Submitted:
05 March 2026
Posted:
06 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
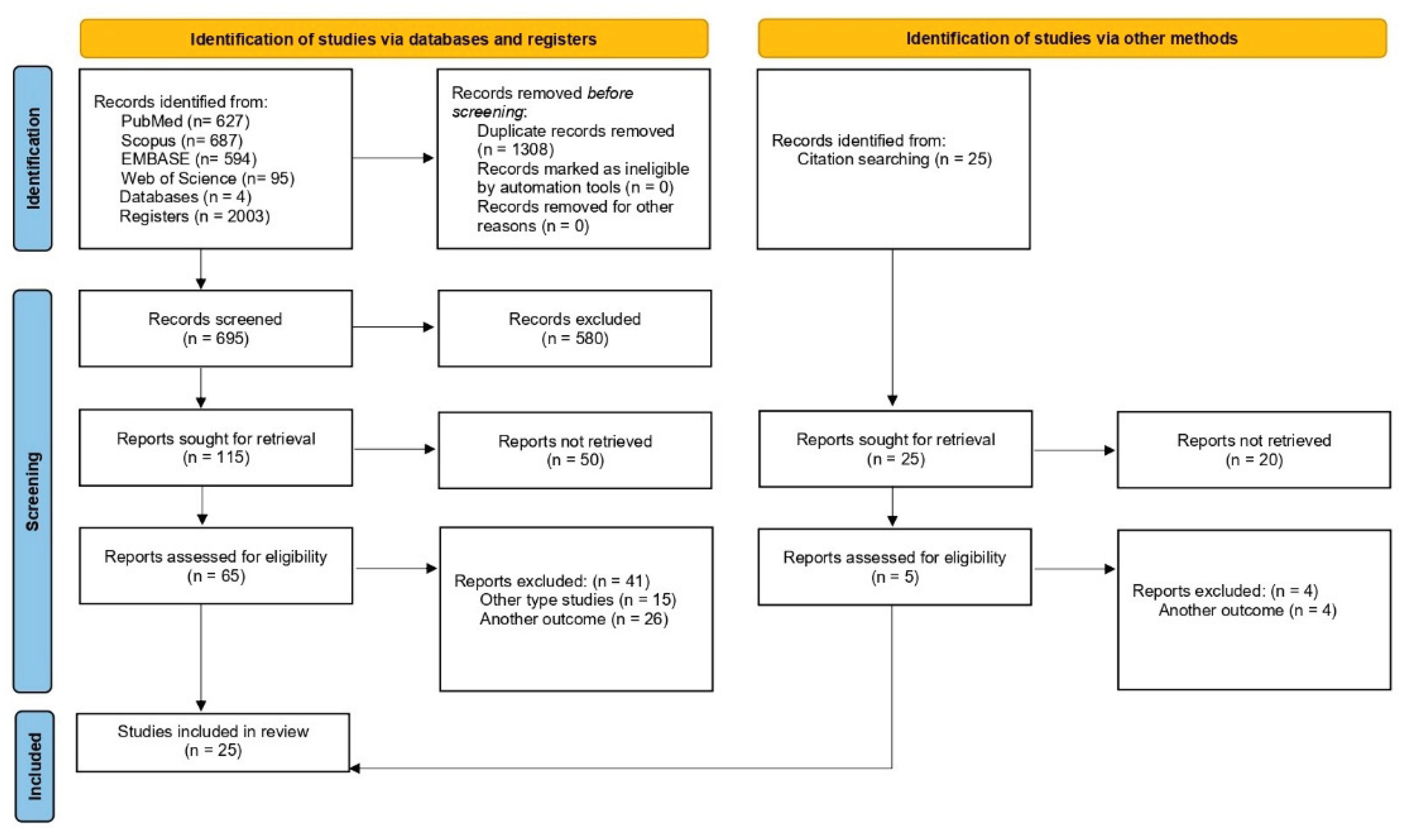
2.1. Protocol and Registration
2.2. Eligibility Criteria
2.3. Information Sources
2.4. Search Strategy
2.5. Selection of Sources of Evidence
2.6. Data Charting Process
2.7. Data Items
2.8. Synthesis of Results
3. Results
3.1. Selection of Sources of Evidence
3.2. Respiratory Syncytial Virus Vaccines
3.3. Pneumococcal Vaccines
3.4. Influenza Vaccines
3.5. COVID-19 Vaccines
4. Discussion
4.1. Respiratory Syncytial Virus Vaccines
4.2. Pneumonia Vaccines
4.3. Influenza Vaccines
4.4. COVID-19 Vaccines
4.5. Limitations
4.6. Recommendations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AMI | Acute myocardial infarction |
| CVD | Cardiovascular diseases |
| RSVpreF | Prefusion F protein–based RSV vaccine |
| RSV | Respiratory syncytial virus |
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 |
References
- Vervoort, D.; Minthor, L.; Pezzella, A.T. Monitoring, evaluation, and disease surveillance for cardiovascular surgical disease. Global Cardiac Surgery Capacity Development in Low and Middle-Income Countries 2022, 107–111. [Google Scholar]
- Pedretti, R.F.E.; Asteggiano, R.; Gevaert, A.B.; Bowen, T.S.; Caselli, S.; Cornelissen, V.A.; et al. Cardiovascular risk factors management in older adults: a clinical consensus statement from the European Association of Preventive Cardiology of the ESC and the ESC Council for Cardiology Practice. Eur J Prev Cardiol 2025, zwaf175. [Google Scholar] [CrossRef]
- Santoro, A.; Bientinesi, E.; Monti, D. Immunosenescence and inflammaging in the aging process: age-related diseases or longevity? Ageing Res Rev 2021, 71, 101422. [Google Scholar] [CrossRef]
- Moffa, L.; Tana, C. Healthcare-associated infections (HAIs) in the elderly: molecular mechanisms of immunosenescence and clinical, nutritional and therapeutic implications. Int J Mol Sci 2025, 26, 9649. [Google Scholar] [CrossRef] [PubMed]
- Sudnik, P.; Walsh, E.E.; Branche, A.R.; Islam, M.R.; Falsey, A.R. Comprehensive analysis of cardiovascular events and risk factors in patients hospitalized with respiratory syncytial virus. Clin Infect Dis 2026, 82, e396–403. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.Q.; Vlasenko, D.; Shetty, A.N.; Zhao, E.; Reid, C.M.; Clothier, H.J.; et al. Systematic review and meta-analysis of respiratory viral triggers for acute myocardial infarction and stroke. Cardiovasc Res 2025, 121, 1330–1344. [Google Scholar] [CrossRef]
- Woodruff, R.C.; Melgar, M.; Pham, H.; Sperling, L.S.; Loustalot, F.; Kirley, P.D.; et al. Acute cardiac events in hospitalized older adults with respiratory syncytial virus infection. JAMA Intern Med 2024, 184, 602–611. [Google Scholar] [CrossRef]
- Rademacher, J.; Therre, M.; Hinze, C.A.; Buder, F.; Böhm, M.; Welte, T. Association of respiratory infections and the impact of vaccinations on cardiovascular diseases. Eur J Prev Cardiol 2024, 31, 877–888. [Google Scholar] [CrossRef] [PubMed]
- Ivey, K.S.; Edwards, K.M.; Talbot, H.K. Respiratory syncytial virus and associations with cardiovascular disease in adults. J Am Coll Cardiol 2018, 71, 1574–1583. [Google Scholar] [CrossRef]
- Stotts, C.; Corrales-Medina, V.F.; Rayner, K.J. Pneumonia-induced inflammation, resolution and cardiovascular disease: causes, consequences and clinical opportunities. Circ Res 2023, 132, 751–774. [Google Scholar] [CrossRef]
- Micheletto, C.; Aliberti, S.; Andreoni, M.; Blasi, F.; Di Marco, F.; Di Matteo, R.; et al. Vaccination strategies in respiratory diseases: recommendation from AIPO-ITS/ETS, SIMIT, SIP/IRS, and SItI. Respiration 2025, 104, 556–574. [Google Scholar] [CrossRef]
- Addario, A.; Célarier, T.; Bongue, B.; Barth, N.; Gavazzi, G.; Botelho-Nevers, E. Impact of influenza, herpes zoster, and pneumococcal vaccinations on the incidence of cardiovascular events in subjects aged over 65 years: a systematic review. Geroscience 2023, 45, 3419–3447. [Google Scholar] [CrossRef] [PubMed]
- Aidoud, A.; Marlet, J.; Angoulvant, D.; Debacq, C.; Gavazzi, G.; Fougère, B. Influenza vaccination as a novel means of preventing coronary heart disease: effectiveness in older adults. Vaccine 2020, 38, 4944–4955. [Google Scholar] [CrossRef] [PubMed]
- Omidi, F.; Zangiabadian, M.; Shahidi Bonjar, A.H.; Nasiri, M.J.; Sarmastzadeh, T. Influenza vaccination and major cardiovascular risk: a systematic review and meta-analysis of clinical trials studies. Sci Rep 2023, 13, 20235. [Google Scholar] [CrossRef] [PubMed]
- Ciszewski, A. Cardioprotective effect of influenza and pneumococcal vaccination in patients with cardiovascular diseases. Vaccine 2018, 36, 202–206. [Google Scholar] [CrossRef]
- Heidecker, B.; Libby, P.; Vassiliou, V.S.; Roubille, F.; Vardeny, O.; Hassager, C.; et al. Vaccination as a new form of cardiovascular prevention: a European Society of Cardiology clinical consensus statement. Eur Heart J 2025, 46, 3518–3531. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev 2015, 4, 1. [Google Scholar] [CrossRef]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; et al. PRISMA extension for scoping reviews (PRISMA-ScR): checklist and explanation. Ann Intern Med 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Lassen, M.C.H.; Johansen, N.D.; Christensen, S.H.; Aliabadi, N.; Skaarup, K.G.; Modin, D.; et al. Bivalent RSV prefusion F protein-based vaccine for preventing cardiovascular hospitalizations in older adults: a prespecified analysis of the DAN-RSV trial. JAMA 2025, 334, 1431–1441. [Google Scholar] [CrossRef]
- Pareek, M.; Lassen, M.C.H.; Johansen, N.D.; Christensen, S.H.; Aliabadi, N.; Skaarup, K.G.; et al. Effectiveness of bivalent respiratory syncytial virus prefusion F protein-based vaccine in individuals with or without atherosclerotic cardiovascular disease: the DAN-RSV trial. Eur Heart J 2025, 46, 4291–4298. [Google Scholar] [CrossRef]
- Tong, X.; Gao, L.; Wong, I.C.K.; Chan, V.K.Y.; Wong, A.Y.S.; Mak, J.C.W.; et al. Effects of sequential vs single pneumococcal vaccination on cardiovascular diseases among older adults: a population-based cohort study. Int J Epidemiol 2024, 53, dyae005. [Google Scholar] [CrossRef]
- Hsu, S.Y.; Chen, F.L.; Liaw, Y.P.; Huang, J.Y.; Nfor, O.N.; Chao, D.Y. A matched influenza vaccine strain was effective in reducing the risk of acute myocardial infarction in elderly persons: a population-based study. Medicine (Baltimore) 2016, 95, e2869. [Google Scholar] [CrossRef]
- Chiang, M.H.; Wu, H.H.; Shih, C.J.; Chen, Y.T.; Kuo, S.C.; Chen, T.L. Association between influenza vaccination and reduced risks of major adverse cardiovascular events in elderly patients. Am Heart J 2017, 193, 1–7. [Google Scholar] [CrossRef]
- Liu, J.C.; Wang, T.J.; Sung, L.C.; Kao, P.F.; Yang, T.Y.; Hao, W.R.; et al. Influenza vaccination reduces hemorrhagic stroke risk in patients with atrial fibrillation: a population-based cohort study. Int J Cardiol 2017, 232, 315–323. [Google Scholar] [CrossRef]
- Mohseni, H.; Kiran, A.; Khorshidi, R.; Rahimi, K. Influenza vaccination and risk of hospitalization in patients with heart failure: a self-controlled case series study. Eur Heart J 2017, 38, 326–333. [Google Scholar] [CrossRef]
- Christiansen, C.F.; Thomsen, R.W.; Schmidt, M.; Pedersen, L.; Sørensen, H.T. Influenza vaccination and 1-year risk of myocardial infarction, stroke, heart failure, pneumonia, and mortality among intensive care unit survivors aged 65 years or older: a nationwide population-based cohort study. Intensive Care Med 2019, 45, 957–967. [Google Scholar] [CrossRef]
- Lam, F.; Chen, T.L.; Shih, C.C.; Lin, C.S.; Yeh, C.C.; Lee, Y.J.; et al. Protective effect of influenza vaccination on outcomes in geriatric stroke patients: a nationwide matched cohort study. Atherosclerosis 2019, 282, 85–90. [Google Scholar] [CrossRef]
- Wu, H.H.; Chang, Y.Y.; Kuo, S.C.; Chen, Y.T. Influenza vaccination and secondary prevention of cardiovascular disease among Taiwanese elders—a propensity score-matched follow-up study. PLoS One 2019, 14, e0219172. [Google Scholar] [CrossRef] [PubMed]
- Gotsman, I.; Shuvy, M.; Tahiroglu, I.; Zwas, D.R.; Keren, A. Influenza vaccination and outcome in heart failure. Am J Cardiol 2020, 128, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Pang, Y.; Wang, Q.; Lv, M.; Yu, M.; Lu, M.; Huang, Y.; et al. Influenza vaccination and hospitalization outcomes among older patients with cardiovascular or respiratory diseases. J Infect Dis 2021, 223, 1196–1204. [Google Scholar] [CrossRef] [PubMed]
- Pang, Y.; Liu, X.; Liu, G.; Lv, M.; Lu, M.; Wu, J.; et al. Effectiveness of influenza vaccination on in-hospital death and recurrent hospitalization in older adults with cardiovascular diseases. Int J Infect Dis 2022, 122, 162–168. [Google Scholar] [CrossRef]
- Saade, E.A.; Abul, Y.; McConeghy, K.; Edward Davidson, H.; Han, L.; Joyce, N.; et al. High-dose influenza vaccines for the prevention of hospitalization due to cardiovascular events in older adults in the nursing home: post-hoc analysis of a cluster-randomized trial. Vaccine 2022, 40, 6700–6705. [Google Scholar] [CrossRef]
- Christensen, J.; Johansen, N.D.; Modin, D.; Janstrup, K.H.; Nealon, J.; Samson, S.; et al. Relative effectiveness of high-dose versus standard-dose quadrivalent influenza vaccine in older adults with cardiovascular disease: a prespecified analysis of the DANFLU-1 randomized clinical trial. Circ Cardiovasc Qual Outcomes 2025, 18, e011496. [Google Scholar] [CrossRef] [PubMed]
- NajafZadeh, M.; Shin, H.; Schneeweiss, S.; Wang, S.V.; Solomon, S.D.; Vardeny, O.; et al. High-dose vs. standard-dose influenza vaccine and cardiopulmonary hospitalization or mortality: emulating the INVESTED trial using insurance claims data. Clin Pharmacol Ther 2024, 115, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Miró, Ò.; Ivars, N.; Lopez-Ayala, P.; Gil, V.; Llauger, L.; Alquézar-Arbé, A.; et al. Effect of flu vaccination on severity and outcome of heart failure decompensations. J Card Fail 2023, 29, 734–744. [Google Scholar] [CrossRef]
- Guo, J.; Wang, T.; Liu, Z.; Zeng, W.; Shen, P.; Sun, Y.; et al. Estimating cardiovascular effects of influenza vaccination in older adults: a target trial emulation using proximal causal inference. EClinicalMedicine 2025, 87, 103449. [Google Scholar] [CrossRef] [PubMed]
- Lei, J.; Zheng, L.Z.; Chen, K.Y.; Yang, X.; Tian, Y.; Qiu, Z.H.; et al. Independent effect of influenza vaccination on all-cause mortality in critically ill patients with atrial fibrillation: a retrospective study from the MIMIC-IV database. Int J Cardiol 2025, 433, 133246. [Google Scholar] [CrossRef]
- Yang, T.; Yang, H.; Zhou, T.; Wu, X.; Ye, L. Influenza vaccination and ischemic stroke risk reduction in elderly stroke survivors: a retrospective cohort study with negative control validation. BMC Geriatr 2025, 25, 984. [Google Scholar] [CrossRef]
- Miró, Ò.; Ivars, N.; Espinosa, B.; Jacob, J.; Alquézar-Arbé, A.; López-Díez, M.P.; et al. Effect of seasonal influenza and COVID-19 vaccination on severity and long-term outcomes of patients with heart failure decompensations. Eur J Heart Fail 2025, 27, 152–165. [Google Scholar] [CrossRef]
- Akbar, U.A.; Thyagaturu, H.; Taha, A.; Atti, L.; Khan, A.; Raina, S.; et al. COVID-19 vaccination and cardiovascular outcomes in older adults with coronary artery disease and heart failure: insights from a large propensity-matched cohort study. J Am Heart Assoc 2025, 14, e044546. [Google Scholar] [CrossRef]
- Johnson, K.W.; Patel, S.; Thapi, S.; Jaladanki, S.K.; Rao, A.; Nirenberg, S.; et al. Association of reduced hospitalizations and mortality rates among COVID-19-vaccinated patients with heart failure. J Card Fail 2022, 28, 1475–1479. [Google Scholar] [CrossRef]
- Sindet-Pedersen, C.; Michalik, F.; Strange, J.E.; Christensen, D.M.; Nouhravesh, N.; Gerds, T.A.; et al. Risk of worsening heart failure and all-cause mortality following COVID-19 vaccination in patients with heart failure: a nationwide real-world safety study. Circ Heart Fail 2023, 16, e010617. [Google Scholar] [CrossRef]
- Ye, X.; Huang, C.; Wei, Y.; Li, S.T.H.; Yan, V.K.C.; Yiu, K.H.; et al. Safety of BNT162b2 or CoronaVac COVID-19 vaccines in patients with heart failure: a self-controlled case series study. Lancet Reg Health West Pac 2023, 30, 100630. [Google Scholar] [CrossRef]
- Obando-Pacheco, P.; Justicia-Grande, A.J.; Rivero-Calle, I.; Rodríguez-Tenreiro, C.; Sly, P.; Ramilo, O.; et al. Respiratory syncytial virus seasonality: a global overview. J Infect Dis 2018, 217, 1356–1364. [Google Scholar] [CrossRef] [PubMed]
- Shi, T.; Denouel, A.; Tietjen, A.K.; Campbell, I.; Moran, E.; Li, X.; et al. Global disease burden estimates of respiratory syncytial virus-associated acute respiratory infection in older adults in 2015: a systematic review and meta-analysis. J Infect Dis 2020, 222, S577–S583. [Google Scholar] [CrossRef] [PubMed]
- Wildenbeest, J.G.; Lowe, D.M.; Standing, J.F.; Butler, C.C. Respiratory syncytial virus infections in adults: a narrative review. Lancet Respir Med 2024, 12, 822–836. [Google Scholar] [CrossRef]
- Savic, M.; Penders, Y.; Shi, T.; Branche, A.; Pirçon, J.Y. Respiratory syncytial virus disease burden in adults aged 60 years and older in high-income countries: a systematic literature review and meta-analysis. Influenza Other Respir Viruses 2023, 17, e13031. [Google Scholar] [CrossRef]
- Cherukuri, A.; Patton, K.; Gasser, R.A.; Zuo, F.; Woo, J.; Esser, M.T.; et al. Adults 65 years old and older have reduced numbers of functional memory T cells to respiratory syncytial virus fusion protein. Clin Vaccine Immunol 2013, 20, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Papi, A.; Ison, M.G.; Langley, J.M.; Lee, D.G.; Leroux-Roels, I.; Martinon-Torres, F.; et al. Respiratory syncytial virus prefusion F protein vaccine in older adults. N Engl J Med 2023, 388, 595–608. [Google Scholar] [CrossRef]
- Ison, M.G.; Papi, A.; Athan, E.; Feldman, R.G.; Langley, J.M.; Lee, D.G.; et al. Efficacy and safety of respiratory syncytial virus (RSV) prefusion F protein vaccine (RSVPreF3 OA) in older adults over 2 RSV seasons. Clin Infect Dis 2024, 78, 1732–1744. [Google Scholar] [CrossRef]
- Ison, M.G.; Papi, A.; Athan, E.; Feldman, R.G.; Langley, J.M.; Lee, D.G.; et al. Efficacy, safety, and immunogenicity of the AS01E-adjuvanted respiratory syncytial virus prefusion F protein vaccine (RSVPreF3 OA) in older adults over three respiratory syncytial virus seasons (AReSVi-006): a multicentre, randomised, observer-blinded, placebo-controlled, phase 3 trial. Lancet Respir Med 2025, 13, 517–529. [Google Scholar] [PubMed]
- Garcia-Carretero, R.; Gil-Prieto, R.; Hernandez-Barrera, V.; Gil-de-Miguel, A. Epidemiological and clinical impact of pneumococcal disease in Spain in 2023: a nationwide retrospective analysis. Hum Vaccin Immunother 2025, 21, 2579385. [Google Scholar] [CrossRef]
- Mrabt, F.; Guedes, S. Systematic review on serotypes distribution of pneumococcal pneumonia in adults and the elderly. BMC Public Health 2025, 25, 1194. [Google Scholar] [CrossRef]
- Africano, H.F.; Serrano-Mayorga, C.C.; Ramirez-Valbuena, P.C.; Bustos, I.G.; Bastidas, A.; Vargas, H.A.; et al. Major adverse cardiovascular events during invasive pneumococcal disease are serotype dependent. Clin Infect Dis 2021, 72, e711–e719. [Google Scholar] [CrossRef]
- Dulfer, E.A.; Serbée, M.J.V.; Dirkx, K.K.T.; Schaars, C.F.; Wertheim, H.F.L.; de Jonge, M.I.; et al. Cardiovascular events after invasive pneumococcal disease: a retrospective cohort study. Int J Infect Dis 2024, 147, 107185. [Google Scholar] [CrossRef]
- Wiese, A.D.; Mitchel, E.; Ndi, D.; Markus, T.M.; Talbot, H.K.; Schaffner, W.; et al. Risk of acute myocardial infarction among patients with laboratory-confirmed invasive pneumococcal disease: a self-controlled case series study. Clin Infect Dis 2023, 76, 2171–2177. [Google Scholar] [CrossRef]
- Bulkhi, A.; Khadawardi, H.A.; Dairi, M.S.; Alwafi, H.; Alim, H.M.; Turkistani, Y.A.; et al. Effectiveness of pneumococcal vaccination in reducing hospitalization and mortality among the elderly: a systematic review and meta-analysis. Hum Vaccin Immunother 2025, 21, 2561315. [Google Scholar] [CrossRef]
- Heidenreich, P.A.; Bhatt, A.; Nazir, N.T.; Schaffner, W.; Vardeny, O. 2025 concise clinical guidance: an ACC expert consensus statement on adult immunizations as part of cardiovascular care: a report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol 2025, 86, 2085–2098. [Google Scholar] [CrossRef] [PubMed]
- Clar, C.; Oseni, Z.; Flowers, N.; Keshtkar-Jahromi, M.; Rees, K. Influenza vaccines for preventing cardiovascular disease. Cochrane Database Syst Rev 2015, 2015, CD005050. [Google Scholar] [PubMed]
- Marra, F.; Zhang, A.; Gillman, E.; Bessai, K.; Parhar, K.; Vadlamudi, N.K. The protective effect of pneumococcal vaccination on cardiovascular disease in adults: a systematic review and meta-analysis. Int J Infect Dis 2020, 99, 204–213. [Google Scholar] [CrossRef]
- Rosero, C.I.; Gravenstein, S.; Saade, E.A. Influenza and aging: clinical manifestations, complications, and treatment approaches in older adults. Drugs Aging 2025, 42, 39–55. [Google Scholar] [CrossRef]
- Cowling, B.J.; Okoli, G.N. Influenza vaccine effectiveness and progress towards a universal influenza vaccine. Drugs 2024, 84, 1013–1023. [Google Scholar] [CrossRef] [PubMed]
- Uyeki, T.M.; Hui, D.S.; Zambon, M.; Wentworth, D.E.; Monto, A.S. Influenza. Lancet 2022, 400, 693–706. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; Zou, Y.; Zhao, D.; Yu, J. Optimising vaccine immunogenicity in ageing populations: key strategies. Lancet Infect Dis 2025, 25, e23–e33. [Google Scholar] [CrossRef]
- Paneni, F.; Diaz Cañestro, C.; Libby, P.; Lüscher, T.F.; Camici, G.G. The aging cardiovascular system: understanding it at the cellular and clinical levels. J Am Coll Cardiol 2017, 69, 1952–1967. [Google Scholar] [CrossRef]
- Iuliano, A.D.; Roguski, K.M.; Chang, H.H.; Muscatello, D.J.; Palekar, R.; Tempia, S.; et al. Estimates of global seasonal influenza-associated respiratory mortality: a modelling study. Lancet 2018, 391, 1285–1300. [Google Scholar] [CrossRef]
- Ferdinands, J.M.; Blanton, L.H.; Alyanak, E.; Chung, J.R.; Trujillo, L.; Taliano, J.; et al. Protection against influenza hospitalizations from enhanced influenza vaccines among older adults: a systematic review and network meta-analysis. J Am Geriatr Soc 2024, 72, 3875–3889. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.K.H.; Lam, G.K.L.; Yin, J.K.; Loiacono, M.M.; Samson, S.I. High-dose influenza vaccine in older adults by age and seasonal characteristics: systematic review and meta-analysis update. Vaccine X 2023, 14, 100327. [Google Scholar] [CrossRef]
- Gupta, R.; Quy, R.; Lin, M.; Mahajan, P.; Malik, A.; Sood, A.; et al. Role of influenza vaccination in cardiovascular disease: systematic review and meta-analysis. Cardiol Rev 2024, 32, 423–428. [Google Scholar] [CrossRef]
- Barbetta, L.M.D.S.; Correia, E.T.; de O, G.R.A.O.C.; Mesquita, E.T. Influenza vaccination as prevention therapy for stable coronary artery disease and acute coronary syndrome: a meta-analysis of randomized trials. Am J Med 2023, 136, 466–475. [Google Scholar] [CrossRef]
- Johansen, N.D.; Modin, D.; Pardo-Seco, J.; Rodriguez-Tenreiro-Sánchez, C.; Loiacono, M.M.; Harris, R.C.; et al. High-dose vs. standard-dose influenza vaccine and cardiovascular outcomes in older adults: the FLUNITY-HD prespecified pooled analysis. Circulation 2025. [Google Scholar] [CrossRef]
- Chen, B.; Farzan, M.; Choe, H. SARS-CoV-2 spike protein: structure, viral entry and variants. Nat Rev Microbiol 2025, 23, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Guzik, T.J.; Mohiddin, S.A.; Dimarco, A.; Patel, V.; Savvatis, K.; Marelli-Berg, F.M.; et al. COVID-19 and the cardiovascular system: implications for risk assessment, diagnosis, and treatment options. Cardiovasc Res 2020, 116, 1666–1687. [Google Scholar] [CrossRef] [PubMed]
- Jone, P.N.; John, A.; Oster, M.E.; Allen, K.; Tremoulet, A.H.; Saarel, E.V.; et al. SARS-CoV-2 infection and associated cardiovascular manifestations and complications in children and young adults: a scientific statement from the American Heart Association. Circulation 2022, 145, e1037–e1052. [Google Scholar] [CrossRef] [PubMed]
- Cenko, E.; Badimon, L.; Bugiardini, R.; Claeys, M.J.; De Luca, G.; de Wit, C.; et al. Cardiovascular disease and COVID-19: a consensus paper from the ESC working groups and ACVC in collaboration with EHRA. Cardiovasc Res 2021. [Google Scholar] [CrossRef]
- Giustino, G.; Pinney, S.P.; Lala, A.; Reddy, V.Y.; Johnston-Cox, H.A.; Mechanick, J.I.; et al. Coronavirus and cardiovascular disease, myocardial injury, and arrhythmia: JACC focus seminar. J Am Coll Cardiol 2020, 76, 2011–2023. [Google Scholar] [CrossRef]
- Liang, C.K.; Lee, W.J.; Peng, L.N.; Meng, L.C.; Hsiao, F.Y.; Chen, L.K. COVID-19 vaccines in older adults: challenges in vaccine development and policy making. Clin Geriatr Med 2022, 38, 605–620. [Google Scholar] [CrossRef]
- la Cruz, R.A.R.D.; Flores-Córdova, J.M.; Calderon-Hernandez, C.C.; Cahuapaza-Gutierrez, N.L.; Ccallalli-Ruiz, N.A.; Runzer-Colmenares, F.M. Humoral and cellular immune responses against SARS-CoV-2 following COVID-19 vaccination in older adults: a systematic review. Vaccines (Basel) 2025, 13, 852. [Google Scholar] [CrossRef]
- Yang, X.H.; Bao, W.J.; Zhang, H.; Fu, S.K.; Jin, H.M. The efficacy of SARS-CoV-2 vaccination in the elderly: a systematic review and meta-analysis. J Gen Intern Med 2023. [Google Scholar] [CrossRef]
- Ip, S.; North, T.L.; Torabi, F.; Li, Y.; Abbasizanjani, H.; Akbari, A.; et al. Cohort study of cardiovascular safety of different COVID-19 vaccination doses among 46 million adults in England. Nat Commun 2024, 15, 6085. [Google Scholar] [CrossRef]
| Author /year | Country | Design | Network | Period | Vaccine type | Total sample | Vaccinated sample | Age (Mean/ Median) | Female | Unvaccinated sample | Age | Female | Outcomes | Follow-up |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lassen et al. 2025 [19] | Denmark | Prespecified secondary analysis of a trial | DAN-RSV | 2024-2025 | RSV Vaccine | 28 662 | 14 377 | 71.8 | 5 186 | 14 285 | 71.8 | 5 038 | Hospitalizations Mortality |
1 year |
| Pareek et al. 2025 [20] | Denmark | Prespecified secondary analysis of a trial | DAN-RSV | 2024-2025 | RSV Vaccine | 131 276 | 65 642 | 71.5 | 21 268 | 65 634 | 69.2 | 21 265 | Effectivity | 1 year |
| Author /year | Vaccinated sample | Cardiovascular disease | Efficacy All-cause mortality |
Hospitalizations | Safety/ MACE |
Conclusions |
|---|---|---|---|---|---|---|
| Lassen et al. 2025 [19] | 14 377 | AF: 10 126 (70.5%) IHD: 9 746 (67.8%) HF: 2 973 (20.7%) |
VE: -56.5% | VE: -4.7%, HF VE: 1.8%, MI VE: 19.4%, Stroke VE: -2.4%, AF |
- | Lower hospitalizations rate in the vaccinated group |
| Pareek et al. 2025 [20] | 65 642 | Prespecified secondary analysis of a trial | DAN-RSV | 2024-2025 | VE: 9.3%, MACE | The effectiveness of the vaccines was similar to that of the controls |
| Author /year | Country | Design | Network | Period | Vaccine type | Total sample | Vaccinated sample | Age (Mean/ Median) | Female | Unvaccinated sample | Age | Female | Outcomes | Follow-up |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hsu et al. 2016 [22] | Taiwan | Retrospective cohort | LHID 2005 |
2007-2008 | Influenza vaccine | 202 058 | 93 051 | 75.91 | 46 243 | 109 007 | 74.55 | 54 069 | Risk of AMI | 9 months |
| Chiang et al. 2017 [23] | Taiwan | Retrospective case-control | NHIRD | 2000-2013 | Influenza vaccine | 160 726 | Case: 29 046 Controls: 33 285 |
76.8 | - | Case: 51 317 Controls: 47 078 |
76.8 | - | MACE | - |
| Liu et al. 2017 [24] | Taiwan | Cohort | NHIRD | 2005-2012 | Influenza vaccine | 6 570 | 2 547 | 74.33 | 1 187 | 4 023 | 72.79 | 1 913 | Risk of HS | - |
| Mohseni et al. 2017 [25] | UK | Self-controlled case series | CPRD | - | Influenza vaccine | 59 202 | 59 202 | 74.7 | 29 553 | - | - | - | Hospitalizations | - |
| Christiansen et al. 2019 [26] | Denmark | Cohort | NHIRD | 2005-2015 | Influenza vaccine | 31 108 | 11 866 | - | 5 192 | 19 242 | - | 67 043 | Hospitalization All-cause mortality |
1 year |
| Lam et al. 2019 [27] | Taiwan | Cohort | NHIRD | 2000 -2009 | Influenza vaccine | 50 496 | 25 248 | - | 11 332 | 25 248 | - | 11 332 | In-hospital mortality Hospitalizations |
30 days |
| Wu et al. 2019 [28] | Taiwan | Retrospective PM-cohort | NHIRD |
2000-2013 | Seasonal influenza vaccine | 8 700 | 4 350 | 76.3 | 1527 | 4 350 | 76.2 | 1 505 | All-cause mortality Hospitalizations |
1 year |
| Gotsman et al. 2020 [29] | Israel | Retrospective cohort | Clalit Health Services | 2017-2018 | Influenza vaccine | 6 435 | 4 440 | 77 | 2 056 | 1 995 | 74 | 970 | All-cause mortality Hospitalizations |
1 year |
| Pang et al. 2021 [30] | China | Retrospective cohort | UEBMI | January 2013– December 2016 | Influenza vaccine | 139 506 | 17 655 | 74 | - | 121 851 | 72.9 | - | In-hospital death | - |
| Pang et al. 2022 [31] | China | Retrospective cohort | UEBMI | January 2013– December 2019 | Influenza vaccine | 713 488 | 95 060 | 74.1 | 53 788 | 618 428 | 72.9 | 343 227 | In-hospital death Hospitalizations |
- |
| Saade et al. 2022 [32] | USA | Post-hoc analysis | CMS | - | Influenza vaccine | 49 175 | 49 175 | 83.8 | 35 674 | - | - | - | Hospitalizations | - |
| Miró et al. 2023 [35] | Spain | Secondary analysis of cohort | EAHFE | January 2018– February 2019 | Influenza vaccine | 6 147 | 1 339 | 85 | 654 | 5 008 | 84 | 2 756 | All-cause mortality Decompensations |
1 year |
| Christensen et al. 2024 [33] | Denmark | Prespecified analysis of randomized clinical trial | DANFLU-1 | 2021-2022 | Influenza vaccine | 2 540 | 2 540 | 72.6 | 909 | - | - | - | All-cause hospitalization Mortality |
1 year |
| NajafZadeh et al. 2024 [34] | USA | Emulator Clinical Trial | Medicare claims data | 2016-2019 | Influenza vaccine | 106 786 | 106 786 | 79.96 | 60 634 | - | - | - | All-cause mortality Hospitalizations |
- |
| Guo et al. 2025 [36] | USA | Target trial emulation | YRHCD | 2020 – 2022 | Influenza vaccine | 339 976 | 169 988 | 72 | 90 414 | 169 988 | 72 | 91 138 | MACE | 2 years |
| Lei et al. 2025 [37] | China | Retrospective PM-cohort | MIMIC-IV | NR | Influenza vaccine | 9 500 | 4 758 | 75.44 | - | 4 742 | 75.76 | - | All-cause mortality | 1 year |
| Miró et al. 2025 [39] | Spain | Secondary analysis of cohort | EAHFE | November - December 2022 | Influenza vaccine | 4 243 | 1 841 | 86 | 1 039 | 2 402 | 84 | 1 359 | All-cause mortality Decompensations |
1 year |
| Yang et al. 2025 [38] | China | Retrospective cohort | RHIP | 2021-2022 | Influenza vaccine | 76 747 | 31 729 | 75 | 15 326 | 45 018 | 76 | 21 851 | Stroke risk |
1 year |
| Author /year | Vaccinated sample | Cardiovascular disease | Efficacy All-cause mortality |
Hospitalizations | Severe decompensations | Safety/MACE | Conclusions |
|---|---|---|---|---|---|---|---|
| Hsu et al. 2016 [22] | 93 051 | Hypertension: 41 371 (44.5%) IHD: 15 904 (17.1%) MI: 313 (0.34%) IS: 6 350 (6.8%) HF: 3 928 (4.2%) |
- | - | - | HR= 0.681, AMI | Vaccination was associated with a reduced risk of AMI |
| Chiang et al. 2017 [23] | 62 331 | Stroke: 46 704 (74.9%) MI: 15 627 (25.1%) |
- | - | - | aOR= 0.80, Stroke aOR= 0.80, MI |
Vaccination is associated with a reduced risk of MACE |
| Liu et al. 2017 [24] | 2 547 | AF: 2 547 (100%) Hypertension: 1 939 (76.13%) CHF: 1 298 (50.96%) |
- | - | - | aHR= 0.72, HS | Vaccination reduces the incidence of hemorrhagic stroke |
| Mohseni et al. 2017 [25] | 59 202 | HF: 59 202 (100%) Hypertension: 38 753 (46%) MI: 41 502 (49.2%) Stroke: 14 522 (17.2%) |
- | Overall IRR= 0.73 | - | - | Vaccination is associated with a lower risk of hospitalizations |
| Christiansen et al. 2019 [26] | 11 866 | MI: 4 067 (11.7%) Stroke: 5 080 (14.6%) CHF: 4 847 (13.9%) Hypertension: 12 773 (36.6%) AF/Flutter: 6 234 (17.9%) |
1 year (aHR= 0.92) |
1 year (aHR= 0.93, MI) 1 year (aHR= 0.98, HF) 1 year (aHR= 0.84, Stroke) |
- | - | Vaccination was associated with a lower risk of stroke and mortality |
| Lam et al. 2019 [27] | 25 248 | Stroke: 25 248 (100%) Hypertension: 9 735 (38.6%) IHD: 1 017 (4.0%) HF: 276 (1.1%) |
30 days (OR= 0.60) |
30 days (OR = 0.91, ICU admission) | - | - | Vaccination associated with reduced post-stroke complications and mortality |
| Wu et al. 2019 [28] | 4 350 | MI: 4 350 (100%) Hypertension: 3 918 (90.07%) HF: 2 062 (47.4%) AF: 660 (15.17%) |
1 year (HR= 0.82) |
HR= 0.83, HF | - | - | Vaccination was associated with a reduced risk of CVD, all-cause mortality and hospitalizations |
| Gotsman et al. 2020 [29] | 4 440 | HF: 4 440 (100%) Hypertension: 3 745 (84%) CHD: 2 992 (67%) MI: 1 904 (43%) AF: 1 743 (39%) Stroke: 1 053 (24%) |
HR= 0.80 | HR=0.83, CVD | - | - | Vaccination was associated with a reduction in deaths and hospitalizations |
| Pang et al. 2021 [30] | 17 655 | NR | aOR= 0.55 | - | - | - | Vaccination was associated with a lower risk of in-hospital death |
| Pang et al. 2022 [31] | 95 060 | IS: 95 060 (100%) IHD: 95 060 (100%) |
aOR= 0.85 | OR= 0.92, IHD OR= 1.04, IS |
- | - | Vaccination was associated with a lower risk of in-hospital death in patients with fewer comorbidities |
| Saade et al. 2022 [32] | 49 175 | HF: 10 160 (20.7%) Hypertension: 39 009 (79.3%) Stroke: 9 813 (20%) |
- | HR= 0.92, MACE HR= 0.96, ACS HR= 0.84, Stroke HR= 0.96, HF |
- | - | Similar reductions in hospitalizations were observed with both doses |
| Miró et al. 2025 [35] | 1 841 | AHF: 1 841 (100%) Hypertension: 1 603 (87.1%) AF: 993 (54%) CAD: 440 (23.9%) |
90 days (HR= 0.831) 1 year (HR= 0.885) |
OR= 0.746, HFD | OR= 0.926 | - | Vaccination is associated with less severe decompensation and lower all-cause mortality |
| Christensen et al. 2024 [33] | 2 540 | Hypertension: 961 (37.8%) IHD: 913 (35.9%) AF: 822 (32.4%) |
IRR= 0.51 | IRR: 0.87, CVD | - | - | All-cause mortality declined in a dose–response pattern |
| NajafZadeh et al. 2024 [34] | 106 786 | Hypertension: 105 315 (98.6%) AF: 63 986 (59.9%) IS: 28 621 (26.8%) AMI: 17 547 (16.4%) |
HR= 0.92 | HR= 0.97 | - | - | All-cause mortality and hospitalizations declined in a dose–response pattern |
| Guo et al. 2025 [36] | 169 988 | Hypertension: 141 085 (83%) HF: 18 441 (10.8%) Stroke: 38 919 (22.9%) ACS: 14 019 (8.2%) |
- | - | - | 1 year (IRR= 0.86, MACE) 1 year (IRR= 0.87, ACS) |
Vaccination was associated with a reduction in MACE and ACS |
| Lei et al. 2025 [37] | 4 758 | AF: 4 758 (100%) Hypertension: 4 084 (76.05%) CHF: 2 447 (45.57%) MI: 1 229 (22.89%) |
1 year (HR= 0.83) |
- | - | - | Vaccination was associated with a reduction in all-cause mortality |
| Miró et al. 2023 [39] | 1 339 | AHF: 1 339 (100%) HVD: 274 (31.5%) |
90 days (HR= 0.885) |
aOR= 0.823 | aOR= 0.934 | - | Vaccination is associated with less severe decompensations and fewer hospitalizations |
| Yang et al. 2025 [38] | 31 729 | Stroke: 31 729 (100%) Hypertension: 17 169 (54.1%) CAD: 2 486 (7.8%) AF: 3 262 (10.3%) |
- | - | - | sHR= 0.84, Stroke recurrence sHR= 0.75, HS sHR= 0.86, IS |
Vaccination associated with reduced ischemic stroke risk |
| Author /year | Country | Design | Network | Period | Vaccine type | Total sample | Vaccinated sample | Age (Mean/ Median) | Female | Unvaccinated sample | Age | Female | Outcomes | Follow-up |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Akbar et al. 2025 [40] | USA | Retrospective PM-cohort | TriNetX US | December 2020-2022 | COVID-19 vaccine | 148 472 | 74 236 | 73.90 | 25 601 | 74 236 | 74.30 | 25 428 | All-cause mortality | 1-2 years |
| Miró et al. 2025 [39] | Spain | Secondary analysis of cohort | EAHFE | November - December 2022 | COVID-19 vaccine | 4 243 | 3 139 | 85 | 1 769 | 1 104 | 85 | 629 | All-cause mortality Decompensations |
1 year |
| Johnson et al. 2022 [41] | USA | Retrospective cohort | - | January 2021 – January 2022 | COVID-19 vaccine | 7 094 | 3 898 | 73.9 | 1 877 | 3 196 | - | - | All-cause mortality Hospitalizations |
- |
| Sindet-Pedersen et al. 2023 [42] | Denmark | Secondary analysis of cohort | - | 2019 – 2021 | COVID-19 vaccine | 87 734 | 43 850 | - | 15 612 | 43 884 | - | 15 624 | All-cause mortality Worsening Safety |
- |
| Ye et al. 2023 [43] | China | Self-controlled case series | - | February 2021 – March 2022 | COVID-19 vaccine | 8 201 | 3 035 | - | 1 523 | 5 166 | - | 2 901 | Hospitalizations MACE |
- |
| Author /year | Vaccinated sample | Dose | Cardiovascular disease | Efficacy All-cause mortality |
Hospitalizations | Revascularization rates | Safety/ MACE |
Conclusions |
|---|---|---|---|---|---|---|---|---|
| Akbar et al. 2025 [40] | 74 236 | 1st: 28 500 2nd: 3 2000 3rd: 13 736 |
CAD: 67 327 (90.70%) HF: 38 017 (51.12%) Hypertension: 57 591 (77.58%) |
1 year (HR= 0.65, 3rd dose) 2 year (HR= 0.40, 3rd dose) |
1 year (HR= 0.85, HF) 2 year (HR= 0.90, HF) 1 year (HR= 0.94, AF) 2 year (HR= 0.93, AF) |
1 year: PCI (HR= 0.86, CAD) 2 year: PCI (HR= 0.87) 1 year: CABG (HR= 0.83) 2 year: CABG (HR= 0.80) |
1 year (HR= 1.43, Myocarditis) 2 year (HR= 1.36, Myocarditis) |
All-cause mortality declined in a dose–response pattern |
| Miró et al. 2025 [39] | 3 139 | - | HF: 3 139 (100%) Hypertension: 2 713 (86.5%) AF: 1 663 (53%) CAD: 717 (22.8%) |
90 days (aHR= 0.829) 1 year (aHR= 0.91) |
aOR= 1.215 | - | - | Vaccination was associated with increased hospitalizations and lower in-hospital mortality |
| Johnson et al. 2022 [41] | 3 898 | 1st: 3 898 2nd: 3 253 3rd: 1 053 |
HF: 3 898 (100%) Hypertension: 2 529 (64.9%) |
HR= 0.87, 1st dose HR= 0.36, 2nd dose |
HR= 0.68 | - | - | Vaccination was associated with a lower likelihood of all-cause hospitalizations and mortality |
| Sindet-Pedersen et al. 2023 [42] | 43 850 | - | HF: 43 850 (100%) Hypertension: 37 712 (86%) AF: 19 401 (44.2%) IHD: 21 055 (48%) AMI: 10 266 (23.4%) Stroke: 5498 (12.5%) |
90 days (Standardized risk= 2.23%) | - | - | 90 days (Standardized risk= 0.01%, Myocarditis) | Vaccination was associated with a slight reduction in mortality. It was not associated with worsening HF or an increased risk of myocarditis |
| Ye et al. 2023 [43] | 3 035 | - | HF: 3 035 (100%) Hypertension: 1 791 (59%) MI: 341 (11.2%) IS: 40 (1.3%) |
- | 0-13 days (IRR= 0.60, HF) 14-27 days (IRR= 0.60, HF) |
- | 0-13 days (IRR= 0.19, MACE) 14-27 days (IRR= 0.10, MACE) |
Hospitalizations and MACE declined in a dose–response pattern |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




