Submitted:
11 March 2026
Posted:
12 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
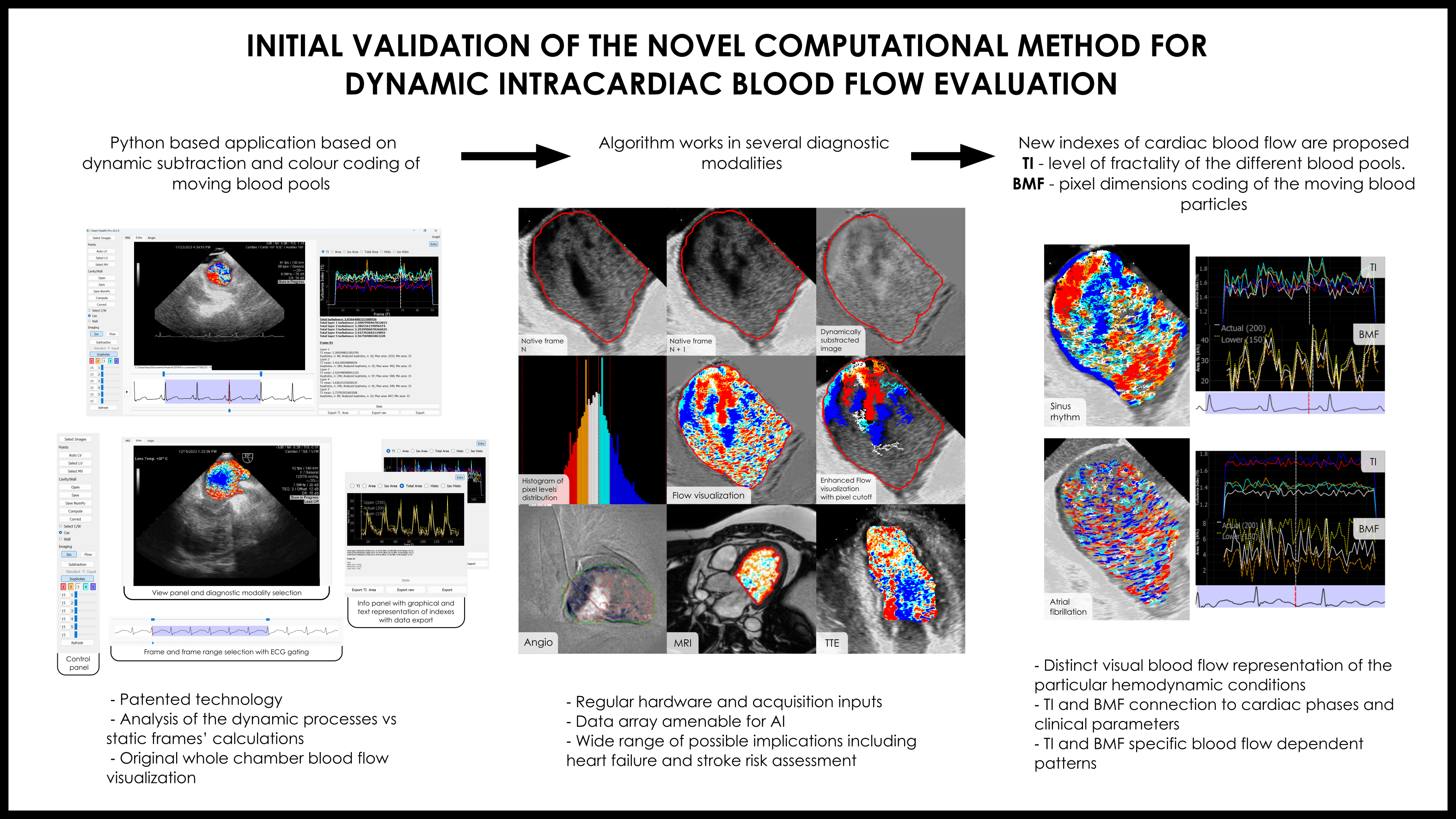
2. Materials and Methods
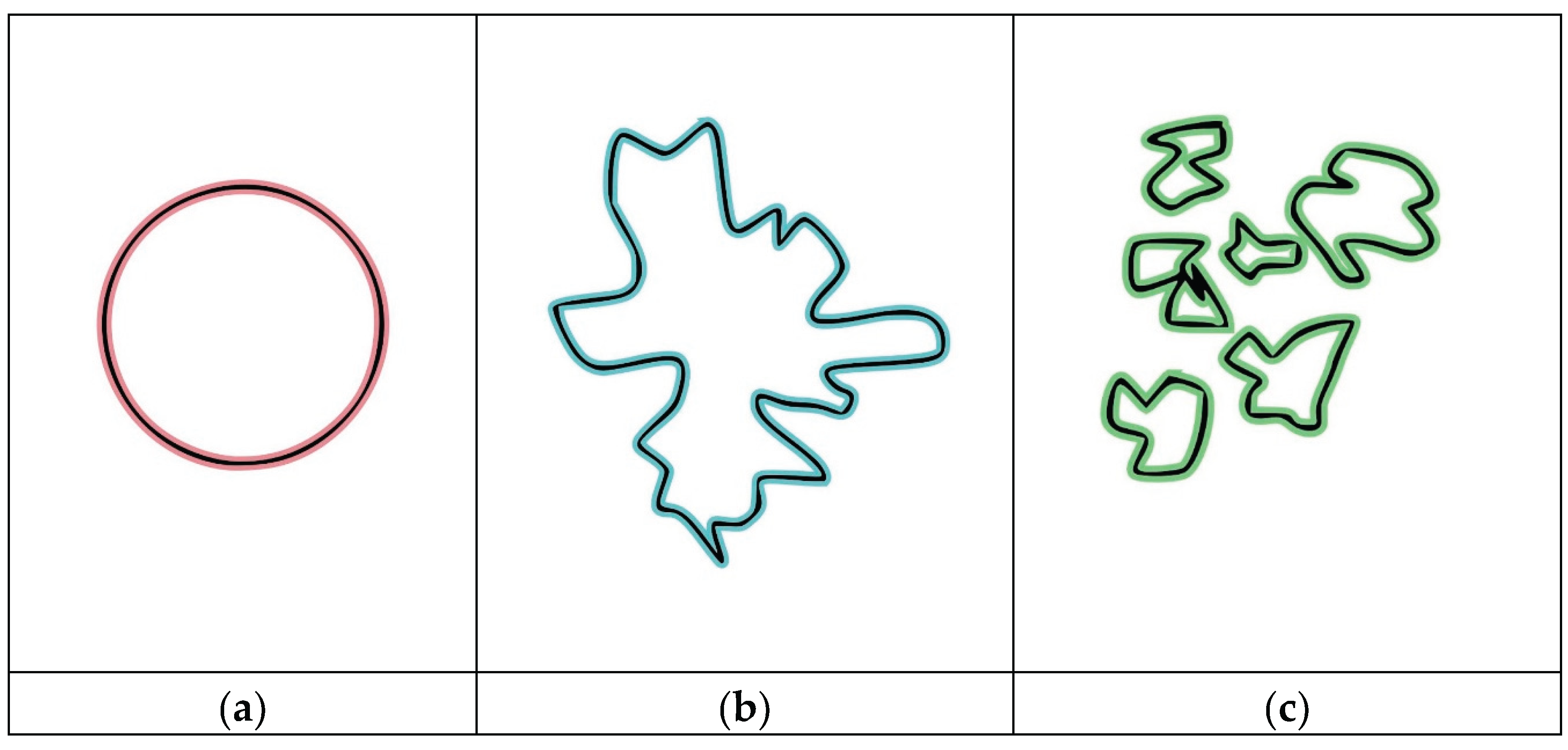
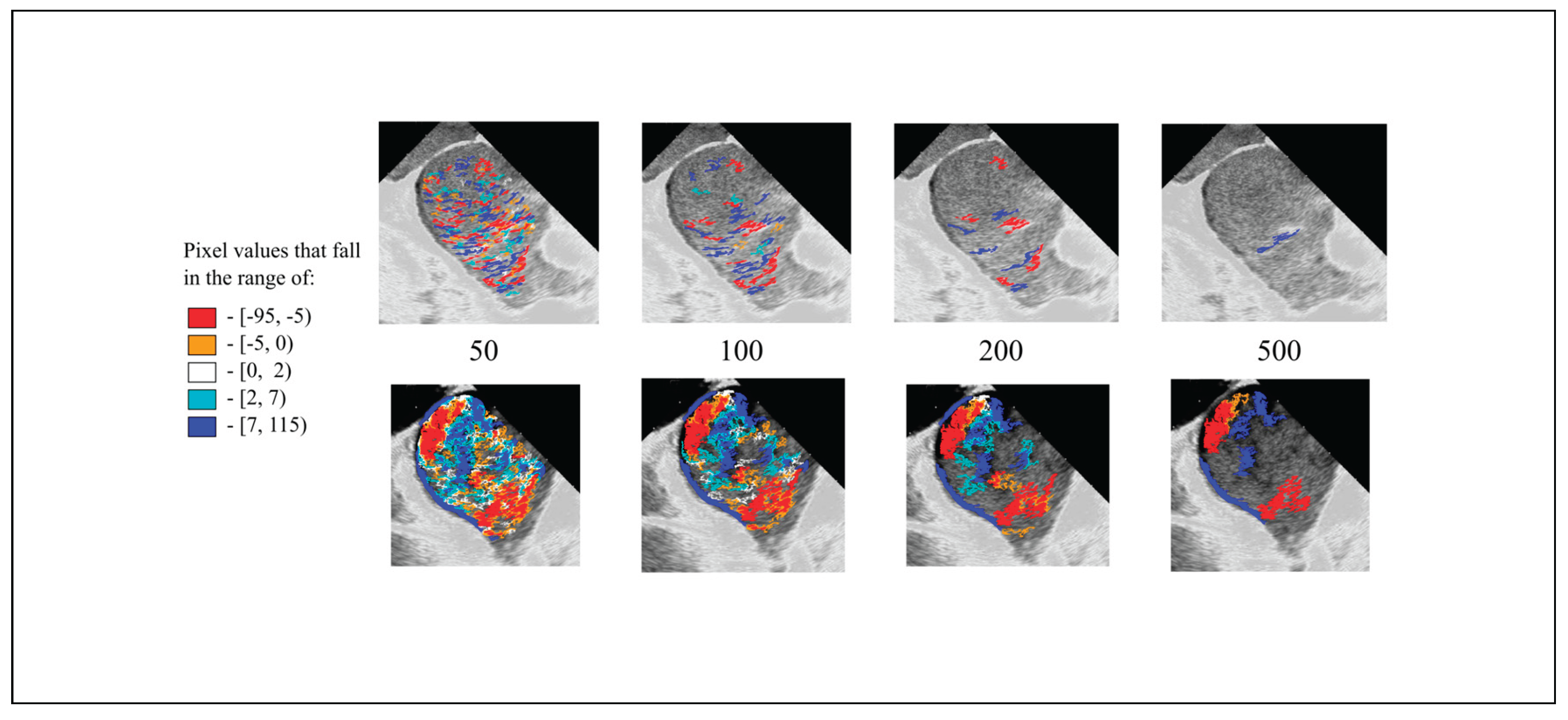
- Turbulence index (TI) - fractality level of the different blood pools;
- Blood mobility fraction (BMF) - pixel dimensions coding of the moving blood particles.
3. Results
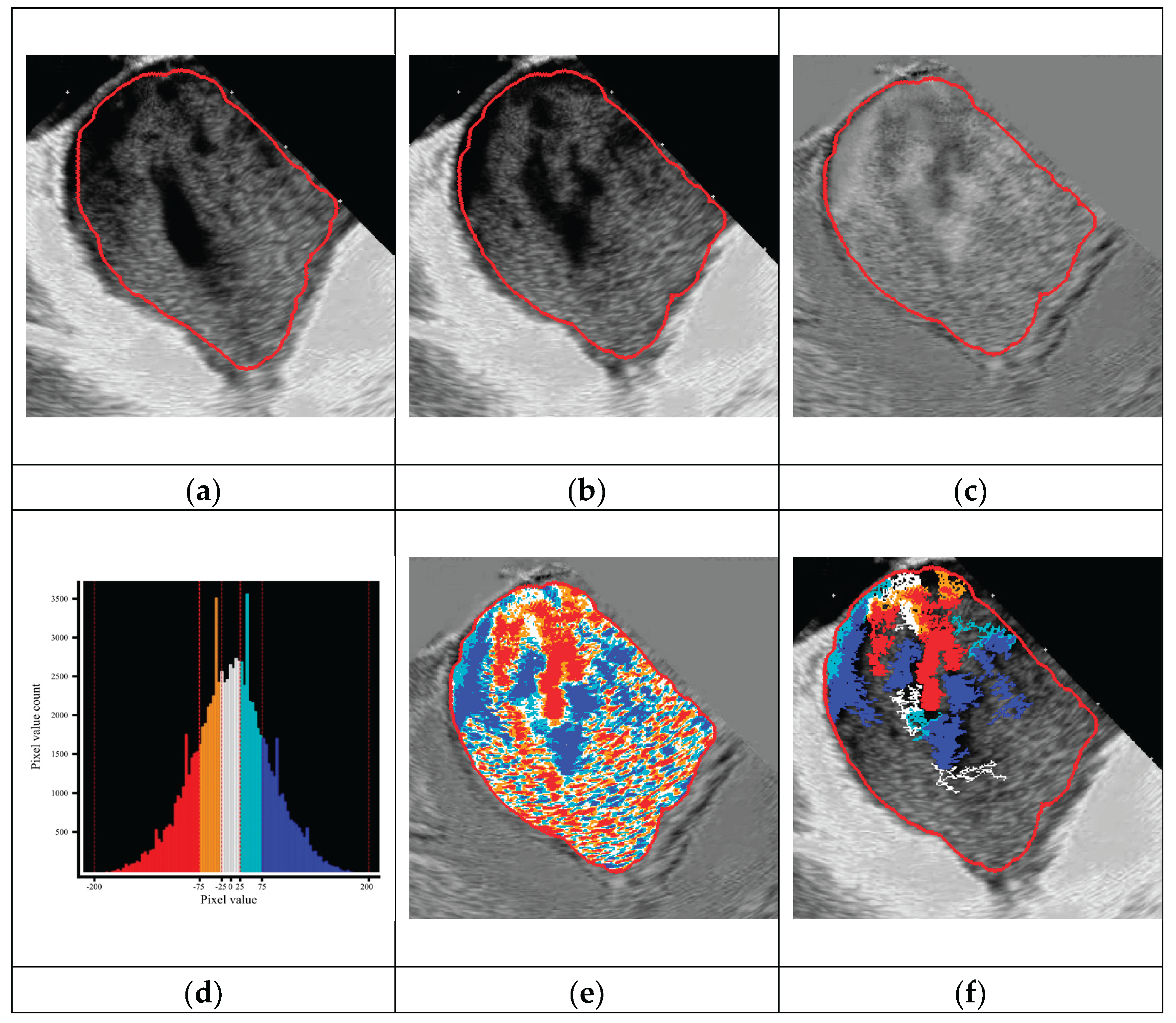
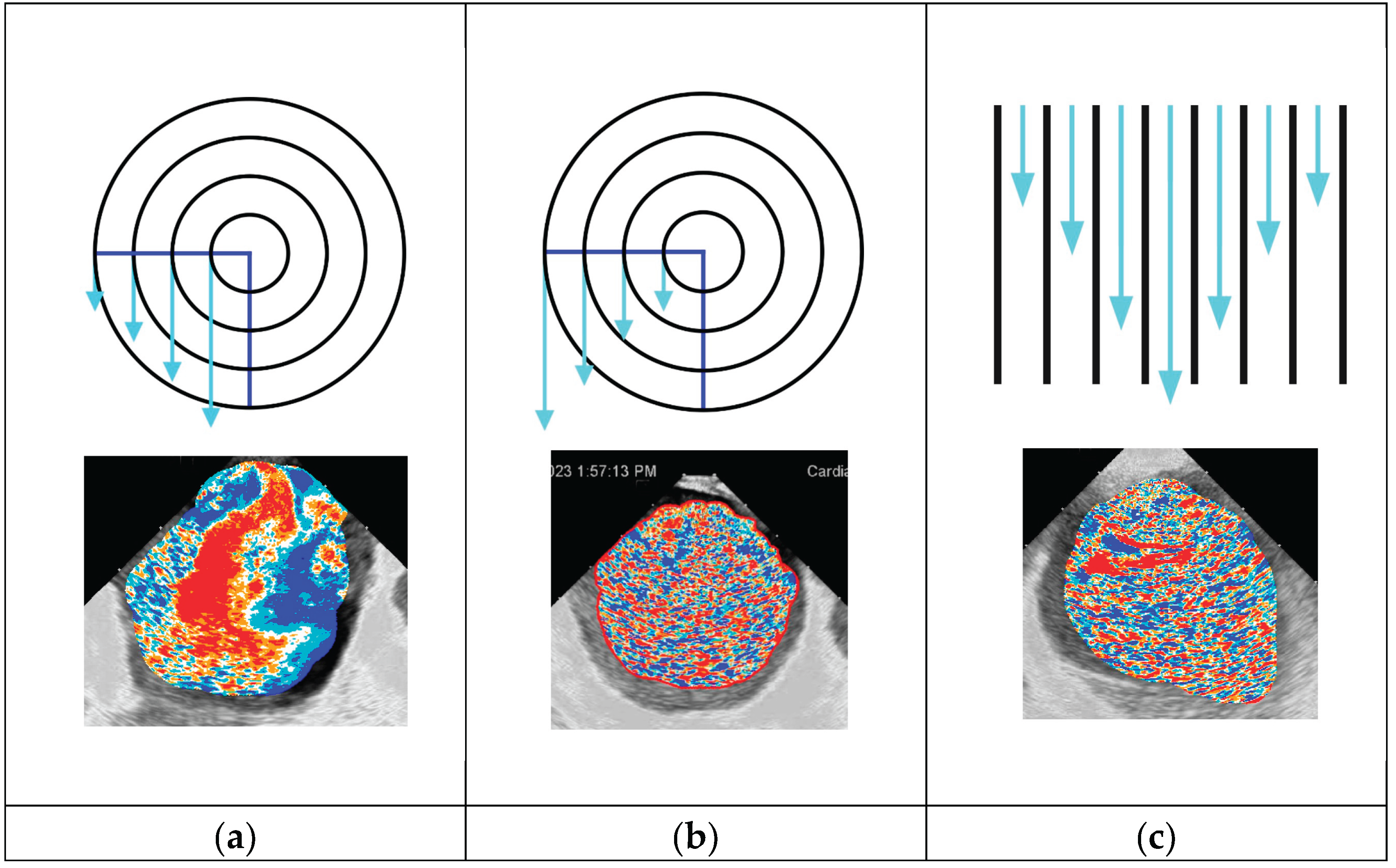
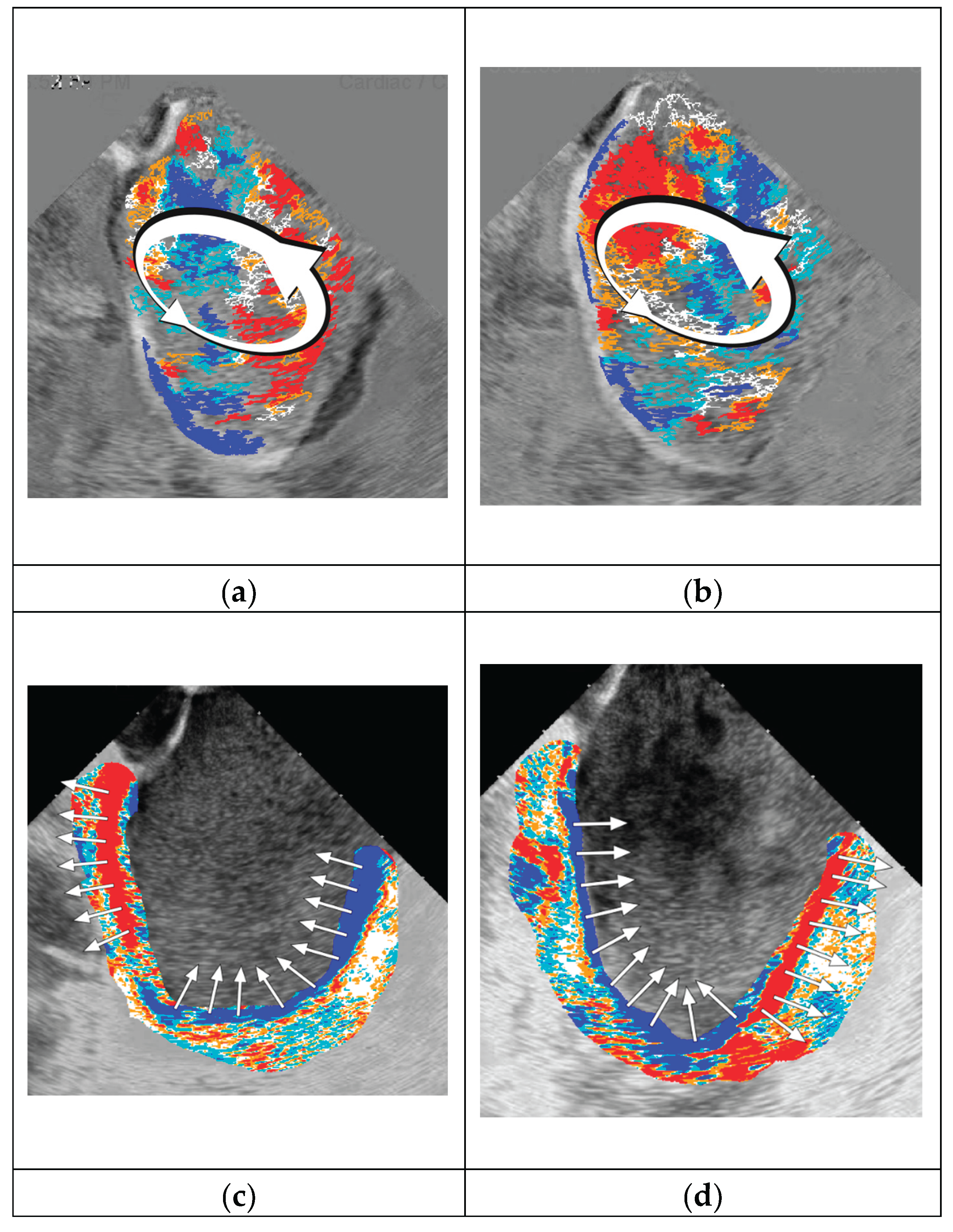
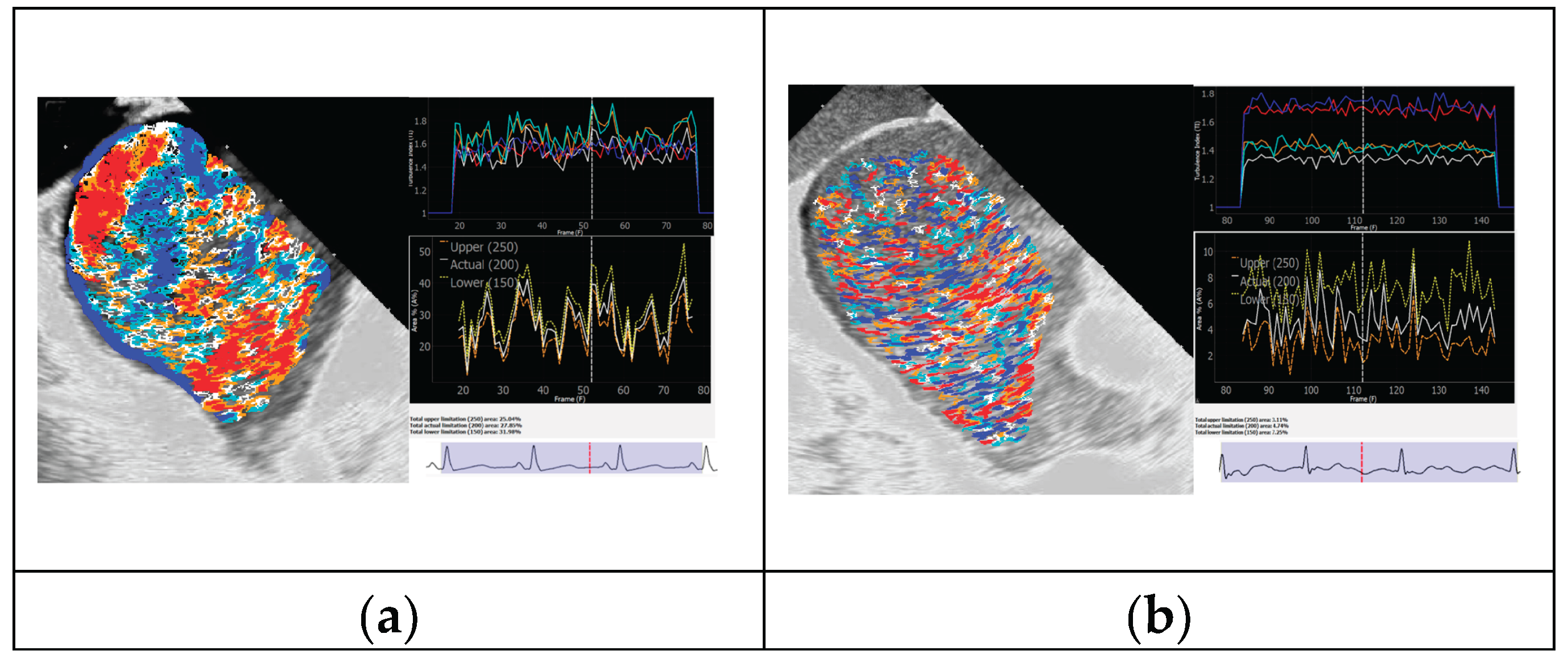
3.1. Visual Representations of the Flow Patterns
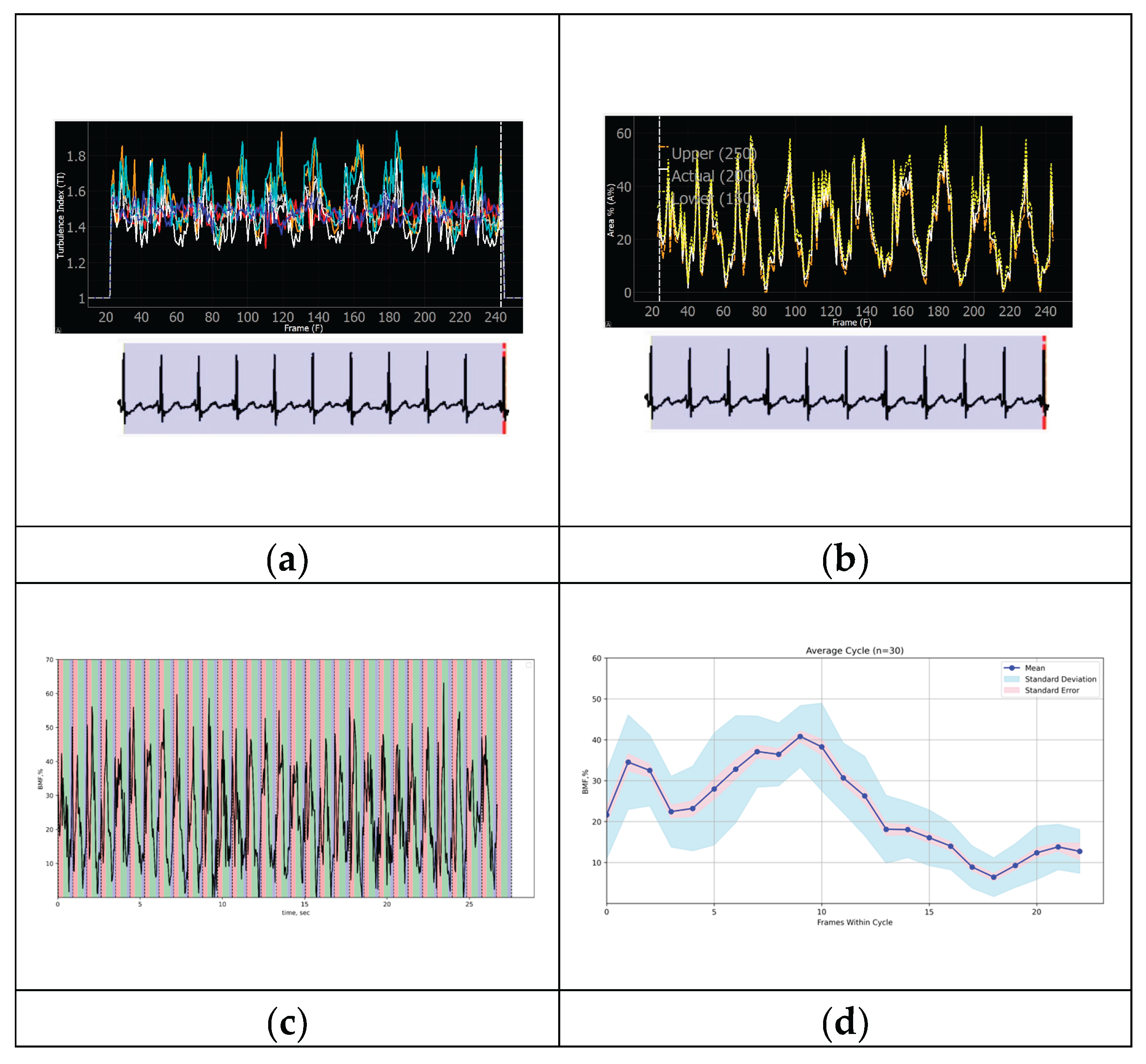
3.2. Program-Derived Data Repetitiveness and Correlation with Cardiac Cycle Phases and Other Parameters
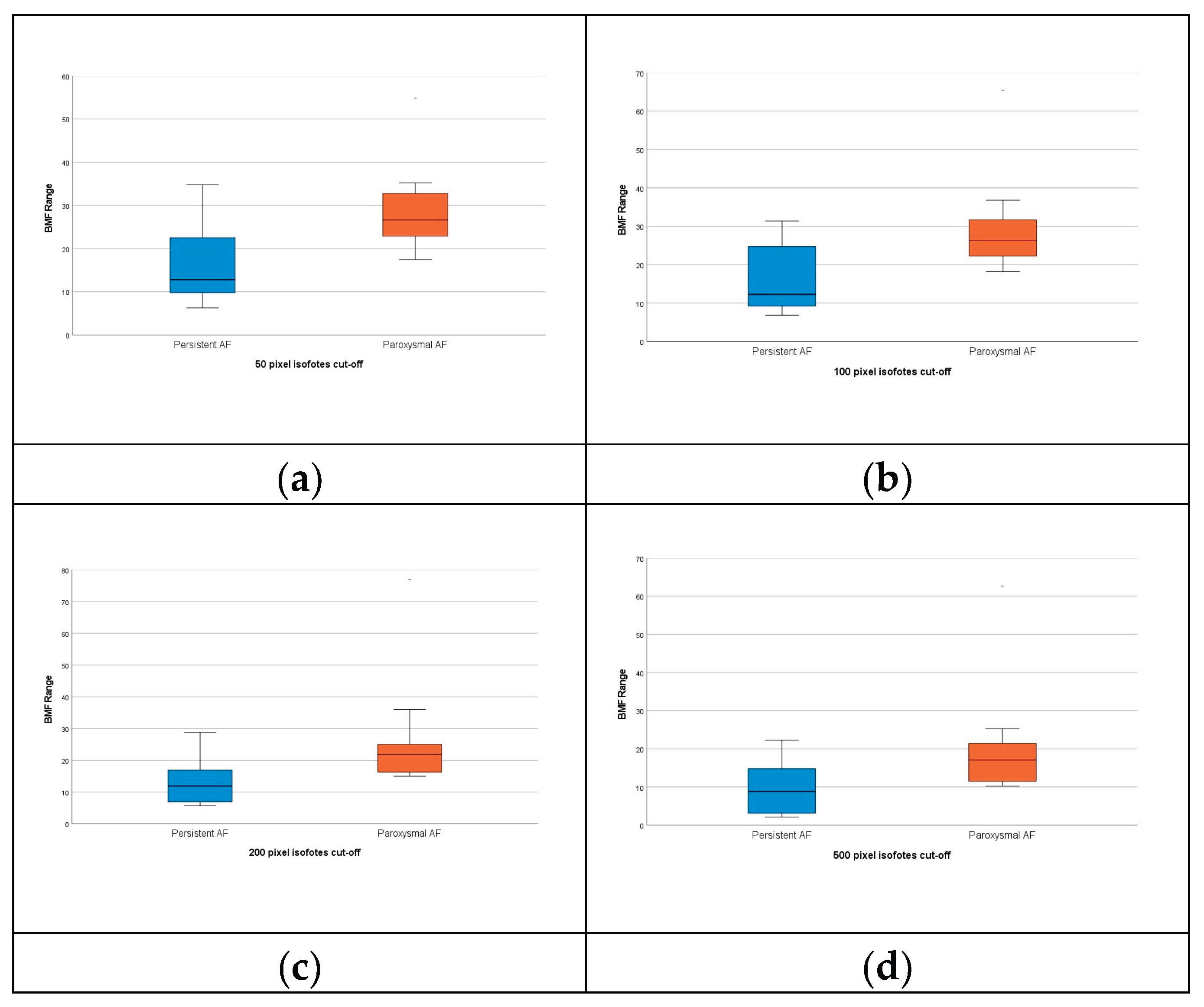
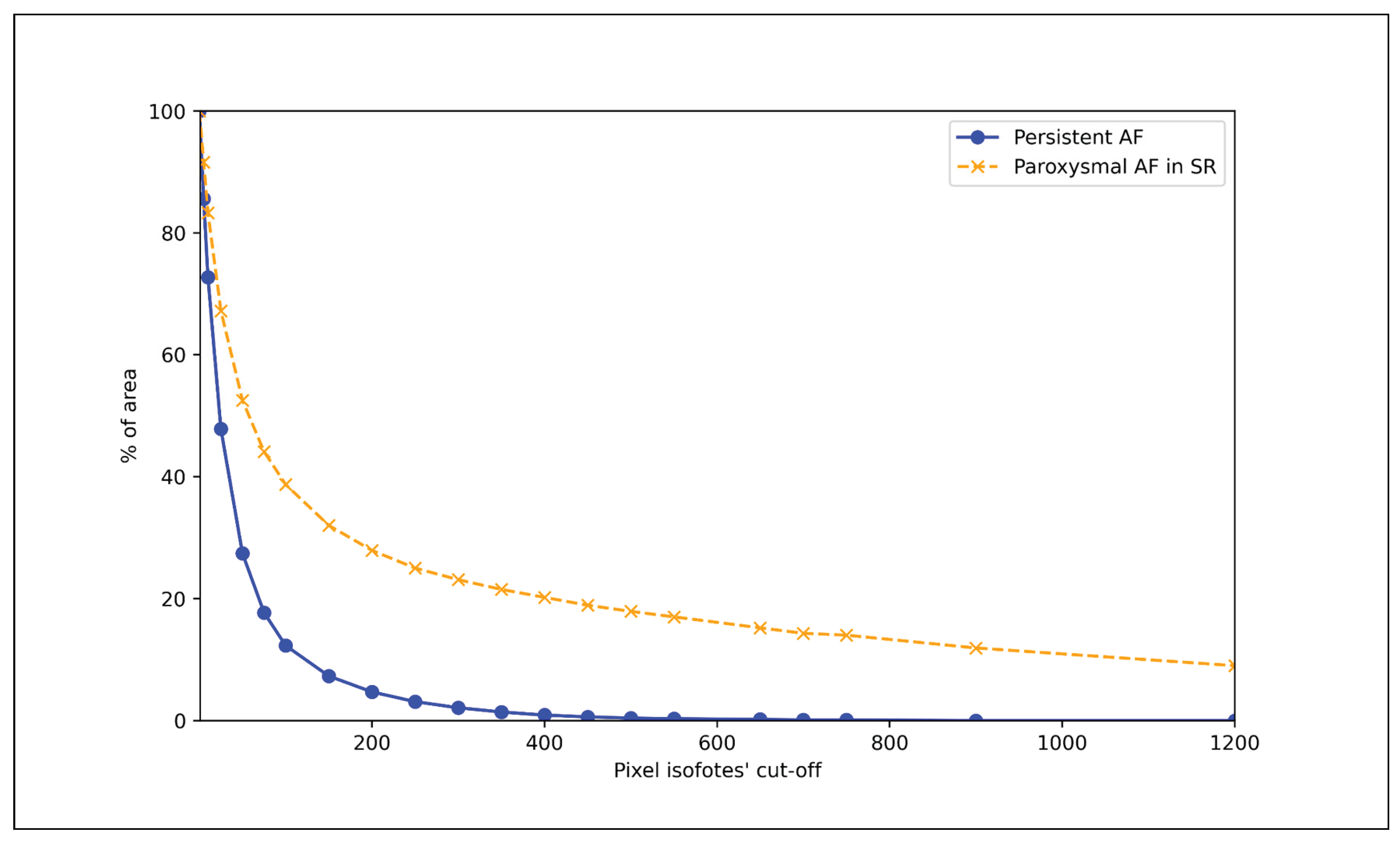
3.3. Comparison of the Blood Flow Patterns Among Study Patients’ Groups
4. Discussion
5. Conclusions
- A Python-based computer program was created to specifically analyze the cyclic intracardiac blood flow phenomena with the focus on the visual surrogates of the turbulence digital calculations.
- The program creates enhanced dynamic color-coded whole chamber visualization of intracardiac blood flow using regular raw radiological data inputs from conventional hardware via different imaging modalities.
- The newly proposed TI and BMF were preliminary validated on a small dataset of ICE images as special dynamic indices of blood flow evaluation and had recognizable patterns in selected SR and AF patients.
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AF | Atrial fibrillation |
| AI | Artificial intelligence |
| BMF (n) | Blood mobility fraction with n-pixel cutoff |
| CW | Clockwise |
| CCW | Counterclockwise |
| ECG | Electrocardiogram |
| ICE | Intracardiac echocardiography |
| LA | Left atrium, left atrial |
| LAA | Left atrial appendage |
| LV | Left ventricle, left ventricular |
| MR | Mitral regurgitation |
| MRI | Magnetic resonance imaging |
| PAF | Paroxysmal AF |
| PersAF | Persistent AF |
| PV(s) | Pulmonary vein(s) |
| RA | Right atrium, right atrial |
| ROI | Region of interest |
| TI | Turbulence index |
| TTE | Transthoracic echocardiography |
| TEE | Transesophageal echocardiography |
| TR | Tricuspid regurgitation |
References
- January CT, Wann LS, Calkins H, Chen LY, Cigarroa JE, Cleveland JC Jr, Ellinor PT, Ezekowitz MD, Field ME, Furie KL, Heidenreich PA, Murray KT, Shea JB, Tracy CM, Yancy CW. 2019 AHA/ACC/HRS Focused Update of the 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society in Collaboration With the Society of Thoracic Surgeons. Circulation. 2019;140(2):e125-e151. [CrossRef]
- E, Lip GY, Galderisi M, Goette A, Shah D, Marwan M, Lederlin M, Mondillo S, Edvardsen T, Sitges M, Grapsa J, Garbi M, Senior R, Gimelli A, Potpara TS, Van Gelder IC, Gorenek B, Mabo P, Lancellotti P, Kuck KH, Popescu BA, Hindricks G, Habib G, Cardim NM, Cosyns B, Delgado V, Haugaa KH, Muraru D, Nieman K, Boriani G, Cohen A. EACVI/EHRA Expert Consensus Document on the role of multi-modality imaging for the evaluation of patients with atrial fibrillation. Eur Heart J Cardiovasc Imaging. 2016;17(4):355-83. [CrossRef]
- Bosi GM, Cook A, Rai R, Menezes LJ, Schievano S, Torii R, Burriesci G Burriesci. Computational Fluid Dynamic Analysis of the Left Atrial Appendage to Predict Thrombosis Risk. Front Cardiovasc Med. 2018;5:34. [CrossRef]
- Spartera M, Pessoa-Amorim G, Stracquadanio A, Von Ende A, Fletcher A, Manley Pet al. Left atrial 4D flow cardiovascular magnetic resonance: a reproducibility study in sinus rhythm and atrial fibrillation. J Cardiovasc Magn Reason. 2021;23:29. [CrossRef]
- Klabunde RE. Cardiovascular physiology concepts. PA: LWW, 3d edition, 2021, 272p.
- K. Kundu, Ira M. Cohen and David R. Dowling. Fluid Mechanics. 6th Edition. Elsevier Inc. 2016. 784p.
- Kaufmann TAS, Graefe R, Hormes M, Schmitz-Rode T, Steinseiferand U. Computational Fluid Dynamics: Theory, Analysis and Applications. P. 109–136.
- Baillargeon B, Rebelo N, Fox DD., Taylor RL, Kuhl E. The Living Heart Project: A robust and integrative simulator for human heart function. European Journal of Mechanics - A/Solids. 2014;48:38-47. [CrossRef]
- Qin JJ, Indja B, Gholipour A, Gök M, Grieve SM. Evaluation of Left Ventricular Function Using Four-Dimensional Flow Cardiovascular Magnetic Resonance: A Systematic Review. J Cardiovasc Dev Dis. 2022;9(9):304. [CrossRef]
- Dyverfeldt P, Bissell M, Barker AJ, Bolger AF, Carlhäll CJ, Ebbers T, Francios CJ, Frydrychowicz A, Geiger J, Giese D, Hope MD, Kilner PJ, Kozerke S, Myerson S, Neubauer S, Wieben O, Markl M. 4D flow cardiovascular magnetic resonance consensus statement. J Cardiovasc Magn Reson. 2015;17(1):72. [CrossRef]
- Fiorencis A, Pepe M, Smarrazzo V, Martini M, Severino S, Pergola V, Evangelista M, Incarnato P, Previtero M, Maglione M, Iliceto S, Pedrizzetti G, Mele D. Noninvasive Evaluation of Intraventricular Flow Dynamics by the HyperDoppler Technique: First Application to Normal Subjects, Athletes, and Patients with Heart Failure. J Clin Med. 2022;11(8):2216. [CrossRef]
- GR, Kim M, Pedrizzetti G, Vannan MA. Current clinical application of intracardiac flow analysis using echocardiography. J Cardiovasc Ultrasound. 2013;21(4):155-62.
- D, Beccari R, Pedrizzetti G. Effect of Aging on Intraventricular Kinetic Energy and Energy Dissipation. J Cardiovasc Dev Dis. 2023;10(7):308.
- J, Eriksson J, Dyverfeldt P, Bolger AF, Ebbers T, Carlhäll CJ. Turbulent kinetic energy in normal and myopathic left ventricles. J Magn Reson Imaging. 2015;41(4):1021-9. [CrossRef]
- Volkov D, Batsak B, Skoryi D. Blood flow turbulence index as a new parameter for cardiac function assessment in various cardiac imaging modalities. European Journal Of Heart Failure. 2022;24:84-85.
- Bäck S, Skoda I, Lantz J, Henriksson L, Karlsson LO, Persson A, Carlhäll CJ, Ebbers T. Elevated atrial blood stasis in paroxysmal atrial fibrillation during sinus rhythm: a patient-specific computational fluid dynamics study. Front Cardiovasc Med. 2023;10:1219021. [CrossRef]
- D, Trevisan F, Fiorencis A, Smarrazzo V, Bertini M, Ferrari R. Current Role of Echocardiography in Cardiac Resynchronization Therapy: from Cardiac Mechanics to Flow Dynamics Analysis. Curr Heart Fail Rep. 2020;17(6):384-396. [CrossRef]
- C, Jin Z, Liu R, Iwata S, Tugcu A, Yoshita Met al. LA volumes and reservoir function are associated with subclinical cerebrovascular disease: the CABL (Cardiovascular Abnormalities and Brain Lesions) study. JACC Cardiovasc Imaging 2013;6:313–23.
- Fyrenius A, Wigström L, Ebbers T, Karlsson M, Engvall J, BolgerAF. Three dimensional flow in the human left atrium. Heart 2001;86:448-55. [CrossRef]
- Park KH, Son JW, Park WJ, Lee SH, Kim U, Park JS, Shin DG, Kim YJ, Choi JH, Houle H, Vannan MA, Hong GR. Characterization of the left atrial vortex flow by two-dimensional transesophageal contrast echocardiography using particle image velocimetry. Ultrasound MedBiol 2013;39:62-71. [CrossRef]
- J, Gupta V, Henriksson L, Karlsson M, Persson A, Carlhäll CJ, Ebbers T. Impact of Pulmonary Venous Inflow on Cardiac Flow Simulations: Comparison with In Vivo 4D Flow MRI. Ann Biomed Eng. 2019;47(2):413-424. [CrossRef]
- Jingquan Z, Deyong L, Huimin C, Hua F, Xuebin H, Chenyang J, Yan L, Xuebin L, Min T, Zulu W, Yumei X, Jinlin Z, Wei Z, Xiaochun Z, Daxin Z, Yun Z, Changsheng M, Zei PC, Di Biase L. Intracardiac echocardiography Chinese expert consensus. Front Cardiovasc Med. 2022;9:1012731. [CrossRef]
- US 10,631,811 B2, United States Patent; А61В6/032, А61В8/06, А61В8/0883 Method and system for processing of medical images for generating of prognosis of cardiac function. Dmytro Volkov, Valerij Boyko, Alexander Bakai; appl. No 15/060,970; filed 04.03.2016; issued 28.04.2020.
- Pedrizzetti G, La Canna G, Alfieri O. et al. The vortex - an early predictor of cardiovascularoutcome? Nat Rev Cardiol. 2014;11:545–553. [CrossRef]
- Spartera M, Stracquadanio A, Pessoa-Amorim G, Von Ende A, Fletcher A, Manley P, Ferreira VM, Hess AT, Hopewell JC, Neubauer S, Wijesurendra RS, Casadei B. The impact of atrial fibrillation and stroke risk factors on left atrial blood flow characteristics. Eur Heart J Cardiovasc Imaging. 2021;23(1):115-123. [CrossRef]
- Cimino S, G. Pedrizzetti, G. Tonti, E. Canali, V. Petronilli, L. De Luca, C. Iacoboni, L. Agati. In vivo analysis of intraventricular fluid dynamics in healthy hearts. European Journal of Mechanics - B/Fluids. 2012;35:40-46. [CrossRef]
- Fadnes S, Wigen MS, Nyrnes SA, Lovstakken L. In Vivo Intracardiac Vector Flow Imaging Using Phased Array Transducers for Pediatric Cardiology. IEEE Transactions On Ultrasonics, Ferroelectrics, And Frequency Control. 2017;64(9):1318-26. [CrossRef]
- Sun Y, Ling Y, Chen Z, Wang Z, Li T, Tong Q, Qian Y. Finding low CHA2DS2-VASc scores unreliable? Why not give morphological and hemodynamic methods a try? Front Cardiovasc Med. 2023 Jan 4;9:1032736.
- Nakagami H, Yamamoto K, Ikeda U, Mitsuhashi T, Goto T, Shimada K. Mitral regurgitation reduces the risk of stroke in patients with nonrheumatic atrial fibrillation. Am Heart J. 1998;136:528-32. [CrossRef]
- Melduni R, Nkomo VT, Wysokinski W, Gersh BJ, Deshmukh A, Padang R, Greene EL, Oh JK, Lee HC. Risk of left atrial appendage thrombus and stroke in patients with atrial fibrillation and mitral regurgitation. Heart. 2022;108(1):29-36. [CrossRef]
- Van Laer SL, Verreyen S, Winkler KM, Miljoen H, Sarkozy A, Heuten H, Saenen J, Van Herck P, Van de Heyning CM, Heidbuchel H, Claeys MJ. Effect of Mitral Regurgitation on Thrombotic Risk in Patients With Nonrheumatic Atrial Fibrillation: A New CHA2DS2-VASc Score Risk Modifier? Am J Cardiol. 2021;145:69-76. [CrossRef]
- Spartera M, Stracquadanio A, Pessoa-Amorim G, Von Ende A, Fletcher A, Manley P, Ferreira VM, Hess AT, Hopewell JC, Neubauer S, Wijesurendra RS, Casadei B. The impact of atrial fibrillation and stroke risk factors on left atrial blood flow characteristics. European Heart Journal - Cardiovascular Imaging. 2022;23:115-123. [CrossRef]
| Patients, n=16 | Paroxysmal AF, n=8 | Persistent AF, n=8 | p-value |
|---|---|---|---|
| Age | 59±19 | 74±8 | 0.0551 |
| BMI | 26.4 | 29.3 | 0.1856 |
| CHA2DS2-VASC score | 2.0 | 3.9 | 0.0205 |
| LVEF, % | 58±7 | 60±10 | 0.5971 |
| LA dimensions, mm | 40±3.8 | 43±3.6 | 0.0523 |
| Mitral max velocity, m/s | 0.66±0.25 | 0.89±0.36 | 0.0657 |
| Mitral deceleration, ms | 198±35 | 183±28 | 0.5619 |
| MR, grade | 1.1±0.4 | 1.4±0.7 | 0.1108 |
| Patients | Paroxysmal AF, n=8 | Persistent AF, n=8 | p-value |
|---|---|---|---|
| ECG with SR | |||
| HR, bpm | 62±15 | 70±17 | 0.5287 |
| P wave, ms | 106±15 | 104±17 | 0.7948 |
| PR, ms | 151±30 | 158±23 | 0.6383 |
| QRS, ms | 97±26 | 90±21 | 0.5287 |
| QT, ms | 412±56 | 397+45 | 0.9601 |
| Peak velocities (ICE), m/s | |||
| LAA | 0.48±0,13 | 0.3±0.11 | 0.0106 |
| Left PVs | 0.47±0,15 | 0.36±0.06 | 0.0929 |
| Right PVs | 0.31±0.16 | 0.4±0.18 | 0.0404 |
| Transmitral flow | 0.46±0.07 | 0.58±0.1 | 0.0209 |
| Mean LA pressure | 13±5 | 19±7 | 0.0930 |
| Patients | BMF Maximum value | BMF Minimum value | BMF Min-Max range | |||
|---|---|---|---|---|---|---|
| PesrAF | PAF | PesrAF | PAF | PersAF | PAF | |
| 1 | 15.4 | 41.8 | 2.8 | 5.8 | 12.6 | 36 |
| 2 | 17.9 | 16.3 | 1.28 | 0.5 | 16.6 | 15.8 |
| 3 | 8.4 | 30.4 | 2.7 | 2.4 | 5.7 | 28 |
| 4 | 19.6 | 21.9 | 2.4 | 0 | 17.2 | 21.9 |
| 5 | 14.2 | 78.6 | 3 | 1.6 | 11.2 | 77 |
| 6 | 9.1 | 15.9 | 2.2 | 0.9 | 6.9 | 15 |
| 7 | 8.7 | 18.7 | 1.8 | 1.9 | 7 | 16.8 |
| 8 | 29.3 | 25.1 | 0.5 | 3.1 | 28.8 | 22 |
| Avg | 15.3 | 31.1 | 2.1 | 2 | 13.3 | 29.1 |
| SD | 7.1 | 21 | 0.8 | 1.8 | 7.7 | 20.6 |
| p-value | 0.0238 | 0.6383 | 0.0315 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




