Submitted:
16 March 2026
Posted:
17 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Inclusion and Exclusion Criteria
- age ≥18 years;
- instrumental assessment of LVEF within the previous 12 months;
- documented cardiac rhythm (sinus rhythm, AF, or other rhythm) confirmed by electrocardiography (ECG) or Holter ECG monitoring;
- availability of sufficiently complete clinical, laboratory, and imaging data required for the analysis.
- an acute cardiovascular event (myocardial infarction, ischemic or hemorrhagic stroke, acute peripheral arterial incident, or other) ≤90 days prior to the time of assessment;
- implanted rhythm-modifying cardiac devices (e.g., permanent pacemakers, implantable cardioverter-defibrillators, or cardiac resynchronization therapy devices);
- significant uncontrolled comorbidities that could influence the analyzed parameters (e.g., active malignancy, severe renal failure, uncontrolled endocrine disorders);
- incomplete medical documentation preventing adequate analysis.
2.3. Electrocardiography and Holter ECG Monitoring
2.4. Echocardiographic Assessment
2.5. Laboratory Investigations
2.6. Ethical Considerations
2.7. Statistical Analysis
3. Results
3.1. Baseline Characteristics of the Study Population
4. Discussion
4.1. Main Findings of the Study
4.2. Interpretation of Predictors in HFpEF
4.3. Predictors of AF in HFmrEF
4.4. Predictors of AF in HFrEF
4.5. Comparison with Previous Studies
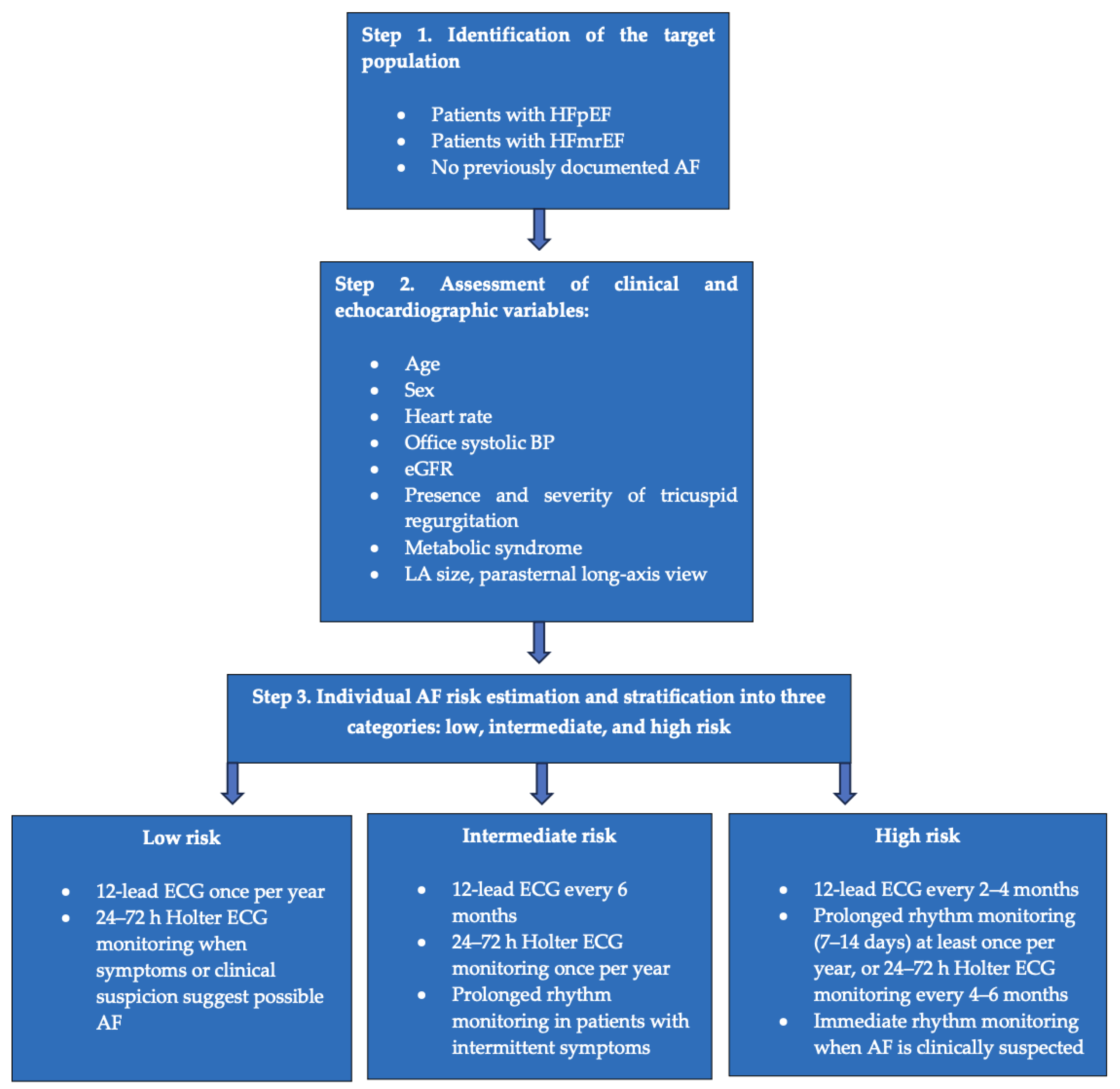
4.6. Clinical Implications
4.7. Highlights
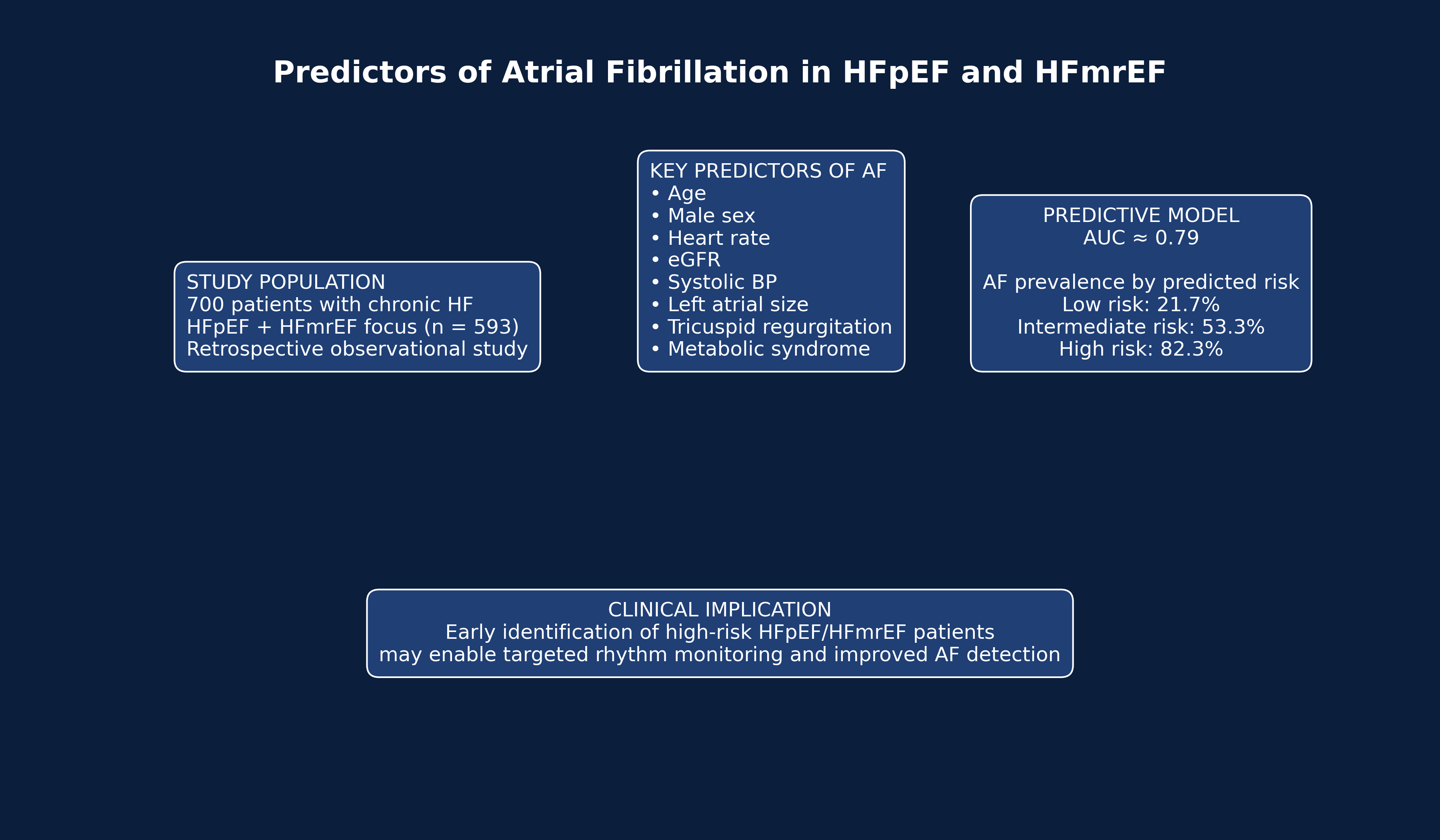
- HFpEF demonstrates the most complex network of clinical and echocardiographic determinants of AF.
- LA enlargement is the most consistent predictor of AF in HFpEF and HFmrEF.
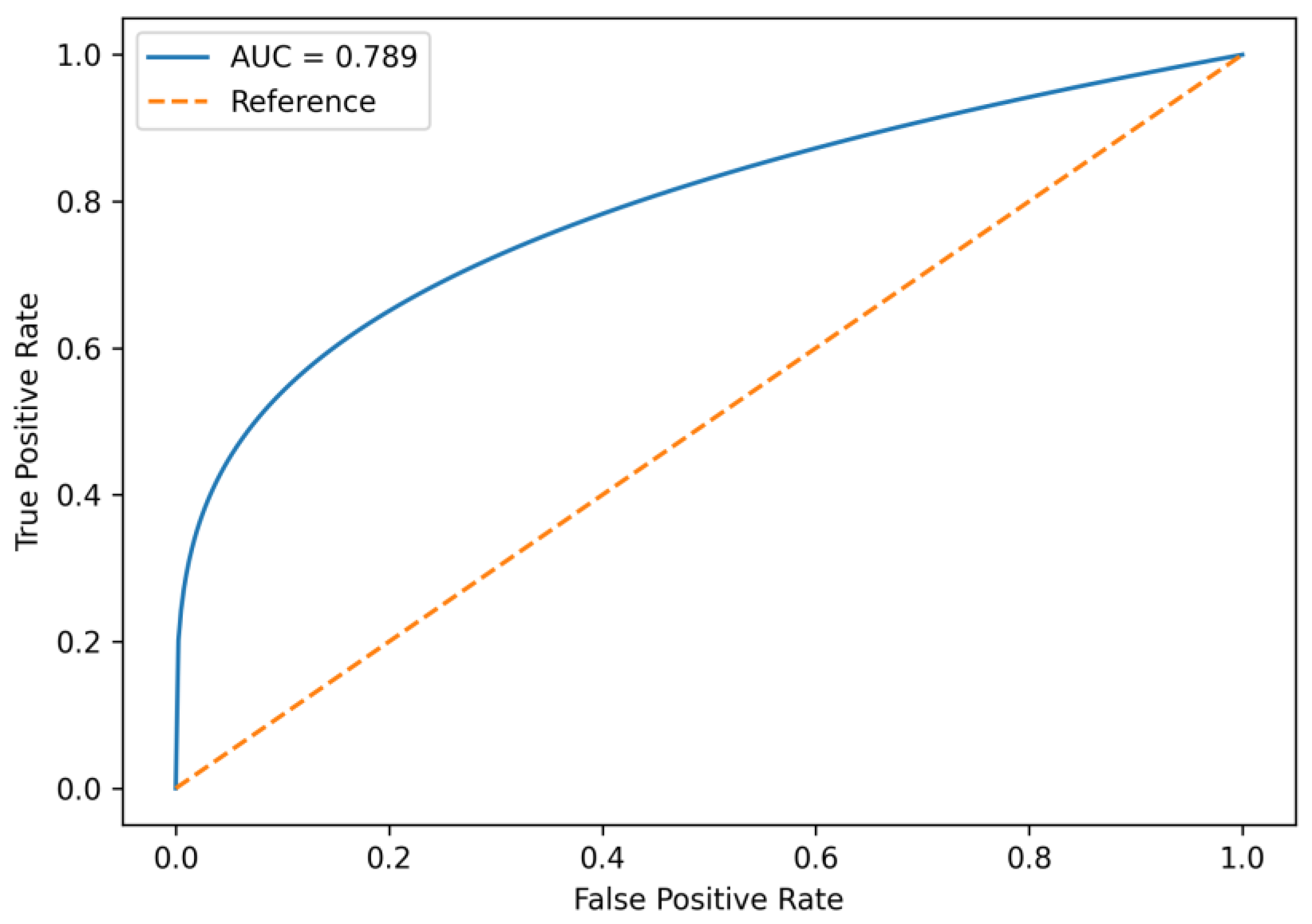
- A multivariable model combining clinical and echocardiographic parameters showed good predictive performance (AUC ≈ 0.79).
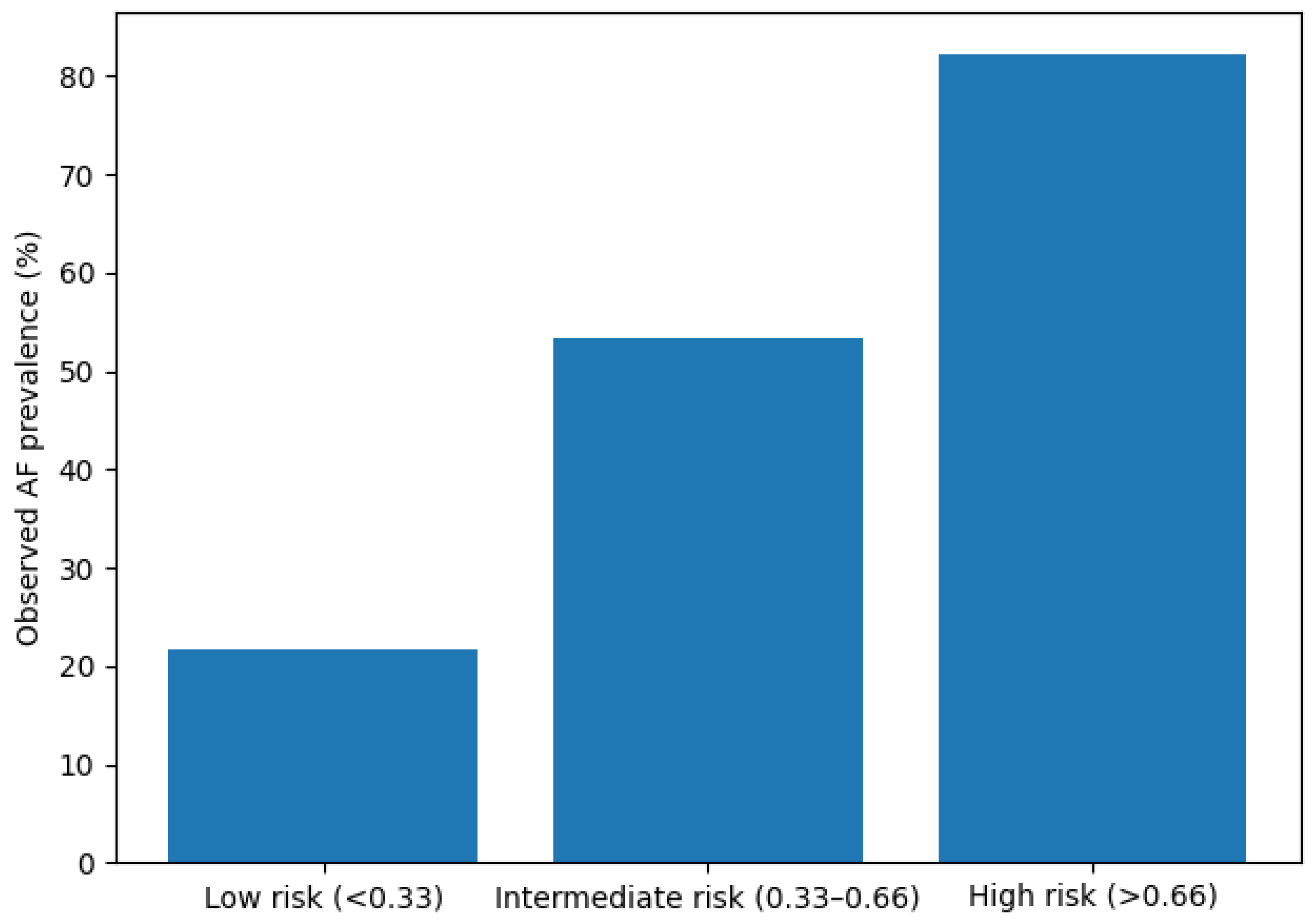
- Risk stratification based on predicted probability effectively discriminated low-, intermediate-, and high-risk groups for AF.
4.8. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AF | Atrial fibrillation |
| BP | Blood pressure |
| eGFR | Estimated glomerular filtration rate |
| ECG | Electrocardiography |
| ESC | European Society of Cardiology |
| HF | Heart failure |
| HFmrEF | Heart failure with mildly reduced ejection fraction |
| HFpEF | Heart failure with preserved ejection fraction |
| HFrEF | Heart failure with reduced ejection fraction |
| LA | Left atrial |
| LV | Left ventricular |
| LVEF | Left ventricular ejection fraction |
References
- Van Gelder IC, Rienstra M, Bunting KV, Casado-Arroyo R, Caso V, Crijns HJGM; et al. 2024 ESC Guidelines for the management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). European Heart Journal. 2024 Sep 29;45(36):3314–414. [CrossRef]
- Joglar JA, Chung MK, Armbruster AL, Benjamin EJ, Chyou JY, Cronin EM; et al. 2023 ACC/AHA/ACCP/HRS Guideline for the Diagnosis and Management of Atrial Fibrillation: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2024 Jan 2;149(1). [CrossRef]
- Karatela MF, Calkins H. The Global Impact of Atrial Fibrillation. Arrhythm Electrophysiol Rev. 2025 Nov 8;14:e28. [CrossRef]
- Alexander B, Tse G, Martinez-Selles M, Baranchuk A. Atrial Conduction Disorders. CCR. 2021 Jan;17(1):68–73. [CrossRef]
- Lippi G, Sanchis-Gomar F, Cervellin G. Global epidemiology of atrial fibrillation: An increasing epidemic and public health challenge. International Journal of Stroke. 2021 Feb;16(2):217–21. [CrossRef]
- Zhang X, Chen Y, Hu W, Lu S, Yu H, Sun Y. Global burden of atrial fibrillation/atrial flutter and its attributable risk factors in adolescents and young adults, 1990–2021: Insights from the global burden of disease study. Annals of Medicine. 2025 Dec 31;57(1):2543524. [CrossRef]
- Bassand JP, Apenteng PN, Atar D, Camm AJ, Cools F, Corbalan R; et al. GARFIELD-AF: A Worldwide Prospective Registry of Patients With Atrial Fibrillation at Risk of Stroke. Future Cardiol. 2021 Jan;17(1):19–38. [CrossRef]
- McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. European Heart Journal. 2021 Sep 21;42(36):3599–726. [CrossRef]
- McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M; et al. 2023 Focused Update of the 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. European Heart Journal. 2023 Oct 1;44(37):3627–39. [CrossRef]
- Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM; et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022 May 3;145(18). [CrossRef]
- Jia Z, Du X. Epidemiology and Burden of Heart Failure. Cardiology Discovery. 2025 Dec;5(4):281–4. [CrossRef]
- Bidaoui G, Assaf A, Marrouche N. Atrial Fibrillation in Heart Failure: Novel Insights, Challenges, and Treatment Opportunities. Curr Heart Fail Rep. 2025 Dec;22(1):3. [CrossRef]
- Gorczyca-Głowacka I, Galas A, Tymińska A, Byczkowska K, Furman-Niedziejko A, Tkaczyszyn M; et al. Atrial fibrillation in patients with heart failure with reduced, mildly reduced, and preserved ejection fraction: A report from the HEart failuRe ObsErvational Study (HEROES). Pol Heart J. 2025 Jun 30;83(6):701–8. [CrossRef]
- Corica B, Romiti GF, Proietti M, Boriani G, Olshansky B, Huisman MV; et al. Heart failure in patients with recently diagnosed atrial fibrillation: Findings from the GLORIA-AF Registry Phase III. European Journal of Preventive Cardiology. 2025 Sep 1;zwaf485. [CrossRef]
- Xia Y, Jiang J, Fan F, Pan Y, Zhou J, Zhang Y; et al. Prevalence, Characteristics, and Treatment Strategy of Different Types of Heart Failure in Patients With Nonvalvular Atrial Fibrillation. JAHA. 2024 Oct;13(19):e033941. [CrossRef]
- Bozkurt B, Ahmad T, Alexander K, Baker WL, Bosak K, Breathett K; et al. HF STATS 2024: Heart Failure Epidemiology and Outcomes Statistics An Updated 2024 Report from the Heart Failure Society of America. Journal of Cardiac Failure. 2025 Jan;31(1):66–116. [CrossRef]
- Gori M, Iacovoni A, Senni M. Haemodynamics of Heart Failure With Preserved Ejection Fraction: A Clinical Perspective. Cardiac Failure Review. 2016. [CrossRef]
- Savarese G, Stolfo D, Sinagra G, Lund LH. Heart failure with mid-range or mildly reduced ejection fraction. Nat Rev Cardiol. 2022 Feb;19(2):100–16. [CrossRef]
- Saksena S, Slee A, Nagarakanti R, Atul Prakash. Atrial Fibrillation (AF) and Heart Failure With Preserved Ejection Fraction (HFpEF): Advances and Challenges. Cardiovasc electrophysiol. 2025 Oct;36(10):2720–45. [CrossRef]
- Ariyaratnam JP, Elliott AD, Mishima RS, Gallagher C, Lau DH, Sanders P. Heart failure with preserved ejection fraction: An alternative paradigm to explain the clinical implications of atrial fibrillation. Heart Rhythm O2. 2021 Dec;2(6):771–83. [CrossRef]
- Gorter TM, Van Veldhuisen DJ, Mulder BA, Artola Arita VA, Van Empel VPM, Manintveld OC; et al. Prevalence and Incidence of Atrial Fibrillation in Heart Failure with Mildly Reduced or Preserved Ejection Fraction: (Additive) Value of Implantable Loop Recorders. JCM. 2023 May 26;12(11):3682. [CrossRef]
- Patel RB, Greene SJ, Xu H, Alhanti B, Peterson P, Yancy CW; et al. Intersection of Atrial Fibrillation and Heart Failure with Mildly Reduced and Preserved Ejection Fraction in >400 000 Participants in the Get With The Guidelines-Heart Failure Registry. European Journal of Heart Failure. 2023 Jan 1;25(1):63–73. [CrossRef]
- Chousou PA, Chattopadhyay R, Tsampasian V, Vassiliou VS, Pugh PJ. Electrocardiographic Predictors of Atrial Fibrillation. Medical Sciences. 2023 Apr 7;11(2):30. [CrossRef]
- Nguyen KT, Vittinghoff E, Dewland TA, Mandyam MC, Stein PK, Soliman EZ; et al. Electrocardiographic Predictors of Incident Atrial Fibrillation. The American Journal of Cardiology. 2016 Sep;118(5):714–9. [CrossRef]
- Atabekov T, Batalov R, Archakov E, Silivanova I, Khlynin M, Kisteneva I; et al. Predictors of Atrial Fibrillation in Heart Failure Patients with Indications for ICD Implantation. JCM. 2025 Jun 18;14(12):4358. [CrossRef]
- Park JJ, Park JH, Hwang IC, Park JB, Cho GY, Marwick TH. Left Atrial Strain as a Predictor of New-Onset Atrial Fibrillation in Patients With Heart Failure. JACC: Cardiovascular Imaging. 2020 Oct;13(10):2071–81. [CrossRef]
- O’Neal WT, Sandesara P, Patel N, Venkatesh S, Samman-Tahhan A, Hammadah M; et al. Echocardiographic predictors of atrial fibrillation in patients with heart failure with preserved ejection fraction. European Heart Journal - Cardiovascular Imaging. 2017 Jul 1;18(7):725–9. [CrossRef]
- Boehmer AA, Ehrlich JR, Nattel S. Atrial Fibrillation and Heart Failure With Reduced Ejection Fraction. JACC: Clinical Electrophysiology. 2025 Dec;11(12):2774–90. [CrossRef]
- A. Al-Shoaibi N, Y. Ashram W, T. Mokhtar A, K. Arfaj Y, G. Alnahari D, S. Ibrahim R; et al. Non-genetic Risk Factors for Atrial Fibrillation in Individuals Aged 60 and Above. J Pioneering Med Sci. 2025 Oct 5;14(09):1–5. [CrossRef]
- Chen M, Ding N, Mok Y, Mathews L, Hoogeveen RC, Ballantyne CM; et al. Growth Differentiation Factor 15 and the Subsequent Risk of Atrial Fibrillation: The Atherosclerosis Risk in Communities Study. Clinical Chemistry. 2022 Jul 27;68(8):1084–93. [CrossRef]
- Bertoni M1*, Traini AM2, Celli A3, Bini C1, Bracciali A1, Foretic M1 and Di Natale ME. Galectin-3 in Heart Failure with Preserved Ejection Fraction and Persistent Atrial Fibrillation Versus Sinus Rhythm. Correlation with Left Atrial Volume and N-Terminal Pro B-Type Natriuretic Peptide [Internet]. Available from: https://austinpublishinggroup.com/cardiovascular-disorders/fulltext/jcd-v7-id1043.php.
- Ianos RD, Iancu M, Pop C, Lucaciu RL, Hangan AC, Rahaian R; et al. Predictive Value of NT-proBNP, FGF21, Galectin-3 and Copeptin in Advanced Heart Failure in Patients with Preserved and Mildly Reduced Ejection Fraction and Type 2 Diabetes Mellitus. Medicina. 2024 Nov 8;60(11):1841. [CrossRef]
- Lancellotti P, Cosyns B, editors. Examination. In: The EACVI Echo Handbook [Internet]. 1st ed. Oxford University PressOxford; 2015 [cited 2026 Mar 8]. p. 1–14. Available from: https://academic.oup.com/esc/book/40751/chapter/348644852. [CrossRef]
- Stevens PE, Ahmed SB, Carrero JJ, Foster B, Francis A, Hall RK; et al. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney International. 2024 Apr;105(4):S117–314. [CrossRef]
- Sagris D, Harrison SL, Lip GYH. Lipids and atrial fibrillation: New insights into a paradox. PLoS Med. 2022 Aug 11;19(8):e1004067. [CrossRef]
- Wang TJ. Obesity and the Risk of New-Onset Atrial Fibrillation. JAMA. 2004 Nov 24;292(20):2471. [CrossRef]
- Sandhu RK, Ezekowitz J, Andersson U, Alexander JH, Granger CB, Halvorsen S; et al. The ‘obesity paradox’ in atrial fibrillation: Observations from the ARISTOTLE (Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation) trial. Eur Heart J. 2016 Oct 7;37(38):2869–78. [CrossRef]
- Carbone S, Canada JM, Billingsley HE, Siddiqui MS, Elagizi A, Lavie CJ. Obesity paradox in cardiovascular disease: Where do we stand? VHRM. 2019 May;Volume 15:89–100. [CrossRef]
- Cheng Y, Deng H, Wei H, Lin W, Liang Z, Chen Y; et al. Association Between Age at Diagnosis of Atrial Fibrillation and Subsequent Risk of Ischemic Stroke. JAHA. 2025 Feb 18;14(4):e038367. [CrossRef]
- Nieuwlaat R, Eurlings LW, Cleland JG, Cobbe SM, Vardas PE, Capucci A; et al. Atrial Fibrillation and Heart Failure in Cardiology Practice: Reciprocal Impact and Combined Management From the Perspective of Atrial Fibrillation. Journal of the American College of Cardiology. 2009 May;53(18):1690–8. [CrossRef]
- Zafrir B, Lund LH, Laroche C, Ruschitzka F, Crespo-Leiro MG, Coats AJS; et al. Prognostic implications of atrial fibrillation in heart failure with reduced, mid-range, and preserved ejection fraction: A report from 14 964 patients in the European Society of Cardiology Heart Failure Long-Term Registry. European Heart Journal. 2018 Dec 21;39(48):4277–84. [CrossRef]
- Ito T, Noda T, Nochioka K, Shiroto T, Yamamoto N, Sato H; et al. Clinical impact of atrial fibrillation progression in patients with heart failure with preserved ejection fraction: A report from the CHART-2 Study. Europace. 2024 Aug 30;26(9):euae218. [CrossRef]
- Chen P, Yin X, Ren L. Construction and validation of a predictive model for the risk of atrial fibrillation in patients with heart failure with preserved ejection fraction: A single-center retrospective analysis. BMC Cardiovasc Disord. 2025 Dec 23;25(1):873. [CrossRef]
- Gopinathannair R, Chen LY, Chung MK, Cornwell WK, Furie KL, Lakkireddy DR; et al. Managing Atrial Fibrillation in Patients With Heart Failure and Reduced Ejection Fraction: A Scientific Statement From the American Heart Association. Circ: Arrhythmia and Electrophysiology. 2021 Jul;14(7). [CrossRef]
| Variable | HFpEF (n = 487; 69.6%) | HFmrEF (n = 106; 15.1%) | HFrEF (n = 107; 15.3%) | Total (n = 700) | p-value |
|---|---|---|---|---|---|
| Sex, n (%) | |||||
| Male | 179 (36.7%) | 64 (60.4%) | 70 (65.4%) | 313 (44.7%) | <0.001 |
| Female | 308 (63.3%) | 42 (39.6%) | 37 (34.6%) | 387 (55.3%) | |
| Age, median (IQR), years | 73 (67–80) | 73 (67–78) | 74 (65–83) | 74 (66–80) | 0.738 |
| Age group, n (%) | |||||
| <40 years | 0 (0%) | 0 (0%) | 1 (0.9%) | 2 (0.3%) | |
| 40–49 years | 0 (0%) | 0 (0%) | 1 (0.9%) | 15 (2.1%) | |
| 50–59 years | 9 (1.8%) | 3 (2.8%) | 3 (2.8%) | 65 (9.3%) | 0.051 |
| 60–69 years | 45 (9.2%) | 8 (7.5%) | 12 (11.2%) | 177 (25.3%) | |
| 70–79 years | 120 (24.6%) | 28 (26.4%) | 29 (27.1%) | 277 (39.6%) | |
| ≥80 years | 196 (40.2%) | 50 (47.2%) | 31 (29.0%) | 164 (23.4%) | |
| COVID-19 period, n (%) | |||||
| Before the pandemic | 210 (43.1%) | 54 (50.9%) | 40 (37.4%) | 304 (43.4%) | 0.132 |
| After the pandemic | 277 (56.9%) | 52 (49.1%) | 67 (62.6%) | 396 (56.6%) |
| Variable | HFpEF (n = 487; 69.6%) | HFmrEF (n = 106; 15.1%) | HFrEF (n = 107; 15.3%) | Total (n = 700) | p-value |
|---|---|---|---|---|---|
| HF duration before study inclusion, median (IQR), years | 1 (1–2) | 1 (1–4) | 1 (1–4) | 1 (1–3) | 0.077 |
| NYHA functional class, n (%) | |||||
| NYHA II | 12 (2.5%) | 1 (0.9%) | 0 (0.0%) | 13 (1.9%) | <0.001 |
| NYHA III | 474 (97.3%) | 105 (99.1%) | 101 (94.4%) | 680 (97.1%) | |
| NYHA IV | 1 (0.2%) | 0 (0%) | 6 (5.6%) | 7 (1.0%) | |
| Heart rhythm, n (%) | |||||
| Sinus rhythm | 249 (51.1%) | 33 (31.1%) | 35 (32.7%) | 317 (45.3%) | |
| Paroxysmal AF | 50 (10.3%) | 10 (9.4%) | 6 (5.6%) | 66 (9.4%) | |
| Persistent AF | 91 (18.7%) | 27 (25.5%) | 23 (21.5%) | 141 (20.1%) | <0.001 |
| Long-standing persistent AF | 1 (0.2%) | 0 (0%) | 0 (0%) | 1 (0.1%) | |
| Permanent AF | 96 (19.7%) | 36 (34.0%) | 43 (40.2%) | 175 (25.0%) |
| Variable | HFpEF (n = 487; 69.6%) | HFmrEF (n = 106; 15.1%) | HFrEF (n = 107; 15.3%) | Total (n = 700) | p-value |
|---|---|---|---|---|---|
| Arterial hypertension | 476 (97.7%) | 102 (96.2%) | 97 (90.7%) | 675 (96.4%) | 0.596 |
| Dyslipidemia | 163 (33.5%) | 33 (31.1%) | 39 (36.8%) | 235 (33.6%) | 0.678 |
| Impaired glucose metabolism | 8 (1.6%) | 1 (0.9%) | 0 (0%) | 9 (1.3%) | 0.834 |
| Impaired glucose tolerance/ impaired fasting glucose | |||||
| Type 2 diabetes mellitus | 142 (29.2%) | 34 (32.1%) | 26 (24.3%) | 202 (28.9%) | |
| Type 1 diabetes mellitus | 5 (1.0%) | 3 (2.8%) | 1 (0.9%) | 9 (1.3%) | |
| Metabolic syndrome | 27 (5.5%) | 2 (1.9%) | 3 (2.8%) | 32 (4.6%) | 0.167 |
| Weight disorders* | |||||
| Overweight | 14 (2.9%) | 1 (0.9%) | 1 (0.9%) | 16 (2.3%) | 0.330 |
| Obesity | 78 (16.0%) | 13 (12.3%) | 14 (13.1%) | 105 (15.0%) | |
| Coronary artery disease | 144 (29.6%) | 45 (42.5%) | 43 (40.2%) | 232 (30.1%) | 0.009 |
| Valvular heart disease# | 468 (96.3%) | 104 (98.1%) | 105 (98.1%) | 677 (96.9%) | 0.455 |
| Congenital heart disease | 2 (0.4%) | 1 (0.9%) | 0 (0%) | 3 (0.4%) | 0.572 |
| Chronic kidney disease | 93 (19.1%) | 23 (21.7%) | 34 (31.8%) | 150 (21.6%) | 0.015 |
| Cerebrovascular disease | 105 (21.6%) | 22 (20.8%) | 31 (29.0%) | 158 (22.6%) | 0.224 |
| Peripheral arterial disease | 0 (0%) | 0 (0%) | 2 (1.9%) | 2 (0.3%) | 0.004 |
| Prior cardiac surgery | 74 (15.2%) | 26 (24.5%) | 33 (30.8%) | 133 (19.0%) | <0.001 |
| Thyroid disease | 84 (17.3%) | 17 (16.0%) | 20 (18.7%) | 121 (17.3%) | 0.876 |
| Chronic pulmonary disease^ | 111 (22.8%) | 25 (23.6%) | 25 (23.4%) | 161 (23.0%) | 0.998 |
| Obstructive sleep apnea | 16 (3.3%) | 6 (5.7%) | 5 (4.7%) | 27 (3.9%) | 0.700 |
| Smoking status | |||||
| Active smoker | 63 (12.9%) | 21 (19.8%) | 19 (17.8%) | 103 (14.7%) | |
| Former smoker | 59 (12.1%) | 20 (18.9%) | 15 (14.0%) | 94 (13.4%) | 0.060 |
| HF Phenotype | Cardiac Rhythm | n (%) | CHA₂DS₂-VA score, median (IQR), points |
p-value |
|---|---|---|---|---|
| HFpEF (≥50%) | Paroxysmal AF | 50 (10.3%) | 4 (3–5) | |
| Persistent AF | 91 (18.7%) | 3 (3–5) | ||
| Long-standing persistent AF | 1 (0.2%) | 4 (4–4) | ||
| Permanent AF | 96 (19.7%) | 4 (3–5) | ||
| HFmrEF (41–49%) | Paroxysmal AF | 10 (9.4%) | 3 (2.8–5) | 0.568 |
| Persistent AF | 27 (25.5%) | 4 (2–5) | ||
| Permanent AF | 36 (34.0%) | 4 (3–5) | ||
| HFrEF (≤40%) | Paroxysmal AF | 6 (5.6%) | 2.5 (1.5–4) | |
| Persistent AF | 23 (21.5%) | 3 (2–5) | ||
| Permanent AF | 43 (40.2%) | 4 (3–5) |
| HF Phenotype | Cardiac Rhythm | n (%) | HAS-BLED score, median (IQR), points |
p-value |
|---|---|---|---|---|
| HFpEF (≥50%) | Paroxysmal AF | 50 (10.3%) | 2 (2–2) | |
| Persistent AF | 91 (18.7%) | 2 (1–2) | ||
| Long-standing persistent AF | 1 (0.2%) | 1 (1–1) | ||
| Permanent AF | 96 (19.7%) | 2 (2–3) | ||
| HFmrEF (41–49%) | Paroxysmal AF | 10 (9.4%) | 2 (1–2.25) | 0.523 |
| Persistent AF | 27 (25.5%) | 2 (2–2) | ||
| Permanent AF | 36 (34.0%) | 2 (2–3) | ||
| HFrEF (≤40%) | Paroxysmal AF | 6 (5.6%) | 1.5 (0.75–2.75) | |
| Persistent AF | 23 (21.5%) | 2 (1–3) | ||
| Permanent AF | 43 (40.2%) | 2 (2–2) |
| Variable | HFpEF (n = 487) | HFmrEF (n = 106) | HFrEF (n = 107) | Total | p-value |
|---|---|---|---|---|---|
| Office systolic BP, median (IQR), mmHg | 135 (120–150) | 130 (120–150) | 125 (110.5–140) | 130 (120–150) | 0.002 |
| Office diastolic BP, median (IQR), mmHg | 80 (75–90) | 80 (75–90) | 80 (70–90) | 80 (75–90) | 0.530 |
| Body mass index, median (IQR), kg/m² | 31 (28–33.75) | 30 (29.25–30.75) | 37 (24.5–41.7) | 31 (28–35) | 0.525 |
| Heart rate, mean ± SD, bpm | 78.3 ± 17.6 | 85.1 ± 20.4 | 89.3 ± 22.9 | 81.0 ± 19.4 | <0.001 |
| Variable | HFpEF (n = 487) | HFmrEF (n = 106) | HFrEF (n = 107) | Total (n = 700) | p-value |
|---|---|---|---|---|---|
| Potassium (K), mmol/L, median (IQR) | 4.00 (4.00–4.91) | 4.00 (4.00–4.75) | 4.00 (4.00–5.00) | 4.00 (4.00–4.97) | 0.691 |
| Sodium (Na), mmol/L, median (IQR) | 141 (138–143) | 141 (138–143) | 140 (138–143) | 141 (138–143) | 0.038 |
| Hemoglobin (Hb), g/L, median (IQR) | 137 (123–147) | 134 (121–148) | 134 (120–146) | 136 (122–147) | 0.673 |
| Creatinine, µmol/L, median (IQR) | 90 (74–112) | 100 (81.5–131) | 108 (86–156) | 93 (77–117) | 0.341 |
| eGFR, mL/min/1.73 m², median (IQR) | 65 (50–85) | 57 (43–85) | 56 (34–73.3) | 62 (45–84) | 0.037 |
| Variable | HFpEF (n = 487) | HFmrEF (n = 106) | HFrEF (n = 107) | p-value |
|---|---|---|---|---|
| LA size, parasternal long-axis view (mm), median (IQR) | 42 (38–47) | 45 (41.5–50) | 48 (43.5–53.5) | <0.001 |
| LA longitudinal diameter, apical four-chamber view (mm), median (IQR) | 54 (50–62) | 59 (53–63.5) | 62 (55.25–69) | <0.001 |
| Interventricular septal thickness, parasternal long-axis view (mm), median (IQR) | 12 (11–13) | 12 (11–13) | 12 (11–13) | 0.003 |
| Posterior LV wall thickness, parasternal long-axis view (mm), median (IQR) | 12 (11–12) | 12 (11–12) | 11 (10–12) | 0.005 |
| LV end-diastolic diameter, parasternal long-axis view (mm), median (IQR) | 48 (44–52) | 52 (46–59) | 57 (51–62) | <0.001 |
| LV end-systolic diameter, parasternal long-axis view (mm), median (IQR) | 31 (28–35) | 38 (32–42.5) | 45 (39–52) | <0.001 |
| LV end-diastolic volume, apical four-chamber view (mL), median (IQR) | 94 (78.25–119) | 116 (81–150) | 137 (102–177.5) | <0.001 |
| LV end-systolic volume, apical four-chamber view (mL), median (IQR) | 35 (27–48.5) | 59.5 (40–85) | 83 (59.5–128.5) | <0.001 |
| LV ejection fraction (%), Simpson method, median (IQR) | 62 (57–67) | 46 (42–48) | 34 (28–39) | <0.001 |
| Variable | OR | 95% CI | p-value |
|---|---|---|---|
| HAS-BLED score (per 1-point increase) | 1.419 | 1.120–1.797 | 0.004 |
| CHA₂DS₂-VA score (per 1-point increase) | 1.324 | 1.161–1.509 | <0.001 |
| HF duration (per 1-year increase) | 1.134 | 1.044–1.231 | 0.003 |
| Age (per 1-year increase) | 1.064 | 1.043–1.086 | <0.001 |
| Heart rate (per 1 bpm increase) | 1.022 | 1.011–1.033 | <0.001 |
| Serum creatinine (per 1 µmol/L increase) | 1.005 | 1.001–1.010 | 0.015 |
| Hemoglobin (per 1 g/L increase) | 0.988 | 0.979–0.997 | 0.010 |
| Office diastolic BP (per 1 mmHg increase) | 0.982 | 0.967–0.998 | 0.028 |
| Office systolic BP (per 1 mmHg increase) | 0.985 | 0.977–0.993 | <0.001 |
| eGFR (per 1 mL/min/1.73 m² increase) | 0.975 | 0.966–0.984 | <0.001 |
| Severe tricuspid regurgitation | 8.936 | 3.296–24.226 | <0.001 |
| Moderate tricuspid regurgitation | 4.516 | 2.263–9.009 | <0.001 |
| Moderate mitral regurgitation | 3.879 | 1.778–8.460 | 0.001 |
| Severe mitral regurgitation | 3.733 | 1.136–12.272 | 0.030 |
| Concomitant CKD | 3.097 | 1.332–7.199 | 0.009 |
| Moderate aortic regurgitation | 2.514 | 1.263–5.003 | 0.009 |
| Mild aortic regurgitation | 1.774 | 1.177–2.673 | 0.006 |
| LA size, parasternal long-axis view (per mm increase) | 1.123 | 1.088–1.159 | <0.001 |
| RA size, apical four-chamber view (per mm increase) | 1.073 | 1.038–1.109 | <0.001 |
| LA longitudinal size, apical four-chamber view (per mm increase) | 1.056 | 1.021–1.092 | 0.001 |
| Mitral inflow E-wave velocity (per 1 m/s increase) | 1.023 | 1.013–1.033 | <0.001 |
| Deceleration time of mitral E-wave (per 1-unit increase) | 0.994 | 0.990–0.999 | 0.013 |
| LV ejection fraction, Simpson method (per 1% increase) | 0.964 | 0.940–0.988 | 0.003 |
| Mineralocorticoid receptor antagonist therapy | 2.204 | 1.282–3.790 | 0.004 |
| Prior cardiac surgery | 2.167 | 1.295–3.626 | 0.003 |
| Angiotensin II receptor blocker therapy | 0.649 | 0.446–0.944 | 0.024 |
| Dihydropyridine calcium channel blocker therapy | 0.599 | 0.414–0.868 | 0.007 |
| Centrally acting antihypertensive therapy | 0.495 | 0.264–0.929 | 0.029 |
| Variable | Adjusted OR | 95% CI | p-value |
|---|---|---|---|
| Severe tricuspid regurgitation | 6.957 | 2.482–19.499 | <0.001 |
| Moderate tricuspid regurgitation | 4.092 | 1.977–8.466 | <0.001 |
| LA size, parasternal long-axis view (per mm increase) | 1.114 | 1.054–1.177 | <0.001 |
| Age (per 1-year increase) | 1.070 | 1.032–1.109 | <0.001 |
| Male sex | 1.680 | 1.076–2.621 | 0.022 |
| Heart rate (per 1 bpm increase) | 1.026 | 1.012–1.039 | <0.001 |
| Metabolic syndrome | 0.358 | 0.130–0.984 | 0.046 |
| eGFR (per 1 mL/min/1.73 m² increase) | 0.983 | 0.973–0.993 | 0.001 |
| Office systolic BP (per 1 mmHg increase) | 0.985 | 0.977–0.993 | <0.001 |
| Variable | OR | 95% CI | p-value |
|---|---|---|---|
| LA size, parasternal long-axis view (per mm increase) | 1.114 | 1.030–1.206 | 0.007 |
| Age (per 1-year increase) | 1.049 | 1.002–1.097 | 0.040 |
| Heart rate (per 1 bpm increase) | 1.030 | 1.005–1.055 | 0.019 |
| Variable | Adjusted OR | 95% CI | p-value |
|---|---|---|---|
| LA size, parasternal long-axis view (per mm increase) | 1.142 | 1.011–1.291 | 0.033 |
| Variable | OR | 95% CI | p-value |
|---|---|---|---|
| Posterior LV wall thickness, parasternal long-axis view (per mm increase) | 1.546 | 1.118–2.138 | 0.008 |
| Age (per 1-year increase) | 1.035 | 1.002–1.070 | 0.035 |
| Heart rate (per 1 bpm increase) | 1.032 | 1.009–1.056 | 0.006 |
| LV end-diastolic volume, apical four-chamber view (per 1 mL increase) | 0.986 | 0.975–0.998 | 0.020 |
| LV end-systolic volume, apical four-chamber view (per 1 mL increase) | 0.983 | 0.969–0.997 | 0.017 |
| Office diastolic BP (per 1 mmHg increase) | 0.952 | 0.917–0.987 | 0.009 |
| Variable | Adjusted OR | 95% CI | p-value |
|---|---|---|---|
| LV end-diastolic volume, apical four-chamber view (per 1 mL increase) | 0.981 | 0.964–0.998 | 0.033 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




