Submitted:
05 March 2026
Posted:
06 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
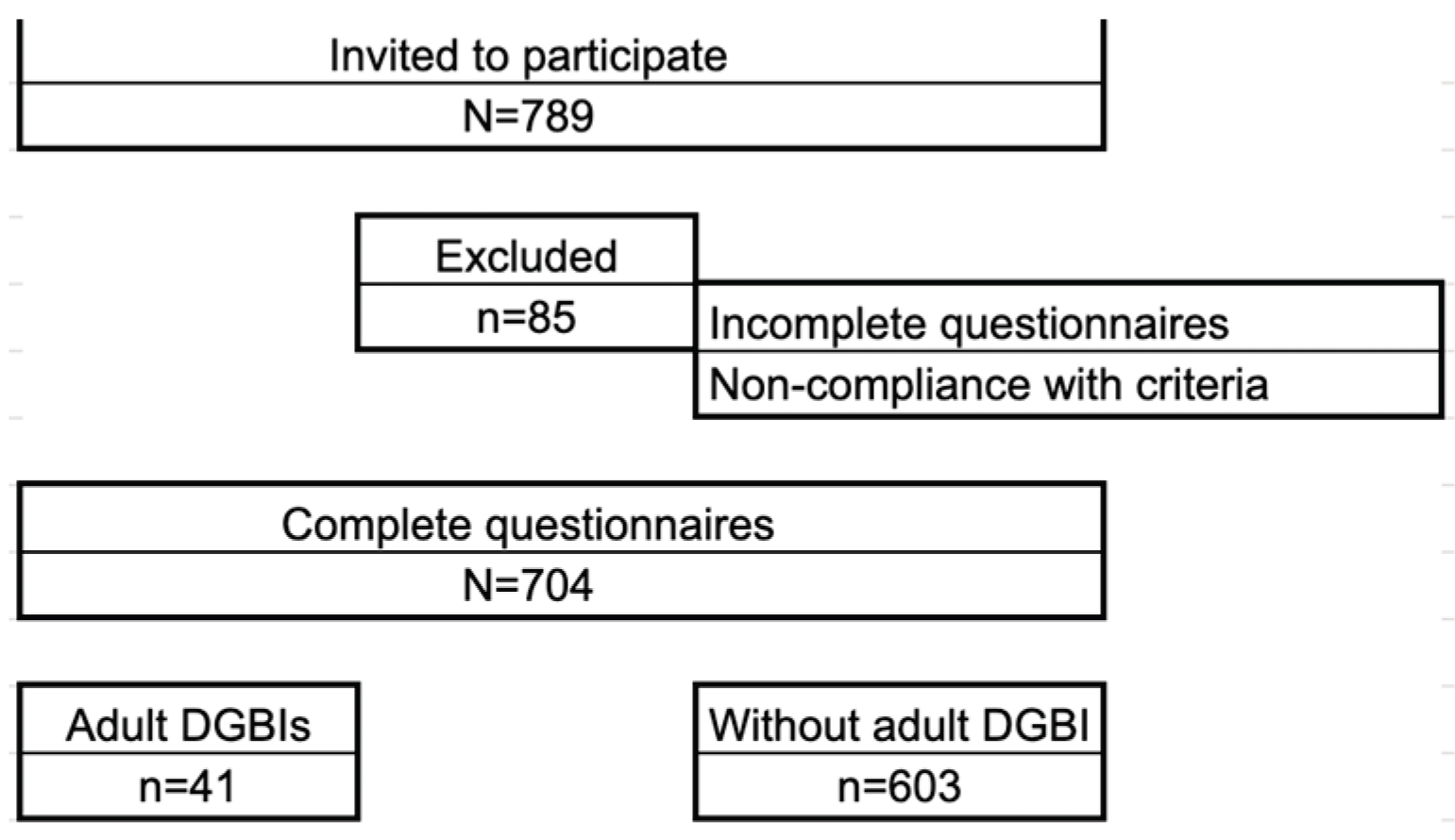
2. Materials and Methods
3. Results
3.1. DGBIs
3.2. Possible Associations with the Presence of Adult DGBIs According to the Rome IV Criteria
3.3. Logistic Regression Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DGBIs | Disorders of the gut–brain interaction |
| QPGS-IV | Questionnaire for Pediatric Gastrointestinal Symptoms Rome IV |
| GERD | Gastroesophageal Reflux Disease |
| PedsQL | Pediatric Quality of Life Inventory™ Questionnaire |
| PROMIS | Patient-Reported Outcomes Measurement Information System |
| 95% Cis | 95% confidence intervals |
| OR | Odds Ratios |
| SD | Standard Deviation |
References
- Ginton, L.; Budhathoki, R.; Saps, M. Reliability of pediatric Rome IV criteria for the diagnosis of disorders of gut–brain interaction. Neurogastroenterol Motil 2024, 36 (7), e14813. [CrossRef]
- Velasco-Benítez, C.A.; Velasco-Suárez, D.A.; Palma, N.; Arrizabalo, S.; Saps, M. The Relationship Between Prematurity and Mode of Delivery with Disorders of Gut-Brain Interaction in Children. Children (Basel) 2025; 12 (6), 799. [CrossRef]
- Vernon-Roberts, A.; Alexander I.; Day, A.S. Systematic Review of Pediatric Functional Gastrointestinal Disorders (Rome IV Criteria). J Clin Med 2021, 10 (21), 5087. [CrossRef]
- Fritz, J.; Coffey, R.; Bloch, J.; Cutler, A.; Gabrielson, S.; DiGiovanni, S.; Faherty, L.J. The relationship between adverse childhood experiences and disorders of the gut-brain interaction. J Pediatr Gastroenterol Nutr 2025, 80 (1), 100–107. [CrossRef]
- Baaleman, D.F.; Velasco-Benítez, C.A.; Méndez-Guzmán, L.M.; Benninga, M.A.; Saps, M. Functional gastrointestinal disorders in children: agreement between Rome III and Rome IV diagnoses. Eur J Pediatr 2021, 180 (7), 2297–2303. [CrossRef]
- Aziz, Q.; Fass, R.; Prakash, C.; Miwa, H.; Pandolfino, J.E.; Zerbib, F. Functional esophageal disorders. Gastroenterology 2016, 150, S0016-5085(16)00178-5. [CrossRef]
- Lanzoni, G.; Sembenini, C.; Gastaldo, S.; Leonardi, L.; Bentivoglio, V.P.; Faggian, G.; Bosa, L.; Gaio, P.; Cananzi, M. Esophageal dysphagia in children: state of the art and proposal for a symptom-based diagnostic approach. Front Pediatr 2022, 10, 885308. [CrossRef]
- Sciumè, G.D.; Berti, G.; Lambiase, C.; Paglianiti, I.; Villanacci, V.; Rettura, F.; Grosso, A.; Ricchiuti, A.; Bortoli, N.; Usai, P.; et al. Misinterpreting diarrhea-predominant irritable bowel syndrome and functional diarrhea: pathophysiological highlights. J Clin Med 2023, 12 (18), 5787. [CrossRef]
- Bhardwaj, R.; Knotts. R.; Khan. A. Functional Chest Pain and Esophageal Hypersensitivity: A Clinical Approach. Gastroenterol Clin North Am 2021, 50 (4), 843-857. [CrossRef]
- Behar, J.; Corazziari, E.; Guelrud, M.; Hogan, W.; Sherman, S.; Toouli, J. Functional gallbladder and sphincter of odi disorders. Gastroenterology 2006, 130 (5), 1498-1509. [CrossRef]
- Carrington, E.V.; Popa, S.L.; Chiarioni, G. Proctalgia Syndromes: Update in Diagnosis and Management. Curr Gastroenterol Rep 2020, 22 (7), 35. [CrossRef]
- Baumann, A.; Katz, P.O. Functional disorders of swallowing. Handb Clin Neurol 2016, 139, 483-488. [CrossRef]
- Drossman, D.A. Rome IV: Functional Gastrointestinal Disorders – Disorders of Gut-Brain Interaction, First edition.; The Rome Foundation: Raleigh (NC), United State, 2016.
- Velasco-Benítez, C.A.; Collazos-Saa, L.I.; García-Perdomo, H.A. A systematic review and meta-analysis in schoolchildren and adolescents with functional gastrointestinal disorders according to Rome IV criteria. Arq Gastroenterol 2022, 59 (2), 304-313. [CrossRef]
- Long, D.; Sintusek, P. Functional constipation in children: What physicians should know. World J Gastroenterol 2023, 29 (8), 1261-1288. [CrossRef]
- Velasco-Benítez, C.A.; Ramírez-Hernández, C.R.; Moreno-Gómez, J.E.; Játiva-Mariño, E.; Zablah, R.; Rodríguez-Reynosa, L.L.; Leyva-Jiménez, S.A.; Chanís, R.; Mejía-Castro, M. Superposición de desórdenes gastrointestinales funcionales en escolares y adolescentes latinoamericanos. Rev Chil Pediatr 2018, 89 (6), 726-731. [CrossRef]
- Flannery, J.; Callaghan, B.; Sharpton, T.; Fisher, P.; Pfeifer, J. Is adolescence the missing developmental link in Microbiome–Gut–Brain axis communication?. Dev Psychobiol 2019, 61 (5), 783-795. [CrossRef]
- Velasco-Benítez, C.A.; Velasco-Suárez, D.A.; Balda, A.; Arrizabalo, S.; Saps, M. Prevalence of functional diarrhea in children and adolescents. Neurogastroenterol Motil 2025, 37 (1), e14950. [CrossRef]
- Sperber, A.D.; Bangdiwala, S.I.; Drossman, D.A.; Ghoshal, U.C.; Simren, M.; Tack, J.; Whitehead, W.E.; Dumitrascu, D.L.; Fang, X.; Fukudo, S. Worldwide prevalence and burden of functional gastrointestinal disorders: results of the Rome Foundation global study. Gastroenterology 2021, 160 (1), 99–114.e3. [CrossRef]
- Sagawa, T.; Okamura, S.; Kakizaki, S.; Zhang, Y.; Morita, K.; Mori, M. Functional gastrointestinal disorders in adolescents and quality of school life. J Gastroenterol Hepatol 2013, 28 (2), 285–290. [CrossRef]
- Horst, S.; Shelby, G.; Anderson, J.; Acra, S.; Polk, D.B.; Saville, B.R.; Garber, J.; Walker, L.S. Predicting persistence of functional abdominal pain from childhood into young adulthood. Clin Gastroenterol Hepatol 2014, 12 (12), 2026–2032. [CrossRef]
- Zanchi, C.; Pintaldi, S.; Di Leo, G.; Ronfani, L.; Zamagni, G.; Viel, M.; Barbi, E.; Cozzi, G. Fifteen-years follow-up in a cohort of children with functional gastrointestinal disorders. Children (Basel) 2021, 8 (10), 838. [CrossRef]
- Azmy, D.J.; Qualia, C.M. Review of abdominal migraine in children. Gastroenterol Hepatol 2020, 16 (12), 632–639.
- Drossman, D.A.; Hasler, W.L. Rome IV—functional GI disorders: disorders of gut-brain interaction. Gastroenterology 2016, 150 (6), 1257–1261. [CrossRef]
- Caram, R. Anorectal functional disorders: towards a better diagnosis and treatment. Arq Gastroenterol 2019, 56 (1), 1–2. [CrossRef]
- Jeyarajah, S.; Purkayastha, S. Proctalgia fugax. CMAJ 2013, 185 (5), 417. [CrossRef]
- Rao, S.S.; Bharucha, A.E.; Chiarioni, G.; Felt-Bersma, R.; Knowles, C.; Malcolm, A.; Wald, A. Functional anorectal disorders. Gastroenterology 2016, S0016-5085(16)00175-X. [CrossRef]
- Palsson, O.S.; Whitehead, W.; Törnblom, H.; Sperber, A.D.; Simren, M. Prevalence of Rome IV functional bowel disorders among adults in the United States, Canada, and the United Kingdom. Gastroenterology 2020, 158 (5), 1262–1273.e3. [CrossRef]
- Na-Na, X.; Wei, J.; Mei-Yun, K.; Hong, X.; Li, T.; Li-Ming, Z.; Sha, Y.; Jiang, J.; Fischer, F. Illness perception of patients with functional gastrointestinal disorders. Front Psychiatry 2018, 9, 122. [CrossRef]
- van Tilburg, M.A.L.; Drossman, D.A.; Knowles, S.R. Psychogastroenterology: the brain-gut axis and its psychological applications. J Psychosom Res 2022, 155, 110684. [CrossRef]
- Reed-Knight, B.; Claar, R.L.; Verrill, J.; van Tilburg, M.A.L. Implementing psychological therapies for functional GI disorders in children and adults. Expert Rev Gastroenterol Hepatol 2016, 10 (9), 981–984. [CrossRef]
- Zia, J.K.; Lenhart, A.; Pei-Lin, Y.; Heitkemper, M.M.; Baker, J.; Keefer, L.; Saps, M.; Cuff, C.; Hungria, G.; Videlock, E. J. Risk factors for abdominal pain-related disorders of gut-brain interaction in adults and children. Gastroenterology 2022, 163 (4), 995–1023.e3. [CrossRef]
- de Bruijn, C.M.A.; Rexwinkel, R.; Gordon, M.; Benninga, M.; Tabbers, M.M. Antidepressants for functional abdominal pain disorders in children and adolescents. Cochrane Database Syst Rev 2021, 2 (2), CD008013. [CrossRef]
- Santucci, N.R.; Velasco-Benítez, C.A.; Cunningham, N.; Li, J.; Fei, L.; Sun, Q.; Saps M. Psychological distress and coping efficacy in children with disorders of gut–brain interaction. Neurogastroenterol Motil 2024, 36 (2), e14724. [CrossRef]
- Liu, L.; Wang, H.; Chen, X.; Zhang, Y.; Zhang, H.; Xie, P. Gut microbiota and its metabolites in depression: from pathogenesis to treatment. EBioMedicine 2023, 91, 104527. [CrossRef]
- Baaleman, D.F.; Velasco-Benítez, C.A.; Méndez-Guzmán, L.M.; Benninga, M.A.; Saps, M. Can we rely on the Rome IV questionnaire to diagnose children with functional gastrointestinal disorders?. J Neurogastroenterol Motil 2021, 27 (4), 626–631. [CrossRef]
- Tersteeg, S.M.; Borowitz, S.M. School absenteeism as a predictor of functional gastrointestinal disorders in children. Front Pediatr 2024, 12, 1503783. [CrossRef]
- Garr, K.; Odar, C.; Flannery, M.; Yacob, D.; Bali, N.; van Diest, A.K. The impact of pediatric disorders of gut-brain interaction on the family. Neurogastroenterol Motil 2025, 37 (6), e70014. [CrossRef]
- Varni, J.W.; Shulman, R.J.; Self, M.M.; Nurko, S.; Saps, M.; Saeed, S.A.; Patel, A.S.; Dark, C.V.; Bendo, C.B.; Pohl, J.F. Gastrointestinal symptoms predictors of health-related quality of life in pediatric patients with functional gastrointestinal disorders. Qual Life Res 2017, 26 (4), 1015–1025. [CrossRef]
- Varni, J.W.; Bendo, C.B.; Nurko, S.; Shulman, R.J.; Self, M.M.; Franciosi, J.P.; Saps, M.; Pohl, J.F.; Pediatric Quality of Life Inventory (PedsQL) Gastrointestinal Symptoms Module Testing Study Consortium. Health-related quality of life in pediatric patients with functional and organic gastrointestinal diseases. J Pediatr 2015, 166 (1), 85–90. [CrossRef]
| Disorder | Definition according to the Rome Criteria in adults |
Questions and answers added to the pediatric QPGS-IV, present during the past month… |
|---|---|---|
| Functional dysphagia | 1. Persistent or recurrent sensation of difficulty in the passage of solid and/or liquid foods 2. Absence of evidence of structural, mucosal, or motor abnormalities explaining the symptom 3. The symptoms are not explained by GERD or major esophageal motility disorders 4. Symptoms must be present during the last 3 months, with onset at least 6 months prior to diagnosis |
Choking sensation Does food or drink get stuck in your chest after swallowing, or does it pass slowly through your chest? How many days? = 4 or more days And For how long? = 2 or more months |
| Functional diarrhea | 1. Predominantly loose or liquid stools without predominant stomachache 2. Increased frequency of bowel movements 3. Absence of criteria for irritable bowel syndrome with diarrhea 4. Symptoms must be present during the last 3 months, with onset at least 6 months prior to diagnosis |
Diarrhea How often does he/she poop? • 3 to 6 times per week, or • Once a day, or • Two to three times a day, or • More than 3 times a day And How is poop according to the Bristol Stool Scale? = Type 6 or 7 And Does it hurt when you poop? = No And What is your main complaint? • Stomachache, or • Loose/mucous stool or stool with undigested food, or • Frequent bowel movements, or • Bloated stomach And Does he/she gain weight normally? = Yes |
| Functional chest pain | 1. Recurrent retrosternal pain or discomfort 2. No evidence of heart disease as the cause of the pain 3. Absence of structural, inflammatory, or motor esophageal disease that explains the symptoms 4. The symptoms are not explained by GERD 5. Symptoms must be present during the last 3 months, with onset at least 6 months prior to diagnosis |
Chest pain or discomfort How many days? = 4 or more days And For how long? = 2 months or more, And How often does he/she experience a burning sensation? = Never And How frequently is it associated with food choking when swallowing? • Occasionally, or • Sometimes, or • Most of the time, or • Always |
| Functional biliary pain | 1. Recurrent episodes of pain located in the epigastrium and/or right upper quadrant 2. The pain reaches a steady intensity and lasts ≥ 30 minutes 3. The pain interferes with daily activities or leads to medical consultation 4. It is not relieved by bowel movements, postural changes, or antacids 5. Absence of gallstones or other structural pathology 6. Symptoms must be present during the last 3 months, with onset at least 6 months prior to diagnosis |
Pain or discomfort in the right upper abdomen quadrant, even if of short duration: How many days? = 4 or more days And For how long? = 2 months or more, And As the hours go by, does he/she improve? = No And As the hours go by, does he/she get worse? = Yes And Does the pain occur at the same time as pooping? = Never And Was the poop softer, or more liquid, than usual? = Never And Was the stool harder or in pieces, than usual? = Never And Does he/she poop more times, than usual? = Never And Does he/she poop less times, than usual? = Never And Does it improve with medications? = Never And Suspension of activities or visit to a doctor or emergency room? = Yes And For how long were the activities suspended? • Between half an hour and 1 hour, or • 1-2 hours, or • 3-4 hours, or • Most of the day, or • All day And Does it improve by changing position from lying down to sitting? = No And/or Does it improve by changing position from sitting to standing? = No And How long does it take before it repeats? • Several hours, or • Several days, or • Several weeks, or • Several months |
| Functional heartburn | 1. Recurrent retrosternal burning sensation 2. Absence of evidence of GERD 3. Absence of esophageal motility disorders or structural disease 4. Symptoms do not respond adequately to antisecretory therapy 5. Symptoms must be present during the last 3 months, with onset at least 6 months prior to diagnosis |
Heartburn (burning, stinging, discomfort) in the chest: How many days? = 4 or more days And For how long? = 2 months or more, And Medications for reflux? = Yes And Does it improve with medication? • Never, or • Occasionally, or • Sometimes, or • Most of the time, or |
| Proctalgia fugax | 1. Recurrent episodes of intense, short-duration rectal pain (seconds to minutes) 2. Absence of anorectal pain between episodes 3. Absence of anorectal structural or inflammatory cause 4. It is not related with bowel movements 5. Symptoms must be present during the last 3 months, with onset at least 6 months prior to diagnosis |
Pain, discomfort, or pressure in the rectum without having a bowel movement: How many days? = 4 or more days And For how long? = 2 months or more, And How long does it last? = More than a minute, but less than 30 minutes And How long does it take before it repeats? • Several hours, or • Several days, or • Several weeks, or • Several months |
| No | 663 (94.2) |
| Yes | 41 (5.8) |
| Proctalgia fugax | 20 (2.8) |
| Functional heartburn | 7 (1.0) |
| Functional diarrhea | 6 (0.9) |
| Functional dysphagia | 4 (0.6) |
| Functional chest pain | 3 (0.4) |
| Functional biliary pain | 1 (0.1) |
| All (n=789) |
Functional chest pain (n=3) |
Functional biliary pain (n=1) |
Proctalgia fugax (n=20) |
Functional heartburn (n=7) |
Functional dysphagia (n=4) |
Functional diarrhea (n=6) |
|
|---|---|---|---|---|---|---|---|
| Sociodemographic variables | |||||||
| Age (years) | |||||||
| X±SD | 13.7±2.8 | 14.2±2.6 | 16.1 | 13.8±3.7 | 13.7±2.5 | 15.3±2.3 | 14.4±1.6 |
| Range | 4-18 | 11-16 | n/a | 4-18 | 10-17 | 13-18 | 12-16 |
| Age groups | |||||||
| Toddlers | 9 (1.1) | 0 (0.0) | 0 (0.0) | 1 (5.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| School-aged children | 214 (27.1) | 1 (33.3) | 0 (0.0) | 4 (20.0) | 3 (42.9) | 0 (0.0) | 0 (0.0) |
| Adolescents | 566 (71.8) | 2 (66.7) | 1 (100.0) | 15 (75.0) | 4 (57.1) | 4 (100.0) | 6 (100.0) |
| Sex | |||||||
| Female | 372 (47.2) | 2 (66.7) | 0 (0.0) | 12 (60.0) | 2 (28.6) | 4 (100.0) | 3 (50.0) |
| Male | 417 (52.8) | 1 (33.3) | 1 (100.0) | 8 (40.0) | 5 (71.4) | 0 (0.0) | 3 (50.0) |
| City | |||||||
| Cucuta | 358 (45.4) | 1 (33.3) | 0 (0.0) | 9 (45.0) | 5 (71.4) | 1 (25.0) | 0 (0.0) |
| Corozal | 270 (34.2) | 1 (33.3) | 0 (0.0) | 9 (45.0) | 0 (0.0) | 0 (0.0) | 6 (100.0) |
| Maicao | 161 (20.4) | 1 (33.3) | 1 (100.0) | 2 (10.0) | 2 (28.6) | 3 (75.0) | 0 (0.0) |
| Race | (n=716) | (n=3) | (n=1) | (n=20) | (n=7) | (n=4) | (n=6) |
| Mixed race | 364 (50.8) | 3 (100.0) | 1 (100.0) | 4 (20.0) | 1 (14.3) | 2 (50.0) | 1 (16.7) |
| White | 221 (30.9) | 0 (0.0) | 0 (0.0) | 9 (45.0) | 6 (85.7) | 1 (25.0) | 2 (33.3) |
| Indigenous | 102 (14.3) | 0 (0.0) | 0 (0.0) | 7 (35.0) | 0 (0.0) | 1 (25.0) | 3 (50.0) |
| Afro-descendant | 29 (4.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Clinical Variables | |||||||
| COVID-19 | (n=704) | (n=3) | (n=1) | (n=20) | (n=7) | (n=4) | (n=6) |
| History of COVID-19 | |||||||
| No | 658 (93.5) | 1 (33.3) | 1 (100.0) | 19 (95.0) | 6 (85.7) | 4 (100.0) | 6 (100.0) |
| Yes | 46 (6.5) | 2 (66.7) | 0 (0.0) | 1 (5.0) | 1 (14.3) | 0 (0.0) | 0 (0.0) |
| COVID-19 vaccination | (n=660) | (n=3) | (n=1) | (n=19) | (n=7) | (n=4) | (n=6) |
| No | 356 (53.9) | 0 (0.0) | 0 (0.0) | 15 (78.9) | 4 (57.1) | 1 (25.0) | 5 (83.3) |
| Yes | 304 (46.1) | 3 (100.0) | 1 (100.0) | 4 (21.1) | 3 (42.9) | 3 (75.0) | 1 (16.7) |
| School/social absenteeism | |||||||
| School absenteeism | (n=704) | (n=3) | (n=1) | (n=20) | (n=7) | (n=4) | (n=6) |
| No | 527 (74.9) | 1 (33.3) | 0 (0.0) | 8 (40.0) | 4 (57.1) | 1 (25.0) | 5 (83.3) |
| Si | 177 (25.1) | 2 (66.7) | 1 (100.0) | 12 (60.0) | 3 (42.9) | 3 (75.0) | 1 (16.7) |
| Reason for school absenteeism | |||||||
| Abdominal pain | 18 (10.2) | 1 (50.0) | 0 (0.0) | 5 (41.7) | 1 (33.3) | 1 (33.3) | 1 (100.0) |
| Headache | 16 (9.0) | 1 (50.0) | 0 (0.0) | 2 (16.7) | 0 (0.0) | 1 (33.3) | 0 (0.0) |
| Nausea | 11 (6.2) | 0 (0.0) | 0 (0.0) | 3 (25.0) | 1 (33.3) | 0 (0.0) | 0 (0.0) |
| Colic | 11 (6.2) | 0 (0.0) | 1 (100.0) | 1 (8.3) | 1 (33.3) | 1 (33.3) | 0 (0.0) |
| Vomiting | 9 (5.1) | 0 (0.0) | 0 (0.0) | 1 (8.3) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Other | 112 (63.3) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Social absenteeism | (n=704) | (n=3) | (n=1) | (n=20) | (n=7) | (n=4) | (n=6) |
| No | 552 (78.4) | 1 (33.3) | 0 (0.0) | 8 (40.0) | 3 (42.9) | 4 (100.0) | 5 (83.3) |
| Yes | 152 (21.6) | 2 (66.7) | 1 (100.0) | 12 (60.0) | 4 (57.1) | 0 (0.0) | 1 (16.7) |
| Social activity | |||||||
| Go out with friends | 35 (23.0) | 0 (0.0) | 0 (0.0) | 4 (20.0) | 1 (25.0) | 0 (0.0) | 0 (0.0) |
| Go out with family | 21 (13.8) | 0 (0.0) | 0 (0.0) | 4 (20.0) | 1 (25.0) | 0 (0.0) | 1 (50.0) |
| Play | 32 (21.0) | 0 (0.0) | 1 (100.0) | 2 (10.0) | 2 (50.0) | 0 (0.0) | 0 (0.0) |
| Play a sport | 37 (24.3) | 1 (50.0) | 0 (0.0) | 2 (10.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Other | 27 (17.8) | 1 (50.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Quality of life | |||||||
| Quality of life according to PedsQL | (n=580) | (n=3) | (n=1) | (n=17) | (n=4) | (n=4) | (n=6) |
| Normal | 414 (71.4) | 0 (0.0) | n/a | 11 (64.7) | 2 (50.0) | 1 (25.0) | 4 (66.7) |
| Altered | 166 (28.6) | 3 (100.0) | 6 (35.3) | 2 (50.0) | 3 (75.0) | 2 (33.3) | |
| Anxiety traits | (n=451) | (n=2) | (n=13) | (n=3) | (n=3) | (n=6) | |
| None to mild | 335 (74.3) | 1 (50.0) | n/a | 6 (46.2) | 1 (33.3) | 0 (0.0) | 2 (33.3) |
| Mild | 80 (17.7) | 1 (50.0) | 3 (23.1) | 1 (33.3) | 1 (33.3) | 3 (50.0) | |
| Moderate | 31 (6.9) | 0 (0.0) | 4 (30.8) | 1 (33.3) | 2 (66.7) | 1 (16.7) | |
| Severe | 5 (1.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| Depressive traits | (n=462) | (n=2) | (n=16) | (n=3) | (n=3) | (n=6) | |
| None to mild | 314 (68.0) | 1 (50.0) | n/a | 6 (37.5) | 2 (66.7) | 0 (0.0) | 2 (33.3) |
| Mild | 98 (21.2) | 0 (0.0) | 7 (43.8) | 0 (0.0) | 1 (33.3) | 4 (66.7) | |
| Moderate | 46 (10.0) | 0 (0.0) | 3 (18.8) | 1 (33.3) | 2 (66.7) | 0 (0.0) | |
| Severe | 4 (0.9) | 1 (50.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| DGBIs in adults | ||||||
| No | Yes | OR | 95% CI | p | ||
| n=663 | n=41 | |||||
| Race | ||||||
| Indigenous | ||||||
| No | 574 (86.6) | 30 (73.2) | 1.00 | 0.0170 | ||
| Yes | 89 (13.4) | 11 (26.8) | 2.36 | 1.02-5.06 | ||
| School absenteeism | ||||||
| No | 508 (76.6) | 19 (46.3) | 1.00 | 0.0000 | ||
| Yes | 155 (23.4) | 22 (53.7) | 3.79 | 1.90-7.60 | ||
| Social absenteeism | ||||||
| No | 535 (80.7) | 17 (41.5) | 1.00 | 0.0000 | ||
| Yes | 128 (19.3) | 24 (58.5) | 5.90 | 2.93-12.03 | ||
| Depressive traits | (n=419) | (n=30) | ||||
| No | 292 (69.7) | 11 (36.7) | 1.00 | 0.0002 | ||
| Yes | 127 (30.3) | 19 (63.3) | 3.97 | 1.73-9.49 | ||
| Quality of life | (n=534) | (n=34) | ||||
| Normal | 388 (72.7) | 18 (52.9) | 1.00 | 0.0135 | ||
| Altered | 146 (27.3) | 16 (47.1) | 2.36 | 1.09-5.04 | ||
| OR | 95% CI | p | |
|---|---|---|---|
| Depressive traits | 4.08 | 1.82-9.12 | 0.001 |
| School absenteeism | 2.51 | 1.06-5.98 | 0.036 |
| Social absenteeism | 4.04 | 1.70-9.62 | 0.002 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




