Submitted:
11 March 2026
Posted:
12 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Uranium Series Imbalance
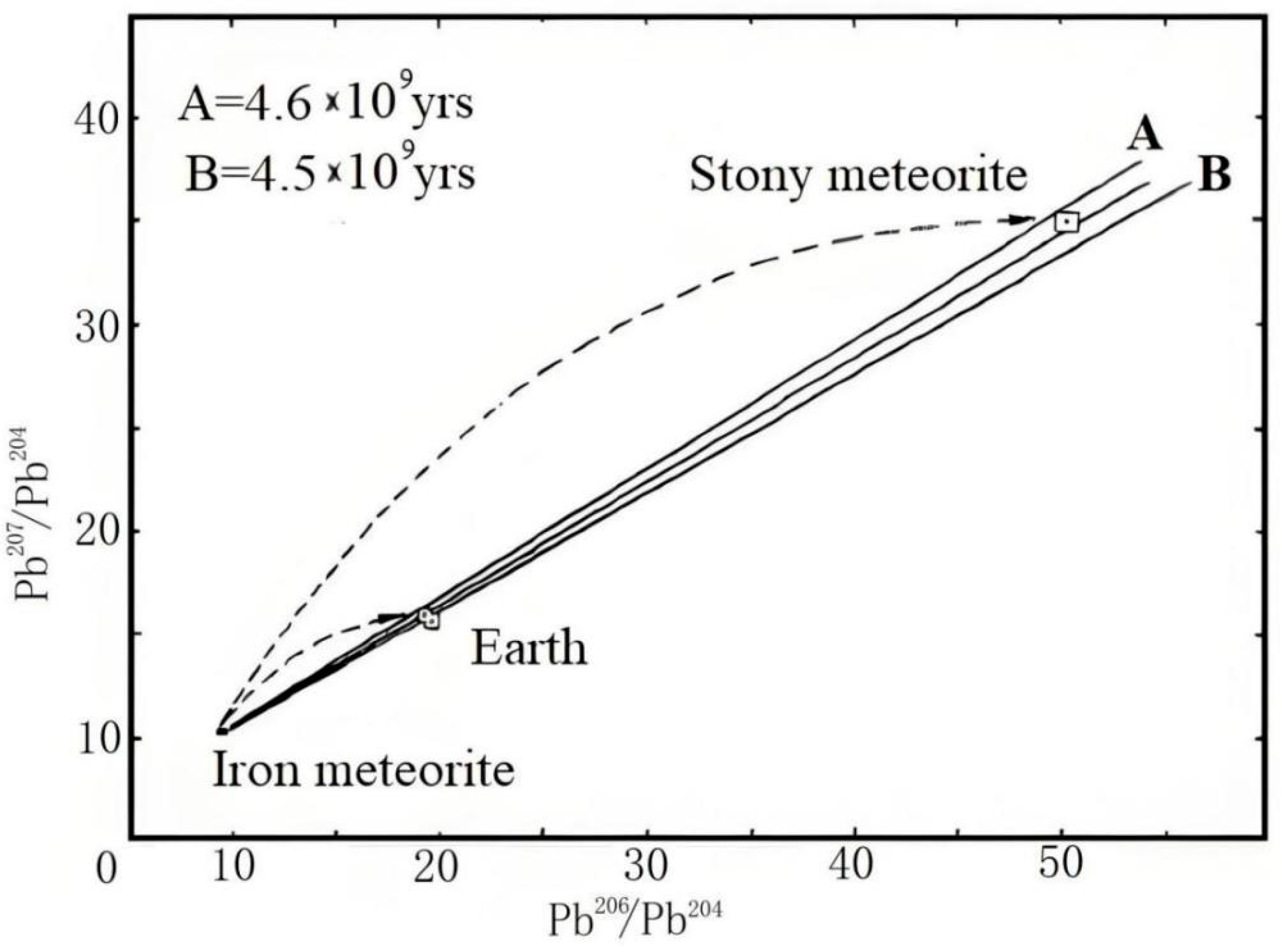
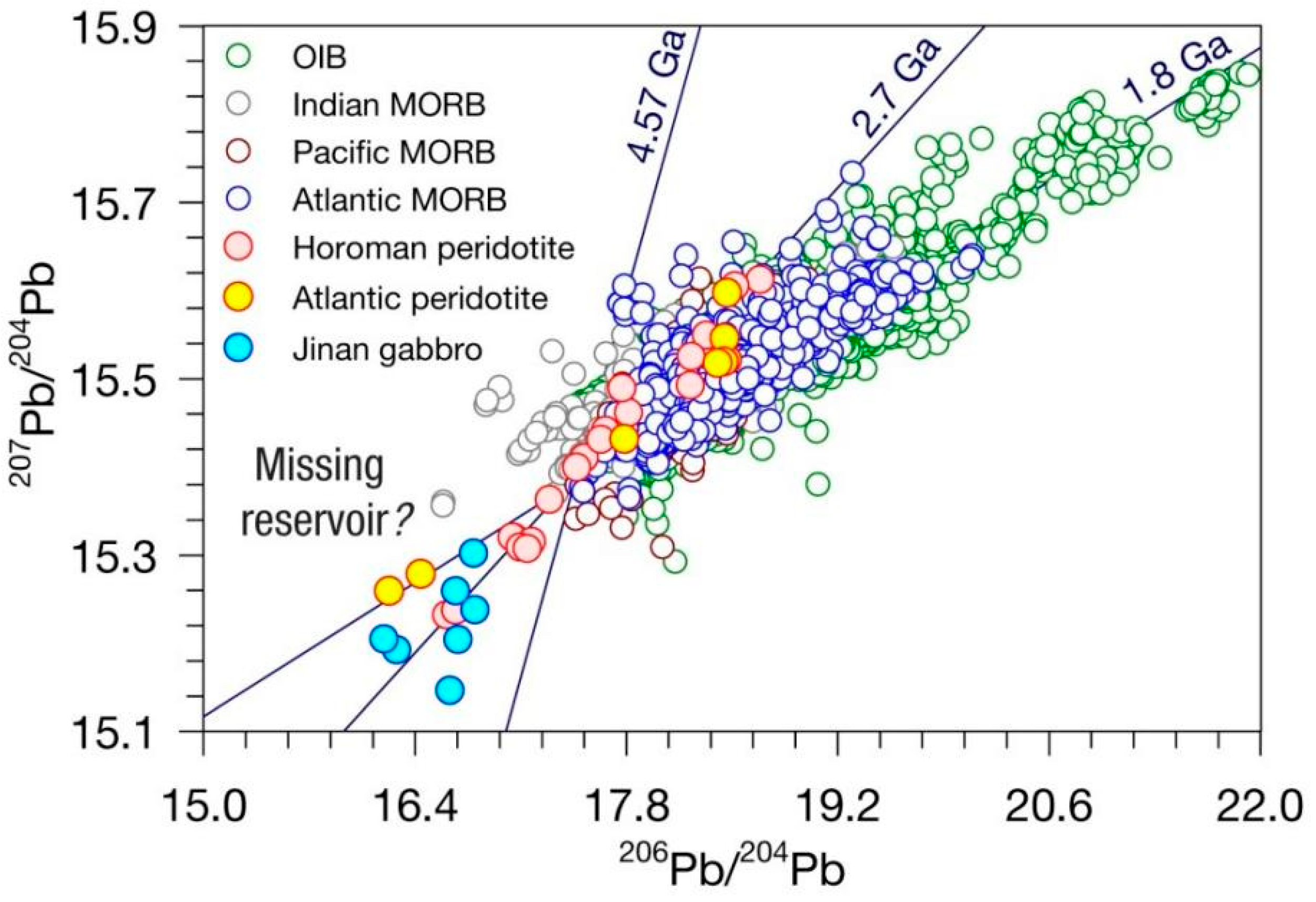
1.2. The Lead Paradox
1.3. Excess Argon
2. Neutrino Oscillation and Magma Formation
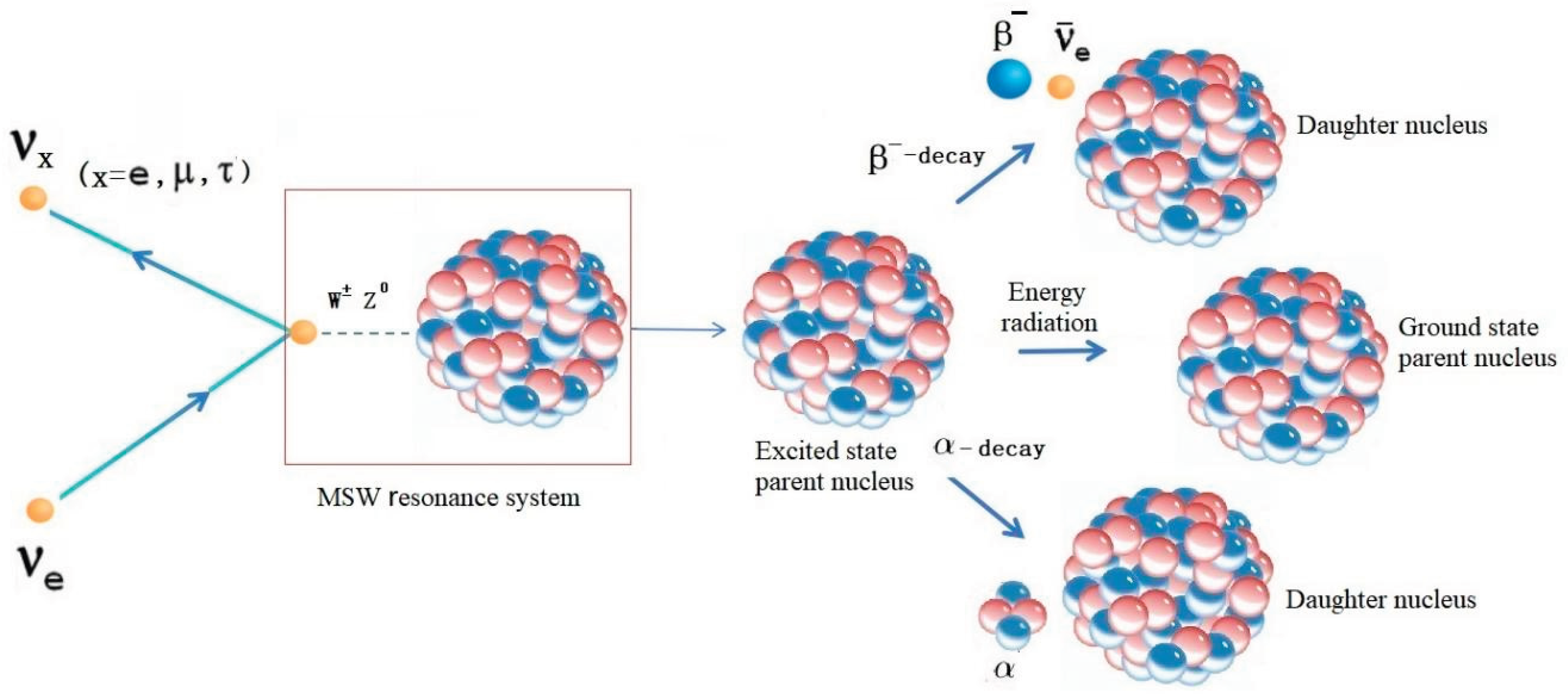
2.1. Neutrino Oscillation-Induced Radioactive Decay
2.2. Formation and Migration of Magma
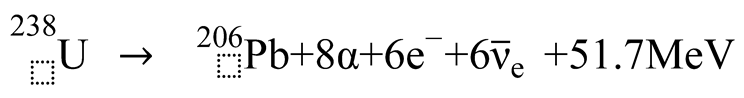
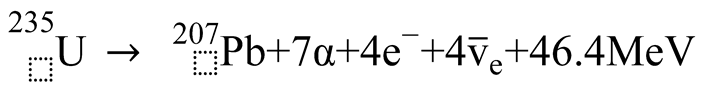
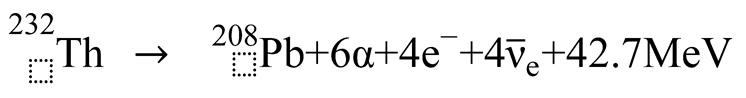
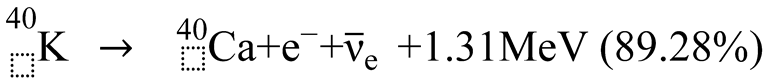
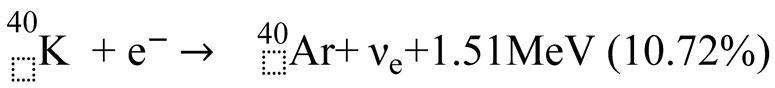
3. Discussion
3.1. Formation of Uranium-Series Imbalance and the Lead Paradox
3.2. Causes of Excess Argon and ⁴⁰K Deficiency
3.3. On the Issue of Radiometric Dating
4. Conclusions
References
- Zhang, GL; Zeng, ZG; Yin, XB. U-series disequilibrium on magma generation and transportation beneath mid-ocean ridge axes. Marine Geology & Quaternary Geology 2008, 28(1), 77–84. [Google Scholar] [CrossRef]
- Doucet, LS; Li, ZX; Fougerouse, D; et al. The global lead isotope system: Toward a new framework reflecting Earth's dynamic evolution. Earth-Science Reviews 2023, 243, 104483. [Google Scholar] [CrossRef]
- Hofmann, AW. The enduring lead paradox. Nature Geosci 2008, 1(12), 812–813. [Google Scholar] [CrossRef]
- Aldrich, LT. Nier AO (1948) Argon 40 in Potassium Minerals. Physical Review 74, 876–877. [CrossRef]
- Li, DP; Chen, YL; Luo, ZH. 40Ar/39Ar dating of biotites and hornblendes in the Kangding complexes and their significances. Acta Petrologica Sinica 2006, 22(11), 2753–2761. Available online: http://ysxb.ac.cn/article/id/aps_2006011294.
- Zhang, GL; Zheng, ZG. Genesis of 230Th excess in basalts from mid-ocean ridges and ocean islands: Constraints from the global U-series isotope database and major and rare earth element geochemistry. Sci China Earth Sci 2010, 40(10), 1389–1398. [Google Scholar] [CrossRef]
- Beattiea, P. The generation of uranium series disequilibria by partial melting of spinel peridotite: Constraints from partitioning studies. Earth Planet Sci Lett 1993, 117, 379–391. [Google Scholar] [CrossRef]
- Lundstrom, CC; Hoernle, K; Gill, J. U-series disequilibria in volcanic rocks from the Canary islands: Plume versus lithospheric melting. Geochim Cosmochim Acta 2003, 67, 4153–4177. [Google Scholar] [CrossRef]
- Peate, DW; Hawkesworth, CJ; Van Calsteren, PW. 238U-230Th constraints on mantle upwelling and plume-ridge interaction along theReykjanes ridge. Earth Planet Sci Lett 2001, 187, 259–272. [Google Scholar] [CrossRef]
- Rubin, KH; Macdougall, JD. 226Ra excesses in mid-ocean-ridge basalts and mantle melting. Nature 1988, 335, 158–161. [Google Scholar] [CrossRef]
- Volpe, AM; Goldstein, SJ. 226Ra-230Th disequilibrium in axial and off-axis mid-ocean ridge basalts. Geochimica Et Cosmochimica Acta 1992, 57, 1233–1241. [Google Scholar] [CrossRef]
- Zou, HB; Fan, QC. Uranium-thorium isotope disequilibrium in young volcanic rocks from China. Acta Petrologica Sinica 2011, 27(10), 2821–2826. [Google Scholar]
- Turner, S; Blundy, J; Wood, B; Hole, M. Large 230Th-excesses in basalts produced by partial melting of spinel lherzolite. Chem Geol 2000, 162, 127–136. [Google Scholar] [CrossRef]
- Wood, B; Blundy, J; Robinson, J. The role of clinopyroxene in generating U-series disequilibrium during mantle melting:Implications for uranium series disequilibria in basalts. Geochim Cosmochim Acta 1999, 63, 1613–1620. [Google Scholar] [CrossRef]
- Lundstrom, CC; Williams, Q; Gill, JB. Investigating solid mantle upwelling rates beneath mid-ocean ridges using U-series disequilibria,1: A global approach. Earth Planet Sci Lett 1998, 157, 151–165. [Google Scholar] [CrossRef]
- Avanzinelli, R; Casalini, M; Conticelli, S. Carbon fluxes from subducted carbonates revealed by uranium excess at Mount Vesuvius, Italy. Geology 2018, 46(3), 259–262. [Google Scholar] [CrossRef]
- Huang, F; Lundstrom, CC; Sigurdsson, H; et al. U-series disequilibria in Kick’em Jenny submarine volcano lavas: A new view of time-scales of magmatism in convergent margins. Geochimica et Cosmochimica Acta 2011, 75, 195–212. [Google Scholar] [CrossRef]
- Patterson, C. Age of meteorites and the earth. Geochim. Cosmochim. Acta 1956, 10, 230–237. [Google Scholar] [CrossRef]
- Bouvier, A; Blichert-Toft, J; Moynier, F; et al. Pb-Pb dating constraints on the accretion and cooling history of chondrites. Geochim. Cosmochim. Acta 2007, 71, 1583–1604. [Google Scholar] [CrossRef]
- Zhang, JB; Liu, YS; Ducea, MN; et al. Archean, highly unradiogenic lead in shallow cratonic mantle. Geology 2020, 48(6), 584–588. [Google Scholar] [CrossRef]
- Galer, SJG; O^Nions, RK. Residence time of thorium, uranium and lead in the mantle with implications for mantle convection. Nature 1985, 316, 778–782. [Google Scholar] [CrossRef]
- Allegre, CJ; Dupre, B; Brevart, O. Chemical aspects of the formation of the core. Phil. Trans. Royal Soc. London 1982, 306, 49–59. [Google Scholar] [CrossRef]
- Kramers, DJ; Tolstikhim, IN. Two terrestrial lead isotope paradoxes, forward transport modeling, core formation and the history of the continental crust. Chem. Geol 1997, 139, 75–110. [Google Scholar] [CrossRef]
- O^Nions, RK; Evenson, NM; Hamilton, PJ. Geochemical modeling of mantle differentiation and crustal growth. J. Geophys Res 1979, 84, 6091–6101. [Google Scholar] [CrossRef]
- Zartman, RE; Haines, S. The plumbotectonic model for Pb isotopic systematics among major terrestrial reservoirs—a case for bi-directional transport. Geochim. Cosmochim. Acta 1988, 52, 1327–1339. [Google Scholar] [CrossRef]
- Chauvel, D; Goldstain, SL; Hofmann, AW. Hydration and dehydration of oceanic crust controls Pb evolution of the mantle. Chem. Geol 1995, 126, 65–75. [Google Scholar] [CrossRef]
- Murphy, DT; Kamber, BS; Collerson, KD. A refined solution to the first terrestrial Pb-isotope paradox. J. Petrol 2002, 44, 39–53. [Google Scholar] [CrossRef]
- Cortini, M. Uranium in mantle processes. In Uranium geochemistry, mineralogy, geology, exploration and resources; De Vivo, B, Ippolito, F, Capaldi, G, Simpson, PR, Eds.; Springer: Dordrecht, 1984. [Google Scholar] [CrossRef]
- Ke, S; Luo, ZH; Mo, XX; et al. The geochronology of Taxkorgan alkalic complex, Pamir syntax. Acta Petrologica Sinica 2008, 24(2), 315–324. Available online: http://ysxb.ac.cn/en/article/id/aps_20080211.
- Li, DM; Chen, WJ; Li, Q. K-Ar age of young volcanic rocks and excess argon-Binary mixing model and quantitative study of excess argon effect. Chinese Science Bulletin 1999, 44(7), 658–663. [Google Scholar] [CrossRef]
- Li, S; Wang, S; Chen, Y. Excess argon in phengite from eclogite:evidence from the dating of eclogite minerals by the Sm-Nd,Rb-Sr and 40Ar/39Ar methods. Chemical Geology 1994, 112(3-4), 343–350. [Google Scholar] [CrossRef]
- Chen, W; Zhang, Y; Wang, QL; et al. Major Advances in the Research on the Genesis of Excess Argon in High Pressure Muscovite. Acta Geoscientica Sinica 2005, 1(6), 495–498. [Google Scholar] [CrossRef]
- Scaillet, S. Excess 40Ar transport scale and mechanism in high-pressure phengites: A case study from an eclogitized metabasite of the Dora-Maira nappe, western Alps. Geochimica et Cosmochimica Acta 1996, 60(6), 1075–1090. [Google Scholar] [CrossRef]
- Zhang, GW; Zhang, M K. Research on Neutrino Oscillation-Induced Radioactive Decay. Modern Physics 2024, 14, 135–144. [Google Scholar] [CrossRef]
- Zhang, GW; Zhang, MK. Effects of Matter in Atmospheric Neutrino Oscillations and the Formation of Magma. Journal of Geoscience and Environment Protection 2024, 12, 270–287. [Google Scholar] [CrossRef]
- Zhang, MK; Zhang, GW. (2025) Dynamical Mechanisms of Melt Migration Beneath Mid-Ocean Ridges. Open Journal of Geology 15, 358–376. [CrossRef]
- Wolfenstein, L. Neutrino Oscillations in Matter. Physical Review D 1978, 17, 2369–2379. [Google Scholar] [CrossRef]
- Mikheyev, SP; Smirnov, AY. Resonant Neutrino Oscillations in Matter. Progress in Particle and Nuclear Physics 1989, 23, 41–136. [Google Scholar] [CrossRef]
- Gurney, RW; Condon, EU. Quantum Mechanics and Radioactive Disintegration. In Selected Scientific Papers of E.U. Condon; Barut, AO, Odabasi, H, van der Merwe, A., Eds.; Springer: New York, NY, 1991. [Google Scholar] [CrossRef]
- Fermi E (1934) Versuch einer Theorie derβ-Strahlen. I. Zeitschrift für Physik 88, 161.
- Upadhyay, AK; Kumar, A; Agarwalla, SK; et al. Locating the core-mantle boundary using oscillations of atmospheric neutrinos. J. High Energ. Phys. 2023, 68. [Google Scholar] [CrossRef]
- Dye, ST. Geoneutrinos and the radioactive power of the Earth. Reviews of Geophysics 2012, 50(3). [Google Scholar] [CrossRef]
- Honda, M; Kajita, T; Kasahara, K. Calculation of the Flux of Atmospheric Neutrinos. Physical Review D 1995, 52, 4985–5005. [Google Scholar] [CrossRef] [PubMed]
- Gaisser, TK; Honda, M. Flux of Atmospheric Neutrinos. Annual Review of Nuclear and Particle Science 2002, 52, 153–199. [Google Scholar] [CrossRef]
- Dziewonski, AM; Anderson, DL. Preliminary reference Earth model. Phys. Earth Planet. Inter 1981, 25, 297–356. [Google Scholar] [CrossRef]
- Liu, JH. Mineralogy and Geological Significance of the Melting Zone in the Jiaohe Upper Mantle Fragment, Jilin. Earth Science Frontiers 2020, 27, 48–60. [Google Scholar] [CrossRef]
- Zhang, MK; Zhang, GW. Mechanisms for the Formation of Metamorphic Temperature and Pressure Gradients in Orogenic Belts. Journal of Geoscience and Environment Protection 2025, 13, 85–101. [Google Scholar] [CrossRef]
- Zhang, MK; Zhang, GW. Neutrino Oscillation, Radioactive Decay, Magmatic Activity and Earthquake Formation. Open Journal of Earthquake Research 2025, 14, 172–193. [Google Scholar] [CrossRef]
- Dalrymple, GB; Moore, JG. Argon-40: Excess in Submarine Pillow Basalts from Kilauea Volcano, Hawaii. Science 1968, 161, 1132–1135. [Google Scholar] [CrossRef]
- Wang, D; Nie, NX; Peters, BJ. Potassium-40 isotopic evidence for an extant pre-giant-impact component of Earth’s mantle. Nat. Geosci 2025, 18, 1174–1179. [Google Scholar] [CrossRef]
- Wan, JL; Zheng, DW. Several notable problems on dating of young volcanic rocks by FT method-illustrated by dating of Changbaishan volcanic rocks. SEISMOLOGY AND GEOLOGY 2000, 22(s1), 19–24. [Google Scholar]
- Wang, SS. Excess argon-bearing phengite from uhpm eclogite: A new geochronological evidence. Chin.Sci.Bull 1998, 43 (Suppl 1), 140. [Google Scholar] [CrossRef]
- Chen, W; Li, SG; Zhang, Y. 40Ar/39Ar Chronological Study of the Muscovite from Qinglongshan High-Pressure Orthogneiss, Donghai County, Su-Lu UHP-Metamorphic Belt. Geological Review 2003, 49(5), 537–543. [Google Scholar] [CrossRef]
- Jourdan, F; Renne, PR; Reimold, WU. The problem of inherited 40Ar* in dating impact glass by the 40Ar/39Ar method: Evidence from the Tswaing impact crater (South Africa). Geochimica et Cosmochimica Acta 2007, 71, 1214–1231. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



