Submitted:
11 March 2026
Posted:
12 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Material and Methods
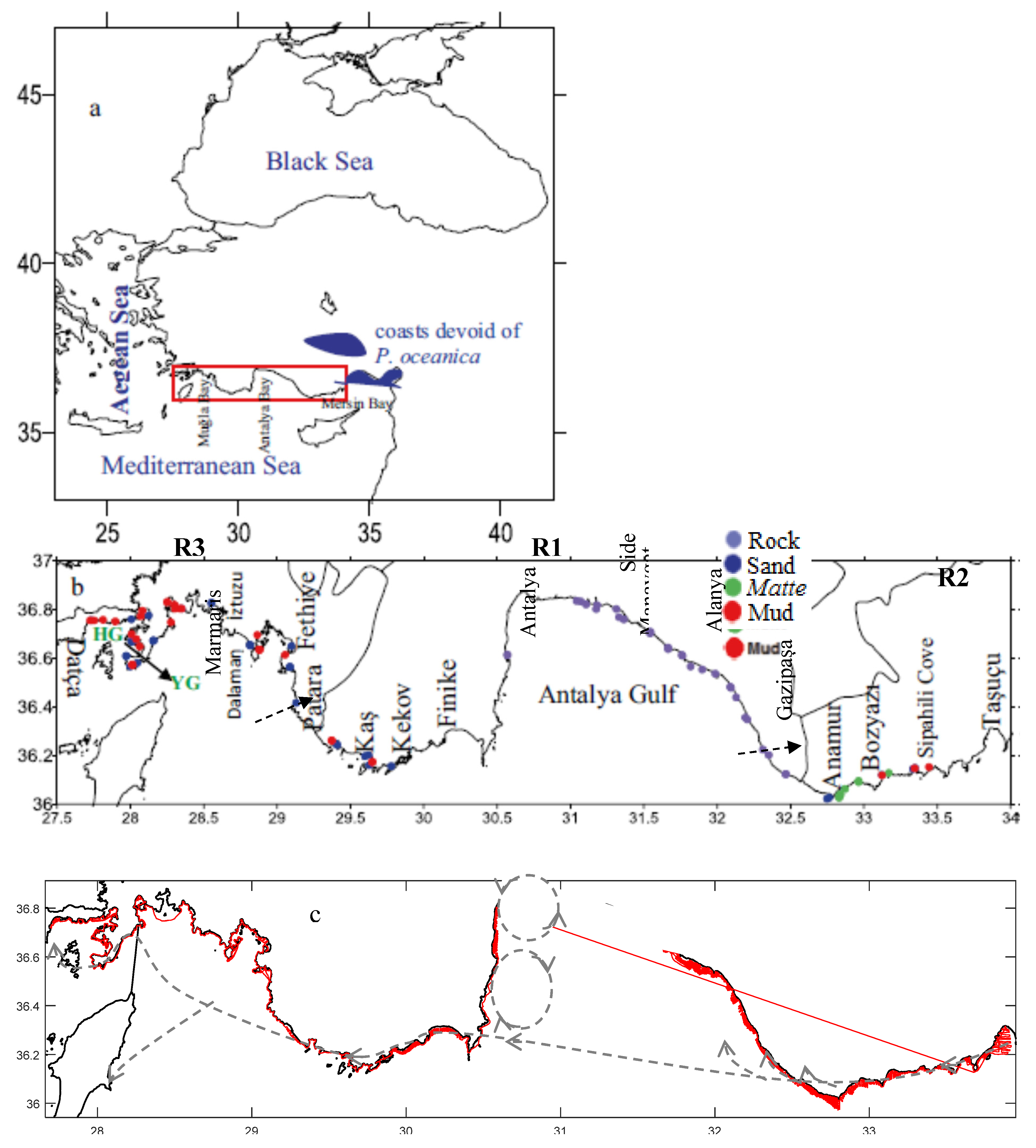
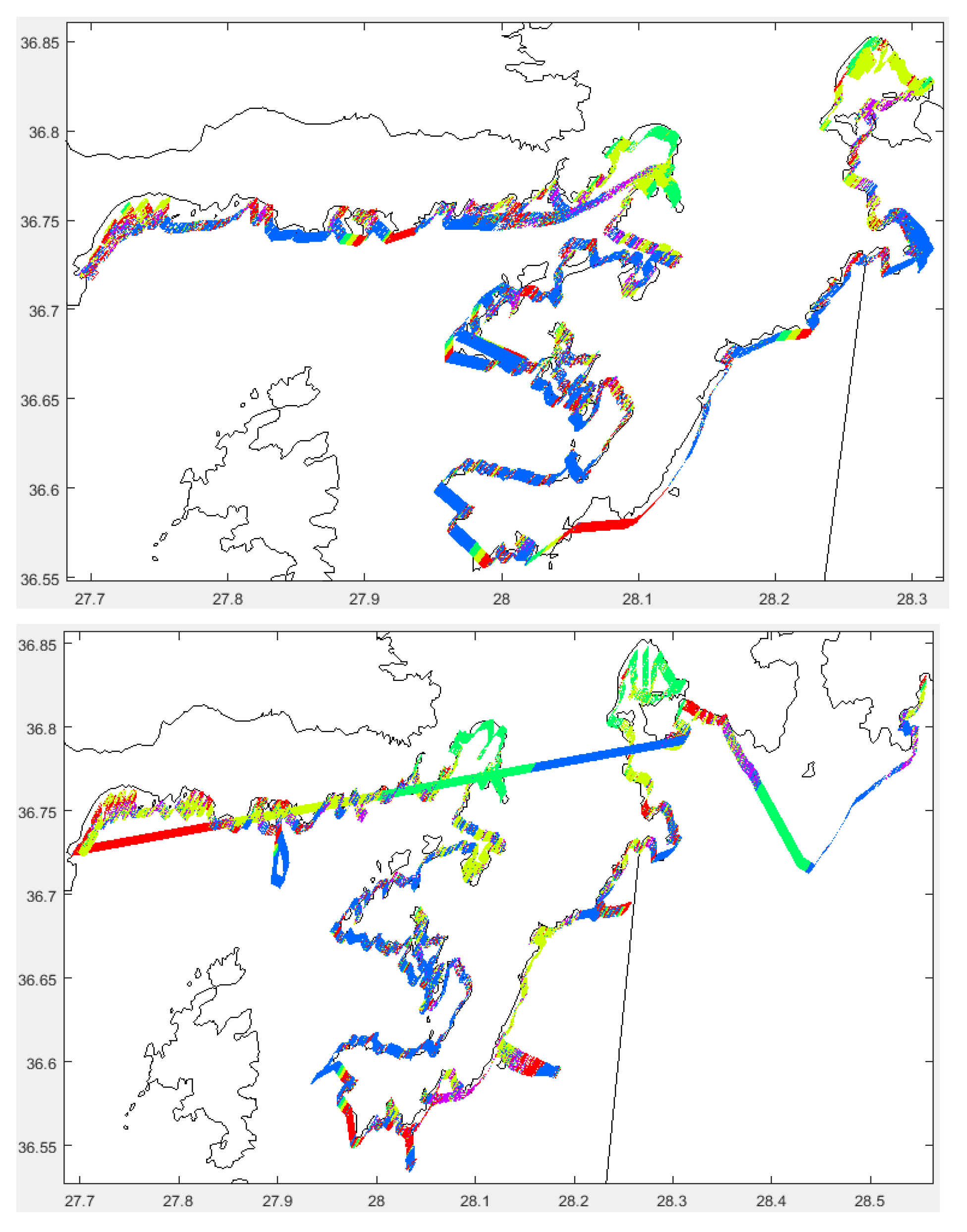
2.1. Study Area Environment
2.2.1. Water Physics
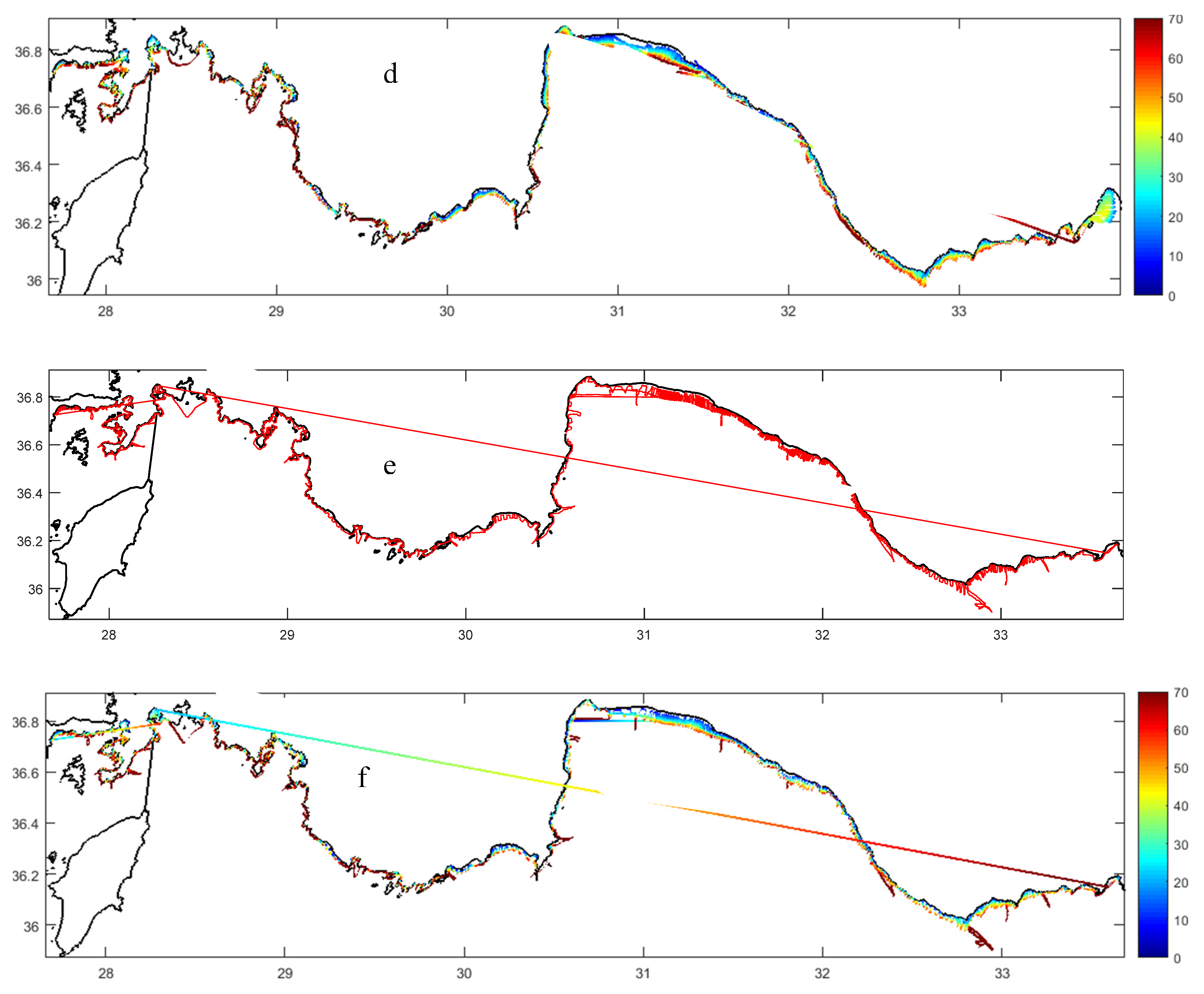
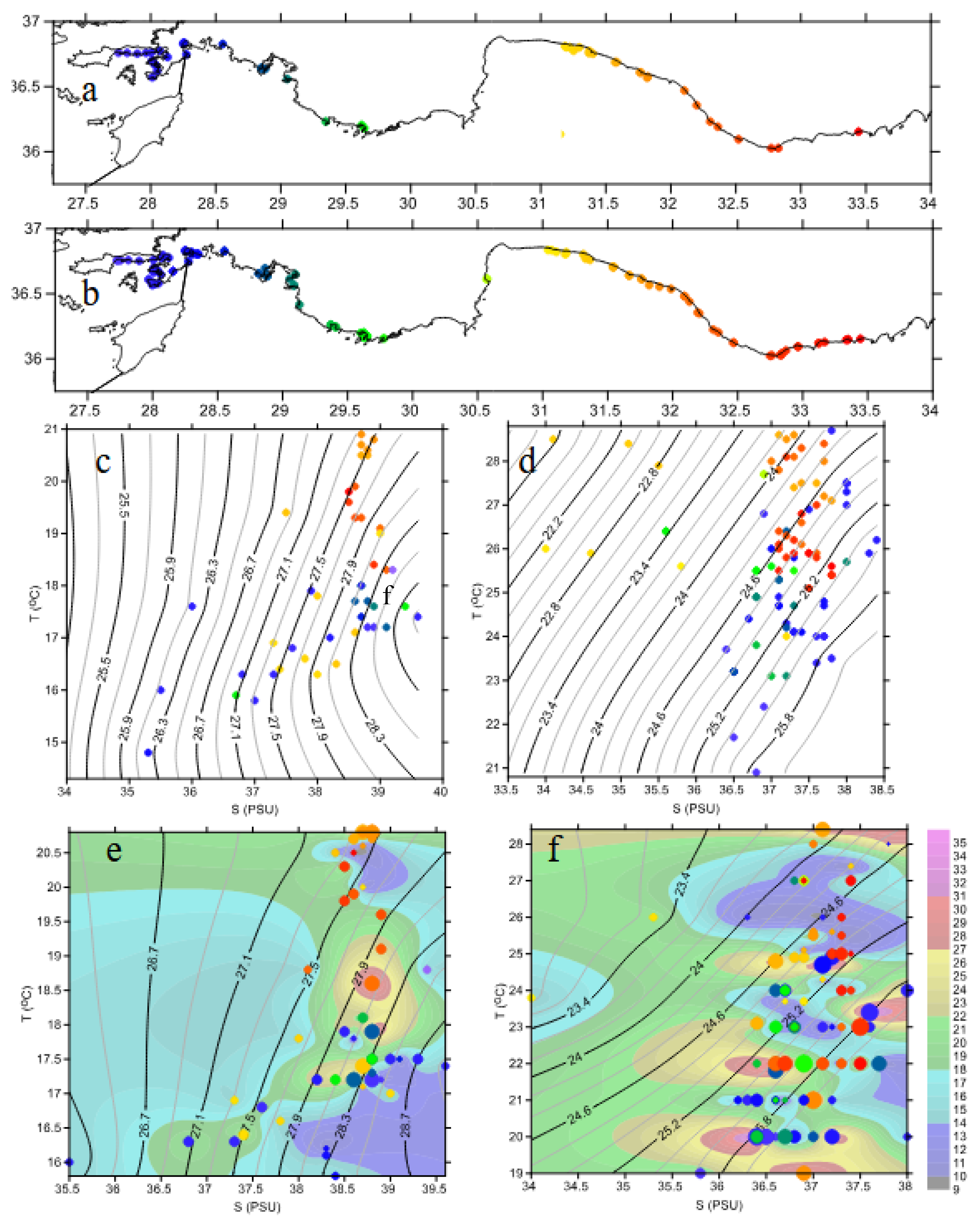
2.2.2. Water Masses
2.2.3. Water Chemistry
2.2.4. Water Optics
2.2.5. General Characteristics
2.2. Acoustical Sampling
2.3. Data Analyses
2.4. Bottom Types and Habitats
2.5. Conversion of Biomass to Biometrics
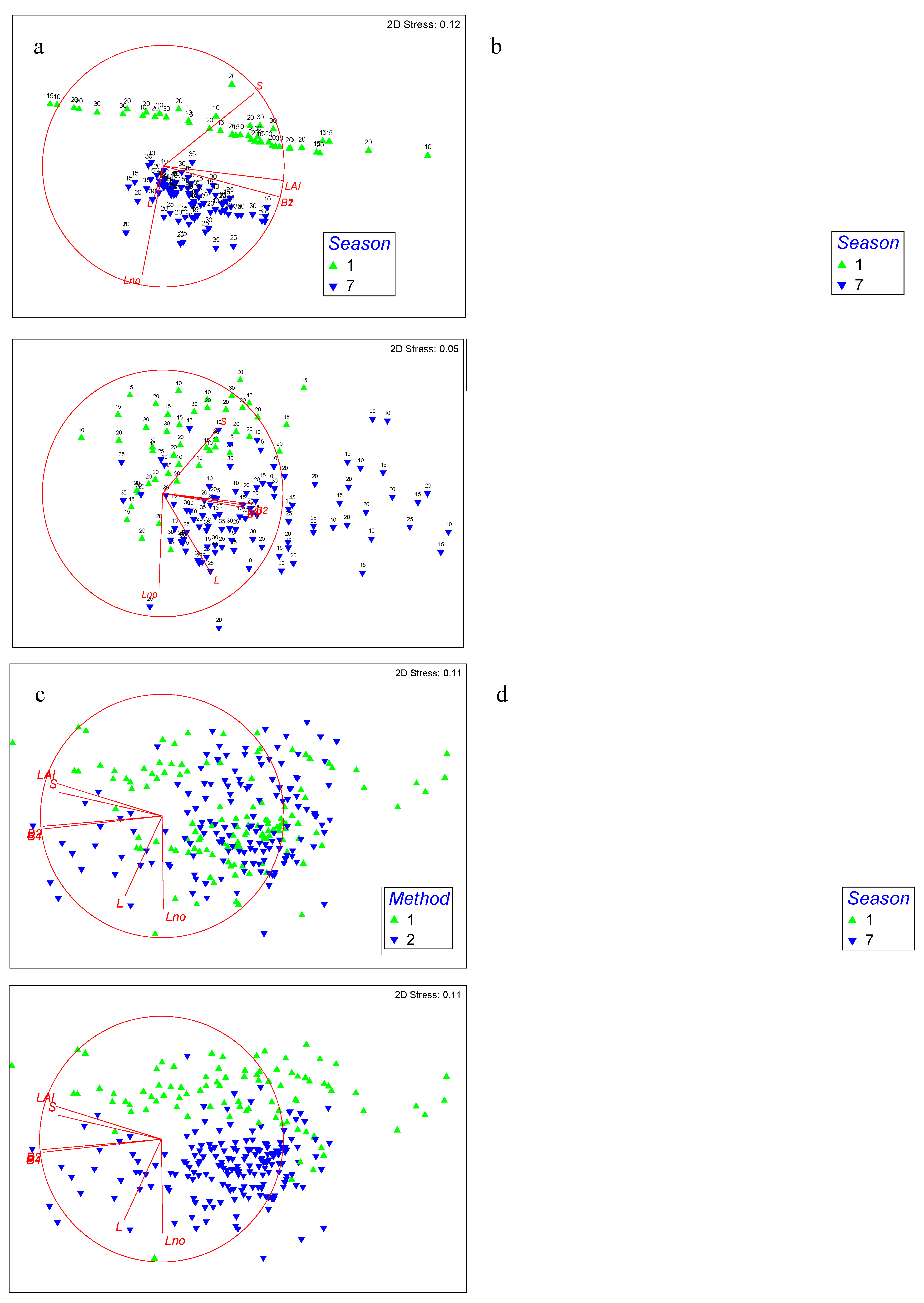
2.6. Statistical Analyses
3. Results
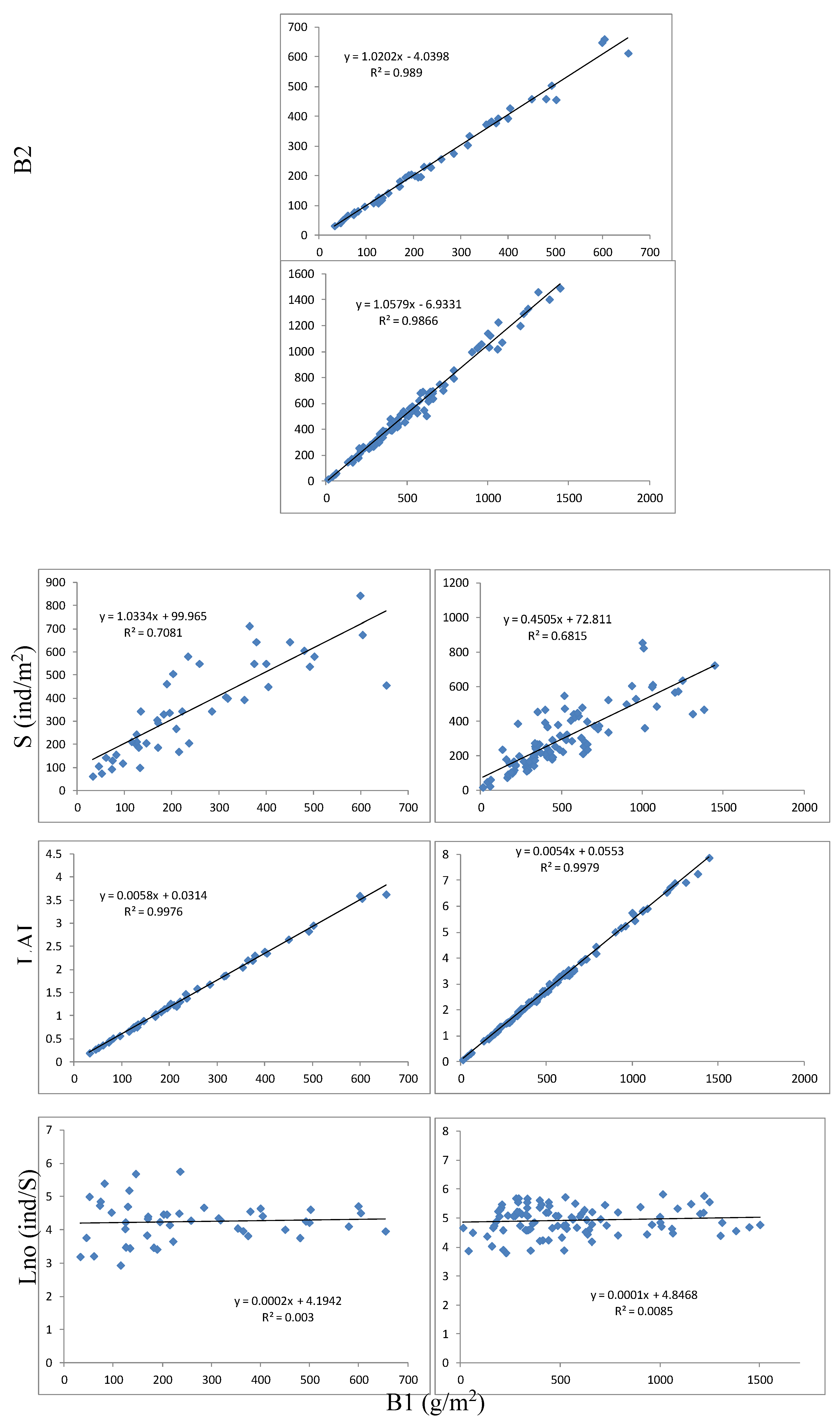
3.1. Acoustic–Biometrics Conversion
3.2. Spatio-Temporal Distribution
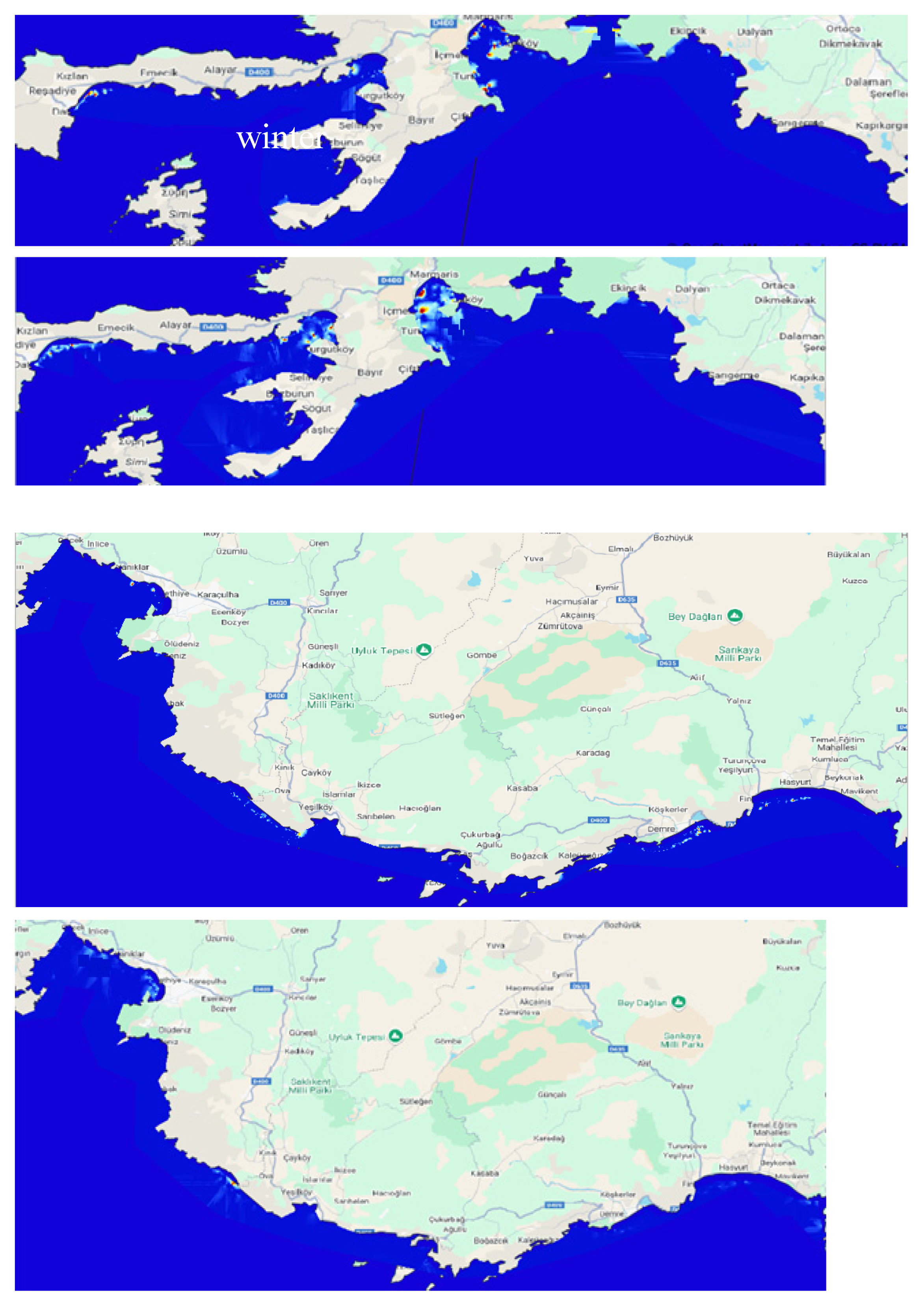
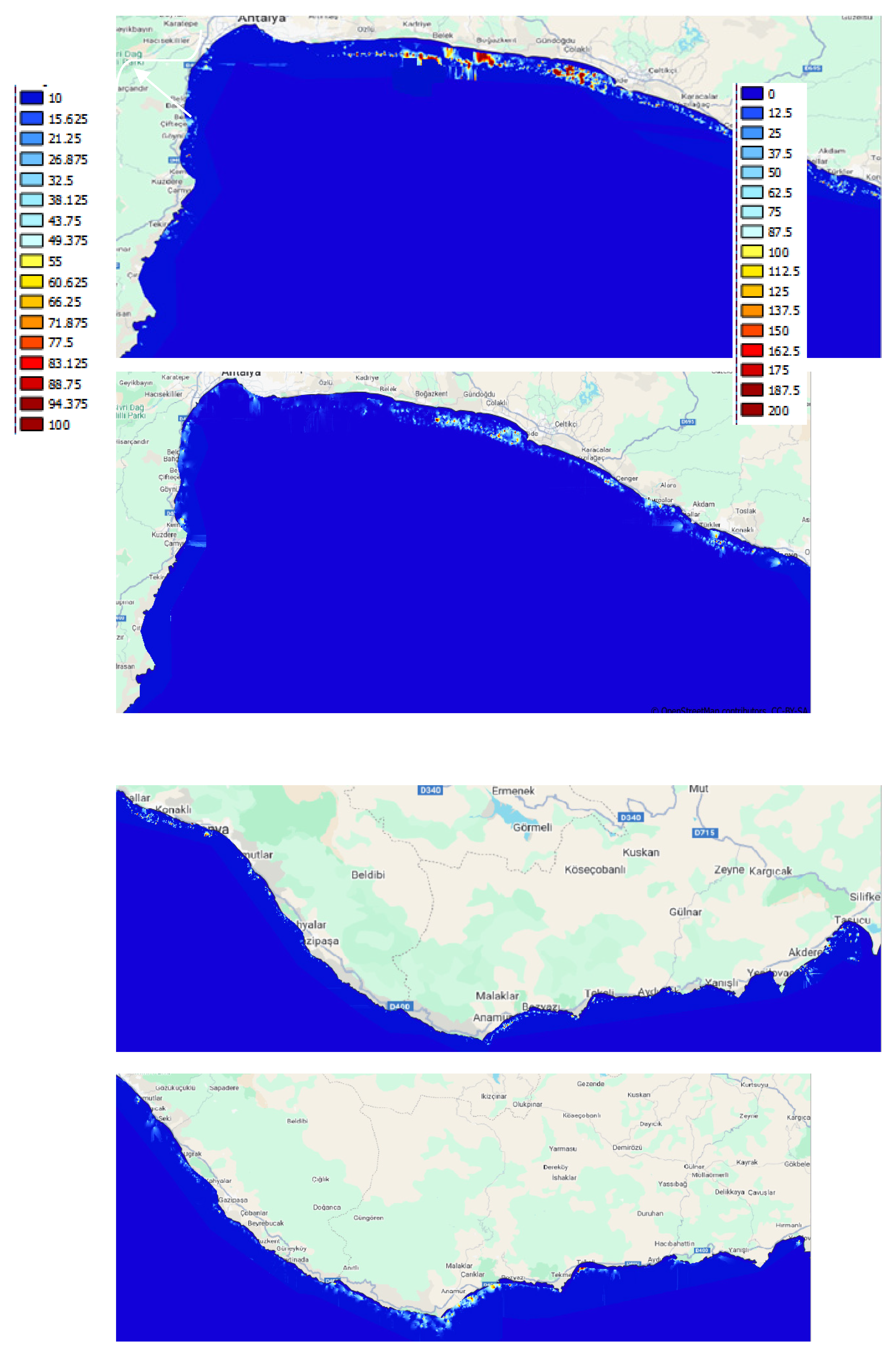
3.2.1. Distribution and Coverage Area

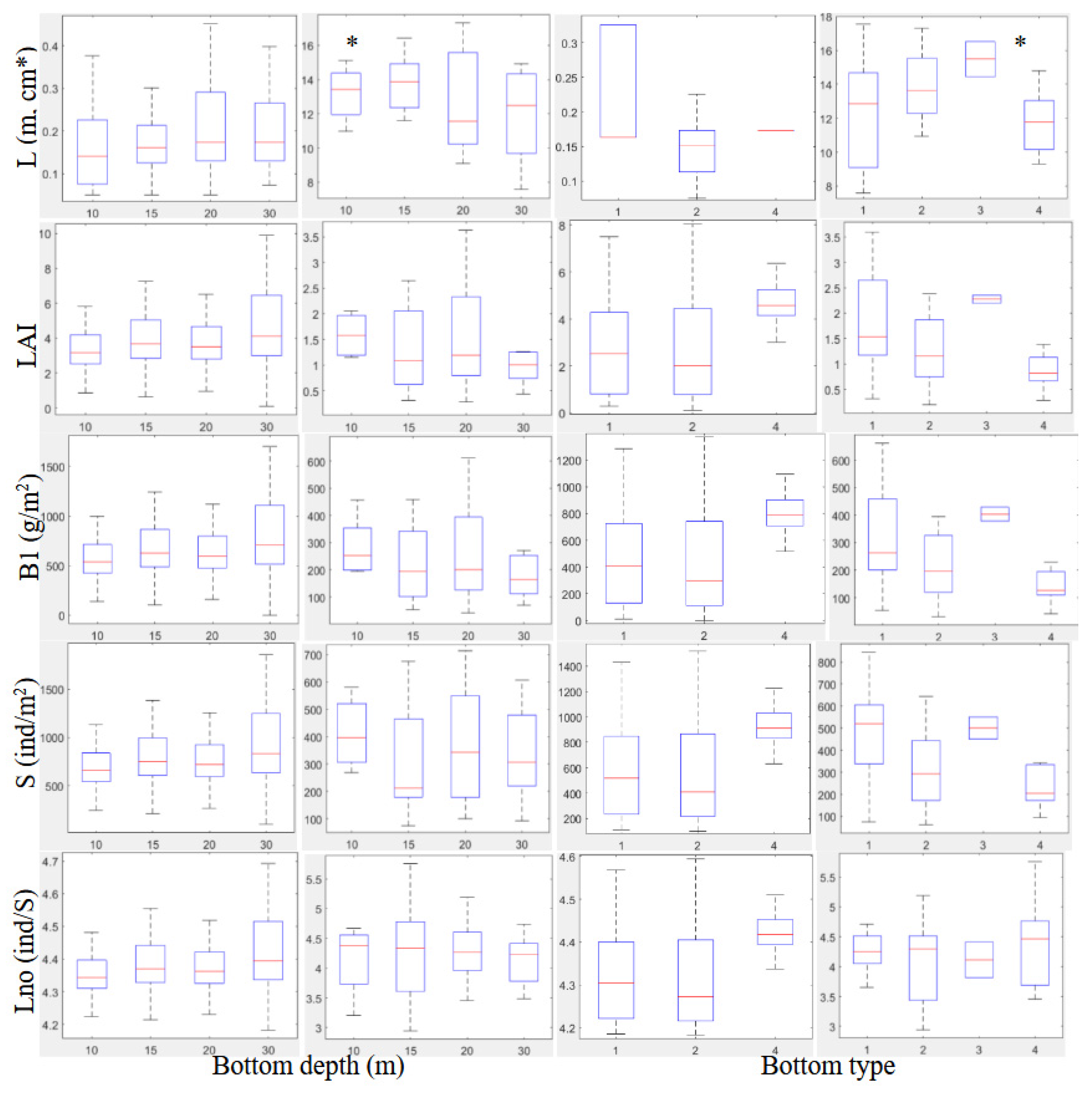
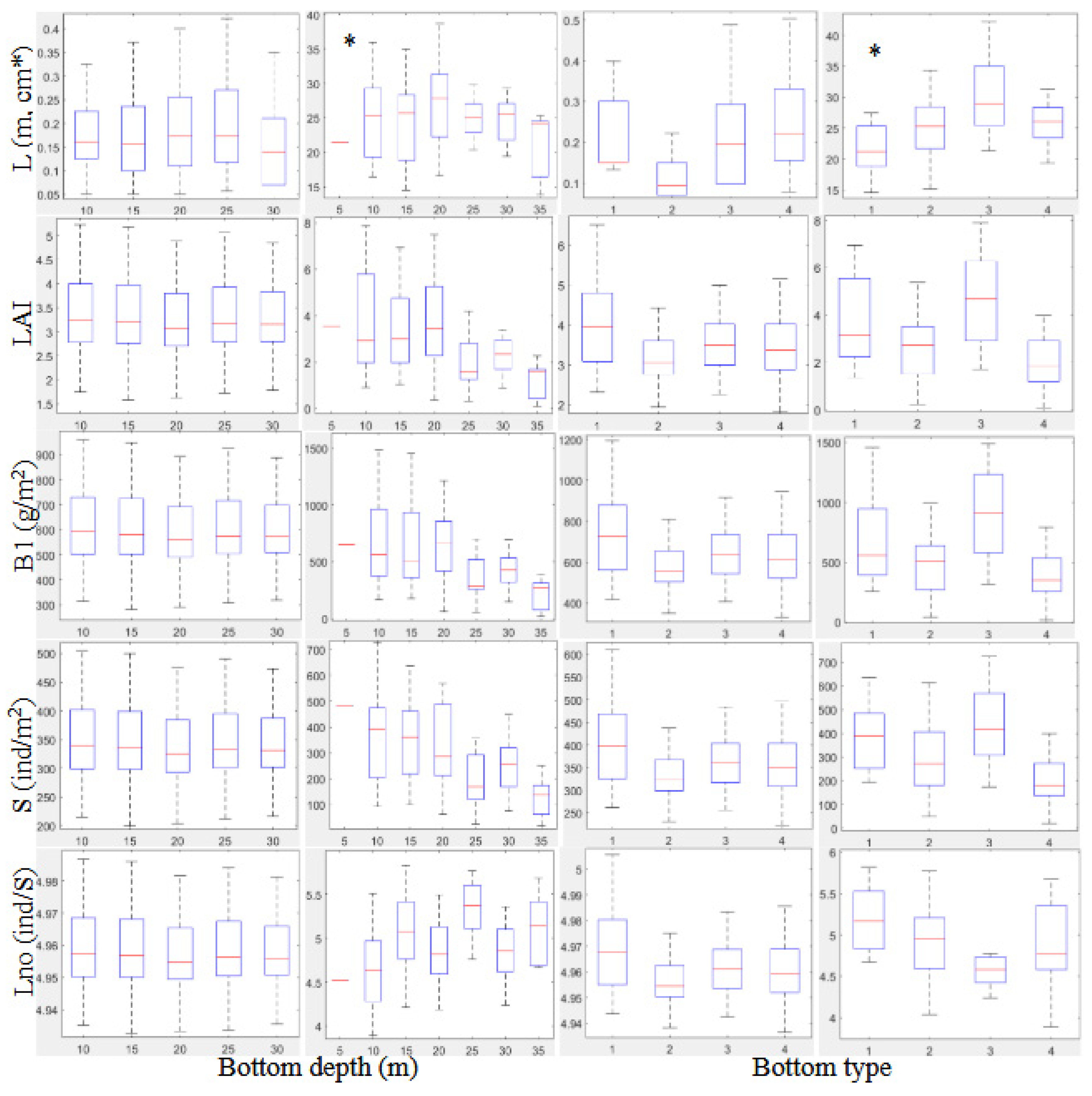
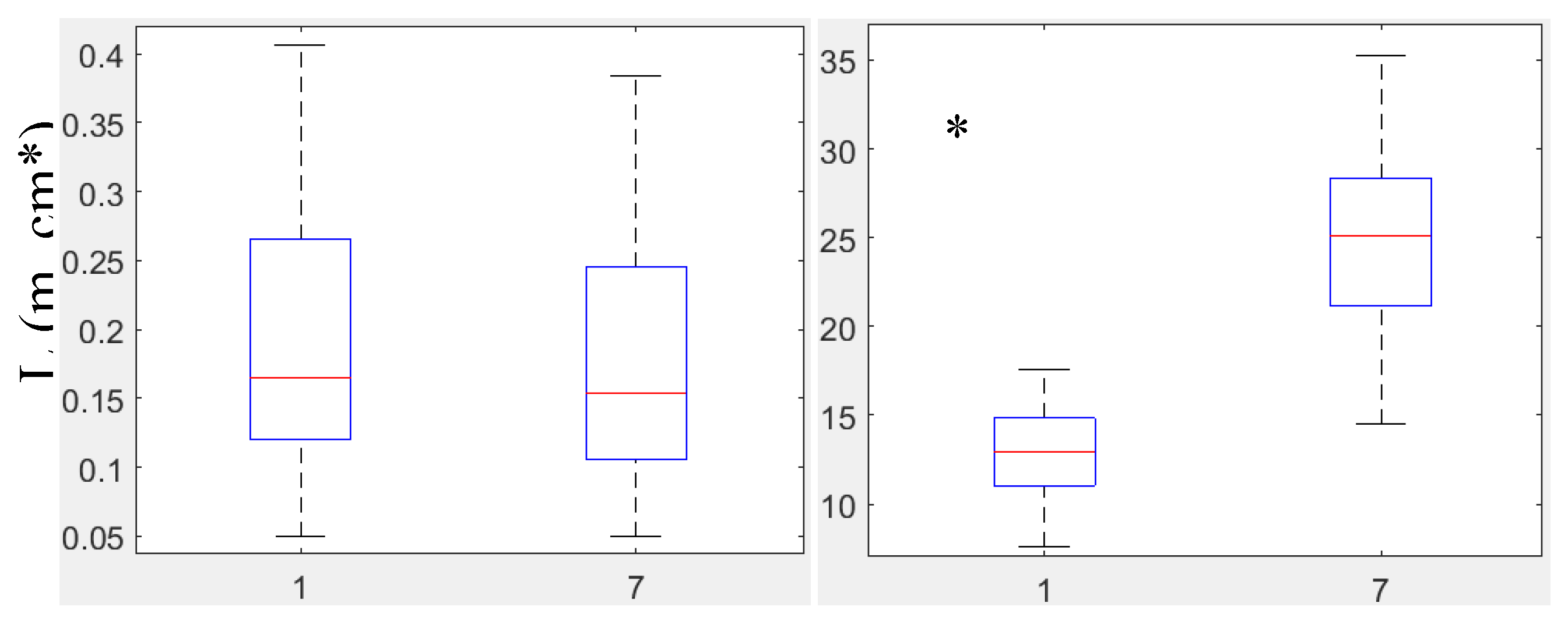
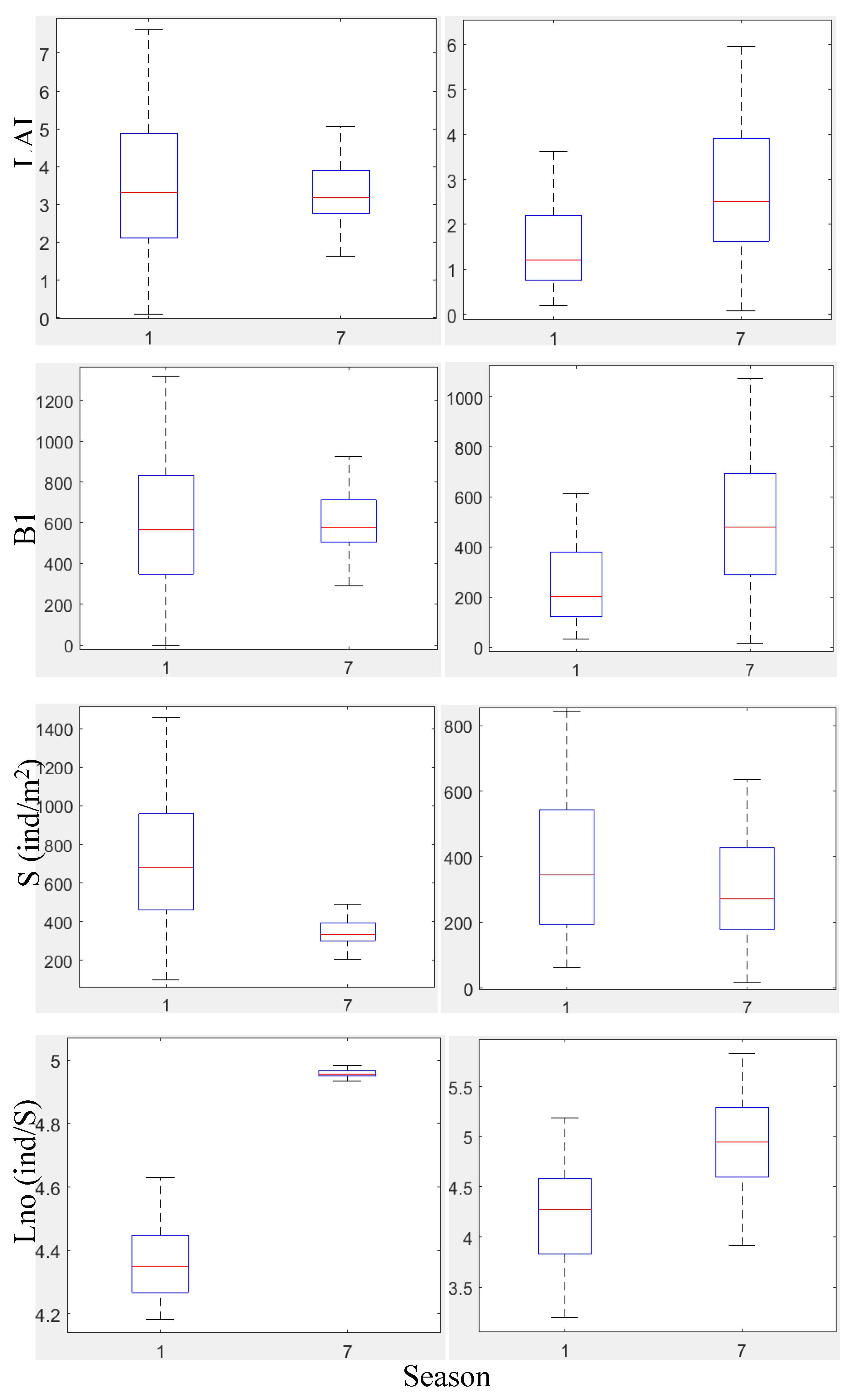
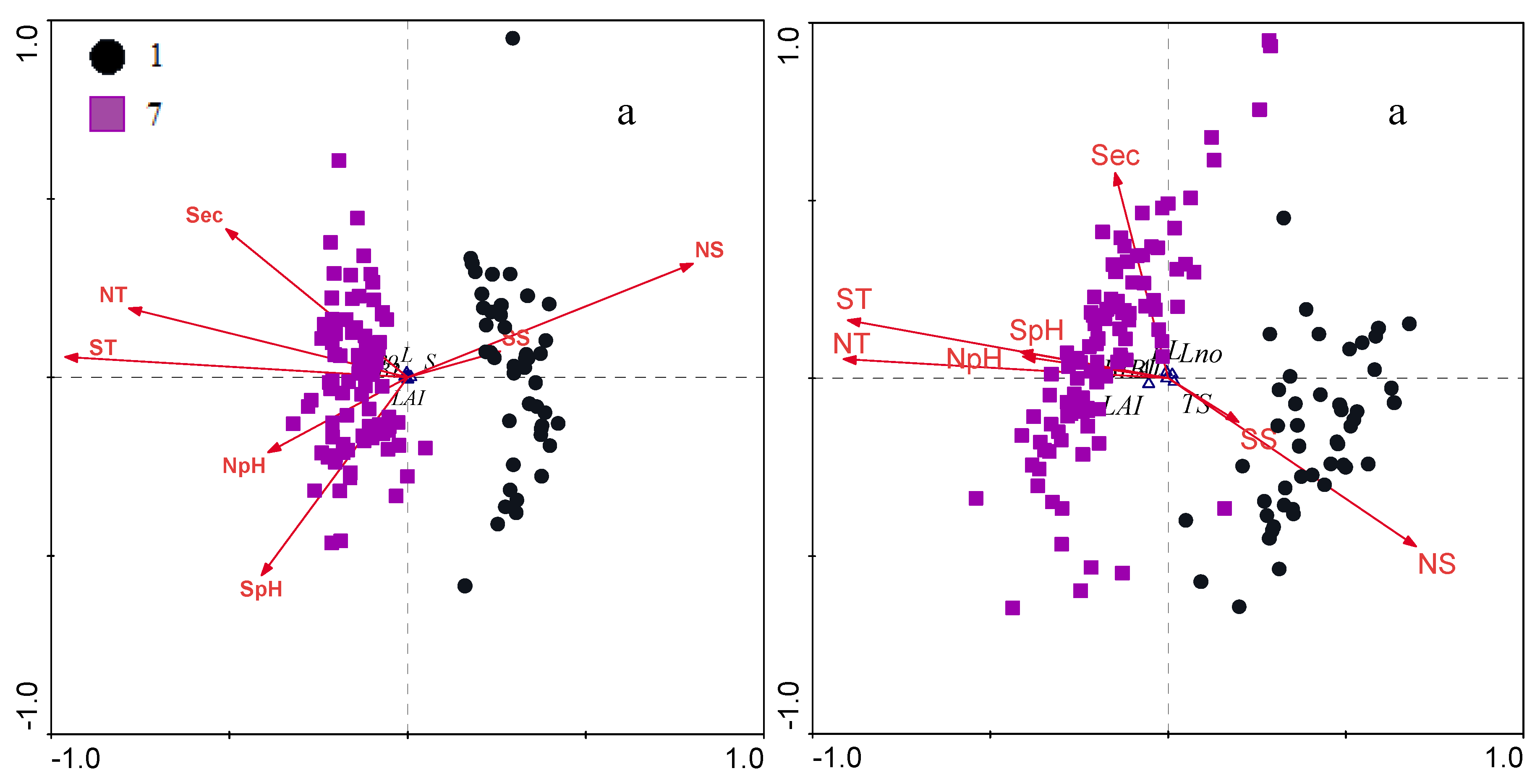
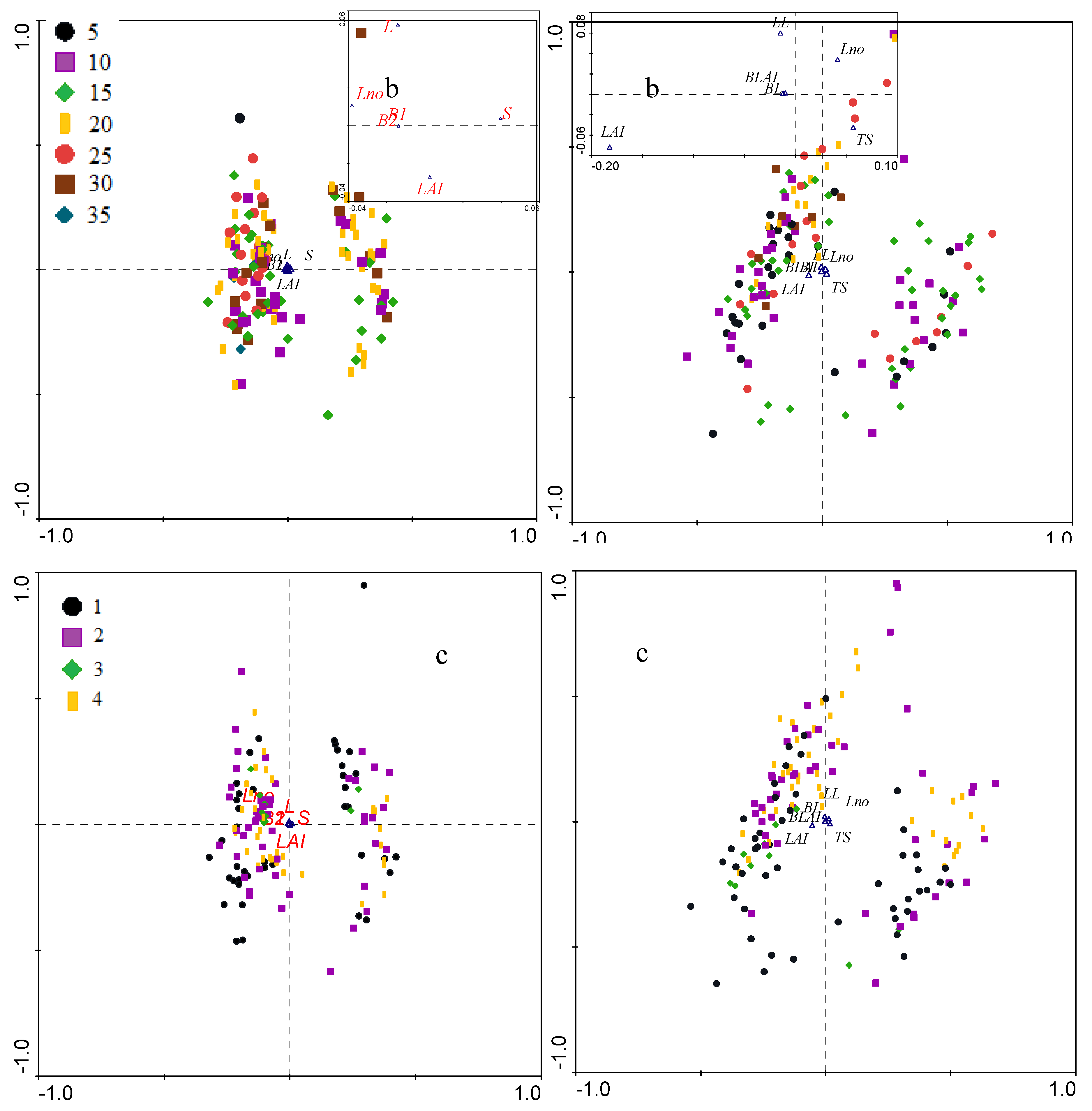
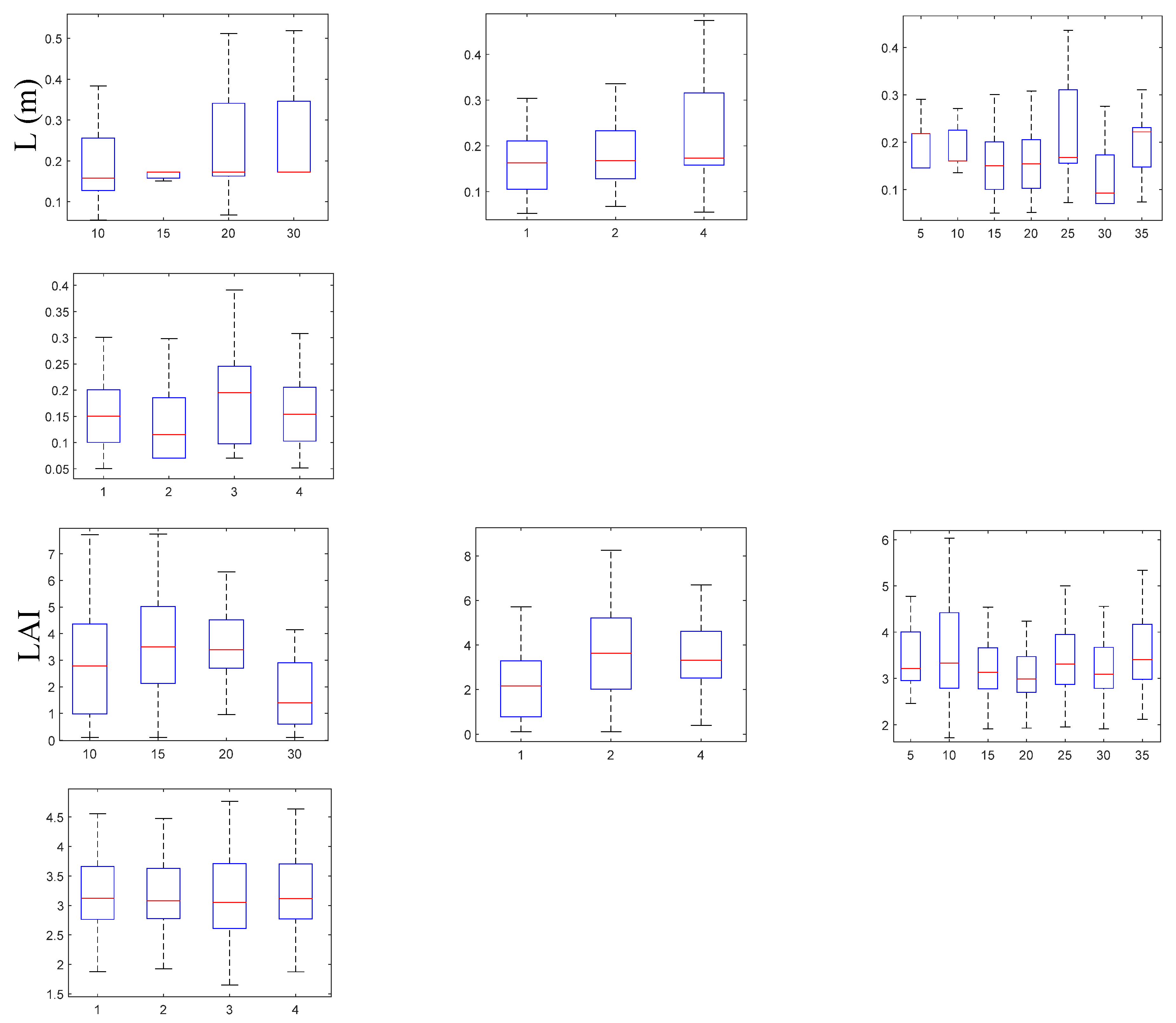
3.2.2. Biometrical Distribution
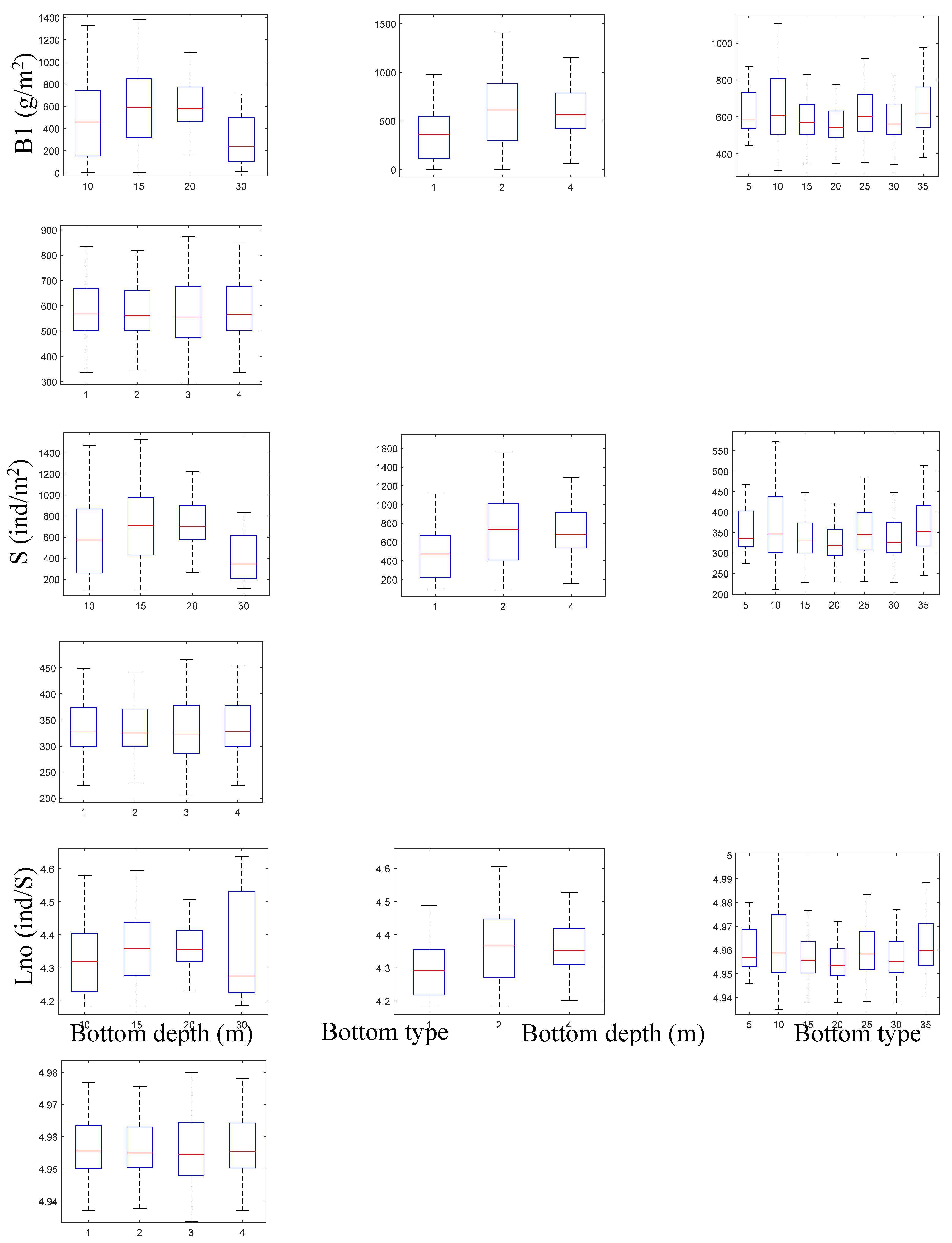
Leaf Biomass, B1 (g m⁻²)
| Acoustics | SCUBA | |||||||||||
| W | L | LAI | B1 | S | Lno | L | LAI | B1 | S | Lno | ||
| D | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.749 | 0.742 | 0.856 | 0.944 | 0.840 | ||
| BT | 0.022 | 0.000 | 0.000 | 0.000 | 0.000 | 0.034 | 0.057 | 0.029 | 0.010 | 0.504 | ||
| S | ||||||||||||
| D | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.494 | 0.011 | 0.034 | 0.001 | 0.012 | ||
| BT | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.087 | 0.006 | 0.002 | 0.000 | 0.038 | ||
| Season | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.198 | 0.000 | ||
Leaf Area Index, LAI (m² Leaf Area m⁻² Bottom Area)
Shoot Density, S (ind. m⁻²)
| Acoustics | SCUBA | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Winter | L* | LAI | B1 | S | Lno | L | LAI | B1 | S | Lno |
| D | ||||||||||
| 10 | 0.17±0.01 | 3.5±0.1 | 594.3±6.5 | 713.2±6.7 | 4.3±0.1 | 13.1±1.1 | 1.5±0.3 | 262.7±60.0 | 386.7±72.0 | 4.1±0.2 |
| 15 | 0.18±0.01 | 3.9±0.1 | 671.6±4.7 | 793.2±4.9 | 4.3±0.1 | 13.5±0.9 | 1.4±0.2 | 255.4±47.0 | 340.3±56.5 | 4.2±0.1 |
| 20 | 0.22±0.01 | 3.7±0.1 | 636.1±4.3 | 757.7±4.4 | 4.3±0.1 | 13.3±0.7 | 1.5±0.2 | 262.3±37.9 | 369.0±45.5 | 4.3±0.1 |
| 30 | 0.20±0.01 | 4.3±0.1 | 750.4±6.8 | 878.8±7.1 | 4.4±0.1 | 11.9±1.2 | 1.1±0.3 | 200.1±64.1 | 338.3±77.0 | 4.1±0.2 |
| BT | ||||||||||
| 1 | 0.23±0.01 | 2.8±0.1 | 485.8±6.9 | 601.5±7.1 | 4.3±0.1 | 12.4±0.7 | 1.8±0.2 | 315.6±36.4 | 462.1±42.5 | 4.3±0.1 |
| 2 | 0.17±0.01 | 2.8±0.1 | 456.4±10.6 | 571.8±10.9 | 4.3±0.1 | 14.8±0.7 | 1.3±0.2 | 234.2±39.9 | 309.5±46.5 | 4.1±0.1 |
| 3 | 15.4±2.1 | 2.2±0.6 | 402.9±109.4 | 500±127.5 | 4.1±0.4 | |||||
| 4 | 0.21±0.01 | 4.8±0.1 | 823.6±9.6 | 950.6±9.9 | 4.4±0.1 | 11.7±0.8 | 0.9±0.2 | 159.2±42.9 | 254.3±50.0 | 4.3±0.1 |
| Summer | ||||||||||
| D | ||||||||||
| 5 | 21.4±6.6 | 3.5±2.0 | 650.9±432.3 | 481.2±174.6 | 4.5±0.5 | |||||
| 10 | 0.17±0.01 | 3.5±0.1 | 642.0±3.6 | 362.0±1.6 | 4.9±2.9e-4 | 26.3±1.5 | 4.1±0.4 | 782.9±99.1 | 417.1±40.1 | 4.6±0.1 |
| 15 | 0.17±0.01 | 3.5±0.1 | 638.2±2.0 | 360.3±0.9 | 4.9±1.6e-4 | 24.7±1.3 | 3.3±0.4 | 637.9±90.1 | 353.5±36.4 | 5.0±0.1 |
| 20 | 0.19±0.01 | 3.3±0.1 | 615.1±2.0 | 349.9±0.9 | 4.9±1.6e-4 | 27.1±1.4 | 3.6±0.4 | 664.2±92.1 | 348.0±37.2 | 4.9±0.1 |
| 25 | 0.19±0.01 | 3.5±0.1 | 639.2±2.4 | 360.7±1.1 | 4.9±1.9e-4 | 25.0±1.7 | 2.2±0.5 | 416.0±111.6 | 217.5±45.1 | 5.3±0.1 |
| 30 | 0.15±0.01 | 3.4±0.1 | 635.4±2.2 | 359.0±1.0 | 4.9±1.8e-4 | 25.0±1.8 | 2.2±0.5 | 421.2±119.9 | 250.0±48.4 | 4.8±0.1 |
| 35 | 21.1±2.5 | 1.2±0.7 | 216.5±163.4 | 129.4±66.0 | 4.9±0.1 | |||||
| BT | ||||||||||
| 1 | 0.21±0.01 | 4.1±0.1 | 742.7±8.9 | 407.3±4.1 | 4.9±7e-4 | 23.6±1.33 | 3.9±0.4 | 775.1±86.7 | 427.8±35.2 | 5.1±0.1 |
| 2 | 0.13±0.01 | 3.3±0.1 | 603.1±2.3 | 344.4±1.1 | 4.9±1e-4 | 24.8±1.0 | 2.8±0.3 | 520.4±68.9 | 293.9±28.0 | 4.8±0.1 |
| 3 | 0.21±0.01 | 3.6±0.1 | 663.3±11.3 | 371.3±5.1 | 4.9±9e-4 | 30.3±2.3 | 4.6±0.7 | 906.2±150.2 | 436.7±61.0 | 4.6±0.1 |
| 4 | 0.23±0.01 | 3.5±0.1 | 652.2±3.8 | 366.6±1.7 | 4.9±3e-4 | 26.0±1.1 | 2.3±0.3 | 413.8±77.5 | 222.7±31.5 | 4.9±0.1 |
| Seaason | ||||||||||
| 1 | 0.20±4.3e-4 | 3.56±0.01 | 603.8±1.1 | 723.3±1.0 | 4.3±3.1e-4 | 13.1±0.8 | 1.4±0.2 | 251.4±55.1 | 359.7±27.7 | 4.2±0.1 |
| 7 | 0.18±3.7e-4 | 3.48±0.01 | 634.3±0.9 | 358.5±0.8 | 4.9±2.9e-4 | 25.3±0.5 | 3.1±0.1 | 580.4±38.2 | 316.1±19.2 | 4.9±0.1 |
Number of Leaves per Shoot, Lno (ind. Shoot⁻¹)
Leaf Length, L (cm)
3.2.3. Habitats
Bottom Types
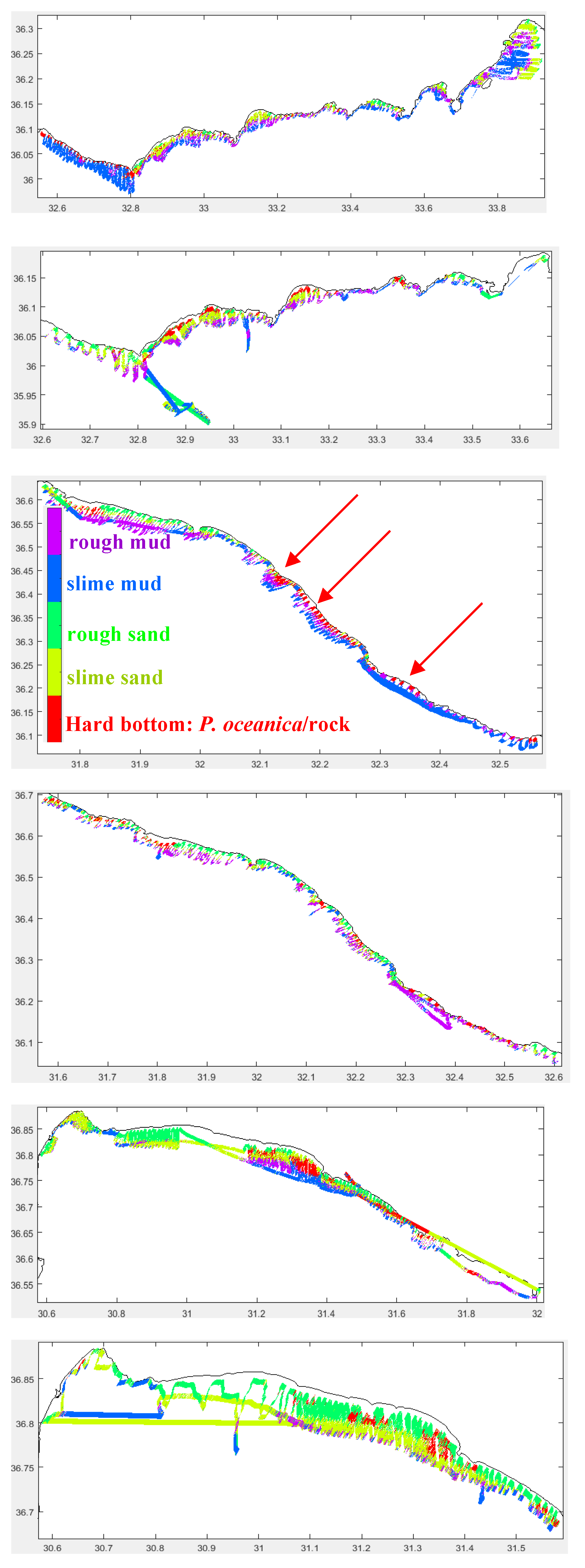
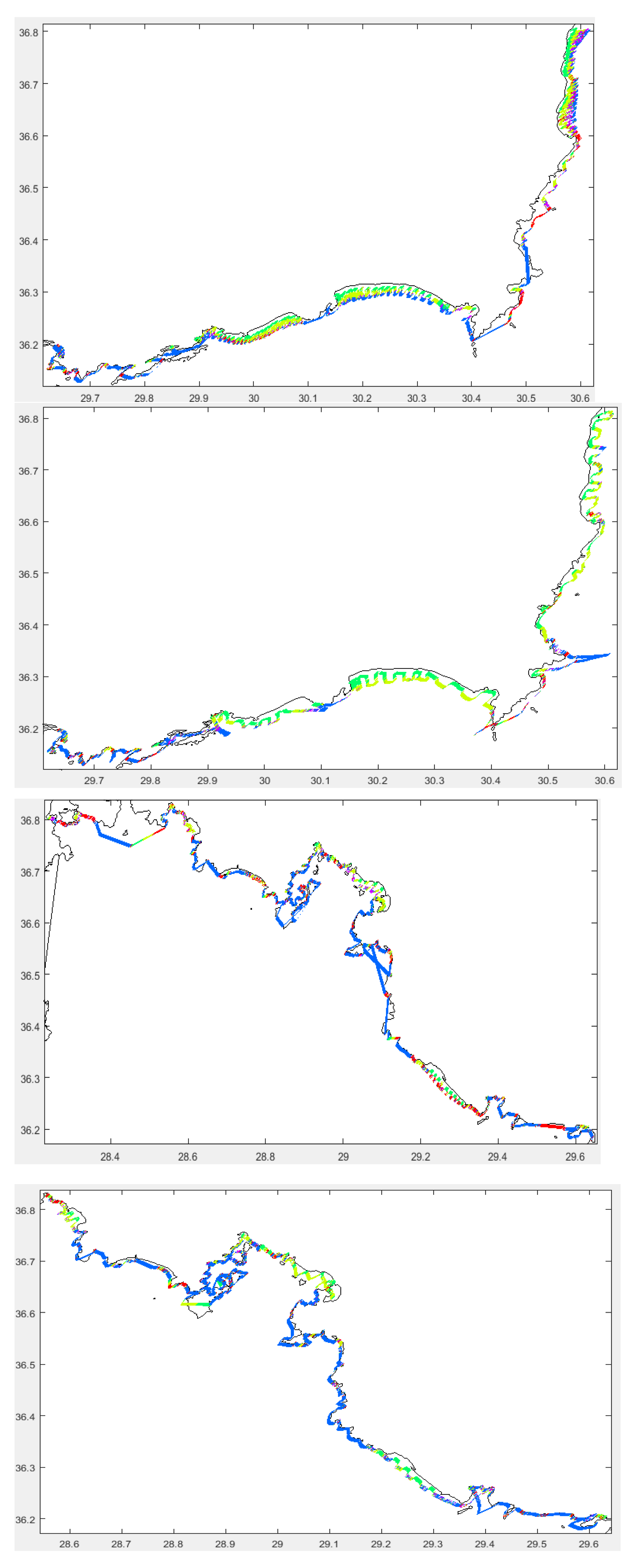
Sediment Thickness
3.2.4. Ecological Evaluation
Ecology
| Acoustic | SCUBA | Ac & Sc | |||||||
| Source | df | p | p(MC) | df | p | p(MC) | df | p | p(MC) |
| Method | 1 | 0.333 | 0.309 | ||||||
| Season | 1 | 0.001 | 0.001 | 1 | 0.001 | 0.001 | 1 | 0.001 | 0.001 |
| Type | 3 | 0.006 | 0.005 | 3 | 0.022 | 0.004 | 3 | 0.184 | 0.182 |
| Depth | 6 | 0.738 | 0.785 | 5 | 0.053 | 0.017 | 6 | 0.368 | 0.383 |
| Method x Season | 1 | 0.003 | 0.002 | ||||||
| Method x Type | 3 | 0.002 | 0.001 | ||||||
| Method x Depth | 5 | 0.079 | 0.055 | ||||||
| Season x Type | 3 | 0.017 | 0.01 | 3 | 0.735 | 0.782 | 3 | 0.036 | 0.026 |
| Season x Depth | 3 | 0.248 | 0.233 | 3 | 0.767 | 0.788 | 3 | 0.304 | 0.34 |
| Type x Depth | 14 | 0.976 | 0.972 | 15 | 0.99 | 0.996 | 15 | 0.989 | 0.994 |
| Method x Season x Type | 3 | 0.16 | 0.148 | ||||||
| Method x Season x Depth | 3 | 0.556 | 0.521 | ||||||
| Method x Type x Depth | 14 | 0.982 | 0.993 | ||||||
| Season x Type x Depth | 6 | 0.887 | 0.911 | 6 | 0.766 | 0.794 | 6 | 0.898 | 0.926 |
| Method x Season x Type x Depth | 6 | 0.816 | 0.819 | ||||||
| Residuals | 104 | 110 | 214 | ||||||
| Total | 140 | 146 | 287 | ||||||
Ecological Status
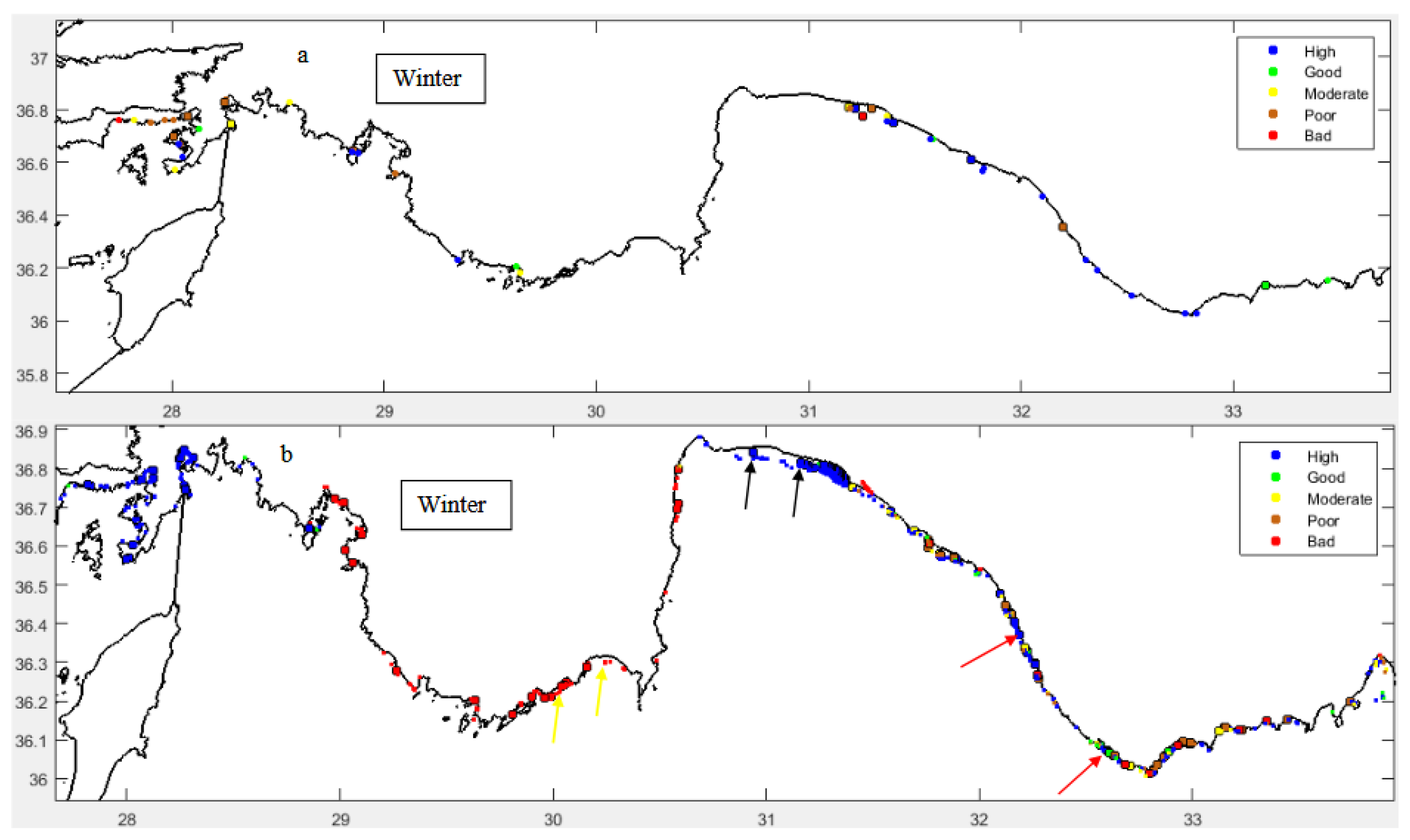
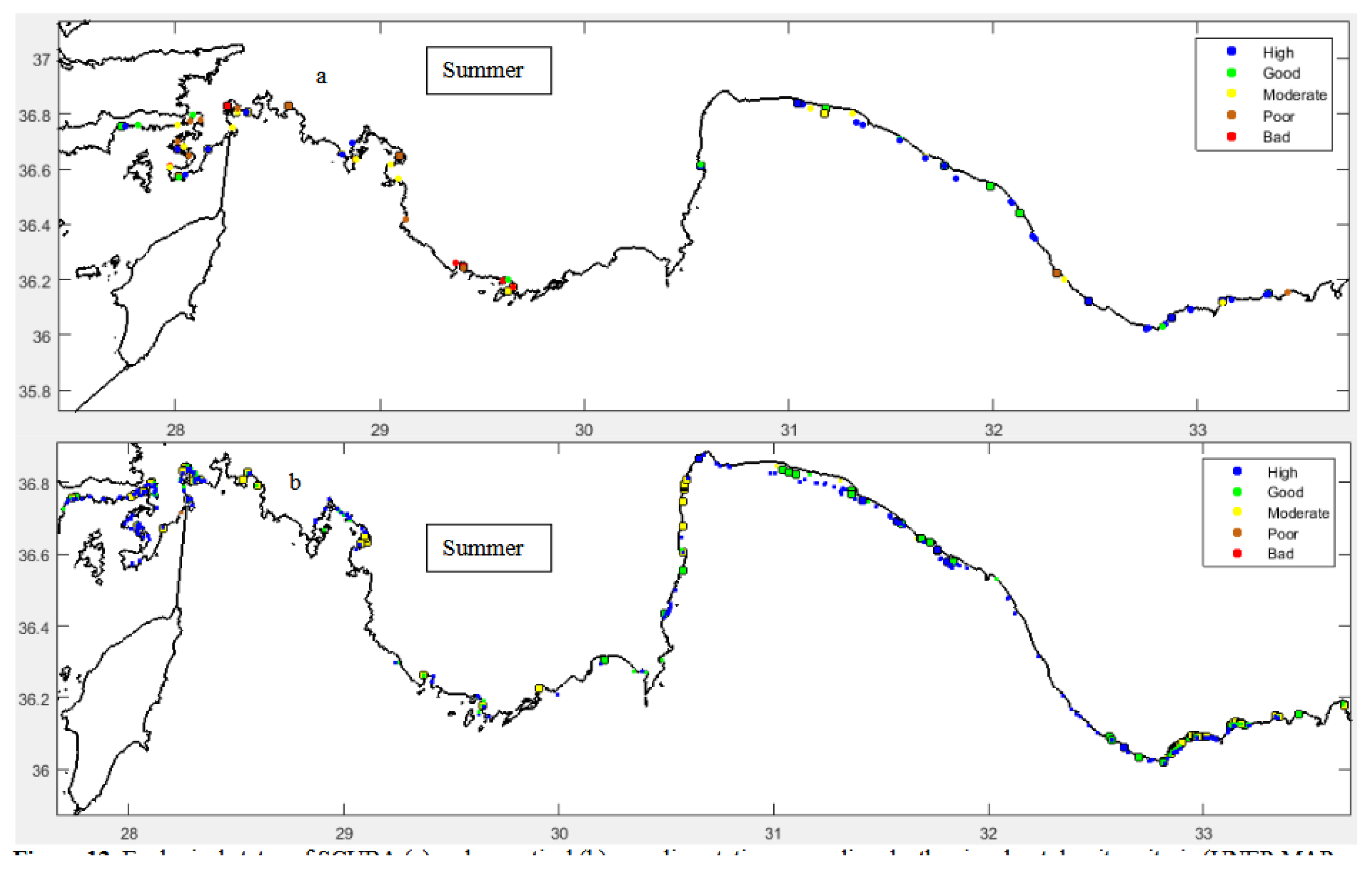
4. Discussion
4.1. Agreement and Bias Between Acoustic and SCUBA-Based Biometrics
4.2. Spatiotemporal Distribution
4.2.1. Biometrics
4.2.2. Habitats
Bottom Types
Sediment Thickness
4.2.3. Ecological Evaluation
Ecology
Ecological Status
5. Conclusions
Supplementary Materials
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix
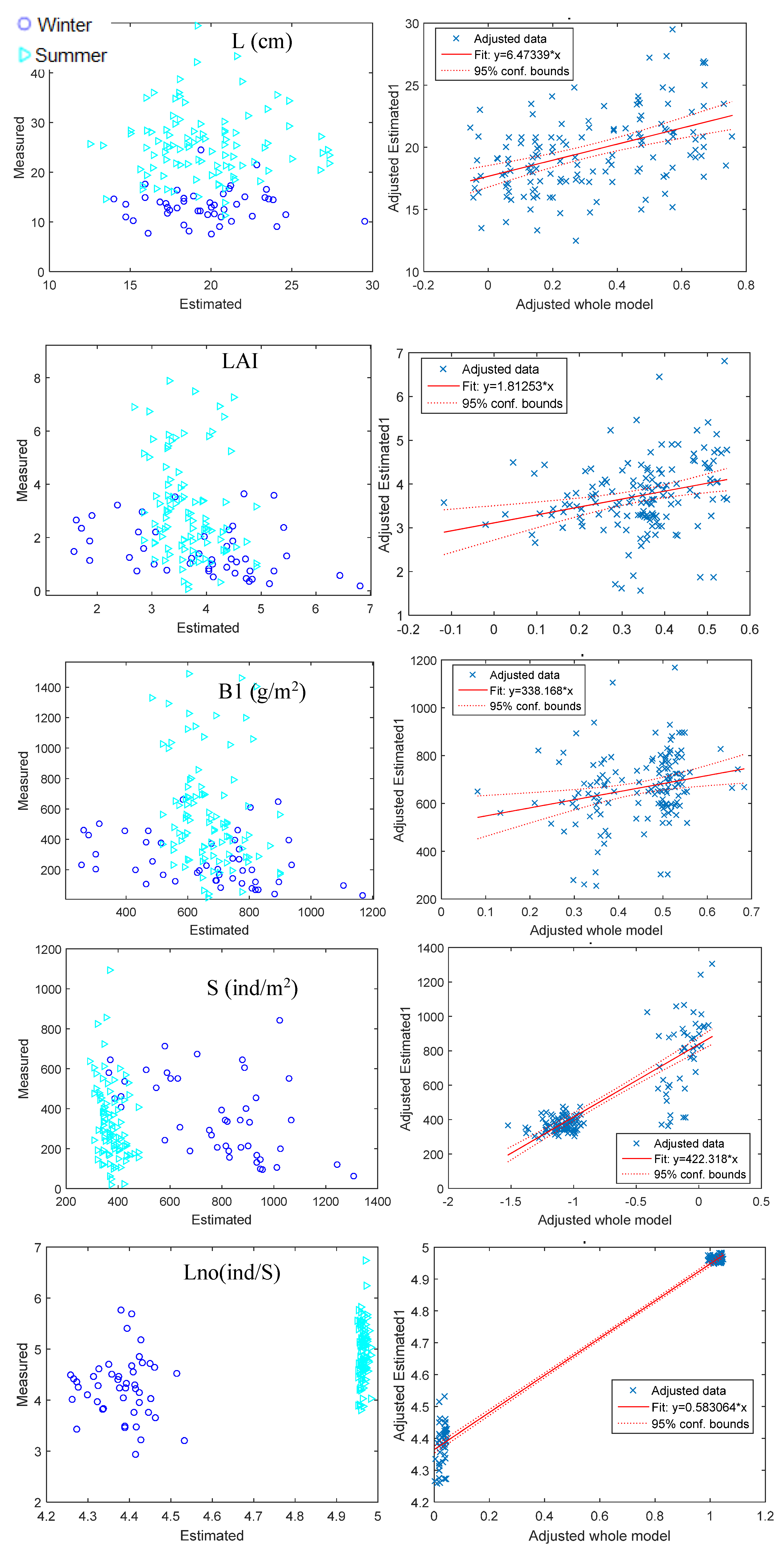
| L | LAI | B1 | S | Lno | ||||||
| Estimate | pValue | Estimate | pValue | Estimate | pValue | Estimate | pValue | Estimate | pValue | |
| (Intercept) | 17.7 | 0.000 | 3.1 | 0.000 | 514.3 | 0.001 | 838.1 | 0.000 | 4.36 | 0.000 |
| Measured | 0.0 | 0.870 | 0.0 | 0.246 | 0.0 | 0.387 | -0.2 | 0.001 | 0.00 | 0.848 |
| Season_S | -0.5 | 0.547 | -0.2 | 0.364 | 12.4 | 0.692 | -416.3 | 0.000 | 0.58 | 0.000 |
| Depth_10 | 1.0 | 0.742 | 0.7 | 0.413 | 125.7 | 0.403 | 39.3 | 0.766 | 0.01 | 0.767 |
| Depth_15 | 0.5 | 0.883 | 0.7 | 0.425 | 123.6 | 0.409 | 32.5 | 0.804 | 0.02 | 0.665 |
| Depth_20 | 3.1 | 0.314 | 0.7 | 0.385 | 133.5 | 0.372 | 35.7 | 0.786 | 0.02 | 0.691 |
| Depth_25 | 4.4 | 0.171 | 0.7 | 0.445 | 124.3 | 0.417 | 6.2 | 0.963 | 0.01 | 0.715 |
| Depth_30 | 1.3 | 0.670 | 0.7 | 0.421 | 130.6 | 0.389 | 12.7 | 0.924 | 0.02 | 0.684 |
| Depth_35 | 2.8 | 0.395 | 0.9 | 0.337 | 166.4 | 0.299 | 2.3 | 0.987 | 0.02 | 0.634 |
| Type_2 | 1.1 | 0.095 | 0.3 | 0.098 | 53.3 | 0.096 | 20.6 | 0.477 | 0.01 | 0.092 |
| Type_3 | 0.0 | 0.973 | -0.1 | 0.740 | -19.5 | 0.712 | -22.6 | 0.624 | -0.01 | 0.523 |
| Type_4 | 0.6 | 0.368 | 0.2 | 0.237 | 43.5 | 0.209 | -2.3 | 0.943 | 0.01 | 0.162 |
| R2 | 0.197 | 0.078 | 0.057 | 0.726 | 0.983 | |||||
| Adjusted R2 | 0.13 | 0.001 | 0.020 | 0.703 | 0.981 | |||||
| pM | 0.001 | 0.433 | 0.701 | 7.1e-32 | 1.8e-110 |
| L | LAI | B1 | S | Lno | ||||||
| Estimate | pValue | Estimate | pValue | Estimate | pValue | Estimate | pValue | Estimate | pValue | |
| (Intercept) | 17.5 | 6.2E-32 | 4.01 | 1.5E-33 | 672.9 | 1.9E-31 | 966.6 | 3.1E-63 | 4.305 | 7.0E-207 |
| Measured | 0.0 | 0.663 | -0.04 | 0.278 | 0.0 | 0.341 | -0.1 | 0.110 | 0.001 | 0.791 |
| Season | -0.1 | 0.439 | -0.06 | 0.010 | -3.9 | 0.3651 | -83.8 | 4.3E-77 | 0.093 | 1.8E-155 |
| Type | 0.1 | 0.516 | 0.03 | 0.527 | 6.0 | 0.523 | -0.6 | 0.916 | 0.001 | 0.622 |
| Depth | 0.1 | 0.014 | 0.00 | 0.585 | 1.1 | 0.505 | 0.6 | 0.477 | 0.000 | 0.363 |
| R2 | 0.051 | 0.093 | 0.035 | 0.919 | 0.996 | |||||
| Adjusted R2 | 0.023 | 0.067 | 0.007 | 0.916 | 0.995 | |||||
| pM | 0.119 | 0.007 | 0.278 | 1.0e-74 | 3.6e-162 |
References
- Strydom, S.; Murray, K.; Moustaka, M.; Hyndes, G.; Wilson, S. Getting edgy: implications of fragmented seagrass meadows for fish assemblages. Landsc. Ecol. 2026, 41, 31. [CrossRef]
- Den Hartog, C. Structure, function, and classification in seagrass communities. In A Scientific Perspective; McRoy, C.P., Helfferich, C., Eds.; Marcel Dekker: New York, 1977; pp. 89–121.
- Boudouresque, C.F.; Meinesz, A. Découverte de l'herbier de Posidonies. Cahier Parc Nation Port-Cros 1982, 4, 79 (in French).
- Edgar, G.J.; Shaw, C. The production and trophic ecology of shallow-water fish assemblages in southern Australia. 3. General relationships between sediments, seagrasses, invertebrates, and fishes. J. Exp. Mar. Biol. Ecol. 1995, 194, 107–131. [CrossRef]
- Peirano, A.; Damasso, V.; Montefalcone, M.; Morri, C.; Bianchi, C.N. Effects of climate, invasive species, and anthropogenic impacts on the growth of the seagrass Posidonia oceanica (L.) Delile in Liguria (NW Mediterranean Sea). Mar. Pollut. Bull. 2005, 50, 817–822. [CrossRef]
- Pergent-Martini, C.; Leoni, V.; Pasqualini, V.; Ardizzone, G.D.; Balestri, E.; Bedini, R.; Belluscio, A.; Belsher, T.; Borg, J.; Boudouresque, C.F.; Boumaza, S.; Bouquegneau, J.M.; Buia, M.C.; Calvo, S.; Cebrian, J.; Charbonnel, E.; Cinelli, F.; Cossu, A.; Maida, G.D.; Dural, B.; Francour, P.; Gobert, S.; Lepoint, G.; Meinesz, A.; Molenaar, H.; Mansour, H.M.; Panayotidis, P.; Peirano, A.; Pergent, G.; Piazzi, L.; Pirrotta, M.; Relini, G.; Romero, J.; Sanchez-Lizaso, J.L.; Semroud, R.; Shembri, P.; Shili, A.; Tomasello, A.; Velimirov, B. Descriptors of Posidonia oceanica meadows: Use and application. Ecol. Indic. 2005, 5, 213–230. [CrossRef]
- UNEP-MAP-RAC/SPA. Rapport sur l’état de mise en œuvre du Protocole ASP/DB. Document de travail pour la neuvièmeréunion des Points focaux pour les ASP, Floriana, Malte, 3–6 juin 2009, UNEP(DEPI)/MED WG.331/03, CAR/ASP édit., Tunis, 2009; 19p.
- Boudouresque, C.F.; Bianchi, C.N. Une idée neuve: la protection des espèces marines. In GIS Posidonie: plus de 30 ans au service de la protection et de la gestion du milieu marin; Le Direach, L.; Boudouresque, C.F., Eds.; GIS Posidonie: Marseille, 2013; pp. 85–91.
- Comte, A.; Barreyre, J.; Monnier, B.; de Rafael, R.; Boudouresque, C.-F.; Pergent, G.; Ruitton, S. Operationalizing blue carbon principles in France: Methodological developments for Posidonia oceanica seagrass meadows and institutionalization. Mar. Pollut. Bull. 2024, 198, 115822. [CrossRef]
- Gobert, S.; Velimirov, B.; Pergent, G.; Pergent-Martini, C.; Walker, D.I. Biology of Posidonia. In Seagrasses: Biology, Ecology and Conservation; Larkum, A.W.D.; Orth, R.J.; Duarte, C.M., Eds.; Springer: The Netherlands, 2006; pp. 387–408.
- UNEP/MAP-RAC/SPA. Guidelines for Standardization of Mapping and Monitoring Methods of Marine Magnoliophyta in the Mediterranean; Pergent-Martini, C., Ed.; RAC/SPA: Tunis, 2015; 48p. + Annexes.
- Mutlu, E.; Olguner, C.; Gökoğlu, M.; Özvarol, Y. Seasonal growth dynamics of Posidonia oceanica in a pristine Mediterranean gulf. Ocean Sci. J. 2022, 57, 381–397. [CrossRef]
- Mutlu, E.; Karaca, D.; Duman, G.S.; Şahin, A.; Özvarol, Y.; Olguner, C. Seasonality and phenology of an epiphytic calcareous red alga, Hydrolithon boreale, on the leaves of Posidonia oceanica (L) Delile in the Turkish waters. Environ. Sci. Pollut. Res. 2023, 30, 17193–17213. [CrossRef]
- Mutlu, E.; Duman, G.S.; Karaca, D.; Özvarol, Y.; Şahin, A. Biometrical variation of Posidonia oceanica with different bottom types along the entire Turkish Mediterranean coast. Ocean Sci. J. 2023, 58, 9. [CrossRef]
- Colantoni, P.; Gallignani, P.; Fresi, E.; Cinelli, F. Patterns of Posidonia oceanica (L.) Delile beds around the Island of Ischia (Gulf of Naples) and in adjacent waters. PSZNI: Mar. Ecol. 1982, 3, 53–74. [CrossRef]
- Vacchi, M.; Montefalcone, M.; Bianchi, C.N.; Morri, C.; Ferrari, M. Hydrodynamic constraints to the seaward development of Posidonia oceanica meadows. Estuar. Coast. Shelf Sci. 2012, 97, 58–65. [CrossRef]
- Tomasello, A.; Perrone, R.; Colombo, P.; Pirrotta, M.; Calvo, S. Root hair anatomy and morphology in Posidonia oceanica (L.) Delile and substratum typology: First observations of a spiral form. Aquat. Bot. 2018, 145, 45–48. [CrossRef]
- Catucci, E.; Scardi, M. Modeling Posidonia oceanica shoot density and rhizome primary production. Sci. Rep. 2020, 10, 16978. [CrossRef]
- Sghaier, Y.R.; Zakhama-Sraieb, R.Y.M.; Charfi-Cheikhrouha, F. Patterns of shallow seagrass (Posidonia oceanica) growth and flowering along the Tunisian coast. Aquat. Bot. 2013, 104, 185–192. [CrossRef]
- Bonacorsi, M.; Pergent-Martini, C.; Breand, N.; Pergent, G. Is Posidonia oceanica regression a general feature in the Mediterranean Sea? Medit. Mar. Sci. 2013, 14, 193–203. [CrossRef]
- Alagna, A.; D'Anna, G.; Musco, L.; Fernandez, T.V.; Gresta, M.; Pierozzi, N.; Badalamenti, F. Taking advantage of seagrass recovery potential to develop novel and effective meadow rehabilitation methods. Mar. Pollut. Bull. 2019, 149, 110578. [CrossRef]
- Pergent-Martini, C.; Acunto, S.; Andre, S.; Barralon, E.; Calvo, S.; Castejon-Silvo, I.; Culioli, J.M.; Lehman, L.; Molenaar, H.; Monnier, B.; Oberti, P.; Pey, A.; Piazzi, L.; Santoni, M.C.; Terrados, J.; Tomasello, A.; Pergent, G. Posidonia oceanica Restoration, a Relevant Strategy after Boat Anchoring Degradation? In Proceedings of the 7th Mediterranean Symposium on Marine Vegetation, Genoa, Italy, 19–20 September 2022; RAC/SPA: Tunis, 2022; pp. 78–83.
- Vacchi, M.; De Falco, G.; Simeone, S.; Montefalcone, M.; Morri, C.; Ferrari, M.; Bianchi, C.N. Biogeomorphology of the Mediterranean Posidonia oceanica seagrass meadows. Earth Surf. Proc. Land. 2017, 42, 42–54. [CrossRef]
- Topouzelis, K.; Makri, D.; Stoupas, N.; Papakonstantinou, A.; Katsanevakis, S. Seagrass mapping in Greek territorial waters using Landsat-8 satellite images. Int. J. Appl. Earth Obs. Geoinf. 2018, 67, 98–111.
- Gerakaris, V.; Papathanasiou, V.; Salomidi, M.; Issaris, Y.; Panayotidis, P. Spatial patterns of Posidonia oceanica structural and functional features in the Eastern Mediterranean (Aegean and E. Ionian Seas) in relation to large-scale environmental factors. Mar. Environ. Res. 2021, 165, 105222.
- Panayotidis, P.; Papathanasiou, V.; Gerakaris, V.; Fakiris, E.; Orfanidis, S.; Papatheodorou, G.; Kosmidou, M.; Georgiou, N.; Drakopoulou, V.; Loukaidi, V. Seagrass meadows in the Greek Seas: presence, abundance and spatial distribution. Botanica Marina 2022, 65, 289–299. [CrossRef]
- Litsi-Mizan, V.; Efthymiadis, P.T.; Gerakaris, V.; Serrano, O.; Tsapakis, M.; Apostolaki, E.T. Decline of seagrass (Posidonia oceanica) production over two decades in the face of warming of the Eastern Mediterranean Sea. New Phytol. 2023, 239, 2126–2137. [CrossRef]
- Litsi-Mizan, V.; García-Escudero, C.A.; Tsigenopoulos, C.S.; Tsiaras, K.; Gerakaris, V.; Apostolaki, E.T. Unravelling the genetic pattern of seagrass (Posidonia oceanica) meadows in the Eastern Mediterranean Sea. Biodivers. Conserv. 2024, 33, 257–280. [CrossRef]
- Pergent, G.; Pergent-Martini, C.; Boudouresque, C.F. Utilisation de l’herbier à Posidonia oceanica comme indicateur biologique de la qualité du milieu littoral en Méditerranée: État des connaissances. Mesogee 1995, 54, 3–27.
- Gobert, S.; Lefebvre, L.; Boissery, P.; Richir, J. A non-destructive method to assess the status of Posidonia oceanica meadows. Ecol. Indic. 2020, 119, 106838.
- Prado, P.; Alcoverro, T.; Romero, J. Influence of nutrients in the feeding ecology of seagrass (Posidonia oceanica L.) consumers: a stable isotopes approach. Mar. Biol. 2010, 157, 715–724. [CrossRef]
- van Rein, H.; Brown, C.J.; Quinn, R.; Breen, J.; Schoeman, D. An evaluation of acoustic seabed classification techniques for marine biotope monitoring over broad-scales (>1 km²) and meso-scales (10 m²–1 km²). Estuar. Coast. Shelf Sci. 2011, 93, 336–349. [CrossRef]
- Fakiris, E.; Zoura, D.; Ramfos, A.; Spinos, E.; Georgiou, N.; Ferentinos, G.; Papatheodorou, G. Object-based classification of sub-bottom profiling data for benthic habitat mapping. Comparison with sidescan and RoxAnn in a Greek shallow-water habitat. Estuar. Coast. Shelf Sci. 2018, 208, 219–234. [CrossRef]
- Lee, W.S.; Lin, C.Y. Mapping of tropical marine benthic habitat: Hydroacoustic classification of coral reefs environment using single-beam (RoxAnn™) system. Cont. Shelf Res. 2018, 170, 1–10.
- Dimas, X.; Fakiris, E.; Christodoulou, D.; Georgiou, N.; Geraga, M.; Papathanasiou, V.; Orfanidis, S.; Kotomatas, S.; Papatheodorou, G. Marine priority habitat mapping in a Mediterranean conservation area (Gyaros, South Aegean) through multi-platform marine remote sensing techniques. Front. Mar. Sci. 2022, 9, 953462. [CrossRef]
- Dattola, L.; Rende, S.F.; Dominici, R.; Lanera, P.; Di Men, R.; Scalise, S.; Cappa, P.; Oranges, T.; Aramini, G. Comparison of Sentinel-2 and Landsat-8 OLI satellite images vs. high spatial resolution images (MIVIS and WorldView-2) for mapping Posidonia oceanica meadows. In Proceedings of SPIE 10784, Remote Sensing of the Ocean, Sea Ice, Coastal Waters, and Large Water Regions, 1078419, 2018. [CrossRef]
- Yücel-Gier, G.; Koçak, G.; Akçalı, B.; İlhan, T.; Duman, M. Evaluation of Posidonia oceanica Map Generated by Sentinel-2 Image: Gülbahçe Bay Test Site. TrJFAS 2020, 20, 571–581. [CrossRef]
- Traganos, D.; Aggarwal, B.; Poursanidis, D.; Topouzelis, K.; Chrysoulakis, N.; Reinartz, P. Towards global-scale seagrass mapping and monitoring using Sentinel-2 on Google Earth Engine: The case study of the Aegean and Ionian Seas. Remote Sens. 2018, 10, 1227. [CrossRef]
- Mederos-Barrera, A.; Marcello, J.; Eugenio, F.; Hernández, E. Seagrass mapping using high resolution multispectral satellite imagery: A comparison of water column correction models. Int. J. Appl. Earth Obs. Geoinf. 2022, 113, 102990. [CrossRef]
- McCarthy, E.; Sabol, B. Acoustic characterization of submerged aquatic vegetation: military and environmental monitoring applications. In Oceans 2000 MTS/IEEE Conference and Exhibition, Providence, USA, 2000; pp. 1957–1961.
- Vis, C.; Hudon, C.; Carignan, R. An evaluation of approaches used to determine the distribution and biomass of emergent and submerged aquatic macrophytes over large spatial scales. Aquat. Bot. 2003, 77, 187–201.
- Brown, C.J.; Smith, S.J.; Lawton, P.; Anderson, J.T. Benthic habitat mapping: A review of progress towards improved understanding of the spatial ecology of the seafloor using acoustic techniques. Estuar. Coast. Shelf Sci. 2011, 92, 502–520.
- Hossain, M.S.; Mazlan, H. Potential of Earth Observation (EO) technologies for seagrass ecosystem service assessments. Int. J. Appl. Earth Obs. Geoinf. 2019, 77, 15–29. [CrossRef]
- Pasqualini, V.; Pergent-Martini, C.; Clabaut, P.; Pergent, G. Mapping of Posidonia oceanica using aerial photographs and side-scan sonar: application to the island of Corsica (France). Estuar. Coast. Shelf Sci. 1998, 47, 359–367.
- Di Maida, G.; Tomasello, A.; Luzzu, F.; Scannavino, A.; Pirrotta, M.; Orestano, C.; Calvo, S. Discriminating between Posidonia oceanica meadows and sand substratum using multibeam sonar. ICES J. Mar. Sci. 2011, 68, 12–19. [CrossRef]
- Mutlu, E. Acoustical identification of the concentration layer of a copepod species, Calanus euxinus. Mar. Biol. 2003, 142, 517–523. [CrossRef]
- Mutlu, E. Diel vertical migration of Sagitta setosa as inferred acoustically in the Black Sea. Mar. Biol. 2006, 149, 573–584. [CrossRef]
- Lavery, A.C.; Wiebe, P.H.; Stanton, T.K.; Lawson, G.L.; Benfield, M.C.; Copley, N. Determining dominant scatterers of sound in mixed zooplankton populations. J. Acoust. Soc. Am. 2007, 122, 3304–3326.
- Mutlu, E. A package of script codes, POSIBIOM, for vegetation acoustics: POSIdonia BIOMass. J. Mar. Sci. Eng. 2023, 11, 1790. [CrossRef]
- Mutlu, E.; Olguner, C. Acoustic scattering properties of seagrass: In/ex situ measurements of Posidonia oceanica. Medit. Mar. Sci. 2023, 24, 272–291. [CrossRef]
- Mutlu, E.; Olguner, C. Acoustic scattering properties of a seagrass, Cymodocea nodosa: In-situ measurements. Bot. Mar. 2023, 6, 491–505. [CrossRef]
- Carbó, R.; Molero, A.C. Scattering strength of a Gelidium biomass bottom. Appl. Acoust. 1997, 51, 343–351. [CrossRef]
- Shao, H.; Minami, K.; Shirakawa, H.; Kawauchi, Y.; Matsukura, R.; Tomiyasu, M.; Miyashita, K. Target strength of a common kelp species, Saccharina japonica, measured using a quantitative echosounder in an indoor seawater tank. Fish. Res. 2019, 214, 110–116. [CrossRef]
- Llorens-Escrich, S.; Tamarit, E.; Hernandis, S.; Sánchez-Carnero, N.; Rodilla, M.; Pérez-Arjona, I.; Moszynski, M.; Puig-Pons, V.; Tena-Medialdea, J.; Espinosa, V. Vertical configuration of a side scan sonar for the monitoring of Posidonia oceanica meadows. J. Mar. Sci. Eng. 2021, 9, 1332. [CrossRef]
- Minami, K.; Kita, C.; Shirakawa, H.; Kawauchi, Y.; Shao, H.; Tomiyasu, M.; Iwahara, Y.; Takahara, H.; Kitagawa, T.; Miyashita, K. Acoustic characteristics of a potentially important macroalgae, Sargassum horneri, for coastal fisheries. Fish. Res. 2021, 240, 105955. [CrossRef]
- Mutlu, E.; Balaban, C. New algorithms for the acoustic biomass estimation of Posidonia oceanica: A study in the Antalya Gulf (Turkey). Fresenius Environ. Bull. 2018, 27, 2555–2561.
- Mutlu, E.; Olguner, C. Density-dependent acoustical identification of two common seaweeds (Posidonia oceanica and Cymodocea nodosa) in the Mediterranean Sea. Thalassas 2023, 39, 1155–1167. [CrossRef]
- Mutlu, E.; Olguner, C.; Özvarol, Y.; Gökoğlu, M. A preliminary study on acoustic-based estimation of biometric parameters of Posidonia oceanica. Thalassas 2026, (under 2nd review).
- Mutlu, E.; Özvarol, Y.; Akçalı, B.; Aslan, B.E.; Seçkiner, S. Determination of distributional maximum biomass (summer) of Posidonia oceanica along the Turkish waters of the Aegean Sea and its seasonal dynamics in Side’s bed (Antalya, Mediterranean Sea) using the acoustic method. TÜBİTAK Project, grant no: 124Y031, 2nd Interim report, Ankara, Turkey, pp. 64 (in progress).
- Mutlu, E.; Olguner, C.; Özvarol, Y.; Gökoğlu, M. Spatiotemporal biometrics of Cymodocea nodosa in a western Turkish Mediterranean coast. Biologia 2022, 77, 649–670. [CrossRef]
- Mutlu, E.; Özvarol, Y.; Akçalı, B.; Aslan, B.E.; Seçkiner, S. Determination of distributional maximum biomass (summer) of Posidonia oceanica along the Turkish waters of the Aegean Sea and its seasonal dynamics in Side’s bed (Antalya, Mediterranean Sea) using the acoustic method. TÜBİTAK Project, grant no: 124Y031, 1st Interim Report, Ankara, Turkey, pp. 127 (in progress).
- Mutlu, E.; Olguner, C.; Gökoğlu, M.; Özvarol, Y. Population dynamics and ecology of Caulerpa prolifera vs Caulerpa taxifolia var. distichophylla within a Levantine Gulf. Thalassas 2022, 38, 1311–1325. [CrossRef]
- Mavko, G.; Mukerji, T.; Dvorkin, J. The Rock Physics Handbook, 2nd ed.; Cambridge University Press: Cambridge, UK, 1998.
- Aleman, P.B. Acoustic impedance inversion of lower Permian carbonate buildups in the Permian Basin, Texas. Master's Thesis, Texas A&M University, 2004; 99.
- Enriquez, S.; Schubert, N. Direct contribution of the seagrass Thalassia testudinum to lime mud production. Nat. Commun. 2004, I, 3835. [CrossRef]
- Balestri, E.; Cinelli, F. Sexual reproductive success in Posidonia oceanica. Aquatic Bot. 2003, 75, 21–32. [CrossRef]
- Balestri, E.; de Battisti, D.; Vallerini, F.; Lardicci, C. First evidence of root morphological and architectural variations in young Posidonia oceanica plants colonizing different substrate typologies. Estuar. Coast. Shelf Sci. 2015, 154, 205–213. [CrossRef]
- Pereda-Briones, L.; Infantes, E.; Orfila, A.; Tomas, F.; Terrados, J. Dispersal of seagrass propagules: Interaction between hydrodynamics and substratum type. Mar. Ecol. Prog. Ser. 2018, 593, 47–59. [CrossRef]
- Marba, N.; Duarte, C.M.; Holmer, M.; Martínez, R.; Basterretxea, G.; Orfila, A.; Jordi, A.; Tintoré, J. Effectiveness of protection of seagrass (Posidonia oceanica) populations in Cabrera National Park (Spain). Environ. Conserv. 2002, 29, 509–518. [CrossRef]
- De Falco, G.; Baroli, M.; Cucco, A.; Simeone, S. Intrabasinal conditions promoting the development of a biogenic carbonate sedimentary facies associated with the seagrass Posidonia oceanica. Cont. Shelf Res. 2008, 28, 797–812. [CrossRef]
- Montefalcone, M.; Vacchi, M.; Archetti, R.; Ardizzone, G.; Astruch, P.; Bianchi, C.N.; Calvo, S.; Criscoli, A.; Fernandez-Torquemada, Y.; Luzzu, F.; Misson, G.; Morri, C.; Pergent, G.; Tomasello, A.; Ferrari, M. Geospatial modelling and map analysis allowed measuring regression of the upper limit of Posidonia oceanica seagrass meadows under human pressure. Estuar. Coast. Shelf Sci. 2019, 217, 148–157. [CrossRef]
- Yalçın, M.G.; Mutlu, E.; Olguner, C.; Atakoğlu, ÖÖ.; Bat, L.; Özkan, E.Y. Spatial geochemical structure of soft sediment on shallow littoral of the Gulf of Antalya, the eastern Mediterranean Sea. Mar. Poll. Bull. 2023, 193, 115155. [CrossRef]
- Milliman, J.D. Production and accumulation of calcium carbonate in the ocean: Budget of a nonsteady state. Glob. Biogeochem. Cyc. 1993, 7, 927–957. [CrossRef]
- Canals, M.; Ballesteros, E. Production of carbonate particles by phytobenthic communities on the Mallorca-Menorca shelf, northwestern Mediterranean Sea. Deep-Sea Res. II 1997, 44, 611–629.
- Gnisci, V.; Martiis, S.C.; Belmonte, A.; Micheli, C.; Piermattei, V.; Bonamano, S.; Marcelli, M. Assessment of the ecological structure of Posidonia oceanica (L.) Delile on the northern coast of Lazio, Italy (central Tyrrhenian, Mediterranean). Ital. Bot. 2020, 9, 1–19. [CrossRef]
- Sandoval-Gil, J.M.; Ruiz, J.M.; Marín-Guirao, L.; Bernardeau-Esteller, J.; Sánchez-Lizaso, J.L. Ecophysiological plasticity of shallow and deep populations of the Mediterranean seagrasses Posidonia oceanica and Cymodocea nodosa in response to hypersaline stress. Mar. Environ. Res. 2014, 95, 39–61. [CrossRef]
- Lal, A.; Arthur, R.; Marba, N.; Lill, A.W.T.; Alcoverro, T. Implications of conserving an ecosystem modifier: increasing green turtle (Chelonia mydas) densities substantially alters seagrass meadows. Biol. Conserv. 2010, 143, 2730–2738. [CrossRef]
- Steele, L.; Darnell, K.M.; Cebrian, J.; Sanchez-Lizaso, J.L. Sarpa salpa herbivory on shallow reaches of Posidonia oceanica beds. Anim. Biodiv. Conserv. 2014, 37, 49–57.
- Marba, N.; Duarte, C.M.; Cebrian, J.; Gallegos, M.E.; Olesen, B.; Sand-Jensen, K. Growth and population dynamics of Posidonia oceanica on the Spanish Mediterranean coast: elucidating seagrass decline. Mar. Ecol. Prog. Ser. 1996, 137, 203–213. [CrossRef]
- Guidetti, P.; Lorenti, M.; Buia, M.C.; Mazzella, L. Temporal dynamics and biomass partitioning in three Adriatic seagrass species, Posidonia oceanica, Cymodocea nodosa, Zostera marina. PSZNI Mar. Ecol. 2002, 23, 51–67. [CrossRef]
- Bay, D. A field study of the growth dynamics and productivity of Posidonia oceanica (L.) Delile in Calvi Bay, Corsica. Aquat. Bot. 1984, 20, 43–64. [CrossRef]
- Wittmann, K.J. Temporal and morphological variations of growth in a natural stand of Posidonia oceanica (L.) Delile. Mar. Ecol. 1984, 5, 301–316. [CrossRef]
- Sgorbini, S.; Peirano, A.; Cocito, S.; Morgigni, M. An underwater tracking system for mapping marine communities: An application to Posidonia oceanica. Oceanol. Acta 2002, 25, 135–138. [CrossRef]
- Gobert, S.; Kyramarios, M.; Lepoint, G.; Pergent-Martini, C.J.; Bouquegneau, J.-M. Variations at different spatial scales of the Posidonia oceanica (L.) Delile meadow; effects on the physicochemical parameters of the sediment. Oceanol. Acta 2003, 26, 199–207.
- Maida, G.D.I.; Tomasello, A.; Sciandra, M.; Pirrotta, M.; Milazzo, M.; Calvo, S. Effect of different substrata on rhizome growth, leaf biometry and shoot density of Posidonia oceanica. Mar. Environ. Res. 2013, 87–88, 96–102. [CrossRef]
- Giovannetti, E.; Lasagna, R.; Montefalcone, M.; Bianchi, C.N.; Albertelli, G.; Morri, C. Inconsistent responses to substratum nature in Posidonia oceanica meadows: An integration through complexity levels? Chem. Ecol. 2008, 24, S83–S91. [CrossRef]
- Touchette, B.W.; Burkholder, J.M. Overview of the physiological ecology of carbon metabolism in seagrasses. J. Exp. Mar. Biol. Ecol. 2000, 250, 169–205. [CrossRef]
- Perez, M.; Duarte, C.M.; Romero, J.; Sand-Jensen, K.; Alcoverro, T. Growth plasticity in Cymodocea nodosa stands: The importance of nutrient supply. Aquat. Bot. 1994, 47, 249–264. [CrossRef]
- Marbà, N.; Duarte, C.M. Rhizome elongation and seagrass clonal growth. Mar. Ecol.-Prog. Ser. 1998, 255, 127–134. [CrossRef]
- Fernandez-Torquemada, Y.; Sanchez-Lizaso, J.L. Effects of salinity on leaf growth and survival of the Mediterranean seagrass Posidonia oceanica (L.) Delile. J. Exp. Mar. Biol. Ecol. 2005, 320, 57–63. [CrossRef]
- Via, J.D.; Sturmbauer, C.; Schonweger, G.; Sotz, E.; Mathekowitsch, S.; Stifter, M.; Rieger, R. Light gradients and meadow structure in Posidonia oceanica: Ecomorphological and functional correlates. Mar. Ecol.-Prog. Ser. 1998, 163, 267–278. [CrossRef]
- Sghaier, Y.R.; Zakhama-Sraieb, R.; Charfi-Cheikhrouha, F. Status of Posidonia oceanica along eastern coast of Tunisia. Biol. Mar. Medit. 2006, 13, 85–91.
- Dalmau, A.; Gubbay, S.; Garcia-Herrero, A. Posidonia beds (Posidonion oceanicae) (1120*). In: Olmeda, C.; Šefferová Stanová, V. (Eds.) Technical guidelines for assessing and monitoring the condition of Annex I habitat types of the Directive 92/43/EEC; Publications Office of the European Union: Luxembourg, 2025; ISBN 978-92-68-32012-9. [CrossRef]
- Mutlu, E.; Gökoğlu, M.; Özvarol, Y.; Balaban, C.; Olguner, M.T. Yaygın deniz çayırlarının akustiksel yoğunluk kalibrasyonu ve dağılımlarının takip edilmesi [Acoustical density-dependent calibration of the dominant sea meadows and seagrasses and monitoring of their distribution]. Final Report, TUBITAK, no: 110Y232, Ankara, Turkey, 2014, pp. 337.
- Mutlu, E.; Meo, I.d.; Miglietta, C.; Deval, M.C. Ecological Indicative Stressors of Native vs Non-Native Fish in an Ultra-Oligotrophic Region of the Mediterranean Sea. Sustainability 2023, 15, 2726. [CrossRef]
- Stanton, T.K.; Chu, D.; Wiebe, P.H. Acoustic scattering characteristics of several zooplankton groups. ICES J. Mar. Sci. 1996, 53, 289–295.
- Simmonds, J.; Maclennan, D. Fisheries acoustics: Theory and practice, 2nd ed.; Blackwell Publishing: 2005; pp. 456.
- Alcoverro, T.; Romero, J.C.M.; Duarte, N.; Lopez, L. Spatial and temporal variations in nutrient limitation of seagrass Posidonia oceanica growth in the NW Mediterranean. Mar. Ecol. Prog. Ser. 1997, 146, 155–161. [CrossRef]
- Pirc, H. Seasonal aspects of photosynthesis in Posidonia oceanica: Influence of depth, temperature, and light intensity. Aquat. Bot. 1986, 26, 203–212. [CrossRef]
- Ruiz, J.M.; Romero, J. Effects of in situ experimental shading on the Mediterranean seagrass Posidonia oceanica. Mar. Ecol.-Prog. Ser. 2001, 215, 107–120. [CrossRef]
- Mateo, M.A.; Romero, J.; Perez, M.; Littler, M.M.; Littler, D.S. Dynamics of millenary organic deposits resulting from the growth of the Mediterranean seagrass Posidonia oceanica. Estuar. Coast. Shelf Sci. 1997, 44, 103–110. [CrossRef]
- Merriam, C.O. Depositional history of lower Permian (Wolfcampian - Leonardian) carbonate buildups, Midland Basin, Upton County, Texas. M.S. thesis, Texas A&M University, 1999.
- Karaca, D.; Mutlu, E.; Uysal, Z. Summer surface phytoplankton assemblages along physically discrete water masses of the entire Turkish Mediterranean coast. Thalassas 2026, (in press).
- Lepoint, G.; Defawe, O.; Gobert, S.; Dauby, P.; Bouquegneau, J.-M. Experimental evidence for nitrogen recycling in the leaves of the seagrass Posidonia oceanica. J. Sea Res. 2002, 48, 173–179. [CrossRef]
- Marín-Guirao, L.; Sandoval-Gil, J.M.; Bernardeau-Esteller, J.; Ruiz, J.M.; Sánchez-Lizaso, J.L. Responses of the Mediterranean seagrass Posidonia oceanica to hypersaline stress duration and recovery. Mar. Environ. Res. 2013, 84, 60–75. [CrossRef]
| Echosounder parameters | Values |
|---|---|
| Manufacturer and model | BioSonics (USA) and DT-X |
| Acoustic frequency | 206 kHz |
| Transducer type and shape | Split and circle |
| Source level | 220.4 dB re µPa at 1 m |
| Receive sensitivity, narrow-beam | -51.0 dB re counts per µPa |
| Receive sensitivity, wide-beam | -56.0 dB re counts per µPa |
| Beam width | 6.8 x 6.8° |
| System noise floor | -140 dB |
| Echosounder settings | |
| Transducer draft | 2.5 m from the surface water |
| Ping rate | 5 pings s-1 |
| Sound speed* | calculated by Visual Acquisition |
| Absorption coefficient* | calculated by Visual Acquisition |
| Data collection threshold | -140 dB |
| Pulse width | 0.1 ms |
| Maximum depth | 70 (to-250) m |
| Source | d.f. | L | LAI | B1 | S | Lno |
|---|---|---|---|---|---|---|
| M | 1 | 0.001 | 0.000 | 0.000 | 0.000 | 0.103 |
| S | 1 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| B | 3 | 0.047 | 0.006 | 0.011 | 0.002 | 0.016 |
| D | 6 | 0.089 | 0.019 | 0.023 | 0.070 | 0.018 |
| M x S | 1 | 0.000 | 0.000 | 0.000 | 0.000 | 0.258 |
| M x B | 3 | 0.039 | 0.000 | 0.000 | 0.000 | 0.018 |
| M x D | 5 | 0.064 | 0.002 | 0.003 | 0.009 | 0.035 |
| S x B | 3 | 0.766 | 0.122 | 0.170 | 0.223 | 0.519 |
| S x D | 3 | 0.867 | 0.525 | 0.425 | 0.489 | 0.693 |
| B x D | 15 | 0.743 | 0.921 | 0.953 | 1.000 | 0.794 |
| M x S x B | 3 | 0.224 | 0.879 | 0.896 | 0.045 | 0.513 |
| M x S x D | 3 | 0.685 | 0.565 | 0.440 | 0.597 | 0.599 |
| M x B x D | 14 | 0.834 | 0.920 | 0.961 | 0.976 | 0.711 |
| S x B x D | 6 | 0.690 | 0.886 | 0.912 | 0.837 | 0.236 |
| M x S x B x D | 6 | 0.743 | 0.767 | 0.773 | 0.887 | 0.285 |
| Error | 214 | |||||
| Total | 287 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




