Submitted:
10 March 2026
Posted:
11 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Study Design and Participants
Ethical Statement
MIRA Technique
Patient Stratification and Treatment Technique
Treatment Modalities
Evaluation Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CDT | Carbon Dioxide Therapy |
| DEC | Dermo-Epidermal Complex |
| EFP | Edematous Fibrosclerotic Panniculopathy |
| IST | Injectable Solution Therapy |
| MIRA | Modulated Insertion of Regenerative Activation |
References
- Sfyri, Ε. Cellulite – Etiology, Epidemiology and Management. 2025, 39, 25–28. [Google Scholar] [CrossRef]
- Tanzi, E.L.; Capelli, C.C.; Robertson, D.W.; LaTowsky, B.; Balcom-Luker, S.; Jacob, C.; Ibrahim, O.; Kaminer, M.S. Improvement in Cellulite Appearance After a Single Treatment Visit With Acoustic Subcision: Long-Term Findings From a Multicenter Clinical Trial. Dermatol Surg 2023, 50, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Bass, L.S.; Hibler, B.P.; Khalifian, S.; Shridharani, S.M.; Klibanov, O.M.; Moradi, A. Cellulite Pathophysiology and Psychosocial Implications. Clin Cosmet Investig Dermatol 2023, 49, S2–S7. [Google Scholar] [CrossRef] [PubMed]
- Hartman, N.; Almukhtar, R.; Wood, E.S.; Fabi, S.G. Collagenase Clostridium Histolyticum-Aaes Injections for Volumetric Change of Cellulite Dimples and Gluteal Contouring. Dermatol Surg 2023, 49, 383–386. [Google Scholar] [CrossRef]
- Nobile, V.; Cestone, E.; Puoci, F.; Ponti, I.D.; Pisati, M.; Michelotti, A. In Vitro and in Vivo Study on Humans of Natural Compound Synergy as a Multifunctional Approach to Cellulite-Derived Skin Imperfections. Cosmetics 2020, 7, 48. [Google Scholar] [CrossRef]
- Bennardo, L.; Fusco, I.; Cuciti, C.; Sicilia, C.; Salsi, B.; Cannarozzo, G.; Hoffmann, K.; Nisticò, S.P. Microwave Therapy for Cellulite: An Effective Non-Invasive Treatment. J Clin Med 2022, 11, 515. [Google Scholar] [CrossRef]
- Mlosek, K.; Malinowska, S. High-Frequency Ultrasound in the Assessment of Cellulite—Correlation Between Ultrasound-Derived Measurements, Clinical Assessment, and Nürnberger–Müller Scale Scores. Diagnostics 2024, 14, 1878. [Google Scholar] [CrossRef]
- Sadick, N.S.; Goldman, M.P.; Liu, G.; Shusterman, N.H.; McLane, M.; Hurley, D.; Young, V.L. Collagenase Clostridium Histolyticum for the Treatment of Edematous Fibrosclerotic Panniculopathy (Cellulite): A Randomized Trial. Dermatol Surg 2019, 45, 1047–1056. [Google Scholar] [CrossRef]
- Sant′Ana, E.M.C.; Pianez, L.R.G.; Custódio, F.S.; Guidi, R.M.; Freitas, J.N.d. Effectiveness of Carboxytherapy in the Treatment of Cellulite in Healthy Women: A Pilot Study. Clin Cosmet Investig Dermatol 2016, Volume 9, 183–190. [Google Scholar] [CrossRef]
- Di Guardo, A.; Solito, C.; Cantisani, V.; Rega, F.; Gargano, L.; Rossi, G.; Musolff, N.; Azzella, G.; Paolino, G.; Losco, L.; et al. Clinical and Ultrasound Efficacy of Topical Hypertonic Cream (Jovita Osmocell(®)) in the Treatment of Cellulite: A Prospective, Monocentric, Double-Blind, Placebo-Controlled Study. Medicina (Kaunas) 2024, 60. [Google Scholar] [CrossRef]
- Pérez Atamoros, F.M.; Alcalá Pérez, D.; Asz Sigall, D.; Ávila Romay, A.A.; Barba Gastelum, J.A.; de la Peña Salcedo, J.A.; Escalante Salgado, P.E.; Gallardo Palacios, G.J.; Guerrero-Gonzalez, G.A.; Morales De la Cerda, R.; et al. Evidence-based treatment for gynoid lipodystrophy: A review of the recent literature. J Cosmet Dermatol 2018, 17, 977–983. [Google Scholar] [CrossRef] [PubMed]
- Bagherani, N.; Smoller, B.R.; Tavoosidana, G.; Ghanadan, A.; Wollina, U.; Lotti, T. An Overview of the Role of Carboxytherapy in Dermatology. J Cosmet Dermatol 2023, 22, 2399–2407. [Google Scholar] [CrossRef] [PubMed]
- Chieregato, F.; Silva, C.N.d.; Carvalho, T.M.D.; Grecco, C.; Moreira, R.G.; Guidi, R.M.; Girola, L.F.; Souza, J.R.d.; Modena, D.A.O. Effects of Multi-Directional Oscillatory Vibration in the Treatment of Cellulite and Body Remodeling. Fisioterapia Brasil 2020, 21, 77–85. [Google Scholar] [CrossRef]
- Melo, D.F.; Barreto, T.d.M.; Plata, G.T.; Araujo, L.R.; Tortelly, V.D. Excellent Response to Mesotherapy as Adjunctive Treatment in Male Androgenetic Alopecia. J Cosmet Dermatol 2019, 19, 75–77. [Google Scholar] [CrossRef]
- Podgórna, K.; Kołodziejczak, A.; Rotsztejn, H. Cutometric Assessment of Elasticity of Skin With Striae Distensae Following Carboxytherapy. J Cosmet Dermatol 2017, 17, 1170–1174. [Google Scholar] [CrossRef]
- Kołodziejczak, A.; Jatczak, A.; Rotsztejn, H. The Correlation Between the Severity of Symptoms and the Thickness of the Fat Fold in Cellulite-affected Areas—A Pilot Study. J Cosmet Dermatol 2022, 21, 5852–5858. [Google Scholar] [CrossRef]
- Tang, Z.; Hu, Y.; Wang, J.; Fan, Z.; Qu, Q.; Miao, Y. Current Application of Mesotherapy in Pattern Hair Loss: A systematic Review. J Cosmet Dermatol 2022, 21, 4184–4193. [Google Scholar] [CrossRef]
- Eldsouky, F.; Ebrahim, H.M. Evaluation and Efficacy of Carbon Dioxide Therapy (Carboxytherapy) Versus Mesolipolysis in the Treatment of Cellulite. Journal of Cosmetic and Laser Therapy 2018, 20, 307–312. [Google Scholar] [CrossRef]
- Bauer, J.; Grabarek, M.; Migasiewicz, A.; Podbielska, H. Non-Contact Thermal Imaging as Potential Tool for Personalized Diagnosis and Prevention of Cellulite. J Therm Anal Calorim 2018, 133, 571–578. [Google Scholar] [CrossRef]
- Intagliata, D.; Garo, M.L. The Role of Ultrasound in the Diagnosis and Treatment of Cellulite: A Systematic Review. J Clin Med 2026, 15, 943. [Google Scholar] [CrossRef]
- Intagliata, D.; Priolo, M.; Molinari, P. High-Frequency Ultrasound Imaging for Stage III Cellulite: A Three-Subtype Structural Classification from an Observational Cohort Study. Dermatology and Therapy 2025. [Google Scholar] [CrossRef] [PubMed]
- Nürnberger, F.; Müller, G. So-Called Cellulite: An Invented Disease. J Dermatol Surg 1978, 4, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Dal'Forno, T.; Del Nero, M.P.; Nunes, F.; Cunha, C.; Haddad, A.; Vilarinho, A.; Nogueira, A.; Tomaz, R. Recomendações clínicas para o uso combinado de ácido poli-L-láctico (PLLA-SCA) e dispositivos baseados em energia: opinião de especialistas e revisão da literatura. Surgical & Cosmetic Dermatology 2025, 17, 1–10. [Google Scholar] [CrossRef]
- Gabriel, A.; Chan, V.; Caldarella, M.; Wayne, T.; O'Rorke, E. Cellulite: Current Understanding and Treatment. Aesthet Surg J Open Forum 2023, 5, ojad050. [Google Scholar] [CrossRef]
- Hexsel, D.M.; Siega, C.; Schilling-Souza, J.; Porto, M.D.; Rodrigues, T.C. A bipolar radiofrequency, infrared, vacuum and mechanical massage device for treatment of cellulite: a pilot study. J Cosmet Laser Ther 2011, 13, 297–302. [Google Scholar] [CrossRef]
- Querleux, B.; Cornillon, C.; Jolivet, O.; Bittoun, J. Anatomy and physiology of subcutaneous adipose tissue by in vivo magnetic resonance imaging and spectroscopy: relationships with sex and presence of cellulite. Skin Res Technol 2002, 8, 118–124. [Google Scholar] [CrossRef]
- Fede, C.; Clair, C.; Pirri, C.; Petrelli, L.; Zhao, X.; Sun, Y.; Macchi, V.; Stecco, C. The Human Superficial Fascia: A Narrative Review. Int J Mol Sci 2025, 26, 1289. [Google Scholar] [CrossRef]
- Rossi, A.B.R.; Vergnanini, A.L. Cellulite: a review. J Eur Acad Dermatol Venereol 2000, 14, 251–262. [Google Scholar] [CrossRef]
- Terranova, F.; Berardesca, E.; Maibach, H. Cellulite: nature and aetiopathogenesis. International Journal of Cosmetic Science 2006, 28, 157–167. [Google Scholar] [CrossRef]
- Cotofana, S.; Kaminer, M.S. Anatomic update on the 3-dimensionality of the subdermal septum and its relevance for the pathophysiology of cellulite. J Cosmet Dermatol 2022, 21, 3232–3239. [Google Scholar] [CrossRef]
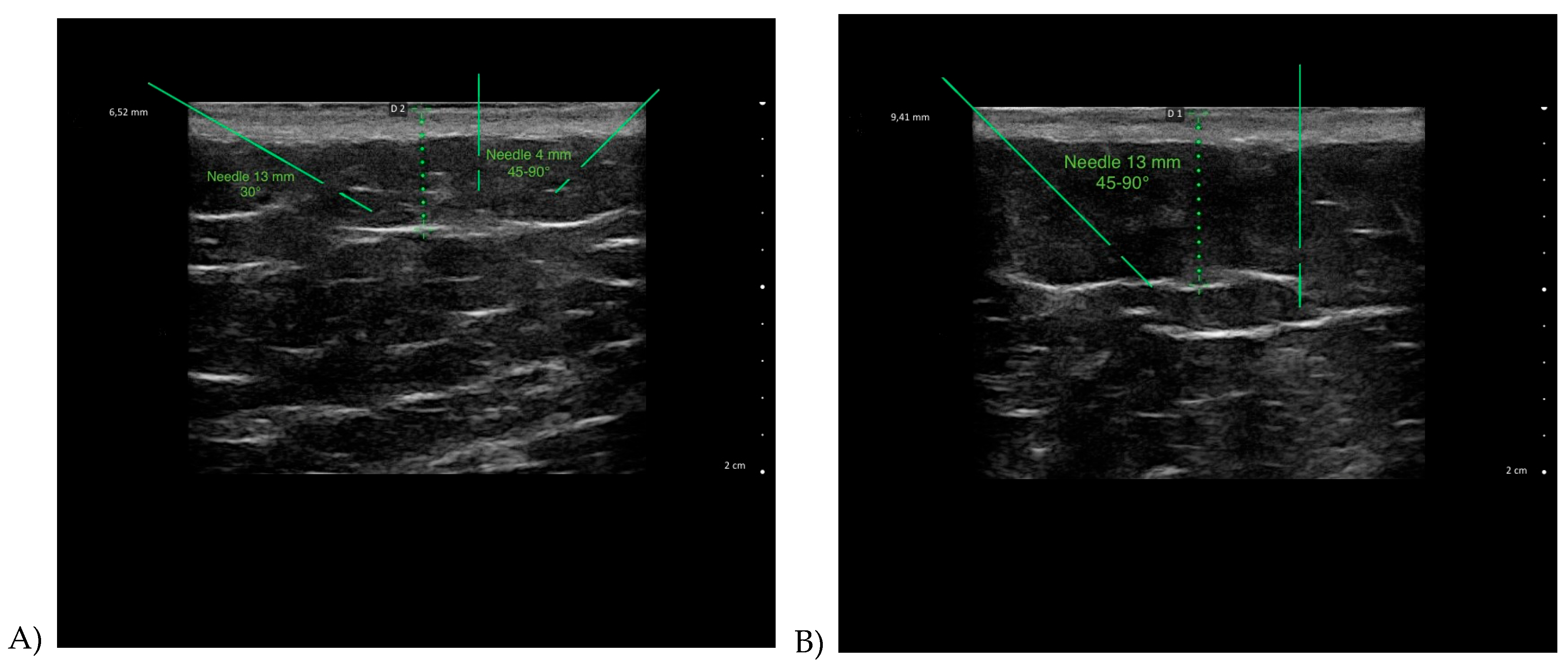
| Ultrasound parameters | Needle length | Injection depth & angle | Clinical rationale |
| Stage 3A | 4–6 mm | Superficial injection at 30°–45° | Targets superficial adipose layer to reduce edema, improve skin turgor, and minimize unnecessary adipose removal |
| Stage 3B | 13 mm | Deep bolus injection at 45°–90° | Acts on deep fibrous septae and excess adipose tissue, improves local drainage, reduces tissue rigidity |
| Stage Mixed | 4–13 mm | 30°–45°–90° | Targets overlapping or intermediate patterns adapting to the specificities of the tissues. |
| Aspect | Standard Technique | MIRA Technique |
| Needle selection | Fixed needle length, independent of patient anatomy | Needle length individualized according to ultrasound-derived subcutaneous thickness |
| Injection angle | Standardized angle (usually fixed for all patients) | Angle adapted to fascial integrity, fibrosis and edema as assessed by ultrasound |
| Treatment planning | Based on clinical staging only | Based on combined clinical and echographic staging (IEC system) |
| Targeting of tissue layers | Approximate, operator-dependent | Precise targeting of adipose lobules and fibrous septa identified on ultrasound |
| Reproducibility | High variability between operators | Reduced operator-dependent variability due to imaging-based guidance |
| Therapeutic goal | General improvement in skin appearance | Patient-specific correction of adipose hypertrophy, septal remodeling, edema reduction |
| Conceptual framework | Empirical aesthetic medicine | Precision aesthetic medicine, outcome-oriented |
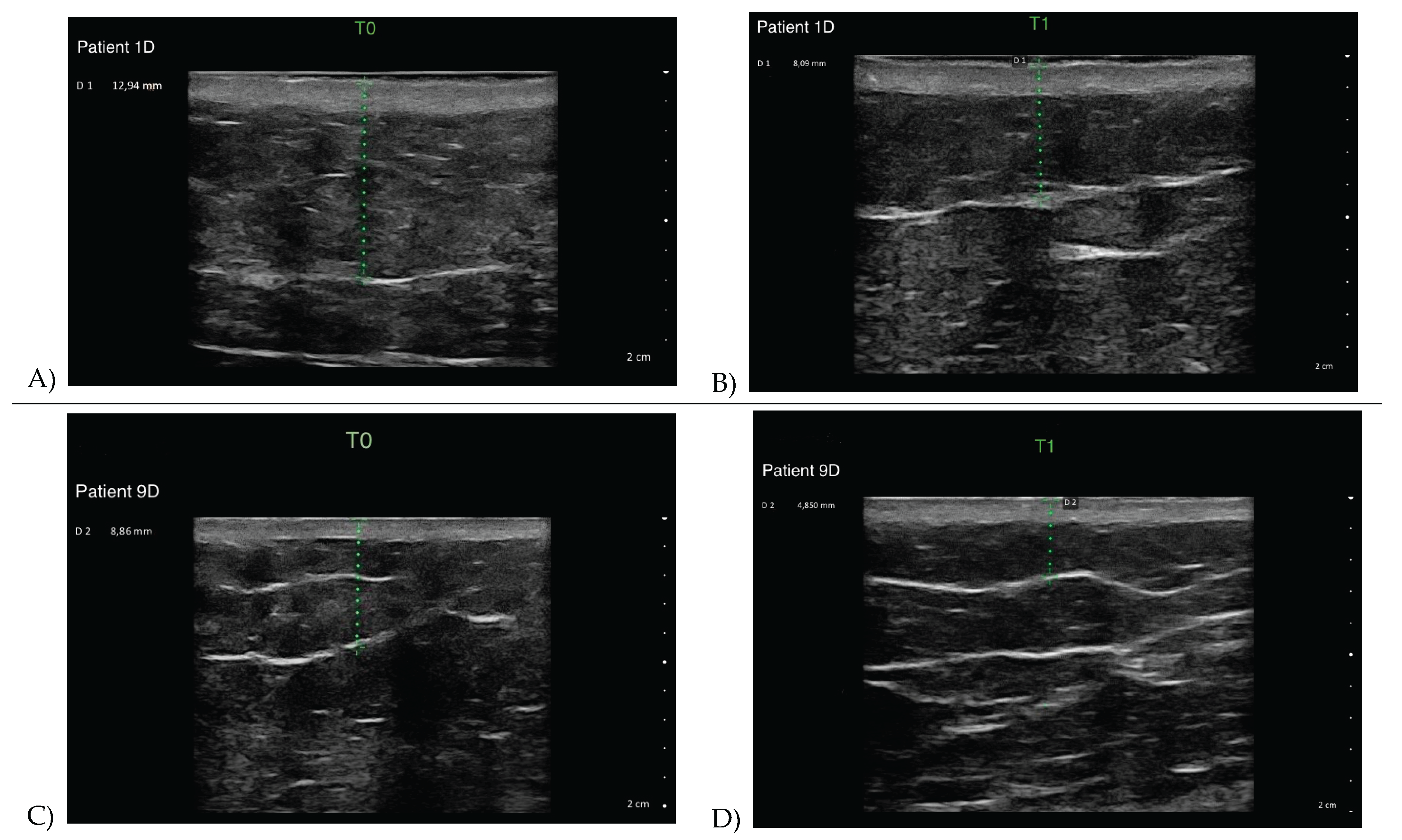
| Step | Criteria / Actions |
| 1. High-resolution ultrasound assessment | Subcutaneous thickness Fibrosis severity Edema grade Fascial integrity |
| 2. Classification (IEC system) | Stage 3A Stage Mixed Stage 3B |
| 3. Treatment type selection | Needle length selection: < 7.5 mm thickness → 4–6 mm needle > 7.5 mm thickness → 13 mm needle Injection angle selection: Superficial → 30°–45° Deep → 45°–90° |
| 4. MIRA guided injection procedure | Execution of the technique based on individualized parameters |
| 5. Follow-up & outcome evaluation | Ultrasound assessment, clinical evaluation, and patient-reported satisfaction |
| CDTMIRA | CDTCTL | p-value | ISTMIRA | ISTCTL | p-value | |
| Age | 37.1 (±9.6) [40, 30;43] |
35.4 (±7.3) [33.5, 29;35.4] |
0.425* | 36.1 (±11) [33.5, 27;45] |
35.9 (±10.3) [33.5, 27;45] |
0.997* |
| BMI (kg/m2) | 23.5 (±3.4) [22.5, 20.8;26.9] |
22.9 (±2.3) [22.8, 20.9;22.9] |
0.716* | 24.3 (±3.3) [24.4, 21.4;27.2] |
25 (±2.6) [25.25, 23.2;27.2] |
0.447* |
| Hormonal Imbalances | 0 (0.0) | 2 (6.7) | 0.492§ | 1 (3.3) | 1 (3.3) | 1.000§ |
| Pharmacological therapies | 1 (3.3) | 2 (6.7) | 1.000§ | 2 (6.7) | 1 (3.3) | 1.000§ |
| CDTMIRA | CDTCTL |
p-value (padj) |
ISTMIRA | ISTCTL |
p-value (padj) |
|
| Baseline | 6.7 (±1.4) [6.95, 5.3;7.9] |
6.9 (±1.0) [6.85, 6.2;6.9] |
0.343 (0.549) |
7.4 (±1.5) [7.75, 6.7;8.5] |
7.1 (±1.6) [7.65, 5.4;8.4] |
0.843 (0.999) |
| Follow-up | 5.2 (±1.1) [5.1, 4.5;6.2] |
6.8 (±0.9) [6.8, 6.2;6.8] |
<0.001 (0.002) |
5.6 (±1.1) [5.55, 5.1;6.2] |
7.1 (±1.5) [7.6, 5.4;8.1] |
<0.001 (0.002) |
| p-value (padj) | <0.001 (0.002) |
0.993 (0.999) |
<0.001 (0.002) |
0.999 (0.999) |
| CDTMIRA | CDTCTL |
p-value (padj) |
ISTMIRA | ISTCTL |
p-value (padj) |
|
| Baseline | 18 (60.0) | 24 (80.0) | 0.158 (0.211) |
16 (53.3) | 16 (53.3) | 1.000 (1.000) |
| Follow-up | 7 (23.3) | 20 (66.7) | 0.002 (0.004) |
1 (3.3) | 16 (53.3) | <0.001 (0.003) |
| p-value (padj) | 0.001 (0.003) |
0.045 (0.072) |
<0.001 (0.003) |
1.000 (1.000) |
| CDTMIRA | CDTCTL | p-value (padj) | ISTMIRA | ISTCTL | p-value (padj) | |
| Baseline | 19 (63.3) | 25 (83.3) | 0.143 (0.179) |
18 (60.0) | 22 (73.3) | 0.412 (0.412) |
| Follow-up | 7 (23.3) | 20 (66.7) | 0.002 (0.004) |
5 (16.7) | 20 (66.7) | <0.001 (0.003) |
| p-value (padj) | <0.001 (0.003) |
0.025 (0.040) |
<0.001 (0.003) |
0.157 (0.179) |
| CDTMIRA | CDTCTL |
p-value (padj) |
ISTMIRA | ISTCTL |
p-value (padj) |
|
| Baseline | ||||||
| Regular | 20 (66.7) | 20 (66.7) | 1.000 (1.000) |
11 (36.7) | 11 (36.7) | 1.000 (1.000) |
| Irregular | 10 (33.3) | 10 (33.3) | 19 (63.3) | 19 (63.3) | ||
| Follow-up | ||||||
| Regular | 26 (86.7) | 15 (50.0) | 0.005 (0.013) |
23 (76.7) | 11 (36.7) | 0.004 (0.013) |
| Irregular | 4 (13.3) | 15 (50.0) | 7 (23.3) | 19 (63.3) | ||
| p-value (padj) | 0.014 (0.028) |
0.096 (0.154) |
0.001 (0.008) |
1.000 (1.000) |
| CDTMIRA | CDTCTL |
p-value (padj) |
ISTMIRA | ISTCTL |
p-value (padj) |
|
| Baseline | 22 (73.3) | 28 (93.3) | 0.080 (0.111) |
27 (90.0) | 26 (86.7) | 1.000 (1.000) |
| Follow-up | 14 (46.7) | 25 (83.3) | 0.006 (0.016) |
19 (63.3) | 26 (86.7) | 0.072 (0.111) |
| p-value (padj) | 0.005 (0.016) |
0.083 (0.111) |
0.005 (0.016) |
1.000 (1.000) |
| CDTMIRA | CDTCTL |
p-value (padj) |
ISTMIRA | ISTCTL |
p-value (padj) |
|
| Baseline | ||||||
| Absent | 4 (13.3) | 8 (26.7) | 0.586 (0.938) |
5 (16.7) | 5 (16.7) | 1.000 (1.000) |
| Mild | 16 (53.3) | 12 (40.0) | 5 (16.7) | 6 (20.0) | ||
| Moderate | 7 (23.3) | 6 (20.0) | 14 (46.7) | 13 (43.3) | ||
| Grave | 3 (10) | 4 (13.3) | 6 (20) | 6 (20) | ||
| Follow-up | ||||||
| Absent | 13 (43.3) | 8 (26.7) | 0.025 (0.050) |
11 (36.7) | 5 (16.7) | 0.006 (0.016) |
| Mild | 16 (53.3) | 12 (40.0) | 13 (43.3) | 7 (23.3) | ||
| Moderate | 1 (3.3) | 6 (20.0) | 6 (20) | 12 (40.0) | ||
| Grave | 0 (0) | 4 (13.3) | 0 (0) | 6 (20.0) | ||
| p-value (padj) (Baseline vs. Follow-up) |
<0.001 (0.004) |
1.000 (1.000) |
<0.001 (0.004) |
1.000 (1.000) |
| CDTMIRA | CDTCTL | p-value (padj) | ISTMIRA | ISTCTL | p-value (padj) | |
| Perceived improvement | 8.1 (±1.6) [8, 7;9] |
6.1 (±0.8) [6, 6;6.1] |
<0.001 (0.002) |
8.5 (±1.4) [9, 8;10] |
6.5 (±0.7) [6, 6;7] |
<0.001 (0.002) |
| Skin quality | ||||||
| As Before | 3 (10.0) | 19 (63.3) | <0.001 (0.002) |
2 (6.7) | 12 (40.0) | <0.001 (0.002) |
| Normal | 4 (13.3) | 7 (23.3) | 3 (10.0) | 12 (40.0) | ||
| Improved | 23 (76.7) | 4 (13.3) | 25 (83.3) | 6 (20.0) | ||
| Cellulite visual appearance T3 | ||||||
| Orange peel skin | 5 (16.7) | 19 (63.3) | <0.001 (0.002) |
6 (20.0) | 15 (50.0) | <0.001 (0.002) |
| Mild waviness | 6 (20.0) | 10 (33.3) | 6 (20.0) | 10 (33.3) | ||
| Absent | 19 (63.3) | 1 (3.3) | 18 (60.0) | 5 (16.7) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




