Submitted:
05 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Background
1.2. Treatment Protocol
1.3. Objective
1.4. Relevance to Insurers and Healthcare Providers
2. Materials and Methods
2.1. Data Overview
2.2. Example Data Field Entry2.3. Data Processing for Clinical Analysis
| Time Spent with Patient: 30 minutes (25 minutes face-to-face) Treatment Number: 4 Placement of Electrodes: Bilateral ankles and toes Symptom Intensity (0–10 Scale): Pain: 0, Numbness: 2, Tingling: 0, Burning: 0, Tightness: 3 Functional Limitation Ratings (1–5 Scale):
Patient Changes Since Start of Treatment: Improvement Recent Functional Issues or Achievements (past 3 days): Good |
2.4. Exclusion Criteria
3. Results
3.1. Data Completeness
3.2. Demographics and Treatment
3.3. Symptom Severity at First Visit
3.4. Functional Limitations at First Visit
3.5. Treatment Completion Patterns
3.6. Baseline Symptom and Functional Differences
3.7. Early Non-Response and Treatment Discontinuation
3.8. Efficacy
3.9. Pain
3.10. Numbness
3.11. Tingling
3.12. Burning
3.13. Tightness
| Comparison | n | Mean Change | % Improved | p-value |
|---|---|---|---|---|
| T1 → T2 | 2311 | -0.254 | 28.4 | < 0.001 |
| T1 → T6 | 1825 | -0.530 | 40.1 | < 0.001 |
| T1 → T12 | 1395 | -0.724 | 45.0 | < 0.001 |
3.14. Symptom Trajectories Across 12 Treatments
3.15. Functional Task Outcomes
3.16. Overall Impact
4. Discussion
Recommendations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TENS | Transcutaneous electrical nerve stimulation |
| PNS | Peripheral nerve stimulation |
| NLP | Natural language processing |
| HPI | History of present illness |
| SNRIs | Serotonin-norepinephrine reuptake inhibitors |
| TCAs | Tricycle antidepressants |
| RFA | Radiofrequency ablation |
References
- Hammi, C.; Yeung, B. Neuropathy. In StatPearls [Internet]; StatPearls Publishing: Treasure Island (FL), Jan 2025–. Updated October 15, 2022; Available online: https://www.ncbi.nlm.nih.gov/books/NBK542220/.
- Schenone, A.; Massucco, S.; Schenone, C.; Venturi, C.B.; Nozza, P.; Prada, V.; Pomili, T.; Di Patrizi, I.; Capodivento, G.; Nobbio, L.; Grandis, M. Basic Pathological Mechanisms in Peripheral Nerve Diseases. Int J Mol Sci. 2025, 26, 3377. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Freeman, M.R. Signaling mechanisms regulating Wallerian degeneration. Curr Opin Neurobiol. 2014, 27, 224–31. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sumner, A.J.; Asbury, A.K. Physiological studies of the dying-back phenomenon. Muscle stretch afferents in acrylamide neuropathy. Brain 1975, 98, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Cashman, C.R.; Höke, A. Mechanisms of distal axonal degeneration in peripheral neuropathies. Neurosci Lett. 2015, 596, 33–50. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kaye, A.D.; Armistead, G.; Amedio, L.S.; Manthei, M.E.; Ahmadzadeh, S.; Bernhardt, B.; Shekoohi, S. Evolving Treatment Strategies for Neuropathic Pain: A Narrative Review. Medicina (Kaunas) 2025, 61, 1063. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cavalli, E.; Mammana, S.; Nicoletti, F.; Bramanti, P.; Mazzon, E. The neuropathic pain: An overview of the current treatment and future therapeutic approaches. Int J Immunopathol Pharmacol. 2019, 33, 2058738419838383. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hallin, R.G.; Torebjörk, H.E. Electrically induced A and C fibre responses in intact human skin nerves. Exp Brain Res. 1973, 16, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Peripheral Nerve Stimulation: A Comprehensive Guide, 1st ed.; Abd-Elsayed, A, Trescot, AM, Eds.; Elsevier: Philadelphia, PA, 2022; ISBN 978-0-323-83007-2. [Google Scholar]
- Papuć, E.; Rejdak, K. The role of neurostimulation in the treatment of neuropathic pain. Ann Agric Environ Med. 2013, 20, 14–17. [Google Scholar]
- Swett, J.E.; Law, J.D. Analgesia with peripheral nerve stimulation: absence of a peripheral mechanism. Pain. 1983, 15, 55–70. [Google Scholar] [CrossRef]
- Deer, T.R.; Jain, S.; Hunter, C.; Chakravarthy, K. Neurostimulation for intractable chronic pain. Brain Sci. 2019, 9, 23. [Google Scholar] [CrossRef]
- Abd-Elsayed, A.; Attanti, S.; Anderson, M.; Dunn, T.; Maloney, J.; Strand, N. Mechanism of Action of Temporary Peripheral Nerve Stimulation. Curr Pain Headache Rep. 2024, 28, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- Abd-Elsayed, A.; D'Souza, R.S. Peripheral Nerve Stimulation: The Evolution in Pain Medicine. Biomedicines 2021, 10, 18. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Manchikanti, L.; Abd-Elsayed, A.; Kaye, A.D.; Sanapati, M.R.; Knezevic, N.N.; Manocha, V.; Hirsch, J.A. Review of Guidelines for Implantable Peripheral Nerve Stimulation (PNS) in the Management of Chronic Pain. Curr Pain Headache Rep. 2025, 29, 89. [Google Scholar] [CrossRef] [PubMed]
- Abd-Elsayed, A. Wireless peripheral nerve stimulation for treatment of peripheral neuralgias. Neuromodulation: Technology at the Neural Interface 2020, 23, 827–830. [Google Scholar] [CrossRef]
- Campbell, J.N.; Long, D.M. Peripheral nerve stimulation in the treatment of intractable pain. J Neurosurg. 1976, 45, 692–9. [Google Scholar] [CrossRef] [PubMed]
- Law, J.D.; Swett, J.; Kirsch, W.M. Retrospective analysis of 22 patients with chronic pain treated by peripheral nerve stimulation. J Neurosurg. 1980, 52, 482–5. [Google Scholar] [CrossRef] [PubMed]
- Waisbrod, H.; Panhans, C.; Hansen, D.; Gerbershagen, H.U. Direct nerve stimulation for painful peripheral neuropathies. J Bone Joint Surg Br. 1985, 67, 470–2. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, E.; Waisbrod, H.; Gerbershagen, H.U. Long-term peripheral nerve stimulation for painful nerve injuries. Clin J Pain. 2004, 20, 143–6. [Google Scholar] [CrossRef] [PubMed]
- Stevanato, G.; Devigili, G.; Eleopra, R.; Fontana, P.; Lettieri, C.; Baracco, C.; Guida, F.; Rinaldo, S.; Bevilacqua, M. Chronic post-traumatic neuropathic pain of brachial plexus and upper limb: a new technique of peripheral nerve stimulation. Neurosurg Rev. 2014, 37, 473–79; discussion 479-80. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lin, E. Peripheral Nerve Stimulation Using High-frequency Electromagnetic Coupling (HF-EMC) Technology to Power an Implanted Neurostimulator With a Separate Receiver for Treating Peripheral Neuropathy. Pain Physician 2024, 27, E725–E730. [Google Scholar] [CrossRef] [PubMed]
- Abd-Elsayed, A.; Gyorfi, M.; Fischman, M.; et al. Reduced Pain and Improved Function Following Short-Term Use of Noninvasive BioWave High Frequency Peripheral Nerve Stimulation for Pain Management. Pain Ther 2023, 12, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Abd-Elsayed, A.; Keith, M.K.; Cao, N.N.; et al. Temporary Peripheral Nerve Stimulation as Treatment for Chronic Pain. Pain Ther 2023, 12, 1415–1426. [Google Scholar] [CrossRef]
- Warner, N.S.; Schaefer, K.K.; Eldrige, J.S.; et al. Peripheral nerve stimulation and clinical outcomes: a retrospective case series. Pain Pract. 2021, 21, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, C.A.; Deer, T.R.; Desai, M.J.; Hopkins, T.J.; Li, S.; DePalma, M.J.; Cohen, S.P.; McGee, M.J.; Boggs, J.W. Durable patient-reported outcomes following 60-day percutaneous peripheral nerve stimulation (PNS) of the medial branch nerves. Interv Pain Med. 2023, 2, 100243. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Shang, K.; Liu, Y.; Qadeer, A. Platelet-rich plasma in peripheral nerve injury repair: a comprehensive review of mechanisms, clinical applications, and therapeutic potential. Exp Biol Med (Maywood) 2025, 250, 10746. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Deepa, G.; Shrikrishna, B.H. The role of stem cells in peripheral nerve regeneration: a narrative review. Cureus 2025, 17, e91459. [Google Scholar] [CrossRef] [PubMed]
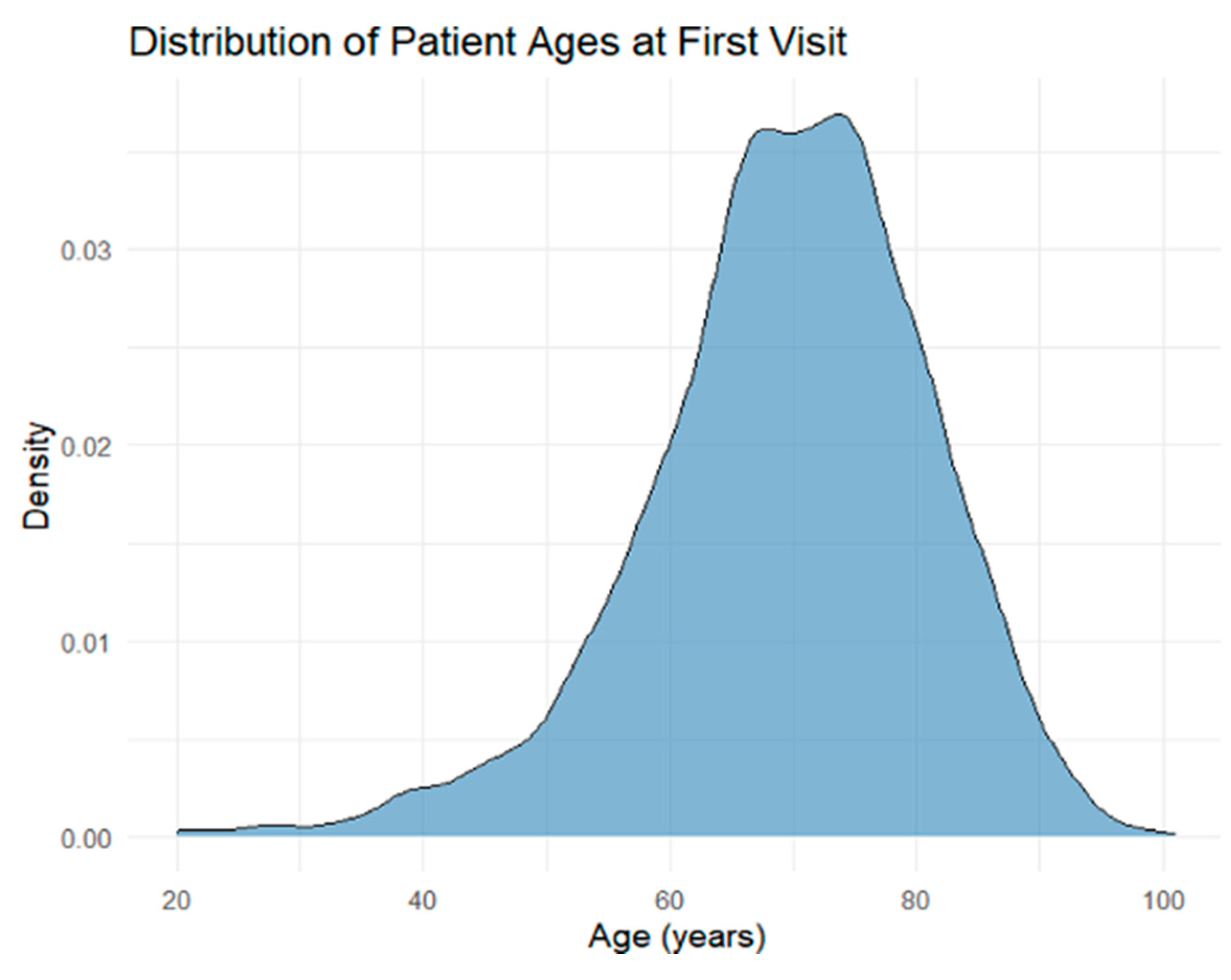
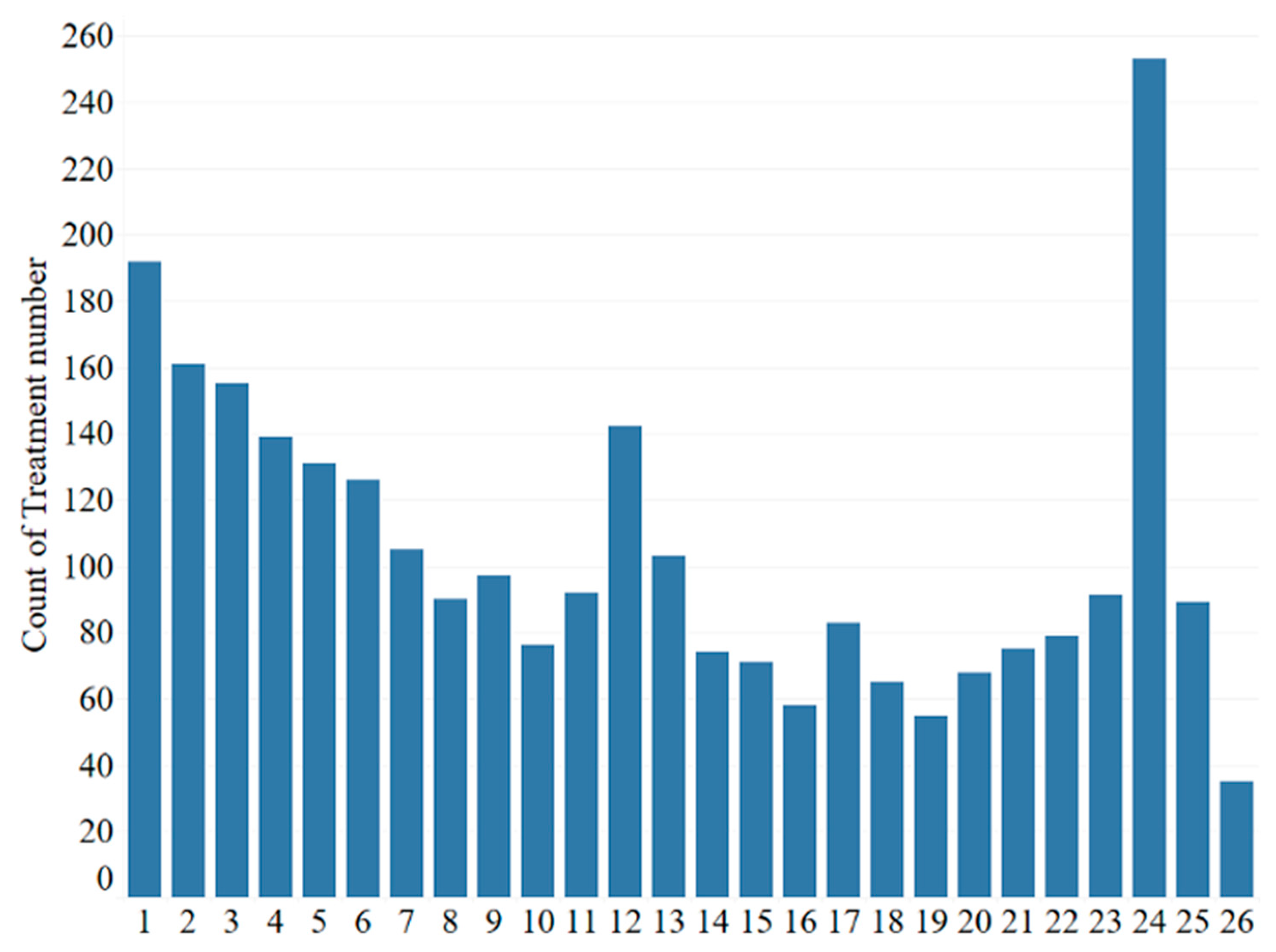
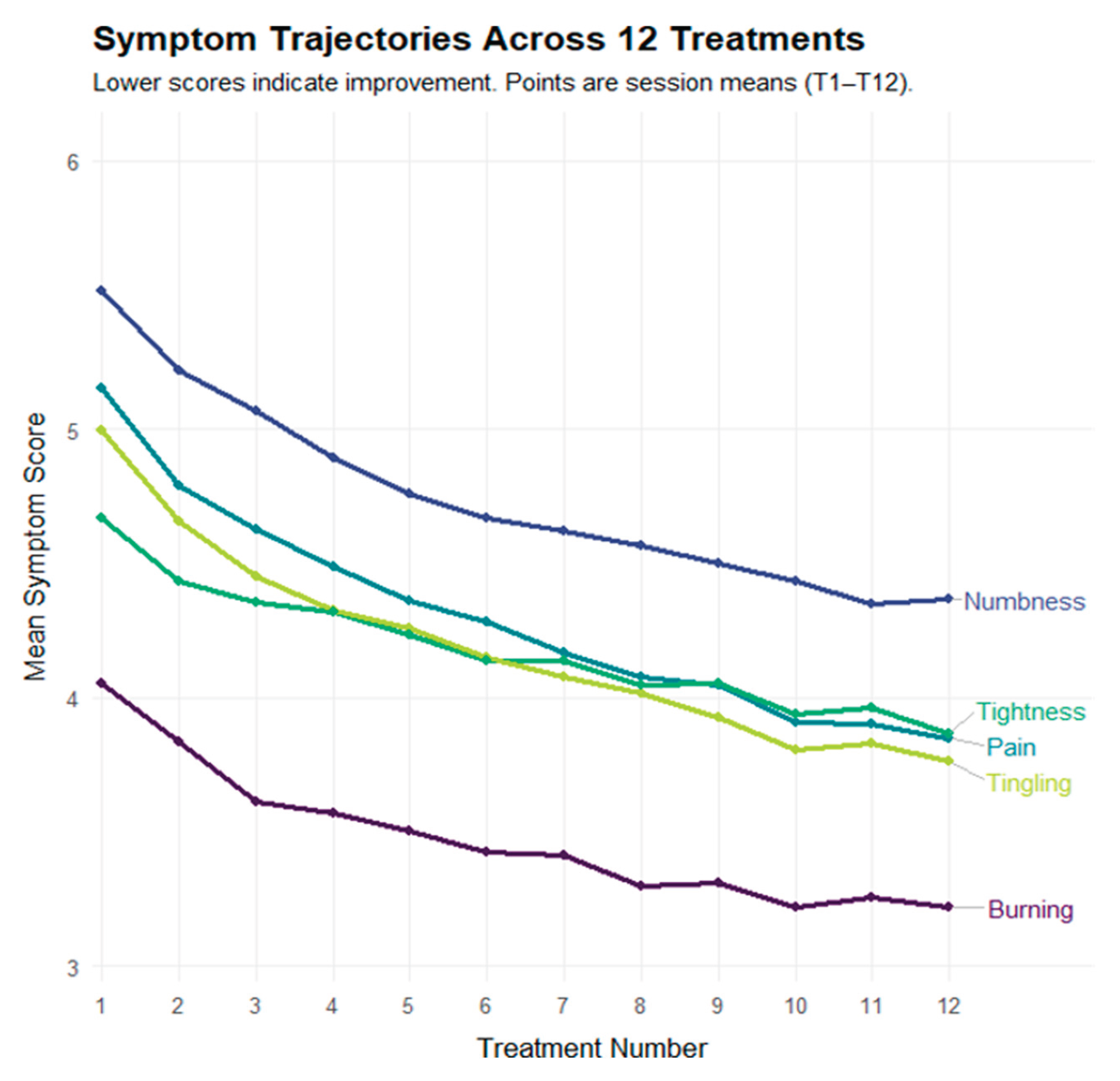
| Symptom or Function Measurement | Percent of Complete Data |
|---|---|
| Pain | 74.51% |
| Numbness | 68.48% |
| Tingling | 67.48% |
| Burning | 68.00% |
| Tightness | 68.12% |
| Bending or stooping | 71.60% |
| Putting on shoes | 65.77% |
| Sleeping | 65.84% |
| Standing up for an hour | 65.65% |
| Going up or down a flight of stairs | 65.47% |
| Walking through a store | 65.95% |
| Driving for an hour | 64.74% |
| Preparing a meal | 65.26% |
| Yard work | 64.17% |
| Picking up items from the floor | 65.41% |
| Comparison | n | Mean Change | % Improved | p-value |
|---|---|---|---|---|
| T1 → T2 | 2512 | -0.311 | 30.6 | < 0.001 |
| T1 → T6 | 1975 | -0.712 | 45.2 | < 0.001 |
| T1 → T12 | 2505 | -0.967 | 47.6 | < 0.001 |
| Comparison | n | Mean Change | % Improved | p-value |
|---|---|---|---|---|
| T1 → T2 | 2365 | -0.304 | 29.9 | < 0.001 |
| T1 → T6 | 1863 | -0.789 | 47.3 | < 0.001 |
| T1 → T12 | 1422 | -1.060 | 53.0 | < 0.001 |
| Comparison | n | Mean Change | % Improved | p-value |
|---|---|---|---|---|
| T1 → T2 | 2315 | -0.335 | 29.2 | < 0.001 |
| T1 → T6 | 1821 | -0.764 | 45.7 | < 0.001 |
| T1 → T12 | 1376 | -1.120 | 50.8 | < 0.001 |
| Comparison | n | Mean Change | % Improved | p-value |
|---|---|---|---|---|
| T1 → T2 | 2347 | -0.231 | 25.4 | < 0.001 |
| T1 → T6 | 1858 | -0.559 | 37.8 | < 0.001 |
| T1 → T12 | 1412 | -0.695 | 41.6 | < 0.001 |
| Task | n | Mean Change | % Improved | p-value |
|---|---|---|---|---|
| Standing up for an hour | 1349 | -0.277 | 30.7 | < 0.001 |
| Bending or stooping | 1435 | -0.194 | 28.4 | < 0.001 |
| Putting on shoes | 1339 | -0.119 | 24.3 | < 0.001 |
| Going up or down a flight of stairs | 1308 | -0.216 | 27.6 | < 0.001 |
| Walking through a store | 1350 | -0.144 | 24.8 | < 0.001 |
| Picking up items from the floor | 1338 | -0.153 | 25.9 | < 0.001 |
| Sleeping | 1335 | -0.101 | 24.0 | 0.002 |
| Preparing a meal | 1328 | -0.0346 | 18.1 | 0.232 |
| Yard work | 1304 | -0.108 | 19.9 | 0.002 |
| Driving for an hour | 1322 | -0.0484 | 19.1 | 0.129 |
| Domain | Measure | n | Mean change | 95% CI | Cohen's d | % Improved (any) |
% Clinically Meaningful |
|---|---|---|---|---|---|---|---|
| Symptom | Burning | 1412 | -0.695 | -0.867, -0.522 |
-0.21 | 41.6 | 60.7 |
| Symptom | Numbness | 1422 | -1.06 | -1.216, -0.909 |
-0.36 | 53 | 60.1 |
| Symptom | Pain | 1505 | -0.967 | -1.122, -0.811 |
-0.31 | 47.6 | 56.9 |
| Symptom | Tightness | 1395 | -0.724 | -0.9, -0.548 |
-0.22 | 45 | 57.9 |
| Symptom | Tingling | 1376 | -1.12 | -1.294, -0.955 |
-0.35 | 50.8 | 60.9 |
| Task | Bending Or Stooping | 1435 | -0.194 | -0.252, -0.135 |
-0.17 | 28.4 | 28.4 |
| Task | Driving For An Hour |
1322 | -0.048 | -0.111, 0.014 | -0.04 | 19.1 | 19.1 |
| Task | Going Up Or Down A Flight Of Stairs |
1308 | -0.216 | -0.28, -0.152 |
-0.18 | 27.6 | 27.6 |
| Task | Picking Up Items From The Floor |
1338 | -0.153 | -0.212, -0.095 |
-0.14 | 25.9 | 25.9 |
| Task | Preparing A Meal | 1328 | -0.035 | -0.091, 0.022 |
-0.03 | 18.1 | 18.1 |
| Task | Putting On Shoes |
1339 | -0.119 | -0.176, -0.061 |
-0.11 | 24.3 | 24.3 |
| Task | Sleeping | 1335 | -0.101 | -0.164, -0.038 |
-0.09 | 24 | 24 |
| Task | Standing Up For An Hour |
1349 | -0.277 | -0.342, -0.211 |
-0.23 | 30.7 | 30.7 |
| Task | Walking Through A Store |
1350 | -0.144 | -0.202, -0.087 |
-0.13 | 24.8 | 24.8 |
| Task | Yard Work | 1304 | -0.108 | -0.178, -0.039 |
-0.08 | 19.9 | 19.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




