Submitted:
17 March 2026
Posted:
18 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
3.1. Patients
3.2. Prevalence of Potassium, Glycemia, BUN and Creatinine Disturbances
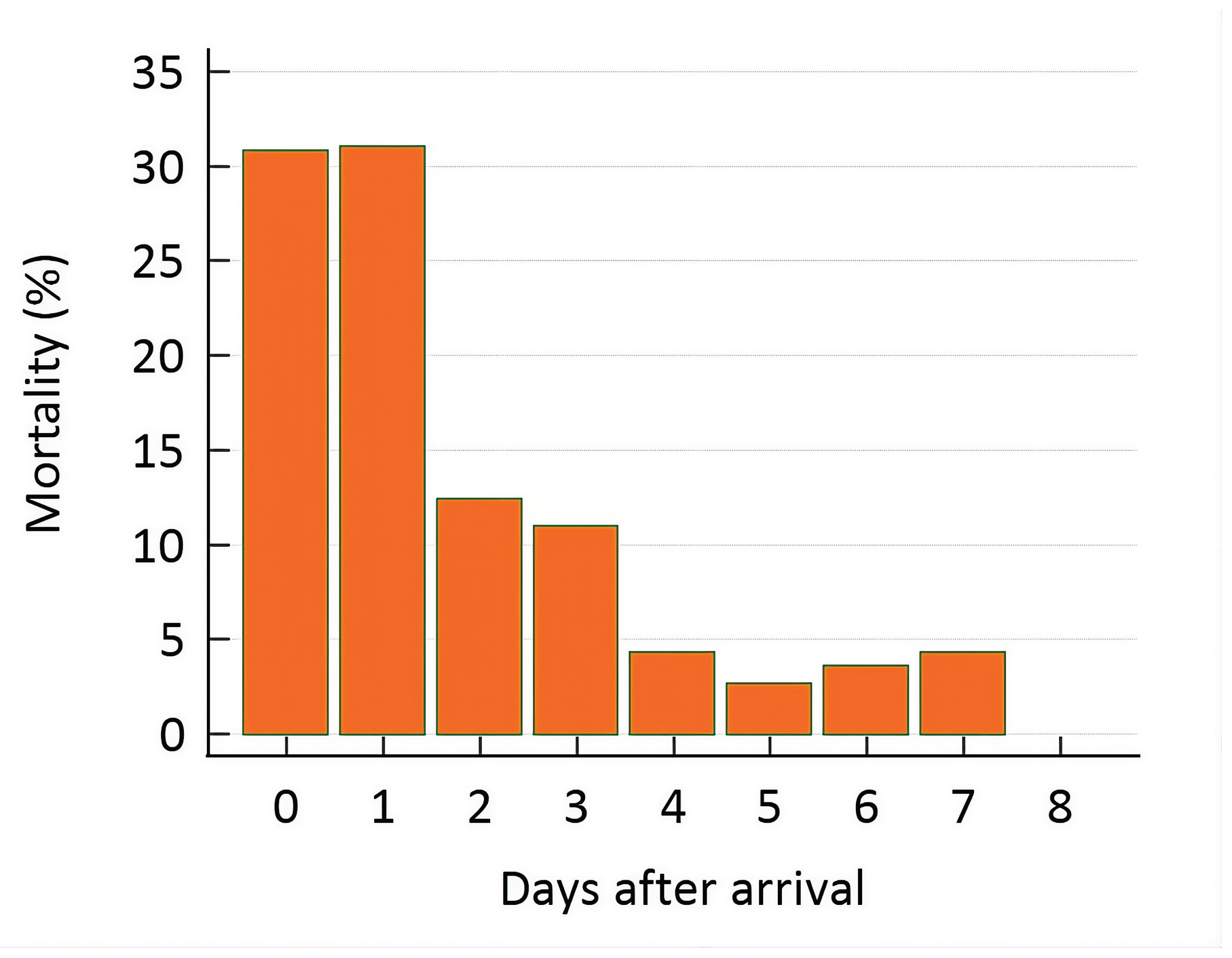
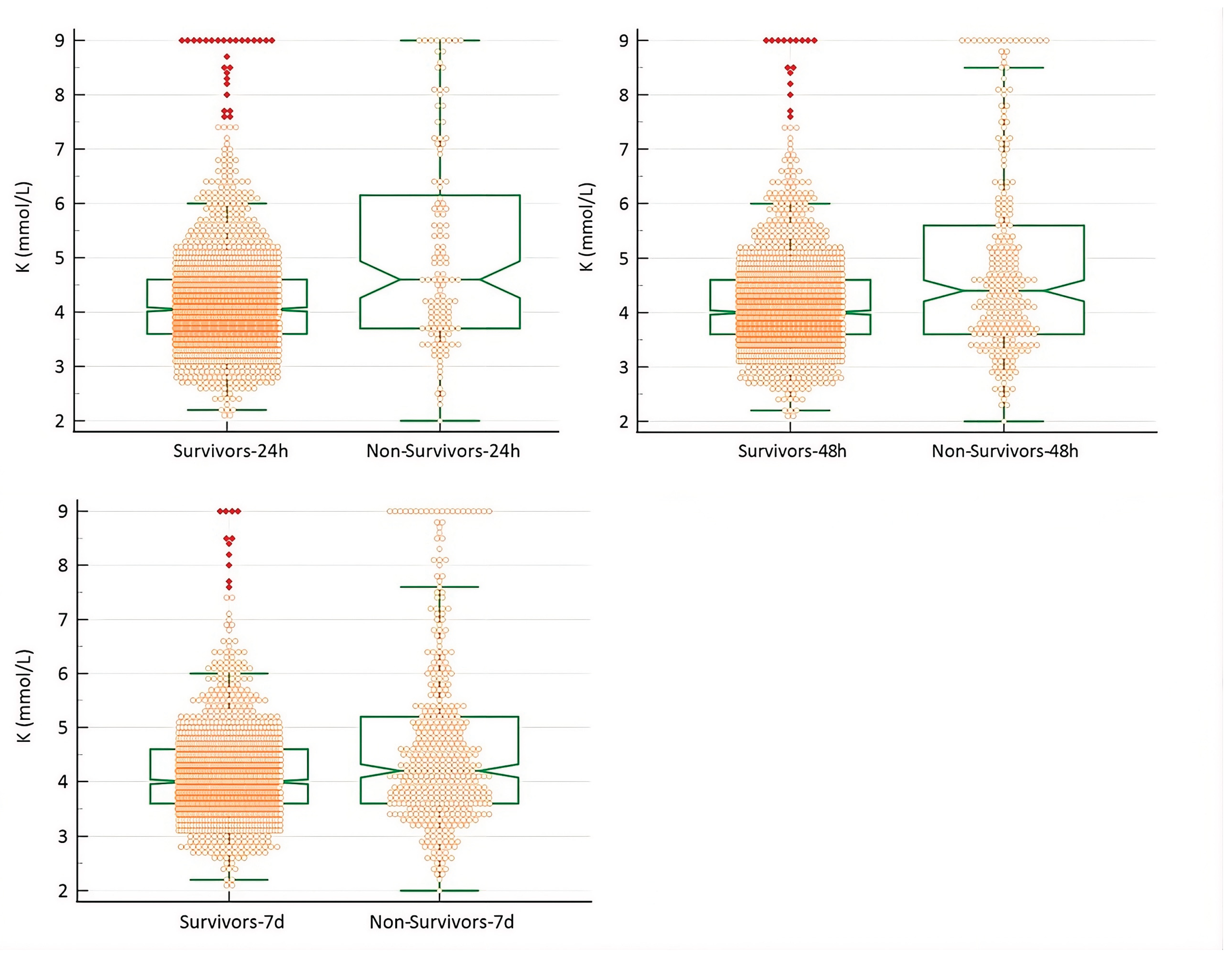
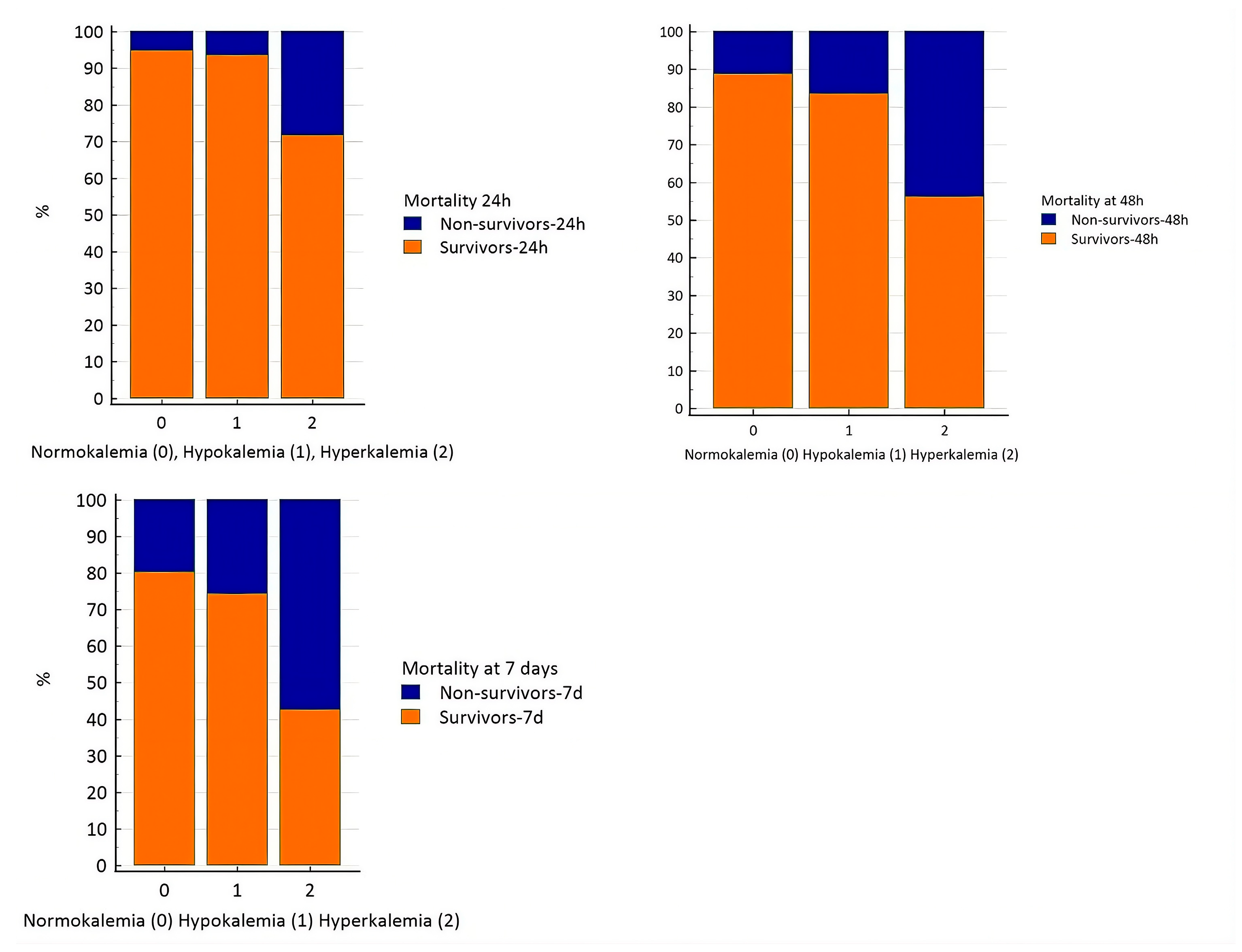
3.2. Potassium Disturbances and Mortality
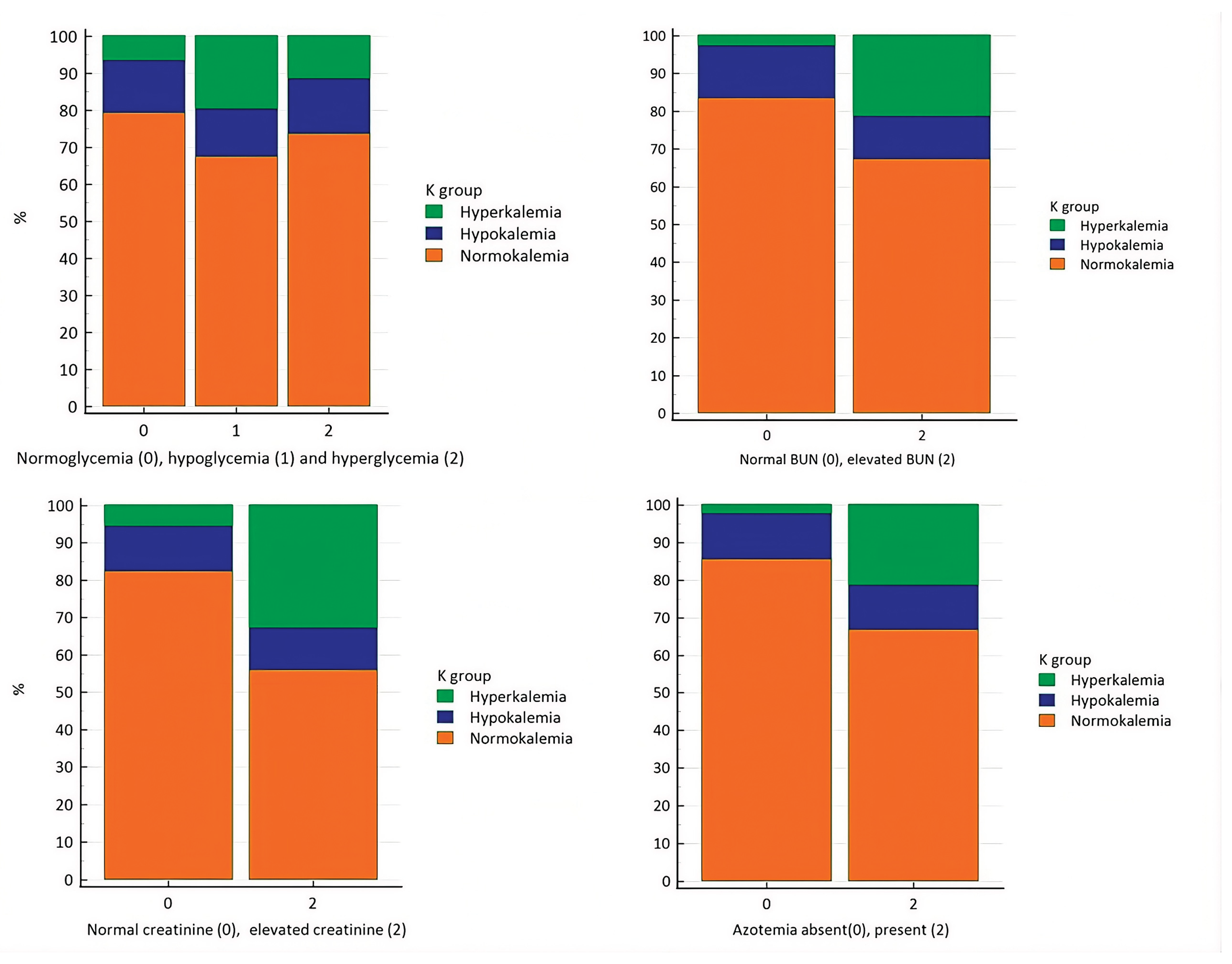
3.2. Potassium Disturbances and Their Association with Glycemia and Renal Disease Markers (BUN and Creatinine)
4. Discussion
5. Conclusions
References
- Willard, M.D. Disorders of Potassium Homeostasis. Vet. Clin. North Am. Small Anim. Pract. 1989, 19, 241–263. [Google Scholar] [CrossRef]
- Palmer, B.F.; Clegg, D.J. Physiology and Pathophysiology of Potassium Homeostasis: Core Curriculum 2019. Am. J. Kidney Dis. 2019, 74, 682–695. [Google Scholar] [CrossRef]
- Hoehne, S.N.; Hopper, K.; Epstein, S.E. Retrospective Evaluation of the Severity of and Prognosis Associated with Potassium Abnormalities in Dogs and Cats Presenting to an Emergency Room (January 2014-August 2015): 2441 Cases. J. Vet. Emerg. Crit. Care. 2019, 29, 653–661. [Google Scholar] [CrossRef]
- Sweeney, R.W. Treatment of Potassium Balance Disorders. Vet. Clin. North Am. Food Anim. Pract. 1999, 15, 609–617. [Google Scholar] [CrossRef]
- Beech, J.; Lindborg, S.; Braund, K.G. Potassium Concentrations in Muscle, Plasma and Erythrocytes and Urinary Fractional Excretion in Normal Horses and Those with Chronic Intermittent Exercise-Associated Rhabdomyolysis. Res. Vet. Sci. 1993, 55, 43–51. [Google Scholar] [CrossRef]
- Kogika, M.M.; de Morais, H.A. A Quick Reference on Hypokalemia. Vet. Clin. North Am. Small Anim. Pract. 2017, 47, 229–234. [Google Scholar] [CrossRef]
- Kettritz, R.; Loffing, J. Potassium Homeostasis – Physiology and Pharmacology in a Clinical Context. Pharmacol. Ther. 2023, 249, 108489. [Google Scholar] [CrossRef]
- Yamada, S.; Inaba, M. Potassium Metabolism and Management in Patients with CKD. Nutrients 2021, 13(6), 1751. [Google Scholar] [CrossRef] [PubMed]
- Schneider, S.; Müller, A.; Wittek, T. Concentration of Potassium in Plasma, Erythrocytes, and Muscle Tissue in Cows with Decreased Feed Intake and Gastrointestinal Ileus. J. Vet. Intern. Med. 2016, 30, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Harcourt-Brown, F.M.; Harcourt-Brown, S. Clinical Value of Blood Glucose Measurement in Pet Rabbits. Vet. Rec. 2012, 170(26), 674. [Google Scholar] [CrossRef] [PubMed]
- Galindo, R.J.; Fayfman, M.; Umpierrez, G.E. Expert Consensus on the Glycemic Management of Critically Ill Patients. J. Intensive Med. 2022, 2, 131–145. [Google Scholar] [CrossRef]
- Godinjak, A.; Iglica, A.; Burekovic, A.; Jusufovic, S.; Ajanovic, A.; Tancica, I.; Kukuljac, A. Hyperglycemia in Critically Ill Patients: Management and Prognosis. Med. Arch. 2015, 69(3), 157–160. [Google Scholar] [CrossRef]
- Shrewsberry, W.C.; MacVicar, R. The Effect of High Potassium Intake on Rabbits. In Proceedings of the Oklahoma Academy of Science, 1953; pp. 170–172. [Google Scholar]
- Gallego, M. Laboratory Reference Intervals for Systolic Blood Pressure, Rectal Temperature, Haematology, Biochemistry and Venous Blood Gas and Electrolytes in Healthy Pet Rabbits. Open Vet. J. 2017, 7, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Grace, S.A.; Munday, K.A.; Noble, A.R. Sodium, Potassium and Water Metabolism in the Rabbit: The Effect of Sodium Depletion and Repletion. J. Physiol. 1979, 292, 407–420. [Google Scholar] [CrossRef]
- Ardiaca, M.; Bonvehí, C.; Montesinos, A. Point-of-Care Blood Gas and Electrolyte Analysis in Rabbits. Vet. Clin. North Am. Exot. Anim. Pract. 2013, 16, 175–195. [Google Scholar] [CrossRef] [PubMed]
- Ardiaca García, M.; Montesinos Barceló, A.; Bonvehí Nadeu, C.; del Caz Calvo, A. Blood Gas and Electrolyte Analysis in Rabbits (N=108) Presented with Acute Gastric Dilatation. In Proceedings of the 5th International Conference on Avian, Herpetological and Exotic Mammal Medicine (online event), Budapest, Hungary, 27-30 March 2022; p. 230. [Google Scholar]
- Bonvehí, C.; Ardiaca, M.; Montesinos, A.; Juan-Sallés, C.; Gómez, A.; Teso, B.; Barbero, S.; Ferrera, S.E. Clinicopathologic Findings of Naturally Occurring Rabbit Hemorrhagic Disease Virus 2 Infection in Pet Rabbits. Vet. Clin. Pathol. 2019, 48, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Tausch, K.A. Untersuchungen Über Den Einsatz Des I-STAT®-Blutanalysegerätes Zur Blutgasanalyse Und Bestimmung Weiterer Labordiagnostischer Parameter Bei Kaninchen Und Meerschweinchen. In Dissertation; Tierärztliche Fakultät: Ludwig Maximilians Universität München, 2011. [Google Scholar] [CrossRef]
- Melillo, A. Rabbit Clinical Pathology. J. Exot. Pet Med. 2007, 16, 135–145. [Google Scholar] [CrossRef]
- Bonvehi, C.; Ardiaca, M.; Barrera, S.; Cuesta, M.; Montesinos, A. Prevalence and Types of Hyponatraemia, Its Relationship with Hyperglycaemia and Mortality in Ill Pet Rabbits. Vet. Rec. 2014, 174, 554–559. [Google Scholar] [CrossRef]
- Johnson, P.J. Electrolyte and Acid-Base Disturbances in the Horse. Vet. Clin. North Am. Equine Pract. 1995, 11, 491–514. [Google Scholar] [CrossRef]
- Boag, A.K.; Coe, R.J.; Martinez, T.A.; Hughes, D. Acid-Base and Electrolyte Abnormalities in Dogs with Gastrointestinal Foreign Bodies. J. Vet. Intern. Med. 2005, 19, 816–821. [Google Scholar] [CrossRef]
- Szmacinski, H.; Lakowicz, J.R. Potassium and Sodium Measurements at Clinical Concentrations Using Phase-Modulation Fluorometry. Sens. Actuators B Chem. 2000, 60(1), 8–18. [Google Scholar] [CrossRef]
- Hart, K.A.; Barton, M.H. Adrenocortical Insufficiency in Horses and Foals. Vet. Clin. North Am. Equine Pract. 2011, 27(1), 19–34. [Google Scholar] [CrossRef] [PubMed]
- Fox, R.R.; Crary, D.D. Genetics and Pathology of Hereditary Adrenal Hyperplasia in the Rabbit: A Model for Congenital Lipoid Adrenal Hyperplasia. J. Hered. 1978, 69, 251–254. [Google Scholar] [CrossRef] [PubMed]
- Jekl, V. Adrenal Disease in Small Mammals. Vet. Clin. North Am. Exot. Anim. Pract. 2025, 28, 87–106. [Google Scholar] [CrossRef]
- Franco-Acevedo, A.; Echavarria, R.; Melo, Z. Sex Differences in Renal Function: Participation of Gonadal Hormones and Prolactin. Endocrines 2021, Vol. 2, 185–202. [Google Scholar] [CrossRef]
- Robert, R.; Ghazali, D.A.; Favreau, F.; Mauco, G.; Hauet, T.; Goujon, J.M. Gender Difference and Sex Hormone Production in Rodent Renal Ischemia Reperfusion Injury and Repair. J. Inflamm. (Lond). 2011, 8, 14–22. [Google Scholar] [CrossRef]
- Iwasa, N.; Takashima, S.; Iwasa, T.; Kumazawa, R.; Nomura, S.; Asami, S.; Shimizu, M.; Kobatake, Y.; Nishii, N. Effect of Age, Sex, and Breed on Serum Cystatin C and Creatinine Concentrations in Dogs. Vet. Res. Commun. 2022, 46, 183–188. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, J.G.; Wong, F.; Reddy, K.R.; Garcia-Tsao, G.; Kamath, P.S.; Biggins, S.W.; Fallon, M.B.; Subramanian, R.M.; Maliakkal, B.; Thacker, L.; et al. Gender-Specific Differences in Baseline, Peak, and Delta Serum Creatinine: The NACSELD Experience. Dig. Dis. Sci. 2017, 62, 768–776. [Google Scholar] [CrossRef] [PubMed]
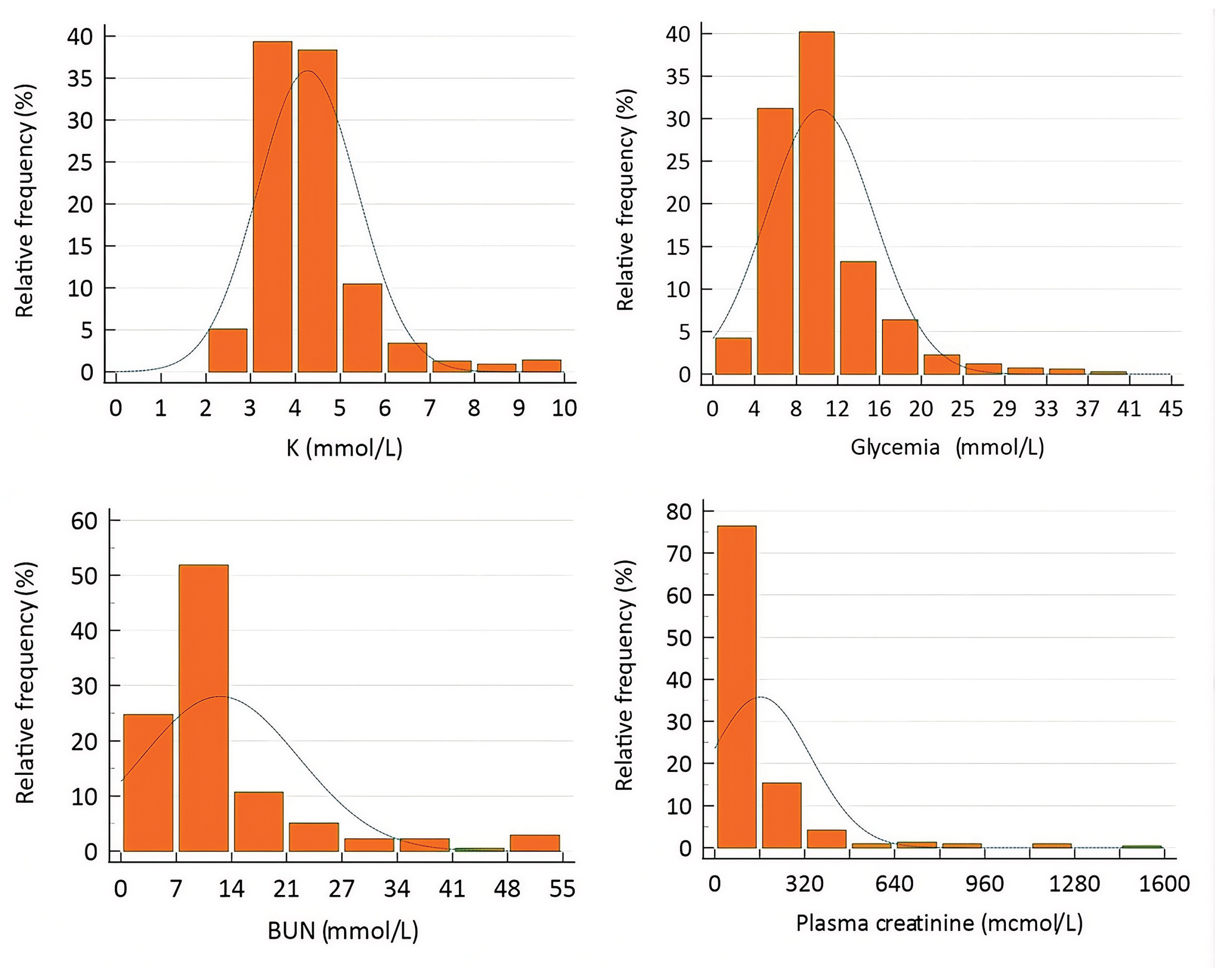
| K+ (mmol/L) | Glucose (mmol/L) |
BUN (mmol/L) |
Crea (µmol/L) |
|
|---|---|---|---|---|
| N | 1773 | 1649 | 1382 | 271 |
| Minimum | 2.0 | 0.83 | 1.1 | 17.7 |
| Maximum | 9.0 | 38.9 | 50.9 | 1644.2 |
| Mean | 4.3 | 10.6 | 13.8 | 205.3 |
| Median | 4.1 | 9.3 | 9.5 | 114.9 |
| Standard deviation | 1.11 | 5.55 | 11.53 | 265.45 |
| Relative standard deviation | 0.26 | 0.52 | 0.84 | 1.29 |
| 25 - 75 Percentiles | 3.6 to 4.7 | 7.3 to 12.2 | 6.9 to 14.9 | 88.4 to 185.6 |
| Kholmogorov-Smirnov test for Normal distribution (p value) | <0.0001 | <0.0001 | <0.0001 | <0.0001 |
| Outliers (Tukey test) N Range |
50 6.4-8.0 |
74 19.6-26.4 |
81 26.9-38.5 |
12 335.9-468.5 |
| Far out values (Tukey test) N Range |
38 8.1-9.0 |
40 27.0-38.9 |
87 39.3-50.9 |
22 477.4-1644.2 |
| N values under lower limit of detection | 1 (<2mmol/L) | 20 (<1.1mmol/L) | 0 | 0 |
| N values over upper limit of detection | 11 (>9mmol/L) | 6 (>38.9mmol/L) | 59 (>50.9mmol/L) | 0 |
| Outcome 24h | Outcome 48h | Outcome 7d | |
|---|---|---|---|
| Hypokalemia Relative risk Odds ratio |
1.2 (P=0.4404) 1.2 (P=0.4424) |
1.5 (P=0.0162)* 1.6 (P=0.0185)* |
1.3 (P=0.0519) 1.4 (P=0.0574) |
| Hyperkalemia Relative risk Odds ratio |
5.4 (P<0.0001)* 7.1 (P<0.0001)* |
3.9 (P<0.0001)* 6.2 (P<0.0001)* |
2.9 (P<0.0001)* 5.4 (P<0.0001)* |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




