Submitted:
07 March 2026
Posted:
09 March 2026
You are already at the latest version
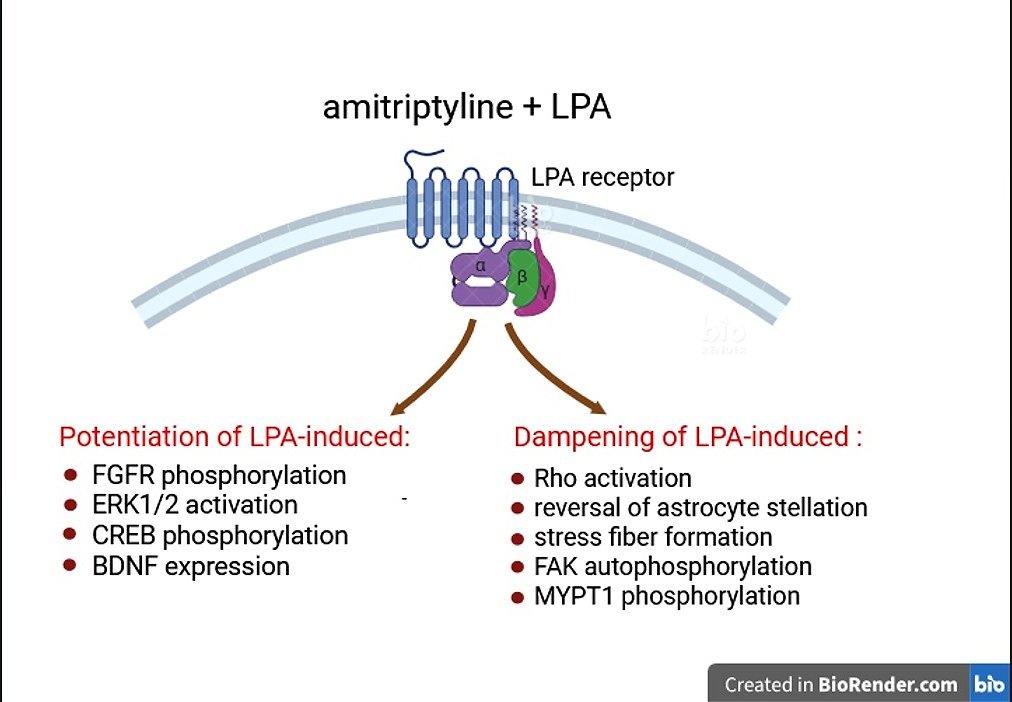
Abstract
Keywords:
1. Introduction
2. Results
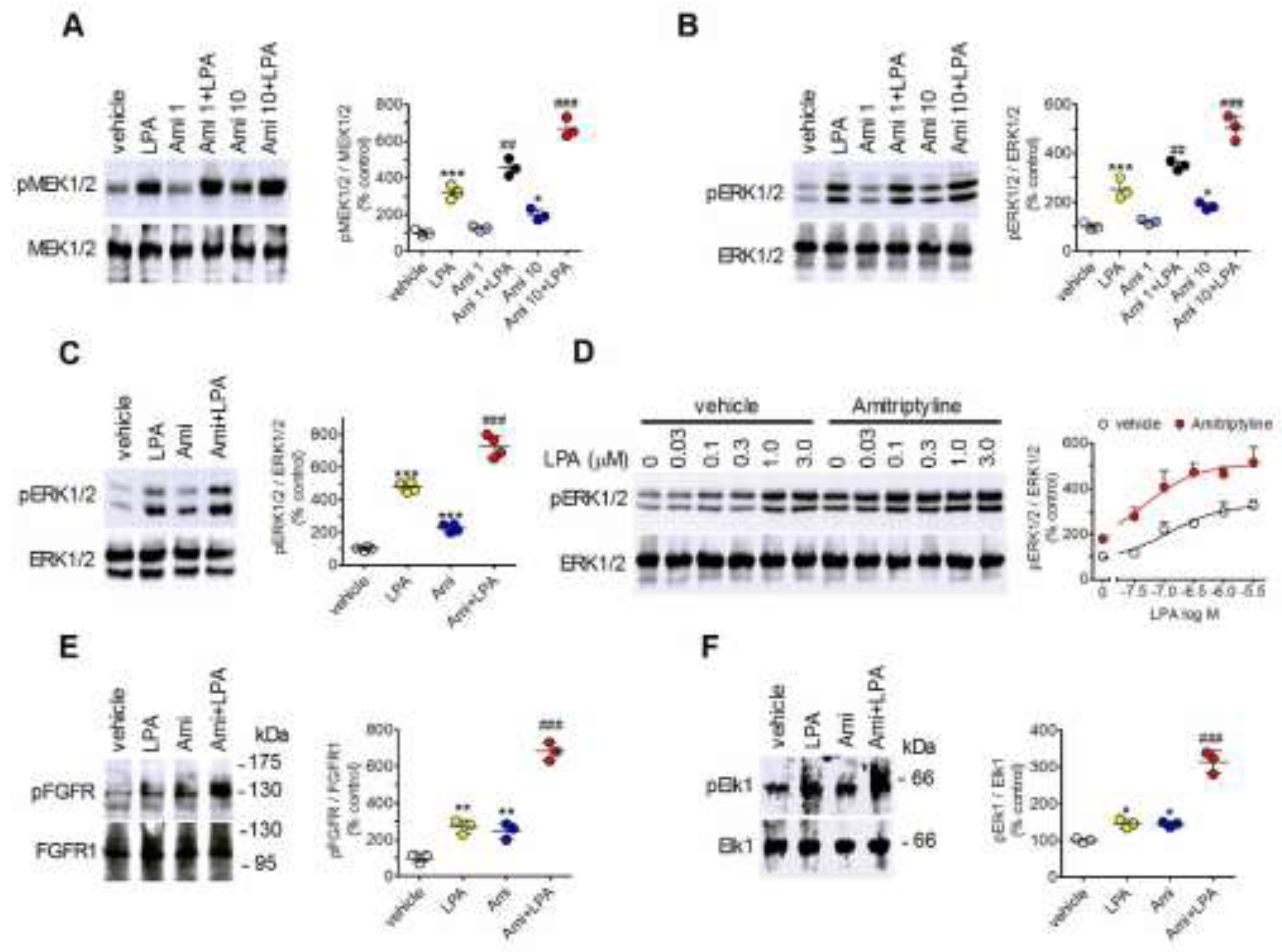
2.1. Amitriptyline Potentiates LPA-Induced ERK1/2 Signaling in Astroglial Cells
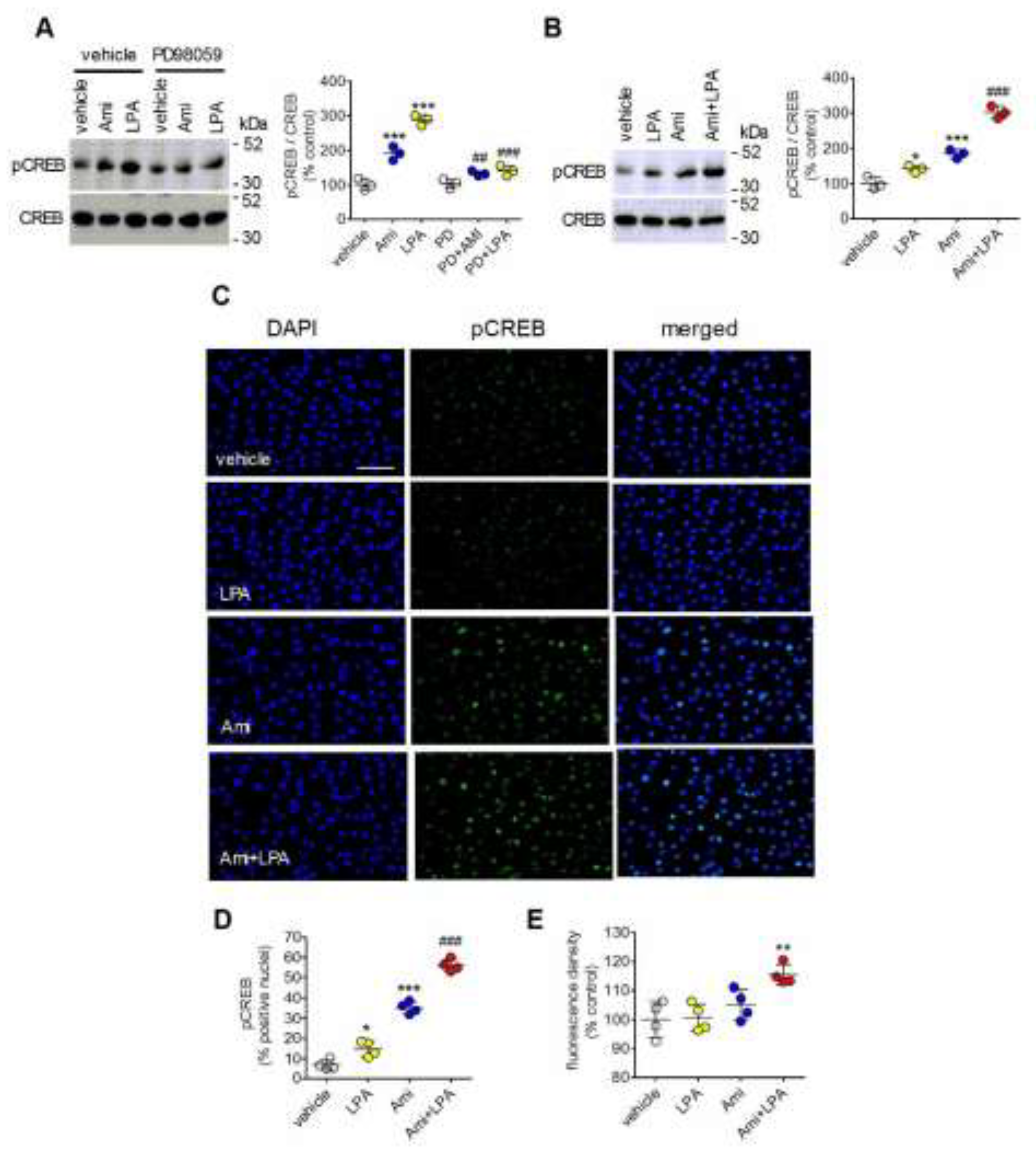
2.2. Synergistic Interaction Between Amitriptyline and LPA in Inducing CREB Phosphorylation
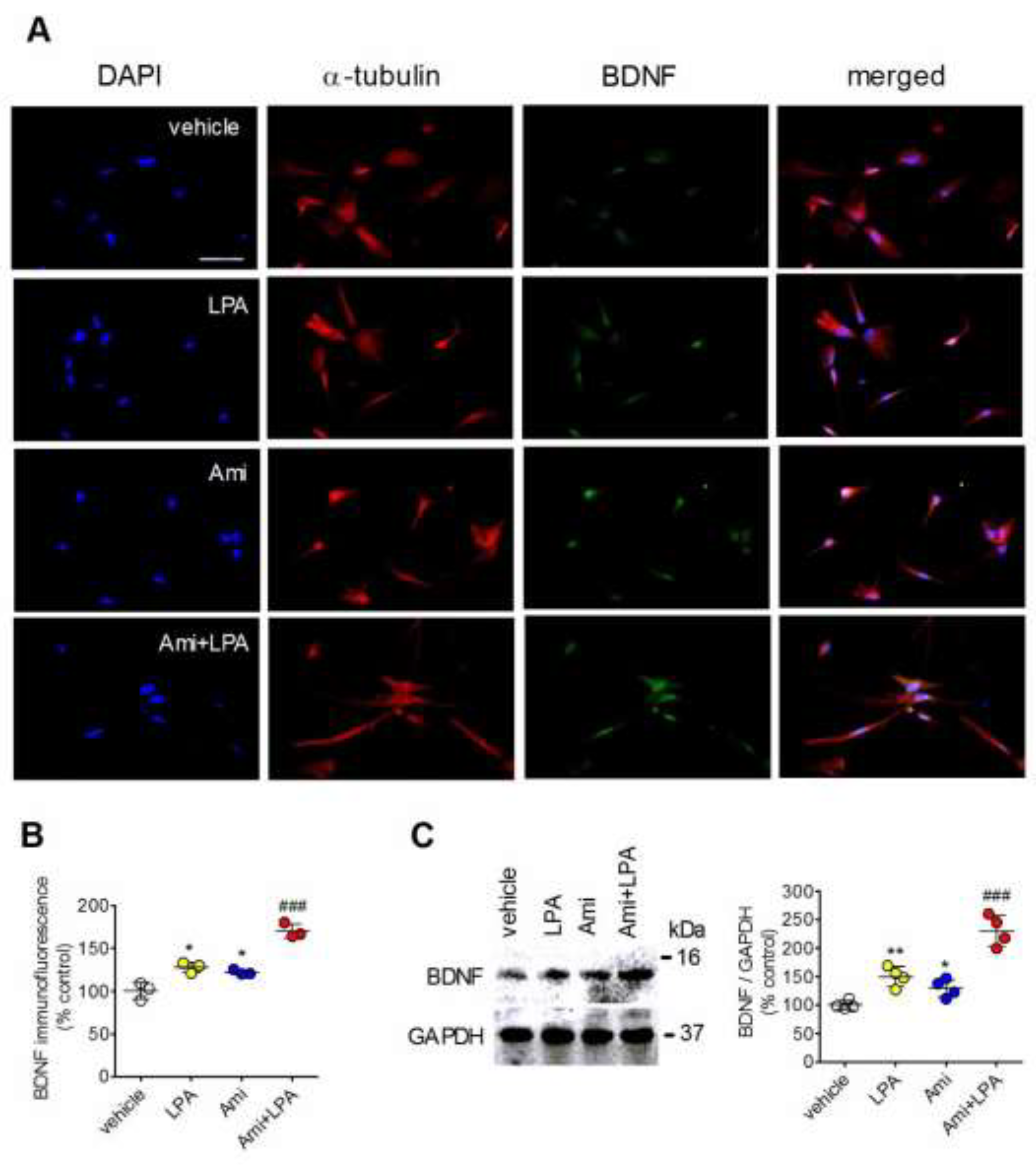
2.3. Amitriptyline Up-Regulates LPA Induction of BDNF Expression
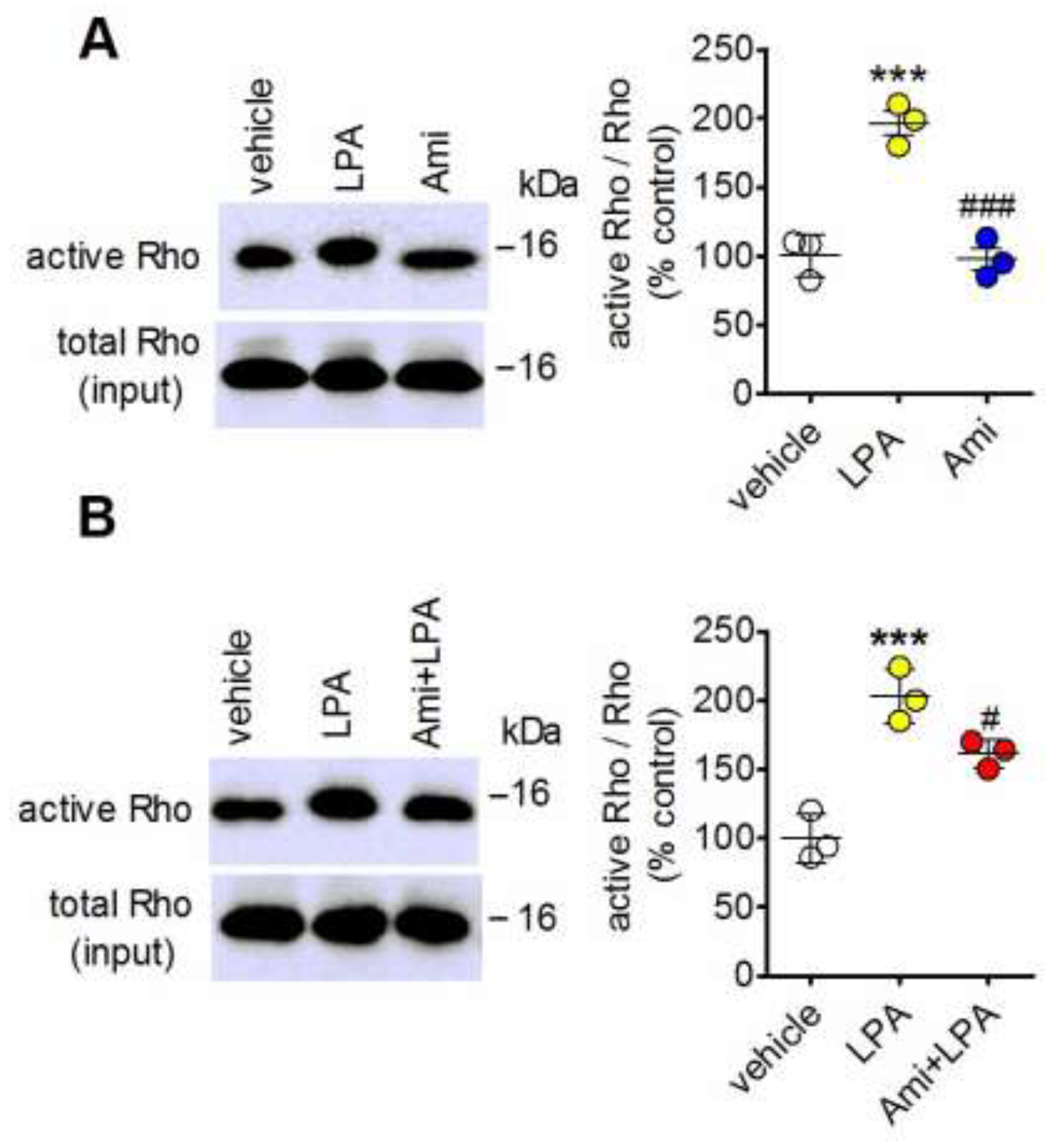
2.4. Amitriptyline Fails to Stimulate Rho and Inhibits Rho Activation Induced by LPA
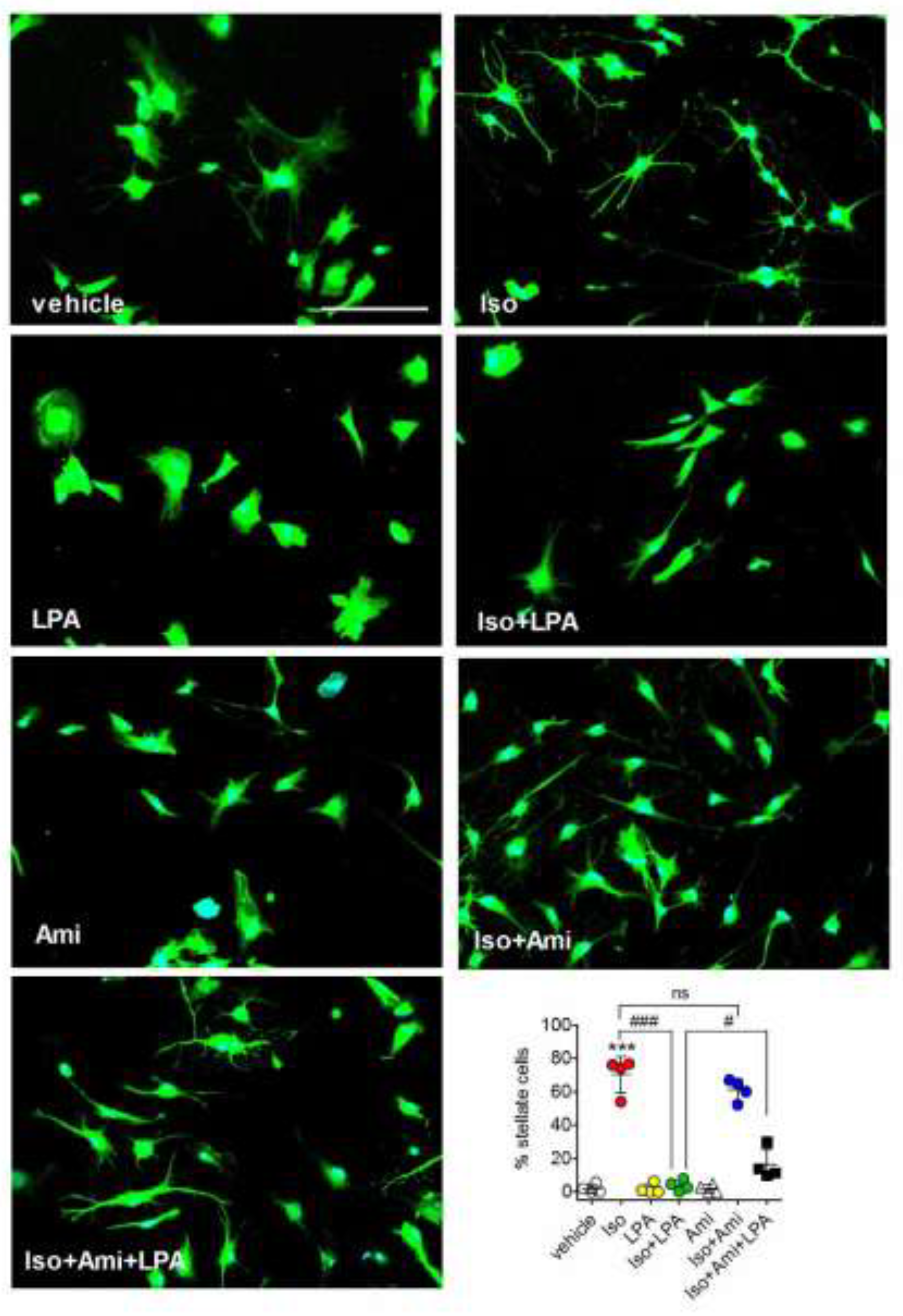
2.5. Amitriptyline Impairs LPA Reversal of Isoproterenol-Induced Morphological Changes in Astrocytes
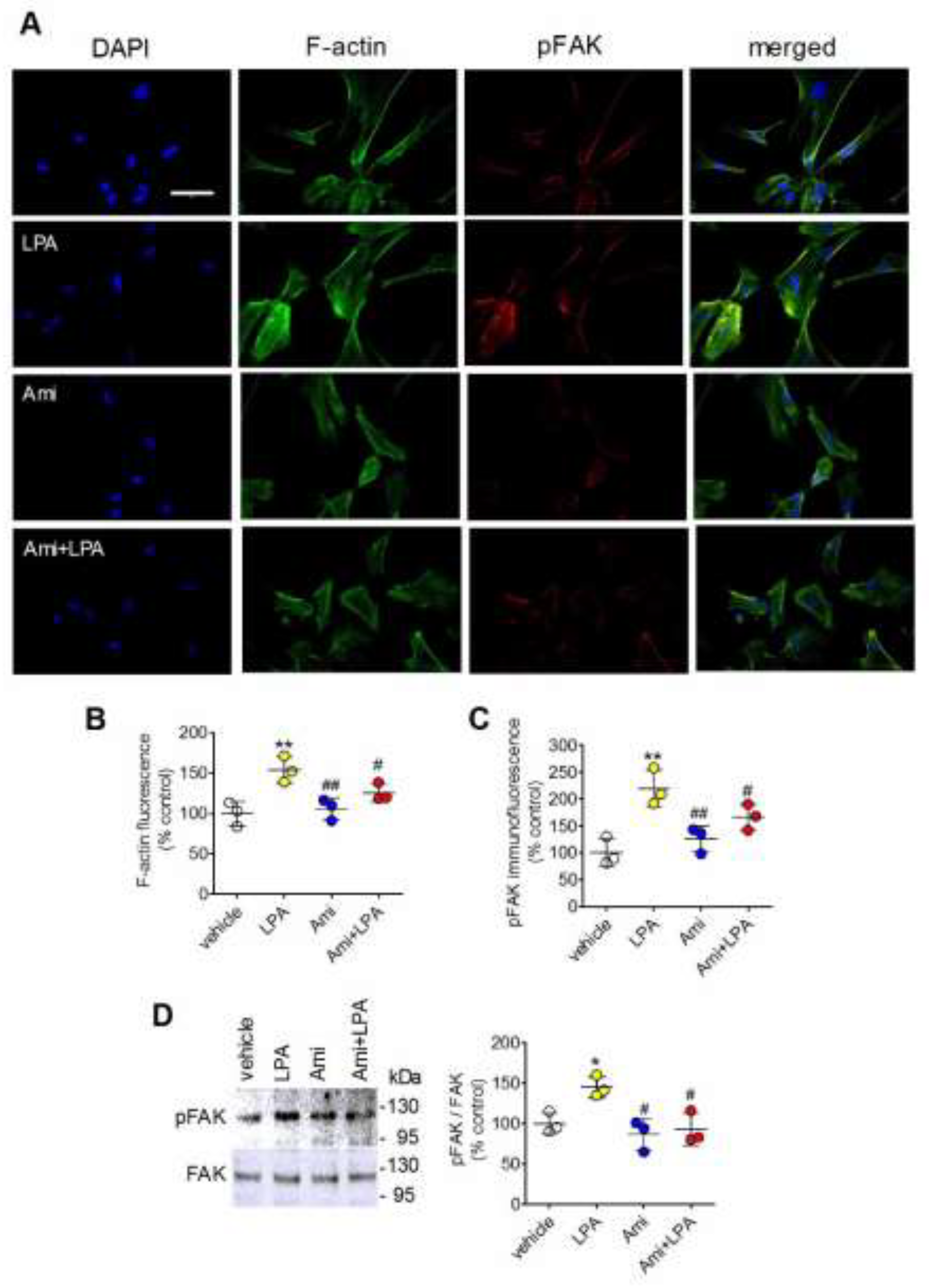
2.6. Amitriptyline Attenuates Stress Fiber Formation and Focal Adhesion Kinase (FAK) Phosphorylation Induced by LPA
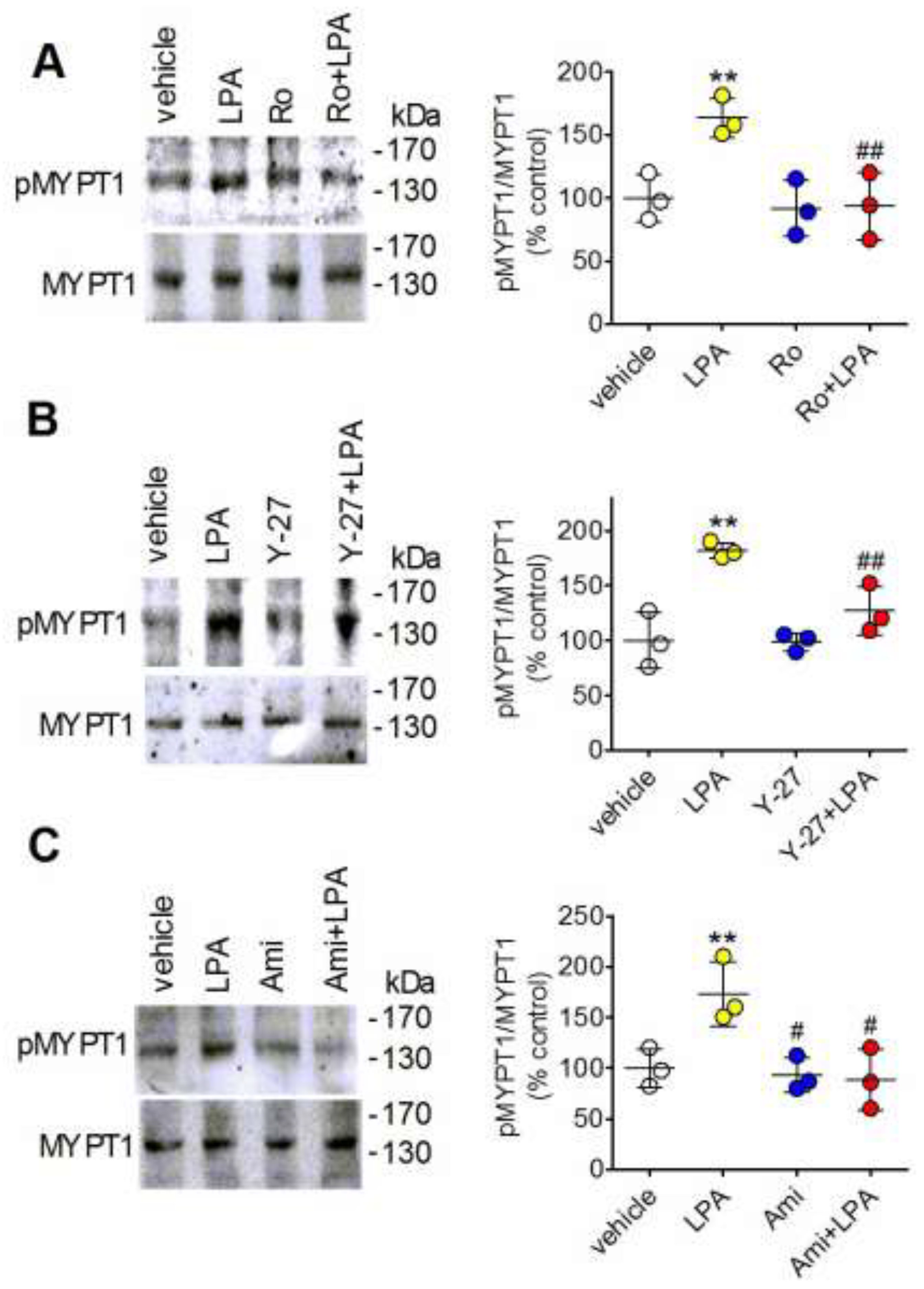
2.7. Amitriptyline Does Not Affect MYPT1 Phosphorylation and Inhibits the Stimulation Induced by LPA
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Culture
4.3. Cell Treatment
4.4. Rho Activation Assay
4.5. Western Blot Analysis
4.6. Immunofluorescence Microscopy
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Harmer, C.J.; Duman, R.S.; Cowen, P.J. How do antidepressant work? New perspectives for refining future treatment approaches. Lancet Psychiatry 2017, 4, 409-418. [CrossRef]
- Gonda, X.; Dome, P.; Neill, J.C.; Tarazi, F.I. Novel antidepressant drugs: Beyond monoamine targets. CNS Spectr. 2023, 28, 6-15. [CrossRef]
- Tigyi, G. Aiming drug discovery at lysophosphatidic acid targets. Br. J. Pharmacol. 2010, 161, 241-270. [CrossRef]
- Kihara, Y.; Maceyka, M.; Spiegel, S.; Chun, J. Lysophospholipid receptor nomenclature review: IUPHAR Review 8. Br. J. Pharmacol. 2014, 171, 3575-3594. [CrossRef]
- Stoddard, N.C.; Chun, J. Promising pharmacological directions in the world of lysophosphatidic acid signaling. Biomol. Ther. 2015, 23, 1-11. [CrossRef]
- Nadhan, R.; Nath, K.; Basu, S.; Isidoro, C.; Song, Y.S.; Dhanasekaran, D.N. Decoding lysophosphatidic acid signaling in physiology and disease; mapping the multimodal and multimodal signaling networks. Signal Transduction and Targeted Therapy 2025, 10:337; [CrossRef]
- Olianas, M.C.; Dedoni, S.; Onali, P. Antidepressants activate the lysophosphatidic acid receptor LPA1 to induce insulin-like growth factor-1 receptor transactivation, stimulation of ERK1/2 signaling and cell proliferation in CHO-K1 fibroblasts. Biochem. Pharmacol. 2015, 95, 311-323. [CrossRef]
- Olianas, M.C.; Dedoni, S.; Onali, P. LPA1 mediates antidepressant-induced ERK1/2 signaling and protection from oxidative stress in glial cells. J. Pharmacol. Exp. Ther. 2016, 359, 340-353. [CrossRef]
- Kajitani, N.; Miyano, K.; Okada-Tsuchioka, M.; Abe, H.; Itagaki, K.; Hisaoka-Nakashima, K.; Morioka, N.; Uezono, Y.; Takebayashi, M. Identification of lysophosphatidic acid receptor 1 in astroglial cells as a target for glial cell-derived neurotrophic factor expression induced by antidepressants. J. Biol. Chem. 2016, 291, 27364-27370. [CrossRef]
- Olianas, M.C.; Dedoni, S.; Onali, P. LPA1 is a key mediator of intracellular signalling and neuroprotection triggered by tetracyclic antidepressants in hippocampal neurons. J. Neurochem. 2017, 143, 183-197. [CrossRef]
- Olianas, M.C.; Dedoni, S.; Onali, P. Inhibition of TNF-α-induced neuronal apoptosis by antidepressants acting through the lysophosphatidic acid receptor LPA1. Apoptosis 2019, 24, 478-498. [CrossRef]
- Banks, D,B,; Chan, G,N,Y,; Evans, R.A.; Miller, D.S.; Cannon, R.E. Lysophosphatidic acid and amitriptyline signal through LPA1R to reduce p-glycoprotein transport at the blood-brain barrier. J. Cereb. Blood Flow Metab. 2018, 38, 857-868. [CrossRef]
- Olianas, M.C.; Dedoni, S.; Onali, P. Antidepressants induce profibrotic responses via the lysophosphatidic acid receptor LPA1. Eur. J. Pharmacol. 2020, 873, 172963. [CrossRef]
- Yung, Y.C.; Stoddard, N.C.; Mirendil, H; Chun, J. Lysophosphatidic acid signaling in the nervous system. Neuron 2015, 85, 669-682. [CrossRef]
- Harrison, S.M.; Reavill, C.; Brown, G.; Brown, J.T.; Cluderay, J.E.; Crook, B.; Davies, C.H.; Dawson, L.A.; Grau, E.; Heidbreder, C; et al. LPA1 receptor-deficient mice have phenotypic changes observed in psychiatric disease. Mol. Cell. Neurosci. 2003, 24, 1170-1179. [CrossRef]
- Castilla-Ortega, E.; Hoyo-Becerra, C.; Pedraza, C.; Chun, J.; Rodriguez De Fonseca, F.; Estivill-Torrus, G.; Santin, L.J. Aggravation of chronic stress effects on hippocampal neurogenesis and spatial memory in LPA1 receptor knockout mice. PLoS ONE 2011, 6, e25522. [CrossRef]
- Santin, L.J.; Bilbao, A.; Pedraza, C.; Matas-Rico, E.; Lopez-Barroso, D.; Castilla-Ortega, E.; Sanchez-Lopez, J.; Riquelme, R.; Varela-Nieto, I.; de la Villa P.; et al. Behavioral phenotype of maLPA1-null mice: increased anxiety-like behavior and spatial memory deficits. Genes Brain Behav. 2009, 8, 772-784. [CrossRef]
- Moreno-Fernandez, R.D.; Perez-Martin, M.; Castilla-Ortega, E.; Rosell Del Valle, C.; Garcia-Fernandez, M.I.; Chun, J.; Estivill-Torrus, G.; Rodriguez de Fonseca, F.; Santin, L.J.; Pedraza, C. maLPA1-null mice as an endophenotype of anxious depression. Transl. Psychiatry 2017, 7, e1077.
- Itagaki, K.; Takebayashi, M.; Abe, H.; Shibasaki, C.; Kajitani, N.; Okada-Tsuchioka, M:; Hattori, K.; Yoshida, S.; Kunugi, H.; Yamawaki, S. Reduced serum and cerebrospinal fluid levels of autotaxin in major depressive disorder. Int. J. Neuropsychoparmacol. 2019, 22, 261-269. [CrossRef]
- Omori, W.; Kano, K.; Hattori, K.; Kajitani, N.; Okada-Tsuchioka, M.; Boku, S.; Kunugi, H.; Aoki, J.; Takebayashi, M. Reduced cerebrospynal fluid levels of lysophosphatidic acid docosahexaenoic acid in patients with major depressive disorder and schizophrenia. Int. J. Neuropsychoparmacol. 2021, 24, 948-955. [CrossRef]
- Olianas, M.C.; Dedoni, S.; Onali, P. Differential targeting of lysophosphatidic acid LPA1, LPA2, and LPA3 receptor signalling by tricyclic and tetracyclic antidepressants. Eur. J. Pharmacol. 2023, 959, 176064. [CrossRef]
- Mohammadi, M.; Dikic, I.; Sorokin, A.; Burgess, W.H.; Jaye, M.; Schlessinger, J. Identification of six novel autophosphorylation sites on fibroblast growth factor receptor 1and elucidation of their importance in receptor activation and signal transduction. Mol. Cell Biol. 1996, 16, 977-989. [CrossRef]
- Roux, P.P.; Blenis, J. ERK and p38 MAPK-activated protein kinases: a family of protein kinases with diverse biological functions. Microbiol. Mol. Biol. Rev. 2004, 68, 320-344. [CrossRef]
- Chrivia, J.C.; Kwok, R.P.; Lamb, N.; Hagiwara, M.; Montminy, M.R.; Goodman, R.H. Phosphorylated CREB binds specifically to the nuclear protein CBP. Nature 1993, 365, 855-859. [CrossRef]
- Zheng, F.; Zhou, X.; Moon, C.; Wang, H. Regulation of brain-derived neurotrophic factor expression in neurons. Int. J. Physiol. Pathophysiol. Pharmacol. 2012, 4, 188-200.
- Shapiro, D.L. Morphological and biochemical alterations in foetal rat brain cells cultured in the presence of monobutyryl cyclic AMP. Nature 1973, 241, 203-204. [CrossRef]
- Tas, P.W.; Koschel, K. Thrombin reverts the β-adrenergic agonist-induced morphological response in rat glioma C6 cells. Exp. Cell Res. 1990, 189, 22-27. [CrossRef]
- Manning, T.J.; Sontheimer, H. Bovine serum albumin and lysophosphatidic acid stimulate calcium mobilization and reversal of cAMP-induced stellation in rat spinal cord astrocytes. Glia 1997, 20, 163-172. [CrossRef]
- Suidan, H.S.; Nobes, C.D.; Hall, A.; Monard, D. Astrocyte spreading in response to thrombin and lysophosphatidic acid is dependent on the Rho GTPase. Glia 1997, 21, 244-252. [CrossRef]
- Ramakers, G.J.A.; Moolenaar, W.H. Regulation of astrocyte morphology by RhoA and lysophosphatidic acid. Exp. Cell Res. 1998, 245, 252-262. [CrossRef]
- Holtje, M; Hoffmann, A.; Hofmann, F.; Mucke, C.; Groβe, G.; Van Rooijen, N.; Kettenmann, H.; Just, I.; Ahnert-Hilger, G. Role of Rho GTPase in astrocyte morphology and migratory response during in vitro wound healing. J. Neurochem. 2005, 95, 1237-1248. [CrossRef]
- Ridley, A.; Hall, A. The small GTP-binding protein rho regulates the assembly of focal adhesions and actin stress fibers in response to growth factors. Cell 1992, 70, 389-399. [CrossRef]
- Chrzanowska-Wodnicka, M.; Burridge, K. Rho-stimulated contractility drives the formation of stress fibers and focal adhesions. J. Cell Biol. 1996, 133, 1403-1415. [CrossRef]
- Burridge, K.; Wennerberg, K. Rho and Rac take center stage. Cell 2004, 116, 167-179. [CrossRef]
- Zeug, A.; Muller, F.E.; Anders, S.; Herde, M.K.; Minge, D.; Ponimaskin, E.; Henneberger, C. Control of astrocyte morphology by Rho GTPases. Brain Res. Bull. 2018, 36, 44-53. [CrossRef]
- Ridley, A.; Hall, A. Signal transduction pathways regulating Rho-mediated stress fiber formation: requirement for a tyrosine kinase. EMBO J. 1994,13, 2600-2610. [CrossRef]
- Katoh, K. Activation of Rho-kinase and focal adhesion kinase regulates the organization of stress fibers and focal adhesions in the central part of fibroblasts, PeerJ 2017 5:e4063; [CrossRef]
- Ito, M.; Nakano, T.; Erdodi, F.; Hartshorne, D.J. Myosin phosphatase: structure, regulation and function. Mol. Cell Biochem. 2004, 259, 197-209. [CrossRef]
- Birukova, A.A.; Smurova, K.; Birukov, K.G.; Usatyuk, P.; Liu, F.; Kaibuchi, K.; Ricks-Cord, A.; Natarajan, V.; Alieva, I.; Garcia, J.G.N.; et al. Microtubule disassembly induces cytoskeletal remodeling and lung vascular barrier dysfunction: role of Rho-dependent mechanisms. J. Cell Physiol. 2004, 201, 55-70. [CrossRef]
- Shano, S.; Moriyama, R; Chun, J.; Fukushima, N. Lysophosphatidic acid stimulates astrocyte proliferation through LPA1. Neurochem. Int. 2008, 52, 216-220. [CrossRef]
- Czeh, B.; Di Benedetto, B. Antidepressants act directly on astrocytes: evidences and functional consequences. Eur. Neuropsychopharmacol. 2013, 23, 171-185. [CrossRef]
- Sanacora, G.; Banasr, M. From pathophysiology to novel antidepressant drugs: glial contributions to the pathology and treatment of mood disorders. Biol. Psychiatry 2013, 73, 1172-1179. [CrossRef]
- Frizzo, M.E.; Ohno, Y. Perisinaptic astrocytes as a potential target for novel antidepressant drugs. J. Pharmacol. Sci. 2021, 145, 60-68. [CrossRef]
- Wang, Q.; Jie, W.; Liu, J.-H.; Yang, J.-M.; Gao, T.-M. An astroglial basis of major depressive disorder? ]An overview. Glia 2017, 65, 1227-1250. [CrossRef]
- Lu, C.-L.; Ren, J.; Cao, X. An astroglial basis of major depressive disorder: molecular, cellular, and circuit features. Biol. Psychiatry 2025, 97, 217-226. [CrossRef]
- Pebay, A.; Torrens, Y.; Toutant, M.; Cordier, J.; Glowinski, J.; Tence, M. Pleiotropic effects of lysophosphatidic acid on striatal astrocytes. Glia 1999, 28, 25-33. [CrossRef]
- Blendy, J.A. The role of CREB in depression and antidepressant treatment. Biol. Psychiatry 2006, 59, 1144-1150. [CrossRef]
- Bjorkholm, C.; Monteggia, L.M. BDNF - a key transducer of antidepressant effects. Neuropharmacology 2016, 102, 72-79. [CrossRef]
- Duman, R.S.; Deyama, S; Fogaca, M.V. Role of BDNF in the pathophysiology and treatment of depression: activity dependent effects distinguish rapid acting antidepressants. Eur. J. Neurosci. 2021, 53, 126-139. [CrossRef]
- Almeida Chiareli, R.; Almeida Carvalho, G.; Lemes Marques, B.; Soares Mota, L.; Oliveira-Lima, O.C.; Mello Gomez, O.; Birbrair, A.; Santiago Gomez, R.; Simao, F.; Klempin, F.; et al. The role of astrocytes in the neurorepair process. Front. Cell Dev. Biol. 2021, 9: 665795. [CrossRef]
- Gomez-Casati, M.E.; Murtie, J.C.; Rio, C.; Stankovic, K.; Liberman, M.C.; Corfas, C. Nonneuronal cells regulate synapse formation in the vestibular sensory epithelium via erbB-dependent BDNF expression. Proc. Natl. Acad. Sci. USA 2010, 107, 17005-17010. [CrossRef]
- Vignoli, B.; Battistini, G.; Melani, R.; Blum, R.; Santi, S.; Berardi, N.; Canossa, M. Peri-synaptic glia recycles brain-derived neurotrophic factor for LTP stabilization and memory retention. Neuron 2016, 92, 873-887. [CrossRef]
- Vaz, S.H.; Jorgensen, T.N.; Cristovao-Ferreira, S.; Duflot, S.; Ribeiro, J.A.; Gether, U.; Sebastiao, A.M. Brain-derived neurotrophic factor (BDNF) enhances GABA transport by modulating the trafficking of GABA transporter-1 (GAT-1) from the plasma membrane of rat cortical astrocytes. J. Biol. Chem. 2011, 286, 40464-40476. [CrossRef]
- Rusakov, D.A.; Dityatev, A.E. Brain circuitry outside the synaptic cleft. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2014, 369, (1654):20130591 doi:.
- Kalman, D.; Gomperts, S.N.; Hardy, S.; Kitamura, M.; Bishop, J.M. Ras family GTPases control growth of astrocyte processes. Mol. Biol.Cell 1999, 10,1665-1683. [CrossRef]
- Cho, S.;Muthukumar, A.K.; Stork,52 T.; Coutinho-Budd, J.C.; Freeman, M.R. Focal adhesion molecules regulate astrocyte morphology and glutamate transporters to suppress seizure-like behavior. Proc. Natl, Acad. Sci. USA 2018, 115, 11316-11321. [CrossRef]
- Hanifa, M.; Singh, M.; Randhawa, P.K.; Jaggi, A. S.; Bali, A. A focus on Rho-ROCK signaling pathway: an emerging therapeutic target in depression. Eur. J. Pharmacol. 2023, 946, 175648. [CrossRef]
- Christopoulos, A. Advances in G protein-coupled receptor allostery: from function to structure. Mol. Pharmacol. 2014, 86, 463-478. [CrossRef]
- Suzuki, S.; Tanaka, K.; Kamegawa, A.; Nishikawa, K.; Suzuki, H.; Oshima, A.; Fujiyoshi, Y. Structural insights into the engagement of lysophosphatidic acid receptor 1 with different G proteins. J. Struct. Biol. 2025, 217, 108164. [CrossRef]
- Shpakov, A.O. Allosteric regulation of G-protein-coupled receptors: from diversity of molecular mechanisms to multiple allosteric sites and their ligands. Int. J. Mol. Sci. 2023, 24, 6187. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



