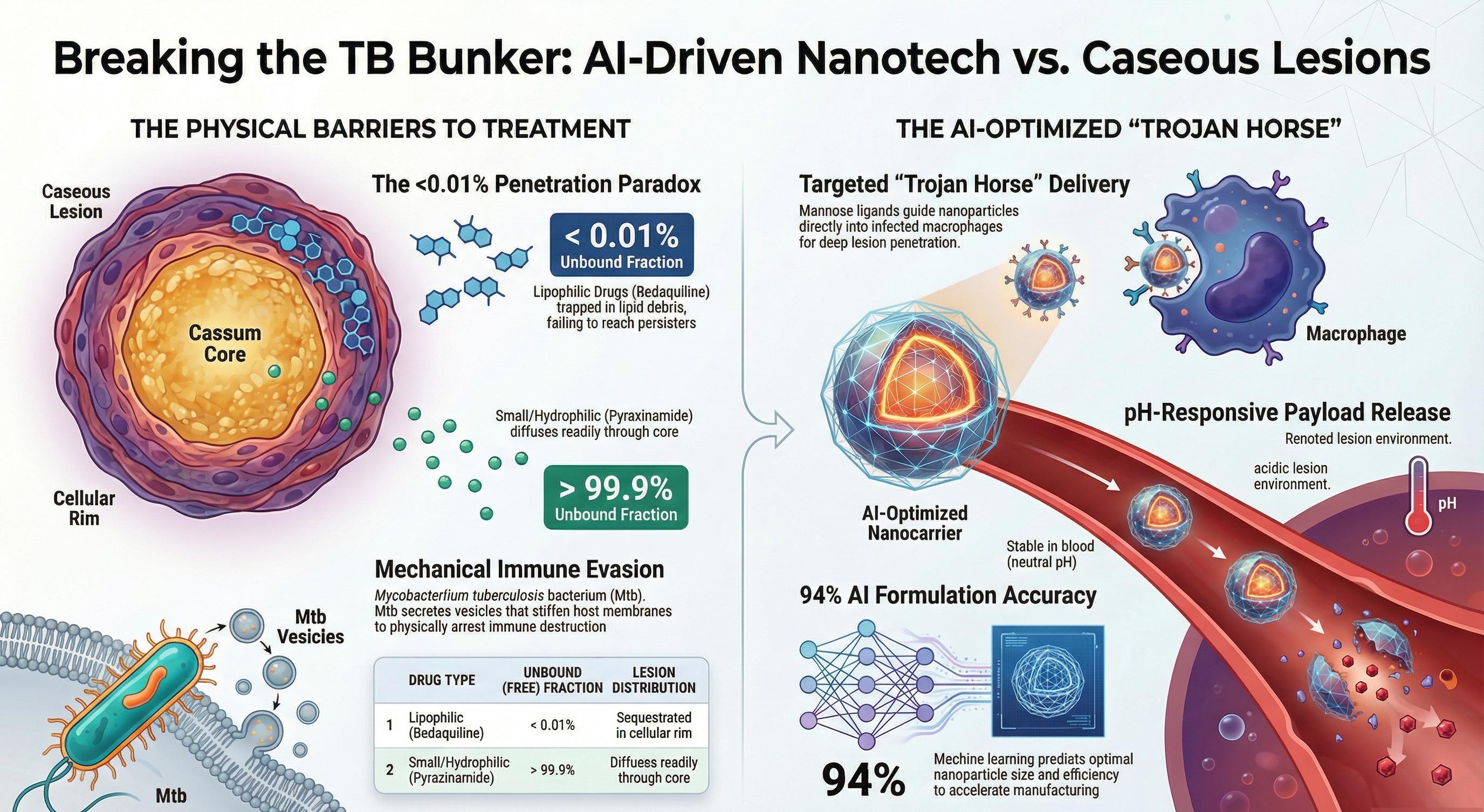
Despite the availability of antibiotics, pulmonary tuberculosis (TB) remains a leading infectious cause of mortality globally. Treatment failure and the emergence of drug-resistant strains are largely driven by the heterogeneous architecture of caseating granulomas and the complex biophysical mechanisms by which Mycobacterium tuberculosis (Mtb) evades host immunity. Highly lipophilic frontline drugs, such as bedaquiline and clofazimine, exhibit severe sequestration within the lipid-rich necrotic caseum, preventing them from reaching the dormant persister bacilli at the lesion's core. Furthermore, recent biophysical discoveries reveal that Mtb utilizes extracellular vesicles and specialized lipids to mechanically stiffen host macrophage membranes, thereby arresting phagosome-lysosome fusion. This review proposes an AI-optimized, "Trojan Horse" hybrid nanocarrier strategy—comprising a lipidic core, a mucoadhesive chitosan shell, mannose-targeted ligands, and pH-responsive release mechanisms—to bypass these dual barriers. By bridging lesion-centric pharmacokinetics ( , ), novel bioorthogonal diagnostic probes, and machine learning formulation designs, we present a translational roadmap aimed at achieving complete sterilization of caseous cavities.