Submitted:
16 March 2026
Posted:
17 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Population and Ethics
2.2. Patient Characteristics and Classification
2.3. Data Collection
2.4. Outcome Measures
2.5. Statistical Analysis
3. Results
3.1. Study Population Characteristics
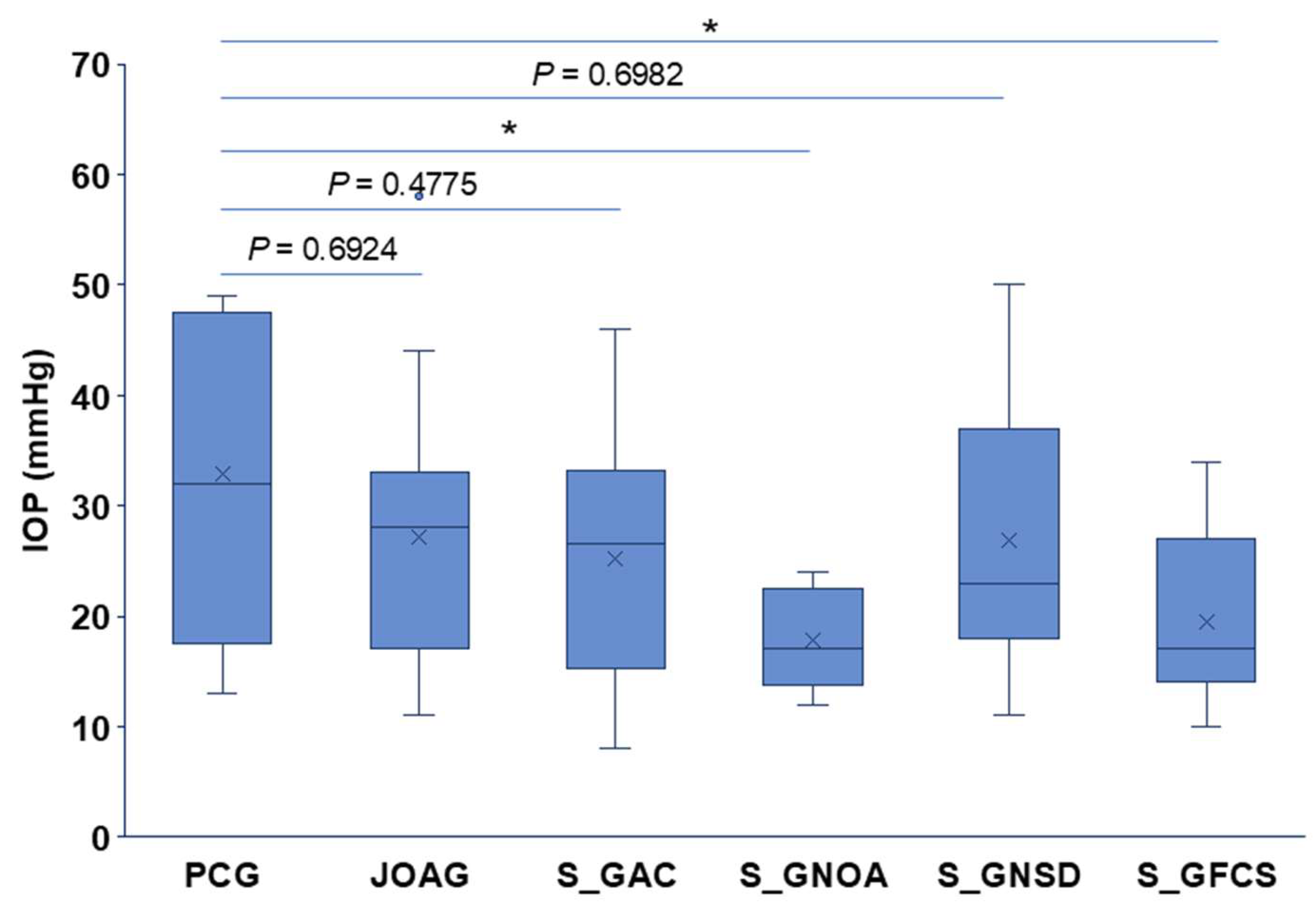
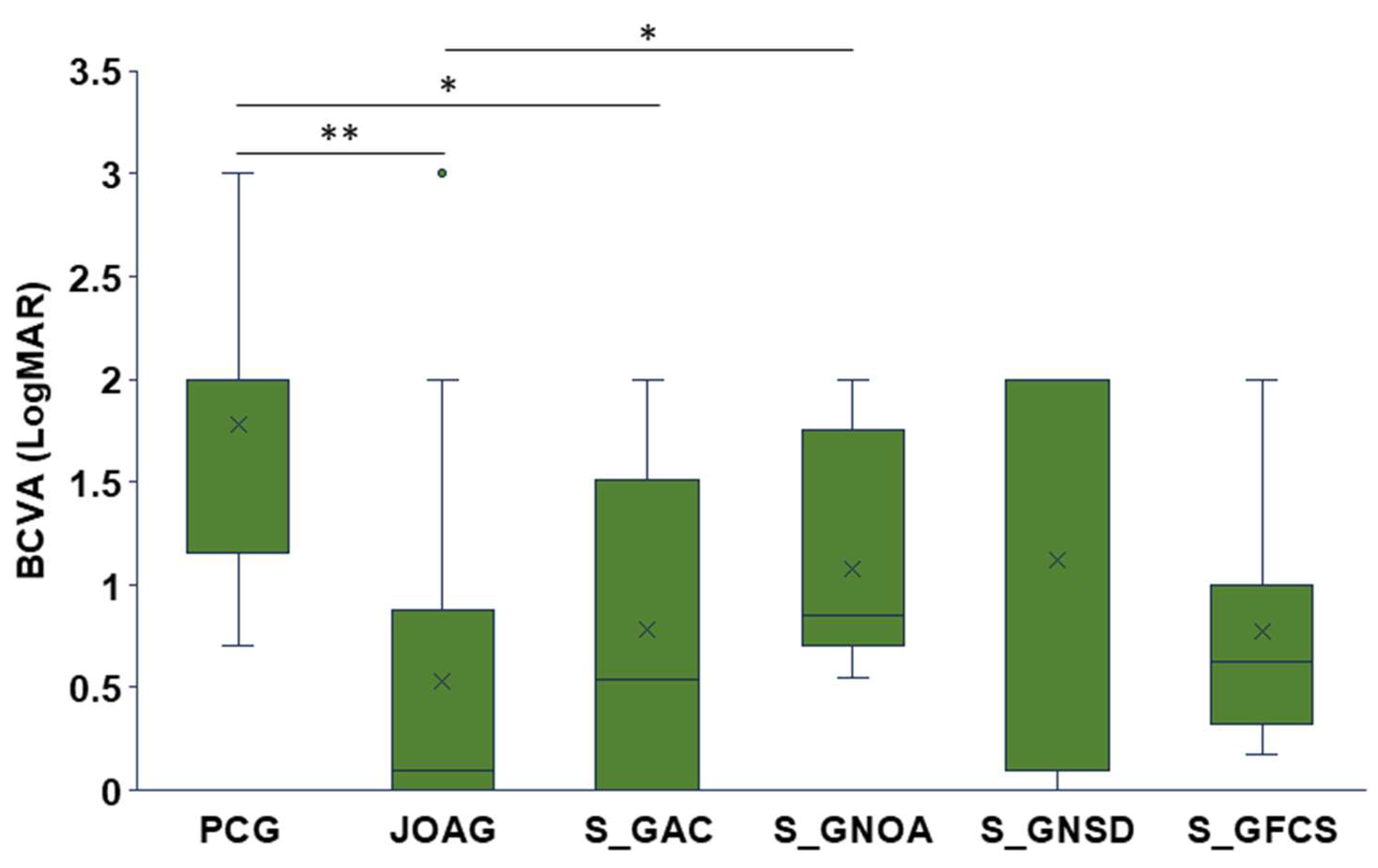
3.2. Baseline Ocular Characteristics
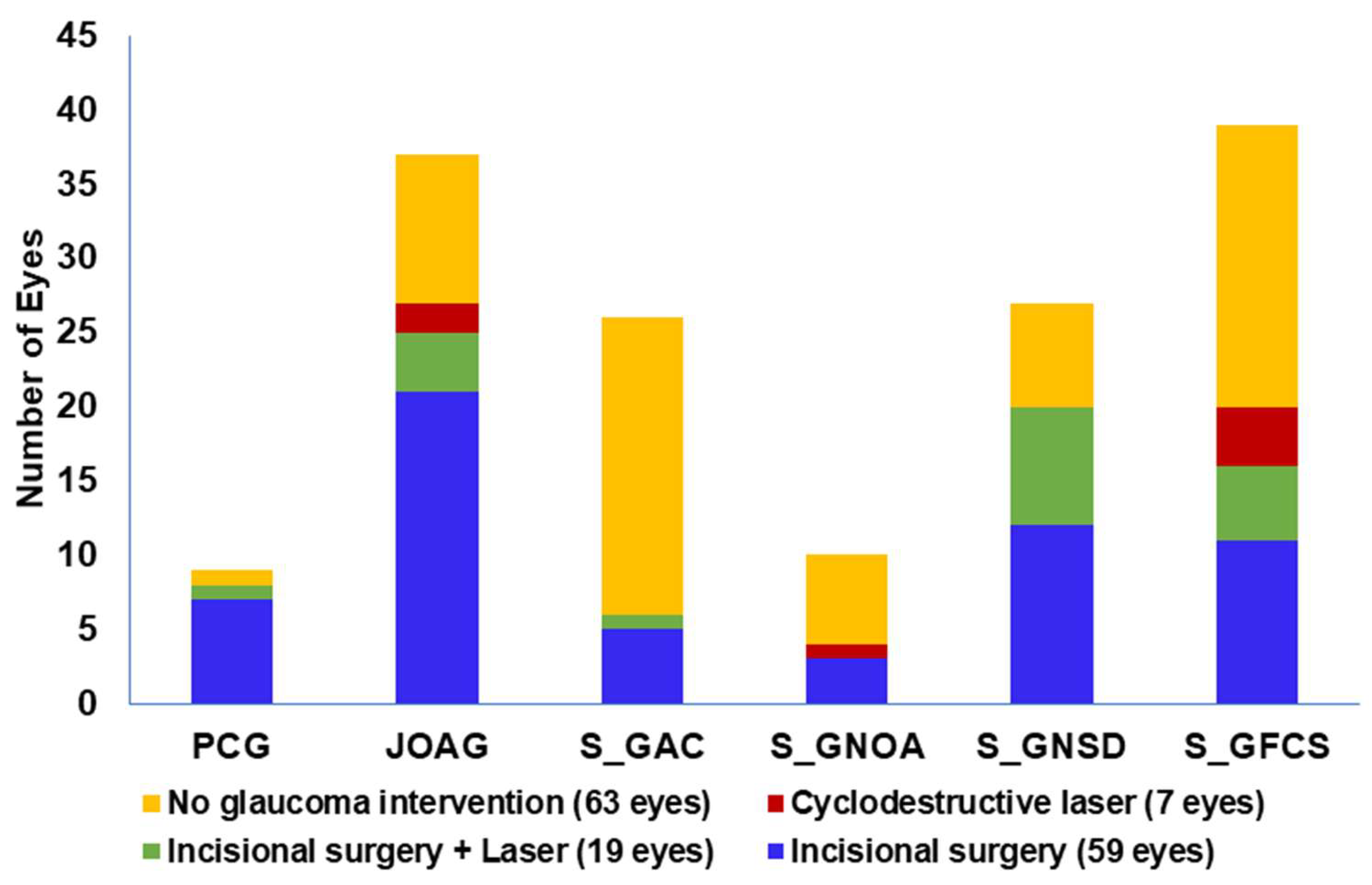
3.3. Surgical Interventions
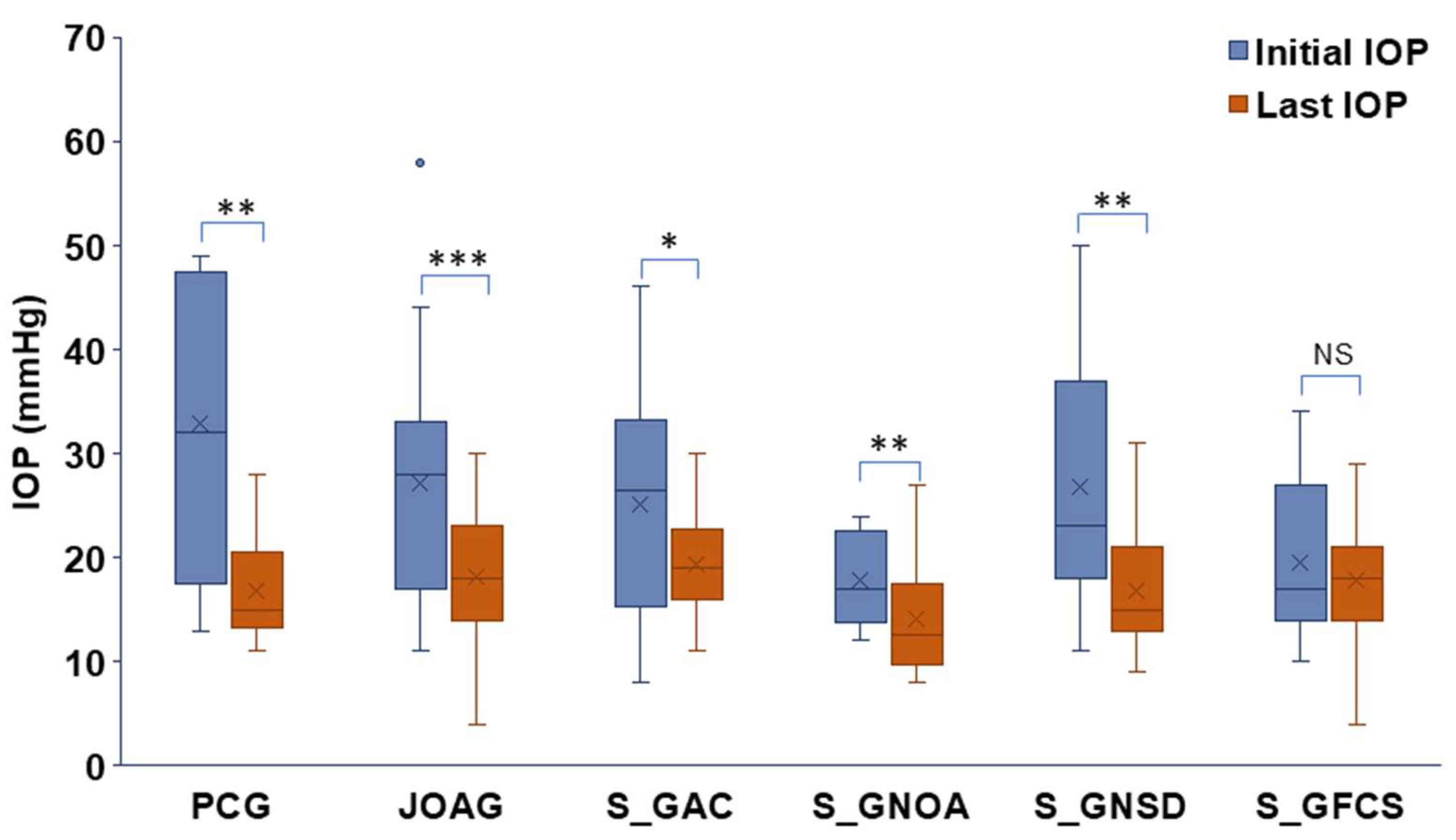
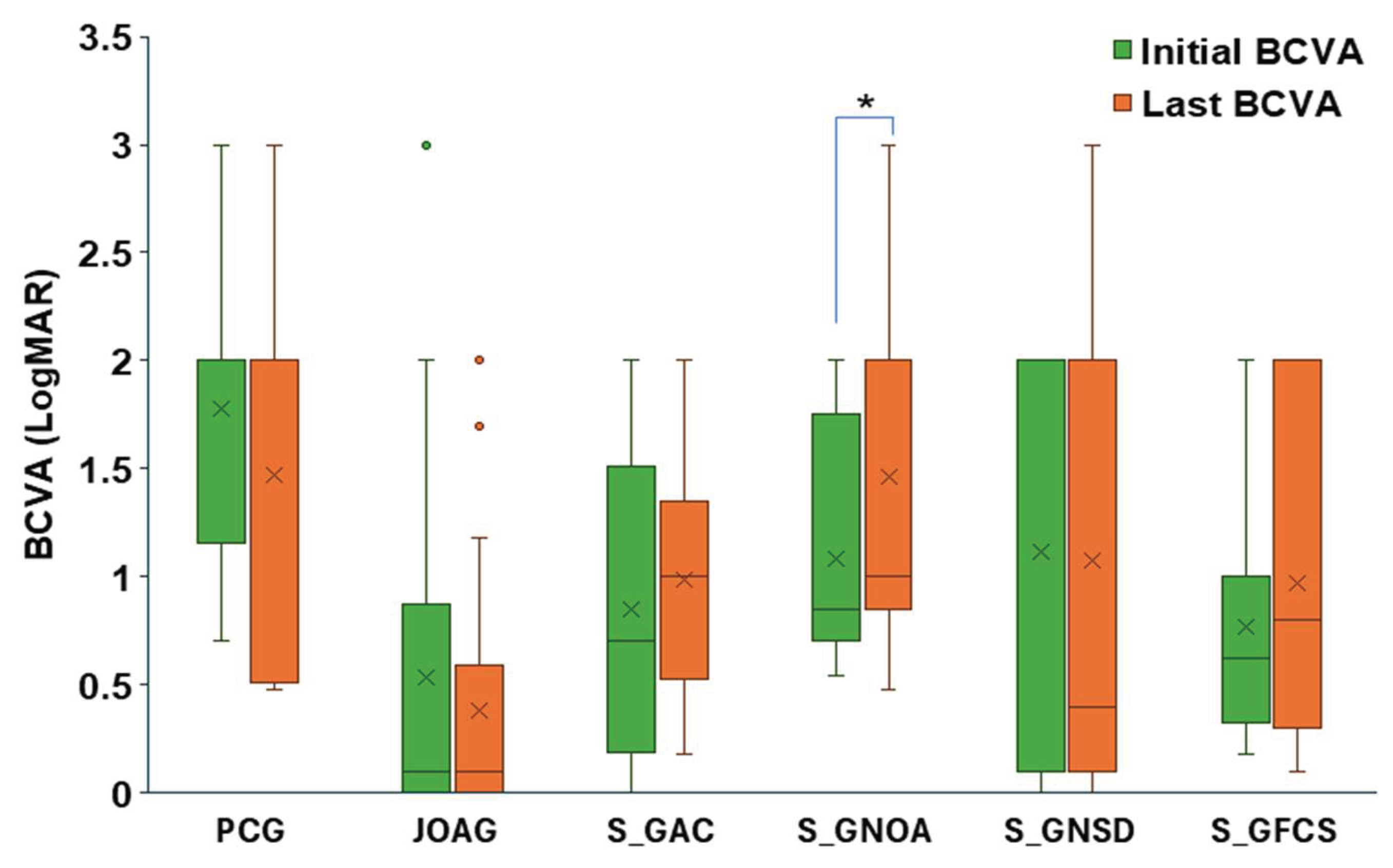
3.4. Follow-Up Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- deLuise, V. P.; Anderson, D. R. Primary infantile glaucoma (congenital glaucoma). Surv Ophthalmol 1983, 28, 1–19. [Google Scholar] [CrossRef]
- Aponte, E. P.; Diehl, N.; Mohney, B. G. Incidence and clinical characteristics of childhood glaucoma: a population-based study. Arch Ophthalmol 2010, 128, 478–82. [Google Scholar] [CrossRef] [PubMed]
- Kipp, M. A. Childhood glaucoma. Pediatr Clin North Am 2003, 50, 89–104. [Google Scholar] [CrossRef]
- Alodhayb, S.; Morales, J.; Edward, D. P.; Abu-Amero, K. K. Update on pediatric glaucoma. Semin Ophthalmol 2013, 28, 131–43. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, M.; Edmunds, B.; Fenerty, C.; Khaw, P. T. Childhood glaucoma surgery in the 21st century. Eye (Lond) 2014, 28, 931–43. [Google Scholar] [CrossRef] [PubMed]
- Lingham, G.; Thakur, S.; Safi, S.; Gordon, I.; Evans, J. R.; Keel, S. A systematic review of clinical practice guidelines for childhood glaucoma. BMJ Open Ophthalmol 2022, 7, e000933. [Google Scholar] [CrossRef]
- Hoguet, A.; Grajewski, A.; Hodapp, E.; Chang, T. C. A retrospective survey of childhood glaucoma prevalence according to Childhood Glaucoma Research Network classification. Indian J Ophthalmol 2016, 64, 118–23. [Google Scholar] [CrossRef]
- Thau, A.; Lloyd, M.; Freedman, S.; Beck, A.; Grajewski, A.; Levin, A. V. New classification system for pediatric glaucoma: implications for clinical care and a research registry. Curr Opin Ophthalmol 2018, 29, 385–394. [Google Scholar] [CrossRef]
- Karaconji, T.; Zagora, S.; Grigg, J. R. Approach to childhood glaucoma: A review. Clin Exp Ophthalmol 2022, 50, 232–246. [Google Scholar] [CrossRef]
- Taylor, R. H.; Ainsworth, J. R.; Evans, A. R.; Levin, A. V. The epidemiology of pediatric glaucoma: the Toronto experience. J AAPOS 1999, 3, 308–15. [Google Scholar] [CrossRef]
- Fung, D. S.; Roensch, M. A.; Kooner, K. S.; Cavanagh, H. D.; Whitson, J. T. Epidemiology and characteristics of childhood glaucoma: results from the Dallas Glaucoma Registry. Clin Ophthalmol 2013, 7, 1739–46. [Google Scholar] [CrossRef]
- Bouhenni, R. A.; Ricker, I.; Hertle, R. W. Prevalence and Clinical Characteristics of Childhood Glaucoma at a Tertiary Care Children’s Hospital. J Glaucoma 2019, 28, 655–659. [Google Scholar] [CrossRef]
- Surukrattanaskul, S.; Suvannachart, P.; Chansangpetch, S.; Manassakorn, A.; Tantisevi, V.; Rojanapongpun, P. Characteristics and long-term outcomes of childhood glaucoma: a retrospective-cohort study. F1000Res 2021, 10, 165. [Google Scholar] [CrossRef] [PubMed]
- Fujita, A.; Vu, D. M.; Aziz, K.; Chang, T. C.; Chung, I. Y.; Ciociola, E. C.; Friedman, D. S.; Lokhande, A.; Lorch, A. C.; Miller, J. W.; Parikh, P.; Rothman, A. L.; Seresirikachorn, K.; Elze, T.; Zebardast, N.; Consortium, I. R. A. C. Treatment Patterns of Childhood Glaucoma in the United States: Analysis of IRIS(Ⓡ) Registry (Intelligent Research in Sight). Am J Ophthalmol 2024, 271, 210–221. [Google Scholar] [CrossRef] [PubMed]
- Holmes, J. M.; Beck, R. W.; Repka, M. X.; Leske, D. A.; Kraker, R. T.; Blair, R. C.; Moke, P. S.; Birch, E. E.; Saunders, R. A.; Hertle, R. W.; Quinn, G. E.; Simons, K. A.; Miller, J. M. Pediatric Eye Disease Investigator, G., The amblyopia treatment study visual acuity testing protocol. Arch Ophthalmol 2001, 119, 1345–53. [Google Scholar] [CrossRef]
- Holladay, J. T. Visual acuity measurements. J Cataract Refract Surg 2004, 30, 287–90. [Google Scholar] [CrossRef]
- Wallace, D. K.; Morse, C. L.; Melia, M.; Sprunger, D. T.; Repka, M. X.; Lee, K. A.; Christiansen, S. P. American Academy of Ophthalmology Preferred Practice Pattern Pediatric Ophthalmology/Strabismus, P., Pediatric Eye Evaluations Preferred Practice Pattern(R): I. Vision Screening in the Primary Care and Community Setting; II. Comprehensive Ophthalmic Examination. Ophthalmology 2018, 125, P184–P227. [Google Scholar] [PubMed]
- Tam, E. K.; Elhusseiny, A. M.; Shah, A. S.; Mantagos, I. S.; VanderVeen, D. K. Etiology and outcomes of childhood glaucoma at a tertiary referral center. J AAPOS 2022, 26, 117 e1–117 e6. [Google Scholar] [CrossRef]
- Beldick, S. R.; Rockter, A.; Beck, A. D.; Levin, A. V. The Robison D. Harley, MD Childhood Glaucoma Research Network International Pediatric Glaucoma Registry: The First 872 Cases. Ophthalmol Glaucoma 2024. [Google Scholar] [CrossRef]
- Jacobson, A.; Bohnsack, B. L. Visual and surgical outcomes of childhood glaucoma following cataract surgery. J AAPOS 2024, 28, 103993. [Google Scholar] [CrossRef]
- Knight, L. S. W.; Ruddle, J. B.; Taranath, D. A.; Goldberg, I.; Smith, J. E. H.; Gole, G.; Chiang, M. Y.; Willett, F.; D’Mellow, G.; Breen, J.; Qassim, A.; Mullany, S.; Elder, J. E.; Vincent, A. L.; Staffieri, S. E.; Kearns, L. S.; Mackey, D. A.; Luu, S.; Siggs, O. M.; Souzeau, E.; Craig, J. E. Childhood and Early Onset Glaucoma Classification and Genetic Profile in a Large Australasian Disease Registry. Ophthalmology 2021, 128, 1549–1560. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, S.; Senthil, S.; Gupta, V.; Balekudaru, S.; Dubey, S.; Ali, H.; Mandal, A. K. Indian Childhood Glaucoma Study, G., Profile of Newly Diagnosed Childhood Glaucoma in India: Indian Childhood Glaucoma Study (ICGS) Group 1. Ophthalmol Glaucoma 2024, 7, 54–65. [Google Scholar] [CrossRef] [PubMed]
- Qiao, C. Y.; Wang, L. H.; Tang, X.; Wang, T.; Yang, D. Y.; Wang, N. L. Epidemiology of hospitalized pediatric glaucoma patients in Beijing Tongren Hospital. Chin Med J (Engl) 2009, 122, 1162–6. [Google Scholar] [PubMed]
- Mokbel, T. H.; El Hefney, E. M.; Hagras, S. M.; AA, A. L.; Badawi, A. E.; Kasem, M. A.; El Shaer, S. M. Childhood glaucoma profile in Dakahelia, Egypt: a retrospective study. Int J Ophthalmol 2018, 11, 674–680. [Google Scholar]
- Lopes, N. L.; Gracitelli, C. P. B.; Rolim-de-Moura, C. Childhood Glaucoma Profile in a Brazilian Tertiary Care Center Using Childhood Glaucoma Research Network Classification. J Glaucoma 2021, 30, 129–133. [Google Scholar] [CrossRef]
- Liu, Q.; Liu, C.; Li, H.; Yang, X.; Dong, Y.; Feng, X.; Cheng, W. Clinical Analysis of Pediatric Glaucoma in Central China. Front Med (Lausanne) 2022, 9, 874369. [Google Scholar] [CrossRef]
- Mulugeta, T.; Gebremichael, G.; Adugna, S. Childhood glaucoma profile in a Southwestern Ethiopia tertiary care center: a retrospective study. BMC Ophthalmol 2024, 24, 31. [Google Scholar] [CrossRef]
- Papadopoulos, M.; Cable, N.; Rahi, J.; Khaw, P. T.; Investigators. B. I. G. E. S., The British Infantile and Childhood Glaucoma (BIG) Eye Study. Invest Ophthalmol Vis Sci 2007, 48, 4100–6. [Google Scholar] [CrossRef]
- Aktas, Z.; Gulpinar Ikiz, G. D. Current surgical techniques for the management of pediatric glaucoma: A literature review. Front Ophthalmol (Lausanne) 2023, 3, 1101281. [Google Scholar] [CrossRef]
| Characteristics | PCG | JOAG | S_GAC | S_GNOA | S_GNSD | S_GFCS | GS | Total |
|---|---|---|---|---|---|---|---|---|
| Number of patients, n (%) | 7 (6.7) | 19 (18.1) | 21 (20) | 7 (6.7) | 16 (15.3) | 24 (22.9) | 11 (10.5) | 105 (100) |
| Number of eyes, n (%) | 9 (5.4) | 37 (22) | 26 (15.5) | 10 (6.0) | 27 (16.1) | 39 (23.2) | 20 (11.9) | 168 (100) |
| Gender (patients, n) | ||||||||
| 11111Male, n (%) | 4 (51.7) | 9 (47.4) | 16 (76.2) | 2 (28.6) | 9 (56.3) | 12 (50) | 4 (36.4) | 56 (53.3) |
| 11111Female, n (%) | 3 (42.8) | 10 (52.6) | 5 (23.8) | 5 (71.4) | 7 (43.7) | 12 (50) | 7 (63.6) | 49 (46.7) |
| Race, Caucasian, n (%) | 5 (1.0) | 14 (13.3) | 11 (10.5) | 5 (1.0) | 11 (10.5) | 17 (16.2) | 9 (8.57) | 72 (68.57) |
| Age at first visit (year)* | 2.98 (1.91) | 8.24 (7.07) | 7.9 (4.95) | 3.25 (4.7) | 3.9 (3.36) | 3.19 (5.37) | 6.8 (4.39) | 5.52 (5.6) |
| Age at last visit (year)* | 4.26 (1.96) | 14.1 (7.83) | 12.8 (4.23) | 9.06 (3.28) | 9.17 (5.3) | 10.08 (5.48) | 12 (3.89) | 10.93 (5.9) |
| Duration follow-up (year)* | 1.29 (1.71) | 5.86 (3.87) | 4.78 (3.05) | 5.81 (3.72) | 5.27 (3.39) | 6.89 (2.67) | 5.2 (3.23) | 5.41 (3.37) |
| Laterality (patients) | ||||||||
| 11111Bilateral, n (%) | 2 (28.6) | 18 (94.7) | 5 (23.8) | 3 (42.9) | 11 (68.8) | 15 (62.5) | 9 (81.8) | 63 (60) |
| 11111Unilateral, n (%) | 5 (71.4) | 1 (5.3) | 16 (71.2) | 4 (57.1) | 5 (31.2) | 9 (37.5) | 2 (11.2) | 42 (40) |
| Glaucoma subtypes |
IOP (mmHg) | Cup-to-Disc Ratio (CDR) | Spherical Equivalent |
| (Mean ± SD) Eyes (n) | (Mean ± SD) Eyes (n) | (Median, IQR) Eyes (n) | |
| PCG | 32.89 ± 14.31 (n = 9) | 0.64 ± 0.25 (n = 8) | - 2.87, 4.5 (n = 9) |
| JOAG | 27.16 ± 10.41 (n = 37) | 0.61 ± 0.19 (n = 30) | - 4.25, 6.5 (n = 37) |
| S_GAC | 25.17 ± 11.56 (n = 18) | 0.38 ± 0.14 (n = 23) | - 0.50, 4.5 (n = 26) |
| S_GNOA | 17.81 ± 4.61 (n = 10) | 0.28 ± 0.17 (n = 7) | + 0.5, 13.87 (n = 10) |
| S_GNSD | 26.87 ± 12.62 (n = 23) | 0.53 ± 0.25 (n = 21) | - 0.75, 4.87 (n = 27) |
| S_GFCS | 20.47 ± 7.72 (n = 19) | 0.37 ± 0.22 (n = 31) | + 9.63, 8.87 (n = 39) |
| GS | 17.22 ± 3.71 (n = 15) | 0.60 ± 0.21 (n = 16) | + 1.0, 2.75 (n = 20) |
| Overall | 24.41 ± 10.85 (n = 131) | 0.46 ± 0.23 (n = 136) | - 0.63, 4.25 (n =168) |
| BCVA (LogMAR) | Unilateral Glaucoma | Bilateral Glaucoma | |||
| Glaucomatous eye | Non-glaucomatous eye | Better seeing eye | Worse seeing eye | Eyes Equal | |
| ≤ 0.3 | 5 | 14 | 4 | 3 | 16 |
| >0.3 - ≤0.6 | 1 | 1 | 4 | 2 | 4 |
| >0.6 - ≤1.0 | 0 | 0 | 1 | 3 | 2 |
| >1.0 | 0 | 0 | 0 | 1 | 0 |
| CSM | 2 | 5 | 2 | 0 | 8 |
| CF or HM | 6 | 1 | 1 | 1 | 0 |
| LP + UFF | 7 | 6 | 1 | 1 | 16 |
| NLP | 2 | 0 | 0 | 0 | 0 |
| Surgery Procedure | PCG | JOAG | S_GAC | S_GNOA | S_GNSD | S_GFCS |
|---|---|---|---|---|---|---|
| eyes, n | eyes, n | eyes, n | eyes, n | eyes, n | eyes, n | |
| Trabeculectomy | 1 | 10 | 2 | 1 | 9 | 3 |
| Trabeculotomy | 1 | 2 | 0 | 0 | 0 | 0 |
| Trabeculectomy + Baerveldt | 0 | 4 | 0 | 0 | 4 | 6 |
| Baerveldt implantation | 2 | 6 | 1 | 1 | 0 | 4 |
| Goniotomy | 3 | 1 | 0 | 0 | 4 | 1 |
| Iridectomy | 0 | 0 | 2 | 1 | 1 | 1 |
| Cyclodestructive laser | 0 | 2 | 0 | 1 | 0 | 4 |
| Baerveldt + Iridex laser | 1 | 2 | 1 | 0 | 2 | 5 |
| Total | 8 | 27 | 6 | 4 | 20 | 20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




