Submitted:
15 March 2026
Posted:
17 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Aims of the Study
- Data Quality Governance: To implement and validate a protocol that is rigorous for “AI-readiness” which is based on the FAIR principles (Findability, Accessibility, Interoperability, and Reusability) while also testing the hypothesis that prioritizing data quality over sheer data volume can yield better diagnostic accuracy, even with a small cohort.
- Integration of Validated Nomograms: To evaluate how much predictive weight is added by combining the Briganti nomogram and ISUP biopsy grades with an MLP architecture and to assess whether this combination is more effective than traditional clinical staging metrics.
- Model Optimization and Stability: To conduct a thorough sensitivity analysis that compares various data partitioning schemes which include 20/80, 34/66, and 39/61 to determine the best configuration for maximum classification accuracy and minimal cross-entropy error.
2. Materials and Methods
2.1. Study Population and Data Collection
2.1.1. Inclusion Criteria
- Histologically confirmed diagnosis of PCa.
- Complete clinical and biochemical records required for D’Amico risk stratification [58], including PSA at diagnosis, ISUP biopsy grade, and clinical TNM staging.
- Comprehensive mpMRI findings (mrT and mrN).
2.1.2. Exclusion Criteria
2.1.2.1. Methodological Note and Limitation
2.1.3. Data Quality and Integrity
- -
- Intrinsic Accuracy and Completeness: Clinical records were verified to make sure data were accurate, reliable, and free from errors.
- -
- Consistency and Mathematical Validity: Consistency means that the data is represented uniformly and there are no contradictions among sources. To keep the mathematical integrity of the Multilayer Perceptron or MLP, the analysis automatically excluded cases where factor levels or dependent variable values in the testing or reserved samples were not present in the training sample. This removal process helps maintain the mathematical validity of the MLP by preventing predictions on unobserved factor levels, but it also introduces some selection bias. By filtering out test cases that have rare categorical attributes (e.g., high-extremity PSA values or specific mrT stages not present in the training subset), the resulting performance metrics reflect the model’s efficacy on a “refined” holdout set instead of a completely unbiased population. Future iterations should utilize one-hot encoding for all categorical variables to allow the model can manage rare levels by using a generic “other” category or zero-weighting, which would ensure that the entire intended sample is assessed.
- -
- Alignment with FAIR Principles: The study adhered to the FAIR principles in order to improve transparency and accountability. Comprehensive documentation of exclusion criteria was maintained to ensure the traceability of the datasets and the credibility of the scientific conclusions.
2.1.4. Sample Size and Statistical Power
2.2. Artificial Neural Network (ANN) Configuration.
2.2.1. Network Architecture.
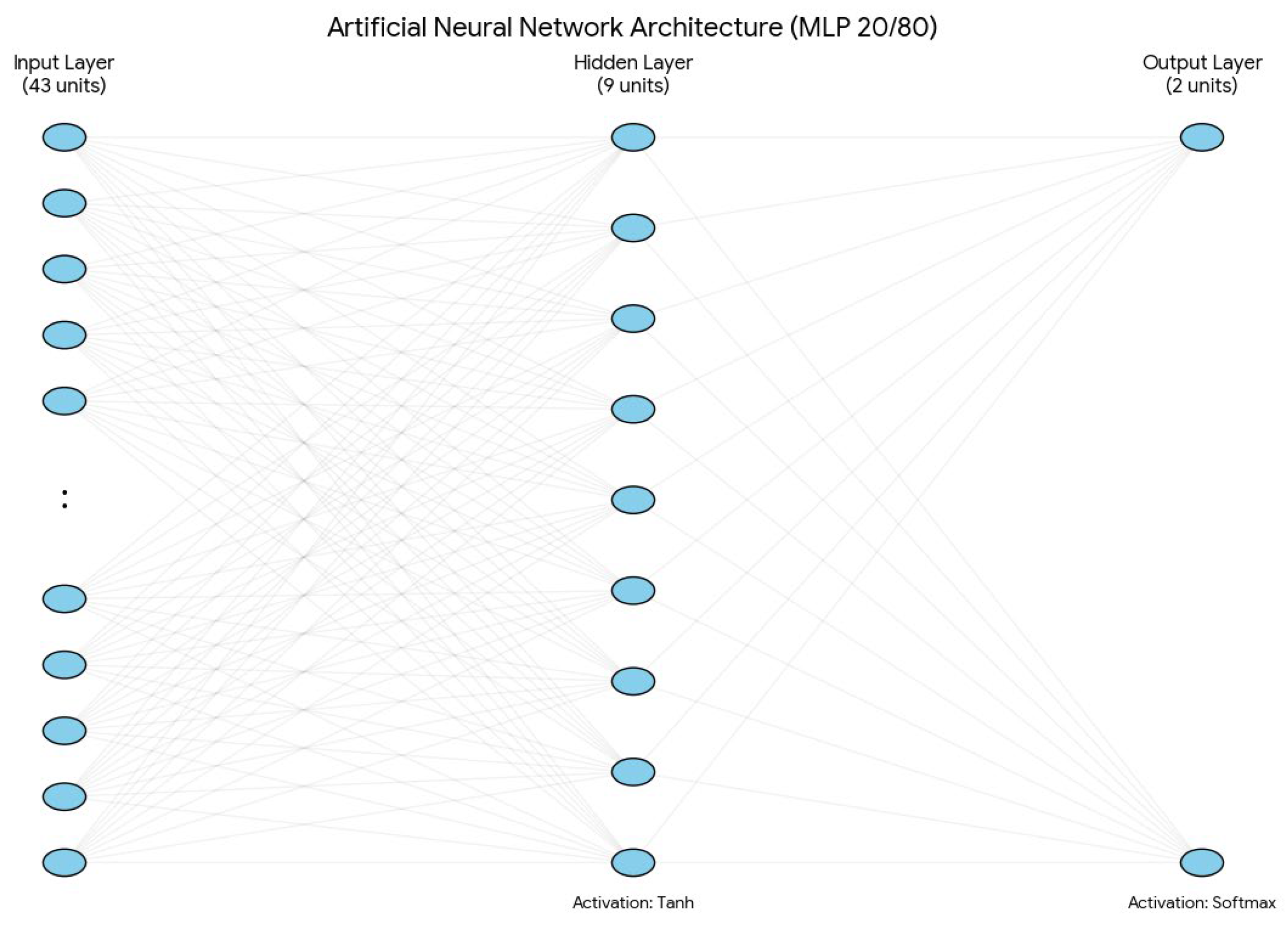
- Input Layer: The network architecture is built on 11 main clinical variables, and it has 43 input units in total for the 20/80 model. This expansion is performed automatically by the IBM SPSS MLP procedure using 1-of-c encoding for the categorical factors. There are seven categorical variables included in this process: PSA at diagnosis, ISUP biopsy grade, biopsy laterality, clinical TNM stage, clinical nodal stage, mrT, and mrN. Under the 1-of-c scheme, these factors are transformed into 39 separate input units (one for each category level). Combined with the four units for continuous covariates, this results in a total of 43 input units.
- For continuous covariates, there are four variables: age, PSA density, prostate volume, and Briganti score. These continuous variables undergo a rescaling procedure by linear normalization, which adjusts them to a standardized numerical range defined by the minimum and maximum values found in the training set. This pre-processing step is crucial, as it facilitates training convergence and prevents variables with larger numerical ranges, such as prostate volume, from disproportionately affecting the network’s weight estimations or causing “weight saturation” in the activation functions.
- Hidden Layer: A single hidden layer was used, with the number of neurons determined via the IBM SPSS MLP automatic architecture selection algorithm. This procedure optimized the size of the hidden layer within a predefined range from 6 to 9 by selecting the configuration that minimized the training cross-entropy error. This architectural constraint acts as a type of structural regularization which creates an ‘information bottleneck’ that prevents the network from memorizing the training set. By restricting the capacity of the hidden layer and combining these limits, the model is forced to prioritize the most influential predictors, such as the ISUP grade and Briganti score, over less significant categorical levels. Furthermore, to prevent ‘over-training,’ the model applied an early stopping rule that ended the iteration process at the first sign of error plateauing where the cross-entropy error did not to decrease anymore.
- Output Layer: The target variable was D’Amico Risk Group. Although the model was initialized to support three categories (High, Intermediate, and Low), the output neurons were dynamically reduced to two (High vs. Intermediate) in the 20/80 configuration. This occurred because the Low-risk group did not have enough representation in the training partition for that specific split, which did not allow for robust category initialization (Figure 1).
2.2.2. Sensitivity Analysis and Validation.
2.2.3. Model Robustness as an Extension of Data Quality Governance.
- Reproducible Initialization and Training: The framework requires the use of a fixed random seed (2,000,000) for all stochastic processes, including initial weight assignment and case selection for data partitions, to guarantee that all training runs can be precisely duplicated by independent researchers. This approach creates a repeatable baseline that can be used to compare subsequent experiments, even though it does not fully capture the range of the model’s stochastic behavior.
- Sensitivity Analysis as a Governance Mandate: The framework requires a sensitivity analysis across several partitioning schemes rather than depending on a single data split, which could yield results that are artifacts of a particularly favorable or unfavorable partition. Three different splits (20/80, 34/66, and 39/61) were assessed for this investigation. In order to determine whether the observed performance is a feature of the architecture’s interaction with high-quality data or just a reflection of a single, lucky patient grouping, this method examines the stability of the network’s learning across various cohort compositions and sizes.
- Reporting Distributions Rather Than Point Estimates: The approach requires that findings be reported as distributions (Mean ± SD) across the sensitivity analysis instead of single peak-performance percentages in order to account for the algorithmic variability inherent in small-sample machine learning. This approach gives readers a more accurate and comprehensive understanding of model stability.
- Early Stopping to Prioritize Generalization: An aggressive early stopping rule, which stops training after one consecutive step without reducing cross-entropy error, is specified by the framework. Given the limited cohort size, it is crucial to prioritize generalization over training-set accuracy, which is why this criterion was selected. Since this would have further lowered the already small training sample (from N=34 to an even smaller size) and impeded the model’s ability to identify stable decision boundaries, a separate validation set for early stopping was not used.
- Accounting for Exclusion Bias: Additionally, the DQG framework requires open reporting of how the final evaluable sample is impacted by its own rules. The valid sample size varied slightly between configurations (n=41 to n=44) because cases with factor levels not present in a particular training split were automatically excluded to maintain mathematical validity within the SPSS MLP framework. This must be stated clearly in the framework: the analysis should be seen as a test of the architecture’s capacity to generalize from a “standardized” clinical signal rather than a straightforward comparison across identical patient subsets.
- Uncertainty Quantification: The framework requires statistical testing against a null hypothesis of a random classifier (p = 0.50) in order to determine whether classification results might be attributed to random chance. Using IBM SPSS v26, a One-Sample Binomial Test was performed for this investigation. Confidence intervals were computed using the Clopper-Pearson exact method, which is the most rigorous and conservative method for small-sample validation, especially for the N=9 independent testing set.
- Benchmarking Against Traditional Methods: Lastly, the framework requires that the “AI-premium”, the neural network’s superior performance above conventional statistical methods, be quantified. Exact Logistic Regression in LogXact-11 [67], the statistical gold standard for small-sample datasets where standard maximum likelihood estimation may be incorrect, was used for this study’s baseline comparison. To enable a direct comparison with the MLP architecture, the baseline model employed the same 11 clinical predictors and the 20/80 data partitioning strategy.
3. Results
3.1. Cohort Curation and Model Performance as a Function of Data Quality
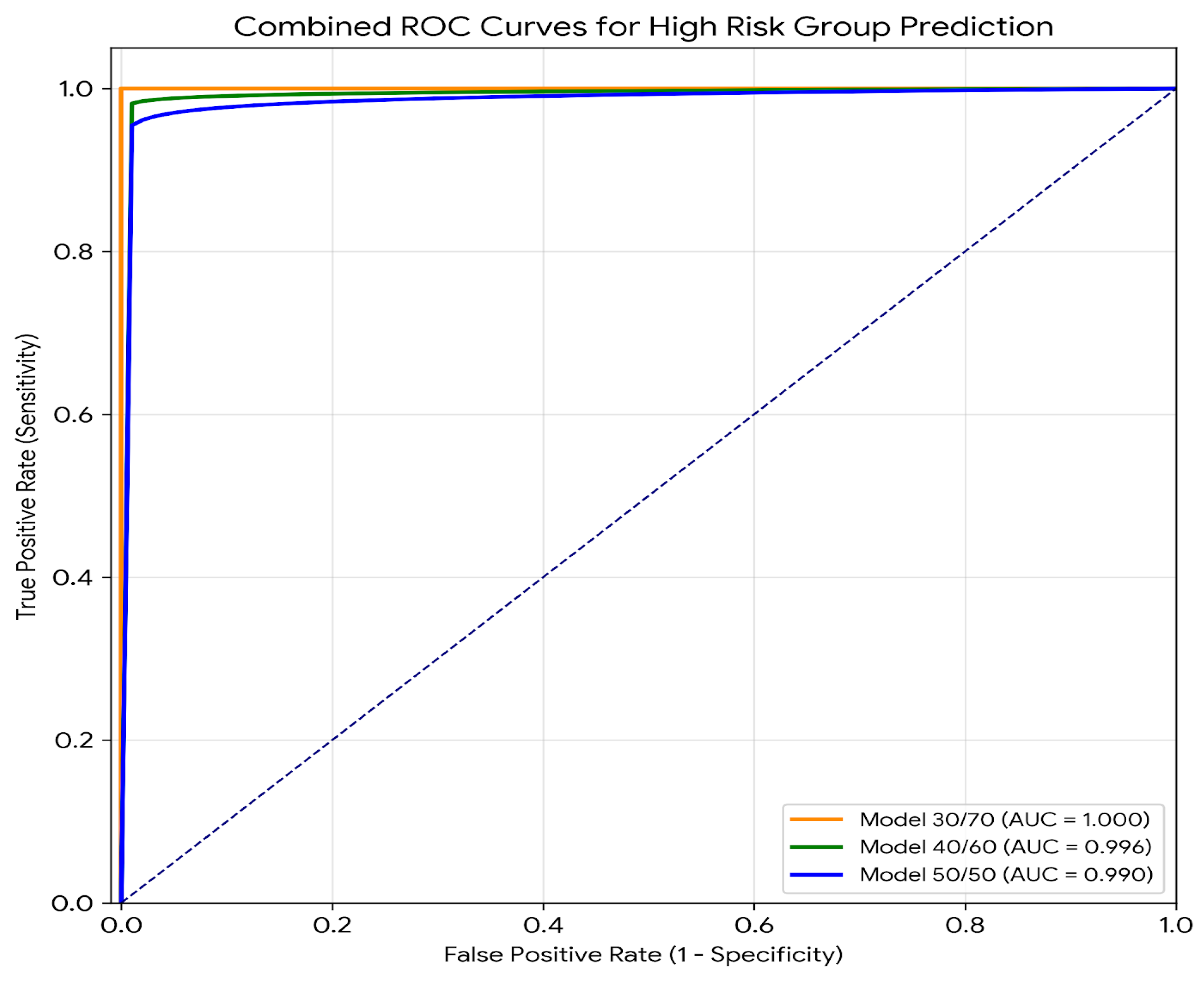
3.2. Discriminatory Capacity: Characterizing the Framework’s Extracted Signal
3.3. Optimal Model Classification: A Detailed View of the Framework’s Signal
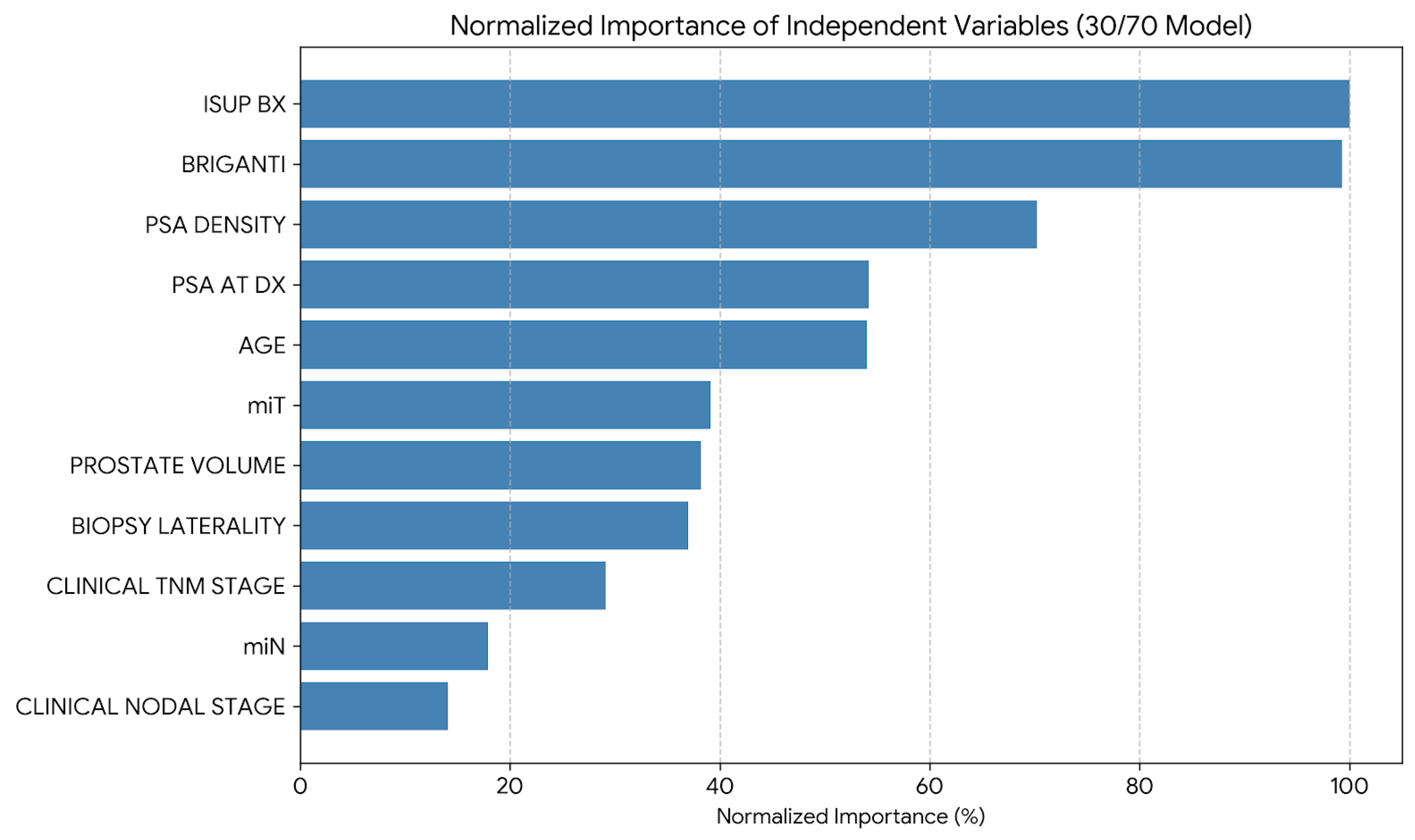
3.4. Independent Variable Importance: The Clinical Signal Preserved by the Framework
3.5. Comparative Benchmark: Evidence for Non-Linear Signal Preservation
4. Discussion
4.1. The Fragility of Perfect Metrics in a Small-Sample Context
4.2. Evidence for an "AI-Premium": Signal Detection or Overfitting?
4.3. Clinical Significance of Predictive Variables
4.4. Data Quality as a Strategic Imperative
4.5. Limitations and Future Directions
4.6. Clinical Translation: A Cautionary Framework, Not a Deployable Tool
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ANN | Artificial Neural Network |
| AUC | Area Under the Curve |
| cT | Clinical tumor stage |
| DQG | Data Quality Governance |
| EHR | Electronic Health Record |
| FAIR | Findability, Accessibility, Interoperability, and Reusability |
| ISUP | International Society of Urological Pathology |
| mrN | Multiparametric imaging nodal stage (or Imaging nodal stage) |
| mrT | Multiparametric imaging tumor stage (or Imaging tumor stage) |
| MLP | Multilayer Perceptron |
| mpMRI | Multiparametric Magnetic Resonance Imaging |
| NPV | Negative Predictive Value |
| PCa | Prostate Cancer |
| PMML | Predictive Model Markup Language |
| PPV | Positive Predictive Value |
| PSA | Prostate-Specific Antigen |
| ROC | Receiver Operating Characteristic |
| SD | Standard Deviation |
| SHAP | SHapley Additive exPlanations |
| SOP | Standard Operating Procedure |
| tanh | Hyperbolic Tangent |
| XML | Extensible Markup Language |
Appendix A: AI-Readiness and Data Quality Governance (DQG) Checklist Corresponding to the Study
References
- Schafer, E. J.; Laversanne, M.; Sung, H.; Soerjomataram, I.; Briganti, A.; Dahut, W.; Bray, F.; Jemal, A. Recent Patterns and Trends in Global Prostate Cancer Incidence and Mortality: An Update. Eur. Urol. 2025, 87(3), 302–313. [Google Scholar] [CrossRef]
- Wang, L.; Lu, B.; He, M.; Wang, Y.; Wang, Z.; Du, L. Prostate Cancer Incidence and Mortality: Global Status and Temporal Trends in 89 Countries From 2000 to 2019. Front. Public Health 2022, 10, 811044. [Google Scholar] [CrossRef]
- Chu, F.; Chen, L.; Guan, Q.; Chen, Z.; Ji, Q.; Ma, Y.; Ji, J.; Sun, M.; Huang, T.; Song, H.; Zhou, H.; Lin, X.; Zheng, Y. Global Burden of Prostate Cancer: Age-Period-Cohort Analysis from 1990 to 2021 and Projections until 2040. World J. Surg. Oncol. 2025, 23(1), 98. [Google Scholar] [CrossRef] [PubMed]
- Kratzer, T. B.; Mazzitelli, N.; Star, J.; Dahut, W. L.; Jemal, A.; Siegel, R. L. Prostate Cancer Statistics, 2025. CA Cancer J. Clin. 2025, 75(6), 485–497. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R. L.; Soerjomataram, I.; Jemal, A. Global Cancer Statistics 2022: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2024, 74(3), 229–263. [Google Scholar] [CrossRef] [PubMed]
- Cornford, P.; Tilki, D.; van den Berghn, R.; Eberlin, D. Prostate Cancer. EAU Guidelines; EAU Guidelines Office: Arnhem, 2025. [Google Scholar]
- Hassanipour-Azgomi, S.; Mohammadian-Hafshejani, A.; Ghoncheh, M.; Towhidi, F.; Jamehshorani, S.; Salehiniya, H. Incidence and Mortality of Prostate Cancer and Their Relationship with the Human Development Index Worldwide. Prostate Int. 2016, 4(3), 118–124. [Google Scholar] [CrossRef] [PubMed]
- Dudipala, H.; Jani, C. T.; Gurnani, S. D.; Morgenstern-Kaplan, D.; Tran, E.; Edwards, K.; Shalhoub, J.; McKay, R. R. Evolving Global Trends in Prostate Cancer: Disparities Across Income Levels and Geographic Regions (1990-2019). JCO Glob. Oncol. 2025, No. 11. [Google Scholar] [CrossRef]
- Huang, J.; Ssentongo, P.; Sharma, R. Editorial: Cancer Burden, Prevention and Treatment in Developing Countries. Front. Public Health 2023, 10. [Google Scholar] [CrossRef]
- Brand, N. R.; Qu, L. G.; Chao, A.; Ilbawi, A. M. Delays and Barriers to Cancer Care in Low- and Middle-Income Countries: A Systematic Review. Oncologist 2019, 24(12), e1371–e1380. [Google Scholar] [CrossRef]
- Daniels, J.; Mosadi, L. E.; Nyantakyi, A. Y.; Ayabilah, E. A.; Tackie, J. N. O.; Kyei, K. A. Metastatic Breast Cancer in Resource-Limited Settings: Insights from a Retrospective Cross-Sectional Study at a Radiotherapy Centre in Sub-Saharan Africa. Ecancermedicalscience 2025, 19. [Google Scholar] [CrossRef] [PubMed]
- Cazap, E.; Magrath, I.; Kingham, T. P.; Elzawawy, A. Structural Barriers to Diagnosis and Treatment of Cancer in Low- and Middle-Income Countries: The Urgent Need for Scaling Up. Journal of Clinical Oncology 2016, 34(1), 14–19. [Google Scholar] [CrossRef]
- Morsi, M. H.; Elawfi, B.; ALsaad, S. A.; Nazar, A.; Mostafa, H. A.; Awwad, S. A.; Abdelwahab, M. M.; Tarakhan, H.; Baghagho, E. Unveiling the Disparities in the Field of Precision Medicine: A Perspective. Health Sci. Rep. 2025, 8(8). [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Feng, X.; Hu, Y.; Yang, J.; Chen, Y.; Bastow, J.; Zheng, Z.-J.; Xu, M. Factors Associated with the Utilization of Diagnostic Tools among Countries with Different Income Levels during the COVID-19 Pandemic. Glob. Health Res. Policy 2023, 8(1), 45. [Google Scholar] [CrossRef] [PubMed]
- Fu, E.; Yager, P.; Floriano, P. N.; Christodoulides, N.; McDevitt, J. T. Perspective on Diagnostics for Global Health. IEEE Pulse 2011, 2(6), 40–50. [Google Scholar] [CrossRef]
- Hubley, J. H. Barriers to Health Education in Developing Countries. Health Educ. Res. 1986, 1(4), 233–245. [Google Scholar] [CrossRef]
- Olah, C.; Mairinger, F.; Wessolly, M.; Joniau, S.; Spahn, M.; Kruithof-de Julio, M.; Hadaschik, B.; Soós, A.; Nyirády, P.; Győrffy, B.; Reis, H.; Szarvas, T. Enhancing Risk Stratification Models in Localized Prostate Cancer by Novel Validated Tissue Biomarkers. Prostate Cancer Prostatic Dis. 2025, 28(3), 773–781. [Google Scholar] [CrossRef]
- D’Amico, AV.; Whittington, R.; Malkowicz, S.; Schulz, D.; Blank, K.; Broderick, G.; Tomaszewski, J.; Renshaw, A.; Kaplan, I.; Beard, C.; Wein, A. D’Amico risk classification for prostate cancer (Version: 1.28) - Evidencio. Available online: https://www.evidencio.com/models/show/300 (accessed on 2026 -02 -10).
- Hernandez, D. J.; Nielsen, M. E.; Han, M.; Partin, A. W. Contemporary Evaluation of the D’Amico Risk Classification of Prostate Cancer. Urology 2007, 70(5), 931–935. [Google Scholar] [CrossRef] [PubMed]
- Cooperberg, M. R. Clinical Risk Stratification for Prostate Cancer: Where Are We, and Where Do We Need to Go? Canadian Urological Association Journal 2017, 11(3–4), 101. [Google Scholar] [CrossRef] [PubMed]
- Rastinehad, A. R.; Baccala, A. A.; Chung, P. H.; Proano, J. M.; Kruecker, J.; Xu, S.; Locklin, J. K.; Turkbey, B.; Shih, J.; Bratslavsky, G.; Linehan, W. M.; Glossop, N. D.; Yan, P.; Kadoury, S.; Choyke, P. L.; Wood, B. J.; Pinto, P. A. D’Amico Risk Stratification Correlates With Degree of Suspicion of Prostate Cancer on Multiparametric Magnetic Resonance Imaging. Journal of Urology 2011, 185(3), 815–820. [Google Scholar] [CrossRef]
- Chierigo, F.; Flammia, R. S.; Sorce, G.; Hoeh, B.; Hohenhorst, L.; Tian, Z.; Saad, F.; Gallucci, M.; Briganti, A.; Montorsi, F.; Chun, F. K. H.; Graefen, M.; Shariat, S. F.; Guano, G.; Mantica, G.; Borghesi, M.; Suardi, N.; Terrone, C.; Karakiewicz, P. I. The Association of the Type and Number of D’Amico High-Risk Criteria with Rates of Pathologically Non-Organ-Confined Prostate Cancer. Cent. European J. Urol. 2023, 76(2), 104–108. [Google Scholar] [CrossRef]
- Coetzer, M. How AI Ready Data Strengthens Clinical Analytics and Outcomes Research. Available online: https://imatsolutions.com/index.php/2025/12/the-role-of-ai-ready-data-in-strengthening-clinical-research-predictive-modeling-and-care-quality/ (accessed on 2026 -02 -10).
- Domagalski, M. J.; Lu, Y.; Pilozzi, A.; Williamson, A.; Chilappagari, P.; Luker, E.; Shelley, C. D.; Dabic, A.; Keller, M. A.; Rodriguez, R. M.; Lawlor, S.; Thangudu, R. R. Preparing Clinical Research Data for Artificial Intelligence Readiness: Insights from the National Institute of Diabetes and Digestive and Kidney Diseases Data Centric Challenge. Journal of the American Medical Informatics Association 2025, 32(10), 1609–1616. [Google Scholar] [CrossRef]
- Bönisch, C.; Schmidt, C.; Kesztyüs, D.; Kestler, H. A.; Kesztyüs, T. Proposal for Using AI to Assess Clinical Data Integrity and Generate Metadata: Algorithm Development and Validation. JMIR Med. Inform. 2025, 13, e60204–e60204. [Google Scholar] [CrossRef]
- Wichard, J. D.; Cammann, H.; Stephan, C.; Tolxdorff, T. Classification Models for Early Detection of Prostate Cancer. Biomed Res. Int. 2008, 2008(1). [Google Scholar] [CrossRef]
- Chen, S.; Jian, T.; Chi, C.; Liang, Y.; Liang, X.; Yu, Y.; Jiang, F.; Lu, J. Machine Learning-Based Models Enhance the Prediction of Prostate Cancer. Front. Oncol. 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Elmarakeby, H. A.; Hwang, J.; Arafeh, R.; Crowdis, J.; Gang, S.; Liu, D.; AlDubayan, S. H.; Salari, K.; Kregel, S.; Richter, C.; Arnoff, T. E.; Park, J.; Hahn, W. C.; Van Allen, E. M. Biologically Informed Deep Neural Network for Prostate Cancer Discovery. Nature 2021, 598(7880), 348–352. [Google Scholar] [CrossRef] [PubMed]
- Shanei, A.; Etehadtavakol, M.; Azizian, M.; Ng, E. Y. K. Comparison of Different Kernels in a Support Vector Machine to Classify Prostate Cancerous Tissues in T2-Weighted Magnetic Resonance Imaging. Explor. Res. Hypothesis Med. 2022, 000(000), 000–000. [Google Scholar] [CrossRef]
- Chiu, P. K.-F.; Shen, X.; Wang, G.; Ho, C.-L.; Leung, C.-H.; Ng, C.-F.; Choi, K.-S.; Teoh, J. Y.-C. Enhancement of Prostate Cancer Diagnosis by Machine Learning Techniques: An Algorithm Development and Validation Study. Prostate Cancer Prostatic Dis. 2022, 25(4), 672–676. [Google Scholar] [CrossRef]
- Singh, S.; Pathak, A. K.; Kural, S.; Kumar, L.; Bhardwaj, M. G.; Yadav, M.; Trivedi, S.; Das, P.; Gupta, M.; Jain, G. Integrating MiRNA Profiling and Machine Learning for Improved Prostate Cancer Diagnosis. Sci. Rep. 2025, 15(1), 30477. [Google Scholar] [CrossRef]
- Jiang, M.; Miao, Z.; Xu, R.; Guo, M.; Li, X.; Li, G.; Luo, P.; Hu, S. Clinical-Radiomics Hybrid Modeling Outperforms Conventional Models: Machine Learning Enhances Stratification of Adverse Prognostic Features in Prostate Cancer. Front. Oncol. 2025, 15. [Google Scholar] [CrossRef] [PubMed]
- Zamo, F. C. D.; Ebongue, A. N.; Bongue, D.; Ndontchueng, M. M.; Njeh, C. F. Classification of PSQA Outcomes in Prostate VMAT Treatments: A Comparative Study of Machine Learning Models. Biomedical Engineering Advances 2026, 11, 100206. [Google Scholar] [CrossRef]
- Nieboer, D.; Vergouwe, Y.; Roobol, M. J.; Ankerst, D. P.; Kattan, M. W.; Vickers, A. J.; Steyerberg, E. W. Nonlinear Modeling Was Applied Thoughtfully for Risk Prediction: The Prostate Biopsy Collaborative Group. J. Clin. Epidemiol. 2015, 68(4), 426–434. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, L.; Zhang, S.; Li, J.; Shi, K.; Yao, D.; Li, Q.; Zhang, T.; Xu, L.; Geng, L.; Sun, Y.; Wan, J. Machine Learning-Based MRI Imaging for Prostate Cancer Diagnosis: Systematic Review and Meta-Analysis. Prostate Cancer Prostatic Dis. 2025. [Google Scholar] [CrossRef]
- Morote, J.; Miró, B.; Hernando, P.; Paesano, N.; Picola, N.; Muñoz-Rodriguez, J.; Ruiz-Plazas, X.; Muñoz-Rivero, M. V.; Celma, A.; García-de Manuel, G.; Servian, P.; Abascal, J. M.; Trilla, E.; Méndez, O. Developing a Predictive Model for Significant Prostate Cancer Detection in Prostatic Biopsies from Seven Clinical Variables: Is Machine Learning Superior to Logistic Regression? Cancers (Basel). 2025, 17(7), 1101. [Google Scholar] [CrossRef]
- Zhang, Y.-D.; Wang, J.; Wu, C.-J.; Bao, M.-L.; Li, H.; Wang, X.-N.; Tao, J.; Shi, H.-B. An Imaging-Based Approach Predicts Clinical Outcomes in Prostate Cancer through a Novel Support Vector Machine Classification. Oncotarget 2016, 7(47), 78140–78151. [Google Scholar] [CrossRef]
- Siech, C.; Wenzel, M.; Grosshans, N.; Cano Garcia, C.; Humke, C.; Koll, F. J.; Tian, Z.; Karakiewicz, P. I.; Kluth, L. A.; Chun, F. K. H.; Hoeh, B.; Mandel, P. The Association Between Lymphovascular or Perineural Invasion in Radical Prostatectomy Specimen and Biochemical Recurrence. Cancers (Basel). 2024, 16(21), 3648. [Google Scholar] [CrossRef] [PubMed]
- Chung, D. H.; Han, J. H.; Jeong, S.-H.; Yuk, H. D.; Jeong, C. W.; Ku, J. H.; Kwak, C. Role of Lymphatic Invasion in Predicting Biochemical Recurrence after Radical Prostatectomy. Front. Oncol. 2023, 13. [Google Scholar] [CrossRef] [PubMed]
- Fajkovic, H.; Mathieu, R.; Lucca, I.; Hiess, M.; Hübner, N.; Al Awamlh, B. A. H.; Lee, R.; Briganti, A.; Karakiewicz, P.; Lotan, Y.; Roupret, M.; Rink, M.; Kluth, L.; Loidl, W.; Seitz, C.; Klatte, T.; Kramer, G.; Susani, M.; Shariat, S. F. Validation of Lymphovascular Invasion Is an Independent Prognostic Factor for Biochemical Recurrence after Radical Prostatectomy. Urologic Oncology: Seminars and Original Investigations 2016, 34(5), 233.e1–233.e6. [Google Scholar] [CrossRef] [PubMed]
- Karwacki, J.; Stodolak, M.; Dłubak, A.; Nowak, Ł.; Gurwin, A.; Kowalczyk, K.; Kiełb, P.; Holdun, N.; Szlasa, W.; Krajewski, W.; Hałoń, A.; Karwacka, A.; Szydełko, T.; Małkiewicz, B. Association of Lymphovascular Invasion with Biochemical Recurrence and Adverse Pathological Characteristics of Prostate Cancer: A Systematic Review and Meta-Analysis. Eur. Urol. Open Sci. 2024, 69, 112–126. [Google Scholar] [CrossRef]
- Schmidhuber, J. Deep Learning in Neural Networks: An Overview. Neural Networks 2015, 61, 85–117. [Google Scholar] [CrossRef] [PubMed]
- Litjens, G.; Kooi, T.; Bejnordi, B. E.; Setio, A. A. A.; Ciompi, F.; Ghafoorian, M.; van der Laak, J. A. W. M.; van Ginneken, B.; Sánchez, C. I. A Survey on Deep Learning in Medical Image Analysis. Med. Image Anal. 2017, 42, 60–88. [Google Scholar] [CrossRef]
- Aggarwal, R.; Sounderajah, V.; Martin, G.; Ting, D. S. W.; Karthikesalingam, A.; King, D.; Ashrafian, H.; Darzi, A. Diagnostic Accuracy of Deep Learning in Medical Imaging: A Systematic Review and Meta-Analysis. NPJ Digit. Med. 2021, 4(1), 65. [Google Scholar] [CrossRef] [PubMed]
- Erdem, E.; Bozkurt, F. A Comparison of Various Supervised Machine Learning Techniques for Prostate Cancer Prediction. European Journal of Science and Technology 2021, No. 21, 610–620. [Google Scholar] [CrossRef]
- Shu, X.; Liu, Y.; Qiao, X.; Ai, G.; Liu, L.; Liao, J.; Deng, Z.; He, X. Radiomic-Based Machine Learning Model for the Accurate Prediction of Prostate Cancer Risk Stratification. Br. J. Radiol. 2023, 96(1143). [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Kumar, M. Prostate Cancer Prognosis Using Multi-Layer Perceptron and Class Balancing Techniques. In 2021 Thirteenth International Conference on Contemporary Computing (IC3-2021); ACM: New York, NY, USA, 2021; pp. 1–6. [Google Scholar] [CrossRef]
- Yang, T.; Zhang, H.; Peng, H.; Niu, X.; Yang, F.; Zhang, J.; Wang, Q.; Fan, J.; Song, Y.; Tao, W. PSMA PET/MRI-based Swin Transformer Architecture for Gleason Score Prediction in Prostate Cancer. Med. Phys. 2026, 53(1). [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.; Hulsey, G.; James, K.; James, T.; Carlson, J. M. Deep Learning Classification of Prostate Cancer Using MRI Histopathologic Data. Radiol. Imaging Cancer 2025, 7(5). [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Han, X.; Gao, J.; Zhang, Q.; Yang, H.; Liao, S.; Guo, H.; Zhang, B. Deep Learning in Prostate Cancer Diagnosis Using Multiparametric Magnetic Resonance Imaging With Whole-Mount Histopathology Referenced Delineations. Front. Med. (Lausanne). 2022, 8. [Google Scholar] [CrossRef]
- He, M.; Cao, Y.; Chi, C.; Yang, X.; Ramin, R.; Wang, S.; Yang, G.; Mukhtorov, O.; Zhang, L.; Kazantsev, A.; Enikeev, M.; Hu, K. Research Progress on Deep Learning in Magnetic Resonance Imaging–Based Diagnosis and Treatment of Prostate Cancer: A Review on the Current Status and Perspectives. Front. Oncol. 2023, 13. [Google Scholar] [CrossRef]
- Quarta, L.; Stabile, A.; Pellegrino, F.; Scilipoti, P.; Longoni, M.; Cannoletta, D.; Zaurito, P.; Santangelo, A.; Viti, A.; Barletta, F.; Scuderi, S.; Leni, R.; Pellegrino, A.; Mazzone, E.; Nocera, L.; Brembilla, G.; De Cobelli, F.; Karnes, R. J.; Rouprêt, M.; Montorsi, F.; Gandaglia, G.; Briganti, A. Tailored Use of PSA Density According to Multiparametric MRI Index Lesion Location: Results of a Large, Multi-Institutional Series. Prostate Cancer Prostatic Dis. 2025. [Google Scholar] [CrossRef]
- Ciobanu-Caraus, O.; Aicher, A.; Kernbach, J. M.; Regli, L.; Serra, C.; Staartjes, V. E. A Critical Moment in Machine Learning in Medicine: On Reproducible and Interpretable Learning. Acta Neurochir. (Wien). 2024, 166(1), 14. [Google Scholar] [CrossRef]
- McDermott, M. B. A.; Wang, S.; Marinsek, N.; Ranganath, R.; Foschini, L.; Ghassemi, M. Reproducibility in Machine Learning for Health Research: Still a Ways to Go. Sci. Transl. Med. 2021, 13(586). [Google Scholar] [CrossRef]
- Guillen-Aguinaga, M.; Aguinaga-Ontoso, E.; Guillen-Aguinaga, L.; Guillen-Grima, F.; Aguinaga-Ontoso, I. Data Quality in the Age of AI: A Review of Governance, Ethics, and the FAIR Principles. Data (Basel). 2025, 10(12), 201. [Google Scholar] [CrossRef]
- Clark, T.; Caufield, H.; Parker, J. A.; Al Manir, S.; Amorim, E.; Eddy, J.; Gim, N.; Gow, B.; Goar, W.; Haendel, M.; Hansen, J. N.; Harris, N.; Hermjakob, H.; Joachimiak, M.; Jordan, G.; Lee, I.-H.; K. McWeeney, S.; Nebeker, C.; Nikolov, M.; Shaffer, J.; Sheffield, N.; Sheynkman, G.; Stevenson, J.; Chen, J. Y.; Mungall, C.; Wagner, A.; Kong, S. W.; Ghosh, S. S.; Patel, B.; Williams, A.; Munoz-Torres, M. C. AI-Readiness for Biomedical Data: Bridge2AI Recommendations. October 25, 2024. [CrossRef]
- Aksenova, A.; Johny, A.; Adams, T.; Gribbon, P.; Jacobs, M.; Hofmann-Apitius, M. Current State of Data Stewardship Tools in Life Science. Front. Big Data 2024, 7. [Google Scholar] [CrossRef] [PubMed]
- D’Amico, A. V. Biochemical Outcome After Radical Prostatectomy, External Beam Radiation Therapy, or Interstitial Radiation Therapy for Clinically Localized Prostate Cancer. JAMA 1998, 280(11), 969. [Google Scholar] [CrossRef] [PubMed]
- Diamand, R.; Oderda, M.; Albisinni, S.; Fourcade, A.; Fournier, G.; Benamran, D.; Iselin, C.; Fiard, G.; Descotes, J.-L.; Assenmacher, G.; Svistakov, I.; Peltier, A.; Simone, G.; Di Cosmo, G.; Roche, J.-B.; Bonnal, J.-L.; Van Damme, J.; Rossi, M.; Mandron, E.; Gontero, P.; Roumeguère, T. External Validation of the Briganti Nomogram Predicting Lymph Node Invasion in Patients with Intermediate and High-Risk Prostate Cancer Diagnosed with Magnetic Resonance Imaging-Targeted and Systematic Biopsies: A European Multicenter Study. Urologic Oncology: Seminars and Original Investigations 2020, 38(11), 847.e9–847.e16. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.; Rink, M.; Bianchi, M.; Kluth, L. A.; Tian, Z.; Ahyai, S. A.; Shariat, S. F.; Briganti, A.; Steuber, T.; Fisch, M.; Graefen, M.; Karakiewicz, P. I.; Chun, F. K. -H. External Validation of the Updated Briganti Nomogram to Predict Lymph Node Invasion in Prostate Cancer Patients Undergoing Extended Lymph Node Dissection. Prostate 2013, 73(2), 211–218. [Google Scholar] [CrossRef]
- Małkiewicz, B.; Ptaszkowski, K.; Knecht, K.; Gurwin, A.; Wilk, K.; Kiełb, P.; Dudek, K.; Zdrojowy, R. External Validation of the Briganti Nomogram to Predict Lymph Node Invasion in Prostate Cancer—Setting a New Threshold Value. 2021, 11(6), 479. [Google Scholar] [CrossRef]
- International Organization for Standardization. ISO/IEC 42001:2023 Information Technology — Artificial Intelligence — Management System; Geneva, 2023.
- Wang, R. Y.; Strong, D. M. Beyond Accuracy: What Data Quality Means to Data Consumers. Journal of Management Information Systems 1996, 12(4), 5–33. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.-G. Statistical Power Analyses Using G*Power 3.1: Tests for Correlation and Regression Analyses. Behav. Res. Methods 2009, 41(4), 1149–1160. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.-G.; Buchner, A. G*Power 3: A Flexible Statistical Power Analysis Program for the Social, Behavioral, and Biomedical Sciences. Behav. Res. Methods 2007, 39(2), 175–191. [Google Scholar] [CrossRef]
- Soe, MM.; Dean AG, S. K. M. OpenEpi: Open Source Epidemiologic Statistics for Public Health, Versión. 2013. [Google Scholar]
- Cytel Inc. LogXact-11: Software for Exact Logistic Regression, A, 2015; Cytel Inc: Waltham, MA, US, 2015. [Google Scholar]
- Sur, P.; Candès, E. J. A Modern Maximum-Likelihood Theory for High-Dimensional Logistic Regression. Proceedings of the National Academy of Sciences 2019, 116(29), 14516–14525. [Google Scholar] [CrossRef] [PubMed]
- Yee, T. W. On the Hauck-Donner Effect in Wald Tests: Detection, Tipping Points, and Parameter Space Characterization. 2022. [Google Scholar] [CrossRef]
- Hauck, W. W.; Donner, A. Wald’s Test as Applied to Hypotheses in Logit Analysis. J. Am. Stat. Assoc. 1977, 72(360a), 851–853. [Google Scholar] [CrossRef]
- Van Calster, B.; Collins, G. S.; Vickers, A. J.; Wynants, L.; Kerr, K. F.; Barreñada, L.; Varoquaux, G.; Singh, K.; Moons, K. G.; Hernandez-Boussard, T.; Timmerman, D.; McLernon, D. J.; van Smeden, M.; Steyerberg, E. W. Evaluation of Performance Measures in Predictive Artificial Intelligence Models to Support Medical Decisions: Overview and Guidance. Lancet Digit. Health 2025, 7(12), 100916. [Google Scholar] [CrossRef]
- Roberts, M.; Driggs, D.; Thorpe, M.; Gilbey, J.; Yeung, M.; Ursprung, S.; Aviles-Rivero, A. I.; Etmann, C.; McCague, C.; Beer, L.; Weir-McCall, J. R.; Teng, Z.; Gkrania-Klotsas, E.; Ruggiero, A.; Korhonen, A.; Jefferson, E.; Ako, E.; Langs, G.; Gozaliasl, G.; Yang, G.; Prosch, H.; Preller, J.; Stanczuk, J.; Tang, J.; Hofmanninger, J.; Babar, J.; Sánchez, L. E.; Thillai, M.; Gonzalez, P. M.; Teare, P.; Zhu, X.; Patel, M.; Cafolla, C.; Azadbakht, H.; Jacob, J.; Lowe, J.; Zhang, K.; Bradley, K.; Wassin, M.; Holzer, M.; Ji, K.; Ortet, M. D.; Ai, T.; Walton, N.; Lio, P.; Stranks, S.; Shadbahr, T.; Lin, W.; Zha, Y.; Niu, Z.; Rudd, J. H. F.; Sala, E.; Schönlieb, C.-B. Common Pitfalls and Recommendations for Using Machine Learning to Detect and Prognosticate for COVID-19 Using Chest Radiographs and CT Scans. Nat. Mach. Intell. 2021, 3(3), 199–217. [Google Scholar] [CrossRef]
- Varoquaux, G.; Cheplygina, V. Machine Learning for Medical Imaging: Methodological Failures and Recommendations for the Future. NPJ Digit. Med. 2022, 5(1), 48. [Google Scholar] [CrossRef]
- Yu, A. C.; Mohajer, B.; Eng, J. External Validation of Deep Learning Algorithms for Radiologic Diagnosis: A Systematic Review. Radiol. Artif. Intell. 2022, 4(3). [Google Scholar] [CrossRef] [PubMed]
- Arun, S.; Grosheva, M.; Kosenko, M.; Robertus, J. L.; Blyuss, O.; Gabe, R.; Munblit, D.; Offman, J. Systematic Scoping Review of External Validation Studies of AI Pathology Models for Lung Cancer Diagnosis. NPJ Precis. Oncol. 2025, 9(1), 166. [Google Scholar] [CrossRef] [PubMed]
- Tseng, A. S.; Shelly-Cohen, M.; Attia, I. Z.; Noseworthy, P. A.; Friedman, P. A.; Oh, J. K.; Lopez-Jimenez, F. Spectrum Bias in Algorithms Derived by Artificial Intelligence: A Case Study in Detecting Aortic Stenosis Using Electrocardiograms. European Heart Journal - Digital Health 2021, 2(4), 561–567. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Gao, X.-S.; Zhang, L.; Miao, Y. Generalizability of Memorization Neural Networks; 2024. [Google Scholar]
- Zhang, C.; Bengio, S.; Hardt, M.; Mozer, M. C.; Singer, Y. Identity Crisis: Memorization and Generalization under Extreme Overparameterization; 2020. [Google Scholar]
- Belkin, M.; Hsu, D.; Ma, S.; Mandal, S. Reconciling Modern Machine-Learning Practice and the Classical Bias–Variance Trade-Off. Proceedings of the National Academy of Sciences 2019, 116(32), 15849–15854. [Google Scholar] [CrossRef] [PubMed]
- Hastie, T.; Tibshiran, R.; Friedman, J. The Elements of Statistical Learning: Data Mining, Inference, and Prediction, 2nd ed.; Springer: New York, 2017. [Google Scholar]
- Crespo Márquez, A. The Curse of Dimensionality. In Digital Maintenance Management; Springer Science and Business Media Deutschland GmbH: New York, 2022; pp 67–86. [CrossRef]
- Tishby, N.; Zaslavsky, N. Deep Learning and the Information Bottleneck Principle. In 2015 IEEE Information Theory Workshop (ITW); IEEE, 2015; pp 1–5. [CrossRef]
- Koch, R. de M.; Ghosh, A. Two-Phase Perspective on Deep Learning Dynamics. Phys. Rev. E 2025, 112(2), 025307. [Google Scholar] [CrossRef]
- Chapman, B. P.; Weiss, A.; Duberstein, P. R. Statistical Learning Theory for High Dimensional Prediction: Application to Criterion-Keyed Scale Development. Psychol. Methods 2016, 21(4), 603–620. [Google Scholar] [CrossRef]
- Yao, Y.; Rosasco, L.; Caponnetto, A. On Early Stopping in Gradient Descent Learning. Constr. Approx. 2007, 26(2), 289–315. [Google Scholar] [CrossRef]
- Lee, F. What is Bias-Variance Tradeoff? Available online: https://www.ibm.com/think/topics/bias-variance-tradeoff (accessed on 2026 -02 -15).
- Hu, Y.; Zhang, X.; Slavin, V.; Belsti, Y.; Tiruneh, S. A.; Callander, E.; Enticott, J. Beyond Comparing Machine Learning and Logistic Regression in Clinical Prediction Modelling: Shifting from Model Debate to Data Quality. J. Med. Internet Res. 2025, 27, e77721. [Google Scholar] [CrossRef] [PubMed]
- Condon, D. Performance of Artificial Neural Networks on Small Structured Datasets. 2019. [Google Scholar]
- Bailly, A.; Blanc, C.; Francis, É.; Guillotin, T.; Jamal, F.; Wakim, B.; Roy, P. Effects of Dataset Size and Interactions on the Prediction Performance of Logistic Regression and Deep Learning Models. Comput. Methods Programs Biomed. 2022, 213, 106504. [Google Scholar] [CrossRef] [PubMed]
- Issitt, R. W.; Cortina-Borja, M.; Bryant, W.; Bowyer, S.; Taylor, A. M.; Sebire, N. Classification Performance of Neural Networks Versus Logistic Regression Models: Evidence From Healthcare Practice. Cureus 2022. [Google Scholar] [CrossRef]
- Nguyen, T.; Nguyen, H.-T.; Nguyen-Hoang, T.-A. Data Quality Management in Big Data: Strategies, Tools, and Educational Implications. J. Parallel Distrib. Comput. 2025, 200, 105067. [Google Scholar] [CrossRef]
- Liu, J.; Zhao, J.; Zhang, M.; Chen, N.; Sun, G.; Yang, Y.; Zhang, X.; Chen, J.; Shen, P.; Shi, M.; Zeng, H. The Validation of the 2014 International Society of Urological Pathology (ISUP) Grading System for Patients with High-Risk Prostate Cancer: A Single-Center Retrospective Study. Cancer Manag. Res. 2019, Volume 11, 6521–6529. [Google Scholar] [CrossRef]
- Offermann, A.; Hupe, M. C.; Sailer, V.; Merseburger, A. S.; Perner, S. The New ISUP 2014/WHO 2016 Prostate Cancer Grade Group System: First Résumé 5 Years after Introduction and Systemic Review of the Literature. World J. Urol. 2020, 38(3), 657–662. [Google Scholar] [CrossRef] [PubMed]
- Epstein, J. I.; Egevad, L.; Amin, M. B.; Delahunt, B.; Srigley, J. R.; Humphrey, P. A. The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma. American Journal of Surgical Pathology 2016, 40(2), 244–252. [Google Scholar] [CrossRef] [PubMed]
- Barberis, A.; Aerts, H. J. W. L.; Buffa, F. M. Robustness and Reproducibility for AI Learning in Biomedical Sciences: RENOIR. Sci. Rep. 2024, 14(1), 1933. [Google Scholar] [CrossRef] [PubMed]
- Colliot, O.; Thibeau-Sutre, E.; Burgos, N. Reproducibility in Machine Learning for Medical Imaging. In Machine Learning for Brain Disorders; Colliot, O., Ed.; Humana, 2023; Vol. 197, pp 631–653. [CrossRef]
- Beam, A. L.; Manrai, A. K.; Ghassemi, M. Challenges to the Reproducibility of Machine Learning Models in Health Care. JAMA 2020, 323(4), 305. [Google Scholar] [CrossRef] [PubMed]
| Quality Dimension | Metric / Target | Operational Validation Rule (Concrete Check) |
|---|---|---|
| Accuracy | 100% Clinical Concordance | Cross-verification of PSA values and ISUP grades between the Electronic Health Record (EHR) and the study database. |
| Completeness | 0% Missingness in Predictors | Exclusion of any case with missing values in the 11 primary clinical variables (Listwise deletion). |
| Validity (Range) | Biological Boundary Checks | PSA: (0.1 to 500 ng/mL); Prostate Volume: (10 to 300 cc); Age: (40 to 90 years). |
| Consistency | Logical Relationship | Staging consistency check: Clinical stage (cT) must not exceed pathological or imaging (mrT) findings in illogical sequences. |
| Integrity | Referential Integrity | All categorical factors must map to the D’Amico classification standards (ISUP 1–5). |
| AI-Readiness | Feature Scaling | Continuous variables must be normalized to a standard numerical range to prevent gradient saturation. |
| Partitioning Scheme | Total Sample | Valid Cases (n) | Excluded Cases (n) | Exclusion Rate (%) | Primary Reason for Exclusion |
|---|---|---|---|---|---|
| Model 20/80 | 49 | 43 | 6 | 12.20% | Factor levels (e.g., PSA values or mrT stages) not present in the training set. |
| Model 34/66 | 49 | 44 | 5 | 10.20% | Factor levels or dependent variable values (Low-risk strata) not present in training. |
| Model 39/61 | 49 | 41 | 8 | 16.30% | Factor levels (clinical outliers) not represented in the training sample. |
| Metric | Model 20/80 | Model 34/66 | Model 39/61 |
|---|---|---|---|
| D’Amico Strata Evaluated | Binary (High/Int) | Ternary (High/Int/Low) | Binary (High/Int) |
| Training Sample n (%) | 34 (79.10%) | 29 (65.90%) | 25 (61.00%) |
| Testing Sample n (%) | 9 (20.9%) | 15 (34.1%) | 16 (39.0%) |
| Training Cross-Entropy Error | 0.161 | 3.842 | 0.209 |
| Testing Cross-Entropy Error | 0.001 | 4.227 | 4.636 |
| Training Incorrect Predictions (%) | 0.00% | 6.90% | 0.00% |
| Testing Incorrect Predictions (%) | 0.00% | 13.30% | 6.30% |
| Overall Training Accuracy (%) | 100% | 93.10% | 100.00% |
| Overall Testing Accuracy (%) | 100% (95% CI: 66.4–100) † |
86.70% (95% CI: 62.1–96.3) |
93.80% (95% CI: 71.7–98.9) |
| Correct Classifications (n/N) | (9/9) | (13/15) | (15/16) |
| Sensitivity (High Risk) | 100% (95% CI: 56.5–100) |
85.7% (95% CI: 48.7–97.4 ) |
87.5% (95% CI: 52.9–97.8) |
| Specificity (Int. Risk) | 100% (95% CI: 51.0–100) |
87.5% (95% CI: 52.9–97.8) |
100% (95% CI: 67.6–100) |
| PPV (Positive Predictive Value) | 100% (95% CI: 56.5–100) |
85.7% (95% CI: 48.7–97.4) |
100% (95% CI: 64.6–100) |
| NPV (Negative Predictive Value) | 100% (95% CI: 51.0–100) |
87.5% (95% CI: 52.9–97.8) |
88.9% (95% CI: 56.5–98.0) |
| Risk Group | AUC Model 20/80 | AUC Model 34/66 |
AUC Model 39/61 |
|---|---|---|---|
| High | 1 | 0.996 | 0.99 |
| Intermediate | 1 | 0.991 | 0.99 |
| Low | N/A | 1 | N/A |
| Sample | Observed Risk Group | Predicted: High | Predicted: Intermediate |
Percent Correct |
|---|---|---|---|---|
| Training | High | 22 | 0 | 100% |
| Intermediate | 0 | 12 | 100% | |
| Testing | High | 5 | 0 | 100% |
| Intermediate | 0 | 4 | 100% | |
| Global Percentage | 55.60% | 44.40% | 100% |
| Variable | Importance | Normalized Importance |
|---|---|---|
| ISUP BX | 0.181 | 100.00% |
| BRIGANTI | 0.180 | 99.30% |
| PSA DENSITY | 0.127 | 70.20% |
| PSA at DX | 0.098 | 54.20% |
| AGE | 0.098 | 54.00% |
| mrT | 0.071 | 39.10% |
| PROSTATE VOLUME c.c. | 0.069 | 38.20% |
| BX LATERALITY | 0.067 | 37.00% |
| Clinical TNM Stage | 0.053 | 29.10% |
| mrN | 0.032 | 17.90% |
| Nodal Stage | 0.025 | 14.10% |
| Feature / Metric | Exact Logistic Regression (Baseline) |
Multilayer Perceptron (MLP 20/80) |
|---|---|---|
| Statistical Engine | Exact Likelihood Estimation (LogXact) | Backpropagation / Softmax (SPSS) |
| Model Type | Linear / Parametric | Non-linear / Connectionist +1 |
| Overall Significance | p<0.001 (Likelihood Ratio) | p=0.002 (Exact Binomial Test) |
| Predictor: ISUP Grade | Significant (p=0.005) | Dominant (100% Importance) |
| Predictor: Briganti Nomogram | Not Significant (p=0.800) | Critical (99.3% Importance) |
| Predictor: PSA Density | Not Significant (p=0.500) | High Impact (70.2% Importance) |
| Predictor: Age | Degenerate Estimate (DEGEN)† | Moderate Impact (54.0% Importance) |
| Testing Accuracy | Outperformed by MLP | 100.00% |
| Testing AUC | < 1.000 | 1 |
| Clinical Interpretation | Limited to linear histological signal. | Captures non-linear feature synergies. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




