Submitted:
06 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
Background: Unanticipated difficult airways remain a leading cause of anesthesia-related morbidity and mortality, with traditional bedside predictors demonstrating limited sensitivity. Point-of-Care Ultrasound (POCUS) has emerged as a non-invasive adjunct offering real-time visualization and quantitative measurement of airway anatomy. This narrative review, structured according to the Scale for the Assessment of Narrative Review Articles (SANRA), synthesizes current evidence on Point-of-Care Ultrasound (POCUS) as an adjunct for airway evaluation. We explore the sonoanatomy of the upper airway, the utility of ultrasound in predicting difficult laryngoscopy and intubation, its critical role in emergency front-of-neck access (FONA), and the verification of endotracheal tube placement. Furthermore, we discuss the integration of Artificial Intelligence (AI) in image interpretation and the necessity of standardized training curricula. Methods: We systematically searched PubMed/MEDLINE, Scopus, and Web of Science for English-language peer-reviewed studies addressing sonographic airway assessment, including sonoanatomy, prediction of difficult laryngoscopy/intubation, guidance for emergency front-of-neck access (FONA) and endotracheal tube confirmation. Results: POCUS enhances visualization of critical anatomical structures, improves predictive accuracy when combined with clinical assessment, and provides real-time guidance during emergency procedures. Integration of Artificial Intelligence shows promise for automated image interpretation. Conclusions: Airway ultrasound represents a paradigm shift toward personalized, safer airway management. However, standardized training protocols and validation in diverse clinical settings remain essential. Future research should focus on developing evidence-based algorithms integrating POCUS into airway management guidelines.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Literature Search Strategy and Inclusion Criteria
2.2. Selection and Synthesis Approach
2.3. Evidence Level and Bias Considerations
3. Results
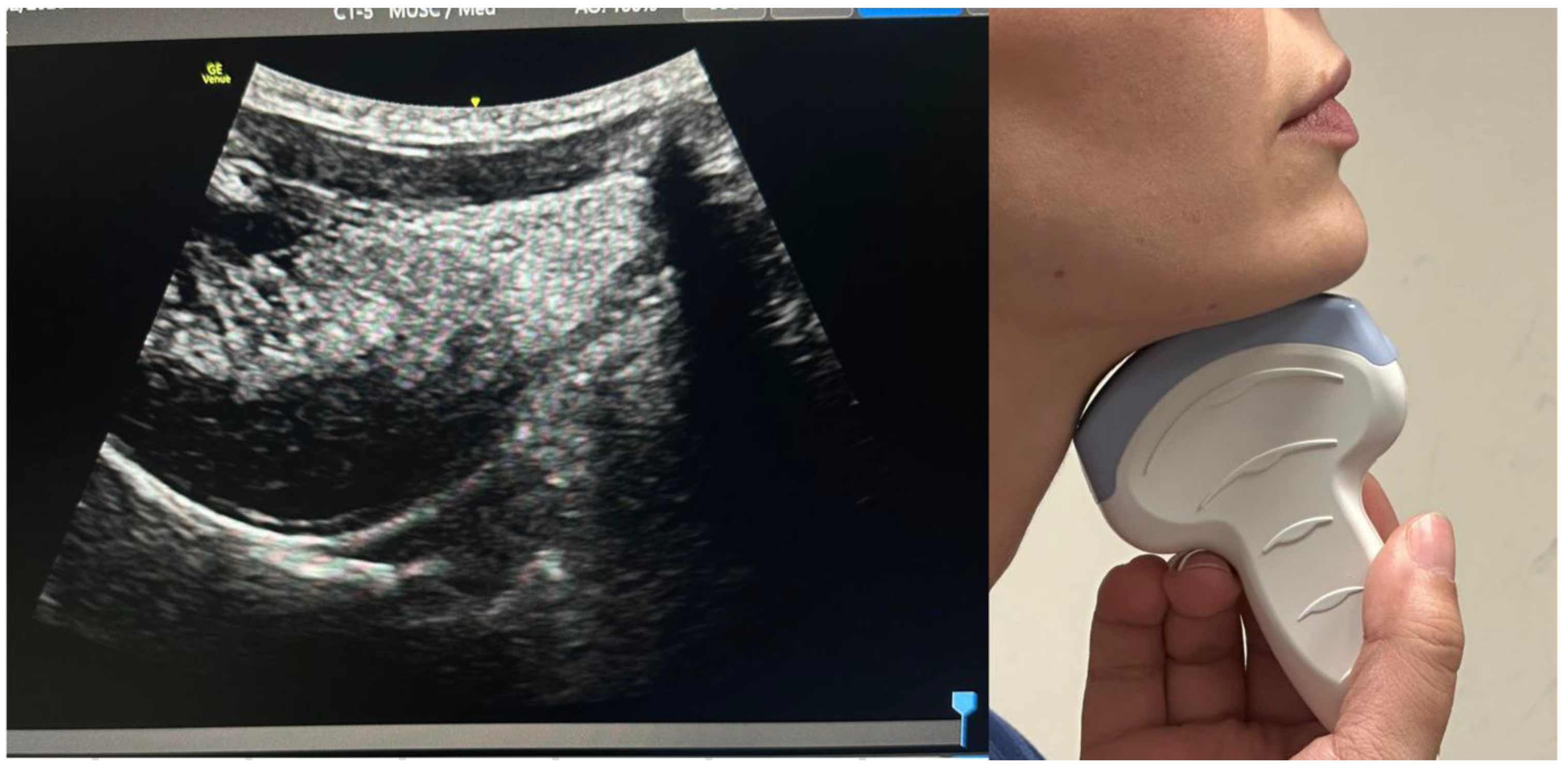
3.1. Ultrasonographic Anatomy of the Upper Airway
3.1. Tongue and Floor of the Mouth
3.2. Epiglottis and Pre-Epiglottic Space
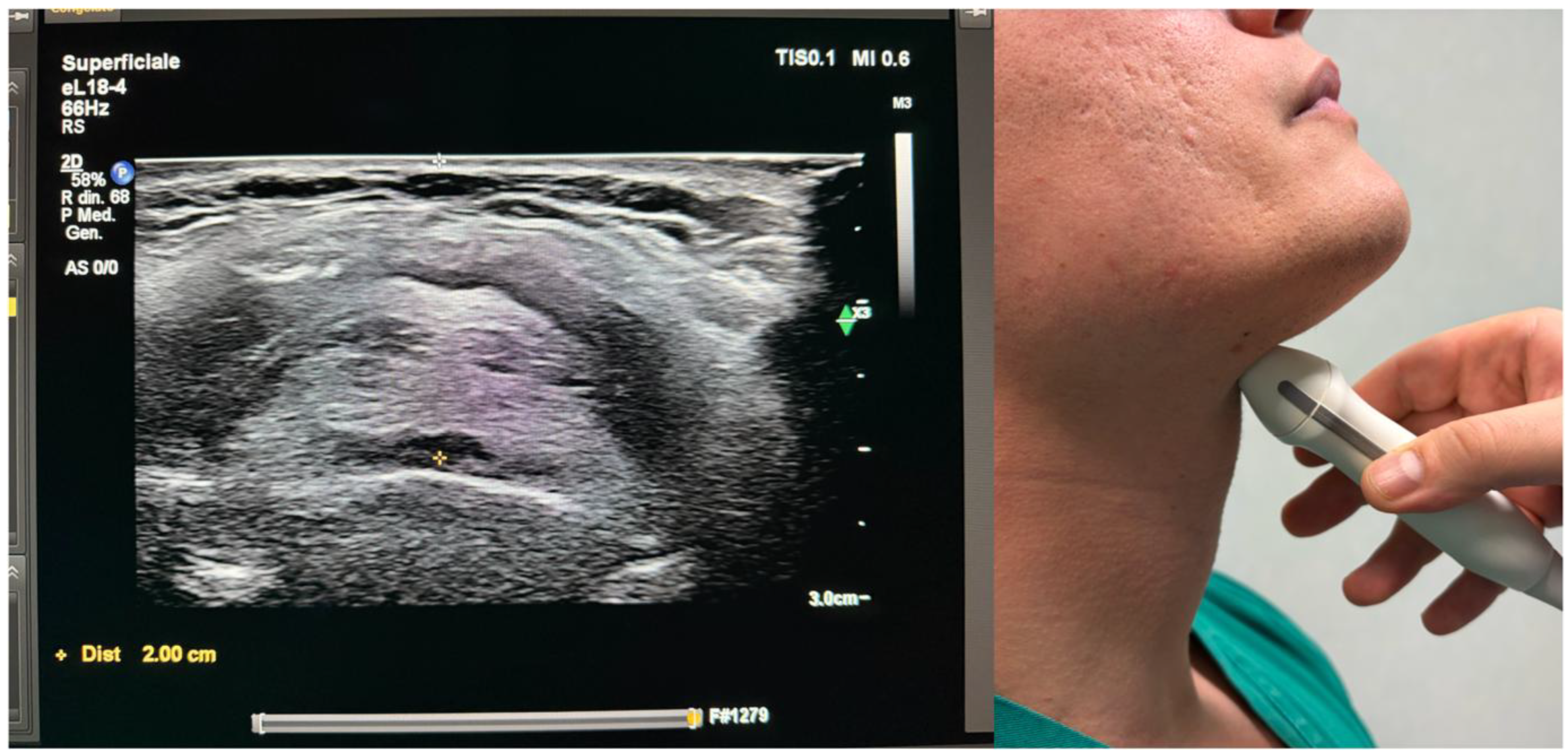
3.3. Hyoid Bone and Laryngeal Cartilages
3.4. Vocal Cords and Glottic Structures
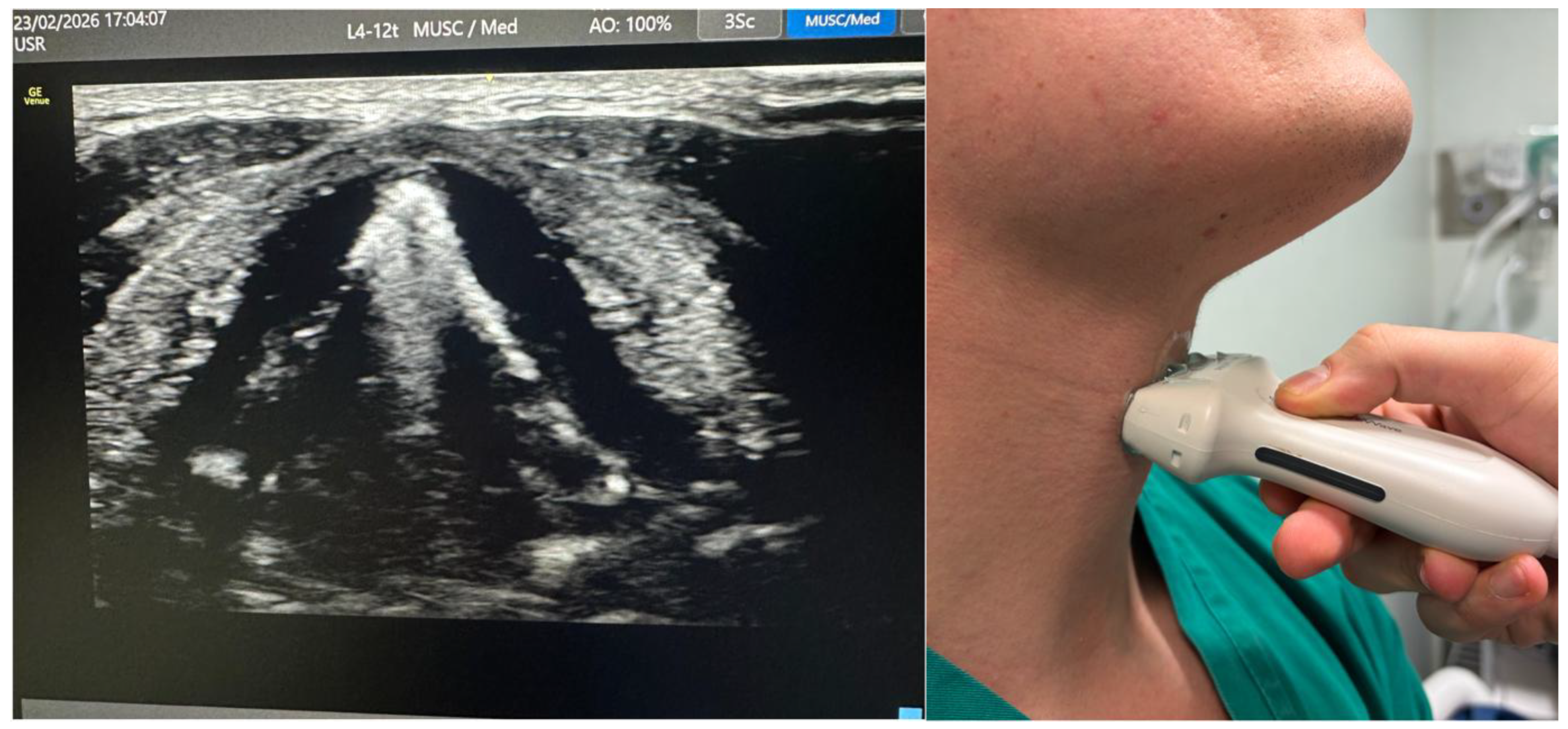
3.5. Trachea and Subglottic Region
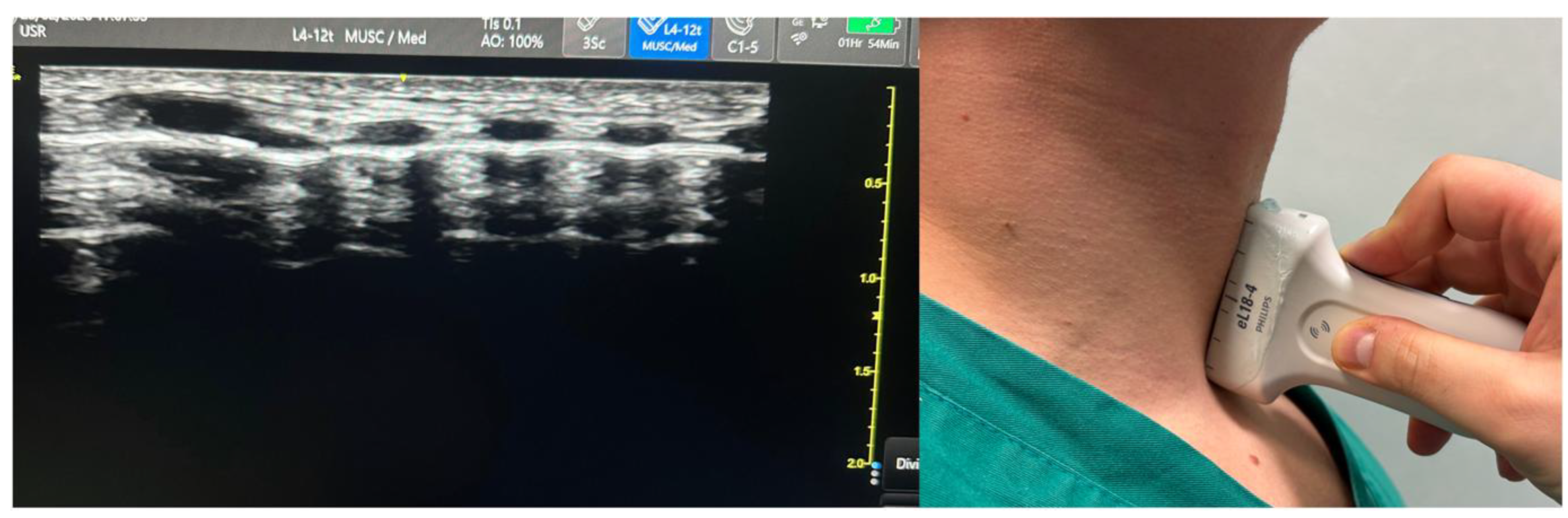
4. Sonographic Predictors of Difficult Intubation
4.1. Quantitative Parameters
4.1.1. Anterior Neck Soft-Tissue Thickness
4.1.2. Hyomental Distance and Ratio
4.1.3. Composite Ultrasound Scores
4.2. Qualitative Markers
4.3. Diagnostic Accuracy Compared with Clinical Prediction Scores
5. Comparative Evidence: US vs Conventional Predictive Tools
5.1. Meta-analyses and Prospective Trials Comparing US and Clinical Assessments
5.2. Integration into Preoperative Screening
6. US-Guided Cricothyroid Membrane Identification
6.1. Techniques and Scanning Protocols
6.2. Accuracy in Normal and Difficult Neck Anatomy
6.3. Role in Emergency Airway Access, Decision-Making
7. Clinical Applications and Training Implications
7.1. Incorporation in Anesthesiology, Emergency Medicine, and Critical Care Practice
7.2. Simulation-Based Education and Credentialing Models
8. Limitations and Controversies
8.1. Operator Variability and Lack of Standardization
8.2. Technological Challenges (Portable Devices, AI-Based Measurement)
9. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Cook, TM; Woodall, N; Frerk, C; Fourth National Audit Project. Major complications of airway management in the UK: results of the Fourth National Audit Project of the Royal College of Anaesthetists and the Difficult Airway Society. Part 1: anaesthesia. Br J Anaesth. 2011, 106(5), 617–31. [Google Scholar] [CrossRef] [PubMed]
- Russotto, V; Sorbello, M. Airway management in critically ill patients. BJA Educ. 2025, 25(9), 375–81. [Google Scholar] [CrossRef] [PubMed]
- Lamperti, M; Romero, CS; Guarracino, F; Cammarota, G; Vetrugno, L; Tufegdzic, B; et al. Preoperative assessment of adults undergoing elective noncardiac surgery: updated guidelines from the European Society of Anaesthesiology and Intensive Care. Eur J Anaesthesiol. 2025, 42(1), 1–35. [Google Scholar] [CrossRef]
- Heidegger, T; Pandit, JJ. Difficult airway management: from the power of prediction to the art of management. Anesth Analg. 2025, 140(2), 290–4. [Google Scholar] [CrossRef]
- Pandit, JJ; Heidegger, T. Putting the 'point' back into the ritual: a binary approach to difficult airway prediction. Anaesthesia 2017, 72(3), 283–8. [Google Scholar] [CrossRef]
- Sorbello, M; Falcetta, S. Time to include ultrasounds in pre-procedural airway evaluation? Trends Anaesth Crit Care 2021, 22, 45–55. [Google Scholar] [CrossRef]
- Lin, J; Bellinger, R; Shedd, A; Wolfshohl, J; Walker, J; Healy, J; et al. Point-of-care ultrasound in airway evaluation and management: a comprehensive review. Diagnostics (Basel) 2023, 13(9), 1541. [Google Scholar] [CrossRef]
- Soni, VM; Pappu, A; Zarabi, S; Khalil, C; You-Ten, KE; Siddiqui, N; et al. Point-of-care ultrasound of the upper airway in difficult airway management: a systematic review and meta-analysis. Anaesthesia 2025, 80(12), 1556–67. [Google Scholar] [CrossRef]
- Carsetti, A; Sorbello, M; Adrario, E; Donati, A; Falcetta, S. Airway ultrasound as predictor of difficult direct laryngoscopy: a systematic review and meta-analysis. Anesth Analg. 2022, 134(4), 740–50. [Google Scholar] [CrossRef] [PubMed]
- Riveros-Perez, E; Avella-Molano, B; Rocuts, A. Airway ultrasound: a narrative review of present use and future applications in anesthesia. Healthcare (Basel) 2025, 13(13), 1502. [Google Scholar] [CrossRef]
- Chrimes, N; Higgs, A; Hagberg, CA; Baker, PA; Cooper, RM; Greif, R; et al. Preventing unrecognised oesophageal intubation: a consensus guideline from the Project for Universal Management of Airways and international airway societies. Anaesthesia 2022, 77(12), 1395–415. [Google Scholar] [CrossRef] [PubMed]
- Amina, S; Rajasree, O; Balakrishnan, M; Philip, FA; George, PS. Efficacy of ultrasound versus video-bronchoscopy in confirming correct placement of i-gel®: a prospective observational study. Indian J Anaesth. 2025, 69(11), 1214–20. [Google Scholar] [CrossRef]
- Pai, SL; Nimma, SR; Beam, WB; VanderWielen, BA; Kalagara, HK; Bettini, LM; et al. Assessment of gastric content using gastric ultrasound in patients on glucagon-like peptide-1 receptor agonists before anesthesia. Anesth Analg Epub ahead of print. 2025. [Google Scholar] [CrossRef]
- De Luis-Cabezón, N; Ly-Liu, D; Renedo-Corcostegui, P; Santaolalla-Montoya, F; Zabala-Lopez de Maturana, A; Herrero-Herrero, JC; et al. A new score for airway assessment using clinical and ultrasound parameters. Front Med (Lausanne) 2024, 11, 1334595. [Google Scholar] [CrossRef]
- Yang, IT; Tung, A; Flores, KS; Berenhaut, KS; Choi, JA; Bryan, YF. Clinical decision-making and process complications during anticipated difficult airway management for elective surgery. Anesth Analg. 2025, 140(2), 295–305. [Google Scholar] [CrossRef]
- Nakazawa, H; Uzawa, K; Tokumine, J; Lefor, AK; Motoyasu, A; Yorozu, T. Airway ultrasound for patients anticipated to have a difficult airway: perspective for personalized medicine. World J Clin Cases 2023, 11(9), 1951–62. [Google Scholar] [CrossRef]
- Baethge, C; Goldbeck-Wood, S; Mertens, S. SANRA—a scale for the assessment of narrative review articles. Res Integr Peer Rev. 2019, 4, 5. [Google Scholar] [CrossRef] [PubMed]
- Nair, AR; Eltohamy, A; Krishnakumar, N. Ultrasound-guided evaluation of airway difficulty in morbid obesity: comparative predictive parameters. Bioinformation 2025, 21(8), 2537–42. [Google Scholar] [CrossRef]
- Hui, CM; Tsui, BC. Sublingual ultrasound as an assessment method for predicting difficult intubation: a pilot study. Anaesthesia 2014, 69(4), 314–9. [Google Scholar] [CrossRef]
- Giordano, G; Alessandri, F; Zulian, A; Bilotta, F; Pugliese, F. Pre-operative ultrasound prediction of difficult airway management in adult patients: a systematic review of clinical evidence. Eur J Anaesthesiol. 2023, 40(5), 313–25. [Google Scholar] [CrossRef]
- Gottlieb, M; Holladay, D; Burns, KM; Nakitende, D; Bailitz, J. Ultrasound for airway management: an evidence-based review for the emergency clinician. Am J Emerg Med. 2020, 38(5), 1007–13. [Google Scholar] [CrossRef]
- Parameshwar, SK; Karna, ST; Waindeskar, V; Kumar, H; Singh, P; Saigal, S. Accuracy of sonographic airway parameters in difficult laryngoscopy prediction: a prospective observational cohort study from Central India. Turk J Anaesthesiol Reanim. 2023, 51(5), 434–42. [Google Scholar] [CrossRef]
- Wojtczak, JA. Submandibular sonography for airway management. J Ultrasound Med. 2012, 31(4), 523–31. [Google Scholar] [CrossRef]
- Nkwocha, O; Enwerem, CJ; Adeniyi, M; Alambrouk, T; Jawad, U; Javed, S; et al. Role of ultrasound in tracheotomy and cricothyrotomy: a research study of midline cervical vasculature in healthy individuals. Cureus 2025, 17(9), e92711. [Google Scholar] [CrossRef]
- Rai, S; Tiku, A; Menon, A; Seth, D; Bajaj, G. Ultrasonographic measurement of glottal area: a potential biomarker study in young normophonic adults. Eur Arch Otorhinolaryngol. 2025, 282(12), 6335–45. [Google Scholar] [CrossRef]
- Falcetta, S; Sorbello, M. Bats, Dracula and Batman: the sixth sense in airway management. Minerva Anestesiol Epub ahead of print. 2021. [Google Scholar] [CrossRef]
- Falcetta, S; Ahmad, I; Paternò, DS; Sorbello, M. Anesthesia management in thyroid surgery. Curr Opin Anaesthesiol. 2025, 38(6), 852–60. [Google Scholar] [CrossRef]
- Richardson, CM; Lam, AS; Nicholas, GE; Wang, X; Sie, KC; Perkins, JA; et al. Tracheal ultrasound for diagnosis of tracheal cartilaginous sleeve in patients with syndromic craniosynostosis. Otolaryngol Head Neck Surg. 2025, 172(1), 307–12. [Google Scholar] [CrossRef] [PubMed]
- Onrubia, X; Frova, G; Sorbello, M. Front of neck access to the airway: a narrative review. Trends Anaesth Crit Care 2018, 22, 45–55. [Google Scholar] [CrossRef]
- Mika, S; Gola, W; Gil-Mika, M; Wilk, M; Misiołek, H. Overview of artificial intelligence in point-of-care ultrasound. New horizons for respiratory system diagnoses. Anaesthesiol Intensive Ther. 2024, 56(1), 1–8. [Google Scholar] [CrossRef] [PubMed]
- La Via, L; Maniaci, A; Gage, D; Cuttone, G; Misseri, G; Lentini, M; et al. Exploring the potential of artificial intelligence in airway management. Trends Anaesth Crit Care 2024, 59, 101512. [Google Scholar] [CrossRef]
- Detsky, ME; Jivraj, N; Adhikari, NK; Friedrich, JO; Pinto, R; Simel, DL; et al. Will this patient be difficult to intubate? The rational clinical examination systematic review. JAMA 2019, 321(5), 493–503. [Google Scholar] [CrossRef]
- Ezri, T; Gewürtz, G; Sessler, DI; Medalion, B; Szmuk, P; Hagberg, C; et al. Prediction of difficult laryngoscopy in obese patients by ultrasound quantification of anterior neck soft tissue. Anaesthesia 2003, 58(11), 1111–4. [Google Scholar] [CrossRef]
- Gomes, SH; Simões, AM; Nunes, AM; Pereira, MV; Teoh, WH; Costa, PS; et al. Useful ultrasonographic parameters to predict difficult laryngoscopy and difficult tracheal intubation—a systematic review and meta-analysis. Front Med (Lausanne) 2021, 8, 671658. [Google Scholar] [CrossRef]
- Saha, S; Rattansingh, A; Viswanathan, K; Saha, A; Martino, R; Yadollahi, A. Ultrasonographic measurement of pharyngeal-airway dimension and its relationship with obesity and sleep-disordered breathing. Ultrasound Med Biol. 2020, 46(11), 2998–3007. [Google Scholar] [CrossRef]
- Petrişor, C; Szabo, R; Constantinescu, C; Prie, A; Trancă, S; Hagău, N. The performance of ultrasound-based quantification of the hyomental distance ratio in predicting difficult airway in anaesthesia: a STARD-compliant prospective diagnostic study. Eur J Anaesthesiol. 2018, 35, 627–8. [Google Scholar] [CrossRef] [PubMed]
- Yao, W; Wang, B. Can tongue thickness measured by ultrasonography predict difficult tracheal intubation? Br J Anaesth. 2017, 118(4), 601–9. [Google Scholar] [CrossRef]
- Wang, B; Yao, W; Xue, Q; Wang, M; Xu, J; Chen, Y; et al. Nomograms for predicting difficult airway based on ultrasound assessment. BMC Anesthesiol. 2022, 22(1), 23. [Google Scholar] [CrossRef]
- Bellizzi, MG; Pace, A; Iannella, G; Maniaci, A; Paternò, DS; Tutino, S; et al. Airway management in obstructive sleep apnea: a comprehensive review of assessment strategies, techniques, and technological advances. Healthcare (Basel) 2025, 13(15), 1823. [Google Scholar] [CrossRef]
- Anushaprasath, U; Kumar, M; Kamal, M; Bhatia, P; Kaloria, N; Paliwal, B; et al. Evaluation and comparison of sonographic difficult airway assessment parameters with clinical airway predictors. J Anaesthesiol Clin Pharmacol. 2024, 40(3), 422–31. [Google Scholar] [CrossRef] [PubMed]
- Benavides-Zora, D; Jaramillo, MC; Townsley, MM; Franco, V; González, S; Hoyos, C; et al. Diagnostic performance of airway ultrasound for the assessment of difficult laryngoscopy: a systematic review and meta-analysis. J Cardiothorac Vasc Anesth. 2023, 37(7), 1101–9. [Google Scholar] [CrossRef] [PubMed]
- Bhargava, V; Rockwell, NA; Tawfik, D; Haileselassie, B; Petrisor, C; Su, E. Prediction of difficult laryngoscopy using ultrasound: a systematic review and meta-analysis. Crit Care Med. 2023, 51(1), 117–26. [Google Scholar] [CrossRef]
- Maniaci, A; Lentini, M; Vaira, L; Lavalle, S; Ronsivalle, S; Rubulotta, FM; et al. The global burden of maxillofacial trauma in critical care: a narrative review of epidemiology, prevention, economics, and outcomes. Medicina (Kaunas) 2025, 61(5), 915. [Google Scholar] [CrossRef]
- Okano, H; Uzawa, K; Watanabe, K; Motoyasu, A; Tokumine, J; Lefor, AK; et al. Ultrasound-guided identification of the cricothyroid membrane in a patient with a difficult airway: a case report. BMC Emerg Med. 2018, 18(1), 5. [Google Scholar] [CrossRef]
- Kristensen, MS; Teoh, WH. Ultrasound identification of the cricothyroid membrane: the new standard in preparing for front-of-neck airway access. Br J Anaesth. 2021, 126(1), 22–7. [Google Scholar] [CrossRef]
- Siddiqui, N; Yu, E; Boulis, S; You-Ten, KE. Ultrasound is superior to palpation in identifying the cricothyroid membrane in subjects with poorly defined neck landmarks: a randomized clinical trial. Anesthesiology 2018, 129(6), 1132–9. [Google Scholar] [CrossRef]
- Rai, Y; You-Ten, E; Zasso, F; De Castro, C; Ye, XY; Siddiqui, N. The role of ultrasound in front-of-neck access for cricothyroid membrane identification: a systematic review. J Crit Care 2020, 60, 161–8. [Google Scholar] [CrossRef] [PubMed]
- Lavelle, A; Drew, T; Fennessy, P; McCaul, C; Shannon, J. Accuracy of cricothyroid membrane identification using ultrasound and palpation techniques in obese obstetric patients: an observational study. Int J Obstet Anesth. 2021, 48, 103205. [Google Scholar] [CrossRef]
- Adhikari, S; Situ-LaCasse, E; Acuña, J; Ray, A; Godwin, M; Blaivas, M. Integration of pre-intubation ultrasound into an airway management course: a novel training program. Indian J Crit Care Med. 2020, 24(3), 179–83. [Google Scholar] [CrossRef]
- Sorbello, M; Parrinello, L; Petrini, F; Frova, G. Ultrasound: not the best 'soundtrack' for a 'cannot ventilate - cannot intubate' scenario. Eur J Anaesthesiol. 2012, 29(6), 295. [Google Scholar] [CrossRef]
- Alerhand, S. Ultrasound for identifying the cricothyroid membrane prior to the anticipated difficult airway. Am J Emerg Med. 2018, 36(11), 2078–84. [Google Scholar] [CrossRef] [PubMed]
- Baker, PA; Dua, G; Iliff, HA. Education in airway management: a narrative review. Br J Anaesth. 2026, 136(1), 308–16. [Google Scholar] [CrossRef] [PubMed]
- Edwards, J; Ahn, D; Alcaraz, D; Chiles, C; Khuu, T; Soni, NJ; et al. Point-of-care ultrasound training among anesthesiology residency programs in the United States. BMC Anesthesiol. 2025, 25(1), 105. [Google Scholar] [CrossRef]
- Sorbello, M; De Jong, A; De Robertis, E; Greif, R. Dubito ergo sum. From philosophy towards prevention of unrecognised oesophageal intubation. Eur J Anaesthesiol. 2023, 40(5), 310–2. [Google Scholar] [CrossRef]
- Gottlieb, M; Alerhand, S; Long, B; Wiant, A. Ultrasound education in emergency medicine residency programs: a national survey. J Ultrasound Med. 2020, 39(5), 1013–9. [Google Scholar] [CrossRef]
- Dinh, VA; Dukes, WS; Prigge, J; Avila, M. Ultrasound integration in undergraduate medical education: comparison of ultrasound proficiency between trained and untrained medical students. J Ultrasound Med. 2015, 34(10), 1819–24. [Google Scholar] [CrossRef]
- Jiang, Y; Xing, F; Wu, Q; Gao, Y; Chen, G; Li, C. Comparison of patient outcomes between video and non-video laryngeal mask airway insertion performed by novices: a prospective randomized controlled study. Front Med (Lausanne) 2025, 12, 1667040. [Google Scholar] [CrossRef]
- Bianchini, A; Felicani, C; Zangheri, E; Bianchin, M; Siniscalchi, A. Point-of-care airway US: a valuable tool in the management of occult over the cuff bleeding and secretions. Ultrasound J 2023, 15(1), 5. [Google Scholar] [CrossRef]
- El Tahan, MR; Tschernko, E; Végh, T; Mourisse, J; Sentürk, M; Lenartova, K; et al. Monitoring in thoracic anesthesia: EACTAIC consensus document. J Cardiothorac Vasc Anesth. Epub ahead of print. 2025, S1053-0770(25)01129-2. [Google Scholar] [CrossRef] [PubMed]
- Sorbello, M; Paternò, DS; Zdravkovic, I; La Via, L. Pharmacological approach to rapid sequence induction/intubation: a contemporary perspective. Curr Opin Anaesthesiol. 2025, 38(4), 369–74. [Google Scholar] [CrossRef]
- Sorbello, M; Morello, G; Pintaudi, S; Cataldo, R. COVID-19: intubation kit, intubation team, or intubation spots? Anesth Analg. 2020, 131(2), e128-30. [Google Scholar] [CrossRef] [PubMed]
- Park, S; Lee, S; Do, HH; Kim, JS; Seo, JS. Effectiveness of limited airway ultrasound education for medical students: a pilot study. Clin Exp Emerg Med. 2019, 6(3), 257–63. [Google Scholar] [CrossRef]
- Hauglum, SD; Crenshaw, NA; Gattamorta, KA; Mitzova-Vladinov, G. Evaluation of a low-cost, high-fidelity animal model to train graduate advanced practice nursing students in the performance of ultrasound-guided central line catheter insertion. Simul Healthc. 2018, 13(5), 341–7. [Google Scholar] [CrossRef]
- Sites, BD; Gallagher, JD; Cravero, J; Lundberg, J; Blike, G. The learning curve associated with a simulated ultrasound-guided interventional task by inexperienced anesthesia residents. Reg Anesth Pain Med. 2004, 29(6), 544–8. [Google Scholar] [CrossRef]
- Shokoohi, H; Armstrong, P; Tansek, R. Emergency department ultrasound improves skillset and knowledge base of internal medicine residents. Cureus 2015, 7(8), e302. [Google Scholar] [CrossRef]
- Haskins, SC; Bronshteyn, Y; Perlas, A; El-Boghdadly, K; Zimmerman, J; Silva, M; et al. American Society of Regional Anesthesia and Pain Medicine expert panel recommendations on point-of-care ultrasound education and training for regional anesthesiologists and pain physicians—part II: recommendations. Reg Anesth Pain Med. 2021, 46(12), 1048–60. [Google Scholar] [CrossRef]
- Quinn, L; Tryposkiadis, K; Deeks, J; De Vet, HCW; Mallett, S; Mokkink, LB; et al. Interobserver variability studies in diagnostic imaging: a methodological systematic review. Br J Radiol. 2023, 96(1148), 20220972. [Google Scholar] [CrossRef] [PubMed]
- Schnittger, T; Breckwoldt, J; Schilling, T; Hinkelbein, J. Ultrasound in anaesthesia: European perspective and training recommendations. Eur J Anaesthesiol. 2020, 37(8), 633–41. [Google Scholar] [CrossRef]
- Ramsingh, D; Rinehart, J; Kain, Z; Strom, S; Canales, C; Alexander, B; et al. Impact assessment of perioperative point-of-care ultrasound training on anesthesiology residents. Anesthesiology 2015, 123(3), 670–82. [Google Scholar] [CrossRef]
- Hall, EA; Showaihi, I; Shofer, FS; Panebianco, NL; Dean, AJ. Ultrasound evaluation of the airway in the ED: a feasibility study. Crit Ultrasound J 2018, 10(1), 3. [Google Scholar] [CrossRef]
- Baloescu, C; Bailitz, J; Cheema, B; Gorenstein, L; Hetherington, T; Shah, P; et al. Artificial intelligence–guided lung ultrasound by nonexperts. JAMA Cardiol. 2025, 10(3), 245–53. [Google Scholar] [CrossRef]
- Tessaro, MO; Salant, EP; Arroyo, AC; Haines, LE; Dickman, E. Tracheal rapid ultrasound saline test (TRUST) for confirming correct endotracheal tube depth in children. Resuscitation 2015, 89, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Lakhal, K; Delplace, X; Cottier, JP; Tranquart, F; Sauvagnac, X; Mercier, C; et al. The feasibility of ultrasound to assess subglottic diameter. Anesth Analg. 2007, 104(3), 611–4. [Google Scholar] [CrossRef] [PubMed]
- Varaganti, M; Vankayalapati, A; Awad, N; Dion, GR; Brattain, LJ. T2ID-CAS: diffusion model and class aware sampling to mitigate class imbalance in neck ultrasound anatomical landmark detection. Annu Int Conf IEEE Eng Med Biol Soc. 2025, 2025, 1–6. [Google Scholar] [CrossRef] [PubMed]
| Domain | Key Findings | Clinical Impact | Evidence Base |
| STRENGTHS | |||
| Objective, Dynamic Assessment | Provides real-time visualization of airway structures (tongue, epiglottis, hyoid, larynx, vocal cords) with quantifiable anatomical measurements during positioning and breathing | • Eliminates inter-observer variability inherent to subjective clinical tests (e.g., Mallampati) • Captures dynamic changes in neck position and tissue compliance not detectable by static bedside examination |
High-frequency US enables real-time assessment; contrast with Mallampati's static, subjective grading |
| Superior Diagnostic Performance | Higher sensitivity (76–86%) and comparable specificity (77–86%) versus conventional predictors; Area Under ROC Curve (AUROC) 0.83–0.87 for key parameters (DSE, HMDR) | • Improves detection of at-risk airways compared to traditional tests (Mallampati sensitivity ~38–52%) • May reduce unanticipated difficult intubations when integrated with clinical assessment |
Multiple meta-analyses; superior sensitivity over Mallampati, thyromental distance, upper lip bite test |
| Practical Accessibility | Non-invasive, portable, bedside technique requiring no ionizing radiation or patient transport | • Enables rapid point-of-care decision-making in preoperative, emergency, and critical care settings • Compact POCUS devices facilitate immediate airway evaluation |
Widespread availability of handheld US; proven feasibility in ED and OR settings |
| LIMITATIONS | |||
| Lack of Standardization | Marked heterogeneity in: probe type (linear vs. curvilinear), scanning planes (transverse vs. sagittal), measurement definitions (e.g., DSE cut-offs range 1.6–2.75 cm), and patient populations (elective vs. emergency, obese vs. non-obese) | • Prevents direct comparison across studies • Precludes definitive meta-analytic conclusions • Hinders development of universal clinical protocols and guidelines |
Systematic reviews highlight inconsistent methodology; no consensus on optimal scanning protocol or thresholds |
| Operator Dependence | Image quality and measurement accuracy strongly influenced by: user experience, probe pressure/angle, technique, and patient anatomy (obesity, short neck, pathology) | • Requires specialized training with steep learning curve • Inter-operator reliability varies without standardized competency assessment • Reduced accuracy in anatomically challenging patients |
Inter-rater ICC ranges from 0.57–0.88; inexperienced operators yield unreliable data |
| Population Variability | Performance of certain parameters (e.g., anterior neck soft-tissue thickness) inconsistent across demographic groups (sex, BMI, ethnicity) and clinical contexts | • Measurement thresholds validated in one population may not generalize to others • Limited data in emergency, distorted anatomy, or pediatric airways |
Few studies include obese, emergency, or anatomically variant cohorts; most focus on elective adult populations |
| Limited Outcome Evidence | Most studies use surrogate endpoints (Cormack–Lehane grade ≥3) rather than patient-centered outcomes (intubation success, complications, hypoxemia, mortality) | • Unclear whether improved prediction translates to better clinical outcomes • Small sample sizes (<200 patients in most trials) limit external validity • Few multicenter or emergency airway datasets |
Evidence base dominated by single-center observational studies; lack of outcome-driven RCTs |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




