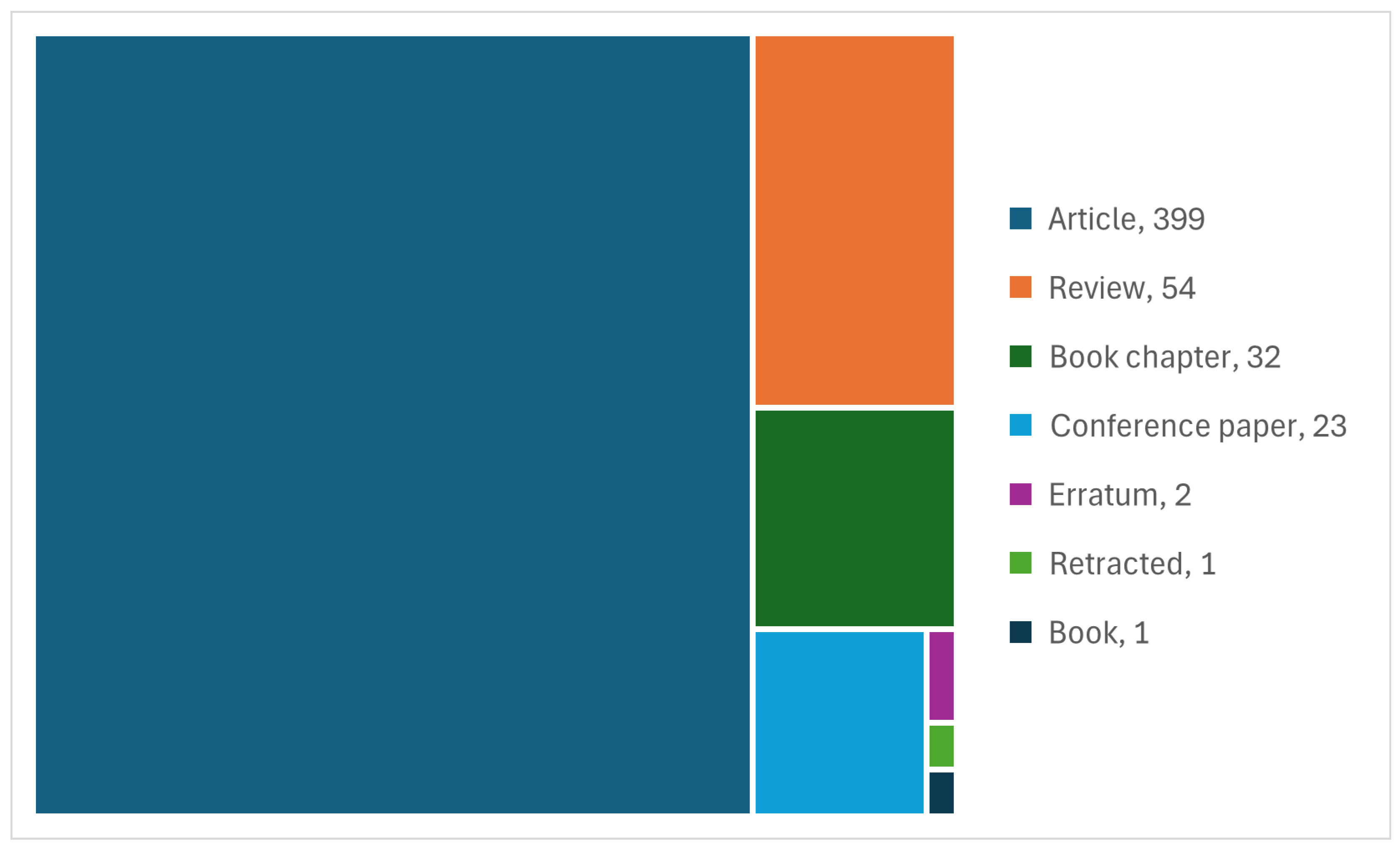
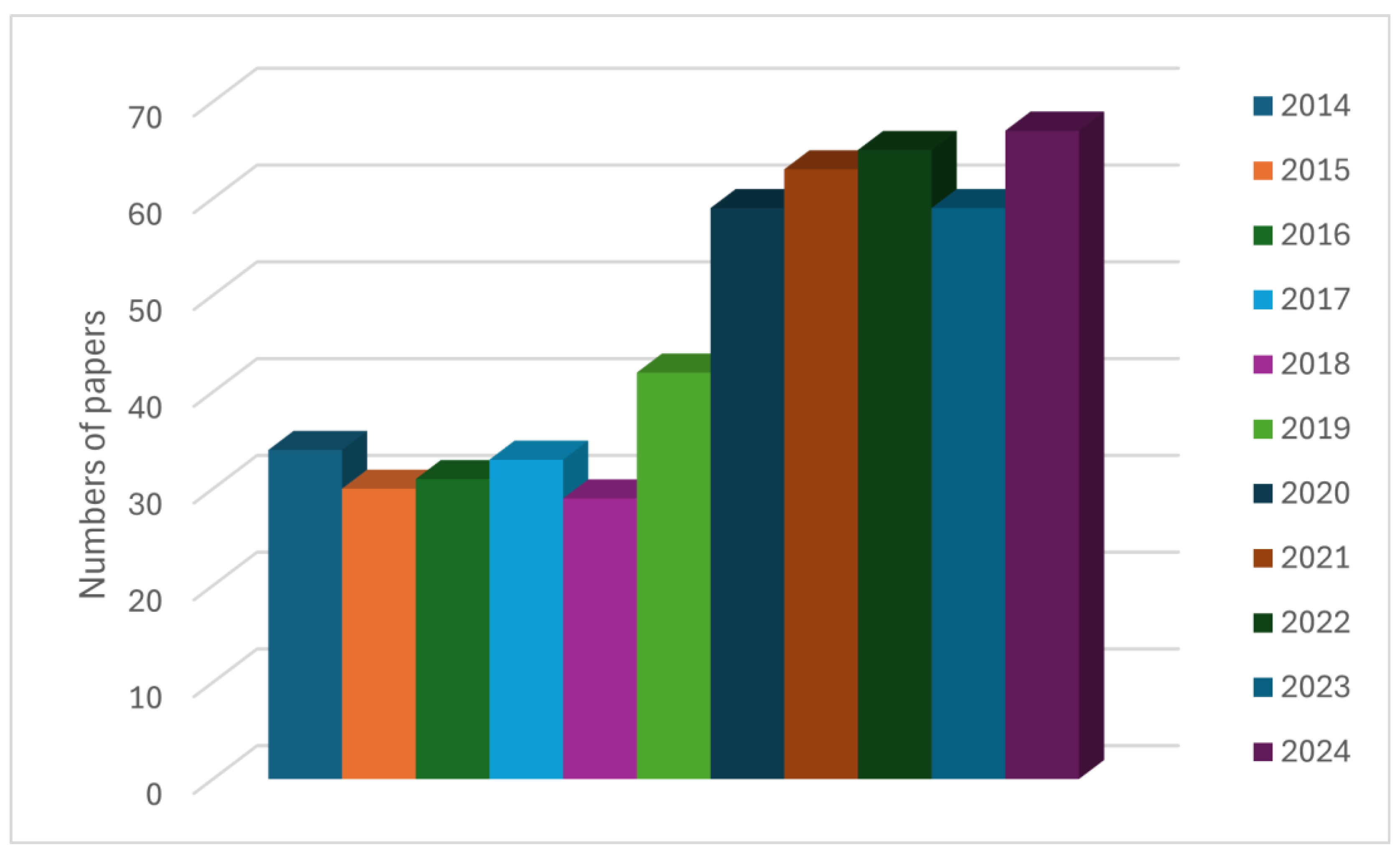
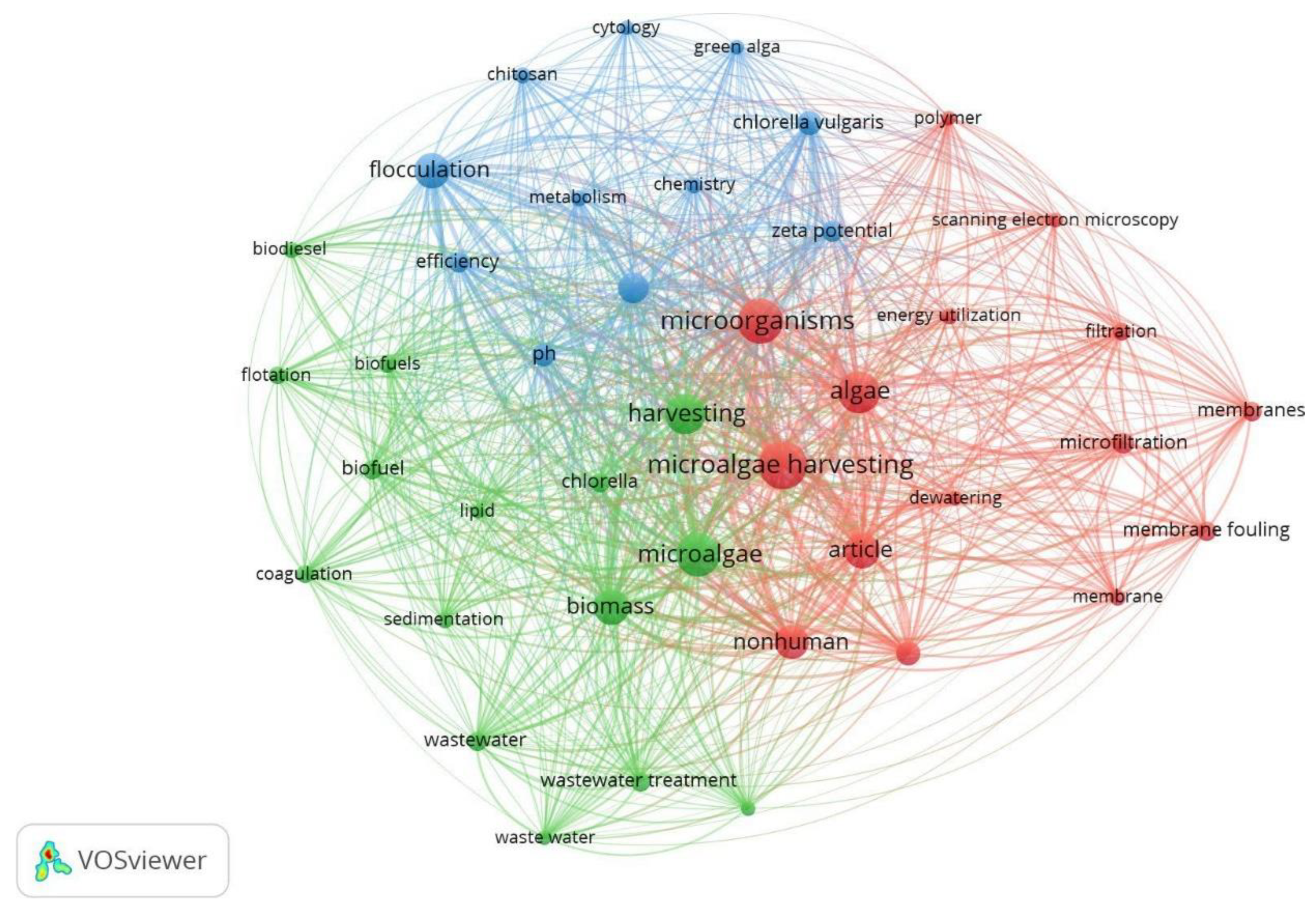
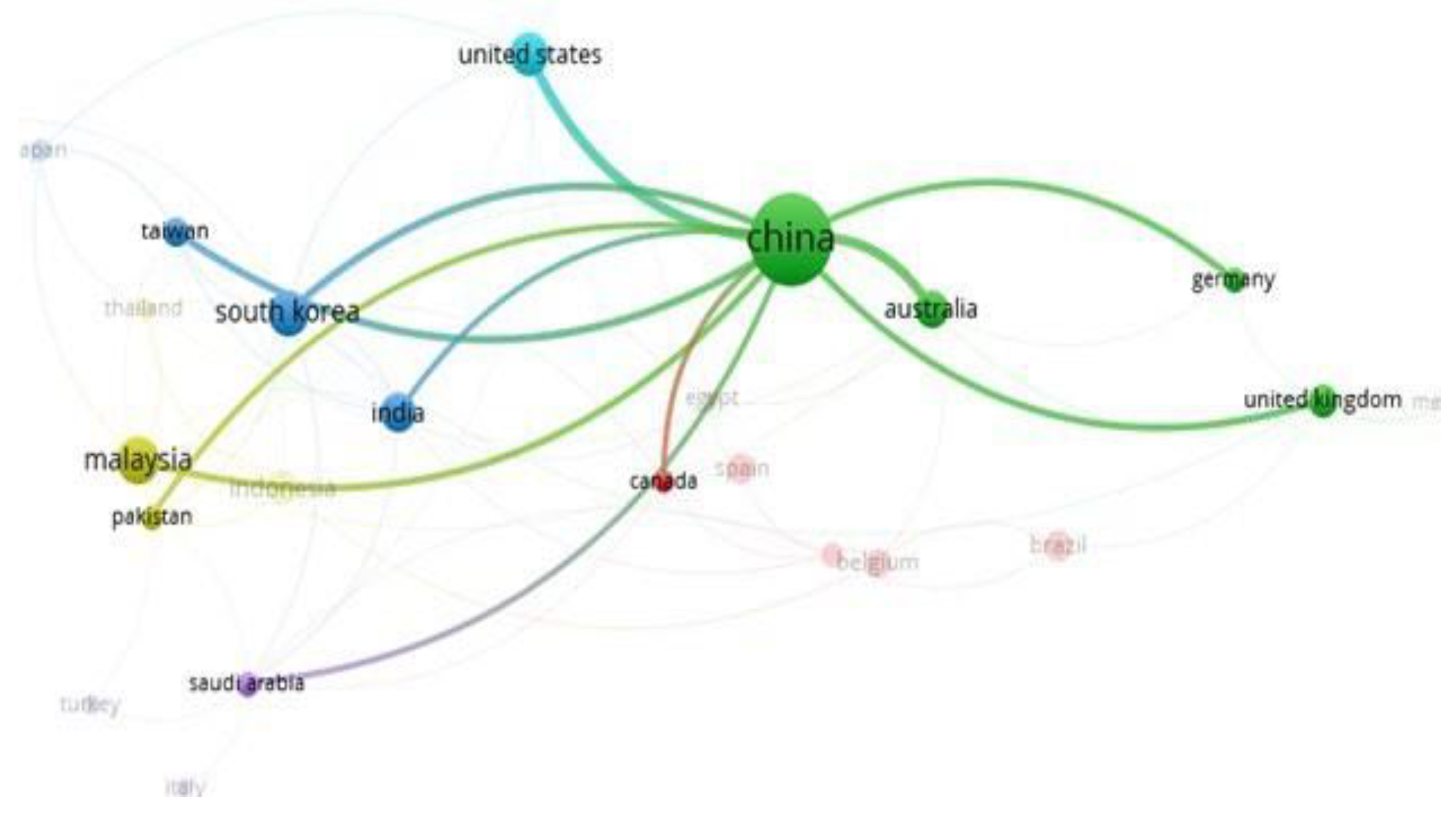
3.6. Technologies for Niomass Separation
This analysis aims to identify the most efficient and sustainable methods, enhancing wastewater treatment and management practices.
Among various harvesting techniques, sedimentation combined with coagulation and flocculation has proven to be the most economically and environmentally advantageous [
9].
Microalgae possess a negative charge due to the presence of carboxyl, hydroxyl, and amine groups on their cell membrane (quote). Consequently, coagulation and flocculation mechanisms can effectively destabilize these particles, facilitating their aggregation and subsequent separation from water. Coagulation offers several advantages: it is a quick, straightforward method suitable for large-scale applications across a wide range of species, causes minimal cell damage, and requires less energy. When bioflocculants are used, coagulation becomes an affordable and sustainable option, in contrast to other techniques that are costly, time-consuming, species-specific, and may even introduce toxicity to the harvested biomass [
11,
16]
A substantial amount of research has focused on flocculation as a critical technique for microalgae harvesting. The literature explores various flocculants, including chitosan,
Moringa oleifera (MO), and tannin-based coagulants, highlighting their impact on harvesting efficiency and biomass composition [
11,
16,
17]. While chitosan is known for its effectiveness, its high cost and potential for downstream contamination raise concerns when compared to more environmentally friendly natural flocculants like
Moringa oleifera. These natural options offer a more sustainable approach, but often require careful optimisation.
Tannin-based coagulants have demonstrated its potential results in the harvesting of microalgal biomass. Their plant-based origin offers a distinct advantage, as they pose minimal risk of metal contamination, making them particularly suitable for biomass reuse. This characteristic has led to growing interest in their application for harvesting microalgae, especially in nutrient-rich environments like domestic sewage. Vargas et al. [
16], evaluated the effectiveness of tannin-based coagulants Tanfloc SG, Tanfloc MTH, and Tanfloc MT for harvesting
Parachlorella kessleri microalgae biomass, cultivated in effluents from the thermal processing industry of chicken. The Tanfloc MT coagulant was the most efficient, reducing turbidity by 99.76% and apparent color by 80%. The harvested biomass presented 39.21% crude protein, surpassing that harvested by centrifugation (28.78%). In addition, the technique demonstrated greater sustainability by avoiding contamination of the biomass with metals, a common characteristic in inorganic coagulants.
Despite the good results recorded by Tanfloc, most papers have shown the use of
Moringa oleifera (MO) seeds, and their derivatives, to harvest specific microalgae species from culture media. Singh and Patidar [
11] examined the effects of MO extract dose, pH, mixing time and rate, settling time, temperature, and biomass concentration. With a dose of 15
mL.
L−1 extract, pH 8 and settling time of 20 minutes, the flocculation efficiency was 92.97%. In addition, turbidity was reduced from 388.16 NTU to 8.39 NTU, and there was a significant improvement in water quality, including the reduction of total nitrogen and total phosphorus by 40% and 21%, respectively.
This approach is essential for identifying optimal conditions to enhance microalgal cell and turbidity removal, thereby improving efficiency and cutting costs in large-scale applications. The results suggest that fine-tuning variables like dose, pH, and settling time can make MO a more feasible and eco-friendly approach for microalgae harvesting.
In recent years, bio-flocculation using other microorganisms has emerged as a viable method for harvesting microalgae. The bio-flocculation of microalgae with filamentous fungi from the genus
Aspergillus is the most extensively documented approach. By utilizing pellet-assisted harvesting, Civzele and Mezule [
14] reported that over 90% removal effi-ciency of Chlorella vulgaris was achieved within 24 hours due to bio-flocculation induced by
Aspergillus niger. Similarly, the concentration of Scenedesmus quadricauda decreased by more than 95% in 48 hours when treated with
Aspergillus fumigatus. Given the general non-toxicity of this method, its potential cost-effectiveness, and high harvesting efficiency, co-cultivating microalgae with filamentous fungi can be regarded as an efficient and optimal strategy for microalgae harvesting. However, despite the well-documented effectiveness of this technology, it faces challenges such as a lack of large-scale testing, relatively long flocculation times (24-48 h), limitations in reusing the biomass obtained after wastewater treatment, and the risk of potential contamination of wastewater with Aspergillus spores, which could lead to environmental leakage [
14].
In the same way, He et al. [
18] demonstrated high harvesting of Chlorella pyrenoidosa using
Citrobacter W4 bacteria, with an optimized bacterial-algal ratio of 4:1, a flocculation velocity gradient of 26.30
s−1, and a processing time of 6 hours, reaching an efficiency of 87.37% and nearing a theoretical maximum of 93.45%. Both studies emphasize the potential of bio-flocculation as a cost-effective, environmentally friendly method for urban wastewater treatment.
Another technique which offers advantages such as low capital and operational costs, minimal energy consumption, and non-disruptive handling of the cells is the sedimentation, one of the most straightforward harvesting methods [
14]. As a gravity-driven solid/liquid separation process, this technique serves a dual purpose: producing large volumes of clari-fied effluent while concentrating microalgal biomass in smaller volumes. This separation is technically challenging due to the small cell size of microalgae (typically 5-20
µm) and their negatively charged cell surfaces, which result in low terminal settling velocities, around 1
cm.
h−1 [
9].
Ortiz et al. [
9] investigated sedimentation coupled with coagulation and flocculation in an inclined plate system, applied in a demonstration-scale unit to treat biomass from pho-tobioreactors. The method used aluminum-based coagulants, such as PAX-18, to form flocs and increase sedimentation efficiency. The technique achieved biomass concentrations be-tween 5 and 20
g.
L−1 and a remarkable clarification efficiency, with effluent turbidity below 8 NTU and suspended solids below 26
mg.
L−1. The scalability of the system was evidenced by its low energy consumption and ability to treat large volumes of effluents. However, the use of chemical coagulants required care regarding the quality of the harvested biomass, especially for applications requiring high purity.
Alternatively, pH-induced sedimentation has gained interest at high-pH environments for microalgae separation. By adding a base such as sodium hydroxide, the pH level rises, creating conditions that facilitate the coprecipitation of inorganic salts promoting sedimentation [
17].
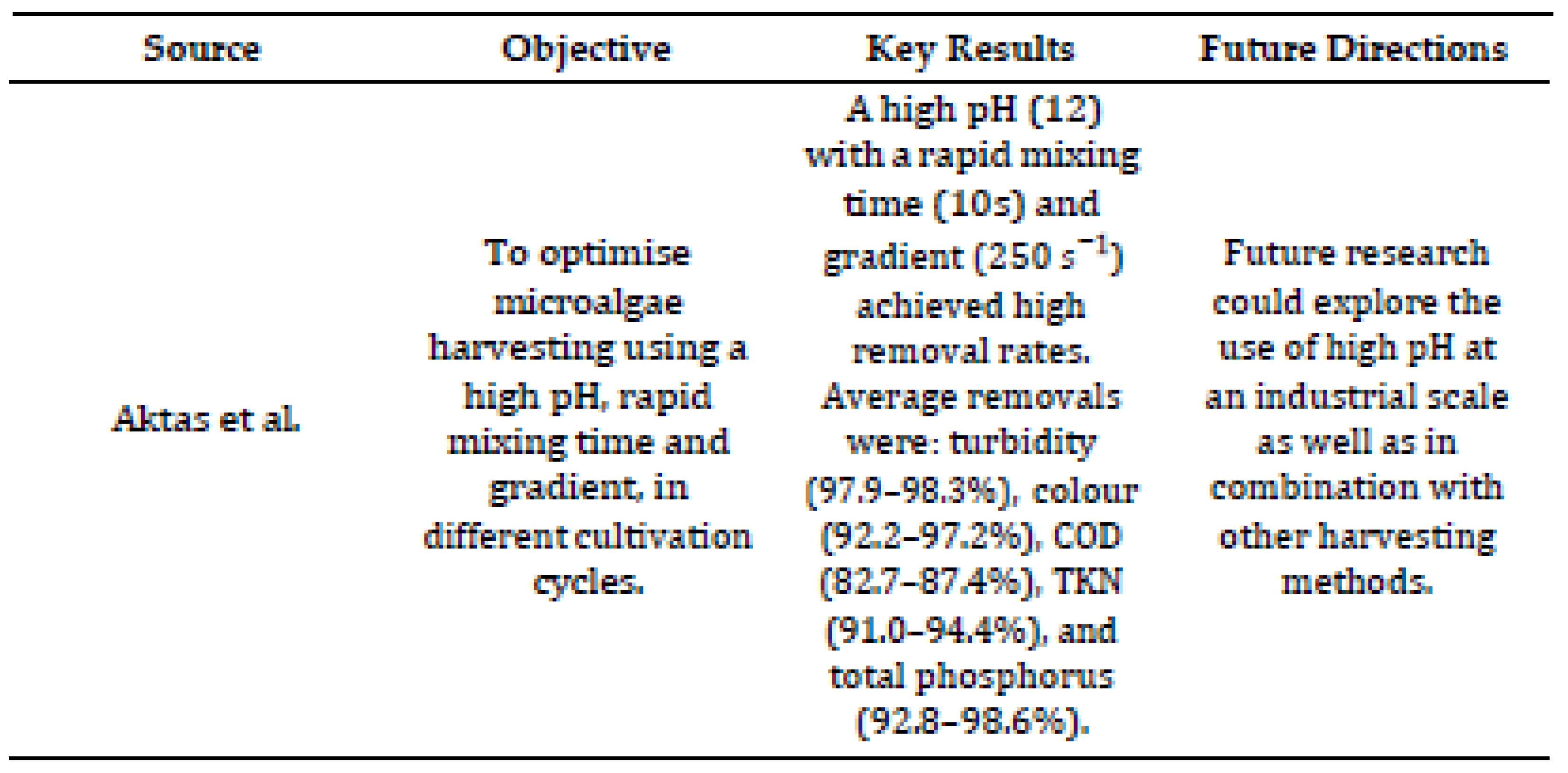
De Souza Leite and Daniel [
7] examined the harvesting of microalgae, specifically Chlorella sorokiniana, using pH-induced sedimentation in wastewater effluents. The microalgae were cultivated in a treated wastewater medium from an up-flow anaerobic sludge blanket (UASB) reactor, using a mixture of effluents from a pig farm and a sewage treatment plant. Under optimized conditions (pH 12, velocity gradient of 250
s−1 and mixing time of 10 seconds), the method achieved a harvesting efficiency of over 97.8%. In addition, the technique significantly improved effluent quality, removing up to 98.6% of phosphorus and 94.4% of nitrogen. The harvested biomass also showed high concentration, increasing up to 123 times when combined with centrifugation. Although efficient, the method requires significant volumes of NaOH for pH adjustment, which can increase operating costs depending on the scale.
These studies highlight that both sedimentation techniques are highly efficient and can be adapted to different contexts. While sedimentation coupled with coagulation and flocculation has proven to be more scalable and simpler for large volumes, pH-induced sedimentation has proven to be a cost-effective solution to improve effluent quality and recover high-purity biomass.
Another process used to harvest is filtration responsible to eliminate the total depen-dence on gravity or kinetic factors, capturing flocs that might escape in a purely gravi-tational process [
15]. In recent years, microalgae harvesting processes have increasingly focused on integrating filtration membranes into photobioreactors (PBRs). This configura tion allows in situ biomass harvesting without interrupting microalgae growth or nutrient flow within the system. The integration of filtration within PBRs facilitates the simultane-ous removal of nutrients, such as nitrogen and phosphorus, while recovering microalgae. This has been demonstrated in studies by Ermis et al. [
12], Larrode-Larretche and Jin [
13], Zhang et al.[
15] and Moglie et al. [
8] who investigated the performance of different module configurations — side-stream and submerged —, and types of membranes integrated with a microalgae PBRs for nutrients removal and microalgae harvesting.
The three studies analyzed presented distinct approaches for the harvesting of microal-gae using filtration techniques, allowing a quantitative comparison of the results. Moglie et al. [
8] investigated the use of microfiltration and ultrafiltration in the treatment of wastewa-ter from olive mills for the cultivation of
Arthrospira platensis. The study demonstrated a reduction of approximately 70% in production costs, standing out as an economical and sustainable solution. However, the presence of phenolic compounds in the treated waters negatively impacted the growth rate of the microalgae, although the quality of the biomass obtained remained satisfactory.
Membrane technologies, such as forward osmosis (FO) and dead-end filtration, show promise for microalgae harvesting. However, challenges related to membrane fouling and the need for process optimisation remain significant hurdles [
12,
13]. The impact of membrane configuration, such as side-stream versus submerged, is also a crucial factor in performance. Different membrane types, such as microfiltration (MF) and ultrafiltration (UF), offer varying degrees of separation and have different operational parameters such as backwashing that can affect membrane fouling and energy consumption.
Zhang et al. [
15] analyzed a combined flocculation and microfiltration process using
Chlorella sorokiniana. The technique achieved a harvest efficiency of 95.2% and an average permeation flux of 55.5
m−3.
m−2.
h−1 under optimized conditions, using 5
mg.
L−1 cationic polyacrylamide (CPAM) and membranes with 25
µm pores.
Natural coagulants, particularly
Moringa oleifera and tannin-based substances, have emerged as effective and environmentally conscious alternatives to chemical coagulants [
11,
16]. These coagulants often present a lower risk of metal contamination compared to their chemical counterparts, but their performance depends on optimising their application conditions, such as dosage, pH, and mixing times [
12]. Variability in composition and performance based on source and processing method is a key challenge that needs to be taken into consideration.
Larronde-Larretche and Jim [
13] explored the Forward Osmosis (FO) technique in two configurations, submerged and side-stream, integrated with photobioreactors for the cultivation of
Chlorella vulgaris. The submerged configuration stood out for its high efficiency in nutrient removal, achieving 92.9% removal of phosphorus ((
PO4)3), 100% of ammonia ((
NH3)
− N) and 98.7% of total nitrogen (TN). In addition, the biomass concentration reached 0.965 g L-1 after seven days of operation, due to in situ dewatering. However, the system showed salt and biomass accumulation in the reactor, which may affect long-term efficiency. The side-stream configuration, in turn, achieved slightly lower nutrient removal efficiencies (82% phosphorus, 96% ammonia and 94.8% total nitrogen), but demonstrated better biofouling control and ease of cleaning, making it more suitable for continuous operations.
Ermis et al. [
12] compared cross-flow and submerged membrane systems for the harvesting of Chlorella vulgaris. The submerged system showed greater efficiency, reaching a biomass concentration of 10.44
g.
L−1 , in contrast to the 6.99
g.
L−1 obtained in the cross-flow system. Both systems used anaerobic digestate as a culture medium, demonstrating large-scale viability, but faced challenges related to membrane clogging. The submerged system was more efficient in biomass concentration, while the cross-flow system showed greater operational robustness.
The studies indicate that the choice of filtration configuration in PBRs depends on bal-ancing harvesting efficiency, maintenance ease, and operational costs. Microfiltration and ultrafiltration were notable for their cost-effectiveness but encountered challenges related to chemical interference. Submerged configurations demonstrated superior efficiency in harvesting and nutrient removal, whereas lateral flow designs offered greater operational reliability, reduced biofouling, and simplified cleaning procedures.
Another promising method for microalgae harvesting is flotation, which is widely recognized for providing high biomass harvesting in short retention times. Among various flotation techniques, dissolved air flotation (DAF) has been shown to be particularly effec-tive. The DAF process works by generating air bubbles that help microalgae flocs rise to the surface of the system, where they accumulate and can be removed, leading to water clarification [
19].
Leite, dos Santos and Daniel [
19], explored the use of dissolved air flotation combined with pH modulation as a harvesting method for
Chlorella sorokiniana cultured in wastewater.
The technique achieved a microalgae harvesting efficiency of 96.5% to 97.9% and nutrient (phosphorus and nitrogen) removal of 91.8% to 98.3%, optimizing parameters such as pH (12), mixing time (30 seconds), and recirculation rate (20%). In addition, the quality of the treated wastewater was significantly improved, demonstrating the feasibility of the method for large-scale applications.
Qi et al. [
20], on the other hand, analyzed electrolytic flotation without coagulation for microalgae species with different degrees of hydrophobicity, a method that generates hydrogen and oxygen bubbles through the electrolysis of water, using inert electrodes.
These bubbles facilitate cell-bubble adhesion, especially in microalgae with high hydropho-bicity, eliminating the need for chemical coagulants. In the study, for Tribonema sp., which is highly hydrophobic, the technique achieved a harvest efficiency of 96.2% with an en ergy consumption of 0.19 kW.h.kg−1 of biomass, making it an efficient and cost-effective approach. In comparison, less hydrophobic microalgae, such as Scenedesmus sp. (55.6%) and Pandorina sp. (42.8%), showed lower harvest efficiency, highlighting the influence of hydrophobicity on cell-bubble adhesion during flotation.
Qi et al. [
10] introduced the concept of autoflotation in a photobioreactor, using
Tribonema sp. as the dominant species. In this method, self-floating microalgae, such as
Tribonema sp., generate oxygen microbubbles through photosynthesis, which adhere to the cells, promoting their natural floating to the surface, without the need for flocculants or external gas supply. The technique achieved a harvest efficiency above 90% in a separation time of 30 minutes. Furthermore, the biomass showed excellent dehydration capacity, with specific filtration resistance (SRF) values of 2.7 × 1011
m.
kg−1 , and nutrient removal efficiency above 98%.
These studies indicate that flotation is a promising technique for the harvesting of microalgae, with the potential to be adapted to different conditions and species. While DAF demonstrates high removal efficiency and improvement of wastewater quality, electrolytic flotation and autoflotation stand out for their energy efficiency and operational simplicity. It is noted that several technologies have been applied for microalgae harvesting, including physical, chemical and biological methods. In some cases, two or more methods combined to obtain the maximum biomass yield. The choice of the most appropriate technique depends on the characteristics of the microalgae, the culture medium and the process objectives, whether they are cost reduction, sustainability or maximization of recovered biomass.