Submitted:
05 March 2026
Posted:
06 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Population and Examination Methods
2.2. Surgical Treatments
2.3. Statistical Analysis
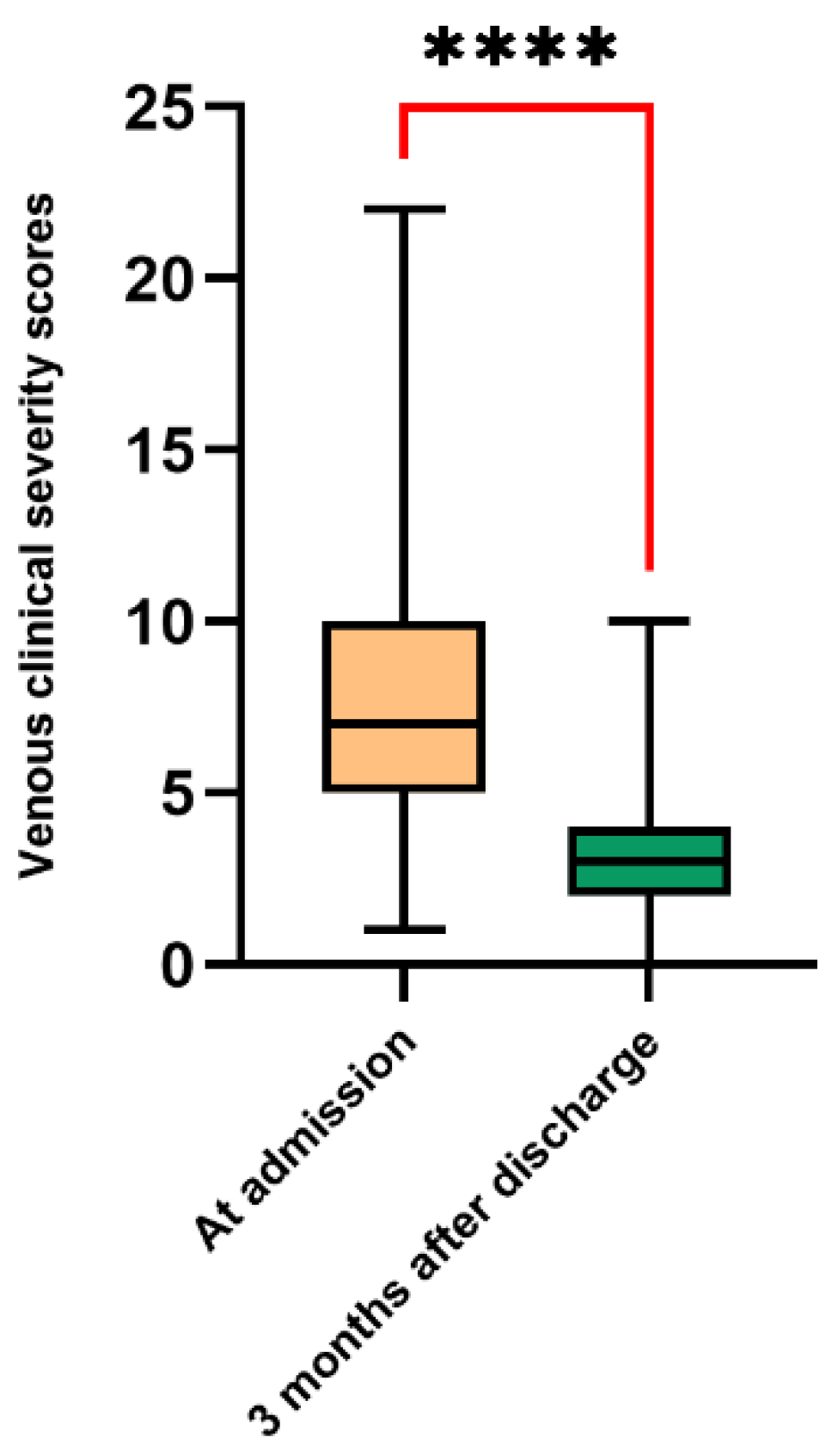
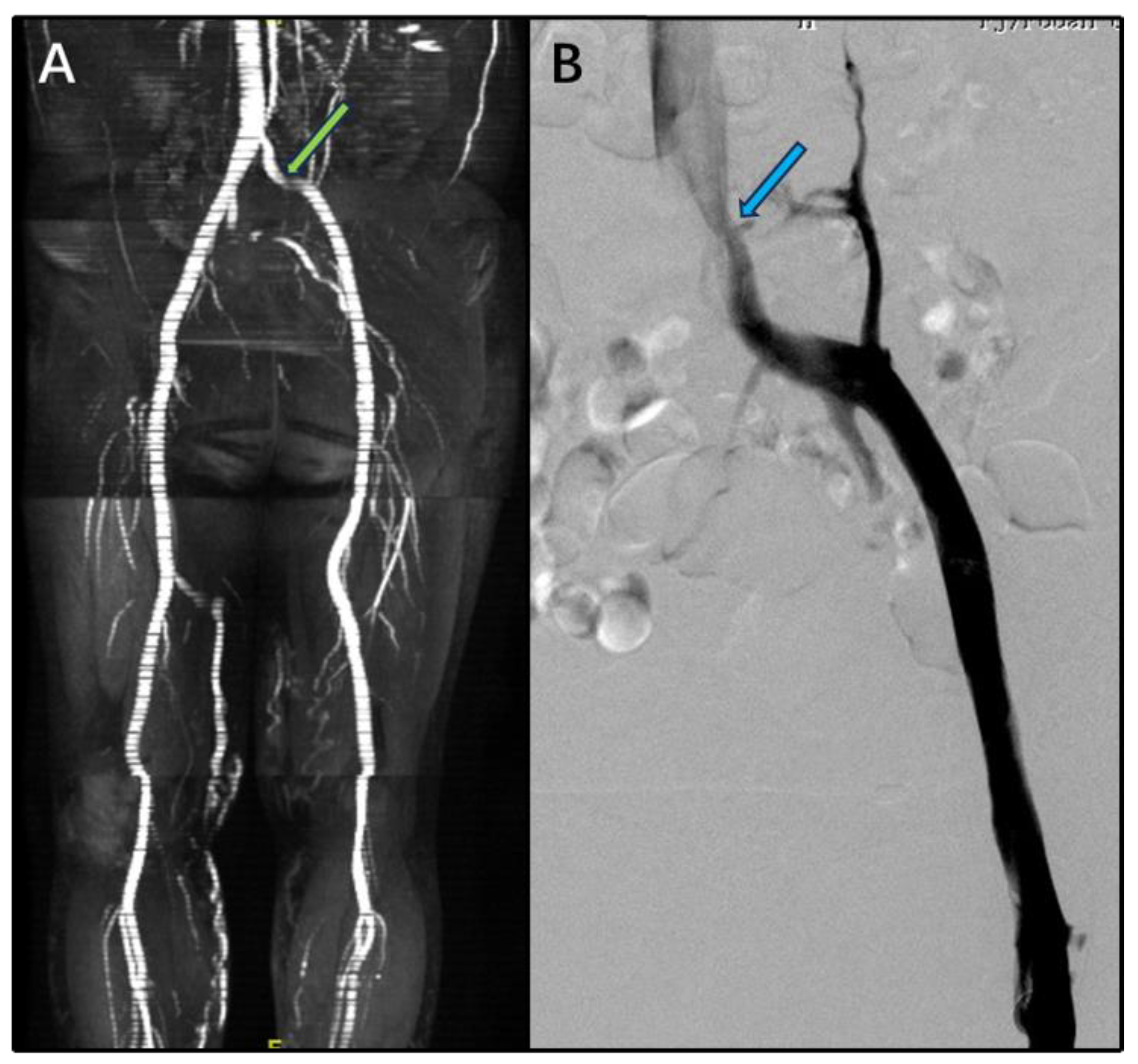
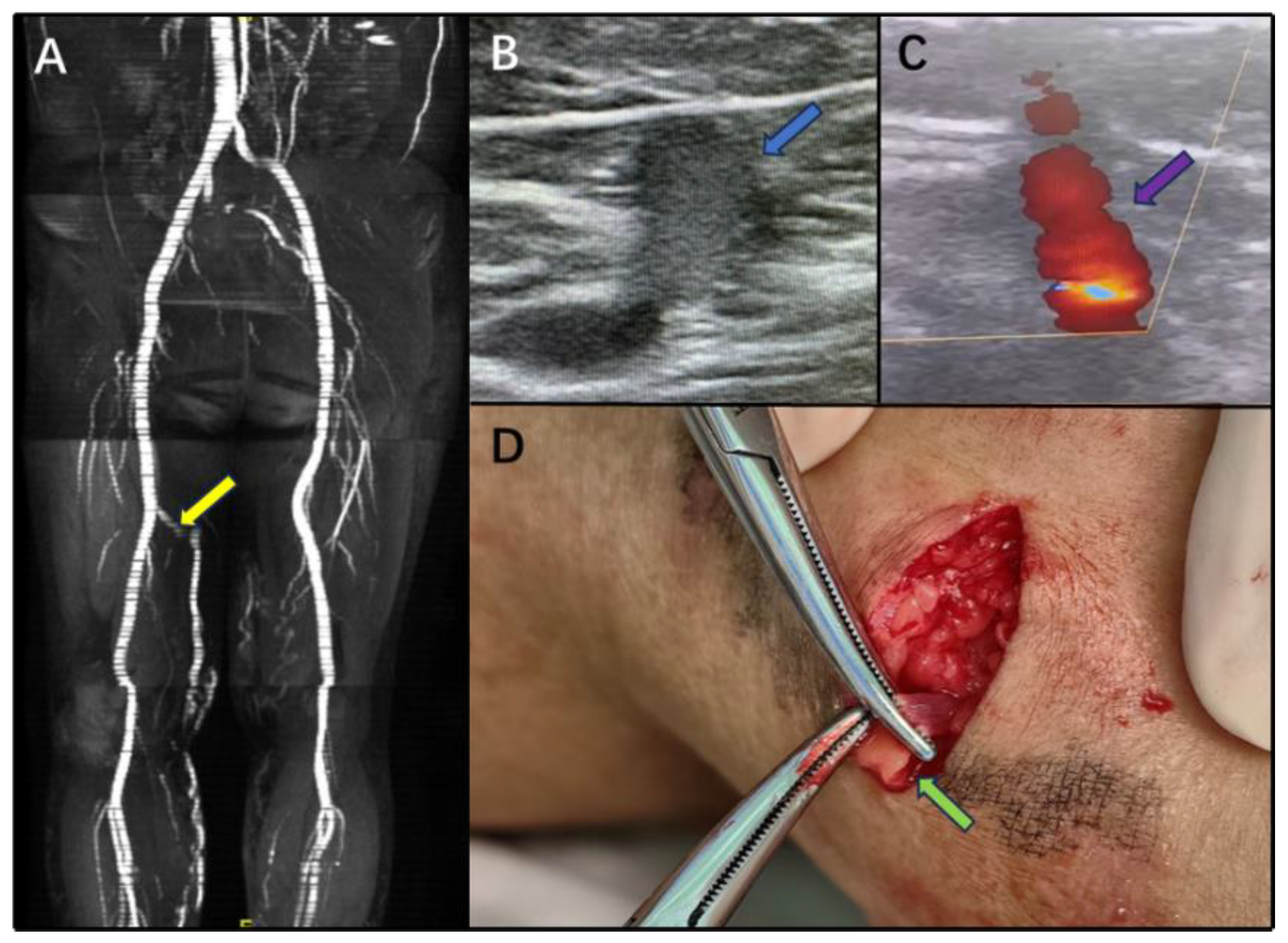
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AASV GSV IPVs LLG NIVCL RLG RVVs SSV HL RFA CEAP RVVs DSA CTV MRV DUS |
anterior accessory saphenous vein great saphenous veins incompetent perforating veins left limbs group nonthrombotic iliac vein compression lesion right limbs group recurrent varicose veins small saphenous vein high ligation of the great saphenous vein radiofrequency ablation of the great saphenous vein clinical, etiological, anatomical, and pathophysiological classification system recurrent varicose veins digital subtraction angiography computed tomography venography magnetic resonance venography duplex ultrasound |
References
- Whing, J.; Nandhra, S.; Nesbitt, C.; et al. Interventions for great saphenous vein incompetence. Cochrane Database Syst Rev 2021, 8(8), CD005624. [Google Scholar] [CrossRef]
- Zhai, Y.; Lu, Y.-M.; Lu, W.; et al. Radiofrequency ablation of the great saphenous vein in the treatment of varicose veins of the lower extremities [J]. Ann Ital Chir 2022, 235. [Google Scholar] [PubMed]
- Nakashima, M.; Kobayashi, M. Endovenous Laser Ablation Combined with Stripping Technique for Large Saphenous Varicose Veins: The Selection of Operation Technique. Annals of Vascular Diseases 2019, 12(4), 514–518. [Google Scholar] [CrossRef]
- Karacalilar, M.; Kutas, H.B. Approach to tortuous vena saphena magna with insufficiency: Foam sclerotherapy. Vascular 2023, 31(5), 1003–1010. [Google Scholar] [CrossRef]
- Zeng, M.; Teng, B.; Zhao, Y.; et al. Effectiveness of iliac vein stenting combined with endovenous laser treatment of recurrent varicose veins associated with iliac vein compression. Quant Imaging Med Surg. 2023, 13(9), 5986–5995. [Google Scholar] [CrossRef]
- Fischer, R.; Chandler, J.G.; Stenger, D.; et al. Patient characteristics and physician-determined variables affecting saphenofemoral reflux recurrence after ligation and stripping of the great saphenous vein. J Vasc Surg. 2006, 43(1), 81–87. [Google Scholar] [CrossRef]
- van Groenendael, L; van der Vliet, JA; Flinkenflögel, L; et al. Treatment of recurrent varicose veins of the great saphenous vein by conventional surgery and endovenous laser ablation. J Vasc Surg. 2009, 50(5), 1106–1113. [Google Scholar] [CrossRef] [PubMed]
- Lv, W.; Wu, X.J.; Collins, M.; et al. Analysis of a series of patients with varicose vein recurrence. J Int Med Res. 2012, 40(3), 1156–1165. [Google Scholar] [CrossRef]
- Svidersky, Y.; Goshchynsky, V.; Migenko, B.; et al. Anterior accessory great saphenous vein as a cause of postoperative recurrence of veins after radiofrequency ablation. J Med Life 2022, 15(4), 563–569. [Google Scholar] [CrossRef] [PubMed]
- Cina, G.; Narese, D.; Amicucci, V.; et al. Recurrent varicose veins of the legs: a single centre experience. Clin Ter. 2014, 165(4), 183–186. [Google Scholar] [CrossRef] [PubMed]
- De Maeseneer, M.G.; Kakkos, S.K.; Aherne, T.; et al. Editor’s Choice - European Society for Vascular Surgery (ESVS) 2022 Clinical Practice Guidelines on the Management of Chronic Venous Disease of the Lower Limbs. Eur J Vasc Endovasc Surg 2022, 63, 184–267. [Google Scholar] [CrossRef]
- Allegra, C.; Antignani, P.L.; Carlizza, A. Recurrent varicose veins following surgical treatment: our experience with five years follow-up. Eur J Vasc Endovasc Surg. 2007, 33(6), 751–756. [Google Scholar] [CrossRef]
- Ozsvath, K; Hager, E; Harlander-Locke, M. Current techniques to treat pathologic perforator veins. J Vasc Surg Venous Lymphat Disord. 2017, 5(2), 293–296. [Google Scholar] [CrossRef]
- Ye, K.; Lu, X.; Li, W.; et al. Long-term outcomes of stent placement for symptomatic nonthrombotic iliac vein compression lesions in chronic venous disease. J Vasc Interv Radiol. 2012, 23(4), 497–502. [Google Scholar] [CrossRef] [PubMed]
- Hua, W.R.; Yi, M.Q.; Jun, W.X.; et al. Causes of recurrent lower limb varicose veins after surgical interventions in 141 limbs - five-year retrospective analysis of two centers. Vascular 2014, 22(4), 267–273. [Google Scholar] [CrossRef] [PubMed]
- Kostas, T.; Ioannou, C.V.; Touloupakis, E.; et al. Recurrent varicose veins after surgery: a new appraisal of a common and complex problem in vascular surgery. Eur J Vasc Endovasc Surg. 2004, 27(3), 275–282. [Google Scholar] [CrossRef]
- Kusagawa, H.; Ozu, Y.; Inoue, K.; et al. Clinical Results 5 Years after Great Saphenous Vein Stripping. Ann Vasc Dis. 2021, 14(2), 112–117. [Google Scholar] [CrossRef] [PubMed]
- BONTINISA; BONTINISV; KOUDOUNASG. Interventions for the treatment of recurrent varicose vein disease arising from the saphenofemoral junction or the great saphenous vein: a systematic review and meta-analysis [J]. Int Angiol 2024, 43(5), 485. [Google Scholar] [CrossRef]
- Gad, M.A.; Saber, A.; Hokkam, E.N. Assessment of causes and patterns of recurrent varicose veins after surgery. N Am J Med Sci. 2012, 4(1), 45–48. [Google Scholar] [CrossRef]
- Zaraca, F.; Ebner, H. Causes and treatment of recurrent varices of the lower limbs. Chir Ital. 2005, 57(6), 761–765. [Google Scholar]
- Musil, D.; Herman, J.; Duda, M. Recurrence of varices in lower extremities. Vnitr Lek. 2004, 50(8), 587–590. [Google Scholar] [PubMed]
- Herman, J.; Musil, D.; Tichy, M.; et al. Recurrent varicose veins: causes and neovascularisation. A 17-year experience. Int Angiol. 2015, 34(1), 53–59. [Google Scholar] [PubMed]
- Cardia, G.; Catalano, G.; Rosafio, I.; et al. Recurrent varicose veins of the legs. Analysis of a social problem. G Chir. 2012, 33(11-12), 450–454. [Google Scholar] [PubMed]
- Perrin, M.; Gillet, J.L. Management of recurrent varices at the popliteal fossa after surgical treatment. Phlebology 2008, 23(2), 64–68. [Google Scholar] [CrossRef]
- Lam, Y.L.; Lawson, J.A.; Toonder, I.M.; et al. Eight-year follow-up of a randomized clinical trial comparing ultrasound-guided foam sclerotherapy with surgical stripping of the great saphenous vein. Br J Surg. 2018, 105(6), 692–698. [Google Scholar] [CrossRef]
| Characteristic | Value |
|---|---|
| Sex [n (%)] | |
| Female | 71 (71.7) |
| Male | 28 (28.3) |
| Mean age (years) | 63.9 ± 9.8 |
| Body mass index | 24.9 ± 2.5 |
| Laterality of RVVs limb [n (%)] | |
| Left | 70 (61.4) |
| Right | 44 (38.6) |
| Clinical CEAP classification [n (%)] | |
| C3 | 16 (14.0) |
| C4 | 74 (64.9) |
| C5 | 4 (3.5) |
| C6 | 20 (17.5) |
| Characteristic | LLG | RLG | t/χ2 | p Value |
|---|---|---|---|---|
| Sex [n (%)] | 0.20 | 0.65 | ||
| Female | 52 (74.3) | 31 (70.5) | ||
| Male | 28 (28.3) | 13 (29.5) | ||
| Mean age (years) | 63.9 ± 9.8 | 63.7 ± 8.7 | −0.18 | 0.81 |
| Body mass index | 24.9 ± 2.5 | 25.6 ± 2.3 | 1.87 | 0.07 |
| Clinical CEAP classification [n (%)] | 1 (2.3) | 3.85 | 0.28 | |
| C3 | 16 (14.0) | 11 (25.0) | ||
| C4 | 74 (64.9) | 31 (70.5) | ||
| C5 | 4 (3.5) | 13 (29.5) | ||
| C6 | 20 (17.5) | 63.7 ± 8.7 |
| Previous surgical treatment | Number of RVVs limbs (%) |
|---|---|
| HL | 56 (49.1) |
| RFA | 3 (2.6) |
| Only varicose vein dissection | 46 (40.3) |
| Only foam sclerotherapy | 9 (8.0) |
| Finding | Number of RVVs limbs (%) |
|---|---|
| Residual main trunk of the GSV | 55 (48.3) |
| Deep venous valves insufficiency | 47 (41.2) |
| NIVCL | 31 (27.2) |
| IPVs | 7 (6.1) |
| AASV insufficiency | 2 (1.8) |
| SSV insufficiency | 2 (1.8) |
| Factor, n (%) | LLG | RLG | t/χ2 | p Value |
|---|---|---|---|---|
| NIVCL | 4.61 | 0.03 | ||
| Yes | 24 (34.3) | 7 (15.9) | ||
| No | 46 (65.7) | 37 (84.1) | ||
| Residual main trunk of the GSV | 1.33 | 0.25 | ||
| Yes | 32 (45.7) | 25 (56.8) | ||
| No | 38 (54.3) | 19 (43.2) | ||
| Deep venous valve insufficiency | 1.51 | 0.22 | ||
| Yes | 32 (45.7) | 15 (34.1) | ||
| No | 38 (54.3) | 29 (65.9) | ||
| IPVs | 0.06 | 0.81 | ||
| Yes | 4 (5.7) | 3 (6.8) | ||
| No | 66 (94.3) | 41 (93.2) | ||
| AASV insufficiency | 0.11 | 0.74 | ||
| Yes | 1 (1.4) | 1 (2.3) | ||
| No | 69 (98.6) | 43 (97.7) | ||
| SSV insufficiency | 1.28 | 0.26 | ||
| Yes | 2 (2.9) | 0 (0) | ||
| No | 68 (97.1) | 44 (100.0) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




