Submitted:
09 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. The Structural Demographic Challenge
1.2. The Insufficiency of Conventional Responses
1.3. Agentic AI Through a Sociotechnical Lens
- −
- RQ1 (Capability): What is the current and near-term evidential basis for AAI across remote monitoring, assistive robotics, diagnostic support, medication adherence, and workflow automation, and how does evidence vary with intervention complexity?
- −
- RQ2 (Implementation): Which regulatory, financial, organisational, and infrastructural factors determine whether AI capability translates into population-level benefit, and how do these vary across national contexts?
- −
- RQ3 (Equity and Sustainability): Under what conditions can AI be adopted to avoid widening inequities and contribute to fiscal moderation, and which governance and evidence gaps prevent validation at scale?
2. Methodology
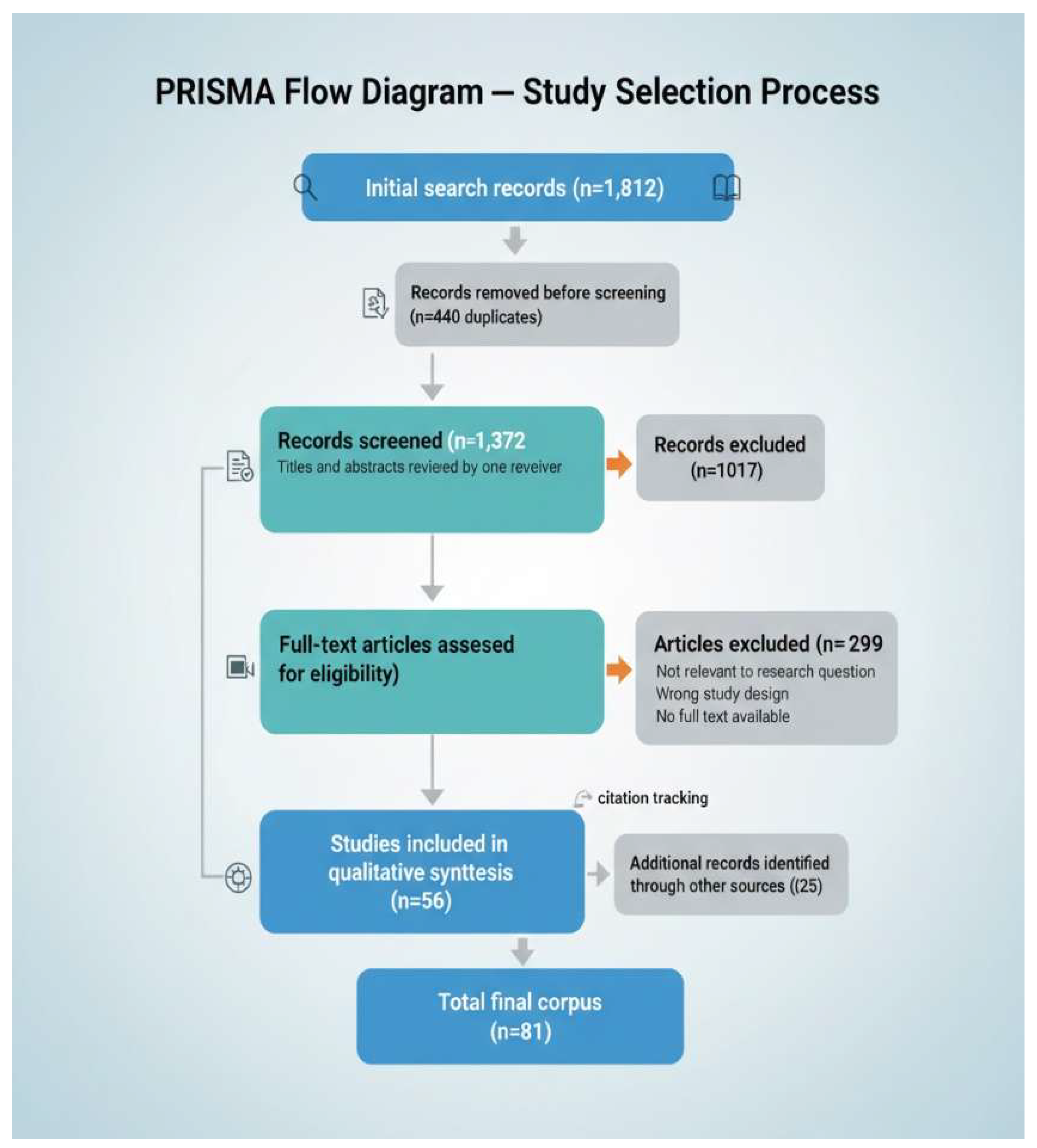
2.1. Search Strategy and Source Selection
2.2. Screening and Selection Process
2.3. Quality Appraisal, Data Extraction and Synthesis
3. Evidence Synthesis: Structural Pressures, AI Applications, and Institutional Constraint
3.1. Structural Pressures on Ageing Health Systems
3.2. AI Applications in Ageing Health Systems
3.3. Economic Evidence: Microeconomic Gains and Macroeconomic Uncertainty
| Economic Layer | Mechanism | Evidence Strength | Key Limitation | Implication |
| Healthcare spending | Reduced admissions through remote monitoring and coordination | Moderate evidence from pilot and controlled studies [19,20] | Effects vary by context and are not validated nationally | Promising but requires system-level evaluation |
| Long-term care costs | Delayed institutionalisation through ageing-in-place technologies | Moderate evidence with sustained deployment [31] | Demographic growth may offset efficiency gains | Scale required for fiscal impact |
| Workforce productivity | Automation of documentation and coordination tasks [41] | Emerging observational evidence | Implementation costs and workflow risks unclear | Near-term productivity gains possible |
| Macro-fiscal impact | AI-augmented care workforce | No validated population-level evidence | Current estimates rely on extrapolation | IMF projects health spending increase of 2.0–6.0% GDP by 2050 [46] |
3.4. Governance, Regulation, and the Algorithmic Bias Problem
3.5. Equity as a Structural Barrier, Not an Afterthought
3.6. Comparative Institutional Readiness
|
Case Study Singapore as a Governance First Benchmark for Ageing System Transformation Singapore provides one of the clearest examples of governance aligned preparation for population ageing. The country faces one of the steepest projected dependency ratios increases globally with the old age dependency ratio expected to rise from 24.2 in 2023 to approximately 85 by 2050. At the same time national healthcare expenditure is projected to reach about S$59 billion by 2030. In response the government has implemented a coordinated national strategy that integrates prevention financing workforce policy digital infrastructure and cross sector governance.[49,54] Preventive System Transformation – The Healthier SG The Healthier SG initiative launched in 2022 restructures primary care around longitudinal preventive management. All residents aged 40 and above are enrolled with a designated primary care physician and receive personalised preventive plans supported by digital monitoring tools including the Healthy 365 application. The government has committed about S$1 billion over five years to support this preventive shift which aims to reduce costly hospital utilisation before the demographic peak.[50] Financing Architecture – The ‘3M’ Framework Singapore financing structure combines three complementary mechanisms. Medisave provides mandatory individual health savings contributions of roughly 8 to 10.5 percent of income. MediShield Life provides universal catastrophic insurance coverage. MediFund operates as a public safety net for patients unable to meet residual costs. This layered design distributes responsibility across individuals’ insurance pools and government while containing fiscal risk and limiting moral hazard.[51] Workforce Longevity Policy - Progressive Retirement Reform To mitigate labour shortages associated with ageing the government has progressively extended workforce participation. The statutory retirement age has been raised to 63 with a planned increase to 65 by 2030 while the re-employment age will rise to 70. Labour force participation among adults aged 65 to 69 increased from 31.4 percent in 2013 to 43.3 percent in 2023 which demonstrates that policy incentives and regulatory reform can significantly extend productive ageing.[52] Digital Health Infrastructure Singapore has established national digital health infrastructure through the National Electronic Health Record system which integrates clinical data across the healthcare system. The Digital Health Blueprint supports nationwide interoperability and data governance. AI enabled diagnostic tools for conditions such as diabetic retinopathy and stroke detection are being deployed while robotic assistants have been piloted in community and long-term care environments. These systems collectively represent operational foundations for the Integrated Perception pillar of the AAF 3.0 framework.[53] Whole of Government Governance – APSA 2023 The Action Plan for Successful Ageing coordinates more than 200 initiatives across health housing, employment, transport and community participation. Active Ageing Centers operate nationwide to support community engagement and care coordination. The Seniors Go Digital initiative has trained more than 200000 older adults in digital literacy which directly addresses digital exclusion risks associated with technology-based healthcare delivery.[54] Implications for Comparative Policy Singapore institutional model is not directly transferable because its city state scale centralised governance and fiscal reserves create enabling conditions that larger federated systems do not possess. Nevertheless, the sequencing of reforms provides a clear policy lesson. Preventive care financing digital infrastructure and cross sector governance were established before demographic pressures reached their peak. In contrast many ageing economies attempt to deploy technological solutions after fiscal and workforce pressures have already intensified. From a NASSS perspective Singapore demonstrates favourable conditions across all six domains including technology readiness value proposition adopter acceptance organisational embedding and system level governance. For larger ageing economies including Japan South Korea Germany and Italy the principal barrier to AI enabled healthcare adoption therefore lies less in algorithmic capability and more in institutional alignment. |
| Country / Region | Digital Health Maturity | Governance Architecture | Strategic Advantage | Structural Constraint | Policy Implication |
| Japan | Very high | Moderately centralised national policy with local implementation | Deep investment in eldercare robotics and monitoring | Severe workforce shortages and fragmented EHR interoperability | Scale automation while prioritising national interoperability standards |
| South Korea | Very high | Highly centralised digital governance | Advanced ICT ecosystem and rapid national deployment capability | Fastest demographic ageing globally compresses policy response window | Accelerate AI assisted care models to offset labour shortages |
| Singapore | Very high | Highly centralised and coordinated governance | Unified national digital health strategy | Small population limits large scale clinical evidence generation | Position as a global regulatory and innovation testbed |
| European Union | High but uneven | Multi-level governance across EU and national authorities | Strong regulatory leadership through the EU AI Act | Fragmented interoperability and extended compliance timelines | Harmonise digital health infrastructure and accelerate cross border data sharing |
| Italy / Southern Europe | Moderate | Decentralised healthcare governance | Demographic urgency creating reform momentum | Fiscal constraints and limited long-term care workforce | Prioritise digital health infrastructure and workforce augmentation through AI |
4. Discussion and Policy Implications for AI Implementation in Ageing Health Systems
4.1. The Central Institutional Thesis
4.2. The Agentic AI Framework (AAF 3.0): Derivation and Differentiation
4.3. Operational Bottlenecks and AI Entry Points in Ageing Health Systems.
4.4. A Sociotechnical Phased Roadmap
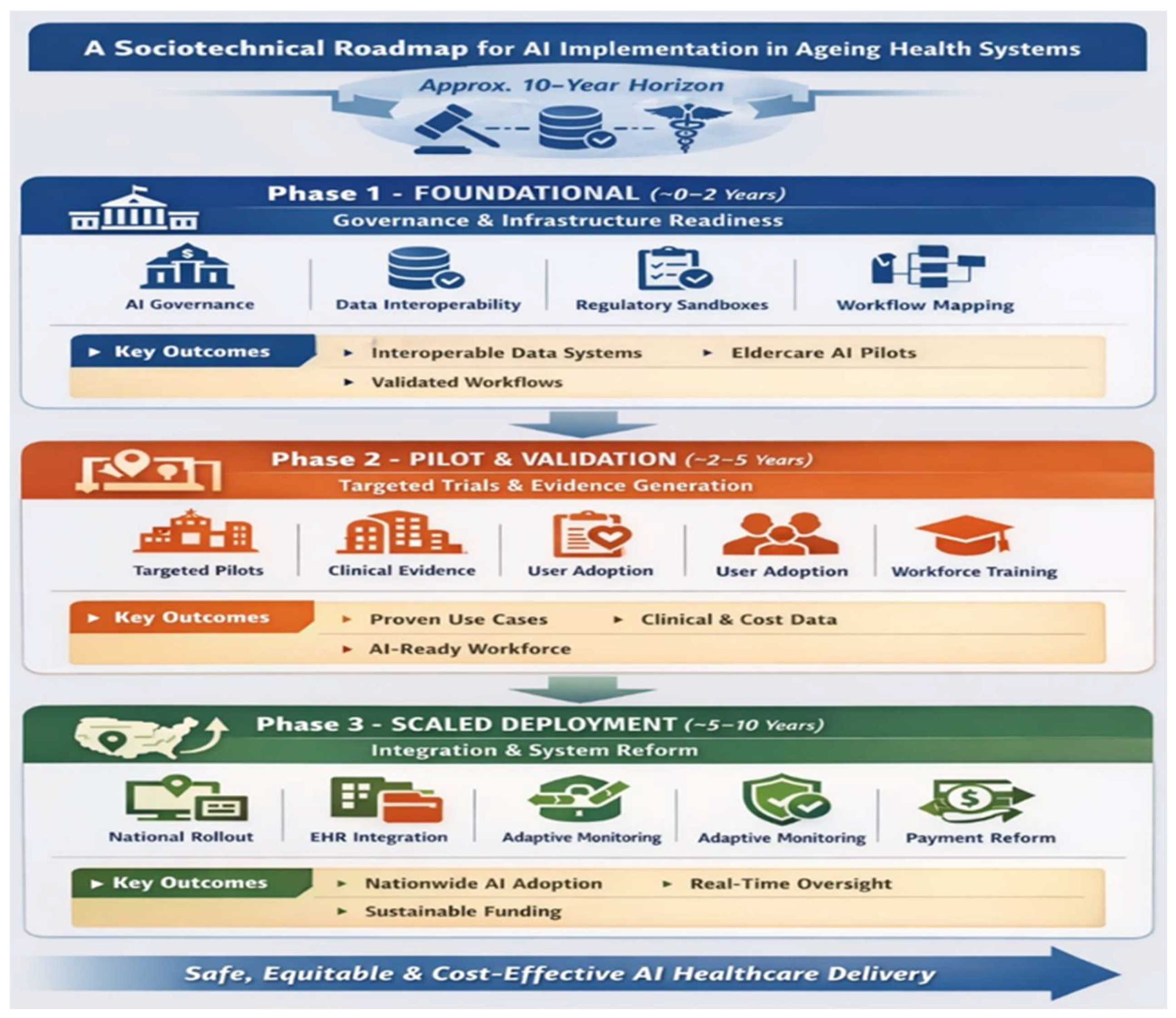
- −
- −
- Data Infrastructure: Mandate HL7 FHIR adoption and secure federated architecture positioning legacy migration as capital investment rather than operating expense[14]
- −
- Regulation: Create regulatory sandboxes for supervised testing of adaptive AI systems and mandate post deployment performance monitoring[34]
- −
- Procurement: Develop AI-specific procurement frameworks requiring interoperability compliance and bias audits.
- −
- Public Engagement: Initiate early co-design with older adults’ caregivers and clinicians to build trust and inform workflow specifications[25]
- −
- Targeted Pilots: Prioritise deployment in structurally constrained settings including rural elderly isolation and long-term care workforce shortages while selecting sites with varying institutional readiness[8]
- −
- −
- Workflow Redesign: Specify required workflow changes prior to deployment and recognise that neutral results without redesign indicate implementation misalignment rather than technological failure
- −
- User Adoption Monitoring: Treat adoption rates, workflow adherence, and clinician override frequency as primary implementation outcomes
- −
- Workforce Development: Integrate AI literacy and human AI collaboration training into professional education and budget training as a core pilot cost[25]
- −
- National Rollout: Link procurement to demonstrate effectiveness and equity appraisal through health technology assessment bodies while sequencing expansion from sites with the highest digital readiness[14]
- −
- Legacy Integration: Require interoperability with dominant national EHR systems as a procurement condition and fund system integration as core health infrastructure
- −
- −
- Payment Reform: Permanently aligned reimbursement with validated AI enabled value-based care models recognising that sustained scale depends on financing reform[9]
4.5. Stakeholder Recommendations
| Stakeholder | Priority Recommendations (Evidence-Derived) |
| Policymakers and Regulators |
1. Replace fixed regulation with adaptive licensing based on a Demographic Urgency Index – A composite index combining dependency ratios, workforce decline, and fiscal capacity should guide regulatory timelines. Countries with higher scores could receive faster approval pathways for eldercare AI, while still requiring post-market monitoring within 12 months. The EU AI Act’s uniform classification system is the main target for reform. (§3.4, §3.6, §4.4) 2. Create sovereign AI-in-health data agreements with shared validation rights – Different privacy laws such as GDPR, PDPA, PIPA, APPI limit cross-border validation of AI systems. Governments should create federated agreements that allow model validation without transferring raw data, including shared audit rights and bias-reporting rules. OECD and WHO should develop a common template. Without such agreements, the evidence base will remain too weak for national-level investment. (§3.4, §4.6, §4.7.3) 3. Replace activity-based reimbursement with Outcome-Linked Technology Contracts – Payment for AI should depend on verified outcomes such as reduced readmissions, delayed institutional care, and clinician time saved. Multi-year contracts should measure these outcomes and be audited annually by an independent health technology body. This approach produces missing economic evidence and aligns commercial incentives with health system sustainability. (§3.3, §3.6, §4.4) |
| Healthcare Systems and Providers |
1. Establish AI Clinical Governance Boards including elder patients and caregivers – Dedicated boards, separate from general informatics committees, should approve AI systems used for patients aged 65+. They should have authority to redesign workflows, suspend poorly performing tools, and request safety reviews. This addresses the governance gap where decisions about replacing or supporting human care are often made by procurement teams rather than clinicians. (§3.2, §4.1, §4.3) 2. Require 90-day shadow deployment before clinical use, with override data reported nationally – Before activation, AI outputs should be reviewed for 90 days but not used in clinical decisions. Override rates and disagreements should be submitted to a national registry. This creates real-world validation data, establishes baseline performance, and triggers review if post-activation results differ significantly. (§3.2, §3.4, §4.4) 3. Replace basic AI literacy training with human-AI teamwork programmes – Training should focus on practical skills: judging when to trust or override AI, maintaining human-led care roles, and monitoring system performance. These competencies should be included in mandatory professional development for geriatricians, nurses, and care coordinators, and required before unit-level deployment approval. (§3.4, §3.5, §4.4) |
| Technology Developers |
1. Publish geriatric-specific model cards showing performance in older populations –Standard model cards hide weaknesses in key groups such as patients aged 80+, people with multiple conditions, and those with cognitive impairment. Developers should report to subgroup performance and training dataset demographics before market entry. This moves bias detection from post-deployment audits to pre-procurement validation. (§3.4, §3.5) 2. Design eldercare AI systems with human-resumable workflows – All multi-step AI processes should allow clinicians to pause, review, or complete tasks manually without losing data. Each handover should generate a plain-language clinical summary. This addresses governance risks in autonomous AI systems and meets EU AI Act human oversight requirements. (§3.4, §4.2, §4.4) 3. Replace proprietary deployment with shared validation partnerships – Real-world performance data should be jointly owned by developers and health systems, with anonymised results published within 24 months. In return, health systems provide controlled access to longitudinal data. This turns commercial deployment into a research partnership and produces the long-term evidence currently missing. (§3.3, §4.6, §4.7.2) |
4.6. Future Research Agenda
4.7. Limitations
4.7.1. Review Methodology Limitations
4.7.2. Implementation and Infrastructure Risks
4.7.3. Global Data Governance Constraints
4.8. Policy Impact and Potential Policy Outcomes
- −
- −
- Clinician administrative burden: Ambient documentation reduces documentation time by at least 15 percent and increases patient facing time[41]. Evidence emerging and mainly observational.
- −
- −
- −
- Healthcare expenditure moderation: If AI interventions reduced projected expenditure growth by 5 to 10 percent relative to IMF projections[46] advanced economies could avoid about 0.1 to 0.35 percentage points of GDP annually by mid-century. Scenario is based on plausible extrapolation from moderate micro level evidence.
- −
- −
- Long-term care costs: Delaying institutionalisation by six to twelve months through ageing in place technologies could generate substantial individual long term care savings[31]. Evidence is moderate at the individual level but dependent on population scale adoption.
- −
- −
- −
- Caregiver burden and rural access: AI enabled telehealth can expand geographic access to care and may reduce caregiving pressure on family networks. Evidence emerges and requires further evaluation.
5. Conclusion
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Appendix A
| Domain | Representative Search Terms |
| Population Ageing | Population ageing, demographic transition, old-age dependency ratio, fertility rate, longevity, elderly population, ageing-in-place, dementia prevalence, healthy ageing, frailty, social isolation in older adults |
| Healthcare System Pressure | Healthcare expenditure, long-term care workforce, eldercare costs, healthcare fiscal sustainability, multimorbidity, geriatric care, caregiver burden, eldercare workforce shortage |
| Artificial Intelligence | Artificial intelligence, agentic AI, machine learning, remote monitoring, assistive robotics, natural language processing, reinforcement learning, digital twin, ambient intelligence, healthcare AI, algorithmic bias, AI ethics |
| Geographic Scope | Japan, Italy, Germany, France, South Korea, Singapore, Portugal, Finland, Greece, Netherlands, OECD nations, EU member states |
Appendix B
| Inclusion Criteria | Exclusion Criteria |
| Published January 2020 – December 2025, with earlier foundational studies included where necessary. | Published before January 2020 (unless foundational) |
| Population ageing, healthcare system pressure, AI in healthcare, or related policy in OECD or advanced economy context | Narrow clinical study unrelated to ageing or AI healthcare integration |
| Peer-reviewed empirical study, systematic review, meta-analysis, or authoritative grey literature (WHO, OECD, UN, national ministries) | Opinion pieces, editorials, or blog posts without empirical basis |
| Available in English or with verified English translation | Unavailable in full text |
| Reports on AI applications in healthcare with outcome or implementation data | AI studies in non-healthcare domains without transferable evidence |
| Classification | Operational Criteria |
| Strong | ≥2 systematic reviews or meta-analyses with low risk of bias (AMSTAR-2 moderate–high), or ≥3 independent RCTs with adequate power for clinically meaningful outcomes |
| Moderate | ≥2 RCTs or well-designed prospective cohort studies; findings consistent across contexts and populations, but with some limitations in generalisability or follow-up duration |
| Emerging | ≥1 controlled study or consistent observational evidence across ≥2 settings; findings directionally consistent but lacking replication at scale |
| Absent | Evidence limited to case reports, expert opinion, commentary, or no peer-reviewed evidence identified in the search |
Appendix C
| Abbreviation | Full Term |
| AI | Artificial Intelligence |
| AAF 3.0 | Agentic AI Framework, version 3.0 |
| CONSORT | Consolidated Standards of Reporting Trials |
| EU | European Union |
| FHIR | Fast Healthcare Interoperability Resources |
| GDP | Gross Domestic Product |
| HIRA | Health Insurance Review & Assessment Service (South Korea) |
| HL7 | Health Level Seven International |
| ICT | Information and Communications Technology |
| LLM | Large Language Model |
| NASSS | Non-adoption, Abandonment, Scale-up, Spread, and Sustainability Framework |
| NEHR | National Electronic Health Record (Singapore) |
| NICE | National Institute for Health and Care Excellence (UK) |
| OECD | Organisation for Economic Co-operation and Development |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| RCT | Randomised Controlled Trial |
| TRIPOD | Transparent Reporting of a Multivariable Prediction Model |
| UN | United Nations |
| WHO | World Health Organization |
Appendix D
| Dimension | Exit Criterion |
| Interoperability | ≥ 1 regional data environment meeting HL7 FHIR compliance across primary, secondary, and long-term care providers |
| Governance | National AI eldercare governance framework published with cross-ministerial mandate and funded secretariat |
| Regulatory | Regulatory sandbox pathway operational with ≥ 2 active supervised AI eldercare pilots enrolled |
| Workflow Mapping | Structured workflow analysis completed for the 3 priority bottleneck domains in ≥ 3 representative care settings |
| Procurement | AI-specific procurement framework published covering modular and integrated platform categories |
| Domain | Clinical Metrics | Operational Metrics | Economic Metrics |
| Ambient Documentation | Clinician-reported documentation accuracy; adverse event documentation completeness | Time-on-documentation reduction ≥ 15%; patient-facing time increase ≥ 10%; adoption rate ≥ 70% at 6 months | Net cost per clinician FTE (training + platform); comparison against baseline documentation costs |
| Discharge Coordination | 30-day readmission rate; medication reconciliation error rate | Care plan completion timeliness; inter-provider handover documentation completeness | Cost-per-readmission prevented; platform vs. manual coordination staffing cost |
| Remote Monitoring | Deterioration detection sensitivity and specificity; emergency department avoidance rate | Sensor adherence rate; alert burden (false positive rate); GP/community nurse response time | Cost-per-hospitalisation avoided; home care cost vs. institutionalisation counterfactual |
Appendix E
| Country | Total Pop 2023 (M) | Pop 65+ (M) | % 65+ (2023) | TFR (2023) | Old-Age Dep. Ratio 2023 | Old-Age Dep. Ratio 2050 (proj.) | % 65+ by 2050 (proj.) | Workforce Trend |
| Japan | 125.7 | 36.6 | 29.1% | 1.20 | 52.1 | 82.6 | 38.7% | −10% by 2030 |
| South Korea | 51.7 | 9.5 | 18.4% | 0.72 | 26.4 | 79.0 | 38.2% | −16% by 2040 |
| Germany | 84.4 | 18.8 | 22.3% | 1.46 | 37.1 | 59.8 | 31.6% | −6M by 2035 |
| Italy | 59.1 | 14.1 | 23.8% | 1.24 | 38.2 | 74.0 | 35.9% | −4M by 2040 |
| France | 68.2 | 14.5 | 21.3% | 1.79 | 35.6 | 52.0 | 29.1% | Moderate decline |
| United States | 335.9 | 58.1 | 17.3% | 1.66 | 27.8 | 40.5 | 23.4% | Slowed by immigration |
| Singapore | 5.9 | 0.93 | 15.8% | 0.97 | 24.2 | 85.0 | 47.0% | −330K by 2030 |
| OECD Average | ~1,380 (38 states) | ~241 | ~17.5% | ~1.63 | ~31.6 | ~48.0 | ~25.0% | Varies by member state |
| Country | Total Health Exp. | Health Exp. % GDP | Per Capita (All) | Per Capita 65+ | Ageing-Related Spend % GDP | Proj. Health % GDP (2050) | Ageing Impact on GDP Growth | Fiscal Risk |
| Japan | $484B | 11.5% | $3,850 | ~$13,500 | ~24% | ~14–15% | −0.8 pp/yr | Very High — Debt 235% GDP |
| South Korea | $183B | 9.7% | $3,540 | ~$12,400 | ~16% | ~13–14% | −0.6 pp/yr | Very High — NPS fund depletion by 2055 projected |
| Germany | $480B | 12.8% | $5,690 | ~$19,900 | ~25% | ~15–16% | −1.2 pp/yr | High — Structural deficit rising; care workforce crisis |
| Italy | $182B | 9.5% | $3,080 | ~$10,800 | ~27% | ~13–14% | −0.9 pp/yr | Critical — Debt 140% GDP; pensions 15.4% GDP |
| United States | $4.86T | 16.9% | $13,500 | ~$19,000 | ~25% | ~22–25% | −0.3 pp/yr | High — Medicare/SS unfunded liabilities exceed $60T |
| Singapore | ~$16B | 6.1% | $2,710 | ~$9,500 | ~10% | ~9–10% | −0.7 pp/yr | Managed — CPF reserves; proactive policy buffers |
| OECD Average | ~$4,986 per capita | ~8.9% | ~$4,986 | ~$17,400 | ~18–20% | ~11–12% | ~−0.5 pp/yr | Varies widely |
References
- United Nations, Department of Economic and Social Affairs, Population Division. (2024). World population prospects 2024. New York: United Nations.
- OECD. (2023). Health at a glance 2023: OECD indicators. Paris: OECD Publishing. https://doi.org/10.1787/7a7afb35-en. [CrossRef]
- OECD. (2024). Society at a glance 2024. Paris: OECD Publishing.
- Statistics Korea. (2024). 2023 birth statistics. Daejeon: Statistics Korea.
- World Health Organization. (2024). World health statistics 2024. Geneva: WHO.
- United Nations. (2019). World population ageing 2019: Highlights. New York: United Nations. [Foundational].
- Kinsella, K., & Phillips, D.R. (2005). Global aging: The challenge of success. Population Bulletin, 60(1), pp.1–40. [Foundational].
- Ministry of Health, Labour and Welfare, Japan. (2023). Annual health, labour and welfare report 2023. Tokyo: MHLW.
- OECD. (2023). Fiscal sustainability and population ageing: OECD economic outlook. Paris: OECD Publishing.
- Livingston, G., et al. (2020). Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. The Lancet, 396(10248), pp.413–446. [CrossRef]
- Vlandas, T. (2024). The political economy of pension reform in France. Journal of European Social Policy, 34(1), pp.12–28.
- AARP Public Policy Institute. (2024). Global aging readiness index 2024. Washington, DC: AARP.
- Wang, L., et al. (2024). A survey on large language model based autonomous agents. Frontiers of Computer Science, 18(6), pp.1–26. [CrossRef]
- Rajpurkar, P., Chen, E., Banerjee, O., & Topol, E.J. (2022). AI in health and medicine. Nature Medicine, 28(1), pp.31–38. [CrossRef]
- European Commission. (2024). The 2024 ageing report: Economic and budgetary projections for EU member states. Luxembourg: Publications Office of the EU.
- Singapore Ministry of Health. (2022). Healthier SG: Envisioning our healthcare system's next bound. Singapore: MOH.
- Esteva, A., et al. (2017). Dermatologist level classification of skin cancer with deep neural networks. Nature, 542(7639), pp.115–118. [Foundational]. [CrossRef]
- Rajpurkar, P., et al. (2017). CheXNet: Radiologist level pneumonia detection on chest X-rays with deep learning. arXiv preprint, arXiv:1711.05225. [Foundational].
- Steventon, A., et al. (2012). Effect of telehealth on use of secondary care and mortality. BMJ, 344, e3874. [Foundational]. [CrossRef]
- Klersy, C., et al. (2009). A meta analysis of remote monitoring of heart failure patients. Journal of the American College of Cardiology, 54(18), pp.1683–1694. [Foundational]. [CrossRef]
- Wada, K., et al. (2008). Robot therapy for dementia patients. Proceedings of WASET, 31, pp.543–548. [Foundational].
- Moyle, W., et al. (2017a). Exploring the effect of companion robots on emotional expression in older adults with dementia. Journal of Gerontological Nursing, 43(5), pp.27–35. [Foundational]. [CrossRef]
- Mesko, B., & Topol, E.J. (2023). The imperative for regulatory oversight of large language models in healthcare. npj Digital Medicine, 6(1), 120.
- Obermeyer, Z., et al. (2019). Dissecting racial bias in an algorithm used to manage the health of populations. Science, 366(6464), pp.447–453. [Foundational].
- Cresswell, K., et al. (2020). NHS Scotland's decision support platform: A formative qualitative evaluation. BMJ Health & Care Informatics, 27(1), e100033. [CrossRef]
- Bjornsson, B., et al. (2020). Digital twins to personalize medicine. Genome Medicine, 12(1), pp.1–4. [CrossRef]
- Raghu, A., et al. (2017). Deep reinforcement learning for sepsis treatment. arXiv preprint, arXiv:1711.09602. [Foundational].
- World Health Organization. (2023). Health and care workforce: The time to act is now. Geneva: WHO.
- Ministry of Health Singapore. (2025). Speech by Mr Ong Ye Kung, Minister for Health, at the MOH Committee of Supply Debate 2025. Singapore: MOH.
- OECD & European Commission. (2024). Health at a glance: Europe 2024. Paris: OECD Publishing.
- OECD. (2025). The economic benefit of promoting healthy ageing and community care. Paris: OECD Publishing.
- OECD. (2024). Adapting health systems to an ageing population. Paris: OECD Publishing.
- OECD. (2024). AI in health: Action plan for responsible deployment. Paris: OECD.
- European Commission. (2024). Regulation laying down harmonised rules on artificial intelligence (EU AI Act). Brussels: European Commission.
- Greenhalgh, T., et al. (2017). Beyond adoption: The NASSS framework. Journal of Medical Internet Research, 19(11). [Foundational].
- CONSORT AI Steering Group. (2020). CONSORT AI extension for clinical trials. Nature Medicine, 26, pp.1364–1374.
- TRIPOD AI Consortium. (2023). TRIPOD AI reporting guideline for prediction models. Available at: https://www.tripod-statement.org/.
- Singapore Ministry of Health. (2023). Action plan for successful ageing. Singapore: MOH.
- Statistics Korea. (2023). Population projections for Korea. Seoul: Statistics Korea.
- Moyle, W., et al. (2017b). Use of a robotic seal as a therapeutic tool to improve dementia symptoms. Journal of the American Medical Directors Association, 18(9), pp.766–773. [Foundational]. [CrossRef]
- Sinsky, C.A., et al. (2021). Metrics for assessing physician activity using electronic health records: The Association of American Medical Colleges approach. Journal of the American Medical Informatics Association, 28(4), pp.840–847.
- Matheny, M.E., et al. (2020). Artificial intelligence in health care: A report from the National Academy of Medicine. JAMA, 323(6), pp.509–510.
- Kelly, C.J., et al. (2019). Key challenges for delivering clinical impact with artificial intelligence. Nature Medicine, 25(1), pp.44–56. [Foundational]. [CrossRef]
- Liu, X., et al. (2019). A comparison of deep learning performance against healthcare professionals. Nature Medicine, 25(6), pp.1009–1015. [Foundational].
- Damschroder, L.J., et al. (2009). Fostering implementation of health services research findings into practice: A consolidated framework for advancing implementation science. Implementation Science, 4(1), 50. [Foundational]. [CrossRef]
- International Monetary Fund (IMF). (2024). Fiscal monitor, April 2024. Washington, DC: IMF.
- Maestas, N., Mullen, K. J., & Powell, D. (2023). The effect of population aging on economic growth, the labor force, and productivity. American Economic Journal: Macroeconomics, 15(2), 306–332. https://doi.org/10.1257/mac.20190196. [CrossRef]
- International Monetary Fund (IMF). (2023). Working paper WP/23/75: The economic impact of ageing populations. Washington, DC: IMF.
- Singapore Ministry of Health (MOH). (2024). Committee of Supply Debate 2024 — Healthcare statistics. Singapore: MOH.
- Ministry of Health Singapore (MOH). (2022). Healthier SG white paper 2022. Singapore: MOH.
- Ministry of Health Singapore (MOH). (2023). Healthcare financing white paper 2023. Singapore: MOH.
- Ministry of Manpower Singapore (MOM). (2023). Labour force in Singapore 2023. Singapore: MOM.
- IHiS (Integrated Health Information Systems). (2023). Digital health blueprint 2023. Singapore: IHiS.
- Ministerial Committee on Ageing. (2023). Action plan for successful ageing 2023 update. Singapore: MCA.
- OECD. (2023). OECD digital government outlook 2023. Paris: OECD Publishing. https://doi.org/10.1787/1a89a067-en. [CrossRef]
- European Commission. (2022). Proposal for a regulation on the European Health Data Space. Brussels: European Commission.
- Personal Information Protection Commission, Republic of Korea. (2023). Personal Information Protection Act (PIPA): Consolidated text as amended 2023. Seoul: PIPC. Available at: https://www.pipc.go.kr/eng/.
| Pressure | Mechanism | Evidence Strength | System Implication | References |
| Age-related expenditure gradient | Per-capita costs rise sharply with age due to multimorbidity, frailty, and long-term care needs | Strong, consistently documented across OECD analyses | Sustained upward fiscal pressure. Demographic ageing alone adds 5–8 percentage points of GDP to public health expenditure by 2060 | [2,9,15] |
| Workforce demand expansion | Growing elderly population increases care demand while working-age population contracts | Strong, supported by workforce projections and policy reports | Persistent staffing shortages. Japan's nursing deficit approaches 250,000 workers by 2026 | [2,8] |
| Infrastructure and service strain | Older patients face longer stays, higher readmissions, and greater long-term care reliance | Moderate, consistent across utilisation studies | Capacity bottlenecks requiring expanded community care models and hospital throughput redesign | [2,32] |
| Care coordination complexity | Multimorbidity requires cross-specialty management across fragmented providers | Strong, documented in geriatric and health systems literature | Increased administrative burden and the need for integrated care pathways and longitudinal coordination | [10,14] |
| Application Domain | System Actors | Multi-Domain Integration | Evidence Classification | Relationship | Note |
| Diagnostic AI (imaging) | Low | Low, single modality | Strong | Strong evidence / low complexity | Benchmark domain |
| Medication adherence tools | Low to Moderate | Low to Moderate | Moderate | Evidence decreases as complexity increases | |
| Remote monitoring / telehealth | Moderate | Moderate | Moderate | Evidence decreases as complexity increases | |
| Workflow automation (ambient documentation) | Moderate | Moderate, single workflow | Emerging | Evidence decreases as complexity increases | |
| Assistive robotics (dementia) | Moderate to High | Moderate | Moderate, mainly specialist settings | Mixed | Ethics complexity high |
| Care coordination (multimorbidity) | High | High, cross-specialty | Emerging to Absent | Weakest evidence at highest complexity | Priority gap |
| Social isolation interventions | High | High | Absent | Inverse relationship strongest | Largest design challenge |
| Country | Proj. Avg GDP Growth 2030–2050 | Health Exp. % GDP | Proj. Health % GDP (2050 est.) | Documented AI Efficiency Gains (from literature) | Illustrative Fiscal Moderation Scenario |
| Japan | 0.5–0.8% p.a. [46] | 11.5% [2] | ~14–15% [9,15] | Remote monitoring reduces avoidable readmissions by 20–30% [19,20]; diagnostic AI reduces imaging costs by 30–40% [17,18,44]; workflow automation reduces documentation time by ~15% [41] | AI-enabled interventions could reduce health spending growth by 5–10% of baseline trajectory, avoiding about 0.5–1.0 percentage points of GDP by 2050 [9,46] |
| Germany | 0.5–1.0% p.a. [46] | 12.8% [2] | ~15–16% [9,15] | DiGA digital therapeutics reimbursement pathway established [16]; AI discharge coordination shows early readmission reduction [19]; ambient documentation reduces administrative burden [41] | AI deployment across LTC and hospitals could reduce spending growth by about 3–7% relative to baseline [9,15] |
| United States | 1.8–2.2% p.a. [46] | 16.9% [2] | ~22–25% [9] | Diagnostic AI reaches specialist-level accuracy [44]; remote monitoring reduces emergency utilisation in chronic disease pilots [19,20]; AI coordination emerging in value-based care [42] | AI-enabled value-based care models could reduce projected Medicare expenditure growth by about 5–10% [33,46] |
| Singapore | 1.5–2.5% p.a. [46,49] | 6.1% [49] | ~9–10% [49] | Healthier SG preventive care model [50]; NEHR-enabled AI diagnostics rollout [53]; robotic eldercare pilots in Active Ageing Hubs [54] | Integrated prevention and digital health strategy aims to keep spending near 9–10% of GDP [46,49,50] |
| Domain | Evidence Status | Outcome Type | Key Limitation | Quality Note |
| Remote monitoring | Established in pilot studies | Reduced admissions and emergency utilisation | Effects vary by context; limited mortality evidence | Steventon 2012 [19] findings qualified by subsequent analyses; see evidence note above |
| Assistive robotics | Established in dementia care settings | Improved mood, agitation, and engagement | Raises ethical questions about replacing human care | |
| Diagnostic AI | Established imaging tasks | Specialist-level diagnostic accuracy | Limited use in complex geriatric clinical reasoning | External validity of benchmark studies limited; see evidence note above |
| Medication adherence | Moderate and mixed evidence | Improved adherence and refill behaviour | Heterogeneous populations and self-reported outcomes | |
| Workflow automation | Early observational evidence | Reduced documentation burden and improved efficiency | Mostly short-term studies |
| Pillar | Pressure Addressed | Present Capability | Near-Term (3–7 yr) | Speculative Horizon | Evidence Status |
| Integrated Perception | Care coordination complexity; information fragmentation | Multimodal EHR + imaging + sensor integration in active study; routine triple-source integration uncommon | Expanded multimodal use in high-acuity settings; broader adoption dependent on HL7 FHIR | Population-scale 'Human Digital Twin' models remain research-based; governance unresolved [26] | Emerging to established in bounded domains; speculative for real-time comprehensive modelling |
| Autonomous Orchestration | Workforce expansion; coordination burden | Ambient documentation deployed with early burden reduction evidence [41]; single-domain scheduling operational | Cross-domain orchestration (2–3 domains) plausible in interoperable systems; full automation without oversight unlikely | Fully autonomous multi-domain coordination requires regulatory structures not yet established | Established for single-domain; emerging for cross-domain; speculative for autonomous authority |
| Adaptive Goal-Seeking | Multimorbidity; static protocol limits | Reinforcement learning explored in sepsis [27]; interpretability and reward challenges persist; no standard eldercare use | Adaptive personalisation plausible; autonomous medication optimisation remains experimental | Autonomous multi-condition optimisation requires advances in interpretability, reward design, and regulation | Experimental in adjacent domains; not established in eldercare; speculative for full optimisation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




