Submitted:
05 March 2026
Posted:
06 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
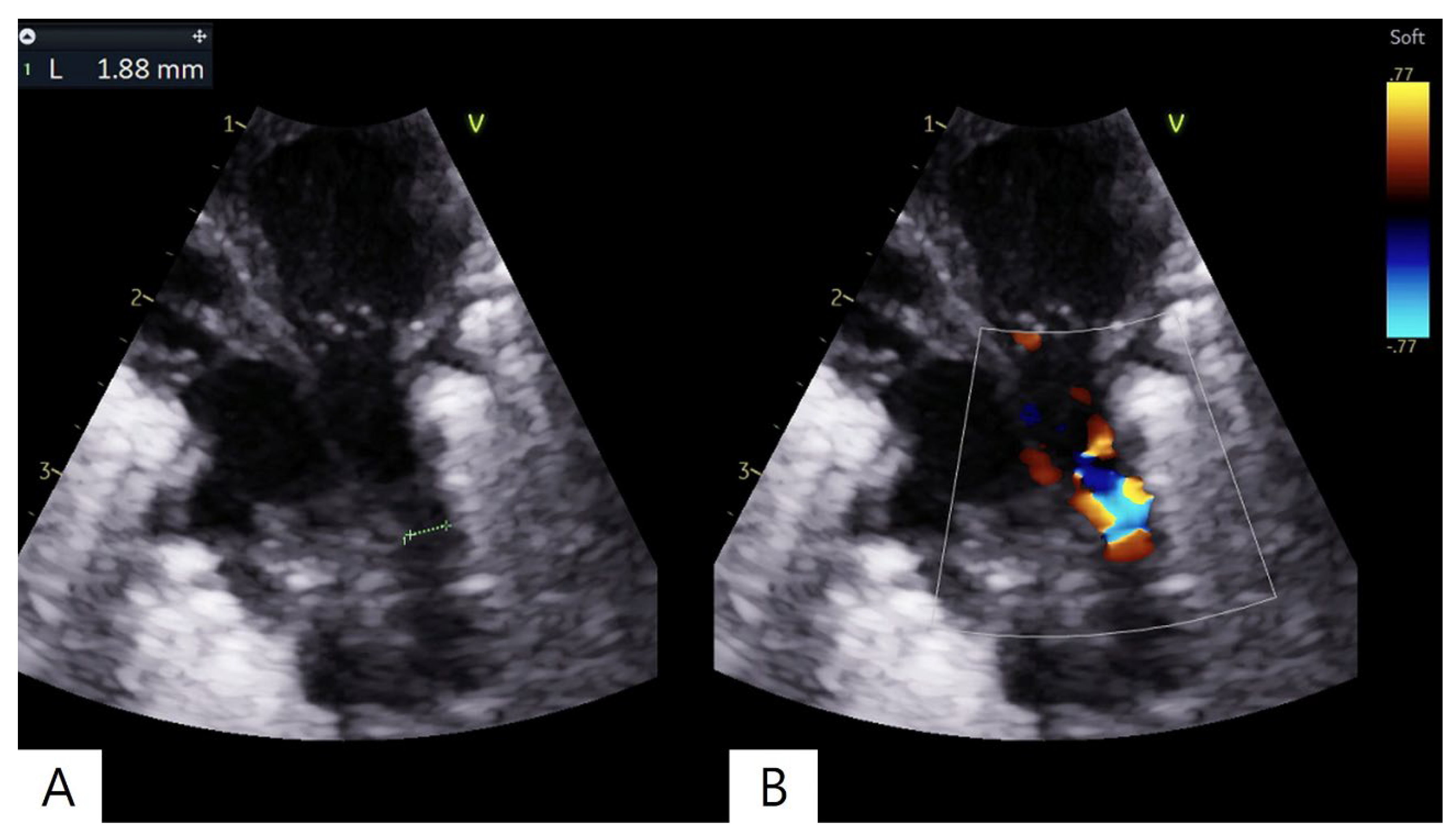
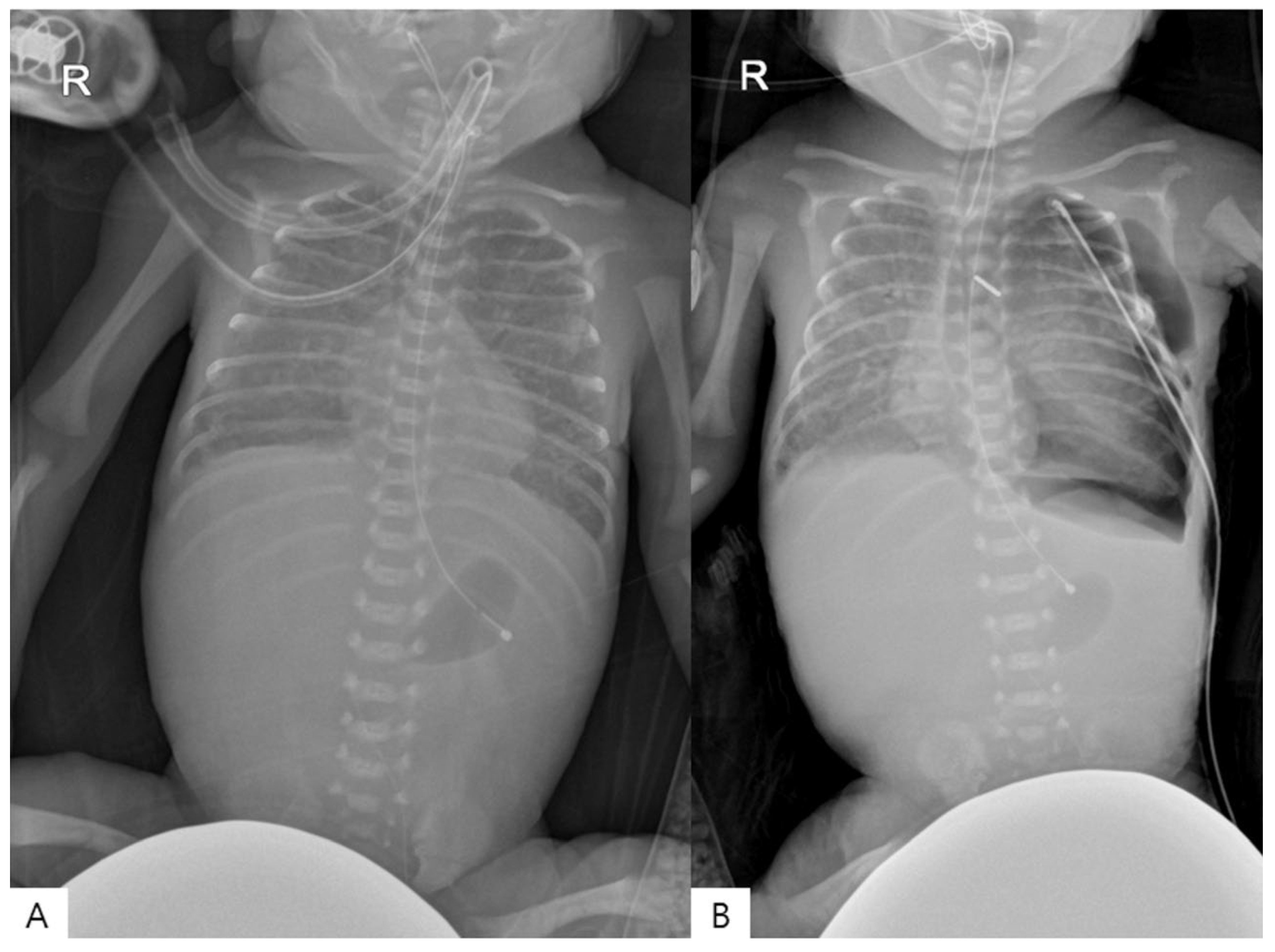
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PDA | Patent ductus arteriosus |
| ELBW | Extremely low birth weight |
| PLCS | Post-ligation cardiac syndrome |
| FiO2 | Fraction of inspired oxygen |
| NIBP | Noninvasive blood pressure |
| NICU | Neonatal intensive care unit |
References
- Wani, G.R.; Parray, N.A.; Lone, M.R.; Ganie, N.A.; Hussain, A.; Islam, J.U.; Mohammad, S.M.; Mohammad, S.M. Association of Patent Ductus Arteriosus (Pda) with Prematurity and Low Birth Weight Neonates. Int. J. Contemp. Pediatrics 2019, 6, 781–785. [Google Scholar] [CrossRef]
- McNamara, P.J.; Sehgal, A. Towards Rational Management of the Patent Ductus Arteriosus: The Need for Disease Staging. Arch. Dis. Child. Fetal Neonatal Ed. 2007, 92, F424–F427. [Google Scholar] [CrossRef] [PubMed]
- Ishida, S.; Yamaguchi, A.; Ooka, M.; Kenmochi, M.; Nakanishi, H. Evaluation of Postoperative Complications for Patent Ductus Arteriosus in Extremely-Low-Birthweight Infants. Pediatr. Int. 2022, 64, e14759. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.S.; Shivananda, S.P.; Stephens, D.; Van Arsdell, G.; McNamara, P.J. Postoperative Cardiorespiratory Instability Following Ligation of the Preterm Ductus Arteriosus Is Related to Early Need for Intervention. J. Perinatol. 2008, 28, 803–810. [Google Scholar] [CrossRef] [PubMed]
- Raval, M.V.; Laughon, M.M.; Bose, C.L.; Phillips, J.D. Patent Ductus Arteriosus Ligation in Premature Infants: Who Really Benefits, and at What Cost? J. Pediatr. Surg. 2007, 42, 69–75, discussion 75. [Google Scholar] [CrossRef] [PubMed]
- Mandhan, P.; Brown, S.; Kukkady, A.; Samarakkody, U. Surgical Closure of Patent Ductus Arteriosus in Preterm Low Birth Weight Infants. Congenit. Heart Dis. 2009, 4, 34–37. [Google Scholar] [CrossRef] [PubMed]
- Noori, S.; Friedlich, P.; Seri, I.; Wong, P. Changes in Myocardial Function and Hemodynamics after Ligation of the Ductus Arteriosus in Preterm Infants. J. Pediatr. 2007, 150, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Bary, M.; Abdel-Baseer, K.A.; Abdel-Latif, A.F.; Abdel-Naser, M.A.; Nafie, M.; Eisa, K.M. Left Ventricular Dysfunction Postsurgical Patent Ductus Arteriosus Ligation in Children: Predictor Factors Analysis. J. Cardiothorac. Surg. 2019, 14, 168. [Google Scholar] [CrossRef] [PubMed]
- Ross, J., Jr. Afterload Mismatch and Preload Reserve: A Conceptual Framework for the Analysis of Ventricular Function. Prog. Cardiovasc. Dis. 1976, 18, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Zarrabi, K.; Ravanfar, P.; Azimifar, A.; Ghaffarpasand, F. Profound Bradycardia Following Patent Ductus Arteriosus Closure; a Rare but Correctable Event. Bull. Emerg. Trauma 2013, 1, 130–132. [Google Scholar] [PubMed]
- Choudhury, M.; Gupta, A.; Krithika, K.; Bharadwaj, V.; Devagourou, V. Ligation of Patent Ductus Arteriosus in Preterm Infant with Comorbidities: An Unforgotten Nightmare. J. Neonatal Crit. Care Anesth. 2024, 1, 46–48. [Google Scholar] [CrossRef]
- Imran, J.B.; Eastman, A.L. Pneumothorax. JAMA 2017, 318, 974. [Google Scholar] [CrossRef] [PubMed]
- Sehgal, A.; Francis, J.V.; Lewis, A.I. Use of Milrinone in the Management of Haemodynamic Instability Following Duct Ligation. Eur. J. Pediatr. 2011, 170, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Samiee-Zafarghandy, S.; Raman, S.R.; van den Anker, J.N.; McHutchison, K.; Hornik, C.P.; Clark, R.H.; Brian Smith, P. Best Pharmaceuticals for Children Act-Pediatric Trials Network Administrative Core, C. Safety of Milrinone Use in Neonatal Intensive Care Units. Early Hum. Dev. 2015, 91, 31–35. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



