Submitted:
11 March 2026
Posted:
12 March 2026
You are already at the latest version
Abstract
Background/Objectives: Trigeminal neuralgia (TN) is a debilitating neurological condition characterized by recurrent, severe pain linked to peripheral and central sensitization within trigeminal pathways. Although current pharmacologic treatments are limited by inadequate efficacy or dose-limiting side effects, botulinum neurotoxin type A (BoNT/A) has emerged as a viable option. However, its potential use in the management of TN is hampered by methodological limitations in existing studies and a lack of pivotal clinical trials. This study investigated the efficacy, optimal treatment site, preventive utility, and duration of effect of incobotulinumtoxinA (Inco/A), a BoNT/A, in a model of TN. Methods: An infraorbital nerve chronic constriction injury model was used to induce mechanical allodynia in male Sprague–Dawley rats, reproducing the trigeminal sensitization seen in TN. The effects of subcutaneous Inco/A (1, 2, and 4U) were measured using the mechanical sensitivity (von Frey) test to evaluate the dose response, effect of injection location, potential preventive nature of treatment, and duration of benefit. Results: Inco/A produced a robust, dose-dependent reduction in mechanical allodynia, predominantly via a local mechanism of action. Both preventive and therapeutic administration of Inco/A was efficacious, with significant reduction of allodynia even when administered up to 28 days before nerve injury. The anti-allodynic effect persisted up to 56 days post-injection. Conclusions: Inco/A is highly effective in alleviating mechanical allodynia in a validated rat model of TN. The findings highlight Inco/A as a promising candidate for clinical translation in TN and related neuropathic pain syndromes and support systematic investigation in well-controlled human trials.
Keywords:
1. Introduction
2. Materials and Methods
Animals
Induction of Neuropathic Trigeminal Pain
Study Treatments
Assessment of Facial Mechanical Allodynia
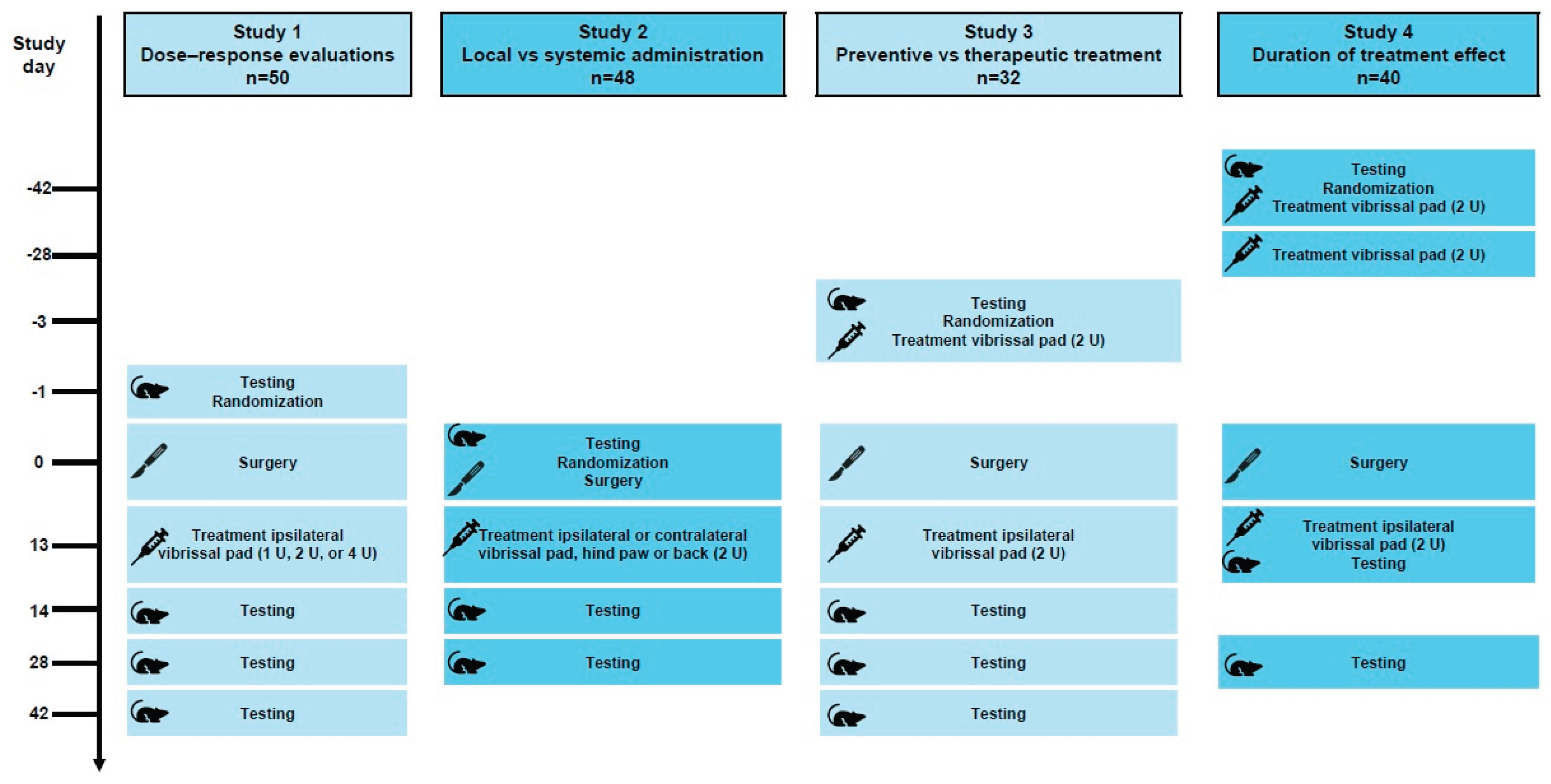
Study Designs
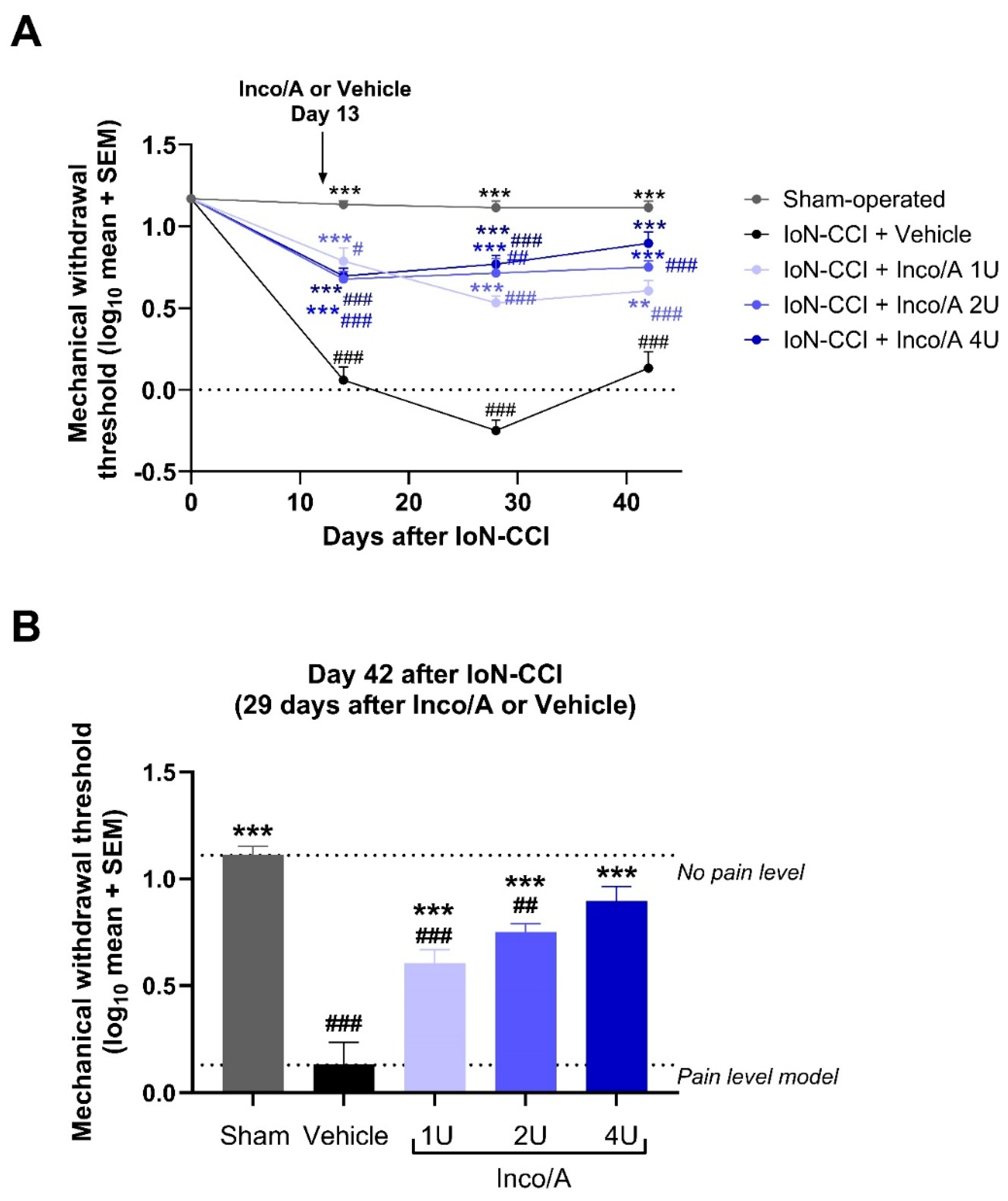
Study 1: Inco/A Administration and Evaluation of Dose–Response
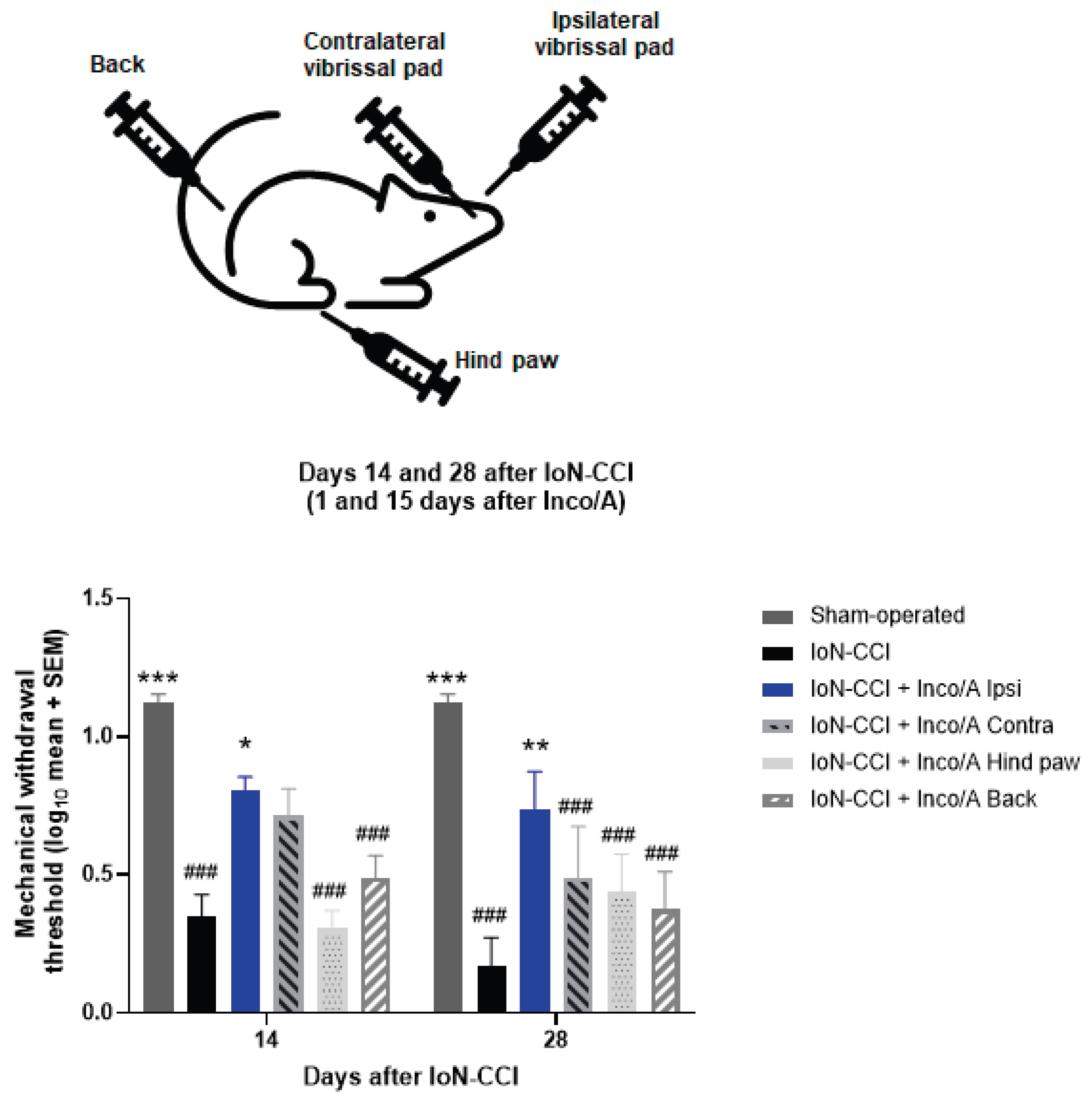
Study 2: Effects of Local Versus Systemic Inco/A Administration
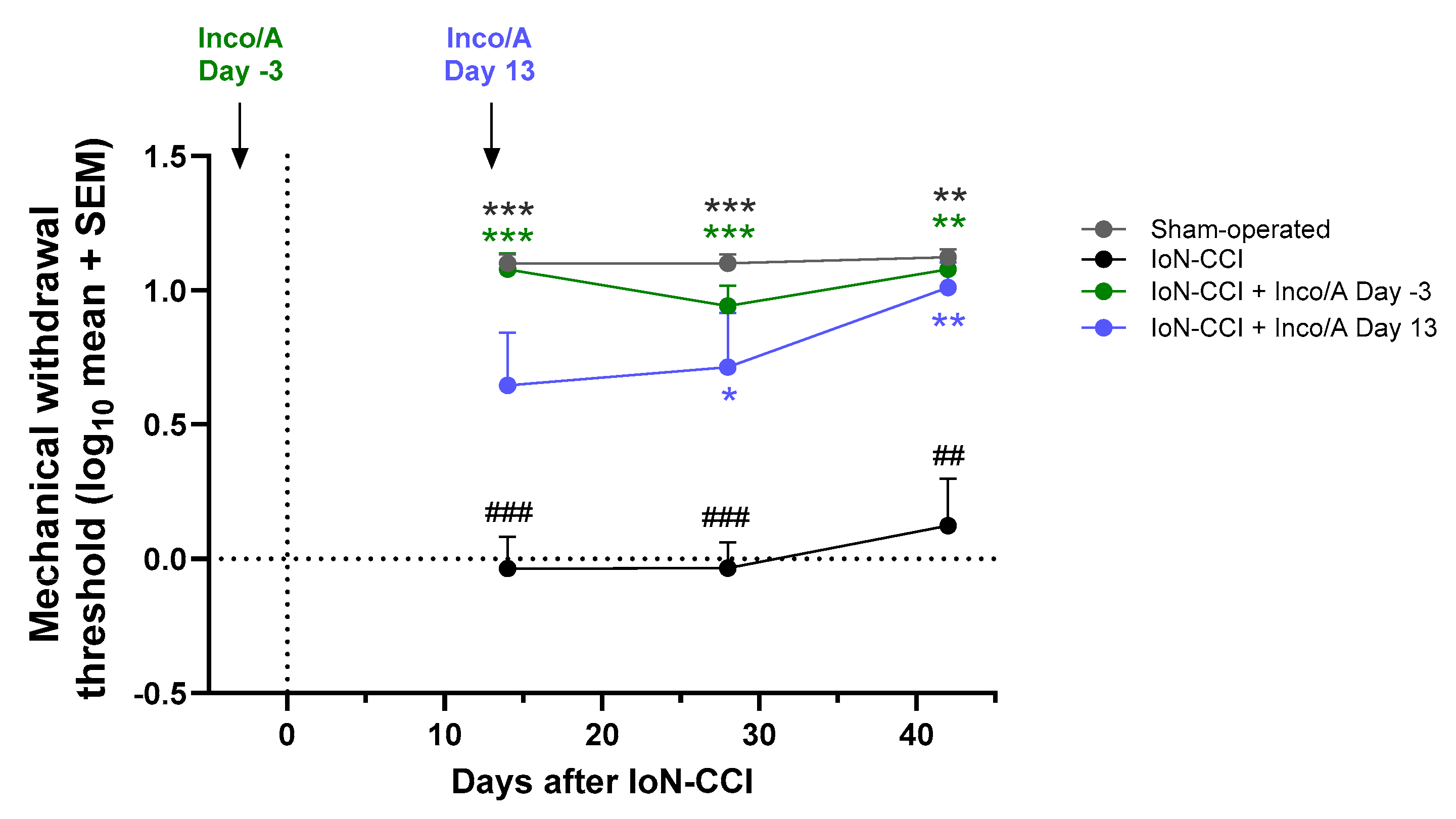
Study 3: Effects of Preventive Versus Therapeutic Inco/A Administration
Study 4: Duration of Inco/A Treatment Effect
Statistical Methods
3. Results
3.1. Study 1: Dose–Response Relationship and Anti-allodynic Effects of Inco/A
3.2. Study 2: Local, Regional, and Systemic Anti-allodynic Effects of Inco/A
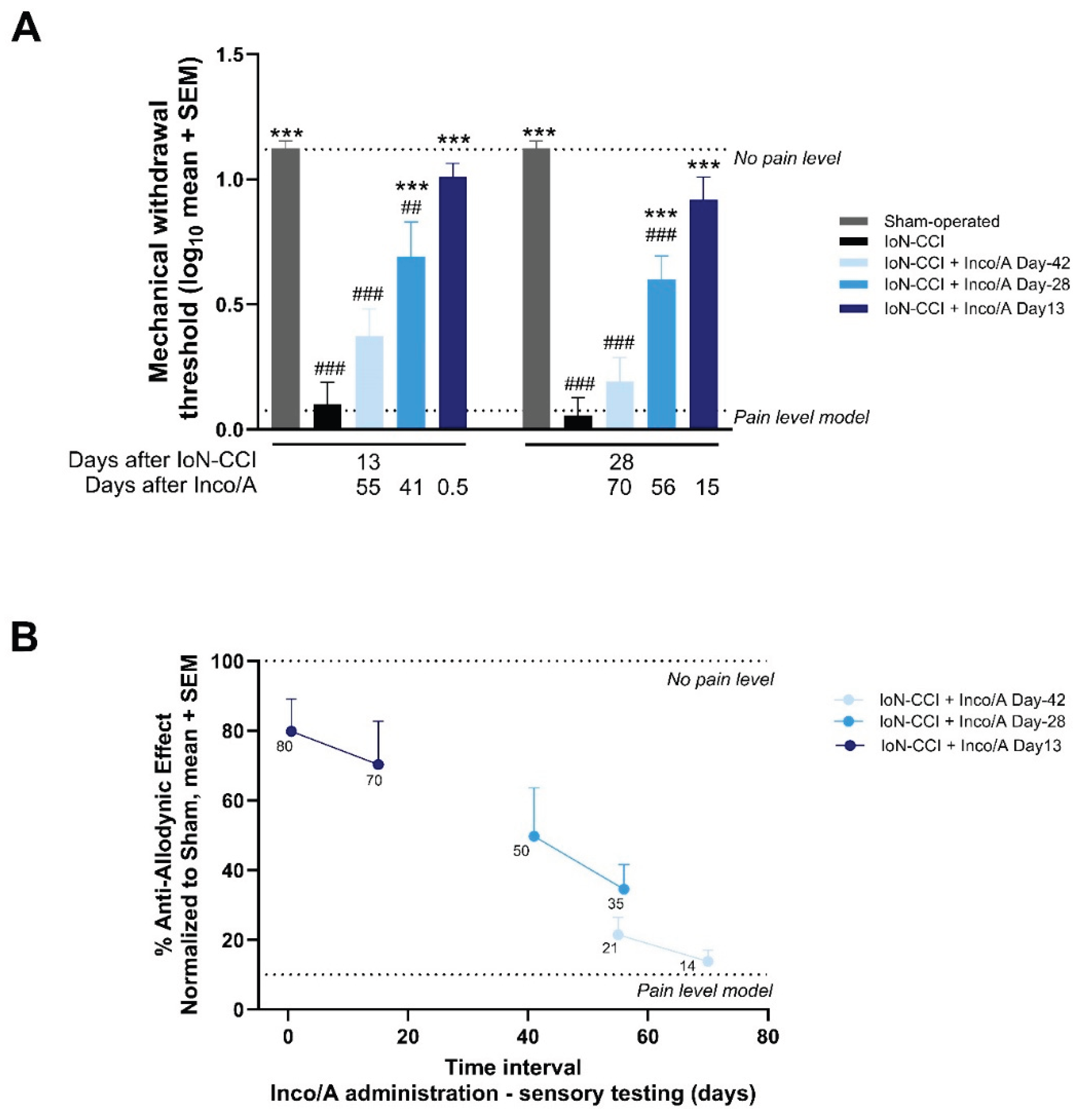
3.3. Study 3: Preventive and Therapeutic Anti-allodynic Effects of Inco/A
3.4. Study 4: Duration of Anti-allodynic Effects of Inco/A
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TN | trigeminal neuralgia |
| BoNT/A | botulinum neurotoxin type A |
| Inco/A | incobotulinumtoxinA |
| MS | multiple sclerosis |
| QoL | Quality of life |
| IoN-CCI | infraorbital nerve chronic constriction injury |
| HDB | HD Bioscience |
| MWT | mechanical withdrawal threshold |
| ANOVA | analysis of variance |
| SEM | standard error of the mean |
References
- No authors listed. Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018, 38, 1–211. [CrossRef]
- Lambru, G.; Zakrzewska, J.; Matharu, M. Trigeminal neuralgia: a practical guide. Pract. Neurol. 2021, 21, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Chong, M.S.; Bahra, A.; Zakrzewska, J.M. Guidelines for the management of trigeminal neuralgia. Cleve. Clin. J. Med. 2023, 90, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Fallata, A.; Salter, A.; Tyry, T.; Cutter, GR.; Marrie, RA. Trigeminal neuralgia commonly precedes the diagnosis of multiple sclerosis. Int. J. MS. Care. 2017, 19, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Mueller, D.; Obermann, M.; Yoon, M.S.; Poitz, F.; Hansen, N.; Slomke, M.A.; et al. Prevalence of trigeminal neuralgia and persistent idiopathic facial pain: a population-based study. Cephalalgia 2011, 31, 1542–1548. [Google Scholar] [CrossRef]
- Zakrzewska, J.M.; Wu, J.; Mon-Williams, M.; Phillips, N.; Pavitt, S.H. Evaluating the impact of trigeminal neuralgia. Pain 2017, 158, 1166–1174. [Google Scholar] [CrossRef]
- Martinelli, R.; Vannuccini, S.; Burattini, B.; D'Alessandris, Q.G.; D'Ercole, M.; Izzo, A.; et al. Psychological assessment in patients affected by trigeminal neuralgia. A systematic review. Neurosurg. Rev. 2025, 48, 414. [Google Scholar] [CrossRef]
- Xu, R.; Xie, M.E.; Jackson, C.M. Trigeminal neuralgia: current approaches and emerging interventions. J. Pain Res. 2021, 14, 3437–3463. [Google Scholar] [CrossRef]
- Pergolizzi, Jr. J.V.; LeQuang, J.A.; El-Tallawy, S.N.; Wagner, M.; Ahmed, R.S.; Varrassi, G. An update on pharmacotherapy for trigeminal neuralgia. Exp. Rev. Neurother. 2024, 24, 773–7786. [Google Scholar] [CrossRef]
- Ahmad, R.M.; Sher, K.; Devi, L.; Abrar, A.; Nisha; Muhammad, FG. Comparison of Oxcarbazepine Versus Carbamazepine in the Management of Trigeminal Neuralgia in Patients Presenting in the Outpatient Department of Neurology, Jinnah Postgraduate Medical Centre. Biol. Clin. Sci. Res. J. 2025, 6, 13–16. [Google Scholar] [CrossRef]
- Tohyama, S.; Datko, M.; Brusaferri, L.; Kinder, L.D.; Schnieders, J.H.; Hyman, M.; et al. Trigeminal nerve microstructure is linked with neuroinflammation and brainstem activity in migraine. Brain 2025, 148, 2551–2562. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, S.; Johnson, K.W.; Ossipov, M.H.; Aurora, S.K. CGRP and the trigeminal system in migraine. Headache 2019, 59, 659–681. [Google Scholar] [CrossRef]
- Lin, K.-H.; Chen, Y.-T.; Fuh, J.-L.; Wang, S.-J. Increased risk of trigeminal neuralgia in patients with migraine: A nationwide population-based study. Cephalalgia 2016, 36, 1218–1227. [Google Scholar] [CrossRef]
- Aurora, S.K.; Dodick, D.W.; Diener, H.C.; DeGryse, R.E.; Turkel, C.C.; Lipton, R.B.; et al. OnabotulinumtoxinA for chronic migraine: efficacy, safety, and tolerability in patients who received all five treatment cycles in the PREEMPT clinical program. Acta Neurol. Scand. 2014, 129, 61–70. [Google Scholar] [CrossRef]
- Domitrz, I.; Slawek, J.; Slowik, A.; Boczarska-Jedynak, M.; Stepien, A.; Rejdak, K.; et al. Onabotulinumtoxin A (ONA-BoNT/A) in the treatment of chronic migraine. Neurol. Neurochir. Pol. 2022, 56, 39–47. [Google Scholar] [CrossRef]
- Argyriou, A.A.; Mitsikostas, D.D.; Mantovani, E.; Vikelis, M.; Tamburin, S. Beyond chronic migraine: a systematic review and expert opinion on the off-label use of botulinum neurotoxin type-A in other primary headache disorders. Exp. Rev. Neurother. 2021, 21, 923–944. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Xia, Y.; Li, J.; Wang, X.; Liu, H.; Hu, J.; et al. Efficacy and safety of botulinum toxin type A in the treatment of trigeminal neuralgia: an update on systematic review with meta-analyses. Clin. J. Pain 2024, 40, 383–392. [Google Scholar] [CrossRef]
- Hosseindoost, S.; Askari Rad, M.; Inanloo, S.H.; Rahimi, M.; Dehghan, S.; Orandi, A.; et al. The analgesic effects of botulinum neurotoxin by modulating pain-related receptors; a literature review. Mol. Pain 2024, 20, 17448069241275099. [Google Scholar] [CrossRef]
- Vos, B.P.; Strassman, A.M.; Maciewicz, R.J. Behavioral evidence of trigeminal neuropathic pain following chronic constriction injury to the rat's infraorbital nerve. J. Neurosci. 1994, 14, 2708–2723. [Google Scholar] [CrossRef]
- Deseure, K.; Hans, G.H. Chronic constriction injury of the rat's infraorbital nerve (IoN-CCI) to study trigeminal neuropathic pain. J. Vis. Exp. 2015, 53167. [Google Scholar] [CrossRef]
- Harriott, A.M.; Strother, L.C.; Vila-Pueyo, M.; Holland, P.R. Animal models of migraine and experimental techniques used to examine trigeminal sensory processing. J. Headache. Pain 2019, 20, 91. [Google Scholar] [CrossRef] [PubMed]
- Noma, N.; Watanabe, K.; Sato, Y.; Imamura, Y.; Yamamoto, Y.; Ito, R.; et al. Botulinum neurotoxin type A alleviates mechanical hypersensitivity associated with infraorbital nerve constriction injury in rats. Neurosci. Lett. 2017, 637, 96–101. [Google Scholar] [CrossRef]
- Wu, C.; Xie, N.; Lian, Y.; Xu, H.; Chen, C.; Zheng, Y.; et al. Central antinociceptive activity of peripherally applied botulinum toxin type A in lab rat model of trigeminal neuralgia. Springerplus 2016, 5, 431. [Google Scholar] [CrossRef]
- Zhang, Y.; Su, Q.; Lian, Y.; Chen, Y. Botulinum toxin type A reduces the expression of transient receptor potential melastatin 3 and transient receptor potential vanilloid type 4 in the trigeminal subnucleus caudalis of a rat model of trigeminal neuralgia. Neuroreport 2019, 30, 735–740. [Google Scholar] [CrossRef]
- Grein, S.; Mander, G.; Fink, K. Stability of botulinum neurotoxin type A, devoid of complexing proteins. Botulinum J. 2011, 2, 49–58. [Google Scholar] [CrossRef]
- US Food and Drug Administration. Highlights of Prescribing Information: XEOMIN (incobotulinumtoxinA) for injection, for intramuscular or intraglandular use. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/125360s097lbl.pdf (accessed on 27 February 2026).
- Percie du Sert, N.; Hurst, V.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; et al. The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. BMJ Open Sci. 2020, 4, e100115. [Google Scholar] [CrossRef] [PubMed]
- Bonin, R.P.; Bories, C.; De Koninck, Y. A simplified up-down method (SUDO) for measuring mechanical nociception in rodents using von Frey filaments. Mol. Pain 2014, 10, 26. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.; Leblond, D.; Joshi, S.; Zhu, C.; Hsieh, G.; Jacobson, P.; et al. Estimating efficacy and drug ED50's using von Frey thresholds: impact of weber's law and log transformation. J. Pain 2012, 13, 519–523. [Google Scholar] [CrossRef]
- Marinelli, S.; Luvisetto, S.; Cobianchi, S.; Makuch, W.; Obara, I.; Mezzaroma, E.; et al. Botulinum neurotoxin type A counteracts neuropathic pain and facilitates functional recovery after peripheral nerve injury in animal models. Neurosci. 2010, 171, 316–328. [Google Scholar] [CrossRef]
- Shi, Y.; Gong, C.; Nan, W.; Zhou, W.; Lei, Z.; Zhou, K.; et al. Intrathecal administration of botulinum toxin type a antagonizes neuropathic pain by countering increased vesicular nucleotide transporter expression in the spinal cord of chronic constriction injury of the sciatic nerve rats. Neuropeptides 2023, 100, 102346. [Google Scholar] [CrossRef]
- Xiao, L.; Cheng, J.; Dai, J.; Zhang, D. Botulinum toxin decreases hyperalgesia and inhibits P2X3 receptor over-expression in sensory neurons induced by ventral root transection in rats. Pain Med. 2011, 12, 1385–1394. [Google Scholar] [CrossRef]
- Yang, K.Y.; Kim, M.J.; Ju, J.S.; Park, S.K.; Lee, C.G.; Kim, S.T.; et al. Antinociceptive effects of botulinum toxin type A on trigeminal neuropathic pain. J. Dent. Res. 2016, 95, 1183–1190. [Google Scholar] [CrossRef]
- Filipovic, B.; Matak, I.; Bach-Rojecky, L.; Lackovic, Z. Central action of peripherally applied botulinum toxin type A on pain and dural protein extravasation in rat model of trigeminal neuropathy. PLoS One 2012, 7, e29803. [Google Scholar] [CrossRef]
- Piovesan, E.J.; Oshinsky, M.; Silberstein, S.; Kowacs, P.A.; Novak, E.M.; Werneck, L.C. Botulinum neurotoxin type-A when utilized in animals with trigeminal sensitization induced a antinociceptive effect. Arq. Neuropsiquiatr. 2016, 74, 462–469. [Google Scholar] [CrossRef]
- Waskitho, A.; Yamamoto, Y.; Raman, S.; Kano, F.; Yan, H.; Raju, R.; et al. Peripherally administered botulinum toxin type A localizes bilaterally in trigeminal ganglia of animal model. Toxins (Basel) 2021, 13, 704. [Google Scholar] [CrossRef] [PubMed]
- Cui, M.; Khanijou, S.; Rubino, J.; Aoki, KR. Subcutaneous administration of botulinum toxin a reduces formalin-induced pain. Pain 2004, 107, 125–133. [Google Scholar] [CrossRef]
- Bach-Rojecky, L.; Lackovic, Z. Antinociceptive effect of botulinum toxin type a in rat model of carrageenan and capsaicin induced pain. Croat. Med. J. 2005, 46, 201–208. [Google Scholar]
- Towal, R.B.; Quist, B.W.; Gopal, V.; Solomon, J.H.; Hartmann, M.J.Z. The morphology of the rat vibrissal array: a model for quantifying spatiotemporal patterns of whisker-object contact. PLoS Comput. Biol. 2011, 7, e1001120. [Google Scholar] [CrossRef]
- Omoto, K.; Maruhama, K.; Terayama, R.; Yamamoto, Y.; Matsushita, O.; Sugimoto, T.; et al. Cross-excitation in peripheral sensory ganglia associated with pain transmission. Toxins (Basel) 2015, 7, 2906–2917. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Guo, R.; Sun, Y.; Li, H.; Ma, D.; Zhang, C.; et al. Botulinum toxin type A and gabapentin attenuate postoperative pain and NK1 receptor internalization in rats. Neurochem. Int. 2018, 116, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.M.; Chung, M.E. Botulinum toxin for neuropathic pain: a review of the literature. Toxins (Basel) 2015, 7, 3127–3154. [Google Scholar] [CrossRef]
- Bagues, A.; Hu, J.; Alshanqiti, I.; Chung, M.K. Neurobiological mechanisms of botulinum neurotoxin-induced analgesia for neuropathic pain. Pharmacol. Ther. 2024, 259, 108668. [Google Scholar] [CrossRef]
- Santos, R.F.; Machado, M.; Ferro, M.; Camões-Barbosa, A. Botulinum toxin type A in trigeminal neuralgia treatment: a case series and literature review. Cureus 2024, 16, e73389. [Google Scholar] [CrossRef]
- Yoshida, K. Effects of botulinum toxin type A on pain among trigeminal neuralgia, myofascial temporomandibular disorders, and oromandibular dystonia. Toxins (Basel) 2021, 13, 605. [Google Scholar] [CrossRef]
- Wu, C.J.; Lian, Y.J.; Zheng, Y.K.; Zhang, H.F.; Chen, Y.; Xie, N.C.; et al. Botulinum toxin type A for the treatment of trigeminal neuralgia: results from a randomized, double-blind, placebo-controlled trial. Cephalalgia 2012, 32, 443–450. [Google Scholar] [CrossRef]
- Bohluli, B.; Motamedi, M.H.K.; Bagheri, S.C.; Bayat, M.; Lassemi, E.; Navi, F.; et al. Use of botulinum toxin A for drug-refractory trigeminal neuralgia: preliminary report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2011, 111, 47–50. [Google Scholar] [CrossRef]
- Shehata, H.S.; El-Tamawy, M.S.; Shalaby, N.M.; Ramzy, G. Botulinum toxin-type A: could it be an effective treatment option in intractable trigeminal neuralgia? J. Headache Pain 2013, 14, 92. [Google Scholar] [CrossRef] [PubMed]
- Türk Börü, Ü.; Duman, A.; Bölük, C.; Coşkun Duman, S.; Taşdemir, M. Botulinum toxin in the treatment of trigeminal neuralgia: 6-Month follow-up. Medicine (Baltimore) 2017, 96, e8133. [Google Scholar] [CrossRef] [PubMed]
- Duran-Hernandez, S.; Herrera-Gonzalez, N.E.; Duran-Hernandez, N.; Carnalla, M.; Castillejos-Lopez, M.J.; Salinas-Lara, C. Single-dose incobotulinumtoxinA in the treatment of early-stage knee osteoarthritis: results from a preliminary single-arm clinical trial. Toxins (Basel) 2025, 17, 216. [Google Scholar] [CrossRef]
- Eisele, K.H.; Fink, K.; Vey, M.; Taylor, H.V. Studies on the dissociation of botulinum neurotoxin type A complexes. Toxicon 2011, 57, 555–565. [Google Scholar] [CrossRef]
- Frevert, J.; Dressler, D. Complexing proteins in botulinum toxin type A drugs: a help or a hindrance? Biologics 2010, 4, 325–332. [Google Scholar] [CrossRef]
- Bolívar, S.; Sanz, E.; Ovelleiro, D.; Zochodne, D.W.; Udina, E. Neuron-specific RNA-sequencing reveals different responses in peripheral neurons after nerve injury. eLife 2024, 12, RP91316. [Google Scholar] [CrossRef] [PubMed]
- Ryu, H.J.; Kwak, S.; Park, M.; Yun, H.Y. Model-based interspecies interpretation of botulinum neurotoxin type A on muscle-contraction inhibition. Biopharm. Drug Dispos. 2024, 45, 190–200. [Google Scholar] [CrossRef]
- Wilcox, M.B.; Jessen, K.R.; Quick, T.J.; Phillips, J.B. The molecular profile of nerve repair: humans mirror rodents. Neural. Regen. Res. 2021, 16, 1440–1441. [Google Scholar] [CrossRef]
- Pellett, S.; Bradshaw, M.; Tepp, W.H.; Pier, C.L.; Whitemarsh, R.C.M.; Chen, C.; et al. The light chain defines the duration of action of botulinum toxin serotype A subtypes. mBio 2018, 9, e00089-18. [Google Scholar] [CrossRef]
- Matak, I.; Bolcskei, K.; Bach-Rojecky, L.; Helyes, Z. Mechanisms of botulinum toxin type A action on pain. Toxins (Basel) 2019, 11, 459. [Google Scholar] [CrossRef]
- Tereshko, Y.; Dal Bello, S.; Lettieri, C.; Belgrado, E.; Gigli, G.L.; Merlino, G.; et al. Botulinum toxin type A for trigeminal neuralgia: a comprehensive literature review. Toxins (Basel) 2024, 16, 500. [Google Scholar] [CrossRef] [PubMed]
- Matak, I.; Tekus, V.; Bolcskei, K.; Lackovic, Z.; Helyes, Z. Involvement of substance P in the antinociceptive effect of botulinum toxin type A: Evidence from knockout mice. Neuroscience 2017, 358, 137–145. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



