Submitted:
12 March 2026
Posted:
16 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design, Setting, and Ethics
2.2. Participants
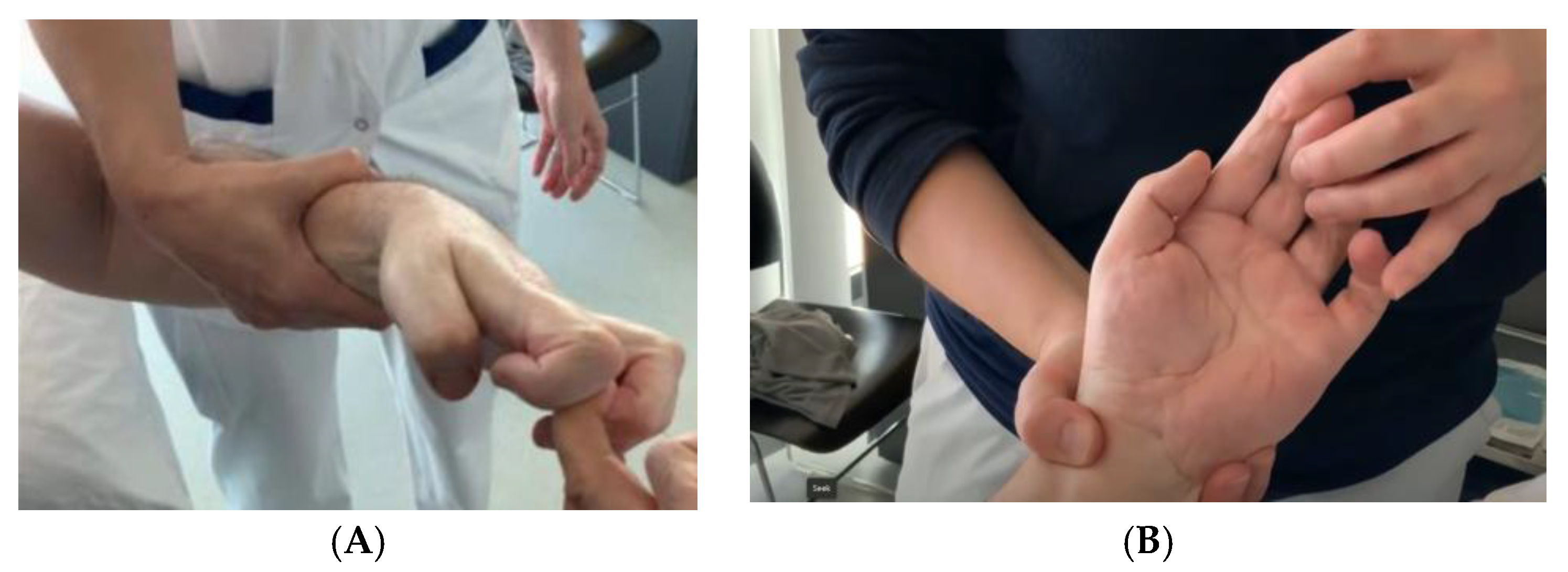
2.3. Pre-Procedure Diagnostic Nerve Block (Target Confirmation)
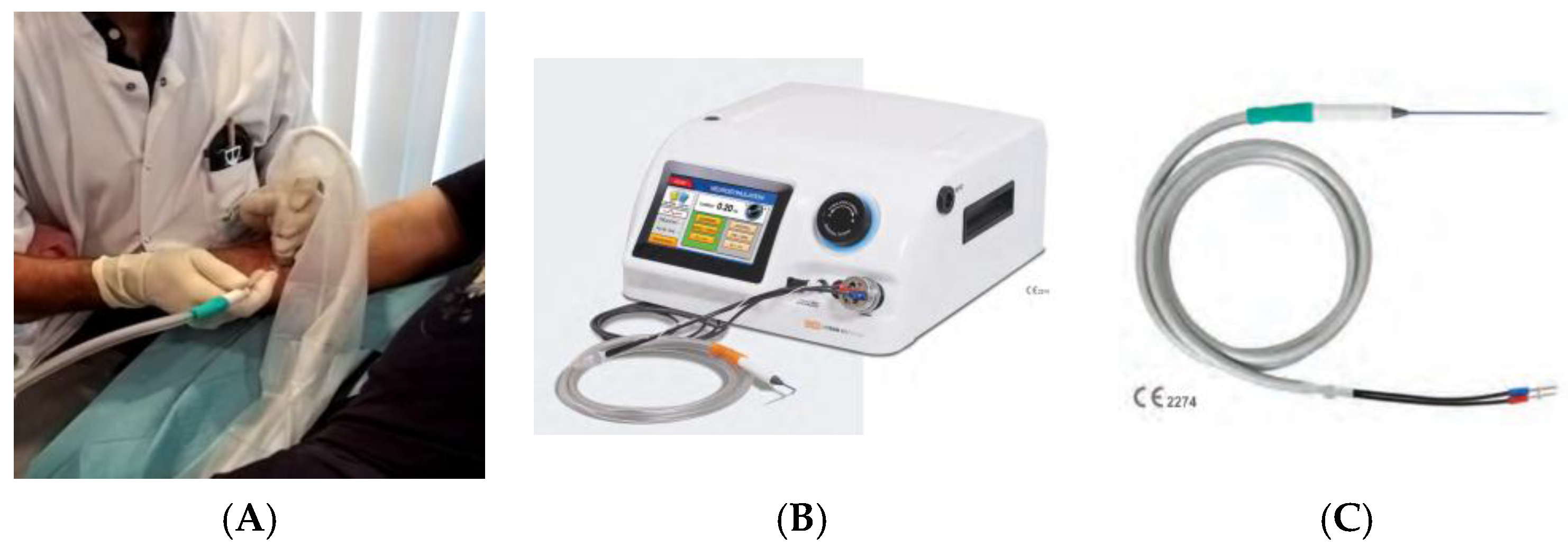
2.4. Cryoneurolysis Procedure
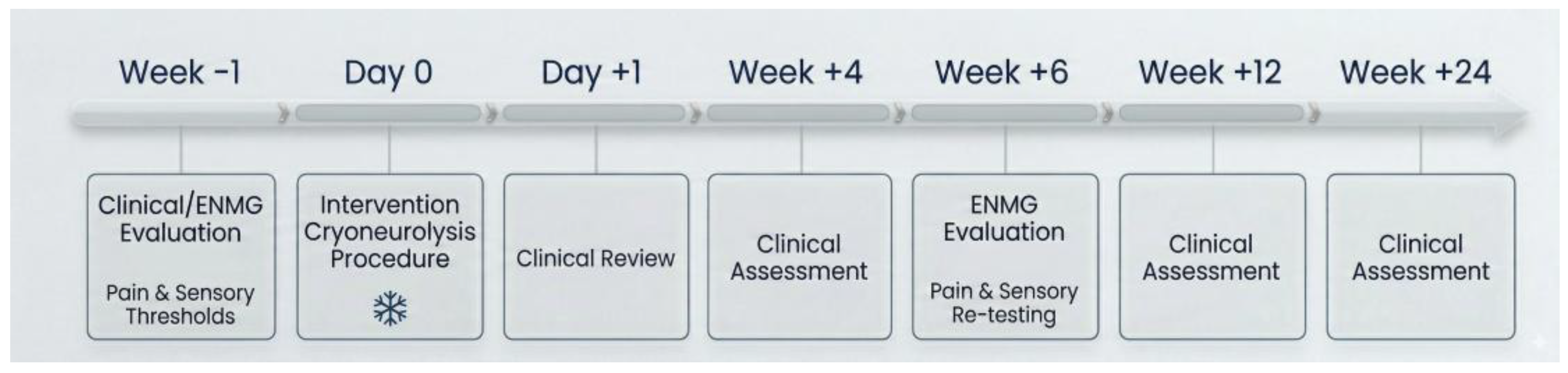
2.5. Outcomes and Follow-Up Schedule
2.6. Pain and Sensory Testing
2.7. Electroneuromyography
2.8. Statistical Analysis
3. Results
3.1. Participant Characteristics and Targets
3.2. Feasibility and Safety Observations
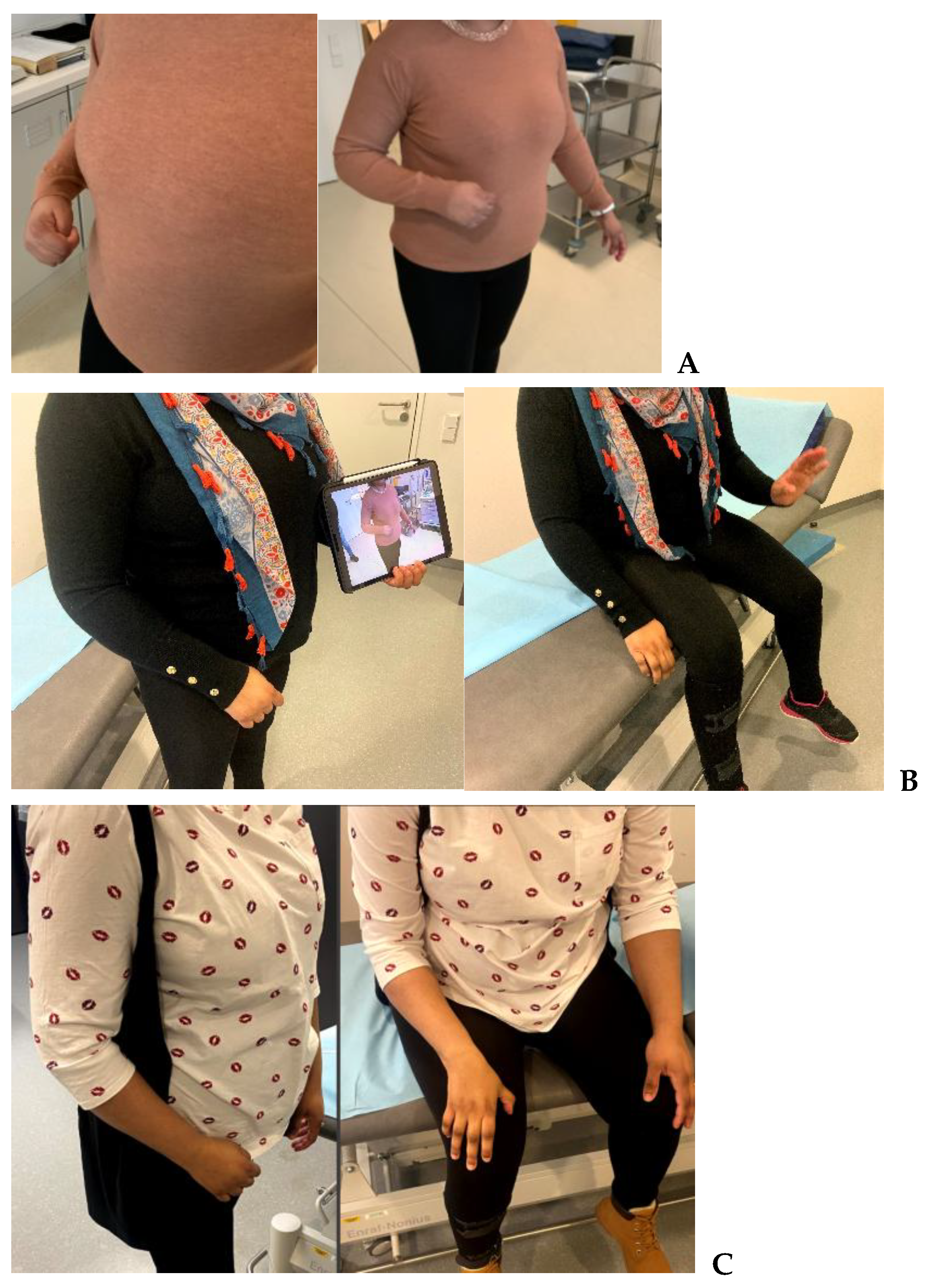
3.3. Clinical Spasticity Outcomes (Tone and Joint Mobility)
3.4. Pain, Sensory Testing and Ease of Care
3.5. Electroneuromyography Findings
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Barnes, M.P.; Johnson, G.R. Upper Motor Neurone Syndrome and Spasticity: Clinical Management and Neurophysiology.
- In Spasticity: Diagnosis and Management, Second edition.; Spasticity: Diagnosis and Management; Brashear, A., Elovic, E., Eds.; Second edition.; Demos: New York, 2016; ISBN 978-1-62070-072-3.
- Sheean, G. The Pathophysiology of Spasticity. Eur J Neurol 2002, 9, 3–9. [CrossRef]
- Nielsen, J.B.; Crone, C.; Hultborn, H. The Spinal Pathophysiology of Spasticity ? From a Basic Science Point of View. Acta Physiol 2007, 189, 171–180. [CrossRef]
- Ward, A.B. A Literature Review of the Pathophysiology and Onset of Post-Stroke Spasticity: Post-Stroke Spasticity: Pathophysiology and Onset. European Journal of Neurology 2012, 19, 21–27. [CrossRef]
- Li, S.; Francisco, G.E. New Insights into the Pathophysiology of Post-Stroke Spasticity. Front. Hum. Neurosci. 2015, 9. [CrossRef]
- Lorentzen, J.; Pradines, M.; Gracies, J.-M.; Bo Nielsen, J. On Denny-Brown’s ‘Spastic Dystonia’ – What Is It and What Causes It? Clinical Neurophysiology 2018, 129, 89–94. [CrossRef]
- Neurological Rehabilitation: Spasticity and Contractures in Clinical Practice and Research; Pandyan, A., Hermens, H.J., Conway, B.A., Eds.; Rehabilitation science in practice series; CRC Press/Taylor & Francis Group: Boca Raton, FL, 2018; ISBN 978-1-4665-6544-9.
- Wafa, H.A.; Wolfe, C.D.A.; Emmett, E.; Roth, G.A.; Johnson, C.O.; Wang, Y. Burden of Stroke in Europe: Thirty-Year Projections of Incidence, Prevalence, Deaths, and Disability-Adjusted Life Years. Stroke 2020, 51, 2418–2427. [CrossRef]
- Pandyan, A.D.; Price, C.I.M.; Rodgers, H.; Barnes, M.P.; Johnson, G.R. Biomechanical Examination of a Commonly Used Measure of Spasticity. Clinical Biomechanics 2001, 16, 859–865. [CrossRef]
- Malhotra, S.; Pandyan, A.; Day, C.; Jones, P.; Hermens, H. Spasticity, an Impairment That Is Poorly Defined and Poorly Measured. Clin Rehabil 2009, 23, 651–658. [CrossRef]
- Pandyan, A.; Gregoric, M.; Barnes, M.; Wood, D.; Wijck, F.V.; Burridge, J.; Hermens, H.; Johnson, G. Spasticity: Clinical Perceptions, Neurological Realities and Meaningful Measurement. Disability and Rehabilitation 2005, 27, 2–6. [CrossRef]
- Fleuren, J.F.M.; Voerman, G.E.; Erren-Wolters, C.V.; Snoek, G.J.; Rietman, J.S.; Hermens, H.J.; Nene, A.V. Stop Using the Ashworth Scale for the Assessment of Spasticity. Journal of Neurology, Neurosurgery & Psychiatry 2010, 81, 46–52. [CrossRef]
- Aloraini, S.M.; Gäverth, J.; Yeung, E.; MacKay-Lyons, M. Assessment of Spasticity after Stroke Using Clinical Measures: A Systematic Review. Disability and Rehabilitation 2015, 37, 2313–2323. [CrossRef]
- Johnson, G.R. Outcome Measures of Spasticity. Euro J of Neurology 2002, 9, 10–16. [CrossRef]
- Alhusaini, A.A.A.; Dean, C.M.; Crosbie, J.; Shepherd, R.B.; Lewis, J. Evaluation of Spasticity in Children With Cerebral Palsy Using Ashworth and Tardieu Scales Compared With Laboratory Measures. J Child Neurol 2010, 25, 1242–1247. [CrossRef]
- Shu, X.; McConaghy, C.; Knight, A. Validity and Reliability of the Modified Tardieu Scale as a Spasticity Outcome Measure of the Upper Limbs in Adults with Neurological Conditions: A Systematic Review and Narrative Analysis. BMJ Open 2021, 11, e050711. [CrossRef]
- Morris, S. Ashworth And Tardieu Scales: Their Clinical Relevance For Measuring Spasticity In Adult And Paediatric Neurological Populations. Physical Therapy Reviews 2002, 7, 53–62. [CrossRef]
- Hanlan, A.; Mills, P.; Lipson, R.; Finlayson, H. Interdisciplinary Spasticity Management Clinic Outcomes Using the Goal Attainment Scale: A Retrospective Chart Review. J Rehabil Med 2017, 49, 423–430. [CrossRef]
- Turner-Stokes, L.; Jacinto, J.; Fheodoroff, K.; Brashear, A.; Maisonobe, P.; Lysandropoulos, A.; group, S. Longitudinal Goal Attainment with Integrated Upper Limb Spasticity Management Including Repeat Injections of Botulinum Toxin A: Findings from the Prospective, Observational Upper Limb International Spasticity (ULIS-III) Cohort Study. J Rehabil Med 2021, 53, jrm00157. [CrossRef]
- Turner-Stokes, L.; Fheodoroff, K.; Jacinto, J.; Lambert, J.; De La Loge, C.; Calvi-Gries, F.; Whalen, J.; Lysandropoulos, A.; Maisonobe, P.; Ashford, S. The Spasticity-Related Quality of Life 6-Dimensions Instrument in Upper-Limb Spasticity: A First Psychometric Evaluation. JRM 2021. [CrossRef]
- Jacinto, J.; Camões-Barbosa, A.; Carda, S.; Hoad, D.; Wissel, J. A Practical Guide to Botulinum Neurotoxin Treatment of Shoulder Spasticity 1: Anatomy, Physiology, and Goal Setting. Front. Neurol. 2022, 13, 1004629. [CrossRef]
- Molteni, F.; Wissel, J.; Fheodoroff, K.; Munin, M.C.; Patel, A.T.; Althaus, M.; Comes, G.; Dekundy, A.; Pulte, I.; Scheschonka, A.; et al. Improvement in Quality-of-Life-Related Outcomes Following Treatment with IncobotulinumtoxinA in Adults with Limb Spasticity: A Pooled Analysis. Toxins 2023, 16, 19. [CrossRef]
- Gasperini, G.; Rota, M.; Guanziroli, E.; Bissolotti, L.; Balestrieri, F.; Chisari, C.; Currà, A.; Del Felice, A.; Farina, N.; Manganotti, P.; et al. Development and Rasch Validation of an Observational Assessment Tool of Upper Limb Functional Impairment in Stroke Survivors: Functional Assessment Test for Upper Limb. Archives of Physical Medicine and Rehabilitation 2023, 104, 597–604. [CrossRef]
- Khan, F.; Amatya, B.; Bensmail, D.; Yelnik, A. Non-Pharmacological Interventions for Spasticity in Adults: An Overview of Systematic Reviews. Annals of Physical and Rehabilitation Medicine 2019, 62, 265–273. [CrossRef]
- Picelli, A.; Santamato, A.; Chemello, E.; Cinone, N.; Cisari, C.; Gandolfi, M.; Ranieri, M.; Smania, N.; Baricich, A. Adjuvant Treatments Associated with Botulinum Toxin Injection for Managing Spasticity: An Overview of the Literature. Annals of Physical and Rehabilitation Medicine 2019, 62, 291–296. [CrossRef]
- Baricich, A.; Picelli, A.; Carda, S.; Smania, N.; Cisari, C.; Santamato, A.; De Sire, A.; Invernizzi, M. Electrical Stimulation of Antagonist Muscles after Botulinum Toxin Type A for Post-Stroke Spastic Equinus Foot. A Randomized Single-Blind Pilot Study. Annals of Physical and Rehabilitation Medicine 2019, 62, 214–219. [CrossRef]
- Esquenazi, A.; Mayer, N.H.; Elia, A.E.; Albanese, A. Botulinum Toxin for the Management of Adult Patients with Upper Motor Neuron Syndrome. Toxicon 2009, 54, 634–638. [CrossRef]
- Das, T.K.; Park, D.M. Effect of Treatment with Botulinum Toxin on Spasticity. Postgraduate Medical Journal 1989, 65, 208–210. [CrossRef]
- Mancini, F.; Sandrini, G.; Moglia, A.; Nappi, G.; Pacchetti, C. A Randomised, Double-Blind, Dose-Ranging Study to Evaluate Efficacy and Safety of Three Doses of Botulinum Toxin Type A (Botox) for the Treatment of Spastic Foot. Neurol Sci 2005, 26, 26–31. [CrossRef]
- Gracies, J.-M. Physiological Effects of Botulinum Toxin in Spasticity. Mov Disord. 2004, 19, S120–S128. [CrossRef]
- Simpson, D.M.; Alexander, D.N.; O’Brien, C.F.; Aswad, A.; Leon, J.M.; Mordaunt, J.M.; Monaghan, E.P. Botulinurn Toxin Type A in the Treatment of Upper Extremity Spasticity:.
- Karri, J.; Mas, M.; Francisco, G.; Li, S. Practice Patterns for Spasticity Management with Phenol Neurolysis. J Rehabil Med 2017, 49, 482–488. [CrossRef]
- Karri, J.; Zhang, B.; Li, S. Phenol Neurolysis for Management of Focal Spasticity in the Distal Upper Extremity. PM&R 2020, 12, 246–250. [CrossRef]
- Wood, K.M. The Use of Phenol as a Neurolytic Agent: A Review: Pain 1978, 5, 205–229. [CrossRef]
- Bakheit, A.; Badwan, D.; McLellan, D. The Effectiveness of Chemical Neurolysis in the Treatment of Lower Limb Muscle Spasticity. Clin Rehabil 1996, 10, 40–43. [CrossRef]
- Sindou, M.P.; Simon, F.; Mertens, P.; Decq, P. Selective Peripheral Neurotomy (SPN) for Spasticity in Childhood. Childs Nerv Syst 2007, 23, 957–970. [CrossRef]
- Dauleac, C.; Sindou, M.; Mertens, P. How I Do It: Selective Tibial Neurotomy. Acta Neurochir 2020, 162, 1921–1923. [CrossRef]
- Sindou, M.P.; Mertens, P. Surgery in the Dorsal Root Entry Zone for Spasticity in Adults. Operative Techniques in Neurosurgery 2004, 7, 157–162. [CrossRef]
- Thomas, S.P.; Addison, A.P.; Curry, D.J. Surgical Tone Reduction in Cerebral Palsy. Physical Medicine and Rehabilitation Clinics of North America 2020, 31, 91–105. [CrossRef]
- Deltombe, T.; Lejeune, T.; Gustin, T. Botulinum Toxin Type A or Selective Neurotomy for Treating Focal Spastic Muscle Overactivity? Annals of Physical and Rehabilitation Medicine 2019, 62, 220–224. [CrossRef]
- Lazorthes, Y.; Sol, J.; Sallerin, B.; Verdié, J. The Surgical Management of Spasticity. Euro J of Neurology 2002, 9, 35–41. [CrossRef]
- Genêt, F.; Denormandie, P.; Keenan, M.A. Orthopaedic Surgery for Patients with Central Nervous System Lesions: Concepts and Techniques. Annals of Physical and Rehabilitation Medicine 2019, 62, 225–233. [CrossRef]
- Hashemi, M.; Sturbois-Nachef, N.; Keenan, M.A.; Winston, P. Surgical Approaches to Upper Limb Spasticity in Adult Patients: A Literature Review. Front. Rehabilit. Sci. 2021, 2, 709969. [CrossRef]
- Filipetti, P.; Decq, P. Interest of anesthetic blocks for assessment of the spastic patient. A series of 815 motor blocks. Neurochirurgie 2003, 49(2-3 Pt 2):226-38.
- Genet, F.; Schnitzler, A.; Droz-Bartholet, F.; Salga, M.; Tatu, L.; Debaud, C.; Denormandie, P.; Parratte, B. Successive Motor Nerve Blocks to Identify the Muscles Causing a Spasticity Pattern: Example of the Arm Flexion Pattern. J. Anat. 2017, 230, 106–116. [CrossRef]
- Yelnik, A.P.; Hentzen, C.; Cuvillon, P.; Allart, E.; Bonan, I.V.; Boyer, F.C.; Coroian, F.; Genet, F.; Honore, T.; Jousse, M.; et al. French Clinical Guidelines for Peripheral Motor Nerve Blocks in a PRM Setting. Annals of Physical and Rehabilitation Medicine 2019, 62, 252–264. [CrossRef]
- Buffenoir, K.; Decq, P.; Lefaucheur, J.-P. Interest of Peripheral Anesthetic Blocks as a Diagnosis and Prognosis Tool in Patients with Spastic Equinus Foot: A Clinical and Electrophysiological Study of the Effects of Block of Nerve Branches to the Triceps Surae Muscle. Clinical Neurophysiology 2005, 116, 1596–1600. [CrossRef]
- Ilfeld, B.M.; Preciado, J.; Trescot, A.M. Novel Cryoneurolysis Device for the Treatment of Sensory and Motor Peripheral Nerves. Expert Review of Medical Devices 2016, 13, 713–725. [CrossRef]
- Bittman, R.W.; Peters, G.L.; Newsome, J.M.; Friedberg, E.B.; Mitchell, J.W.; Knight, J.M.; Prologo, J.D. Percutaneous Image-Guided Cryoneurolysis. American Journal of Roentgenology 2018, 210, 454–465. [CrossRef]
- Gage, A.A.; Baust, J.M.; Baust, J.G. Experimental Cryosurgery Investigations in Vivo. Cryobiology 2009, 59, 229–243. [CrossRef]
- Amoils, S.P. The Joule Thomson Cryoprobe. Archives of Ophthalmology 1967, 78, 201–207. [CrossRef]
- Whittaker, D.K. Ice Crystals Formed in Tissue during Cryosurgery. Cryobiology 1974, 11, 202–217. [CrossRef]
- Sunderland, S. A CLASSIFICATION OF PERIPHERAL NERVE FNJURIES PRODUCING LOSS OF FUNCTION.
- Shah, S.B.; Bremner, S.; Esparza, M.; Dorn, S.; Orozco, E.; Haghshenas, C.; Ilfeld, B.M.; Gabriel, R.A.; Ward, S. Does Cryoneurolysis Result in Persistent Motor Deficits? A Controlled Study Using a Rat Peroneal Nerve Injury Model. Reg Anesth Pain Med 2020, 45, 287–292. [CrossRef]
- Myers, R.R.; Powell, H.C.; Heckman, H.M.; Costello, M.L.; Katz, J. Biophysical and Pathological Effects of Cryogenic Nerve Lesion. Annals of Neurology 1981, 10, 478–485. [CrossRef]
- Evans, P.J.D. Cryoanalgesia: The Application of Low Temperatures to Nerves to Produce Anaesthesia or Analgesia. Anaesthesia 1981, 36, 1003–1013. [CrossRef]
- Trescot, A.M. Cryoanalgesia in Interventional Pain Management. Pain Phys 2003, 3;6, 345–360. [CrossRef]
- Biel, E.; Aroke, E.N.; Maye, J.; Zhang, S.J. The Applications of Cryoneurolysis for Acute and Chronic Pain Management. Pain Practice 2023, 23, 204–215. [CrossRef]
- Finneran, J.J.; Kobayashi, L.; Costantini, T.W.; Weaver, J.L.; Berndtson, A.E.; Haines, L.; Doucet, J.J.; Adams, L.; Santorelli, J.E.; Lee, J.; et al. Ultrasound-Guided Percutaneous Cryoneurolysis for the Treatment of Pain after Traumatic Rib Fracture: A Randomized, Active-Controlled, Participant- and Observer-Masked Study. Anesthesiology 2025, 142, 532–545. [CrossRef]
- Ilfeld, B.M.; Finneran, J.J.; Swisher, M.W.; Said, E.T.; Gabriel, R.A.; Sztain, J.F.; Khatibi, B.; Armani, A.; Trescot, A.; Donohue, M.C.; et al. Preoperative Ultrasound-Guided Percutaneous Cryoneurolysis for the Treatment of Pain after Mastectomy: A Randomized, Participant- and Observer-Masked, Sham-Controlled Study. Anesthesiology 2022, 137, 529–542. [CrossRef]
- Radnovich, R.; Scott, D.; Patel, A.T.; Olson, R.; Dasa, V.; Segal, N.; Lane, N.E.; Shrock, K.; Naranjo, J.; Darr, K.; et al. Cryoneurolysis to Treat the Pain and Symptoms of Knee Osteoarthritis: A Multicenter, Randomized, Double-Blind, Sham-Controlled Trial. Osteoarthritis and Cartilage 2017, 25, 1247–1256. [CrossRef]
- Müller, L.C.; Salzer, G.M.; Ransmayr, G.; Neiss, A. Intraoperative Cryoanalgesia for Postthoracotomy Pain Relief.
- Cheng, J.-G. Cryoanalgesia for Refractory Neuralgia. 2015.
- Ilfeld, B.M.; Gabriel, R.A.; Trescot, A.M. Ultrasound-Guided Percutaneous Cryoneurolysis for Treatment of Acute Pain: Could Cryoanalgesia Replace Continuous Peripheral Nerve Blocks? British Journal of Anaesthesia 2017, 119, 709–712. [CrossRef]
- Winston, P.; Mills, P.B.; Reebye, R.; Vincent, D. Cryoneurotomy as a Percutaneous Mini-Invasive Therapy for the Treatment of the Spastic Limb: Case Presentation, Review of the Literature, and Proposed Approach for Use. Archives of Rehabilitation Research and Clinical Translation 2019, 1, 100030. [CrossRef]
- Winston, P.; Mills, P.; Ganzert, C.; Reebye, R.; Vincent, D. Cryoneurotomy as a Novel Adjunct to Botulinum Toxin Treatment for the Spastic Elbow: A Case Study. Toxicon 2018, 156, S114–S115. [CrossRef]
- Scobie, J.; Winston, P. Case Report: Perspective of a Caregiver on Functional Outcomes Following Bilateral Lateral Pectoral Nerve Cryoneurotomy to Treat Spasticity in a Pediatric Patient With Cerebral Palsy. Front. Rehabilit. Sci. 2021, 2, 719054. [CrossRef]
- Rubenstein, J.; Harvey, A.W.; Vincent, D.; Winston, P. Cryoneurotomy to Reduce Spasticity and Improve Range of Motion in Spastic Flexed Elbow: A Visual Vignette. Am J Phys Med Rehabil 2021, 100, e65–e65. [CrossRef]
- David, R.; Hashemi, M.; Schatz, L.; Winston, P. Multisite Treatment with Percutaneous Cryoneurolysis for the Upper and Lower Limb in Long-Standing Post-Stroke Spasticity. Eur J Phys Rehabil Med 2024, 60. [CrossRef]
- Hashemi, M.; MacRae, F.; Boissonnault, È.; Vincent, D.; Song, J.; Shi, S.; Winston, P. Measuring the Efficacy of Percutaneous Cryoneurolysis in Participants With Refractory or Plateaued Shoulder, Elbow, Wrist, or Finger Spasticity. Am J Phys Med Rehabil 2025, 104, 896–905. [CrossRef]
- MacRae, F.; Brar, A.; Boissonnault, E.; Winston, P. Cryoneurolysis of Anterior and Posterior Divisions of the Obturator Nerve. Am J Phys Med Rehabil 2023, 102, e1–e2. [CrossRef]
- Pick, A.; Dye, R.; Fleming, M.K. Cryoneurolysis: A Novel Treatment for Management of Spasticity. Presentation of a Case Series. Advances in Rehabilitation Science and Practice 2025, 14, 27536351251340216. [CrossRef]
- Chantraine, F.; Pereira, J.A.; Schreiber, C.; Classen, T.; Areno, G.; Dierick, F. Targeted and Sequential Cryoneurolysis Improves Gait After Botulinum-Toxin Unresponsiveness in Post-Stroke Spasticity: A Laboratory-Verified Case. Neurology International 2026, 18, 13. [CrossRef]
- Agha, R.A.; Sohrabi, C.; Mathew, G.; Franchi, T.; Kerwan, A.; O’Neill, N.; Thoma, A.; Beamish, A.J.; Noureldin, A.; Rao, A.; et al. The PROCESS 2020 Guideline: Updating Consensus Preferred Reporting Of CasE Series in Surgery (PROCESS) Guidelines. International Journal of Surgery 2020, 84, 231–235. [CrossRef]
- Hefter, H.; Jost, W.H.; Reissig, A.; Zakine, B.; Bakheit, A.M.; Wissel, J. Classification of Posture in Poststroke Upper Limb Spasticity: A Potential Decision Tool for Botulinum Toxin A Treatment? International Journal of Rehabilitation Research 2012, 35, 227–233. [CrossRef]
- Winston, P.; Mills, P.B.; Reebye, R.; Vincent, D. Cryoneurotomy as a Percutaneous Mini-Invasive Therapy for the Treatment of the Spastic Limb: Case Presentation, Review of the Literature, and Proposed Approach for Use. Archives of Rehabilitation Research and Clinical Translation 2019, 1, 100030. [CrossRef]
- Bouhassira, D.; Attal, N.; Alchaar, H.; Boureau, F.; Brochet, B.; Bruxelle, J.; Cunin, G.; Fermanian, J.; Ginies, P.; Grun-Overdyking, A.; et al. Comparison of Pain Syndromes Associated with Nervous or Somatic Lesions and Development of a New Neuropathic Pain Diagnostic Questionnaire (DN4). Pain 2005, 114, 29–36. [CrossRef]
- Boissonnault, È.; Pick, A.; Pereira, J.; Li, S.; Winston, P. Emerging Role of Cryoneurolysis in Spasticity Management. Physical Medicine and Rehabilitation Clinics of North America 2026, S1047965126000094. [CrossRef]
| Patient | Sex |
Age (years) |
Type of stroke | Affected side |
Time delay between stroke and CNL (months) |
| #1 | man | 45 | hemorrhagic | right | 25 |
| #2 | woman | 34 | hemorrhagic | right | 36 |
| #3 | man | 58 | ischemic | left | 22 |
| Participant | Video | Nerves treated |
Neuropathic pain (DN4) (pre→6 wks) |
Pain VAS (pre→6 wks) |
Sensory testing (6 wks) Pressure (g); 2-point (mm); Thermal |
Spasticity/mobility |
ENMG (6 wks) Median |
ENMG (6 wks) Ulnar / Musculocutaneous |
| #1 | Musculocutaneous; Median | 0→0 | 0→0 | 1.0→0.4 g; 48→48 mm; thermal preserved | MAS ↓ >2 points (elbow flexors, pronators, wrist/finger flexors); Passive mobility improved (no video) | No change (baseline normal); no EMG rest activity post | Ulnar: not treated / not assessed Musculocutaneous: no denervation reported |
|
| #2 |  |
Musculocutaneous; Median; Ulnar | 6→5 | 5.5→0 | 0.7→0.7 g; 25→25 mm; thermal preserved | MAS ↓ >2 points; Passive mobility improved (video 1. Figure 5) |
No change (baseline normal) | Ulnar: no change (baseline normal) Needle EMG: interpretation limited by spasms (rest activity not excluded) |
| #3 |  |
Musculocutaneous; Median; Ulnar | 0→0 | 0→0 | 0.7→0.7 g; no 2-point discrimination pre/post; hot sensation absent pre/post | MAS ↓ >2 points; Passive mobility improved (video 2and? Figure 6) | No change (baseline normal); EMG FDI/APB normal | Ulnar SNAP amplitude reduction progressed (~30%→~60%); F-wave latency normal; ADM EMG normal Musculocutaneous: biceps EMG showed denervation activity post |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




