] [CrossRef]
- Jang, JK; Choi, SH; Lee, JS; Kim, SY; Lee, SS; Kim, KW. Accuracy of the ultrasound attenuation coefficient for the evaluation of hepatic steatosis: a systematic review and meta-analysis of prospective studies. Ultrasonography 2022, 41(1), 83–92. [Google Scholar] [CrossRef] [PubMed]
- Victor de Lédinghen, Julien Vergniol, Maylis Capdepont, Faiza Chermak, Jean-Baptiste Hiriart, Christophe Cassinotto, Wassil Merrouche, Juliette Foucher, Le Bail Brigitte. Controlled attenuation parameter (CAP) for the diagnosis of steatosis: A prospective study of 5323 examinations. Journal of Hepatology, Volume 60, Issue 5, 1026 - 1031.
- Bota, S.; Sporea, I.; Şirli, R.; Popescu, A.; Jurchiş, A. Factors Which Influence the Accuracy of Acoustic Radiation Force Impulse (ARFI) Elastography for the Diagnosis of Liver Fibrosis in Patients With Chronic Hepatitis C. Ultrasound in Medicine & Biology 2013, 39(3), 407–412. [Google Scholar] [CrossRef] [PubMed]
- Özercan M, Melekoğlu Ellik Z, Parmaksız A, Gümüşsoy M, Duman S, Örmeci N. Liver stiffness and steatosis measurements with iLivTouch and FibroScan: A comparative study. Turk J Gastroenterol. Published online June 6, 2024. [CrossRef]
- Wang, M.; Tang, S.; Li, G.; et al. Comparative study of ultrasound attenuation analysis and controlled attenuation parameter in the diagnosis and grading of liver steatosis in non-alcoholic fatty liver disease patients. BMC Gastroenterol 2024, 24, 81. [Google Scholar] [CrossRef] [PubMed]
- Juan Francisco Rozas, Katherine González, Camila Estay, Maximo Cattaneo, Alvaro Urzúa, Juan Pablo Roblero, Alexandra Sandoval, Jaime Poniachik. Concordance between ecography and the Continuous Attenuation Parameter (CAP) by transient elastography for the diagnosis of liver steatosis. Annals of Hepatology,Volume 24, Supplement 1, 2021, 100422, ISSN 1665-2681 https://doi.org/10.1016/j.aohep.2021.100422.
- Tamaki Kobayashi et all - Diagnostic performance of two-dimensional shear wave elastography and attenuation imaging for fibrosis and steatosis assessment in chronic liver disease - Journal of Medical Ultrasonics (2025) 52:95–103 https://doi.org/10.1007/s10396-024-01473-5.
- Mengyun Wang, Shuzhen Tang, Guoqiu Li, Zhibin Huang, Sijie Mo, Keen Yang, Jing Chen, Baishan Du, Jinfeng Xu, Zhimin Ding, Fajin Dong - Comparative study of ultrasound attenuation analysis and controlled attenuation parameter in the diagnosis and grading of liver steatosis in non-alcoholic fatty liver disease patients -BMC Gastroenterology (2024) 24:81 https://doi.org/10.1186/s12876-024-03160-8.
- Andrea Salmi, Luigi di Filippo, Clarissa Ferrari, Stefano Frara, Andrea Giustina - Endocrine. 2022 Nov;78(2):262-269. doi: 10.1007/s12020-022-03157-x. Epub 2022 Aug 18.
- Rosanna Villani, Pierluigi Lupo, Moris Sangineto, Antonino Davide Romano and Gaetano Serviddio - Liver Ultrasound Elastography in Non-Alcoholic Fatty Liver Disease: A State-of-the-Art Summary - Diagnostics 2023, 13(7), 1236; https://doi.org/10.3390/diagnostics13071236.
- Roxana Sirli, Ioan Sporea - Controlled Attenuation Parameter for Quantification of Steatosis: Which Cut-Offs to Use? - Can J Gastroenterol Hepatol. 2021 Mar 26;2021:6662760. doi:10.1155/2021/6662760.
- Dorotea Bozic, Kristian Podrug, Ivana Mikolasevic, Ivica Grgurevic - Ultrasound Methods for the Assessment of Liver Steatosis: A Critical Appraisal - Diagnostics (Basel). 2022 Sep 22;12(10):2287. doi: 10.3390/diagnostics12102287.
- Giovanna Ferraioli et all - Liver Fat Quantification With Ultrasound: Depth Dependence of Attenuation Coefficient - April 2023Journal of ultrasound in medicine: official journal of the American Institute of Ultrasound in Medicine 42(10). DOI:10.1002/jum.16242.
- Chi-Cervera LA, Montalvo GI, Icaza-Chávez ME, Torres-Romero J, Arana-Argáez V, Ramírez-Camacho M, Lara-Riegos J. Clinical relevance of lipid panel and aminotransferases in the context of hepatic steatosis and fibrosis as measured by transient elastography (FibroScan®). J Med Biochem. 2021 Jan 26;40(1):60-66. doi: 10.5937/jomb0-24689. PMID: 33584141; PMCID: PMC7857856.
- Csonka, C., Baranyai, T., Tiszlavicz, L. et al. Isolated hypercholesterolemia leads to steatosis in the liver without affecting the pancreas. Lipids Health Dis 16, 144 (2017). https://doi.org/10.1186/s12944-017-0537-z.
- Sporea, I.; Mare, R.; Popescu, A.; Nistorescu, S.; Baldea, V.; Sirli, R.; Braha, A.; Sima, A.; Timar, R.; Lupusoru, R. Screening for Liver Fibrosis and Steatosis in a Large Cohort of Patients with Type 2 Diabetes Using Vibration Controlled Transient Elastography and Controlled Attenuation Parameter in a Single-Center Real-Life Experience. J. Clin. Med. 2020, 9, 1032. https://doi.org/10.3390/jcm9041032.
- Sweta Tandra, Matthew M. Yeh, Elizabeth M. Brunt, Raj Vuppalanchi, Oscar W. Cummings, Aynur Ünalp-Arida, Laura A. Wilson, Naga Chalasani - Presence and significance of microvesicular steatosis in nonalcoholic fatty liver disease, Journal of Hepatology, Volume 55, Issue 3, 2011, Pages 654-659, ISSN 0168- 8278, https://doi.org/10.1016/j.jhep.2010.11.021. Issue 3.
- Tomizawa M, Kawanabe Y, Shinozaki F, Sato S, Motoyoshi Y, Sugiyama T, Yamamoto S, Sueishi M. Triglyceride is strongly associated with nonalcoholic fatty liver disease among markers of hyperlipidemia and diabetes. Biomed Rep. 2014 Sep;2(5):633-636. doi: 10.3892/br.2014.309. Epub 2014 Jul 1. PMID: 25054002; PMCID: PMC4106613.
- Chang, Madeleine & Chang, Devon & Kodali, Sudha & Harrison, Stephen & Ghobrial, Mark & Alkhouri, Naim. (2024). Degree of Discordance Between FIB-4 and Transient Elastography: An Application of Current Guidelines on General Population Cohort. Clinical gastroenterology and hepatology : the official clinical practice journal of the American Gastroenterological Association. 22. 10.1016/j.cgh.2024.02.008.
- Moini, M., Onofrio, F., Hansen, B.E. et al. Combination of FIB-4 with ultrasound surface nodularity or elastography as predictors of histologic advanced liver fibrosis in chronic liver disease. Sci Rep 11, 19275 (2021). https://doi.org/10.1038/s41598-021-98776-1.
- Chen, Mingxi, Guo, Chang, Ouyang, Ke Liu, Na. Diagnostic role of the fibrosis-4 index and nonalcoholic fatty liver disease fibrosis score as a noninvasive tool for liver fibrosis scoring. Medicine 103(43): p e40214, October 25, 2024. | DOI: 10.1097/MD.0000000000040214. [CrossRef]
- Damjanovska S, Karb DB, Tripathi A, Asirwatham J, Delozier S, Perez JA, Falck-Ytter Y, Cohen S. Accuracy of Ultrasound Elastography and Fibrosis-4 Index (FIB-4) in Ruling Out Cirrhosis in Obese Non-Alcoholic Fatty Liver Disease (NAFLD) Patients. Cureus. 2022 Sep 22;14(9):e29445. doi:10.7759/cureus.29445. PMID: 36299964; PMCID: PMC9587692.
- Kim BK, Bernstein N, Huang DQ, Tamaki N, Imajo K, Yoneda M, Sutter N, Jung J, Nguyen K, Nguyen L, Le T, Madamba E, Richards L, Valasek MA, Behling C, Sirlin CB, Nakajima A, Loomba R. Clinical and histologic factors associated with discordance between steatosis grade derived from histology vs. MRI-PDFF in NAFLD. Aliment Pharmacol Ther. 2023 Jul;58(2):229-237. doi: 10.1111/apt.17564. Epub 2023 Jun 2. PMID: 37269117; PMCID: PMC10330628.
- Lee CM, Kim M, Kang BK, Jun DW, Yoon EL. Discordance diagnosis between B-mode ultrasonography and MRI proton density fat fraction for fatty liver. Sci Rep. 2023 Sep 20;13(1):15557. doi: 10.1038/s41598-023-42422-5. PMID: 37730972; PMCID: PMC10511436.
| Parameter |
Total of patients N=130 |
|
Gender Male gender Female gender |
62 ( 47.69%) 68 (52.31 ) |
|
Urban area Rural area |
72 (55.38 %) 58 ( 44.62%) |
|
Age (years) - < 50 -50-69 ≥ 70 |
30 (23.07 %) 72 ( 55.38%) 28 (21.55% ) |
|
Etiology - viral -alcoholic -autoimmune - other |
24 (18.46% ) 18 (13.84% ) 2 (1.53% ) 86 (66,17%) |
|
Fibrosis (measured byTE) -F0 -F1 -F2 -F3 -F4 |
39 ( 30%) 62 (47.69% ) 9 ( 6.92%) 6 (4.61% ) 14 (10.78%) |
| BMI (kg/m2) -<20 -20-24.9 -25-29.9 ≥30 |
6 (4.61%) 24 (18.46%) 52 (40% ) 48 ( 36.93%) |
| Type 2 Diabetes mellitus | 43 (33.07% ) |
| Cardiac comorbidities | 21 (16.15% ) |
| Arterial Hypertension | 72 ( 55.38%) |
| Hypothyroidism | 16 (12.30% ) |
| ALAT > ULN | 23 ( 17.69%) |
| GGT > ULN | 32 ( 24.61%) |
| Total Serum Cholesterol >200 mg/dl | 47 (36.15% ) |
| Triglycerides > 150 mg/dl | 37 (28.46% ) |
| FIB 4 - < 1.45 -1,45 -3.25 - > 3.25 |
69 ( 53.07%) 46 (35.38% ) 15 ( 11.5%) |
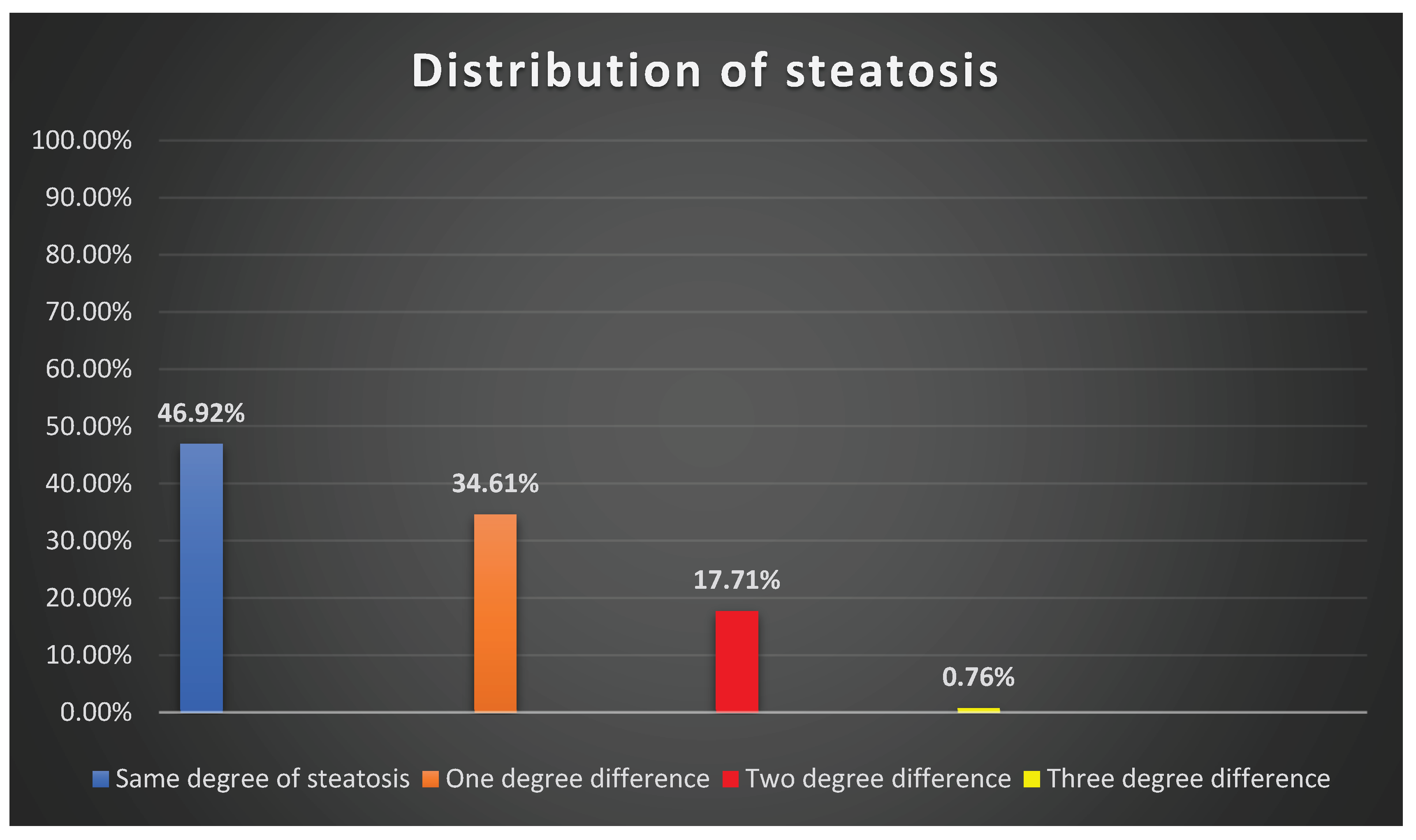
| Ultrasound grade | CAP S0 | CAP S1 | CAP S2 |
CAP S3 | Total |
| S0 | 27 | 3 | 0 | 1 | 31 |
| S1 | 12 | 3 | 4 | 3 | 22 |
| S2 | 19 | 13 | 7 | 10 | 49 |
| S3 | 0 | 0 | 3 | 25 | 28 |
| Total | 58 | 19 | 14 | 39 | 130 |
| Parameter | Same degree Steatosis US and TE (n=61) Group 1 | Diferrent degree Steatosis US and TE (n=69) Group 2 | p value | |
| Male gender | 33 (54.1%) | 29 (42.1%) | 0.17 | |
| Urban area | 33 (54.1%) | 39 (56.5%) | 0.78 | |
| Age (years) - < 50 -50-69 ≥ 70 |
16 (26.2%) 30 (49.2%) 15 (24.6%) |
14 (20.3%) 42 (60.8%) 13 (18,9%) |
0.42 0.18 0.43 |
|
| Etiology – viral -alcoholic -autoimmune |
6 (9.8%) 7 (11.4%) 1 (1.6%) |
18 (26.1%) 11 (15.9%) 1 (1.5%) |
0.01 0.45 0.96 |
|
| Fibrosis (TE) -F0 -F1 -F2 -F3 -F4 |
20 (32.8%) 32 (52.4%) 2 (3.3%) 2 (3.3%) 5 (8.2%) |
19 (27.6%) 30 (43.4%) 7 (10.1%) 4 (5.8%) 9 (13.1%) |
0,52 0.30 0.12 0.50 0.37 |
|
| Fibrosis (TE) ≥ 2 | 9 (14.8%) | 20 (29%) | 0.055 | |
| BMI (kg/m2) -<20 -20-24,9 -25-29,9 ≥30 |
4 (6.6%) 8 (13.1%) 24 (39.4%) 25 (40.9%) |
2 (2.9%) 16 (23.2%) 28 (40.6%) 23 (33.3%) |
0.31 0.14 0.88 0.37 |
|
| Type 2 Diabetes mellitus | 22 (36.1%) | 21 (30.4%) | 0.49 | |
| Cardiac comorbidities | 8 (13.1%) | 13 (18.8%) | 0.43 | |
| Arterial Hypertension | 34 (55.7%) | 38 (55.1%) | 0.94 | |
| ALAT > ULN | 10 (16.4%) | 13 (18.8%) | 0.72 | |
| GGT > ULN | 13 (21.3%) | 19 (27.5%) | 0.41 | |
| Total Serum Cholesterol >200 mg/dl | 28 (45.9%) | 19 (27.5%) | 0.02 | |
| Serum Triglycerides > 150 mg/dl | 22 (36.1%) | 15 (21.7%) | 0.07 | |
|
FIB 4 - < 1.45 -1.45 -3.25 - > 3.25 |
30 (49.2%) 27 (44.2%) 4 (6.6%) |
39 (56.6%) 19 (27.5%) 11 (15.9%) |
0.40 0.04 0.09 |
| Variable | Odds Ratio (OR) | 95% CI | p value |
| Viral etiology | 0.59 | 0.18–1.95 | 0.37 |
| Cholesterol >200 mg/dl | 2.23 | 1.07–4.66 | 0.03 |
| FIB 4 | 0.98 | 0.59–1.63 | 0.92 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




