Submitted:
02 March 2026
Posted:
16 March 2026
You are already at the latest version
Abstract
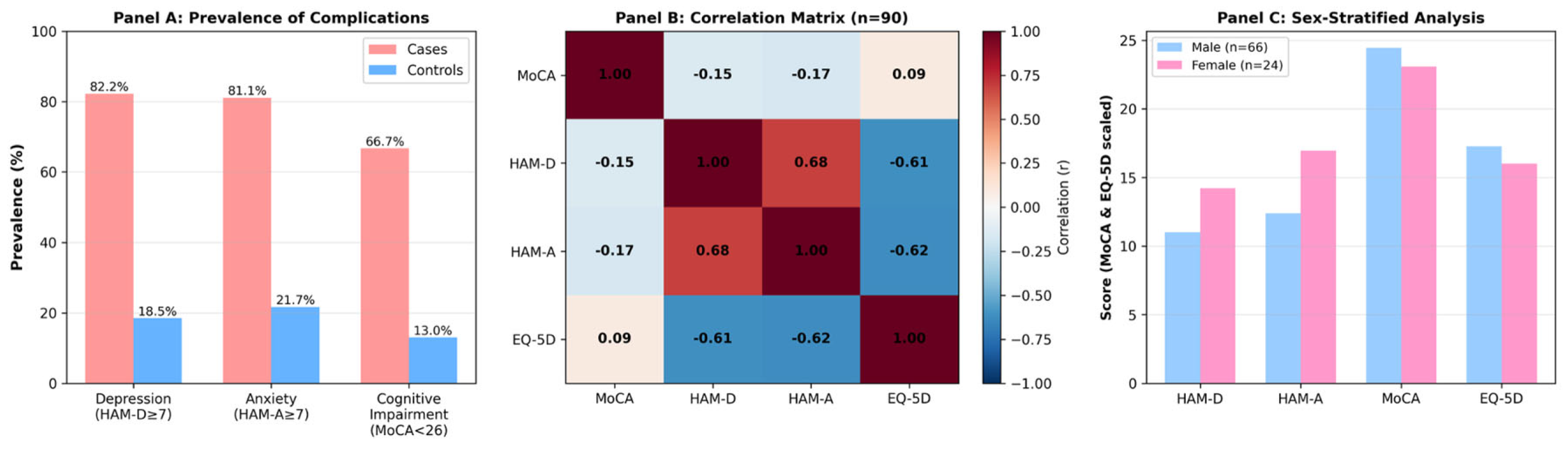
Background. Transient ischemic attack (TIA) and minor stroke often result in excellent functional recovery but are frequently followed by substantial psychological morbidity. It remains unclear whether mood disturbances or cognitive impairment are the primary contributors to reduced health-related quality of life (HRQoL) in this population. Methods. We conducted a prospective observational case–control study including 90 patients with acute TIA or minor stroke confirmed by diffusion-weighted imaging, and 92 age-matched healthy controls. At 90 days, participants completed the Hamilton Depression Rating Scale, Hamilton Anxiety Rating Scale, Montreal Cognitive Assessment, and the EQ-5D-5L. Hierarchical multiple regression using standardized z-scores identified independent predictors of HRQoL. Bias-corrected bootstrapped mediation analyses (5,000 iterations) assessed whether cognitive impairment mediated the relationship between mood symptoms and HRQoL. Results: Compared with controls, patients exhibited markedly higher rates of depression (82.2% vs. 18.5%), anxiety (81.1% vs. 21.7%), and cognitive impairment (66.7% vs. 13.0%) (all p<0.001). Psychopathological variables explained an additional 36.6% of HRQoL variance, whereas cognitive and neuroimaging variables contributed only 1.7% (ΔR2=0.017; p=0.523). Anxiety showed the strongest predictive value (β=–0.055; p=0.064), while cognitive impairment had negligible effects (β=–0.001; p=0.947). Mediation analyses revealed no significant indirect effects, indicating that mood and cognitive complications arise independently rather than sequentially. Conclusions: Following TIA or minor stroke, depressive and anxiety symptoms are highly prevalent, persist despite good neurological recovery, and exert a disproportionately negative impact on HRQoL. Anxiety appears particularly influential in determining patient-reported outcomes, underscoring the need for routine mood screening and targeted psychological management in this population.
Keywords:
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| TIA | transient ischemic attack |
| mRS | modified Rankin Scale |
| HRQoL | health-related quality of life |
| DWI | diffusion-weighted imaging |
| NIHSS | National Institutes of Health Stroke Scale |
| HDRS-17 | Hamilton Depression Rating Scale |
| HAM-A | Hamilton Anxiety Rating Scale |
| MoCA | Montreal Cognitive Assessment |
Appendix A
| Outcome measure | Cases (n=90) | Controls (n=92) | pvalue | Cohen’s d | OR (95% CI)† |
| DEPRESSION | |||||
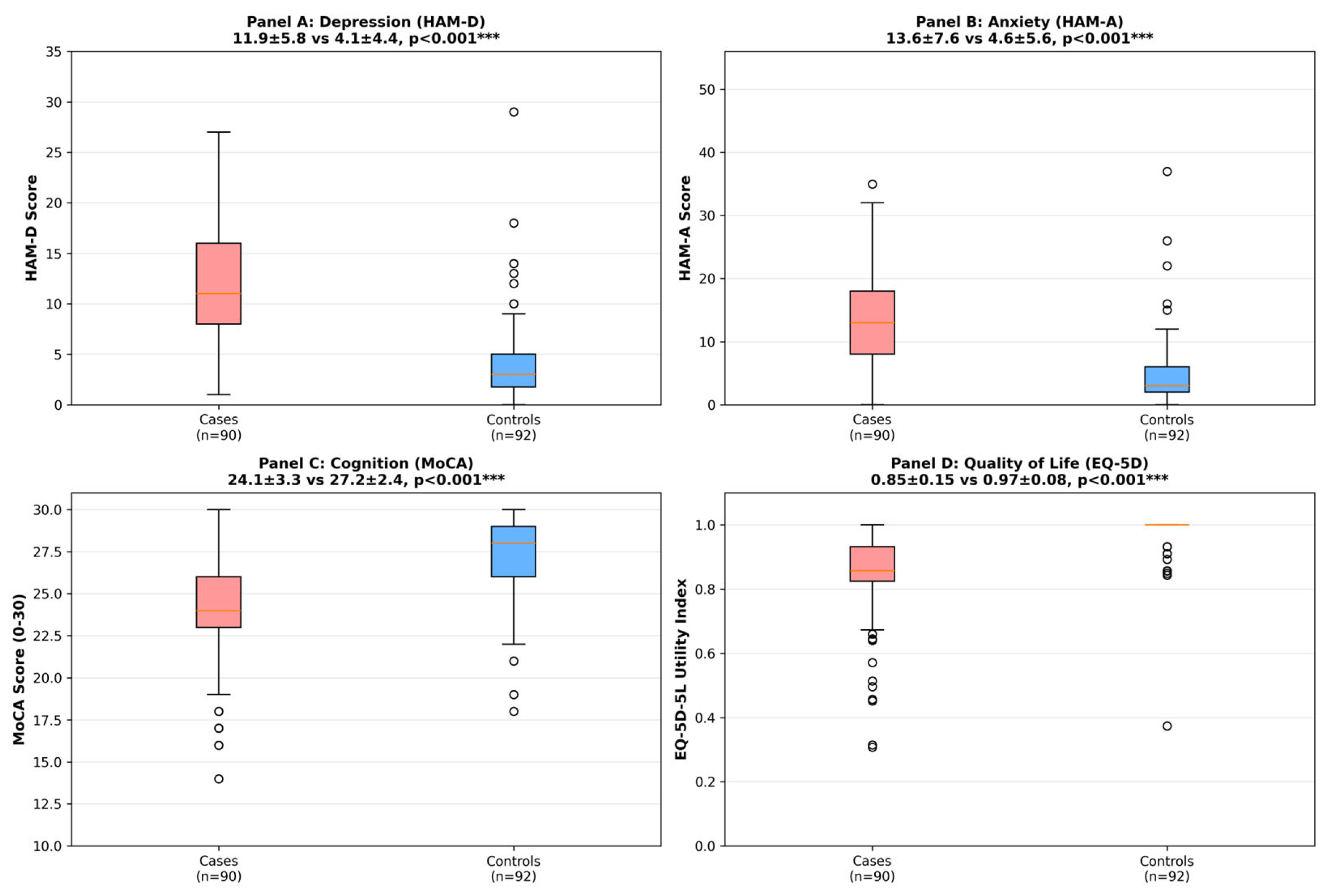
| HDRS-17, mean ± SD | 11.9 ± 5.8 | 4.1 ± 4.4 | <0.001*** | 1.50 | — |
| Depressive symptoms (HDRS-17 ≥ 7), n (%) | 74 (82.2) | 17 (18.5) | <0.001*** | — | 20.41 (9.60–43.42) |
| ANXIETY | |||||
| HAM-A, mean ± SD | 13.6 ± 7.6 | 4.6 ± 5.6 | <0.001*** | 1.35 | — |
| Anxiety symptoms (HAM-A ≥ 7), n (%) | 73 (81.1) | 20 (21.7) | <0.001*** | — | 15.46 (7.50–31.87) |
| COGNITIVE FUNCTION | |||||
| MoCA, mean ± SD | 24.1 ± 3.3 | 27.2 ± 2.4 | <0.001*** | −1.10 | — |
| Cognitive impairment (MoCA < 26), n (%) | 60 (66.7) | 12 (13.0) | <0.001*** | — | 13.33 (6.31–28.21) |
| QUALITY OF LIFE | |||||
| EQ-5D-5L utility index, mean ± SD | 0.85 ± 0.15 | 0.97 ± 0.08 | <0.001*** | −1.06 | — |
| EQ-VAS, mean ± SD | 51 ± 17 | 79 ± 13 | <0.001*** | −1.94 | — |
| Variable pair | r (type) | p value | q (FDR) | Strength |
| MoCA ↔ HDRS-17 | −0.146 (Pearson) | 0.162 | 0.216 | Weak/NS |
| MoCA ↔ HAM-A | −0.170 (Pearson) | 0.112 | 0.186 | Weak/NS |
| HDRS-17 ↔ HAM-A | 0.681 (Pearson) | <0.001*** | <0.001 | Strong |
| HDRS-17 ↔ EQ-5D-5L | −0.612 (Spearman) | <0.001*** | <0.001 | Strong |
| HAM-A ↔ EQ-5D-5L | −0.625 (Spearman) | <0.001*** | <0.001 | Strong |
| MoCA ↔ EQ-5D-5L | 0.092 (Spearman) | 0.372 | 0.372 | Negligible |
| Age ↔ HDRS-17 | −0.103 (Pearson) | 0.314 | 0.357 | Weak/NS |
| Age ↔ MoCA | −0.081 (Pearson) | 0.445 | 0.445 | Weak/NS |
| DWI lesion (0/1) ↔ HDRS-17 | 0.137 (Point-biserial) | 0.190 | 0.216 | Weak/NS |
| DWI lesion (0/1) ↔ MoCA | −0.191 (Point-biserial) | 0.078 | 0.117 | Weak/trend |
| Pathway | Coefficient (β) | SE | 95% CI | p value |
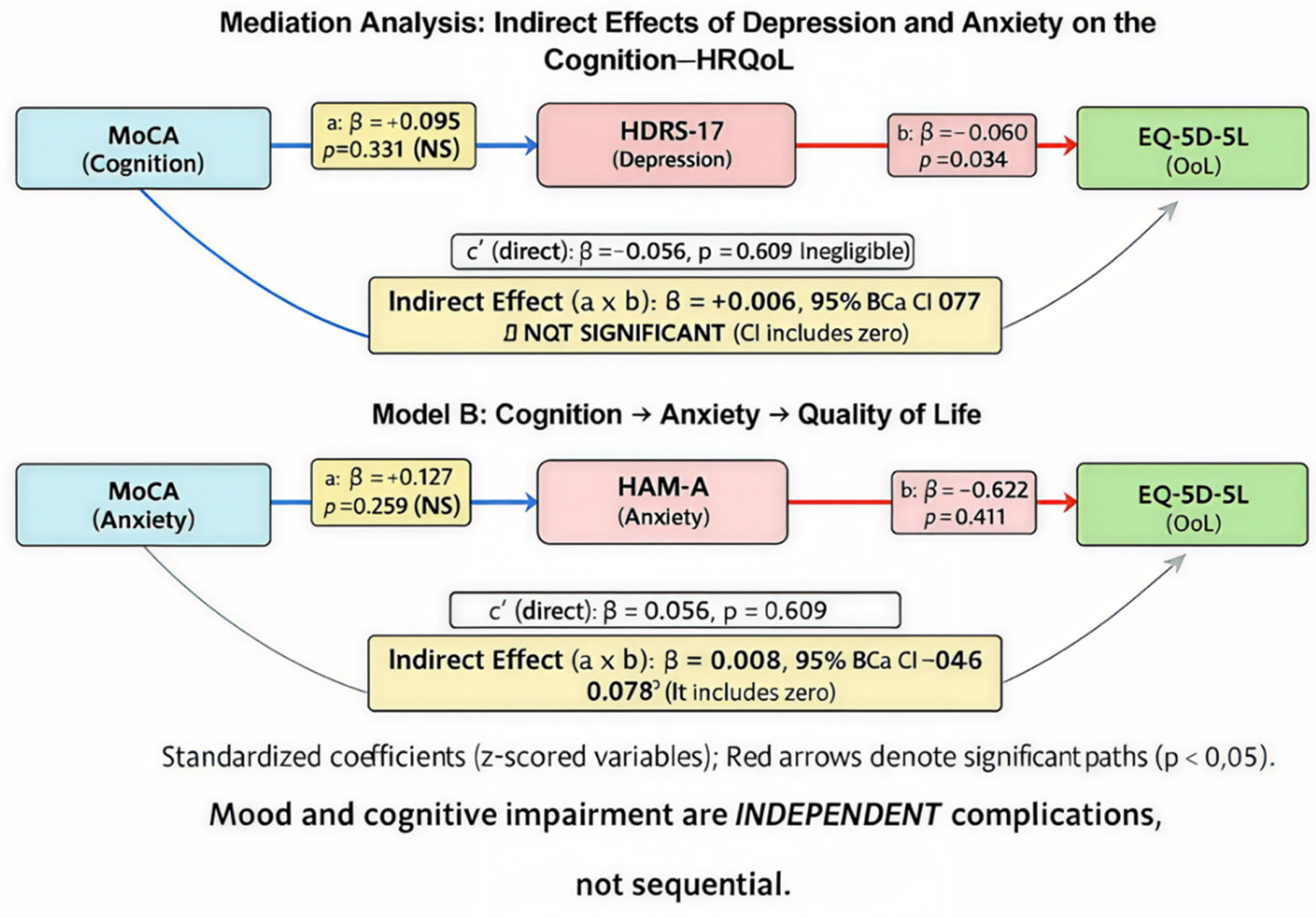
| c (Total effect) | 0.0560 | 0.108 | [−0.123, 0.235] | 0.609 |
| a (Cognition → Depression) | −0.0951 | 0.108 | [−0.310, 0.120] | 0.381 |
| c′ (Direct effect) | 0.0572 | 0.109 | [−0.130, 0.245] | 0.602 |
| b (Depression → HRQoL) | −0.0604 | 0.028 | [−0.116, −0.005] | 0.034* |
| a×b (Indirect effect) | −0.0057 | — | [−0.068, 0.047] | — |
| Pathway | Coefficient (β) | SE | 95% CI | p value |
| c (Total effect) | 0.0560 | 0.108 | [−0.123, 0.235] | 0.609 |
| a (Cognition → Anxiety) | −0.1272 | 0.107 | [−0.343, 0.088] | 0.239 |
| c′ (Direct effect) | 0.0787 | 0.110 | [−0.145, 0.303] | 0.476 |
| b (Anxiety → HRQoL) | −0.0620 | 0.030 | [−0.121, −0.003] | 0.041* |
| a×b (Indirect effect) | −0.0079 | — | [−0.046, 0.078] | — |
Appendix B
Appendix C
| Section/Item | Nº | STROBE recommendation | Where in the manuscript |
| Title/Abstract | 1 | (a) Indicate the study design with a commonly used term in the title or abstract; (b) Provide an informative, balanced summary of methods and findings. | Title (“…Case–Control Study”); Abstract (structured IMRaD). |
| Introduction | 2 | Background/rationale: explain scientific context and rationale. | Introduction, paragraphs 1–4. |
| 3 | Objectives: state specific aims/prespecified hypotheses. | Introduction, final paragraph (study aims). | |
| Methods | 4 | Study design: present key elements early. | Methods—Study Design and Participants (“single-center, prospective, observational case–control”). |
| 5 | Setting: locations, relevant dates; recruitment and assessment windows. | Methods—Neuroimaging Acquisition (acute MRI; mean 1.3 ± 0.8 days post-event) and Assessments at 90 ± 14 days; recruiting hospital(s). | |
| 6 | Participants: eligibility, sources/methods of case ascertainment and control selection; rationale; (b) matching criteria and number of controls per case if applicable. | Methods—Study Design and Participants (18–70 y; TIA/minor stroke NIHSS ≤ 4; DWI-confirmed; age-matched controls; exclusions: prior dementia, pre-morbid mRS > 1, non-Spanish speakers). | |
| 7 | Variables: clearly define outcomes, exposures, predictors, potential confounders/effect modifiers; diagnostic criteria. | Methods—Psychopathological, Cognitive, and HRQoL Assessment (HDRS-17, HAM-A, MoCA, EQ-5D-5L; cut-offs); Neuroimaging variables; clinical covariates (age, sex, mRS, social risk). | |
| 8 | Data sources/measurement: sources and details of assessment methods; comparability between groups. | Methods—Assessments at 90 ± 14 days (trained, blinded evaluators; standardized protocol); Neuroimaging (1.5 T sequences, blinded neuroradiologists); EQ-5D-5L Spanish value set | |
| 9 | Bias: describe efforts to address potential sources of bias. | Methods—Blinding/Training; age-matching for controls; multiplicity control (FDR), HC3 robust SEs, beta-regression sensitivity. | |
| 10 | Study size: explain how size was arrived at. | Methods—Regression Modeling (≈10:1 participants:predictors); Limitations (post-hoc power 1−β = 0.84). | |
| 11 | Quantitative variables: how handled; groupings and rationale. | Methods—Regression Modeling (z-scoring; binary coding 0/1); cut-offs for HDRS-17, HAM-A, MoCA; EQ-5D-5L utilities. | |
| 12 | Statistical methods: (a) all methods incl. confounding control; (b) subgroups/interactions; (c) missing data; (d) matching; (e) sensitivity analyses. | Methods—Statistical Analysis (t/Mann–Whitney/χ2/Fisher; Cohen’s d, OR with 95% CI; Pearson/Spearman; hierarchical multiple regression with ΔR2 and incremental F; mediation via PROCESS v3.5, 5,000 BCa; handling of one missing EQ-5D-5L; HC3 & beta-regression sensitivity). | |
| Results | 13 | Participants: numbers at each stage; reasons for non-participation; consider flow diagram. | Results—Sample Characteristics (N = 182; 90 cases/92 controls; n = 89 in regression due to one EQ-5D missing). Optional flow diagram in Supplement. |
| 14 | Descriptive data: characteristics of participants; exposures/confounders; missing data. | Table 1 (baseline); Results—Sample Characteristics; Table S1 (group contrasts and ORs); note on missing EQ-5D-5L in regression set. | |
| 15 | Outcome data: numbers/summaries per group. | Results—Psychopathological, Cognitive, and HRQoL; Figure 1 & Table S1. | |
| 16 | Main results: estimates with precision (e.g., 95% CI); adjusted and unadjusted; report category boundaries when categorizing continuous variables. | Results—Regression (Models 1–3; R2/ΔR2/p; coefficients in Table 3, Table S5); category thresholds for HDRS-17/HAM-A/MoCA. | |
| 17 | Other analyses: subgroups/interactions; sensitivity analyses. | Sex-stratified analysis (Figure S1 Panel C; Table S1 sex block); sensitivity (Tables S6–S7); mediation (Tables S3A–S3B; Figure 3). | |
| Discussion | 18 | Key results summarized with respect to objectives. | Discussion—Principal Findings. |
| 19 | Limitations: discuss sources of potential bias/precision; direction/magnitude. | Discussion—Limitations (case–control/single time point; measures; unmeasured confounding incl. medications/fatigue; generalizability). | |
| 20 | Interpretation: cautious overall interpretation considering objectives, limitations, multiplicity, results from similar studies, and other evidence. | Discussion—Comparison with Prior Work/Mechanisms/Implications. | |
| 21 | Generalisability (external validity). | Discussion— |
References
- Feigin, VL; Stark, BA; Johnson, CO; Roth, GA; Bisignano, C; Abady, GG; et al. Global, regional, and national burden of stroke and its risk factors, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021, 20(10), 795–820. [Google Scholar] [CrossRef]
- Díaz-Guzmán, J; Egido, JA; Gabriel-Sánchez, R; Barberá-Comes, G; Fuentes-Gimeno, B; Fernández-Pérez, C. Stroke and transient ischemic attack incidence rate in Spain: The IBERICTUS study. Cerebrovasc Dis. 2012, 34(4), 272–81. [Google Scholar] [CrossRef]
- Amarenco, P. Transient ischemic attack. N Engl J Med. 2020, 382(20), 1933–41. [Google Scholar] [CrossRef]
- Hobeanu, C; Lavallée, PC; Charles, H; Labreuche, J; Albers, GW; Caplan, LR; et al. Risk of subsequent disabling or fatal stroke in patients with transient ischaemic attack or minor ischaemic stroke: an international, prospective cohort study. Lancet Neurol. 2022, 21(10), 889–98. [Google Scholar] [CrossRef]
- Coutts, SB; Modi, J; Patel, SK; Aram, H; Demchuk, AM; Goyal, M; et al. What causes disability after transient ischemic attack and minor stroke? Stroke 2012, 43(11), 3018–22. [Google Scholar] [CrossRef]
- van Rooij, FG; Kessels, RPC; Richard, E; de Leeuw, FE; van Dijk, EJ. Cognitive impairment in transient ischemic attack patients: a systematic review. Cerebrovasc Dis. 2016, 42(1–2), 1–9. [Google Scholar] [CrossRef]
- Carnes-Vendrell, A; Deus, J; Molina-Seguin, J; Pifarré, J; Purroy, F. Depression and apathy after transient ischemic attack or minor stroke: prevalence, evolution and predictors. Sci Rep. 2019, 9(1), 1–9. [Google Scholar] [CrossRef]
- Shi, Y; Xiang, Y; Yang, Y; Zhang, N; Wang, S; Ungvari, GS; et al. Depression after minor stroke: prevalence and predictors. J Psychosom Res. 2015, 79(2), 143. [Google Scholar] [CrossRef] [PubMed]
- Broomfield, NM; Quinn, TJ; Abdul-Rahim, AH; Walters, MR; Evans, JJ. Depression and anxiety symptoms post-stroke/TIA: prevalence and associations in cross-sectional data from a regional stroke registry. BMC Neurol. 2014, 14, 198. [Google Scholar] [CrossRef]
- Hackett, ML; Pickles, K. Part I: Frequency of depression after stroke: An updated systematic review and meta-analysis of observational studies. Int J Stroke 2014, 9(8), 1017–25. [Google Scholar] [CrossRef]
- Maaijwee, NAMM; Tendolkar, I; Rutten-Jacobs, LCA; Arntz, RM; Schaapsmeerders, P; Dorresteijn, LD; et al. Long-term depressive symptoms and anxiety after transient ischaemic attack or ischaemic stroke in young adults. Eur J Neurol. 2016, 23(8), 1262–8. [Google Scholar] [CrossRef]
- Kapoor, A; Si, K; Yu, AYX; Lanctôt, KL; Herrmann, N; Murray, BJ; et al. Younger age and depressive symptoms predict high risk of generalized anxiety after stroke and transient ischemic attack. Stroke 2019, 50(9), 2359–63. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, GC; Roy, D; Kontos, N; Beach, SR. Post-stroke depression: a 2020 updated review. Gen Hosp Psychiatry 2020, 66, 70–80. [Google Scholar] [CrossRef]
- Robinson, RG; Jorge, RE; Starkstein, SE. Poststroke depression: an update. J Neuropsychiatry Clin Neurosci. 2024, 36(1), 22–35. [Google Scholar] [CrossRef]
- Ebaid, D; Bird, LJ; McCambridge, LJE; Werden, E; Bradshaw, J; Cumming, T; et al. Mood and cognitive trajectories over the first year after mild ischemic stroke. J Stroke Cerebrovasc Dis. 2022, 31(4), 106323. [Google Scholar] [CrossRef]
- Kusec, A; Demeyere, N. Relationship of subjective and objective cognition with post-stroke mood differs between early and long-term stroke. Clin Neuropsychol. 2025, 39(6), 1651–72. [Google Scholar] [CrossRef]
- Hackett, ML; Köhler, S; O’Brien, JT; Mead, GE. Neuropsychiatric outcomes of stroke. Lancet Neurol. 2014, 13(5), 525–34. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, M. A rating scale for depression. J Neurol Neurosurg Psychiatry 1960, 23(1), 56–62. [Google Scholar] [CrossRef]
- Aben, I; Verhey, F; Lousberg, R; Lodder, J; Honig, A. Validity of the Beck Depression Inventory, Hospital Anxiety and Depression Scale, SCL-90, and Hamilton Depression Rating Scale as screening instruments for depression in stroke patients. Psychosomatics 2002, 43(5), 386–93. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, M. The assessment of anxiety states by rating. Br J Med Psychol. 1959, 32(1), 50–5. [Google Scholar] [CrossRef]
- Xiao, M; Huang, G; Feng, L; Luan, X; Wang, Q; Ren, W; et al. Impact of sleep quality on post-stroke anxiety in stroke patients. Brain Behav. 2020, 10(12), e01813. [Google Scholar] [CrossRef]
- Zhu, BL; Hu, AY; Huang, GQ; Qiu, HH; Hong, XC; Hu, PL; et al. Association between obesity and post-stroke anxiety in patients with acute ischemic stroke. Front Nutr. 2021, 8, 749958. [Google Scholar] [CrossRef] [PubMed]
- Nasreddine, ZS; Phillips, NA; Bédirian, V; Charbonneau, S; Whitehead, V; Collin, I; et al. The Montreal Cognitive Assessment (MoCA): a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005, 53(4), 695–9. [Google Scholar] [CrossRef]
- Ramírez-Moreno, JM; Bartolomé Alberca, S; Muñoz Vega, P; Guerrero Barona, EJ. Detección del deterioro cognitivo con la Evaluación Cognitiva de Montreal en pacientes españoles con ictus minor o ataque isquémico transitorio. Neurología 2022, 37(1), 38–44. [Google Scholar] [CrossRef]
- Herdman, M; Gudex, C; Lloyd, A; Janssen, MF; Kind, P; Parkin, D; et al. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res. 2011, 20(10), 1727–36. [Google Scholar] [CrossRef] [PubMed]
- Moran, GM; Fletcher, B; Feltham, MG; Calvert, M; Sackley, C; Marshall, T. Fatigue, psychological and cognitive impairment following transient ischaemic attack and minor stroke: a systematic review. Eur J Neurol. 2014, 21(10), 1258–67. [Google Scholar] [CrossRef]
- Xiao, W; Liu, Y; Huang, J; Lan, Huang; Bian, Y; Zou, G. Analysis of factors associated with depressive symptoms in stroke patients based on a national cross-sectional study. Sci Rep. 2024, 14(1), 9268. [Google Scholar] [CrossRef] [PubMed]
- Altieri, M; Maestrini, I; Mercurio, A; Troisi, P; Sgarlata, E; Rea, V; et al. Depression after minor stroke: prevalence and predictors. Eur J Neurol. 2012, 19(3), 517–21. [Google Scholar] [CrossRef]
- McColl, AJ; Luengo-Fernandez, R; Vaughan-Fowler, ER; Downer, MB; Pendlebury, ST; Binney, LE; et al. Prevalence, predictors, and prognosis of depression after transient ischemic attack: a population-based study. Stroke 2026, 57(1), 125–33. [Google Scholar] [CrossRef]
- Vitturi, BK; Mitre, LP; Kim, AIH; Gagliardi, RJ. Prevalence and predictors of fatigue and neuropsychiatric symptoms in patients with minor ischemic stroke. J Stroke Cerebrovasc Dis. 2021, 30(9), 105964. [Google Scholar] [CrossRef]
- Randolph, S; Lee, Y; Nicholas, ML; Connor, LT. The mediating effect of anxiety on the association between residual neurological impairment and post-stroke participation among persons with and without post-stroke depression. Neuropsychol Rehabil. 2024, 34(2), 181–95. [Google Scholar] [CrossRef] [PubMed]
- Li, W; Xiao, WM; Chen, YK; Qu, JF; Liu, YL; Fang, XW; et al. Anxiety in patients with acute ischemic stroke: risk factors and effects on functional status. Front Psychiatry 2019, 10, 257. [Google Scholar] [CrossRef] [PubMed]
- Liu, L; Xu, M; Marshall, IJ; Wolfe, CDA; Wang, Y; O’Connell, MDL. Prevalence and natural history of depression after stroke: a systematic review and meta-analysis. PLoS Med. 2023, 20(3), e1004200. [Google Scholar] [CrossRef]
- Naghedi, A; Delgado-Mederos, R; Vives-Bauza, C. Stroke survivors have almost three times higher risk of depression: a systematic review and meta-analysis. J Clin Med. 2025, 14(23), 8410. [Google Scholar] [CrossRef]
- Ebbesen, BH; Modrau, B; Kontou, E; et al. Lasting impairments following transient ischemic attack and minor stroke: a systematic review protocol. Front Neurol. 2023, 14, 1177309. [Google Scholar] [CrossRef]
- Del Bene, VA; Howard, G; Gropen, TI; et al. Cognitive decline after first-time transient ischemic attack. JAMA Neurol. 2025, 82(4). Available online: https://jamanetwork.com/journals/jamaneurology/fullarticle/2830012 (accessed on 29 January 2026). [CrossRef]
- Sonoda, K; Wakabayashi, M. Interventions for treatment of poststroke depression. Am Fam Physician. 2023, 108(5), Online. Available online: https://www.aafp.org/pubs/afp/issues/2023/1100/mbtn-poststroke-depression.html (accessed on 29 January 2026). [PubMed]
| Variable | Cases (n = 90) | Controls (n = 92) | pvalue |
| Demographic characteristics | |||
| Male sex, n (%) | 66 (73.3) | 42 (45.7) | <0.001 |
| Age, years, mean ± SD | 59.3 ± 8.2 | 58.6 ± 7.8 | 0.562 |
| Urban residence, n (%) | 43 (47.8) | 57 (62.0) | 0.055 |
| Educational level, n (%) | 0.005 | ||
| ─ Literate without formal education | 37 (41.1) | 20 (21.7) | |
| ─ Primary or secondary education | 50 (55.6) | 61 (66.3) | |
| ─ University education | 3 (3.3) | 11 (12.0) | |
| Social situation, n (%) | 0.037 | ||
| ─ Good or acceptable | 59 (65.6) | 73 (79.3) | |
| ─ Social risk | 31 (34.4) | 19 (20.7) | |
| Cardiovascular risk factors | |||
| Hypertension, n (%) | 52 (57.8) | 33 (35.9) | 0.003 |
| Dyslipidemia, n (%) | 38 (42.2) | 29 (31.5) | 0.135 |
| Diabetes mellitus, n (%) | 26 (28.9) | 15 (16.3) | 0.042 |
| Atrial fibrillation, n (%) | 10 (11.1) | 8 (8.7) | 0.585 |
| Prior ischemic heart disease, n (%) | 11 (12.2) | 1 (1.1) | 0.002 |
| Current tobacco use, n (%) | 57 (64.0) | 35 (38.0) | <0.001 |
| Alcohol consumption, n (%) | 50 (56.2) | 39 (42.4) | 0.064 |
| Family history | |||
| History of stroke, n (%) | 25 (27.8) | 27 (29.3) | 0.815 |
| History of ischemic heart disease, n (%) | 29 (32.2) | 30 (32.6) | 0.956 |
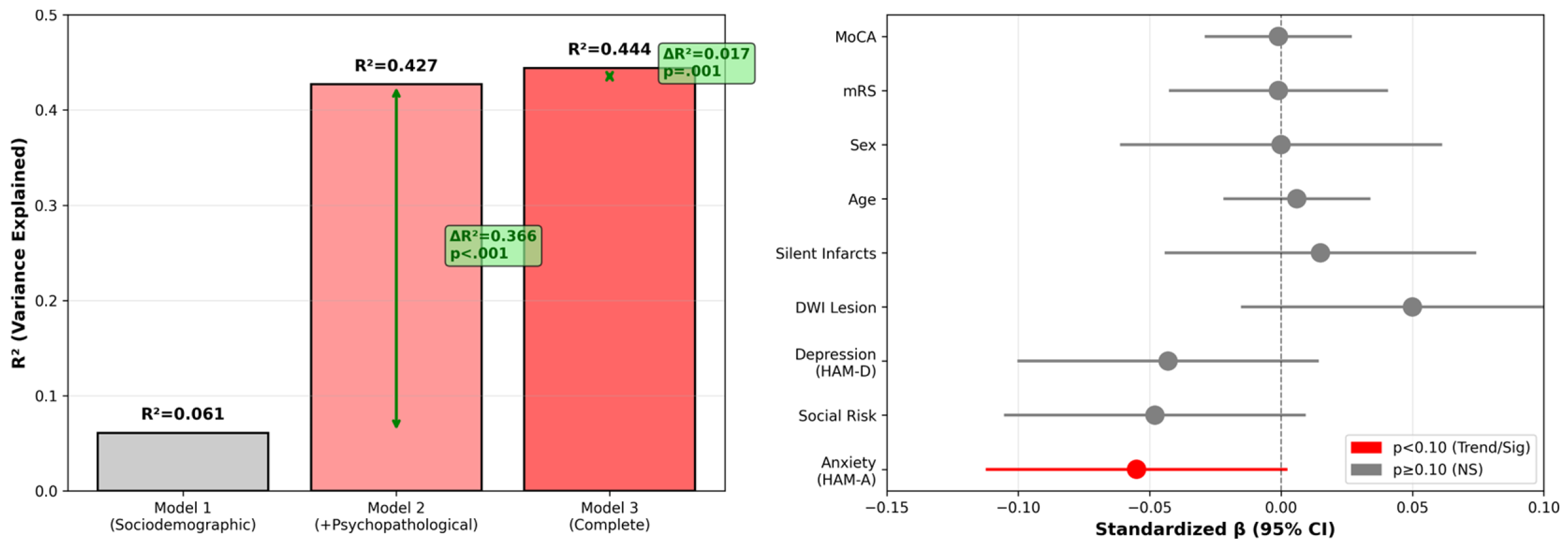
| Model | Predictors included | R2 | Adjusted R2 | Fstatistic | pvalue | ΔR2 |
| Model 1 | Age, sex, mRS, social risk | 0.061 | 0.016 | 1.36 | 0.256 | — |
| Model 2 | Model 1 + HDRS-17, HAM-A | 0.427 | 0.385 | 10.18 | <0.001 | 0.366 |
| Model 3 | Model 2 + MoCA, DWI lesion burden, number of infarcts | 0.444 | 0.381 | 7.02 | <0.001 | 0.017 |
| Predictor | Standardized β | SE | 95% CI | pvalue |
| Anxiety (HAM-A) | −0.055 | 0.029 | −0.114 to 0.003 | 0.064† |
| Social risk | −0.048 | 0.029 | −0.105 to 0.009 | 0.100 |
| Depression (HDRS-17) | −0.043 | 0.029 | −0.102 to 0.016 | 0.147 |
| DWI lesion burden | 0.050 | 0.033 | −0.016 to 0.116 | 0.133 |
| Silent infarcts | 0.015 | 0.030 | −0.046 to 0.075 | 0.627 |
| Age | 0.006 | 0.014 | −0.022 to 0.033 | 0.686 |
| Sex | −0.000 | 0.031 | −0.062 to 0.061 | 0.990 |
| mRS | −0.001 | 0.021 | −0.044 to 0.041 | 0.947 |
| MoCA | −0.001 | 0.014 | −0.029 to 0.027 | 0.947 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




