Submitted:
17 March 2026
Posted:
18 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. YF Virus Overview
3. YF in Non-Human Primates
4. Clinical Findings in New and Old World NHPs
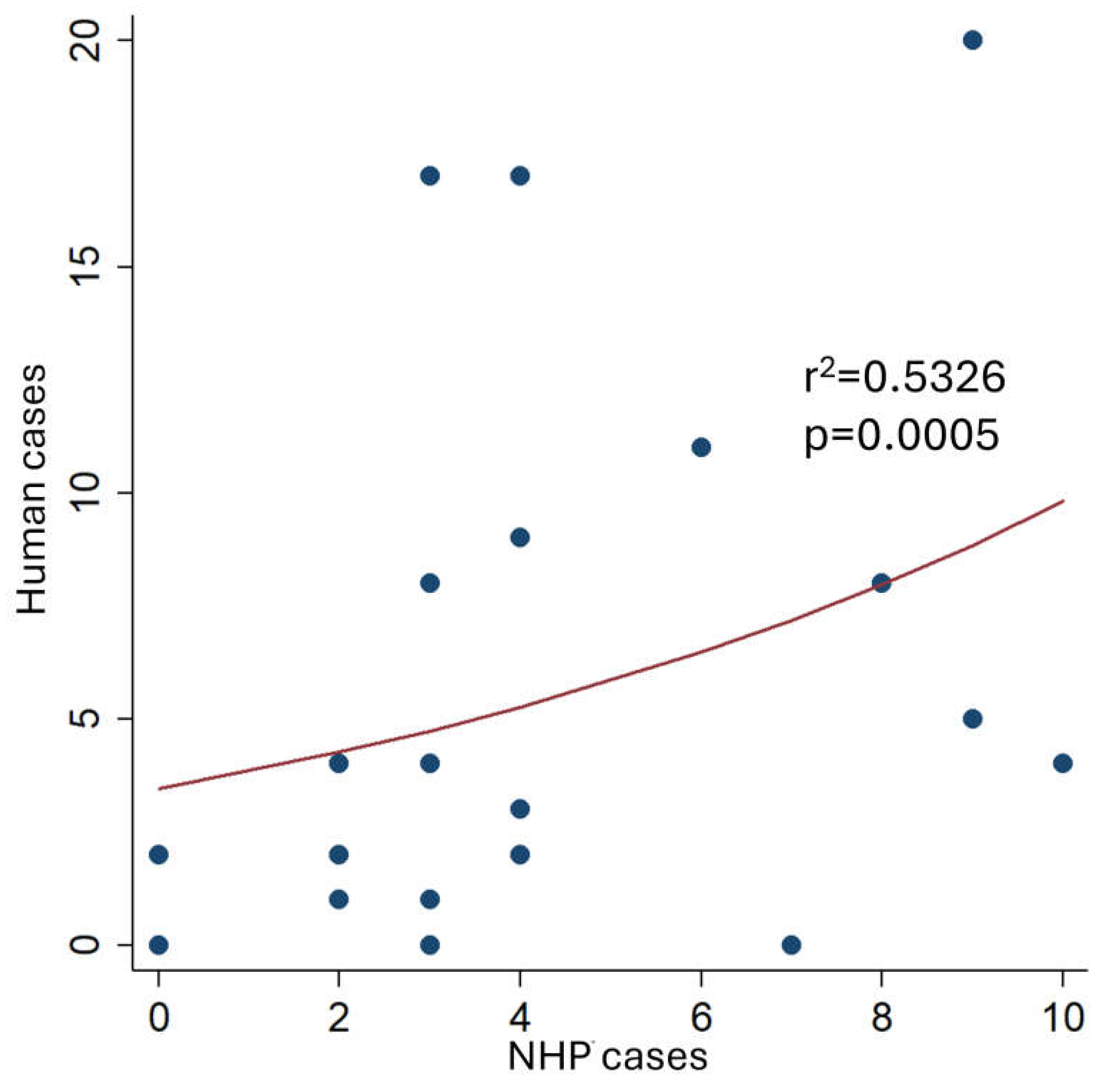
5. Epizootics in the Last Decade
6. Genomic Surveillance of YF in the Context of Epizootics
7. Importance of One Health Surveillance in YF Virus and Non-Human Primates
8. Prevention Mechanisms of the YF Virus in Non-Human Primates
9. Surveillance and Response Protocols for Suspected YF in Non-Human Primates
10. Limitations
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Valderrama, A.; Díaz, Y.; López-Vergès, S. Interaction of Flavivirus with their mosquito vectors and their impact on the human health in the Americas. Biochemical and biophysical research communications 2017, 492, 541–547. [Google Scholar] [CrossRef]
- Burdmann, E.A. Flaviviruses and Kidney Diseases. Advances in chronic kidney disease 2019, 26, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Cuéllar-Sáenz, J.A.; Rodríguez-Morales, A.J.; Faccini-Martínez Á, A. Reemergence of Yellow Fever, Magdalena Valley, Colombia, 2024-2025. Emerging infectious diseases 2025, 31, 2216–2224. [Google Scholar] [CrossRef]
- Srivastava, S.; Jayaswal, N.; Gupta, P.; Sridhar, S.B.; Jaiswal, P.; Tariq, M.; Rao, G.; Mohanty, A.; Sah, S.; Mehta, R.; et al. The Yellow Fever Vaccine Journey: Milestones and Future Directions. Vaccines 2026, 14. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Morales, A.J. Deforestation and zoonotic arboviral diseases: A One Health challenge in the context of Oropouche and Yellow Fever re-emergence in Latin America. German Journal of Veterinary Research 2026, 6, 9–15. [Google Scholar] [CrossRef]
- Bonilla-Aldana, D.K.; Bonilla-Aldana, J.L.; Castellanos, J.E.; Rodriguez-Morales, A.J. Importance of Epizootic Surveillance in the Epidemiology of Yellow Fever in South America. Current Tropical Medicine Reports 2025, 12, 16. [Google Scholar] [CrossRef]
- de Oliveira Figueiredo, P.; Stoffella-Dutra, A.G.; Costa, G.B.; de Oliveira, J.S.; Amaral, C.D.; Alves, P.A.; Filho, J.D.A.; Paz, G.F.; Tonelli, G.B.; Kroon, E.G.; et al. Absence of yellow fever virus circulation in wildlife rodents from Brazil. Brazilian journal of microbiology: [publication of the Brazilian Society for Microbiology] 2022, 53, 647–654. [Google Scholar] [CrossRef]
- Escalera-Antezana, J.P.; Aviles-Sarmiento, J.L.; Montenegro-Narvaez, C.M.; Castro-Calderon, H.A.; Bonilla-Aldana, J.L.; Bonilla-Aldana, D.K.; Rodriguez-Morales, A.J. Yellow fever among captive non-human primates in La Paz, Bolivia, 2025. New Microbes and New Infections 2026, 69, 101695. [Google Scholar] [CrossRef]
- Santos, D.O.D.; de Oliveira, A.R.; de Lucena, F.P.; de Mattos, S.A.; de Carvalho, T.P.; Costa, F.B.; Moreira, L.G.A.; Paixão, T.A.D.; Santos, R.L. Histopathologic Patterns and Susceptibility of Neotropical Primates Naturally Infected With Yellow Fever Virus. Veterinary pathology 2020, 57, 681–686. [Google Scholar] [CrossRef]
- Tavares da Silva Fernandes, A.; Moreira, S.B.; Gaspar, L.P.; Simões, M.; Cajaraville, A.; Pereira, R.C.; Gomes, M.P.B.; Linhares, J.H.R.; Santos, V.O.; Santos, R.T.; et al. Safety and immunogenicity of 17DD attenuated yellow fever vaccine in howler monkeys (Alouatta spp.). Journal of medical primatology 2021, 50, 36–45. [Google Scholar] [CrossRef]
- MacDonald, A.J.; Sipin, T.J.; Ring, K. Spillover risk for yellow fever virus is amplified by forest-urban interface in the Amazon. Biol Lett 2025, 21. [Google Scholar] [CrossRef]
- Costa, G.R.d.T.; Passos, P.H.d.O.; Sousa, D.E.R.d.; Macêdo, I.L.d.; Ramos, D.G.; Cavalcante, K.R.L.J.; Romano, A.P.M.; Martins Filho, A.J.; Casseb, L.M.N.; Martins, L.C.; et al. Surveillance Service of Yellow Fever in Non-Human Primates in the Federal District, Brazil, 2008-2022. Revista da Sociedade Brasileira de Medicina Tropical 2026, 59. [Google Scholar] [CrossRef]
- Cunha, M.S.; Guerra, J.M.; Siconelli, M.J.L.; Fonseca, B.; Dias, J.C.A.; Freitas, G.D.; Sabino, E.C.; Manuli, E.R.; Pereira, G.M.; Valença, I.N.; et al. Yellow fever virus resurgence in Sao Paulo State, Brazil, 2024-2025. Rev Inst Med Trop Sao Paulo 2026, 68, e13. [Google Scholar] [CrossRef]
- Salomón, O.D.; Arias, A.R. The second coming of urban yellow fever in the Americas: looking the past to see the future. Anais da Academia Brasileira de Ciencias 2022, 94, e20201252. [Google Scholar] [CrossRef]
- Johnson, E.; Sunil Kumar Sharma, R.; Ruiz Cuenca, P.; Byrne, I.; Salgado-Lynn, M.; Suraya Shahar, Z.; Col Lin, L.; Zulkifli, N.; Dilaila Mohd Saidi, N.; Drakeley, C.; et al. Landscape drives zoonotic malaria prevalence in non-human primates. eLife 2024, 12. [Google Scholar] [CrossRef] [PubMed]
- Couto-Lima, D.; Madec, Y.; Bersot, M.I.; Campos, S.S.; Motta, M.A.; Santos, F.B.D.; Vazeille, M.; Vasconcelos, P.; Lourenço-de-Oliveira, R.; Failloux, A.B. Potential risk of re-emergence of urban transmission of Yellow Fever virus in Brazil facilitated by competent Aedes populations. Scientific reports 2017, 7, 4848. [Google Scholar] [CrossRef]
- Saad, L.D.C.; Chiaravalloti-Neto, F. Reemergence of yellow fever in the state of São Paulo: the structuring role of surveillance of epizootics in non-human primates in a one health approach. Revista brasileira de epidemiologia = Brazilian journal of epidemiology 2024, 27, e240064. [Google Scholar] [CrossRef] [PubMed]
- Pérez Pérez, A.; Arranz Izquierdo, J.; Navarro Beltrá, M. [The impact of global change on infectious disease epidemiology]. Atencion primaria 2026, 58, 103375. [Google Scholar] [CrossRef]
- Salvarani, F.M.; Oliveira, H.; Correa, L.Y.S.; Soares, A.A.L.; Ferreira, B.C. The Importance of Studying Infectious and Parasitic Diseases of Wild Animals in the Amazon Biome with a Focus on One Health. Veterinary sciences 2025, 12. [Google Scholar] [CrossRef] [PubMed]
- Arsevska, E.; Hellal, J.; Mejri, S.; Hammami, S.; Marianneau, P.; Calavas, D.; Hénaux, V. Identifying Areas Suitable for the Occurrence of Rift Valley Fever in North Africa: Implications for Surveillance. Transboundary and emerging diseases 2016, 63, 658–674. [Google Scholar] [CrossRef]
- Mares-Guia, M.; Horta, M.A.; Romano, A.; Rodrigues, C.D.S.; Mendonça, M.C.L.; Dos Santos, C.C.; Torres, M.C.; Araujo, E.S.M.; Fabri, A.; de Souza, E.R.; et al. Yellow fever epizootics in non-human primates, Southeast and Northeast Brazil (2017 and 2018). Parasites & vectors 2020, 13, 90. [Google Scholar] [CrossRef]
- Sacchetto, L.; Drumond, B.P.; Han, B.A.; Nogueira, M.L.; Vasilakis, N. Re-emergence of yellow fever in the neotropics - quo vadis? Emerging topics in life sciences 2020, 4, 399–410. [Google Scholar] [CrossRef]
- Handari, B.D.; Aldila, D.; Dewi, B.O.; Rosuliyana, H.; Khosnaw, S.H.A. Analysis of yellow fever prevention strategy from the perspective of mathematical model and cost-effectiveness analysis. Mathematical biosciences and engineering: MBE 2022, 19, 1786–1824. [Google Scholar] [CrossRef]
- Mere Roncal, C.; Bowler, M.; Gilmore, M.P. The ethnoprimatology of the Maijuna of the Peruvian Amazon and implications for primate conservation. Journal of ethnobiology and ethnomedicine 2018, 14, 19. [Google Scholar] [CrossRef]
- Reno, E.; Quan, N.G.; Franco-Paredes, C.; Chastain, D.B.; Chauhan, L.; Rodriguez-Morales, A.J.; Henao-Martínez, A.F. Prevention of yellow fever in travellers: an update. Lancet Infect Dis 2020, 20, e129–e137. [Google Scholar] [CrossRef] [PubMed]
- Peinado, R.D.S.; Eberle, R.J.; Pacca, C.C.; Arni, R.K.; Coronado, M.A. Review of -omics studies on mosquito-borne viruses of the Flavivirus genus. Virus research 2022, 307, 198610. [Google Scholar] [CrossRef]
- Rifakis, P.M.; Benitez, J.A.; De-la-Paz-Pineda, J.; Rodriguez-Morales, A.J. Epizootics of yellow fever in Venezuela (2004-2005): an emerging zoonotic disease. Ann N Y Acad Sci 2006, 1081, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Fiebre amarilla en América del Sur: casos y vectores, 2025. Actualizaciones en Sida e Infectología 2025, 33. [CrossRef]
- Forero-Delgadillo, A.J.; Morales-Olivera, J.A.; Celis-Guzman, J.F.; Zapata-Diaz, O.E.; Gonzalez-Varona, G.A.; Acevedo-Bedoya, C.A.; Salazar-Fernandez, R.; Ordonez, J.O.; Robayo-Amortegui, H.; Quintero-Altare, A.; et al. Colombian consensus on the care of critically ill patients with suspected or confirmed severe yellow fever. Lancet Reg Health Am 2025, 48, 101144. [Google Scholar] [CrossRef]
- Srivastava, S.; Jayaswal, N.; Gupta, P.; Sridhar, S.B.; Jaiswal, P.; Tariq, M.; Rao, G.S.N.K.; Mohanty, A.; Sah, S.; Mehta, R.; et al. The Yellow Fever Vaccine Journey: Milestones and Future Directions. Vaccines 2026, 14, 65. [Google Scholar] [CrossRef]
- Nederlof, R.A.; Virgilio, T.; Stemkens, H.J.J.; da Silva, L.; Montagna, D.R.; Abdussamad, A.M.; Chipangura, J.; Bakker, J. Yellow Fever in Non-Human Primates: A Veterinary Guide from a One Health Perspective. Veterinary sciences 2025, 12. [Google Scholar] [CrossRef]
- Shinde, D.P.; Plante, J.A.; Plante, K.S.; Weaver, S.C. Yellow Fever: Roles of Animal Models and Arthropod Vector Studies in Understanding Epidemic Emergence. Microorganisms 2022, 10. [Google Scholar] [CrossRef]
- Rezende, I.M.; Sacchetto, L.; Munhoz de Mello, É.; Alves, P.A.; Iani, F.C.M.; Adelino TÉ, R.; Duarte, M.M.; Cury, A.L.F.; Bernardes, A.F.L.; Santos, T.A.; et al. Persistence of Yellow fever virus outside the Amazon Basin, causing epidemics in Southeast Brazil, from 2016 to 2018. PLoS neglected tropical diseases 2018, 12, e0006538. [Google Scholar] [CrossRef]
- Ortiz-Martínez, Y.; Patiño-Barbosa, A.M.; Rodriguez-Morales, A.J. Yellow fever in the Americas: the growing concern about new epidemics. F1000Res 2017, 6, 398. [Google Scholar] [CrossRef]
- Caleiro, G.S.; Vilela, L.O.; Nuevo, K.M.B.; Tubaki, R.M.; de Menezes, R.M.T.; Mucci, L.F.; Telles-de-Deus, J.; Bergo, E.S.; Araújo, E.L.L.; Cunha, M.S. Yellow Fever Virus (YFV) Detection in Different Species of Culicids Collected During an Outbreak in Southeastern Brazil, 2016-2019. Tropical medicine and infectious disease 2025, 10. [Google Scholar] [CrossRef] [PubMed]
- Althouse, B.M.; Vasilakis, N.; Sall, A.A.; Diallo, M.; Weaver, S.C.; Hanley, K.A. Potential for Zika Virus to Establish a Sylvatic Transmission Cycle in the Americas. PLoS neglected tropical diseases 2016, 10, e0005055. [Google Scholar] [CrossRef]
- Nunes Neto, J.P.; Dias, D.D.; Nascimento, B.; da Silva, S.P.; da Silva, S.L.S.; Reis, L.A.M.; Reis, H.C.F.; da Silva, F.S.; da Silva, E.S.L.H.; Vieira, D.B.R.; et al. Molecular Detection of Yellow Fever Virus in Haemagogus janthinomys Mosquitoes (Diptera: Culicidae) in a Rural Settlement in the State of Pará, Brazilian Amazon, 2024. Viruses 2025, 17. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, S.R.M.; Cruz, E.; Prestes, N.G.O.; da Silva, F.S.; de Araújo, M.T.F.; Amador Neto, O.P.; Lima, M.L.G.; de Alcântara, B.N.; Dias, D.D.; Sousa, J.R.; et al. Histopathological Changes and Immune Response Profile in the Brains of Non-Human Primates Naturally Infected with Yellow Fever Virus. Viruses 2025, 17. [Google Scholar] [CrossRef] [PubMed]
- Paula, N.F.; Vieira, A.D.; Santos, D.O.D.; Souza, L.D.R.; Coelho, C.M.; Tinoco, H.P.; Lima, P.C.S.; Motta, R.O.C.; Pereira, V.D.S.; Carvalho, M.P.N.; et al. Safety and Immunogenicity of the Attenuated Yellow Fever Vaccine in Several Neotropical Primate Species. Vaccines 2025, 13. [Google Scholar] [CrossRef]
- Sanchez-Rojas, I.C.; Bonilla-Aldana, D.K.; Solarte-Jimenez, C.L.; Bonilla-Aldana, J.L.; Belisario-Tovar, M.; Ortega-Gómez, S.; Zambrano-Quenan, V.M.; Perafan-Gomez, J.C.; Gomez-Ocampo, C.H.; Delgado-Cajigas, M.; et al. Fatal yellow fever among captive non-human primates in southern Colombia, 2025. Frontiers in veterinary science 2025, 12, 1655474. [Google Scholar] [CrossRef]
- Fernandes, N.; Cunha, M.S.; Guerra, J.M.; Diaz-Delgado, J.; Ressio, R.A.; Cirqueira, C.S.; Kanamura, C.T.; Fuentes-Castillo, D.; Catão-Dias, J.L. Yellow Fever as Cause of Death of Titi Monkeys (Callicebus Spp.). Veterinary pathology 2021, 58, 730–735. [Google Scholar] [CrossRef] [PubMed]
- Bailey, A.L.; Kang, L.I.; de Assis Barros D'Elia Zanella, L.G.F.; Silveira, C.G.T.; Ho, Y.L.; Foquet, L.; Bial, G.; McCune, B.T.; Duarte-Neto, A.N.; Thomas, A.; et al. Consumptive coagulopathy of severe yellow fever occurs independently of hepatocellular tropism and massive hepatic injury. Proceedings of the National Academy of Sciences of the United States of America 2020, 117, 32648–32656. [Google Scholar] [CrossRef]
- Cunha, M.S.; da Costa, A.C.; de Azevedo Fernandes, N.C.C.; Guerra, J.M.; Dos Santos, F.C.P.; Nogueira, J.S.; D'Agostino, L.G.; Komninakis, S.V.; Witkin, S.S.; Ressio, R.A.; et al. Epizootics due to Yellow Fever Virus in São Paulo State, Brazil: viral dissemination to new areas (2016-2017). Sci Rep 2019, 9, 5474. [Google Scholar] [CrossRef]
- Selemane, I. Epidemiological monitoring of the last outbreak of yellow fever in Brazil - An outlook from Portugal. Travel medicine and infectious disease 2019, 28, 46–51. [Google Scholar] [CrossRef]
- de Azevedo Fernandes, N.C.C.; Guerra, J.M.; Díaz-Delgado, J.; Cunha, M.S.; Saad, L.D.; Iglezias, S.D.; Ressio, R.A.; Dos Santos Cirqueira, C.; Kanamura, C.T.; Jesus, I.P.; et al. Differential Yellow Fever Susceptibility in New World Nonhuman Primates, Comparison with Humans, and Implications for Surveillance. Emerging infectious diseases 2021, 27, 47–56. [Google Scholar] [CrossRef]
- Jorge, T.R.; Mosimann, A.L.P.; Noronha, L.; Maron, A.; Duarte Dos Santos, C.N. Isolation and characterization of a Brazilian strain of yellow fever virus from an epizootic outbreak in 2009. Acta tropica 2017, 166, 114–120. [Google Scholar] [CrossRef]
- Wilk-da-Silva, R.; Prist, P.R.; Medeiros-Sousa, A.R.; Laporta, G.Z.; Mucci, L.F.; Marrelli, M.T. The role of forest fragmentation in yellow fever virus dispersal. Acta tropica 2023, 245, 106983. [Google Scholar] [CrossRef]
- Guan, J.; Qi, Y.; Huang, Y.; Shao, R.; Zhou, D.; Xi, C.; Yin, X. Contrasting pathogenicity of an epizootic hemorrhagic disease virus serotype 7 isolate: high virulence in IFNAR((-/-)) mice versus a negligible role of sheep in EHDV-7 epidemiology. Archives of virology 2025, 170, 204. [Google Scholar] [CrossRef] [PubMed]
- Aliaga-Samanez, A.; Real, R.; Segura, M.; Marfil-Daza, C.; Olivero, J. Yellow fever surveillance suggests zoonotic and anthroponotic emergent potential. Communications biology 2022, 5, 530. [Google Scholar] [CrossRef]
- Cano, M.E.; Marti, G.A.; Balsalobre, A.; Muttis, E.; Bruno, E.A.; Rossi, G.; Micieli, M.V. Database of Sabethes and Haemagogus (Diptera: Culicidae) in Argentina: Sylvatic Vectors of the Yellow Fever Virus. Journal of medical entomology 2021, 58, 1762–1770. [Google Scholar] [CrossRef] [PubMed]
- Cunha, M.S.; Tubaki, R.M.; de Menezes, R.M.T.; Pereira, M.; Caleiro, G.S.; Coelho, E.; Saad, L.D.C.; Fernandes, N.; Guerra, J.M.; Nogueira, J.S.; et al. Possible non-sylvatic transmission of yellow fever between non-human primates in São Paulo city, Brazil, 2017-2018. Scientific reports 2020, 10, 15751. [Google Scholar] [CrossRef]
- Natani, T.; Culot, L.; da Cunha, R.G.T. Potential effects of brown howler monkey extinction on dispersal services in fragmented forests void of large dispersers. Primates; journal of primatology 2024, 65, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Delatorre, E.; de Abreu, F.V.S.; Ribeiro, I.P.; Gómez, M.M.; Dos Santos, A.A.C.; Ferreira-de-Brito, A.; Neves, M.; Bonelly, I.; de Miranda, R.M.; Furtado, N.D.; et al. Distinct YFV Lineages Co-circulated in the Central-Western and Southeastern Brazilian Regions From 2015 to 2018. Frontiers in microbiology 2019, 10, 1079. [Google Scholar] [CrossRef] [PubMed]
- Hill, S.C.; de Souza, R.; Thézé, J.; Claro, I.; Aguiar, R.S.; Abade, L.; Santos, F.C.P.; Cunha, M.S.; Nogueira, J.S.; Salles, F.C.S.; et al. Genomic Surveillance of Yellow Fever Virus Epizootic in São Paulo, Brazil, 2016 - 2018. PLoS pathogens 2020, 16, e1008699. [Google Scholar] [CrossRef]
- Abreu, F.V.S.; de Andreazzi, C.S.; Neves, M.; Meneguete, P.S.; Ribeiro, M.S.; Dias, C.M.G.; de Albuquerque Motta, M.; Barcellos, C.; Romão, A.R.; Magalhães, M.; et al. Ecological and environmental factors affecting transmission of sylvatic yellow fever in the 2017-2019 outbreak in the Atlantic Forest, Brazil. Parasites & vectors 2022, 15, 23. [Google Scholar] [CrossRef]
- Ribeiro, I.P.; Delatorre, E.; de Abreu, F.V.S.; Dos Santos, A.A.C.; Furtado, N.D.; Ferreira-de-Brito, A.; de Pina-Costa, A.; Neves, M.; de Castro, M.G.; Motta, M.A.; et al. Ecological, Genetic, and Phylogenetic Aspects of YFV 2017-2019 Spread in Rio de Janeiro State. Viruses 2023, 15. [Google Scholar] [CrossRef]
- Andrade, M.S.; Campos, F.S.; Oliveira, C.H.; Oliveira, R.S.; Campos, A.A.S.; Almeida, M.A.B.; Fonseca, V.S.; Simonini-Teixeira, D.; Sevá, A.D.P.; Temponi, A.O.D.; et al. Fast surveillance response reveals the introduction of a new yellow fever virus sub-lineage in 2021, in Minas Gerais, Brazil. Memorias do Instituto Oswaldo Cruz 2022, 117, e220127. [Google Scholar] [CrossRef]
- das Graças Dantas Andrade, V.; Ribeiro Adelino, T.; Fonseca, V.; Melo Farias Moreno, K.; Ribeiro Tomé, L.M.; Pereira, L.A.; Lima de La-Roque, D.G.; Bispo de Filippis, A.M.; Garkauskas Ramos, D.; Brock Ramalho, D.; et al. Reemergence of yellow fever virus in forest and periurban settings in Brazil. Communications medicine 2025, 6, 35. [Google Scholar] [CrossRef]
- Giovanetti, M.; de Mendonça, M.C.L.; Fonseca, V.; Mares-Guia, M.A.; Fabri, A.; Xavier, J.; de Jesus, J.G.; Gräf, T.; Dos Santos Rodrigues, C.D.; Dos Santos, C.C.; et al. Yellow Fever Virus Reemergence and Spread in Southeast Brazil, 2016-2019. Journal of virology 2019, 94. [Google Scholar] [CrossRef]
- Srivastava, S.; Dhoundiyal, S.; Kumar, S.; Kaur, A.; Khatib, M.N.; Gaidhane, S.; Zahiruddin, Q.S.; Mohanty, A.; Henao-Martinez, A.F.; Krsak, M.; et al. Yellow Fever: Global Impact, Epidemiology, Pathogenesis, and Integrated Prevention Approaches. Le infezioni in medicina 2024, 32, 434–450. [Google Scholar] [CrossRef]
- Almeida, M.A.B.; Santos, E.D.; Cardoso, J.D.C.; Noll, C.A.; Lima, M.M.; Silva, F.A.E.; Ferreira, M.S.; Martins, L.C.; Vasconcelos, P.; Bicca-Marques, J.C. Detection of antibodies against Icoaraci, Ilhéus, and Saint Louis Encephalitis arboviruses during yellow fever monitoring surveillance in non-human primates (Alouatta caraya) in southern Brazil. Journal of medical primatology 2019, 48, 211–217. [Google Scholar] [CrossRef]
- Larsen, R.S.; Moresco, A.; Karabatsos, N.; Dolz, G.; Glander, K.E. Serosurvey of Arboviruses in Free-Ranging Mantled Howler Monkeys (Alouatta palliata) in Costa Rica. Journal of wildlife diseases 2025, 61, 225–228. [Google Scholar] [CrossRef]
- Sousa, D.E.R.; Wilson, T.M.; Macêdo, I.L.; Romano, A.P.M.; Ramos, D.G.; Passos, P.H.O.; Costa, G.R.T.; Fonseca, V.S.; Mares-Guia, M.; Giovanetti, M.; et al. Case report: Urbanized non-human primates as sentinels for human zoonotic diseases: a case of acute fatal toxoplasmosis in a free-ranging marmoset in coinfection with yellow fever virus. Frontiers in public health 2023, 11, 1236384. [Google Scholar] [CrossRef] [PubMed]
- Dubankova, A.; Boura, E. Structure of the yellow fever NS5 protein reveals conserved drug targets shared among flaviviruses. Antiviral research 2019, 169, 104536. [Google Scholar] [CrossRef]
- Judson, S.D.; Schroeder, L.; Asiedu-Bekoe, F.; Laryea, D.O.; Boateng, G.; Gudjinu, H.; Ossom, R.; Danquah, J.F.; Dowdy, D.W.; Kenu, E. Timeliness of Yellow Fever Specimen Collection and Transport in Ghana, 2018-2022. medRxiv : the preprint server for health sciences 2025, 10.1101/2025.06.19.25329877, doi:10.1101/2025.06.19.25329877.
- Gaye, A.; Ndione, M.H.D.; Ndiaye, E.H.; Dieng, M.; Gaye, M.; Doukanda, S.F.M.; Sankhe, S.; Fall, G.; Weaver, S.C.; Diagne, M.M.; et al. Endemicity of a Yellow Fever Virus Strain Revealed by Entomological Surveillance in Southeastern Senegal. The American journal of tropical medicine and hygiene 2026, 114, 536–539. [Google Scholar] [CrossRef]
- Jiang, X.; Fan, Z.; Li, S.; Yin, H. A Review on Zoonotic Pathogens Associated with Non-Human Primates: Understanding the Potential Threats to Humans. Microorganisms 2023, 11. [Google Scholar] [CrossRef] [PubMed]
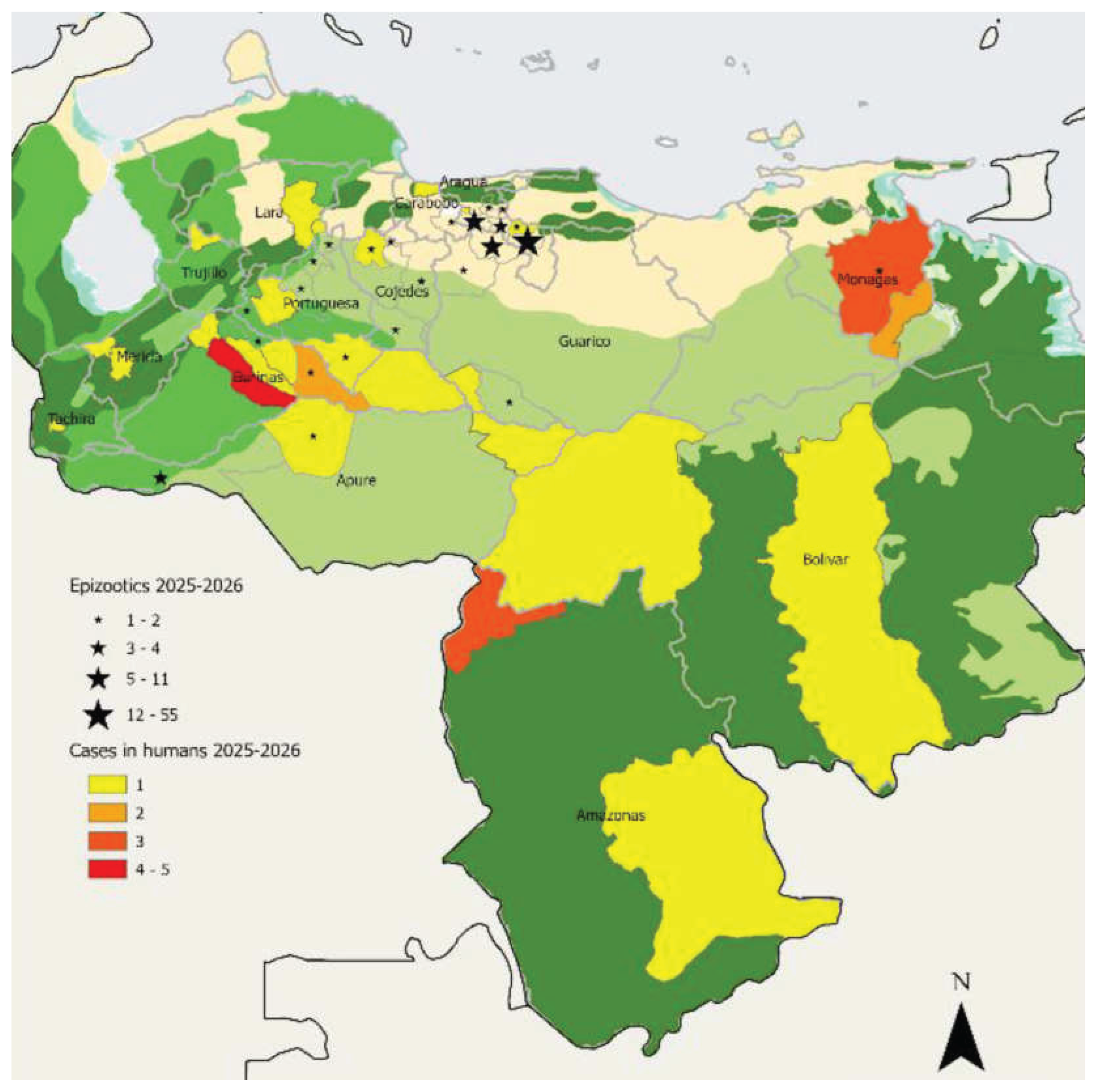
- Rodriguez-Morales, A.J.; Navarro, J.-C.; Forero-Peña, D.A.; Romero-Alvarez, D. Reemergence of yellow fever in Venezuela, 2025/2026. New Microbes and New Infections 2026, 70, 101737. [Google Scholar] [CrossRef]

| Genus of the non-human primate | Common Name | Distribution | Susceptibility to YF virus | Lethality in epizootics | Epidemiological role |
|---|---|---|---|---|---|
| Alouatta | Howler Monkey | Central America and much of South America | Very high | Very high (often >80%) | Key Sentinel and Amplifying Host |
| Sapajus | Robust Capuchin Jumpsuit | Brazil, Paraguay, Bolivia, Northern Argentina | Moderate | Moderate | Occasional amplifying host, sentinel |
| Cebus | Graceful Capuchin Monkey | Amazon and the tropical regions of South America | Moderate | Low to moderate | Secondary amplifier and sentinel |
| Saimiri | Squirrel Monkey | Amazonia (Brazil, Colombia, Peru, Bolivia, Venezuela) | Low to moderate | Generally low | Possible amp host with subclinical infections |
| Aotus | Night jumpsuit or night monkey | Amazon and the tropical regions of South America | Variable / poorly documented | Low | Possible Incidental Host |
| Callithrix | Tití or marmoset | Brazil (mainly Southeast and Central) | Moderate | Moderate | Sentinel in urban-wild shoots in Brazil |
| Feature | Primates of the New World | Primates of the Old World |
|---|---|---|
| Clinical Findings (General) | Marked lethargy, progressive weakness, noticeable decrease in activity, anorexia, dehydration, and rapid deterioration of general condition; In many epizootic episodes, animals are found dead in the forest with no obvious previous signs due to rapid clinical evolution. | They frequently present subclinical infection or mild symptoms; Occasionally, lethargy, mild fever, temporary reduction in activity, and slight anorexia are observed, although many individuals remain asymptomatic and continue with apparently normal behavior. |
| Manifestations of severe illness | High susceptibility in species such as Alouatta and some Atelidae, mainly; the disease can progress rapidly, with elevated viremia, visible jaundice, severe prostration, extreme weakness, and, in some cases, neurological signs such as tremors or disorientation. | Severe disease is rare under natural conditions; however, some species may develop systemic disease in experimental infections, with fever, moderate liver involvement, and transient clinical signs. |
| Complications | Extensive hepatocellular necrosis, acute hepatic failure, internal bleeding, metabolic abnormalities, and multiorgan dysfunction; These complications are associated with high mortality rates during epizootics in highly susceptible primate populations. | Clinical complications are rare in natural populations; when they do occur, they can include moderate liver involvement, systemic inflammation, or transient febrile illness, usually with lower mortality and a higher chance of recovery. |
| AL1 | AL2 | N | AL1 | AL2 | N | AL1 | AL2 | N |
|---|---|---|---|---|---|---|---|---|
| Goias | Abadia De Goias | 2 | Sao Paulo | Colina | 1 | Putumayo | Mocoa | 6 |
| Firminopolis | 1 | Cravinhos | 1 | Orito | 2 | |||
| Goiania | 2 | Descalvado | 1 | Total | 8 | |||
| Guapo | 1 | Guarulhos | 1 | Tolima | Ataco | 10 | ||
| Total | 6 | Itatiba | 1 | Chaparral | 19 | |||
| Minas Gerais | Albertina | 1 | Joanopolis | 3 | Cunday | 9 | ||
| Baependi | 1 | Louveira | 1 | Planadas | 6 | |||
| Belo Horizonte | 1 | Luis Antonio | 6 | Prado | 3 | |||
| Bueno Brandao | 2 | Mairipora | 2 | Purificacion | 1 | |||
| Camanducaia | 1 | Osasco | 1 | Rioblanco | 4 | |||
| Corrego do Bom Jesus | 1 | Pedra Bela | 3 | San Antonio | 5 | |||
| Delfim Moreira | 1 | Pinhalzinho | 2 | Villarrica | 3 | |||
| Estiva | 2 | Pitangueiras | 2 | Total | 60 | |||
| Extrema | 1 | Ribeirao Preto | 39 | Total Colombia | 77 | |||
| Ipuiuna | 4 | Salto | 1 | La Paz | Abel Iturralde | 1 | ||
| Paraisopolis | 1 | Santa Rita do Passa Quatro | 1 | Nor Yungas | 1 | |||
| Poco Fundo | 1 | Santo Antonio Do Pinhal | 1 | Total | 2 | |||
| Pocos De Caldas | 1 | Serra Azul | 1 | Total Bolivia | 2 | |||
| Ponte Nova | 1 | Serra Negra | 2 | Apure | Ujnknown | 5 | ||
| Santa Rita De Caldas | 1 | Socorro | 1 | Aragua | Ujnknown | 74 | ||
| Sapucai-Mirim | 2 | Valinhos | 7 | Barinas | Ujnknown | 2 | ||
| Toledo | 1 | Total | 87 | Carabobo | Ujnknown | 2 | ||
| Virginia | 2 | Tocantins | Palmas | 4 | Cojedes | Ujnknown | 7 | |
| Total | 25 | Total | 4 | Guarico | Ujnknown | 11 | ||
| Roraima | Alto Alegre | 2 | Total Brazil | 124 | Lara | Ujnknown | 1 | |
| Total | 2 | Huila | Aipe | 2 | Monagas | Ujnknown | 1 | |
| Sao Paulo | Amparo | 2 | Neiva | 2 | Portuguesa | Ujnknown | 6 | |
| Atibaia | 1 | Palermo | 4 | Total Venezuela | 109 | |||
| Braganca Paulista | 2 | Total | 8 | |||||
| Cacapava | 1 | Meta | Villavicencio | 1 | Total South America | 312 | ||
| Campinas | 3 | Total | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




