Submitted:
16 March 2026
Posted:
17 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Data Collection and Variables
2.3. Outcomes and Definitions
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics and Comorbidities
3.2. Laboratory Parameters
3.3. Oncological Characteristics
3.4. Intraoperative Factors
3.5. Postoperative Course
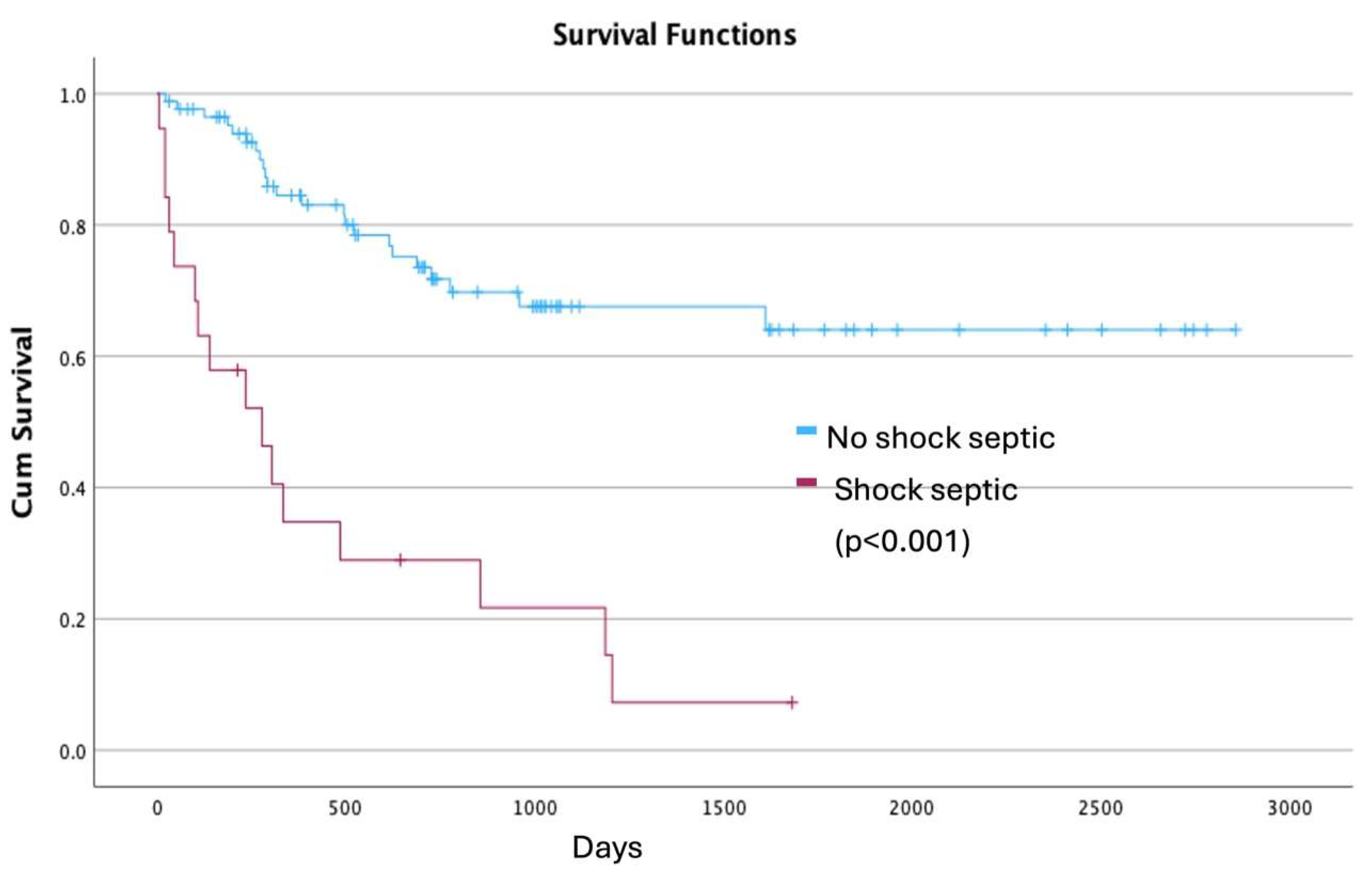
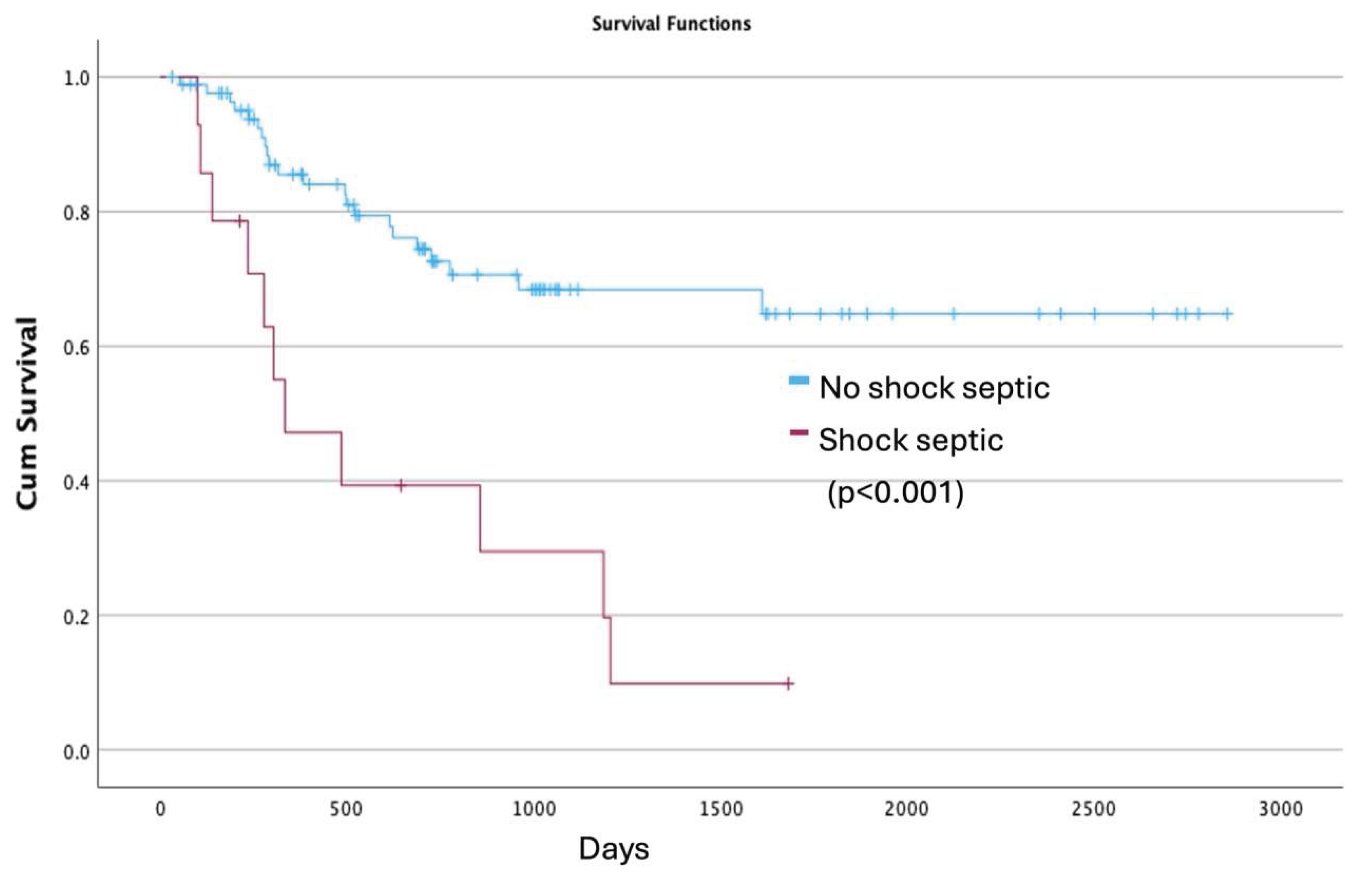
3.6. Multivariate Analysis and Survival
4. Discussion
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bona D, Manara M, Bonitta G, Guerrazzi G, Guraj J, Lombardo F, et al. Long-Term Impact of Severe Postoperative Complications after Esophagectomy for Cancer: Individual Patient Data Meta-Analysis. Cancers (Basel). 2024;16:1468. [CrossRef]
- Liu CQ, Ma YL, Qin Q, Wang PH, Luo Y, Xu PF, et al. Epidemiology of esophageal cancer in 2020 and projections to 2030 and 2040. Thorac Cancer. 2022; 14:3.
- Low DE, Alderson D, Cecconello I, Chang AC, Darling GE, D’Journo XB, et al. International consensus on standardization of data collection for complications associated with esophagectomy: Esophagectomy Complications Consensus Group (ECCG). Ann Surg. 2015 30; 262:286–94.
- Manara M, Bona D, Bonavina L, Aiolfi A. Impact of pulmonary complications following esophagectomy on long-term survival: multivariate meta-analysis and restricted mean survival time assessment. Updates Surg. 2024; 76:757–67. [CrossRef]
- Yamashita K, Makino T, Miyata H, Miyazaki Y, Takahashi T, Kurokawa Y, et al. Postoperative Infectious Complications are Associated with Adverse Oncologic Outcomes in Esophageal Cancer Patients Undergoing Preoperative Chemotherapy. Ann Surg Oncol. 2016; 23:2106–14. [CrossRef]
- Aiolfi A, Griffiths EA, Sozzi A, Manara M, Bonitta G, Bonavina L, et al. Effect of Anastomotic Leak on Long-Term Survival After Esophagectomy: Multivariate Meta-analysis and Restricted Mean Survival Times Examination. Ann Surg Oncol. 2023; 30:5564–72.
- Booka E, Takeuchi H, Nishi T, Matsuda S, Kaburagi T, Fukuda K, et al. The Impact of Postoperative Complications on Survivals After Esophagectomy for Esophageal Cancer. Medicine. 2015; 94(33):e1369. [CrossRef]
- Nuytens F, Dabakuyo-Yonli TS, Meunier B, Gagnière J, Collet D, D’Journo XB, et al. Five-Year Survival Outcomes of Hybrid Minimally Invasive Esophagectomy in Esophageal Cancer: Results of the MIRO Randomized Clinical Trial. JAMA Surg. 2021; 156:323–32.
- Fumagalli Romario U, de Pascale S. Esophagectomy-prevention of complications-tips and tricks for the preoperative, intraoperative and postoperative stage. 2023; 75:343–55.
- Kataoka K, Takeuchi H, Mizusawa J, Igaki H, Ozawa S, Abe T, et al. Prognostic impact of postoperative morbidity after esophagectomy for esophageal cancer: Exploratory analysis of JCOG9907. Ann Surg. 2017; 265:1152–7.
- Yamashita K, Makino T, Miyata H, Miyazaki Y, Takahashi T, Kurokawa Y, et al. Postoperative Infectious Complications are Associated with Adverse Oncologic Outcomes in Esophageal Cancer Patients Undergoing Preoperative Chemotherapy. Ann Surg Oncol. 2016; 23:2106–14. [CrossRef]
- Rahmel TI, Schmitz S, Nowak H, Schepanek K, BergmannID L, Halberstadt P, et al. Long-term mortality and outcome in hospital survivors of septic shock, sepsis, and severe infections: The importance of aftercare. PLoS One. 2020;15(2):e0228952. [CrossRef]
- Shankar-Hari M, Phillips GS, Levy ML, Seymour CW, Liu VX, Deutschman CS, et al. Developing a New Definition and Assessing New Clinical Criteria for Septic Shock: For the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016; 315:775–87.
- Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, et al. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017; 67:93–9.
- Singer M, Deutschman CS, Seymour C, Shankar-Hari M, Annane D, Bauer M, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016; 315:801–10. [CrossRef]
- Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021; 47:1181–247. [CrossRef]
- Booka E, Takeuchi H, Suda K, Fukuda K, Nakamura R, Wada N, et al. Meta-analysis of the impact of postoperative complications on survival after oesophagectomy for cancer. BJS Open. 2018; 2:276-84. [CrossRef]
- Blencowe NS, Strong S, McNair AGK, Brookes ST, Crosby T, Griffin SM, et al. Reporting of short-term clinical outcomes after esophagectomy: A systematic review. Ann Surg. 2012; 255:658-66.
- Markar S, Gronnier C, Duhamel A, Mabrut JY, Bail JP, Carrere N, et al. The Impact of Severe Anastomotic Leak on Long-term Survival and Cancer Recurrence After Surgical Resection for Esophageal Malignancy. Ann Surg. 2015; 262:972–80. [CrossRef]
- Rutegård M, Lagergren P, Rouvelas I, Lagergren J. Intrathoracic anastomotic leakage and mortality after esophageal cancer resection: A population-based study. Ann Surg Oncol. 2012; 19:99–103. [CrossRef]
- Ward ST, Dimick JB, Zhang W, Campbell DA, Ghaferi AA. Association between Hospital Staffing Models and Failure to Rescue. Ann Surg. 2019; 270:91–4. [CrossRef]
- Ponten JEH, van der Horst S, Nieuwenhuijzen GAP, Elenbaas TWO, van Hillegersberg R, Luyer MDP. Early severe mediastinal bleeding after esophagectomy: a potentially lethal complication. J Thorac Dis 2013; 5(2):E58.
- Rieff EA, Hendriks T, Rutten HJT, Nieuwenhuijzen GAP, Gosens MJEM, Van Den Brule AJC, et al. Neoadjuvant Radiochemotherapy Increases Matrix Metalloproteinase Activity in Healthy Tissue in Esophageal Cancer Patients. Ann Surg Oncol. 2009 May;16(5):1384-9. [CrossRef]
- Steenhagen E. Preoperative nutritional optimization of esophageal cancer patients. J Thorac Dis. 2019; 11(Suppl 5):S645-S653. [CrossRef]
- Godi I, Feltracco P, Lorenzoni G, Antonelli A, Salvador R, Gregori D, et al. Incidence, Risk Factors, and Consequences of Acute Kidney Injury in Patients Undergoing Esophageal Cancer Surgery: A Historical Cohort. Kidney and Dialysis. 2024; 4:93–104. [CrossRef]
- Bihorac A, Yavas S, Subbiah S, Hobson CE, Schold JD, Gabrielli A, et al. Long-term risk of mortality and acute kidney injury during hospitalization after major surgery. Ann Surg. 2009; 249:851–8. [CrossRef]
- Aoyama T, Atsumi Y, Hara K, Kazama K, Tamagawa H, Tamagawa A, et al. Postoperative bleeding after esophagectomy for esophageal cancer in patients receiving antiplatelet and anticoagulation treatment. Anticancer Res. 2020; 40:2359–64. [CrossRef]
- Nguyen DD, Pham B Van, Tran MD, Nguyen TD, Thai AD, Le K Van, et al. Characteristics of lymph node metastasis and short-term outcome of esophageal squamous-cell carcinoma undergoing minimally invasive esophagectomy: a prospective cross-sectional study (with video). Annals of Medicine & Surgery. 2024; 86:5739–43.
- Gu YM, Yang YS, Hu WP, Wang WP, Yuan Y, Chen LQ. Prognostic value of lymphovascular invasion in patients with esophageal squamous cell carcinoma. Ann Transl Med. 2019; 7:256–256. [CrossRef]
- Prescott HC, Angus DC. Enhancing Recovery From Sepsis: A Review. JAMA. 2018; 319:62–75.
- Iwashyna TJ, Ely EW, Smith DM, Langa KM. Long-term Cognitive Impairment and Functional Disability Among Survivors of Severe Sepsis. JAMA. 2010; 304:1787–94. [CrossRef]
- Tverskov V, Wiesel O, Solomon D, Orgad R, Kashtan H. The impact of cervical anastomotic leak after esophagectomy on long-term survival of patients with esophageal cancer. Surgery. 2022; 171:1257–62. [CrossRef]
- Martin LW, Swisher SG, Hofstetter W, Correa AM, Mehran RJ, Rice DC, et al. Intrathoracic leaks following esophagectomy are no longer associated with increased mortality. Ann Surg. 2005;242:392-9. [CrossRef]
| Variable | Total (n=106) | No Septic Shock (n=87) | Septic Shock (n=19) | p value |
|---|---|---|---|---|
| Age, years | 59.5 (51.7–68.0) | 60 (52–68) | 58.0 (52–68) | 0.789 |
| Weight, kg | 72.4 (61.6–83.5) | 74 (63–85) | 65 (58.7–73.0) | 0.046 |
| Height, cm | 170 (166–175) | 170 (167–175) | 168 (160–170) | 0.096 |
| BMI , kg/m2 | 24.9 (22.3–28.5) | 25.5 (22.5–28.2) | 23.4 (20.4–26.2) | 0.036 |
| Male sex, n (%) | 92 (86.8) | 77 (88.5) | 15 (78.9) | 0.789 |
| Active alcohol use, n (%) | 15 (14.1) | 11 (12.8) | 4 (21.1) | 0.272 |
| Former alcohol use, n (%) | 11 (10.3) | 10 (11.6) | 1 (5.3) | 0.368 |
| Active smoker, n (%) | 33 (31.1) | 26 (30.2) | 7 (36.8) | 0.574 |
| Former smoker, n (%) | 37 (34.9) | 31 (36.0) | 6 (31.6) | 0.712 |
| Preoperative corticosteroid therapy, n (%) | 8 (7.5) | 7 (8.1) | 1 (5.3) | 0.556 |
| ASA physical status, n (%) | 0.078 | |||
| • ASA I | 1 (0.9) | 1 (1.2) | 0 (0) | |
| • ASA II | 40 (37.7) | 28 (32.6) | 12 (63.2) | |
| • ASA III | 61 (57.5) | 54 (62.8) | 6 (31.6) | |
| • ASA IV | 4 (3.7) | 3 (3.5) | 1 (5.3) | |
| COPD, n (%) | 4 (3.7) | 3 (3.5) | 1 (5.3) | 0.556 |
| OSA, n (%) | 2 (1.8) | 2 (2.3) | 0 (0) | 0.502 |
| DMNID, n (%) | 13 (12.2) | 13 (15.1) | 0 (0) | 0.062 |
| DMID, n (%) | 3 (2.8) | 2 (2.3) | 1 (5.3) | 0.454 |
| CKD, n (%) | 6 (5.6) | 5 (5.8) | 1 (5.3) | 0.703 |
| Hypertension, n (%) | 44 (41.5) | 37 (43.0) | 7 (36.8) | 0.621 |
| Dyslipidemia, n (%) | 41 (38.6) | 32 (37.2) | 9 (47.4) | 0.411 |
| Ischemic heart disease, n (%) | 6 (5.6) | 6 (7.0) | 0 (0) | 0.292 |
| CHF, n (%) | 1 (0.9) | 1 (1.2) | 0 (0) | 0.819 |
| Atrial fibrillation, n (%) | 5 (4.7) | 5 (5.8) | 0 (0) | 0.361 |
| Hypothyroidism, n (%) | 1 (0.9) | 1 (1.2) | 0 (0) | 0.819 |
| Cerebrovascular disease, n (%) | 3 (2.8) | 3 (3.5) | 0 (0) | 0.546 |
| Chronic liver disease, n (%) | 4 (3.7) | 3 (3.5) | 1 (5.3) | 0.556 |
| PVD, n (%) | 8 (7.5) | 7 (8.1) | 1 (5.3) | 0.556 |
| Variable | Total (n=106) | No Septic Shock (n=87) | Septic Shock (n=19) | p value |
|---|---|---|---|---|
| Preoperative | ||||
| Hemoglobin, g/dL | 12.6 (11.8-13.7) | 12.65 (11.8-13.6) | 12.6 (11.0-14.1) | 0.745 |
| Platelets, ×109/L | 210 (149-254) | 211.5 (160.0-254.0) | 209.0 (140.0-254.0) | 0.705 |
| INR | 1.02 (0.97-1.09) | 1.02 (0.97-1.10) | 1.00 (0.94-1.07) | 0.617 |
| Fibrinogen, mg/dL | 496 (437-583) | 495 (438-578) | 505 (432-760) | 0.509 |
| Lymphocytes, ×109/L | 1.2 (0.9-1.8) | 1.3 (0.9-1.8) | 1.0 (0.7-1.4) | 0.155 |
| Leukocytes, ×109/L | 6.20 (4.66-7.90) | 5.95 (4.66-7.90) | 6.98 (4.20-10.10) | 0.446 |
| Glucose, mg/dL | 97.0 (89.0-113.5) | 97 (89-113) | 99 (89-121) | 0.863 |
| Creatinine, mg/dL | 0.77 (0.65-0.89) | 0.79 (0.65-0.89) | 0.73 (0.62-0.92) | 0.714 |
| Albumin, g/dL | 4.1 (3.7-4.3) | 4.10 (3.79-4.30) | 3.85 (3.50-4.10) | 0.039 |
| Total proteins, g/dL | 6.6 (6.3-7.0) | 6.60 (6.30-7.00) | 6.70 (6.20-7.20) | 0.551 |
| C-reactive protein, mg/L | 1.50 (0.20-6.70) | 0.80 (0.20-6.20) | 10.75 (0.85-26.85) | 0.496 |
| Procalcitonin, ng/mL | 0.07 (0.03-0.11) | 0.05 (0.03-0.10) | 0.11 (0.07-0.12) | 0.179 |
| Neutrophil-to-lymphocyte ratio | 4.87 (3.50–8.00) | 4.66 (3.31–6.18) | 6.80 (4.05–10.10) | 0.049 |
| Platelet-to-lymphocyte ratio | 166.67 (104.21–262.86) | 158.89 (103.53–236.00) | 237.78 (110.00–310.00) | 0.227 |
| Albumin-to-lymphocyte ratio | 3.28 (2.19–4.50) | 3.19 (2.15–4.40) | 3.60 (2.25–5.11) | 0.398 |
| PICU admission | ||||
| Hemoglobin, g/dL | 11.6 (10.5–12.6) | 11.55 (10.60–12.40) | 11.80 (9.60–13.10) | 0.780 |
| Platelets, ×109/L | 182 (147–224) | 185 (152–223) | 166 (119–248) | 0.632 |
| INR | 1.11 (1.03–1.21) | 1.11 (1.03–1.21) | 1.10 (0.97–1.23) | 0.492 |
| Fibrinogen, mg/dL | 497 (431–609) | 511 (446–597) | 438 (357–693) | 0.326 |
| Lymphocytes, ×109/L | 0.6 (0.4–1.0) | 0.7 (0.4–1.1) | 0.5 (0.3–0.8) | 0.094 |
| Leukocytes, ×109/L | 10.20 (7.95–12.50) | 10.10 (7.90–12.70) | 10.59 (8.60–11.97) | 0.822 |
| Glucose, mg/dL | 144 (119–165) | 144 (115–166) | 147 (124–165) | 0.334 |
| Creatinine, mg/dL | 0.76 (0.58–0.91) | 0.75 (0.57–0.92) | 0.80 (0.66–0.87) | 0.206 |
| Albumin, g/dL | 3.20 (3.00–3.50) | 3.27 (3.06–3.50) | 3.10 (2.30–3.40) | 0.024 |
| Total proteins, g/dL | 5.50 (5.00–5.70) | 5.50 (5.10–5.70) | 5.40 (4.70–6.00) | 0.914 |
| C-reactive protein, mg/L | 21.7 (4.4–60.7) | 21.7 (5.7–60.1) | 57.6 (0.6–114.6) | 0.956 |
| Procalcitonin, ng/mL | 0.10 (0.04–0.17) | 0.07 (0.03–0.14) | 0.19 (0.15–2.17) | 0.044 |
| Lactate, mmol/L | 2.05 (1.30–3.60) | 1.90 (1.30–3.50) | 2.80 (1.80–3.70) | 0.306 |
| Neutrophil-to-lymphocyte ratio | 15.33 (9.82–25.75) | 14.75 (9.34–24.50) | 17.50 (13.82–27.75) | 0.064 |
| Albumin-to-lactate ratio | 1.58 (0.90–2.42) | 1.68 (0.95–2.47) | 1.03 (0.76–1.84) | 0.064 |
| Platelet-to-lymphocyte ratio | 274.29 (175.45–475.56) | 252.43 (173.64–446.00) | 380.00 (181.18–595.00) | 0.145 |
| Albumin-to-lymphocyte ratio | 5.33 (2.95–7.40) | 4.93 (2.91–7.20) | 5.75 (3.00–10.50) | 0.446 |
| Change preoperative to admission PICU | ||||
| Albumin-to-lymphocyte ratio (preop/PICU) | 0.80 (0.50–1.10) | 0.80 (0.49–1.10) | 0.70 (0.50–1.44) | 0.729 |
| Creatinine change, mg/dL | 0.04 (−0.06–0.13) | 0.05 (−0.04–0.14) | 0.06 (−0.19–0.13) | 0.041 |
| Variable | Total (n=106) | No Septic Shock (n=87) | Septic Shock (n=19) | p value |
|---|---|---|---|---|
| Chemotherapy Neoadjuvant n(%) | 68 (64.1) | 55 (83.3) | 13 (81.3) | 0.547 |
| Radiotherapy Neoadjuvant n(%) | 40 (37.7) | 30 (34.9) | 10 (52.6) | 0.149 |
| Tumor stage (T), n (%) | 0.791 | |||
| • T1 | 9 (8.5) | 8 (9.2) | 1 (5.3) | |
| • T2 | 35 (33) | 27 (31) | 8 (42.1) | |
| • T3 | 48 (45.3) | 40 (46) | 8 (42.1) | |
| • T4 | 14 (13.2) | 12 (13.8) | 2 (10.5) | |
| Nodal stage (N), n (%) | 0.009 | |||
| • N0 | 14 (13.2) | 13 (14.9) | 1 (5.3) | |
| • N1 | 31 (29.2) | 29 (33.3) | 2 (10.5) | |
| • N2 | 42 (39.6) | 34 (39.1) | 8 (42.1) | |
| • N3 | 19 (17.9) | 11 (12.6) | 8 (42.1) | |
| Metastatic stage (M), n (%) | 0.367 | |||
| • M0 | 94 (88.7) | 78 (89.7) | 16 (84.2) | |
| • M1 | 12 (11.3) | 9 (10.3) | 3 (15.8) | |
| Tumor location within esophagus, n (%) | 0.763 | |||
| • Distal | 79 (74.5) | 65 (74.7) | 14 (73.7) | |
| • Middle–distal | 14 (13.2) | 12 (13.8) | 2 (10.5) | |
| • Middle | 7 (6.6) | 6 (6.9) | 1 (5.3) | |
| • Proximal | 6 (5.7) | 4 (4.6) | 2 (10.5) | |
| Variable | Total (n=106) | No Septic Shock (n=87) | Septic Shock (n=19) | p value |
|---|---|---|---|---|
| Surgical approach, n (%) | 0.594 | |||
| • Open | 17 (16.0) | 12 (14.0) | 5 (26.3) | |
| • Hybrid | 44 (41.5) | 35 (40.7) | 9 (47.4) | |
| •Minimally invasive | 45 (42.5) | 40 (46.5) | 5 (26.3) | |
| Feeding jejunostomy, n (%) | 86 (81.1) | 70 (81.4) | 16 (88.9) | 0.354 |
| Intraoperative radiotherapy, n (%) | 13 (12.3) | 10 (11.9) | 3 (15.8) | 0.430 |
| Type of esophagectomy, n (%) | 0.37 | |||
| • Ivor Lewis | 60 (56.6) | 51(58.6) | 9 (47.4) | |
| • McKeown | 46 (43.4) | 36 (41.4) | 10 (52.6) | |
| Duration of surgery, min | 450 (370–525) | 454 (384–528.5) | 435 (320–470) | 0.192 |
| Duration of anesthesia, min | 579.5 (509–663) | 594 (510–667) | 567 (450–663) | 0.317 |
| Use One-lung ventilation | 81 (76.4) | 69 (79.3) | 12 (63.2) | 0.133 |
| Bronchial blocker, n (%) | 59 (55.7) | 48 (55.2) | 11 (57.9) | 0.956 |
| Epidural analgesia n(%) | 74 (69.8) | 61 (70.1) | 13 (68.4) | 0.828 |
| Use of vasoactive drugs, n (%) | 35 (33.0) | 26 (29.9) | 9 (47.4) | 0.142 |
| Extubated in the operating room, n (%) | 89 (84.0) | 74 (85.1) | 15 (78.9) | 0.36 |
| Red blood cell transfusion, n (%) | 2 (1.9) | 1 (1.2) | 1 (5.3) | 0.342 |
| Crystalloids administered, mL | 2200 (1500–2500) | 2100 (1500–2500) | 2500 (2000–2500) | 0.858 |
| Colloids administered, mL | 250 (0–500) | 150 (0–500) | 500 (500–500) | 0.1 |
| Variable | Total (n=106) | No Septic Shock (n=87) | Septic Shock (n=19) | p value |
|---|---|---|---|---|
| Atelectasis n(%) | 12 (11.3) | 9 (10.3) | 3 (15.8) | 0.367 |
| Pleural effusion n(%) | 38 (35.8) | 23 (26.4) | 15 (78.9) | <0.001 |
| Hospital-acquired pneumonia n(%) | 35 (33.0) | 26 (29.9) | 9 (47.4) | 0.142 |
| Ventilator-associated pneumonia n(%) | 8 (7.5) | 3 (3.4) | 5 (26.3) | 0.004 |
| Empyema n(%) | 8 (7.5) | 5 (5.7) | 3 (15.8) | 0.152 |
| Pulmonary embolism n(%) | 2 (1.9) | 2 (2.3) | 0 (0) | 0.672 |
| Acute respiratory distress syndrome n(%) | 10 (9.4) | 4 (4.6) | 6 (31.6) | <0.001 |
| Acute myocardial infarction n(%) | 1 (0.9) | 1 (1.1) | 0 (0) | 0.821 |
| New-onset atrial fibrillation n(%) | 14 (13.2) | 6 (6.9) | 8 (42.1) | <0.001 |
| Cardiac arrest n(%) | 2 (1.9) | 0 (0) | 2 (10.5) | 0.031 |
| Ileus n(%) | 11 (10.4) | 7 (8.0) | 4 (21.1) | 0.107 |
| Biliary leak n(%) | 2 (1.9) | 1 (1.1) | 1 (5.3) | 0.328 |
| Acute kidney failure n(%) | 17 (16.0) | 9 (10.3) | 8 (42.1) | <0.001 |
| Bacteremia n(%) | 8 (7.5) | 5 (5.7) | 3 (15.8) | 0.152 |
| Sepsis (non-shock) n(%) | 33 (31.1) | 26 (29.9) | 7 (36.8) | 0.553 |
| Stroke n(%) | 1 (0.9) | 1 (1.1) | 0 (0) | 0.821 |
| Delirium n(%) | 15 (14.2) | 9 (10.3) | 6 (31.6) | 0.016 |
| Anastomotic leak/dehiscence n(%) | 40 (37.7) | 25 (28.7) | 15 (78.9) | <0.001 |
| Bronchoesophageal fistula n(%) | 7 (6.6) | 5 (5.7) | 2 (10.5) | 0.367 |
| Chylothorax n(%) | 3 (2.8) | 1 (1.1) | 2 (10.5) | 0.082 |
| Pneumothorax n(%) | 8 (7.5) | 6 (6.9) | 2 (10.5) | 0.438 |
| Surgical site infection n(%) | 9 (8.5) | 7 (8.0) | 2 (10.5) | 0.506 |
| Hemorrhagic shock n(%) | 6 (5.7) | 2 (2.3) | 4 (21.1) | 0.004 |
| PICU stay (days) | 5 (2-10.5) | 4 (2-7.3) | 8 (4-26) | 0.008 |
| Hospital length stay (days) | 21 (13-36) | 19 (13-31.5) | 37 (21-72) | 0.011 |
| PICU readmission n(%) | 25 (23.6) | 17 (19.5) | 8 (42.1) | 0.036 |
| Mortality intrahospital n(%) | 7 (6.6) | 1 (1.1) | 6 (31.6) | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




