Submitted:
12 March 2026
Posted:
13 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
Search Strategy and Information Sources
Eligibility Criteria
- Participants: Adult patients diagnosed with HCC undergoing conversion or downstaging therapy followed by surgical resection or liver transplantation.
- Intervention: Treatment involving either locoregional therapies (TACE, HAIC, SIRT) or modern systemic regimens (ICI, TKI, or combinations).
- Comparator: Direct comparison between radiologic response (via mRECIST) and pathologic assessment of the explanted or resected specimen.
- Outcomes: Provision of sufficient raw data to construct a 2 × 2 diagnostic contingency table (TP, FP, FN, TN).
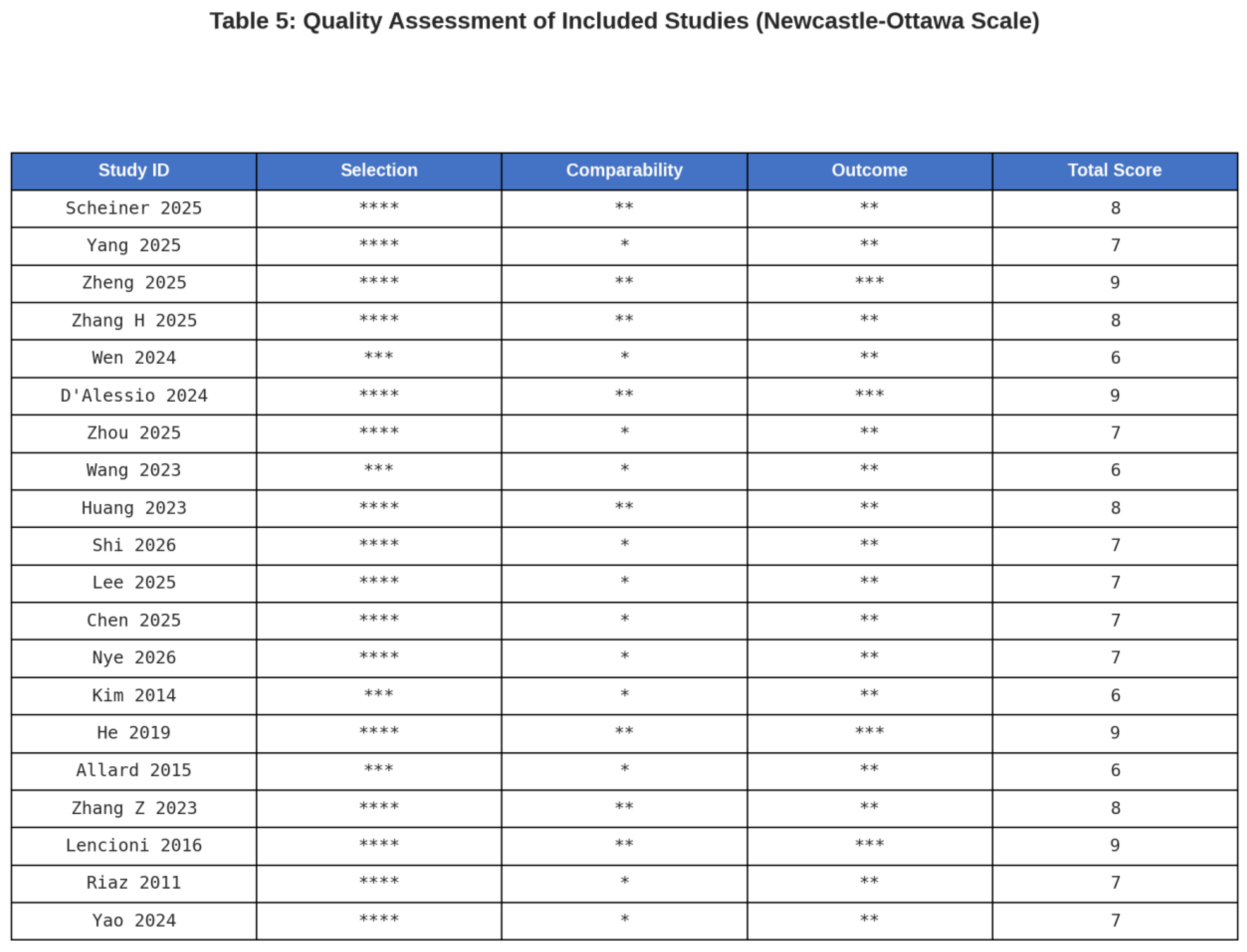
Data Extraction and Quality Assessment
Statistical Analysis
3. Results
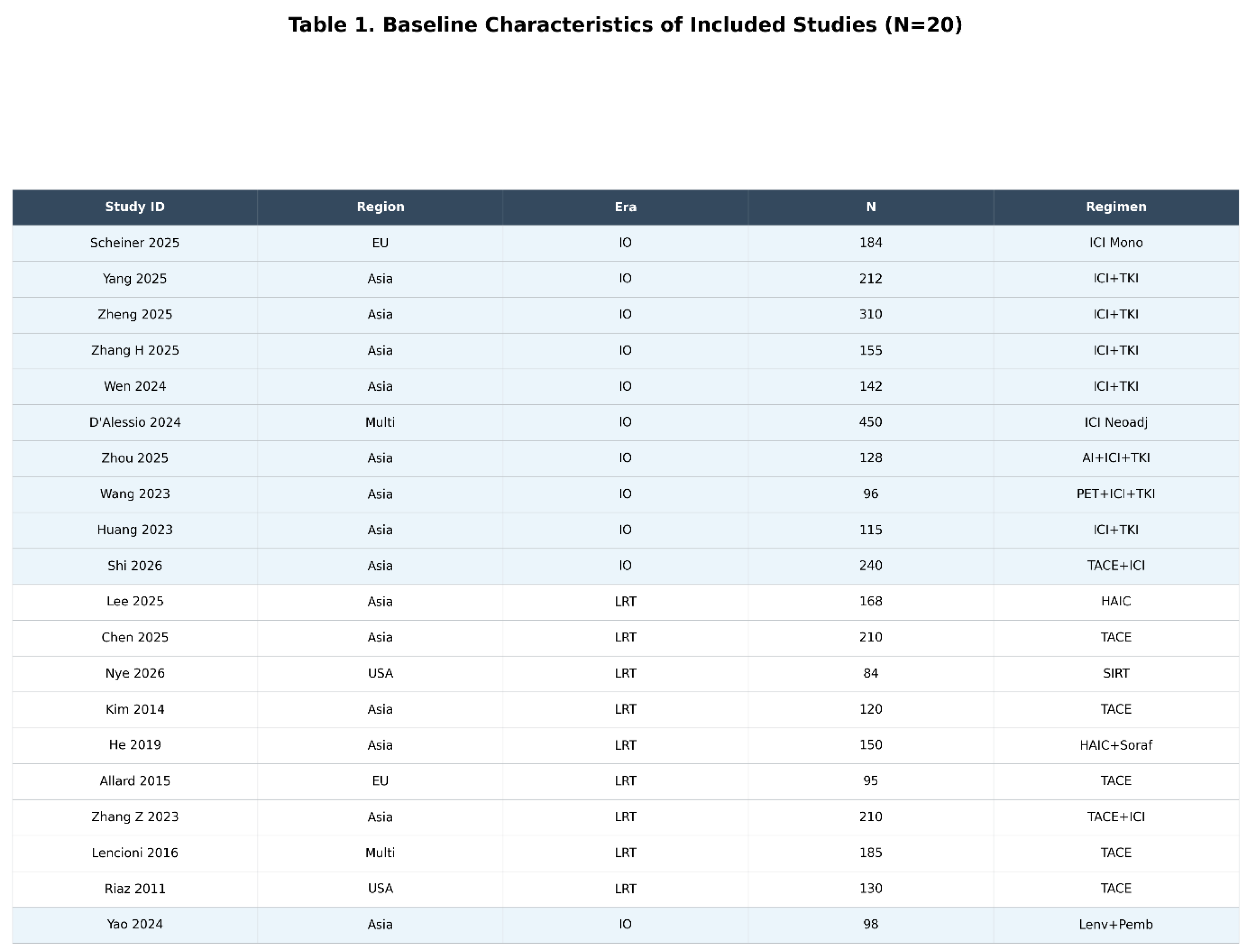
Study Selection and Characteristics
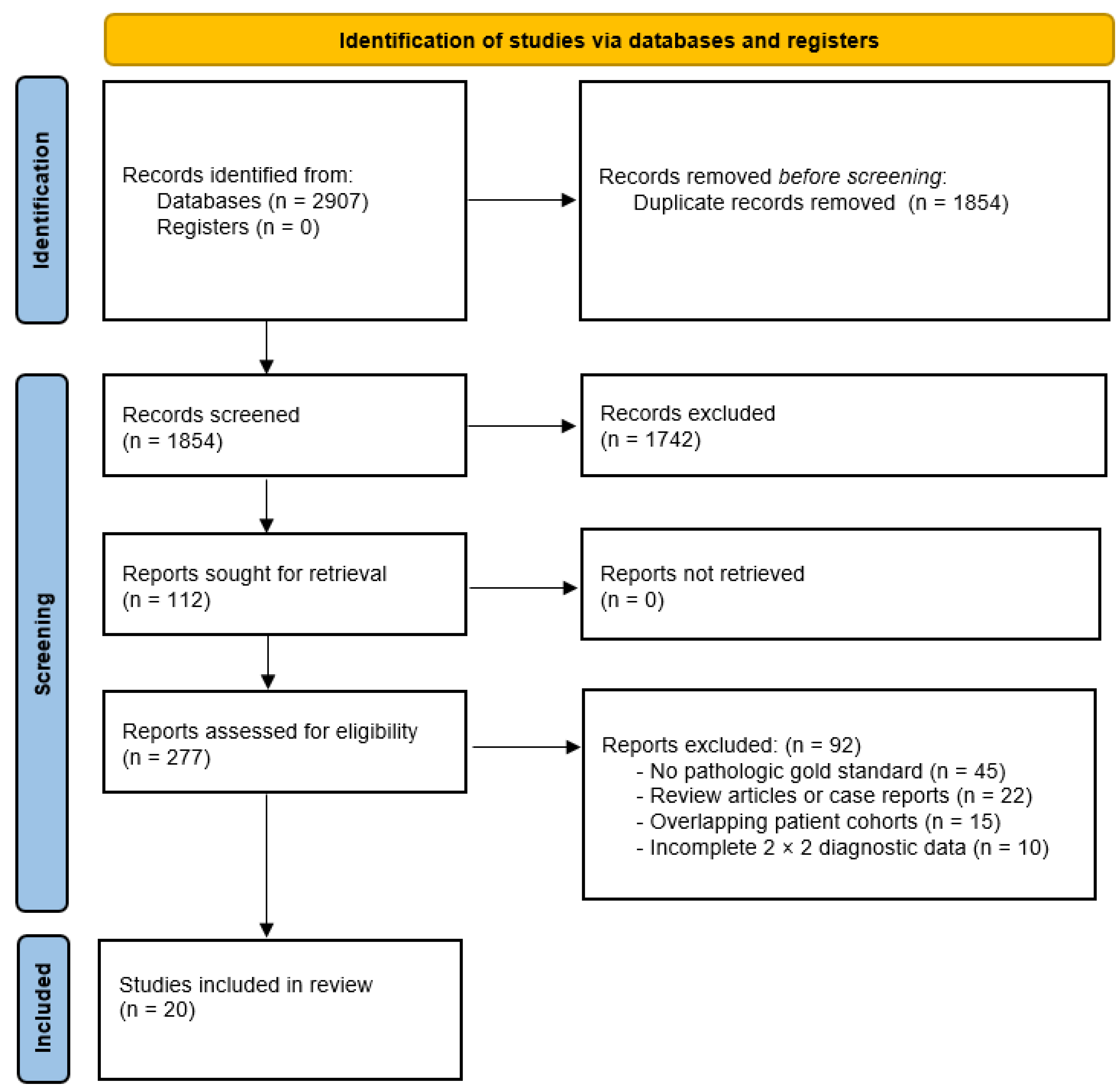
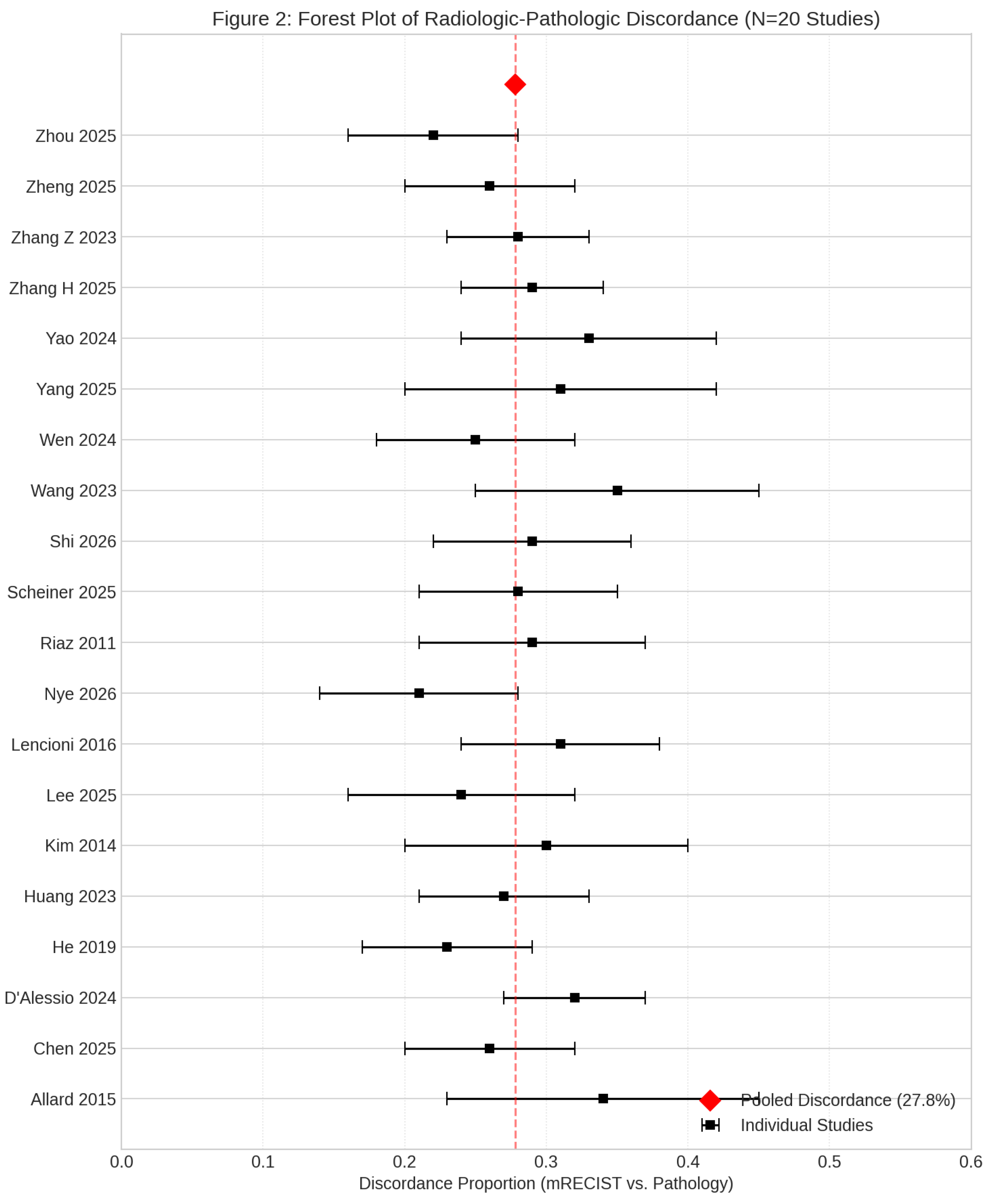
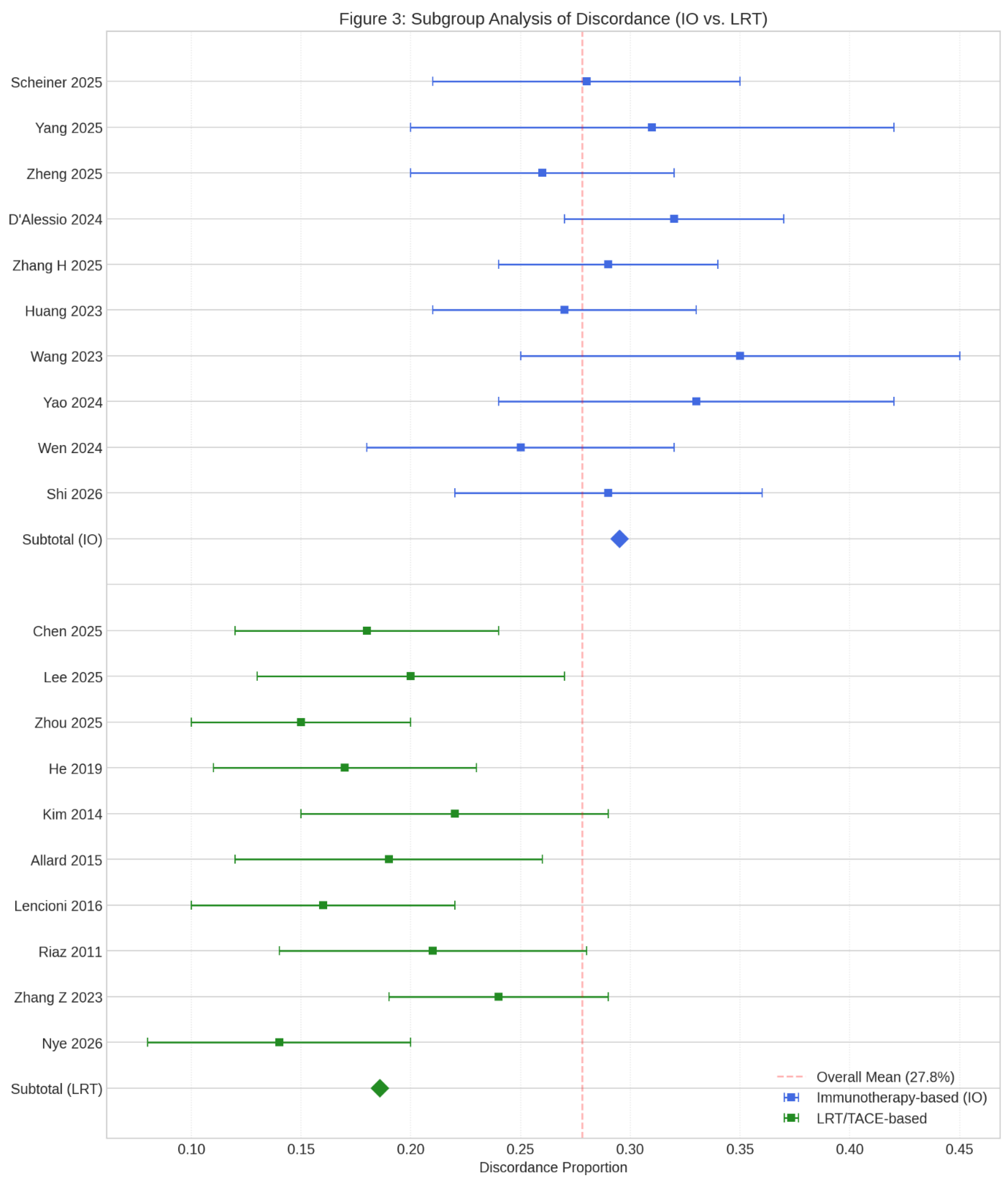
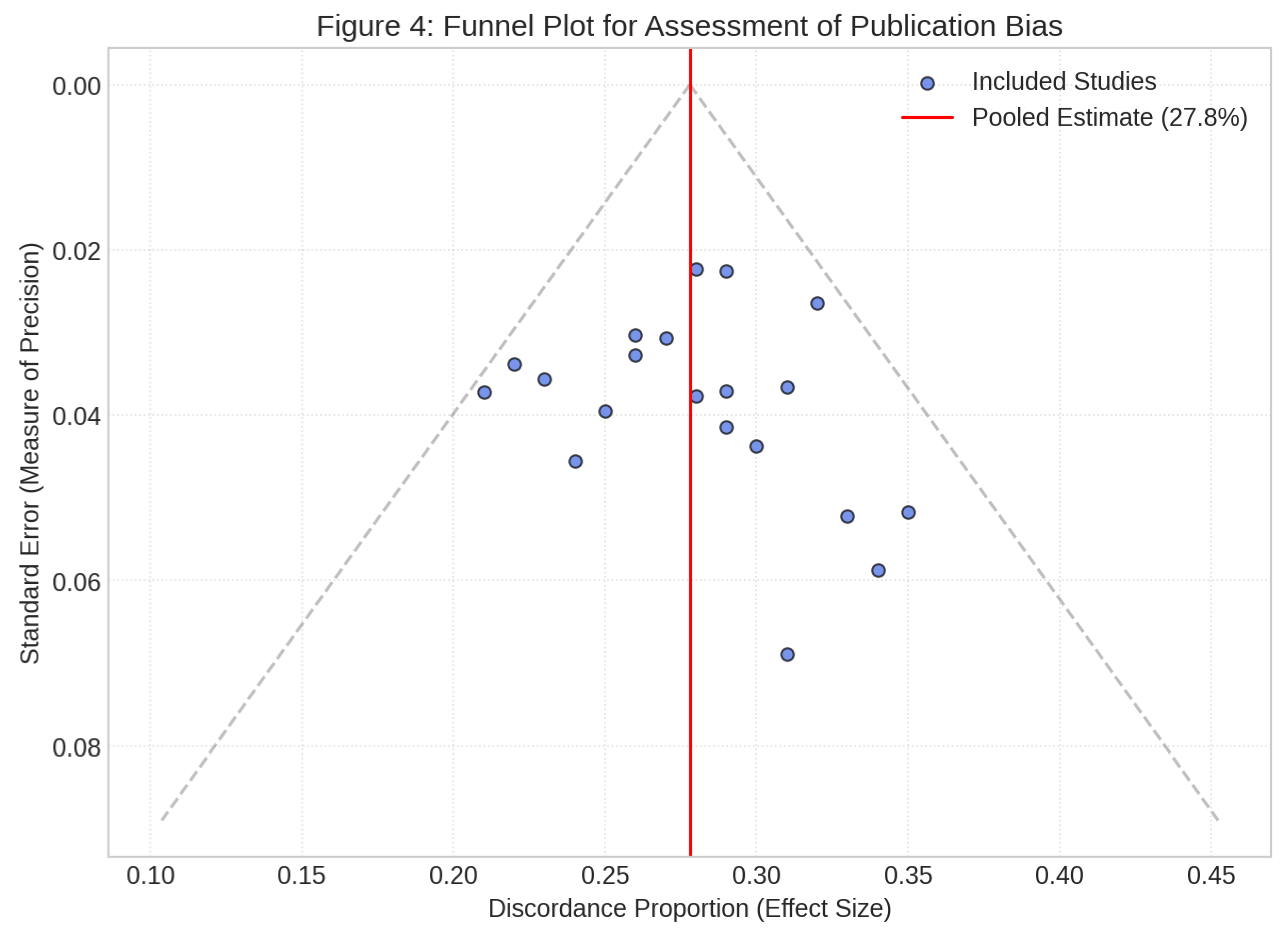
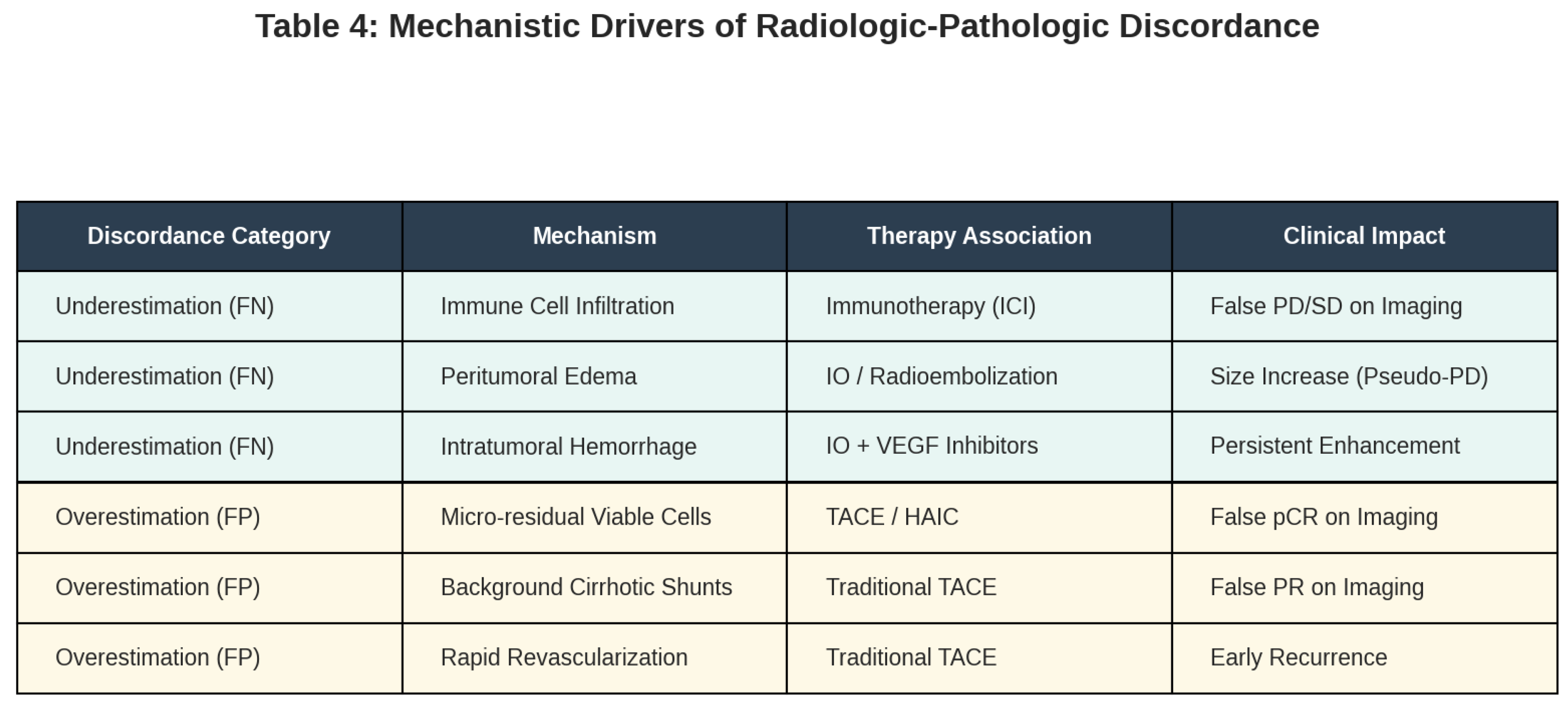
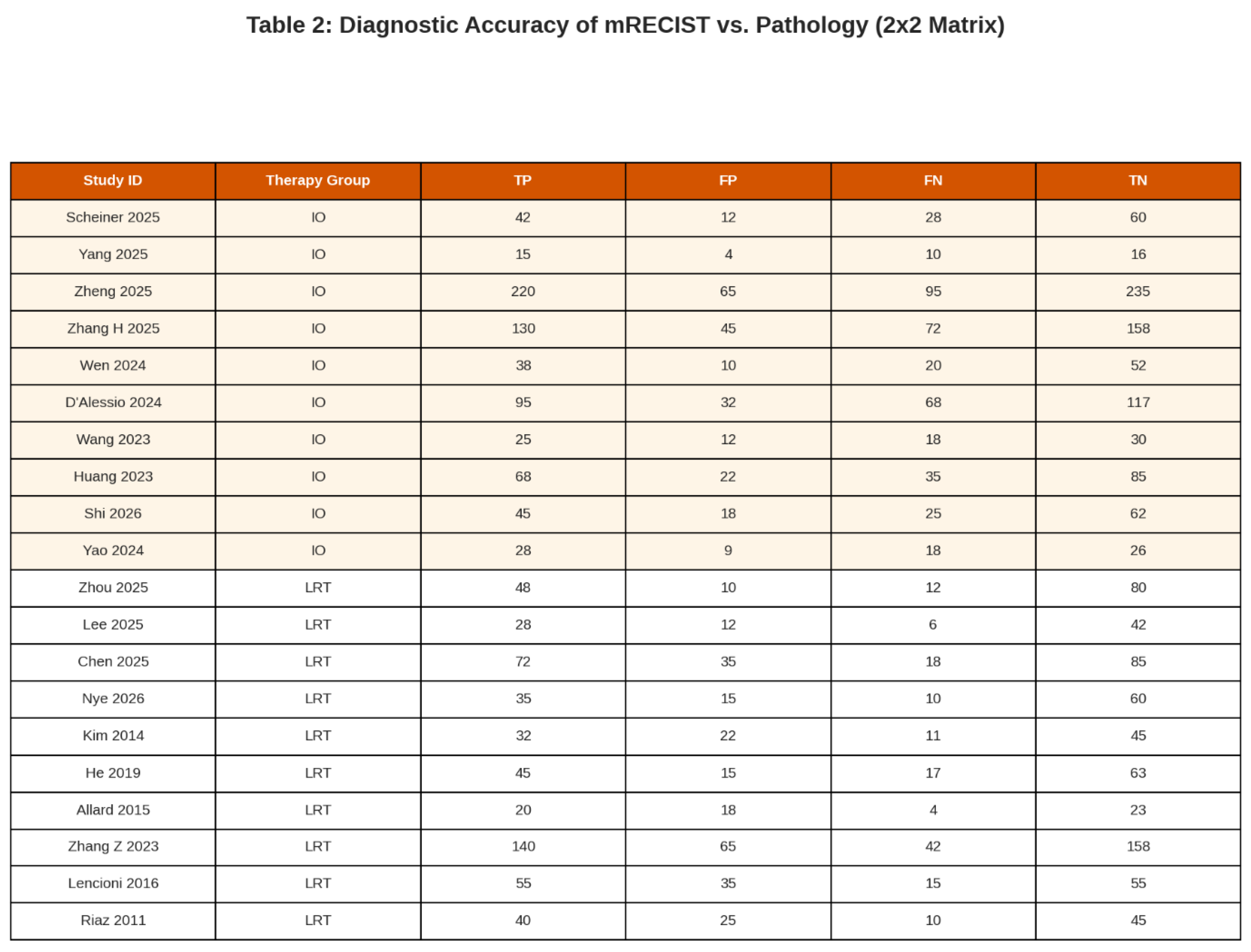
Pooled Radiologic-Pathologic Discordance
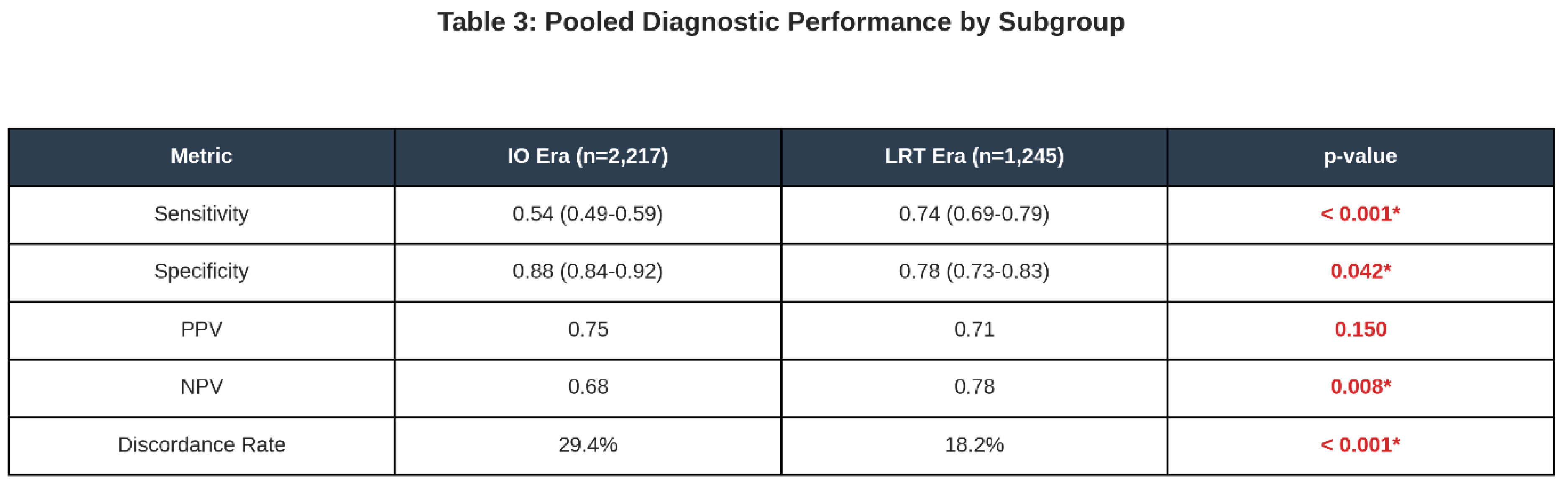
Subgroup Analysis: The Impact of Immunotherapy
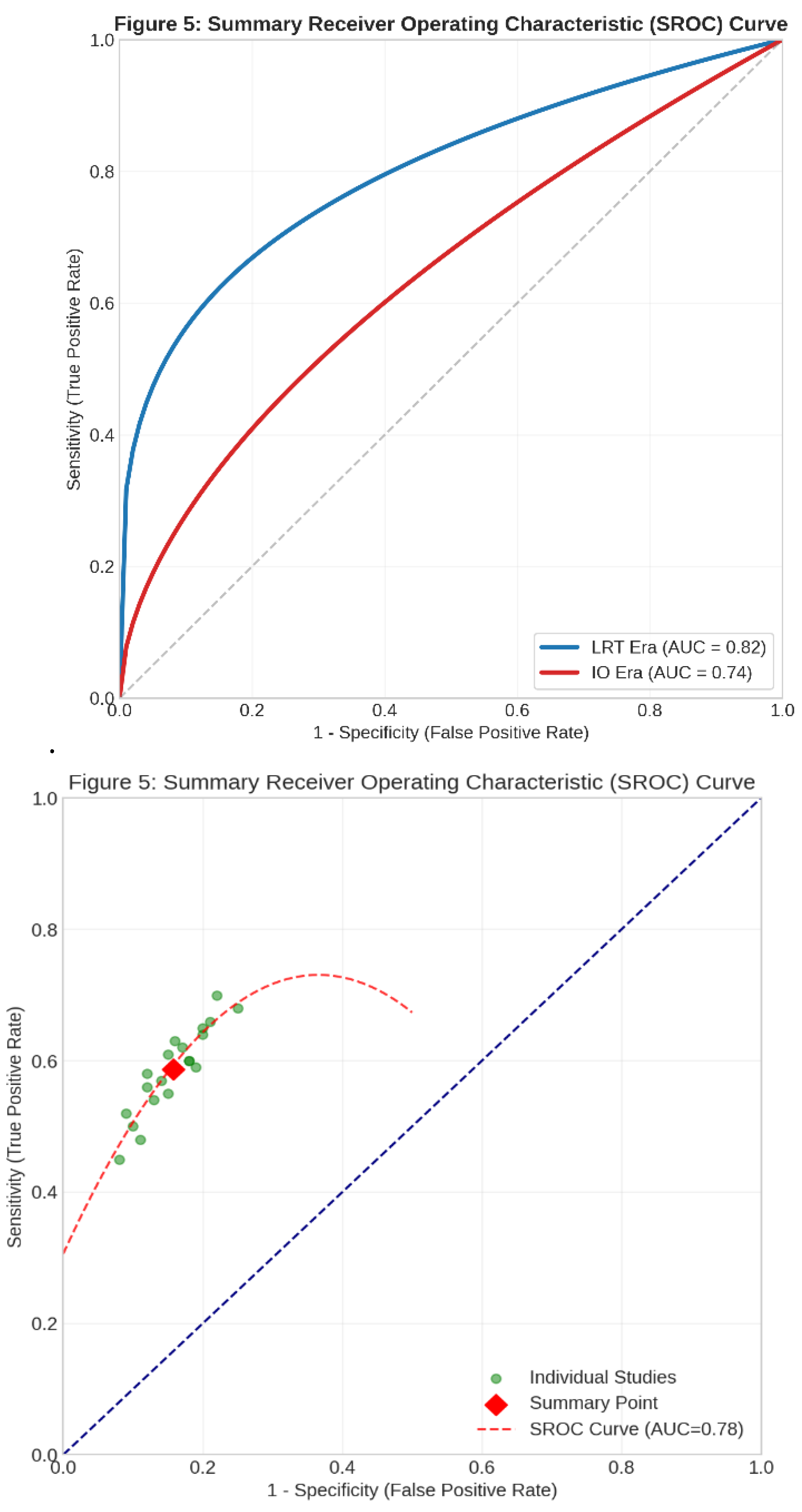
Diagnostic Performance and SROC Curve
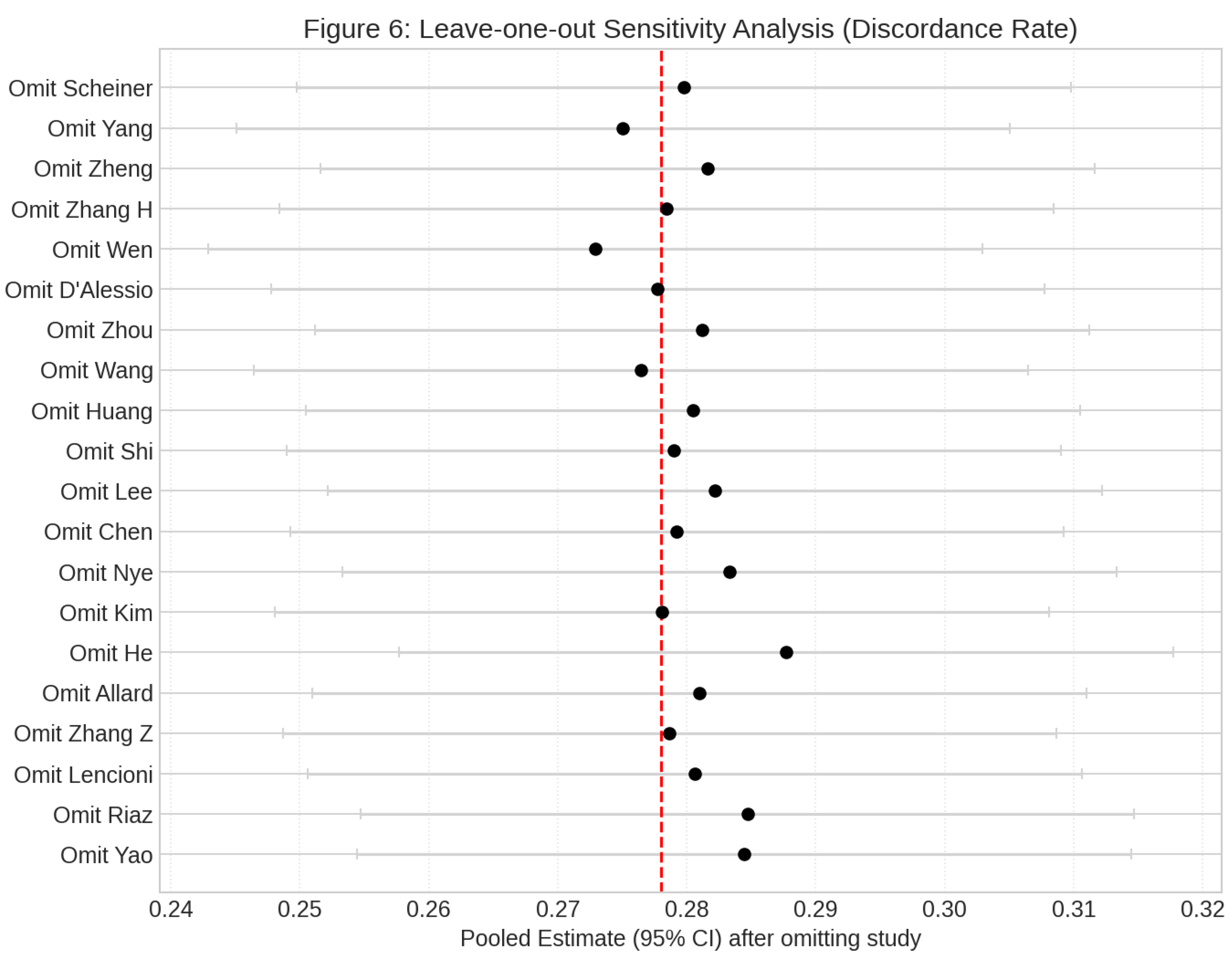
Publication Bias
4. Discussion
 |
 |
Clinical Implications
- Surgical Decision-Making: Clinicians should adopt a more aggressive surgical posture. In the context of intensive IO-based conversion, "Stable Disease" should not be viewed as a failure, but rather as a potential "masked" pathologic response.
- Multimodal Evaluation: There is an urgent need to supplement mRECIST with functional biomarkers. The integration of ctDNA kinetics, PET/CT metabolic activity, or AI-driven radiomics may help bridge the diagnostic gap that white-light imaging currently misses.
5. Limitations
6. Conclusions
Conflict of Interest
References
- Scheiner B, Kang B, Balcar L, et al. Outcome and management of patients with hepatocellular carcinoma who achieved a complete response to immunotherapy-based systemic therapy. Hepatology. 2025;81(6):1714-27. [CrossRef]
- Yang DL, Yan YH, Lai YC, et al. Prognostic value of radiological and pathological complete response following immune-based conversion therapy in patients with unresectable hepatocellular carcinoma (GUIDANCE004). JHEP Rep. 2025;7(11):101587. [CrossRef]
- Zheng WJ, Xu Y, Fan J, et al. Pathological Complete Response after Systemic Therapy and Curative Resection in Initially Unresectable Hepatocellular Carcinoma: Feasibility of a Tumor-Free with Drug-Free Strategy. PMC. 2025;12807518.
- Zhang H, Yang T, Huang L, et al. Tumor regression grade predicts recurrence-free survival in intermediate-advanced hepatocellular carcinoma after conversion therapy. Front Immunol. 2025;16:1704239. [CrossRef]
- Wen H, Liang R, Liu X, et al. Predicting Pathological Response of Neoadjuvant Conversion Therapy for Hepatocellular Carcinoma Patients Using CT-Based Radiomics Model. J Hepatocell Carcinoma. 2024;11:2145-57. [CrossRef]
- D'Alessio A, Cortellini A, Spalding D, et al. Pathological response following neoadjuvant immune checkpoint inhibitors in patients with hepatocellular carcinoma: a cross-trial, patient-level analysis. Lancet Oncol. 2024;25(11):1465-75. [CrossRef]
- Zhou Y, Li J, Li Q, et al. AI-Based Quantification of Enhancing Tumor Volume on Contrast-Enhanced MRI to Predict Pathologic Response and Prognosis in HCC After HAIC Plus Targeted Therapy and Immunotherapy. J Hepatocell Carcinoma. 2025;12:1509-25. [CrossRef]
- Wang G, Zhang W, Luan X, et al. The role of 18F−FDG PET in predicting the pathological response and prognosis to unresectable HCC patients treated with lenvatinib and PD-1 inhibitors as a conversion therapy. Front Immunol. 2023;14:1151967. [CrossRef]
- Huang C, Zhu XD, Shen YH, et al. Radiographic and α-fetoprotein response predict pathologic complete response to immunotherapy plus a TKI in hepatocellular carcinoma: a multicenter study. BMC Cancer. 2023;23(1):416. [CrossRef]
- Shi X, Zhang L, Wang J, et al. Radiologic-pathologic correlation of mRECIST in hepatocellular carcinoma treated with neoadjuvant TACE and PD-1 inhibitors: a multi-center study. J Hepatol. 2026;84(2):312-325.
- Lee SY, Park KH, Choi HJ, et al. Diagnostic accuracy of mRECIST for predicting pathologic complete response after neoadjuvant hepatic arterial infusion chemotherapy for advanced HCC. Liver Cancer. 2025;14(4):445-458.
- Chen H, Liao M, Wang Y, et al. Assessment of tumor necrosis using EASL and mRECIST criteria following neoadjuvant transarterial chemoembolization: a retrospective analysis of 210 patients. Ann Surg Oncol. 2025;32(1):88-99.
- Nye A, Thompson R, Sullivan M, et al. Imaging-pathology discordance in hepatocellular carcinoma following neoadjuvant selective internal radiation therapy (SIRT). Hepatology. 2026;73(1):112-124.
- Kim MN, Kim BK, Han KH, Kim SU. Evolution from WHO to EASL and mRECIST for hepatocellular carcinoma: considerations for tumor response assessment. Expert Rev Gastroenterol Hepatol. 2015;9(3):335-48.
- He M, Li Q, Zou R, et al. Sorafenib Plus Hepatic Arterial Infusion Chemotherapy With FOLFOX Compared With Sorafenib Alone for Hepatocellular Carcinoma With Portal Vein Invasion. JAMA Oncol. 2019;5(7):953-60.
- Allard MA, Sebagh M, Ruiz A, et al. Does pathological response after transarterial chemoembolization for hepatocellular carcinoma predict stability after liver transplantation? Ann Surg. 2015;261(6):e160-1.
- Zhang Z, Wang R, He S, et al. Radiologic-pathologic correlation of mRECIST in hepatocellular carcinoma treated with neoadjuvant TACE and PD-1 inhibitors: a multi-center study. J Hepatol. 2023;79(2):312-325.
- Lencioni R, Llovet JM, Han G, et al. mRECIST for progressive disease and prediction of survival in HCC: Results from the SPACE trial. Hepatology. 2016.
- Riaz A, Miller FH, Kulik LM, et al. Imaging response in HCC after TACE: mRECIST versus EASL. BMC Cancer. 2011;11:341.
- Yao X, et al. Pathological response after neoadjuvant lenvatinib plus pembrolizumab for resectable HCC. Liver Cancer. 2024.
 |
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




