Submitted:
17 March 2026
Posted:
18 March 2026
You are already at the latest version
Abstract
Background/Objectives: Varicosities of the vortex vein ampulla are transient dilations of vortex vein ampullae that appear as red-brown choroidal masses. The purpose of this manuscript is to describe a retrospective case series of 53 patients with varicosities of the vortex vein ampulla and perform a literature review on this topic. Methods: Our case series demonstrates the clinical features of a large cohort of varicosities of the vortex vein ampulla, including their locations in the ocular fundus, sizes when congested, direction of gaze that resulted in detection, frequency of multiple lesions in a single eye, and frequency of bilateral cases. The literature review utilized PubMed and Embase libraries and included all studies published through December 2025. Results: The literature review yielded 44 articles, of which 37 were deemed relevant. Several studies described the appearance of these lesions using imaging modalities, including B-scan ultrasonography, optical coherence tomography, and indocyanine green angiography. Others underscored the potential for these lesions to be mistaken for other types of choroidal masses, such as choroidal melanomas. Conclusions: This extensive series demonstrates that these lesions are most often located nasally, sometimes multiple or bilateral, and often mistaken for choroidal nevi or melanomas, highlighting the importance of understanding clinical characteristics for appropriate diagnosis. In addition, some studies described possible associations with conditions such as nodular scleritis and Donnai-Barrow syndrome.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Literature Review
3. Results
3.1. Case Series
3.2. Literature Review
3.2. Figures and Tables
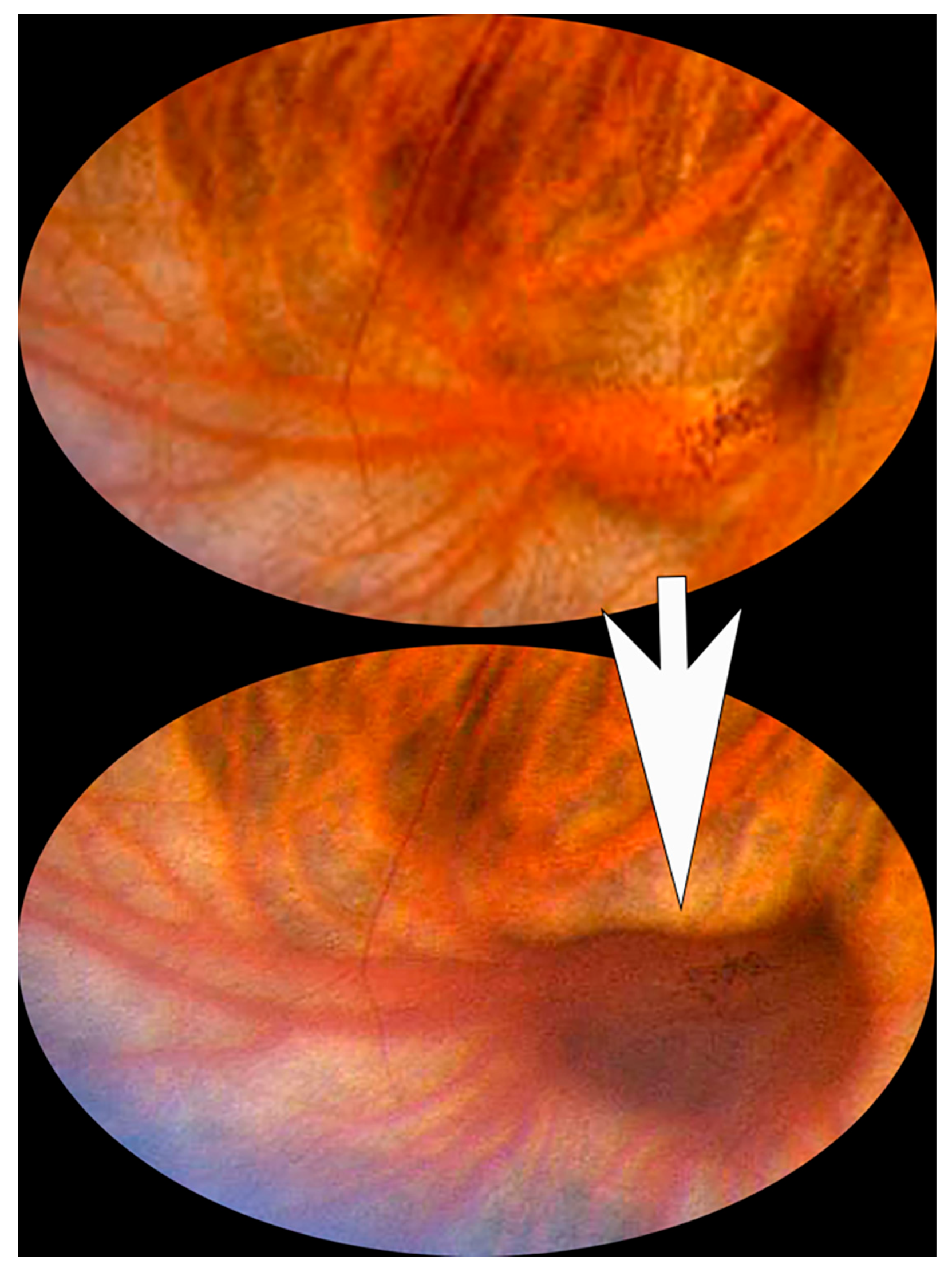
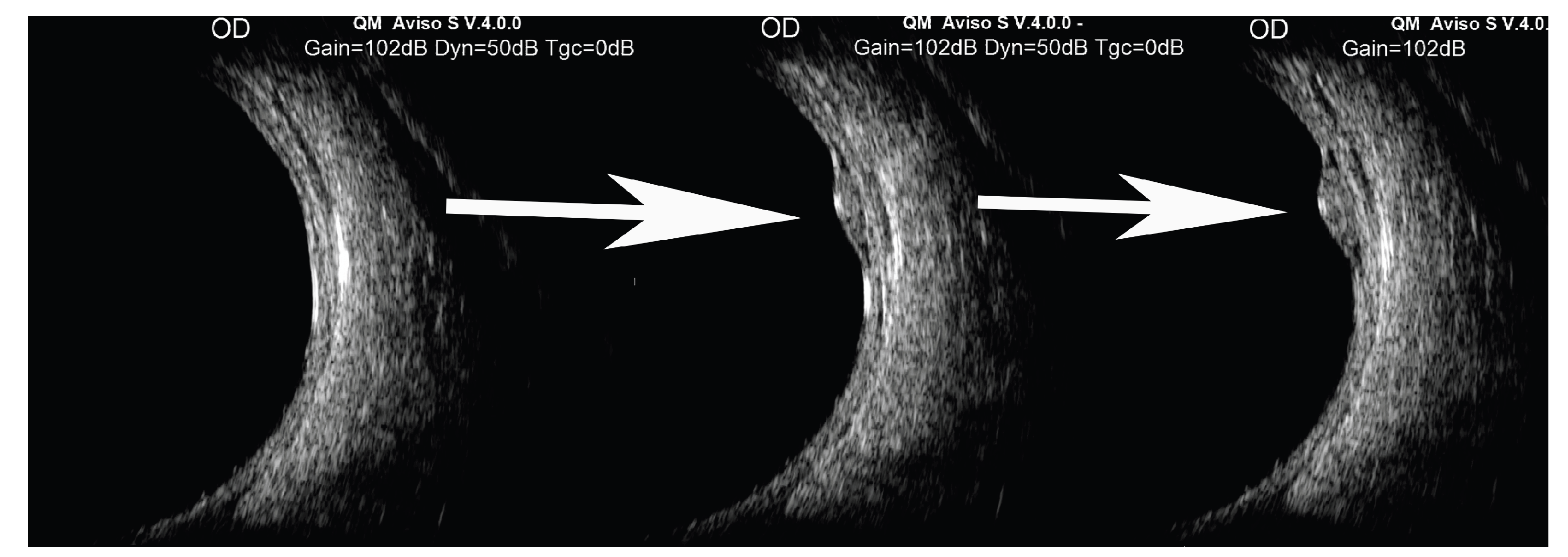
 |
| direction of ocular gaze that resulted in vortex vein ampulla’s congestion (N) | |||||||||
| quadrant location of lesion |
S | SN | N | IN | I | IT | T | ST | all |
| SN (superonasal) | 5 | 18 | 0 | 0 | 0 | 0 | 0 | 0 | 1b |
| IN (inferonasal) | 0 | 0 | 0 | 20 | 0 | 4 | 0 | 0 | 0 |
| IT (inferotemporal) | 0 | 0 | 0 | 0 | 1 | 9 | 0 | 0 | 0 |
| ST (superotemporal) | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 2 | 0 |
| Study Author, year [ref] |
Study Type | N (patients) | N (eyes) | Eye(s) involved |
Lesion location |
Multiplicity | Laterality | Dynamic Behavior |
Advanced imaging modalities | Associated/mimicked conditions | Key notes |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Zhang et al., 2018 [22] | Case report | 1 | 1 | OD | Superotemporal | Single | Unilateral | Collapse with globe pressure | B-scan | Anterior scleritis | |
| Wolfensberger et al, 1997 [11] | Case report | 1 | 1 | OS | Superior | Single | Unilateral | Collapse with globe pressure | FA | Choroidal melanoma mimic | Article not in English, data abstraction relied on English-translated abstract |
| Weidmayer et al, 2021 [29] | Case report | 1 | 1 | OS | Superotemporal | Single | Unilateral | Collapse with globe pressure | OCT, B-scan, FAF | Lesion that spontaneously appeared and resolved, even when patient looked towards direction of lesion | |
| Veronese et al, 2019 [5] | Descriptive imaging series | 8 | 8 | Mixed (7 OD, 1 OS) | Multiple | Single | Unilateral | Collapse with globe pressure | OCT, FA, ICGA, FAF, IR | ||
| Vahdani et al, 2010 [7] | Case report | 1 | 1 | OS | Inferonasal | Multiple | Unilateral | Gaze evoked intumescence; collapse with globe pressure | Not described | ||
| Suetov et al, 2022 [8] | Case series | 2 | Unable to assess - article not in English | Unable to assess - article not in English | Unable to assess due to article not in English | Multiple | Bilateral | Unable to assess - article not in English | Unable to assess - article not in English | Article not in English so data abstraction relied on English-translated abstract | |
| Spiess et al, 2022 [19] | Case report | 1 | 1 | OD | Superonasal | Single | Unilateral | Collapse with globe pressure | OCT | ||
| Snyers et al, 2002 [30] | Descriptive imaging series | Unable to assess due to article not being in English | Unable to assess due to article not being in English | Unable to assess due to article not being in English | Unable to assess-article not in English | Unable to assess due to article not being in English | Unable to assess due to article not being in English | Gaze evoked intumescence; Valsalva evoked intumescence | ICGA | ||
| Singh et al, 1993 [31] | Imaging case report | 1 | 1 | OS | Inferonasal | Single | Unilateral | Gaze evoked intumescence; Valsalva evoked intumescence | B-scan, ICGA | Lesion simulated choroidal melanoma | |
| Siddiqui et al, 2019 [21] | Imaging case report | 1 | 1 | OD | Inferonasal | Single | Unilateral | Collapse with globe pressure | Intraoperative OCT | Choroidal hemorrhage mimic during pars plana vitrectomy for vitreous opacities | |
| Shields et al. 1995 [17] | Descriptive imaging study | 1 | 1 | Not described | Not described | Single | Unilateral | Gaze evoked intumescence | ICGA | ||
| Rubegni et al. 2025 [15] | Imaging case report | 1 | 1 | OS | Inferotemporal | Single | Unilateral | Collapse with globe pressure | OCT | ||
| Rimsaite et al. 2015 [14] | Case report | 1 | 1 | OS | Superotemporal | Single | Unilateral | Collapse with globe pressure | OCT, ICGA | Lesion simulated choroidal melanoma | Article not in English. Data transcribed using English-translated version. |
| Rayess et al. 2015 [20] | Imaging case report | 1 | 1 | OD | Superonasal | Single | Unilateral | Not described | OCT, B-scan, FA | Authors noted that OCT was the only imaging modality in this case that diagnosed the lesion. | |
| Osher et al. 1981 [1] | Descriptive case series | 5 | 5 | Mixed (1 OD, 1 OU, 3 not specified) | Multiple (3 superonasal, 2 inferior, 1 unspecified) | Single | One patient with bilateral | Gaze evoked intumescence; Valsalva evoked intumescence; collapse with globe pressure | B-scan, FA | ||
| Niu et al. 2025 [32] | Imaging case report | 1 | 1 | OD | Inferotemporal | Single | Unilateral | Gaze evoked intumescence; collapse with globe pressure | OCT, B-scan, ICGA | ||
| Nitta et al. 2024 [33] | Imaging case report | 1 | 1 | OS | Inferonasal | Single | Unilateral | Gaze evoked intumescence; collapse with globe pressure | OCT, FA, ICGA | Patient with a vortex vein ampulla varix in OS and a macular vortex vein in OD | |
| Murtagh et al. 2021 [34] | Case report | 1 | 1 | OS | Superonasal | Single | Unilateral | Collapse with globe pressure | OCT | ||
| Milani et al. 2022 [27] | Case report | 1 | 1 | OD | Inferotemporal | Single | Unilateral | Not described | OCT, B-scan, FA ICGA | Suprachoroidal hemorrhage found adjacent to a vortex vein ampulla varix. Authors hypothesized that the varix predisposed the hemorrhage. | |
| Lopez et al. 1986 [35] | Case series | 3 | Unable to access | Unable to access | Unable to access article | Unable to access | Unable to access | Collapse with globe pressure | Unable to access article | Unable to access complete article | |
| Levy et al. 2005 [13] | Case series | 3 | 3 | Mixed (2 OS, 1 OD) | Multiple (1 inferonasal, 2 superonasal) | Multiple | Unilateral | Gaze evoked intumescence; collapse with globe pressure | B-scan | Choroidal melanoma mimic | |
| Khan et al. 2007 [36] | Prospective study | 1 | Not described | Not described | Not described | Not described | Not described | Not described | Not described | Choroidal melanoma mimic | Study assessing accuracy of referrals for suspected choroidal melanoma |
| Kang et al. 2017 [37] | Imaging case report | 1 | 1 | OD | Superonasal | Single | Unilateral | Gaze evoked intumescence; collapse with globe pressure | OCT, FA, ICGA | Description of vortex vein ampulla dynamics using real-time angiography. | |
| Kang et al. 2000 [12] | Imaging case report | 1 | 1 | OD | Superonasal | Single | Unilateral | Gaze evoked intumescence; collapse with globe pressure | FA, ICGA | ||
| Ismail et al. 2011 [6] | Imaging case report | 1 | 1 | OD | Superonasal | Single | Unilateral | Collapse with globe pressure | OCT, ICGA | Subretinal hemorrhage mimic | |
| Hunter et al. 1983 [38] | Case series | 2 | 2 | Mixed (1 OD, 1 OS) | Multiple (1 superonasal, 1 superior) | Single | Unilateral | Gaze evoked intumescence; collapse with globe pressure | |||
| Higham et al. 2022 [24] | Case series | 2 | 4 | OU | Macular | Multiple | Bilateral | Not described | OCT, ICGA | Association with Donnai-Barrow syndrome | |
| Gunduz et al. 1998 [39] | Case series | 4 | 4 | Not described | Multiple | One patient with multiple varix | Unilateral | Gaze evoked intumescence | B-scan, FA, ICGA | Choroidal melanoma mimic | |
| Ghadiali et al. 2017 [26] | Case report | 1 | 1 | OD | Superotemporal | Single | Unilateral | Collapse with globe pressure | OCT, ICGA | Choroidal melanoma mimic | |
| Garcia-Bardera et al. 2024 [40] | Imaging case report | 1 | 1 | OD | Superonasal | Single | Unilateral | Gaze evoked intumescence; valsalva evoked intumescence; collapse with globe pressure | OCT | Unable to access complete article | |
| Levin et al. 2025 [9] | Imaging case series | 22 | 22 | Mixed (12 OS, 10 OD) | Multiple locations reported | Single | Unilateral | Gaze evoked intumescence; valsalva evoked intumescence; collapse with globe pressure | OCT, B-scan, ICGA, IR | ||
| de Carlo et al. 2021 [18] | Imaging case series | 4 | 4 | Mixed (2 OD, 2 OS) | Multiple (2 inferotemporal, 2 superonasal, 1 superotemporal) | One patient with multiple | Unilateral | Valsalva evoked intumescence; collapse with globe pressure | B-scan | Choroidal nevus, choroidal hemorrhage, & choroidal neoplasm mimic | |
| da Cruz et al. 1994 [41] | Case report | 1 | 1 | OD | Multiple (1 superior, 1 superonasal) | Multiple | Unilateral | Gaze evoked intumescence; position dependent; collapse with globe pressure | Choroidal metastases mimic | ||
| Cabral et al. 2022 [23] | Case report | 1 | 1 | Unable to access complete article | Inferior | Single | Unilateral | Unable to access complete article | OCT, B-scan, FA, ICGA | Possible association with nodular posterior scleritis | Unable to access complete article |
| Buettner et al. 1990 [4] | Case report | 1 | 1 | OD | Superonasal | Single | Unilateral | collapse w/ globe pressure; no change in intumescence with gaze change or patient positioning | B-scan | Choroidal melanoma mimic | Patient presenting with persistently engorged vortex vein ampulla varix. |
| Al-Dahmash et al. 2021 [42] | Case report | 1 | Unable to assess due to article not being in English | OD | Unable to assess due to article not being in English | Multiple | Unable to assess due to article not being in English | Unable to assess due to article not being in English | Unable to assess due to article not being in English | Choroidal metastases mimic | Article not in English so data abstraction relied on english-translated abstract |
| Adam et al. 2013 [43] | Case report | 1 | 1 | Unable to access complete article | Parafoveal | Single | Unilateral | Unable to access complete article | OCT, FA, ICGA | Unable to access complete article. Description of a submacular varix. |
4. Discussion
4.1. Literature on the Topic of Multimodal Imaging
4.2. Literature on the Topic of Associated Systemic and Ocular Conditions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| B-scan | Standard B-scan ultrasound |
| ICGA | Indocyanine green angiography |
| FA | Fluorescein Angiography |
| OCT | Optical Coherence Tomography |
| OCT-A | Optical coherence tomography-angiography |
References
- Osher, R.H.; Abrams, G.W.; Yarian, D.; Armao, D. Varix of the vortex ampulla. Am J Ophthalmol. 1981, 92(5), 653–60. [Google Scholar] [CrossRef]
- Verma, A.; Maram, J.; Alagorie, A.R.; Gupta Nittala, M.; van Hemert, J.; Keane, D.; et al. Distribution and Location of Vortex Vein Ampullae in Healthy Human Eyes as Assessed by Ultra-Widefield Indocyanine Green Angiography. Ophthalmol Retina 2020, 4(5), 530–4. [Google Scholar] [CrossRef]
- He, G.; Zhang, X.; Zhuang, X.; Zeng, Y.; Gan, Y.; Su, Y.; et al. A Novel Exploration of the Choroidal Vortex Vein System: Incidence and Characteristics of Posterior Vortex Veins in Healthy Eyes. Invest Ophthalmol Vis Sci. 2024, 65(2), 21. [Google Scholar] [CrossRef] [PubMed]
- Buettner, H. Varix of the vortex ampulla simulating a choroidal melanoma. Am J Ophthalmol. 1990, 109(5), 607–8. [Google Scholar] [CrossRef]
- Veronese, C.; Staurenghi, G.; Pellegrini, M.; Maiolo, C.; Primavera, L.; Morara, M.; et al. Multimodal Imaging in Vortex Vein Varices. Retin Cases Brief Rep. 2019, 13(3), 260–5. [Google Scholar] [CrossRef]
- Ismail, R.A.; Sallam, A.; Zambarakji, H.J. Optical coherence tomographical findings in a case of varix of the vortex vein ampulla. Br J Ophthalmol. 2011, 95(8), 1169–70, 82. [Google Scholar] [CrossRef]
- Vahdani, K.; Kapoor, B.; Raman, V.S. Multiple vortex vein ampulla varicosities. BMJ Case Rep. 2010. [Google Scholar] [CrossRef]
- Suetov, A.A.; Boiko, E.V.; Izmaylov, A.S.; Molodkina, N.A. Vortex vein varix (clinical observations). Vestn Oftalmol. 2022, 138(4), 81–6. [Google Scholar] [CrossRef] [PubMed]
- Fogel Levin, M.; Hostovsky, A.; Fossataro, C.; Caputo, C.G.; Feo, A.; Romano, M.R.; et al. Vortex Vein Varix: Multimodal Imaging and Clinical Correlates. Ophthalmol Retina 2025. [Google Scholar] [CrossRef] [PubMed]
- Rutnin, U. Fundus appearance in normal eyes. I. The choroid. Am J Ophthalmol. 1967, 64(5), 821–39. [Google Scholar] [CrossRef] [PubMed]
- Wolfensberger, T.J. [Varix of the vortex ampulla: an unusual differential choroid tumor diagnosis]. Klin Monbl Augenheilkd 1997, 210(5), 334–6. [Google Scholar] [CrossRef]
- Kang, H.K.; Beaumont, P.E.; Chang, A.A. Indocyanine green angiographic features of varix of the vortex vein ampulla. Clin Exp Ophthalmol. 2000, 28(4), 321–3. [Google Scholar] [CrossRef]
- Levy, J.; Yagev, R.; Shelef, I.; Lifshitz, T. Varix of the vortex vein ampulla: a small case series. Eur J Ophthalmol. 2005, 15(3), 424–7. [Google Scholar] [CrossRef]
- Rimsaite, A.; Andersen, C.U. [A varix of the vortex vein ampulla is a rare differential diagnosis of malignant melanoma of the choroid]. Ugeskr Laeger 2015, 177(8). [Google Scholar]
- Rubegni, G.; Bacci, T.; Tosi, G. Vortex Vein Varix changes with digital pressure: Ultra-Wide-Field imaging and Peripheral OCT report. Am J Ophthalmol. 2025, 272, e5–e6. [Google Scholar] [CrossRef]
- Hu, Y.; Wang, S.; Dong, Y.; Zhou, X.; Yu, W.; et al. Imaging Features of Varix of the Vortex Vein Ampulla: A Small Case Series. J Clinic Experiment Ophthalmol 2011, 2, 173. [Google Scholar] [CrossRef]
- Shields, C.L.; Shields, J.A.; De Potter, P. Patterns of indocyanine green videoangiography of choroidal tumours. Br J Ophthalmol. 1995, 79(3), 237–45. [Google Scholar] [CrossRef] [PubMed]
- de Carlo, T.E.; Mieler, W. Dynamic Echography of Varix of the Vortex Vein Ampulla. Retin Cases Brief Rep. 2021, 15(5), 548–51. [Google Scholar] [CrossRef]
- Spiess, K.; Elgohary, M.A. Diagnosis of Vortex Varix Using Optical Coherence Tomography and Scleral Indentation. Retin Cases Brief Rep. 2022, 16(3), 362–4. [Google Scholar] [CrossRef]
- Rayess, H.; Ehlers, J.P. Utilization of peripheral optical coherence tomography to optimize diagnosis of a vortex vein varix. Retina 2015, 35(4), 838–9. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, M.Z.; Sanders, R.; Sallam, A.B. Utilization of Intraoperative OCT for the Diagnosis of a Case of Varix of the Vortex Vein. Ophthalmol Retina 2019, 3(6), 522. [Google Scholar] [CrossRef]
- Zhang, X.; Olson, D.J.; DiBernardo, C.; Davis, R.M.; Gordon, K.G.B. Scleritis-associated vortex vein varix masquerading as choroidal melanoma. Can J Ophthalmol. 2018, 53(6), e260–e2. [Google Scholar] [CrossRef] [PubMed]
- Cabral, D.; Nogueira, V. Varix of a Vortex Vein Ampulla Induced by Nodular Scleritis. Retin Cases Brief Rep. 2022, 16(3), 325–8. [Google Scholar] [CrossRef] [PubMed]
- Higham, A.; Hildebrand, G.D.; Graham-Evans, K.A.J.; Gilbert, R.D.; Horton, R.; Hunt, D.; et al. Ectopic vortex veins and varices in Donnai Barrow syndrome. Ophthalmic Genet. 2022, 43(2), 248–52. [Google Scholar] [CrossRef]
- Gass, J.D. Uveal effusion syndrome. A new hypothesis concerning pathogenesis and technique of surgical treatment. Retina 1983, 3(3), 159–63. [Google Scholar] [CrossRef]
- Ghadiali, Q.; Tan, A.; Freund, K.B. Unusual Posterior Varix of a Vortex Vein Ampulla. Retin Cases Brief Rep. 2017, 11 Suppl 1, S163–S5. [Google Scholar] [CrossRef]
- Milani, P.; Mazzola, M.; Bergamini, F. Suprachoroidal haemhorrage and vortex vein varix: A potential association. Eur J Ophthalmol. 2022, 32(1), NP130–NP3. [Google Scholar] [CrossRef]
- Chung, S.E.; Kang, S.W.; Kim, J.H.; Kim, Y.T.; Park, D.Y. Engorgement of vortex vein and polypoidal choroidal vasculopathy. Retina 2013, 33(4), 834–40. [Google Scholar] [CrossRef]
- Weidmayer, S.L.; Demirci, H. The spontaneous resolution of a vortex vein varix: case report. BMC Ophthalmol. 2021, 21(1), 101. [Google Scholar] [CrossRef]
- Snyers, B.; De Potter, P. [Advantages of digital indocyanine green angiography for diagnosing choroidal tumors]. J Fr Ophtalmol. 2002, 25(2), 212–20. [Google Scholar] [PubMed]
- Singh, A.D.; De Potter, P.; Shields, C.L.; Shields, J.A. Indocyanine green angiography and ultrasonography of a varix of vortex vein. Arch Ophthalmol. 1993, 111(9), 1283–4. [Google Scholar] [CrossRef] [PubMed]
- Niu, T.T.; Xiao, Y. Varix of the vortex vein ampulla: a case report and imaging correlation. Front Med (Lausanne) 2025, 12, 1685952. [Google Scholar] [CrossRef]
- Nitta, K.; Akiyama, H. Different Vortex Vein Anomalies Observed in a Single Case: Macular Vortex Vein in One Eye and Varix of Vortex Vein Ampulla in the Other Eye. Cureus 2024, 16(7), e63668. [Google Scholar] [CrossRef]
- Murtagh, P.; O'Dwyer, G.; Horgan, N. Vortex Vein Ampulla. Ophthalmology 2021, 128(12), 1707. [Google Scholar] [CrossRef]
- Lopez, P. Varix of the vortex vein ampulla. J Am Optom Assoc. 1986, 57(2), 104–8. [Google Scholar]
- Khan, J.; Damato, B.E. Accuracy of choroidal melanoma diagnosis by general ophthalmologists: a prospective study. Eye (Lond) 2007, 21(5), 595–7. [Google Scholar] [CrossRef]
- Kang, T.D.; Douglass, A.M.; Ferenczy, S.R.; Say, E.A.; Shields, C.L. In Vivo Hemodynamic Changes of Vortex Vein Varix on Real-Time Video Angiography. Retina 2017, 37(2), e8–e9. [Google Scholar] [CrossRef]
- Hunter, J.E. Vortex vein varix. Am J Optom Physiol Opt. 1983, 60(12), 995–6. [Google Scholar] [CrossRef]
- Gunduz, K.; Shields, C.L.; Shields, J.A. Varix of the vortex vein ampulla simulating choroidal melanoma: report of four cases. Retina 1998, 18(4), 343–7. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Bardera, J.; Montolio-Marzo, E.; Etxabe-Avila, H.; Lorenzo-Castro, J.; Garcia-Caride, S. Valsalva-induced changes in vortex vein varices: A comprehensive imaging exploration. J Fr Ophtalmol. 2024, 47(8), 104250. [Google Scholar] [CrossRef] [PubMed]
- da Cruz, L.; James, B.; Gray, R.; Elston, J. Multiple vortex vein varices masquerading as choroidal secondaries. Br J Ophthalmol. 1994, 78(10), 800–1. [Google Scholar] [CrossRef] [PubMed]
- Al-Dahmash, S.A.; AlBloushi, A.F.; Alsarhani, W.K. Multiple giant vortex vein varices masquerading as choroidal metastases. J Fr Ophtalmol. 2021, 44(1), e31–e3. [Google Scholar] [CrossRef] [PubMed]
- Adam, C.R.; Sigler, E.J.; Randolph, J.C.; Calzada, J.I. Submacular choroidal varix simulating chorioretinal folds with metamorphopsia. Ophthalmic Surg Lasers Imaging Retina 2013, 44(6), 596–8. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




