Submitted:
04 March 2026
Posted:
05 March 2026
You are already at the latest version
Abstract
Keywords:
Background
Methods
Results
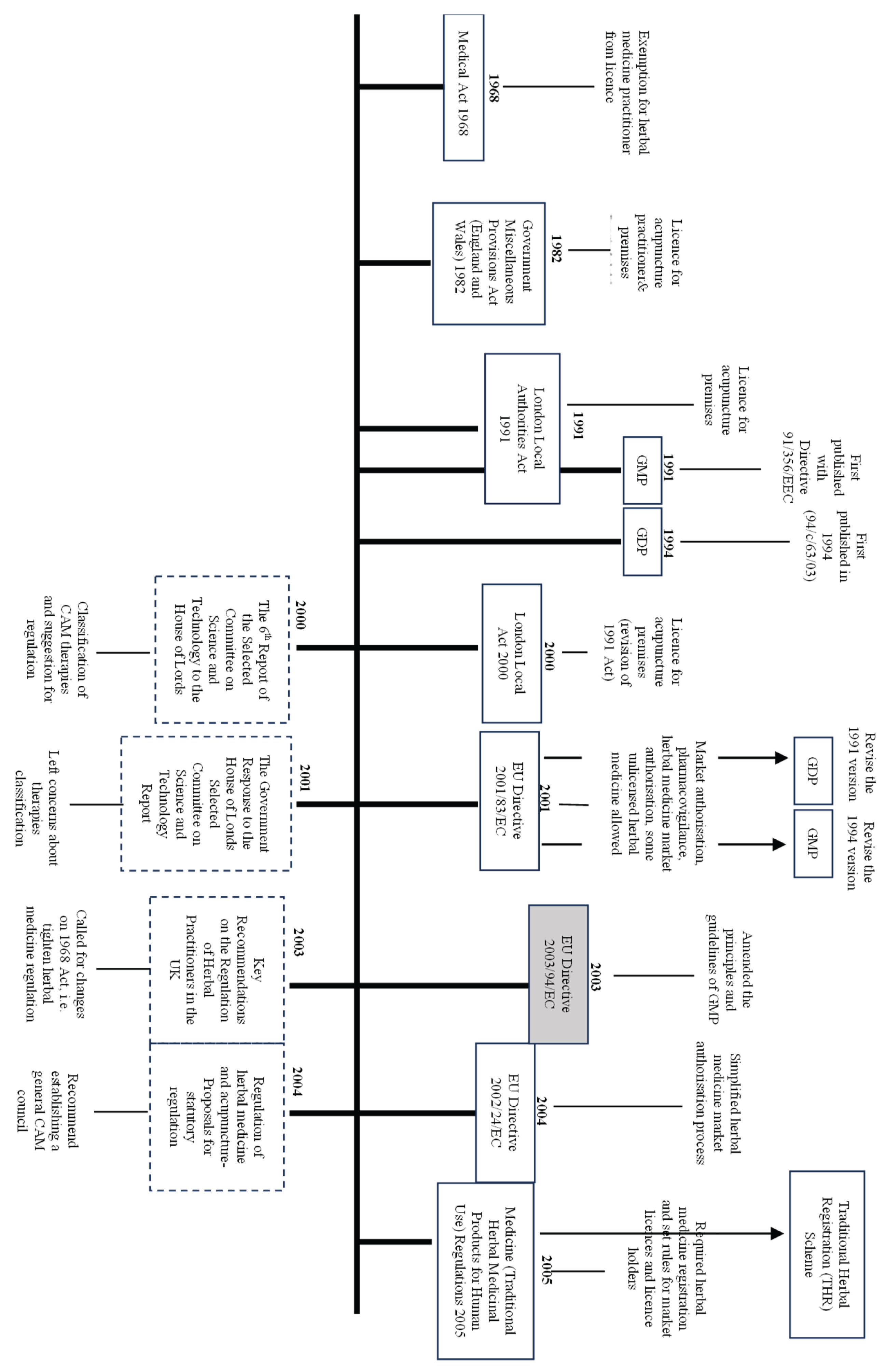
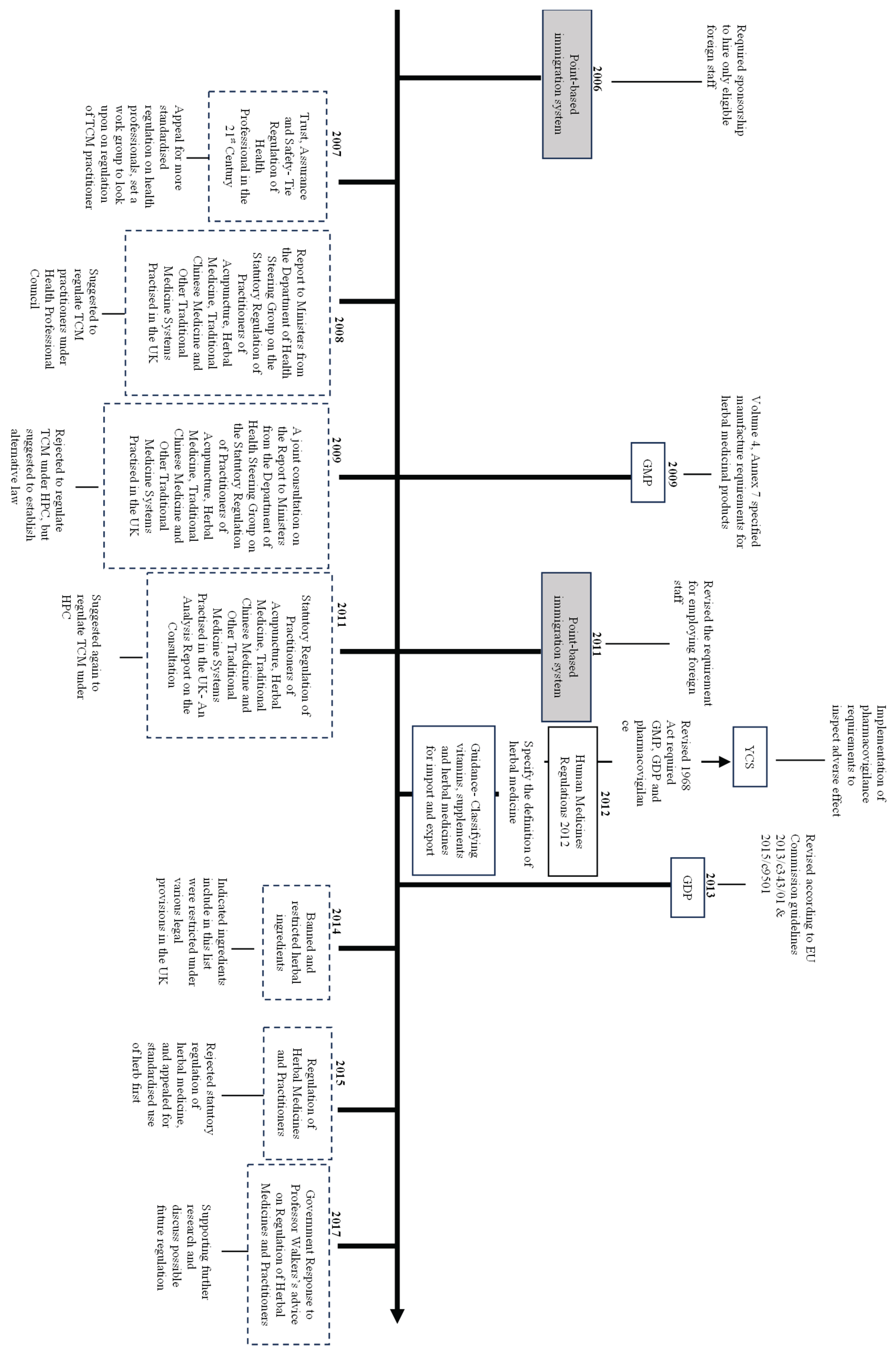
The Historical Development of TCM Regulations in the UK
Discussion
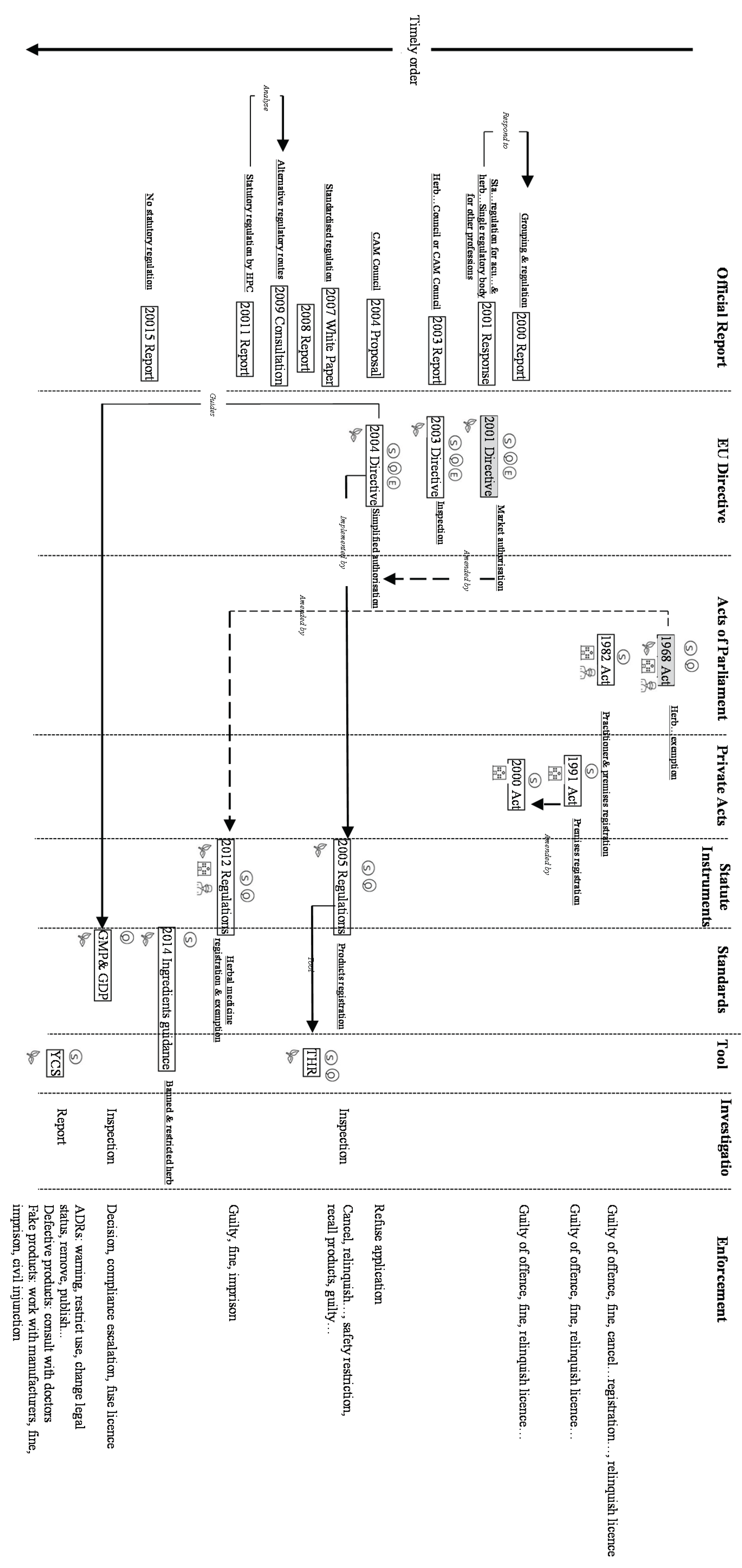
Classification of the Existing Regulations
- All TCM-related regulations have been published to stakeholders and the public as a warning approach to prevent non-compliance. The European Commission has issued specialised guidance within the GMP and GDP frameworks to help the industry comply with the latest rules on medicine manufacturing and distribution [61].
- The 1982 Act, the 2004 Directive, and THR Scheme requires evidence to identify and assess compliance, while the GMP and GDP take inspection as an identification approach.
- The THR Scheme follows the principles of the 2005 Regulations to investigate non-compliance by checking related documents; the Yellow Card Scheme involves a more systematic investigation procedure conducted by the MHRA, manufacturers or medical specialists.
- All TCM regulations have sanctions prepared for non-compliance. Sanctions include being guilty of an offence and facing a fine and changes to licence status. The 2005 Regulations can enforce urgent safety restrictions and the withdrawal of products. The 2012 Regulations can impose infringement notices and sentence offenders with penalties applicable to medicines breaching provisions, pharmacovigilance regulations and information and labelling requirements; the 2012 Regulations and its related Yellow Card Scheme can apply penalties and prison sentences for breaching the requirements of the THR, labelling, and advertisements for traditional herbal medicinal products. The GMP and GDP have a compliance escalation procedure to ensure satisfactory improvements in manufacturing and distribution.
- As outcomes, all the regulations expect that illegal activities will cease and, if possible, that no repeat offences occur. Only the two MHRA leading regulatory schemes, the THR and the Yellow Card Scheme, can be mapped across all five stages set by the enforcement framework, but all the regulations clearly define their sanctions for non-compliance to guarantee implementation.
Summary of the Regulatory Structure
Conclusions
References
- Ryan, A. Globalisation and the ‘Internal Alchemy’ In Chinese Martial Arts: The Transmission of Taijiquan to Britain. East Asian Science Technology and Society an International Journal 2008, 2. [Google Scholar] [CrossRef]
- Zhuang, G. The Chinese Immigration for the Recent 30 Years: The Example of Fuzhou. World Ethno-National Studies in Chinese. 2006, 3, 77–83. [Google Scholar]
- Latham, K.; Wu, B. Chinese Immigration into the EU: New Trends, Dynamics and Implications; Europe China Research and Advice Network: London, 2013. [Google Scholar]
- Uzuner, Ö.; Solti, I.; Cadag, E. Extracting medication information from clinical text. Journal of the American Medical Informatics Association 2010, 17(no. 5). [Google Scholar] [CrossRef] [PubMed]
- Liu, Y. Barriers and Countermeasures in Developing Traditional Chinese Medicine in Europe. Frontiers of Medicine 2016, 10, 360–76. [Google Scholar] [CrossRef] [PubMed]
- Fung, F. Y.; Linn, Y. C. Developing Traditional Chinese Medicine in the Era of Evidence-Based Medicine: Current Evidences and Challenges. In Evidence-Based Complementary and Alternative Medicine; 2015. [Google Scholar]
- Wang, S.; Kong, D. The Current Status of TCM in Different Countries and Regions of the World. Asian Traditional Medicine in Chinese. 2006, 8, 5–22. [Google Scholar]
- Mills, S. Y. Regulation in Complementary and Alternative Medicine. BMJ 2001, 322(no. 7279), 158–60. [Google Scholar] [CrossRef]
- Denham, M. A.; Onwuegbuzie, A. J. Beyond Words: Using Nonverbal Communication Data in Research to Enhance Thick Description and Interpretation. International Journal of Qualitative Methods 2013, 12(no. 1), 670–96. [Google Scholar] [CrossRef]
- Brown, P. S. The Vicissitudes of Herbalism in Late Nineteenth- and Early Twentieth-Century Britain. Medical History 1985, 29, 71–92. [Google Scholar] [CrossRef]
- Pharmacy and Medicines Bill, 1941 (UK).
- Medicines Act 1968, c. 67 (UK).
- Wilkinson. Professional Perspectives in Health Care; Bloomsbury Publishing: London, 2007. [Google Scholar]
- Dixon, L. Moving in from the Fringes: The Regulation of Complementary and Alternative Medical Practitioners in the UK. PhD diss., London School of Economics and Political Science, 2007. Available online: https://ethos.bl.uk/OrderDetails.do?uin=uk.bl.ethos.443796 (accessed on June 2021).
- Cloatre, E.; Ramas, F. S. The Regulation of Acupuncture in France and the UK: Shifts and Fragmentation in Contrasting Healthcare Systems. Medical Law International 2019, 19, 235–57, at 247. [Google Scholar] [CrossRef] [PubMed]
- Local Government (Miscellaneous Provisions) Act 1982, c. 30 (UK).
- London Local Authorities Act 1991, c. xlv (UK).
- London Local Authorities Act 2000, c. vii (UK).
- UK, House of Lords, Science and Technology Committee, Sixth Report (1999–2000 Session), HL Paper 123. Available online: https://publications.parliament.uk/pa/ld199900/ldselect/ldsctech/123/12301.htm (accessed on June 2021).
- UK, Department of Health, Government Response to the House of Lords Science and Technology Committee’s Report on Complementary and Alternative Medicine (London: Department of Health, 2001). Available online: https://publications.parliament.uk/pa/ld199900/ldselect/ldsctech/123/123resp.htm (accessed on June 2021).
- Directive 2001/83/EC of the European Parliament and of the Council of 6 November 2001 on the Community code relating to medicinal products for human use, 2001 O.J. (L 311) 67.
- UK, Herbal Medicine Regulatory Working Group. A Summary Report from the Herbal Medicine Regulatory Working Group: Key Recommendations on the Regulation of Herbal Practitioners in the UK; Department of Health: London, 2003. [Google Scholar]
- Commission Directive 2003/94, 2003 O.J. (L 262) 22 (EC).
- Directive 2004/24, 2004 O.J. (L 136) 85 (EC).
- European Commission. EudraLex, Volume 4: Good Manufacturing Practice Guidelines (2022). Available online: https://health.ec.europa.eu/system/files/2022-02/20220225_gmp_guidelines_vol4_en.pdf.
- European Commission. Guidelines on Good Distribution Practice of Medicinal Products for Human Use (2013/C 343/01), 2013 O.J. (C 343) 1.
- European Commission. Guidelines on Principles of Good Distribution Practice of Active Substances for Medicinal Products for Human Use (2015/C 95/01), 2015 O.J. (C 95) 1.
- UK Government, Good Manufacturing Practice and Good Distribution Practice (2020). Available online: https://www.gov.uk/guidance/good-manufacturing-practice-and-good-distribution-practice (accessed on Mar. 2021).
- U.K. Dep’t of Health, Regulation of Herbal Medicine and Acupuncture: Proposals for Statutory Regulation; Mar 2004.
- SI 2005/2750 (UK); Medicines (Traditional Herbal Medicinal Products for Human Use) Regulations 2005.
- Medicines & Healthcare Products Regulatory Agency (MHRA), Herbal Medicines Granted a Traditional Herbal Registration (THR) (2014). Available online: https://www.gov.uk/guidance/apply-for-a-traditional-herbal-registration-thr (accessed on June 2021).
- Medicines; Healthcare Products Regulatory Agency (MHRA). Herbal Medicines Granted a Traditional Herbal Registration (THR) (2019). Available online: https://www.gov.uk/government/publications/herbal-medicines-granted-a-traditional-herbal-registration-thr/herbal-medicines-granted-a-traditional-herbal-registration (accessed on June 2021).
- Barber, S. Regulation of Herbal Medicines, House of Commons Library, Science and Environment Section, Standard Note SN/SC/6002; House of Commons Library: London, 4 Apr 2014; p. 4. [Google Scholar]
- Home Office, A Points-Based System: Making Migration Work for Britain; Home Office: Norwich, 2006.
- Department of Health and Social Care, Trust, Assurance and Safety – The Regulation of Health Professionals in the 21st Century; The Stationery Office: London, 2007.
- Chief Medical Officer, Good Doctors, Safer Patients; Department of Health: London, 2006.
- Department of Health, The Regulation of the Non-Medical Healthcare Professions: A Review by the Department of Health; Department of Health: London, 2006.
- Department of Health. Report to Ministers from the Department of Health Steering Group on the Statutory Regulation of Practitioners of Acupuncture, Herbal Medicine, Traditional Chinese Medicine and Other Traditional Medicine Systems Practised in the UK; Department of Health: London, 2008. [Google Scholar]
- Department of Health. Joint Consultation on the Report to Ministers from the Department of Health Steering Group on the Statutory Regulation of Practitioners of Acupuncture, Herbal Medicine, Traditional Chinese Medicine and Other Traditional Medicine Systems Practised in the UK; Department of Health: London, 2009. [Google Scholar]
- Department of Health; Professional Standards Division. Statutory Regulation of Practitioners of Acupuncture, Herbal Medicine, Traditional Chinese Medicine and Other Traditional Medicine Systems Practised in the UK: An Analysis Report on the Consultation; Department of Health: London, 2011. [Google Scholar]
- British Acupuncture Council (BAcC). Statutory Regulation for Acupuncture (2011). Available online: https://www.acupuncture.org.uk/public-content/public-regulation-updates/1323-statutory-regulation-for-acupuncture.html (accessed on January 2022).
- Human Medicines Regulations 2012, SI 2012/1916 (UK).
- Medicines and Healthcare products Regulatory Agency (MHRA), MHRA-GMDP Database. Available online: https://cms.mhra.gov.uk/mhra (accessed on September 2025).
- Medicines and Healthcare products Regulatory Agency (MHRA), Yellow Card Scheme (YCS). Available online: https://yellowcard.mhra.gov.uk/ (accessed on September 2025).
- Gov.uk. Guidance- Classifying vitamins, supplements and herbal medicines for import and export. 2012. Available online: https://www.gov.uk/guidance/classifying-vitamins-supplements-and-herbal-medicines-for-import-and-export.
- Medicines and Healthcare products Regulatory Agency (MHRA), Banned and Restricted Herbal Ingredients (2014). Available online: https://www.gov.uk/government/publications/list-of-banned-or-restricted-herbal-ingredients-for-medicinal-use/banned-and-restricted-herbal-ingredients (accessed on June 2021).
- Walker, Report on the Regulation of Herbal Medicines and Practitioners; Department of Health and the Medicines and Healthcare products Regulatory Agency: London, 2015.
- Blackwood, N. Government Response to Professor Walker’s Advice on Regulation of Herbal Medicines and Practitioners (2017). Available online: https://questions-statements.parliament.uk/written-statements/detail/2017-02-28/HCWS505 (accessed on January 2022).
- European Commission. Better Regulation Toolbox. n.d. Available online: https://ec.europa.eu/info/law/law-making-process/planning-and-proposing-law/better-regulation-why-and-how/better-regulation-guidelines-and-toolbox/better-regulation-toolbox-0_en (accessed on January 2022).
- Hatzopoulos, V. From Hard to Soft: Governance in the EU Internal Market. Cambridge Yearbook of European Legal Studies 2013, 15, 101–37. [Google Scholar] [CrossRef]
- Versluis. “Compliance Problems in the EU—What Potential Role for Agencies in Securing Compliance”. paper presented at the 3rd ECPR General Conference, Budapest, September 8–10, 2005. [Google Scholar]
- European Union (Withdrawal Agreement) Act 2020.
- WHO. Effective Medicines Regulation: Ensuring Safety, Efficacy and Quality; World Health Organization: Geneva, 2003; Available online: https://apps.who.int/iris/handle/10665/68391 (accessed on June 2021).
- House of Commons, Safety, Quality, Efficacy: Regulating Medicines in the UK, Twenty-Sixth Report of Session 2002–03; The Stationery Office Limited: London, 2003; pp. 3–5.
- National Institute for Health and Care Excellence (NICE). Chronic Pain: Assessment and Management—Evidence Review for Acupuncture, NICE Guideline NG193 (London: NICE, 2020). Available online: https://www.nice.org.uk/guidance/ng193/documents/evidence-review-7 (accessed on June 2021).
- NHS, Acupuncture. n.d. Available online: https://www.nhs.uk/conditions/acupuncture/ (accessed on January 2022).
- Cloatre; Ramas, F. S. The Regulation of Acupuncture in France and the UK: Shifts and Fragmentation in Contrasting Healthcare Systems. Medical Law International 2019, 19, 235–57. [Google Scholar] [CrossRef] [PubMed]
- Boxtel, C. J.; Santoso, B.; Edwards, I. R. Drug Benefits and Risks: International Textbook of Clinical Pharmacology, rev. 2nd ed.; IOS Press: Amsterdam, 2008. [Google Scholar]
- Regulatory Enforcement and Sanctions Act 2008, c. 13 (UK).
- Medicines and Healthcare products Regulatory Agency (MHRA), Enforcement Strategy; MHRA: London, 2010.
- Medicines and Healthcare products Regulatory Agency (MHRA), New Essential Orange and Green Guides 2017 – Out Now. 2017. Available online: https://www.gov.uk/government/news/new-essential-orange-and-green-guides-2017-out-now--2 (accessed on June 2021).
- World Health Organization. General Guidelines for Methodologies on Research and Evaluation of Traditional Medicine, WHO/EDM/TRM/2000.1; World Health Organization: Geneva, 2000. [Google Scholar]

|
Traditional Chinese medicine OR Chinese medicine OR Chinese traditional Medicine OR Herbal medicine OR Acupuncture OR Complementary medicine OR Alternative medicine OR Complementary and alternative medicine |
AND |
OR Develop OR Regulatory system OR Regulatory regime OR Legislation OR Law |
AND |
the UK OR British OR Europe OR European Union |
| Regulation level | Regulatory instruments | Regulation |
| EU level | Hard regulation —Directive | EU Directive 2001/83/EC & 2004/24/EC |
| Soft regulation —self regulation | Industry self-regulation | |
| Soft regulation —technical standards | GMP | |
| GDP | ||
| Soft regulation —OMC | Centralised and decentralised market authorisation | |
| Education and information | Part of hard or soft regulations | |
| Economic instruments | Part of hard or soft regulations | |
| UK level | Acts of parliament | The Local Government (Miscellaneous Provision) Act 1982 |
| Private acts | The London Local Authorities Act 1991 & 2000 | |
| Statutory instruments (Secondary law) | The Medicines (Traditional Herbal Medicinal Products for Human Use) Regulation 2005 | |
| The Human Medicines Regulations 2012 | ||
| Schemes | THR Scheme | |
| Yellow Card Scheme |
| Regulations | Safety | Quality | Efficacy |
| Local Government (Miscellaneous Provision) Act 1982 | ✓ | ||
| London Local Authorities Act 1991 | ✓ | ||
| London Local Authorities Act 2000 | ✓ | ||
| EU Directive 2004/24/EC | ✓ | ✓ | ✓ |
| The Medicines Regulations 2005 | ✓ | ✓ | |
| Traditional Herbal Registration (THR) Scheme | ✓ | ✓ | |
| Human Medicines Regulations 2012 | ✓ | ✓ | |
| GMP | ✓ | ||
| GDP | ✓ | ||
| Yellow Card Scheme | ✓ |
| Regulation | Practice | Products | People | Premises |
| Local Government Act 1982 | ✓ | ✓ | ||
| London Local Authorities Act 1991 | ✓ | |||
| London Local Authorities Act 2000 | ✓ | |||
| EU Directive 2004/24/EC | ✓ | |||
| The Medicines Regulations 2005 | ✓ | |||
| Traditional Herbal Registration (THR) Scheme |
✓ | |||
| Human Medicines Regulations 2012 | ✓ | ✓ exemption | ✓ exemption | |
| GMP | ✓ | ✓sub-requirements | ✓ sub-requirements | |
| GDP | ✓ | |||
| Yellow Card Scheme | ✓ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




