Submitted:
12 March 2026
Posted:
13 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Study Design and Data Source
Cohort Definitions
Propensity Score Matching
Outcome Definitions and Follow-up
Statistical Analysis
3. Results
Cohort Characteristics
Primary Outcomes
Dementia and Sarcopenia
Parkinson Disease
C-Reactive Protein Elevation
Cataract Positive Control
4. Discussion
Principal Findings
Neuroprotection and the Ambulation Hypothesis
Differential Inflammatory Resolution
Postoperative Delirium, Dementia, and Surgery Type
Implications for the Companion Series
Brain Metal Accumulation and Amyloid Pathology
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BDNF | Brain-Derived Neurotrophic Factor |
| CRP | C-Reactive Protein |
| EHR | Electronic Health Record |
| GDNF | Glial Cell Line-Derived Neurotrophic Factor |
| HCO | Health Care Organization |
| HR | Hazard Ratio |
| ICD-10 | International Classification of Diseases, 10th Revision |
| KM | Kaplan–Meier |
| POD | Postoperative Delirium |
| PSM | Propensity Score Matching |
| SMD | Standardized Mean Difference |
| THA | Total Hip Arthroplasty |
| TJA | Total Joint Arthroplasty |
| TKA | Total Knee Arthroplasty |
| TSR | Total Shoulder Replacement |
References
- Prince, M.; Wimo, A.; Guerchet, M.; Ali, G.C.; Wu, Y.T.; Prina, M. World Alzheimer Report 2015: The Global Impact of Dementia. Alzheimer’s Disease International: London, UK, 2015.
- Dorsey, E.R.; Constantinescu, R.; Thompson, J.P.; Biglan, K.M.; Holloway, R.G.; Kieburtz, K.; Marshall, F.J.; Ravina, B.M.; Schifitto, G.; Siderowf, A.; et al. Projected number of people with Parkinson disease in the most populous nations, 2005 through 2030. Neurology 2007, 68, 384–386. [CrossRef]
- Anker, S.D.; Morley, J.E.; von Haehling, S. Welcome to the ICD-10 code for sarcopenia. J. Cachexia Sarcopenia Muscle 2016, 7, 512–514. [CrossRef]
- Sloan, M.; Premkumar, A.; Sheth, N.P. Projected volume of primary total joint arthroplasty in the U.S., 2014 to 2030. J. Bone Joint Surg. Am. 2018, 100, 1455–1460. [CrossRef]
- Shichman, I.; Roof, M.; Askew, N.; Nherera, L.; Rozell, J.C.; Seyler, T.M.; Schwarzkopf, R. Projections and epidemiology of primary hip and knee arthroplasty in Medicare patients to 2040–2060. JBJS Open Access 2023, 8, e22.00112. [CrossRef]
- Kurtz, S.; Ong, K.; Lau, E.; Mowat, F.; Halpern, M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J. Bone Joint Surg. Am. 2007, 89, 780–785. [CrossRef]
- Robinson, H.; Yassin, M.; Robinson, D.; Qawasmi, F.; Mahamid, A.; Khatib, M. Lower-limb total joint arthroplasty reduces incident dementia and Parkinson disease: A propensity-matched analysis of a national federated electronic health record database. J. Orthop. Surg. Res. 2026, submitted.
- Robinson, D.; Robinson, H.; Yassin, M.; Qawasmi, F.; Mahamid, A.; Khatib, M. Differential neuroprotective effects of total hip versus total knee arthroplasty: A propensity-matched TriNetX analysis. Geriatr. Orthop. 2026, submitted.
- Hshieh, T.T.; Fong, T.G.; Marcantonio, E.R.; Inouye, S.K. Cholinergic deficiency hypothesis in delirium: A synthesis of current evidence. J. Gerontol. A Biol. Sci. Med. Sci. 2008, 63, 764–772. [CrossRef]
- Chen, Z.R.; Huang, J.B.; Yang, S.L.; Hong, F.F. Role of cholinergic signaling in Alzheimer’s disease. Molecules 2022, 27, 1816. [CrossRef]
- Inacio, M.C.S.; Hansen, C.; Pratt, N.L.; Graves, S.E.; Roughead, E.E. Risk factors for persistent and new chronic opioid use in patients undergoing total hip arthroplasty: A retrospective cohort study. BMJ Open 2016, 6, e010664. [CrossRef]
- Coscia, A.C.; Matar, R.N.; Espinal, E.E.; Shah, N.S.; Grawe, B.M. Does preoperative diagnosis impact patient outcomes following reverse total shoulder arthroplasty? A systematic review. J. Shoulder Elbow Surg. 2021, 30, 1458–1470. [CrossRef]
- Viramontes, O.; Luan Erfe, B.M.; Erfe, J.M.; Brovman, E.Y.; Boehme, J.; Bader, A.M.; Bateman, B.T. Cognitive impairment and postoperative outcomes in patients undergoing primary total hip arthroplasty: A systematic review. J. Clin. Anesth. 2019, 54, 121–134. [CrossRef]
- Ahluwalia, S.S.; Lugo, J.D.; Gordon, A.M.; Golub, I.J.; Razi, A.E.; Feliccia, J. The association of dementia on perioperative complications following primary total hip arthroplasty for femoral neck fractures. Eur. J. Orthop. Surg. Traumatol. 2023, 33, 971–976. [CrossRef]
- Hernandez, N.M.; Cunningham, D.J.; Jiranek, W.A.; Bolognesi, M.P.; Seyler, T.M. Total hip arthroplasty in patients with dementia. J. Arthroplasty 2020, 35, 1667–1670. [CrossRef]
- Ebner, B.A.; Erdahl, S.A.; Lundgreen, C.S.; Bui, B.N.; Bao, X.; Bhatt, J.K.; Ecklund, K.A.; Holley, A.S.; Jentoft, M.E.; Hines, S.L.; et al. Brain tissue metal concentrations and Alzheimer’s disease neuropathology in total joint arthroplasty patients versus controls. Acta Neuropathol. 2025, 149, 18. [CrossRef]
- Ahlskog, J.E.; Geda, Y.E.; Graff-Radford, N.R.; Petersen, R.C. Physical exercise as a preventive or disease-modifying treatment of dementia and brain aging. Mayo Clin. Proc. 2011, 86, 876–884. [CrossRef]
- Cotman, C.W.; Berchtold, N.C.; Christie, L.A. Exercise builds brain health: Key roles of growth factor cascades and inflammation. Trends Neurosci. 2007, 30, 464–472. [CrossRef]
- Wang, P.; Velagapudi, R.; Kong, C.; Bhatt, D.L.; Bhatt, D.L.; Tang, Y.; Bhatt, D.L. Neurovascular and immune mechanisms that regulate postoperative delirium superimposed on dementia. Alzheimers Dement. 2020, 16, 734–749. [CrossRef]
- Price, C.C.; Levy, S.A.; Tanner, J.; Schmalfuss, I.; Lamar, M.; Heilman, K.M.; Bhatt, D.L. Orthopedic surgery and post-operative cognitive decline in idiopathic Parkinson disease: Considerations from a pilot study. J. Parkinsons Dis. 2015, 5, 893–905. [CrossRef]
- Leng, K.; Maze, M.; Barreto Chang, O.L. Emerging biomarkers of postoperative delirium at the intersection of neuroinflammation and neurodegeneration. Front. Aging Neurosci. 2025, 17, 1632947. [CrossRef]
- Hampel, H.; Mesulam, M.M.; Cuello, A.C.; Farlow, M.R.; Giacobini, E.; Grossberg, G.T.; Bhatt, D.L. The cholinergic system in the pathophysiology and treatment of Alzheimer’s disease. Brain 2018, 141, 1917–1933. [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscles, exercise and obesity: Skeletal muscle as a secretory organ. Nat. Rev. Endocrinol. 2012, 8, 457–465. [CrossRef]
- Pedersen, B.K. Physical activity and muscle–brain crosstalk. Nat. Rev. Neurosci. 2019, 20, 419–430. [CrossRef]
- Perez-Lloret, S.; Barrantes, F.J. Deficits in cholinergic neurotransmission and their clinical correlates in Parkinson’s disease. NPJ Parkinsons Dis. 2016, 2, 16001. [CrossRef]
- Kunicki, Z.J.; Ngo, L.H.; Marcantonio, E.R.; Jones, R.N.; Habtemariam, D.; Fong, T.G.; Bhatt, D.L. Six-year cognitive trajectory in older adults following major surgery and delirium. JAMA Intern. Med. 2023, 183, 442–450. [CrossRef]
- Vasunilashorn, S.M.; Ngo, L.H.; Inouye, S.K.; Libermann, T.A.; Jones, R.N.; Alsop, D.C.; Bhatt, D.L. Postoperative delirium is associated with increased plasma neurofilament light. Ann. Neurol. 2020, 88, 984–994.
- Rong, X.; Ding, Z.C.; Yu, H.D.; Yao, S.Y.; Zhou, Z.K. Risk factors of postoperative delirium in the knee and hip replacement patients: A systematic review and meta-analysis. J. Orthop. Surg. Res. 2021, 16, 76. [CrossRef]
- Inouye, S.K.; Westendorp, R.G.; Saczynski, J.S. Delirium in elderly people. Lancet 2014, 383, 911–922. [CrossRef]
- Zhao, Y.; Liang, G.; Hong, K.; Pan, J.; Luo, M.; Liu, J.; Bhatt, D.L. Risk factors for postoperative delirium following total hip or knee arthroplasty: A meta-analysis. Front. Psychol. 2022, 13, 993136. [CrossRef]
- Johnson, A.H.; Brennan, J.C.; King, P.J.; Turcotte, J.J.; MacDonald, J.H. Comparison of postoperative outcomes of patients undergoing total hip and total knee arthroplasty following a diagnosis of dementia: A TriNetX database study. Arthroplast. Today 2024, 27, 101359. [CrossRef]
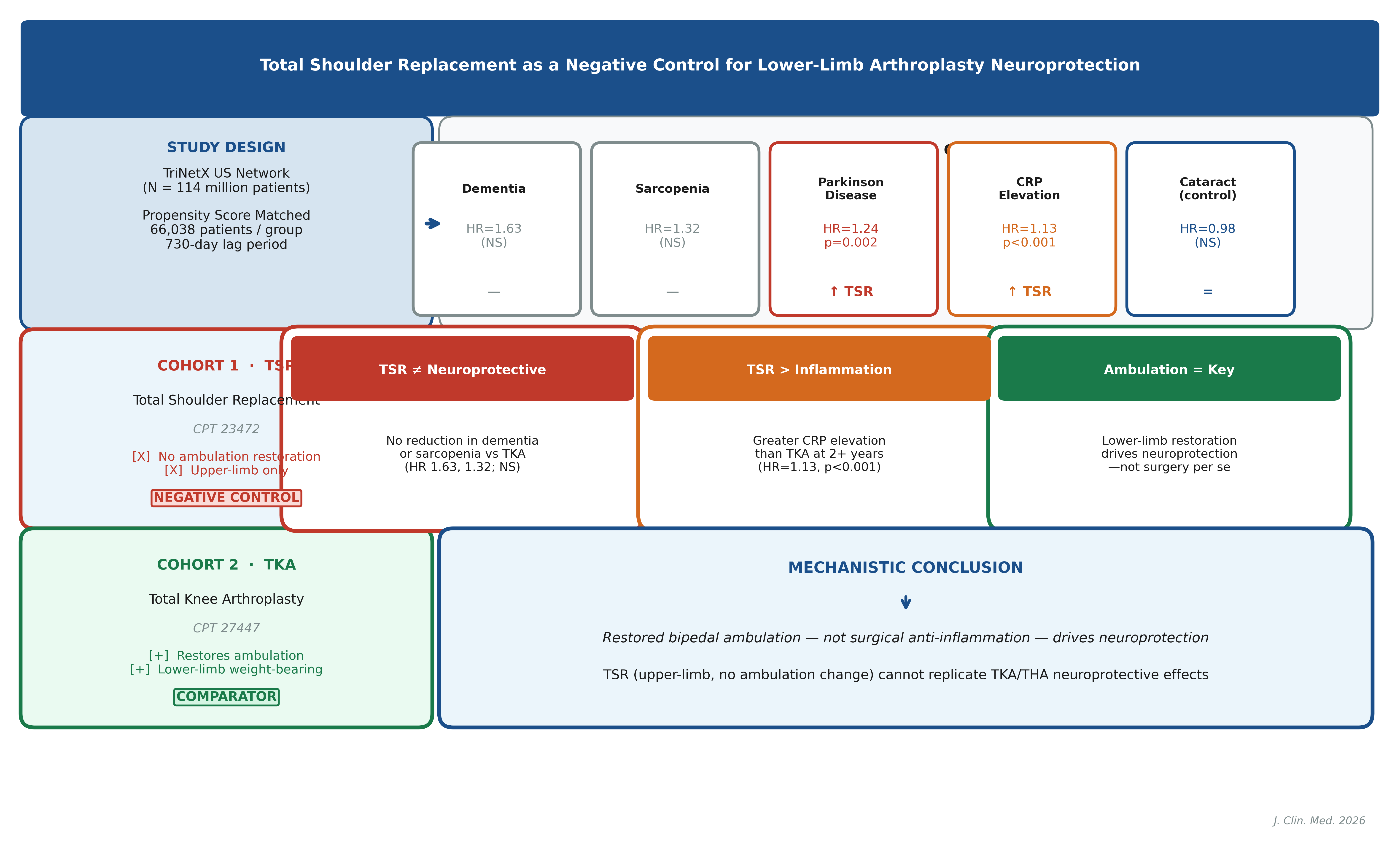
| Outcome | TSR Events (n = 66,038) | TKA Events (n = 66,038) | Hazard Ratio (95% CI) | p-Value |
|---|---|---|---|---|
| Incident Dementia | 15 | 11 | 1.63 (0.75–3.55) | 0.217 |
| Incident Sarcopenia | 22 | 19 | 1.32 (0.72–2.45) | 0.369 |
| Incident Parkinson Disease * | 457 | 401 | 1.24 (1.08–1.41) | 0.002 |
| Elevated CRP * | 1665 | 1655 | 1.13 (1.06–1.21) | <0.001 |
| Cataract (positive control) | 823 | 937 | 0.98 (0.89–1.08) | 0.698 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




