Submitted:
16 February 2026
Posted:
05 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Material and Methods
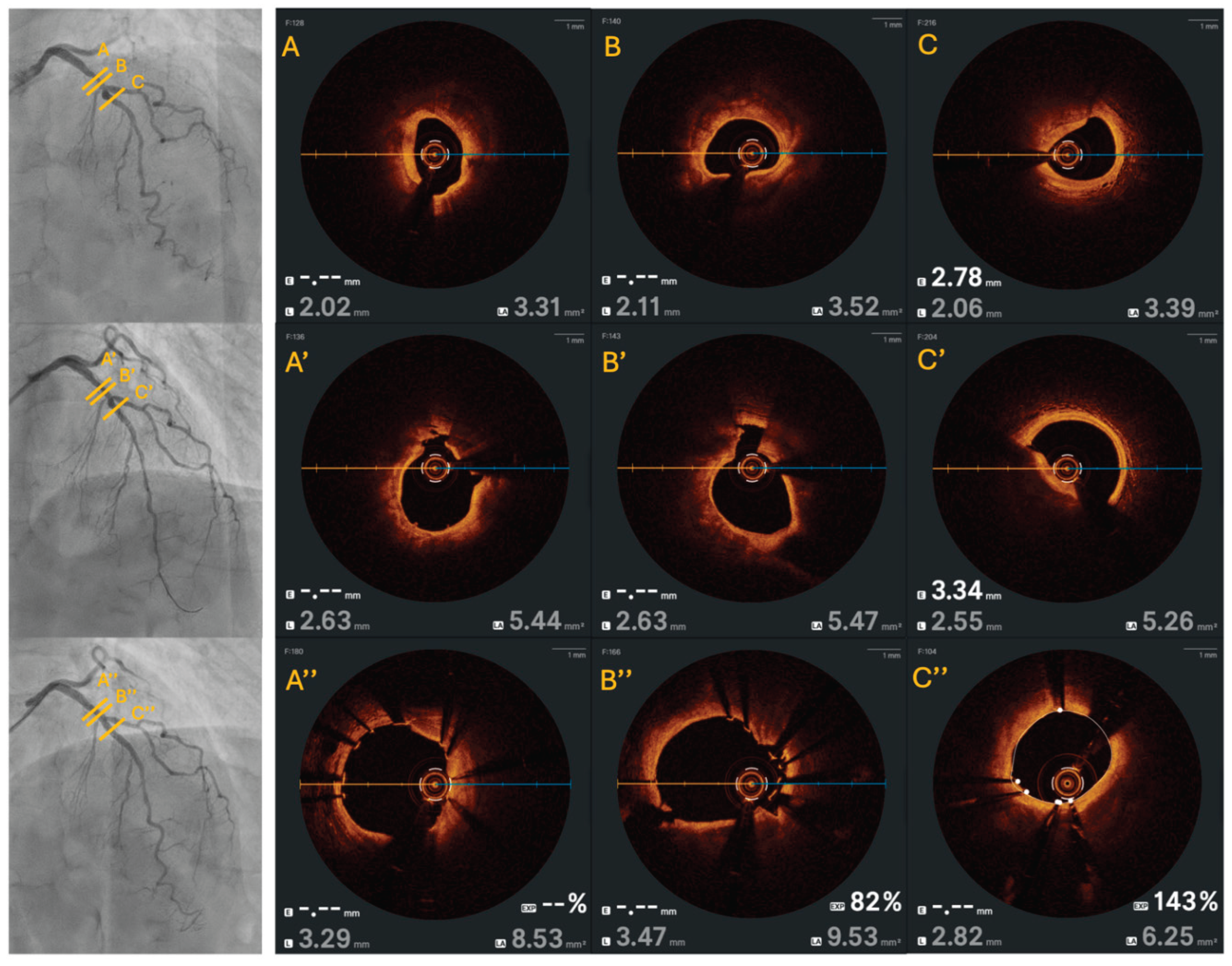
Results
| N of patients = 16 | |
| Pre-discharge ECG modifications, n (%) | 0 (0) |
| Post-procedural MI type 4a, n (%) | 0 (0) |
| In-hospital MACE, n (%) | 0 (0) |
Discussion
Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| PCI | percutaneous coronary intervention |
| OCT | optical coherence tomography |
| IQR | Interquartile range (IQR) |
| BMI | Body Mass index |
| CAD | coronary artery disease |
| MI | myocardial infarction |
| PCI | Percutaneous coronary intervention |
| CABG | coronary artery bypass grafting |
| STEMI | ST elevation myocardial infarction |
| NSTEMI | non-ST elevation myocardial infarction |
| UA | unstable angina |
| CCS | chronic coronary syndrome |
| LVEF | left ventricular ejection fraction. |
| RCA | right coronary artery disease |
| LAD | left anterior descending artery |
| ACC/AHA | American College of Cardiology/American Heart Association |
| DES | Drug eluting stent |
| atm | atmosphere |
| DCB | drug coated balloon |
| IVL | intravascular lithotripsy |
| SC | semi-compliant |
| NC | non-compliant. |
| QCA | quantitative coronary analysis |
| MLD | minimal lumen diameter |
| RVD | reference vessel diameter. |
| MLA | minimal lumen area |
| MLD | minimal lumen diameter |
| EEL | external elastic lamina |
| MSA | minimal stent area. |
| CK | creatine kinase |
| CK-MB | creatine kinase MB. |
| ECG | electrocardiogram |
| MI | myocardial infarction |
| MACE | major adverse cardiovascular events. |
References
- Allison, MA; Criqui, MH; Wright, CM. Patterns and risk factors for systemic calcified atherosclerosis. Arterioscler Thromb Vasc Biol. 2004, 24(2), 331–6. [Google Scholar] [CrossRef] [PubMed]
- Madhavan, MV; Tarigopula, M; Mintz, GS; Maehara, A; Stone, GW; Généreux, P. Coronary artery calcification: pathogenesis and prognostic implications. J Am Coll Cardiol. 2014, 63(17), 1703–14. [Google Scholar] [CrossRef]
- Généreux, P; Redfors, B; Witzenbichler, B; Arsenault, MP; Weisz, G; Stuckey, TD; et al. Two-year outcomes after percutaneous coronary intervention of calcified lesions with drug-eluting stents. Int J Cardiol. 2017, 231, 61–7. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y; Okura, H; Kume, T; Yamada, R; Kobayashi, Y; Fukuhara, K; et al. Impact of target lesion coronary calcification on stent expansion. Circ J. 2014, 78(9), 2209–14. [Google Scholar] [CrossRef] [PubMed]
- Guedeney, P; Claessen, BE; Mehran, R; Mintz, GS; Liu, M; Sorrentino, S; et al. Coronary Calcification and Long-Term Outcomes According to Drug-Eluting Stent Generation. JACC Cardiovasc Interv. 2020, 13(12), 1417–28. [Google Scholar] [CrossRef] [PubMed]
- Raggi, P; Gongora, MC; Gopal, A; Callister, TQ; Budoff, M; Shaw, LJ. Coronary artery calcium to predict all-cause mortality in elderly men and women. J Am Coll Cardiol. 2008, 52(1), 17–23. [Google Scholar] [CrossRef]
- Chen, NX; Moe, SM. Vascular calcification: pathophysiology and risk factors. Curr Hypertens Rep. 2012, 14(3), 228–37. [Google Scholar] [CrossRef]
- Généreux, P; Lee, AC; Kim, CY; Lee, M; Shlofmitz, R; Moses, JW; et al. Orbital Atherectomy for Treating De Novo Severely Calcified Coronary Narrowing (1-Year Results from the Pivotal ORBIT II Trial). Am J Cardiol. 2015, 115(12), 1685–90. [Google Scholar] [CrossRef]
- Kini, AS; Vengrenyuk, Y; Pena, J; Motoyama, S; Feig, JE; Meelu, OA; et al. Optical coherence tomography assessment of the mechanistic effects of rotational and orbital atherectomy in severely calcified coronary lesions. Catheter Cardiovasc Interv. 2015, 86(6), 1024–32. [Google Scholar] [CrossRef]
- Pinilla-Echeverri, N; Bossard, M; Hillani, A; Chavarria, JA; Cioffi, GM; Dutra, G; et al. Treatment of Calcified Lesions Using a Dedicated Super-High Pressure Balloon: Multicenter Optical Coherence Tomography Registry. Cardiovasc Revasc Med. 2023, 52, 49–58. [Google Scholar] [CrossRef]
- Cioffi, GM; Vercelli, L; Avdijaj, D; Madanchi, M; Göldi, T; Attinger-Toller, A; et al. Rotational Atherectomy Combined With Super High-Pressure Noncompliant Balloon in Severe or Recurrent In-Stent Restenosis. Journal of Interventional Cardiology 2025, 2025(1), 8869530. [Google Scholar] [CrossRef]
- Gissler, MC; Kaier, K; Rahimi, F; Bacmeister, L; Rilinger, J; Heger, LA; et al. In-hospital outcomes of intravascular lithotripsy compared to rotational atherectomy and cutting/scoring balloon angioplasty. Clin Res Cardiol 2026. [Google Scholar] [CrossRef]
- Jurado-Román, A; Gonzálvez, A; Galeote, G; Jiménez-Valero, S; Moreno, R. RotaTripsy: Combination of Rotational Atherectomy and Intravascular Lithotripsy for the Treatment of Severely Calcified Lesions. JACC Cardiovasc Interv. 2019, 12(15), e127–e9. [Google Scholar] [CrossRef]
- Brinton, TJ; Ali, ZA; Hill, JM; Meredith, IT; Maehara, A; Illindala, U; et al. Feasibility of Shockwave Coronary Intravascular Lithotripsy for the Treatment of Calcified Coronary Stenoses. Circulation 2019, 139(6), 834–6. [Google Scholar] [CrossRef]
- Kereiakes, DJ; Hill, JM; Ben-Yehuda, O; Maehara, A; Alexander, B; Stone, GW. Evaluation of safety and efficacy of coronary intravascular lithotripsy for treatment of severely calcified coronary stenoses: Design and rationale for the Disrupt CAD III trial. Am Heart J. 2020, 225, 10–8. [Google Scholar] [CrossRef]
- Salazar, CH; Gonzalo, N; Aksoy, A; Tovar Forero, MN; Nef, H; Van Mieghem, NM; et al. Feasibility, Safety, and Efficacy of Intravascular Lithotripsy in Severely Calcified Left Main Coronary Stenosis. JACC: Cardiovascular Interventions 2020, 13(14), 1727–9. [Google Scholar] [CrossRef]
- Wong, B; El-Jack, S; Newcombe, R; Glenie, T; Armstrong, G; Khan, A. Calcified Coronary Lesions Treated With Intravascular Lithotripsy: One-Year Outcomes. J Invasive Cardiol. 2020, 32(7), E200–e1. [Google Scholar] [CrossRef] [PubMed]
- Karimi Galougahi, K; Patel, S; Shlofmitz, RA; Maehara, A; Kereiakes, DJ; Hill, JM; et al. Calcific Plaque Modification by Acoustic Shock Waves: Intravascular Lithotripsy in Coronary Interventions. Circ Cardiovasc Interv. 2021, 14(1), e009354. [Google Scholar] [CrossRef] [PubMed]
- Wiens, EJ; Sklar, JC; Wei, YH; Aleem, Q; Minhas, K. Real-world outcomes in treatment of highly calcified coronary lesions with intravascular shockwave lithotripsy. Indian Heart J. 2021, 73(5), 653–5. [Google Scholar] [CrossRef]
- Ali, ZA; Kereiakes, D; Hill, J; Saito, S; Di Mario, C; Honton, B; et al. Safety and Effectiveness of Coronary Intravascular Lithotripsy for Treatment of Calcified Nodules. JACC: Cardiovascular Interventions 2023, 16(9), 1122–4. [Google Scholar] [CrossRef] [PubMed]
- Kereiakes, DJ; Hill, JM; Shlofmitz, RA; Klein, AJ; Riley, RF; Price, MJ; et al. Intravascular Lithotripsy for Treatment of Severely Calcified Coronary Arteries: 2-Year Results—Disrupt CAD III Study. JACC: Cardiovascular Interventions 2023, 16(19), 2472–4. [Google Scholar]
- Visinoni, ZM; Jurewitz, DL; Kereiakes, DJ; Shlofmitz, R; Shlofmitz, E; Ali, Z; et al. Coronary intravascular lithotripsy for severe coronary artery calcification: The Disrupt CAD I-IV trials. Cardiovascular Revascularization Medicine 2024, 65, 81–7. [Google Scholar] [CrossRef]
- Ellis, SG; Vandormael, MG; Cowley, MJ; DiSciascio, G; Deligonul, U; Topol, EJ; et al. Coronary morphologic and clinical determinants of procedural outcome with angioplasty for multivessel coronary disease. Implications for patient selection. Multivessel Angioplasty Prognosis Study Group. Circulation 1990, 82(4), 1193–202. [Google Scholar] [CrossRef]
- Moushmoush, B; Kramer, B; Hsieh, AM; Klein, LW. Does the AHA/ACC task force grading system predict outcome in multivessel coronary angioplasty? Cathet Cardiovasc Diagn 1992, 27(2), 97–105. [Google Scholar]
- Mintz, GS; Popma, JJ; Pichard, AD; Kent, KM; Satler, LF; Chuang, YC; et al. Patterns of calcification in coronary artery disease. A statistical analysis of intravascular ultrasound and coronary angiography in 1155 lesions. Circulation 1995, 91(7), 1959–65. [Google Scholar] [CrossRef]
- Fujino, A; Mintz, GS; Matsumura, M; Lee, T; Kim, SY; Hoshino, M; et al. A new optical coherence tomography-based calcium scoring system to predict stent underexpansion. EuroIntervention 2018, 13(18), e2182–e9. [Google Scholar] [CrossRef]
- Sato, T; Matsumura, M; Yamamoto, K; Sugizaki, Y; Shlofmitz, E; Moses, JW; et al. A Revised Optical Coherence Tomography–Derived Calcium Score to Predict Stent Underexpansion in Severely Calcified Lesions. JACC: Cardiovascular Interventions 2025, 18(5), 622–33. [Google Scholar] [CrossRef] [PubMed]
- Valgimigli, M; Bueno, H; Byrne, RA; Collet, JP; Costa, F; Jeppsson, A; et al. 2017 ESC focused update on dual antiplatelet therapy in coronary artery disease developed in collaboration with EACTS: The Task Force for dual antiplatelet therapy in coronary artery disease of the European Society of Cardiology (ESC) and of the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2018, 39(3), 213–60. [Google Scholar] [PubMed]
- Vrints, C; Andreotti, F; Koskinas, KC; Rossello, X; Adamo, M; Ainslie, J; et al. 2024 ESC Guidelines for the management of chronic coronary syndromes. Eur Heart J. 2024, 45(36), 3415–537. [Google Scholar]
- Prati, F; Romagnoli, E; Burzotta, F; Limbruno, U; Gatto, L; La Manna, A; et al. Clinical Impact of OCT Findings During PCI: The CLI-OPCI II Study. JACC: Cardiovascular Imaging 2015, 8(11), 1297–305. [Google Scholar] [PubMed]
- Räber, L; Mintz, GS; Koskinas, KC; Johnson, TW; Holm, NR; Onuma, Y; et al. Clinical use of intracoronary imaging. Part 1: guidance and optimization of coronary interventions. An expert consensus document of the European Association of Percutaneous Cardiovascular Interventions. EuroIntervention 2018, 14(6), 656–77. [Google Scholar] [CrossRef]
- Chambers, JW; Feldman, RL; Himmelstein, SI; Bhatheja, R; Villa, AE; Strickman, NE; et al. Pivotal Trial to Evaluate the Safety and Efficacy of the Orbital Atherectomy System in Treating De Novo, Severely Calcified Coronary Lesions (ORBIT II). JACC: Cardiovascular Interventions 2014, 7(5), 510–8. [Google Scholar] [CrossRef]
- Hill, JM; Kereiakes, DJ; Shlofmitz, RA; Klein, AJ; Riley, RF; Price, MJ; et al. Intravascular Lithotripsy for Treatment of Severely Calcified Coronary Artery Disease. J Am Coll Cardiol. 2020, 76(22), 2635–46. [Google Scholar] [CrossRef]
- Kereiakes, DJ; Di Mario, C; Riley, RF; Fajadet, J; Shlofmitz, RA; Saito, S; et al. Intravascular Lithotripsy for Treatment of Calcified Coronary Lesions: Patient-Level Pooled Analysis of the Disrupt CAD Studies. JACC: Cardiovascular Interventions 2021, 14(12), 1337–48. [Google Scholar] [PubMed]
- Kereiakes, DJ; Hill, JM; Shlofmitz, RA; Klein, AJ; Riley, RF; Price, MJ; et al. Intravascular Lithotripsy for Treatment of Severely Calcified Coronary Lesions: 1-Year Results From the Disrupt CAD III Study. Journal of the Society for Cardiovascular Angiography & Interventions 2022, 1(1), 100001. [Google Scholar] [CrossRef]
- Neleman, T; Ziedses des Plantes, AC; Daemen, J. Coronary lithotripsy – a state of the art review. Trends in Cardiovascular Medicine 2023, 33(4), 215–22. [Google Scholar] [CrossRef] [PubMed]
- Phagu, AAS; van Oort, MJH; Oliveri, F; Bingen, BO; Mincione, G; Paradies, V; et al. Comparison of Procedural and Clinical Outcomes of Angiography- Versus Imaging-Guided Percutaneous Coronary Intervention With Intravascular Lithotripsy. Catheter Cardiovasc Interv. 2025, 106(2), 869–78. [Google Scholar] [CrossRef] [PubMed]
- Saito, S; Yamazaki, S; Takahashi, A; Namiki, A; Kawasaki, T; Otsuji, S; et al. Intravascular Lithotripsy for Vessel Preparation in Severely Calcified Coronary Arteries Prior to Stent Placement - Primary Outcomes From the Japanese Disrupt CAD IV Study. Circ J. 2021, 85(6), 826–33. [Google Scholar] [CrossRef]
- Sekimoto, T; Fujiyoshi, K; Kawakami, R; Madra, A; Tanaka, T; Shin, D; et al. Comparison of Vascular Injury From Intravascular Lithotripsy, Cutting, or Ultra-High-Pressure Balloons During Coronary Calcium Modification. JACC Cardiovasc Interv. 2025, 18(17), 2093–104. [Google Scholar] [CrossRef] [PubMed]
- Emori, H; Shiono, Y; Kuriyama, N; Honda, Y; Ebihara, S; Kadooka, K; et al. Calcium Fracture After Intravascular Lithotripsy as Assessed With Optical Coherence Tomography. Circ J 2023, 87(6), 799–805. [Google Scholar] [CrossRef]
- Kawai, K; Sato, Y; Hokama, JY; Kawakami, R; Konishi, T; Ghosh, SKB; et al. Histology, OCT, and Micro-CT Evaluation of Coronary Calcification Treated With Intravascular Lithotripsy: Atherosclerotic Cadaver Study. JACC Cardiovasc Interv. 2023, 16(17), 2097–108. [Google Scholar] [CrossRef] [PubMed]
- Ali, ZA; Nef, H; Escaned, J; Werner, N; Banning, AP; Hill, JM; et al. Safety and Effectiveness of Coronary Intravascular Lithotripsy for Treatment of Severely Calcified Coronary Stenoses: The Disrupt CAD II Study. Circ Cardiovasc Interv. 2019, 12(10), e008434. [Google Scholar] [CrossRef] [PubMed]

| N of patients = 16 | |
| Age, n (±SD) | 76.4 ± 7 |
| Female, n (%) | 5 (31) |
| BMI, kg/m2 [IQR] | 28 [11.4] |
| Diabetes mellitus, n (%) | 4 (25) |
| Dyslipidemia, n (%) | 15 (94) |
| Arterial hypertension, n (%) | 14 (88) |
| Active smoking, n (%) | 4 (25) |
| Family history for CAD, n (%) | 3 (19) |
| Chronic alcohol consumption, n (%) | 2 (13) |
| Previous MI, n (%) | 2 (13) |
| Previous PCI, n (%) | 8 (50) |
| Previous CABG, n (%) | 1 (6) |
| Clinical presentation, n (%) | |
| STEMI | 1 (6) |
| NSTEMI/UA | 3 (19) |
| CCS | 12 (75%) |
| LVEF, % [IQR] | 60 [17] |
| N of patients = 16 | |
| Number of vessels treated per procedure, n [IQR] | 1 [0] |
| Vessel treated, n (%) | |
| RCA, n (%) | 10 (63) |
| LAD, n (%) | 6 (37) |
| ACC/AHA coronary lesion calcification | |
| Severe calcification, n (%) | 16 (100) |
| Type B2, n (%) | 3 (19) |
| Type C, n (%) | 13 (81) |
| DES implantation, n (%) | 14 (88) |
| Number of DES implanted, n [IQR] | 2 [2] |
| Maximum DES diameter, mm [IQR] | 3 [4] |
| DES total length, mm (±SD) | 43±27 |
| DES implantation pressure, atm (±SD) | 12±6 |
| DCB use, n (%) | 2 (13) |
| Predilation before IVL | |
| SC balloon, n (%) | 5 (31) |
| SC balloon max. diameter, mm [IQR] | 1.5 [1] |
| SC balloon max. pressure, atm [IQR] | 14 [4] |
| NC balloon, n (%) | 12 (75) |
| NC balloon max. diameter, mm [IQR] | 2.5 [0.375] |
| NC balloon max. pressure, atm [IQR] | 16 [7] |
| ShockFast™, n (%) | 16 (100) |
| ShockFast™ max. diameter, mm [IQR] | 2.5 [1] |
| ShockFast™ max. pressure, atm [IQR] | 6 [0] |
| SchockFast™ pulses, pulses [IQR] | 120 [35] |
| Postdilatation, n (%) | 16 (100) |
| N of patients = 16 | |
| Initial MLD, mm [IQR] | 1.21 [1.06] |
| Distal reference diameter (RVD), mm [IQR] | 2.5 [0.46] |
| Initial diameter stenosis, % [IQR] | 89.5 [17] |
| Lesion length, mm [IQR] | 28.9 [11.8] |
| Post-ShockFast™ MLD, mm [IQR] | 2.1 [0.64] |
| Post-ShockFast™ residual diameter stenosis, % [IQR] | 60.5 [32] |
| Final MLD, mm [IQR] | 3.1 [0.53] |
| Acute Lumen Gain, mm [IQR] | 1.86 [0.62] |
| Final diameter stenosis, % [IQR] | 11.5 [9] |
| N of patients = 8 | |
| Initial MLA, mm2 [IQR] | 3.1 [1.6] |
| Initial MLD, mm [IQR] | 1.94 [0.86] |
| Proximal reference diameter, mm [IQR] | 3.5 [0.90] |
| Proximal EEL reference diameter, mm [IQR] | 4.36 [0.41] |
| Distal reference diameter, mm [IQR] | 2.5 [0.36] |
| Distal EEL reference diameter, mm [IQR] | 3.27 [0.47] |
| Lesion length, mm [IQR] | 41.5 [28.2] |
| Calcified nodule, n (%) | 3 (38) |
| Eruptive calcified nodule, n (%) | 3 (100) |
| Calcium arc, degrees [IQR] | 274 [90] |
| Calcium thickness, mm [IQR] | 1.75 [0.29] |
| Calcium longitudinal length, mm [IQR] | 25.4 [21] |
| Revised OCT Calcium Score | |
| Score 2, n (%) | 1 (13) |
| Score 3, n (%) | 7 (87) |
| Original OCT Calcium Score | |
| Score 4, n (%) | 8 (100) |
| Presence of calcium fractures after ShockFast™, n (%) | 8 (100) |
| Calcium fractures > 2, n (%) | 5 (63) |
| Post ShockFast™ MLA, mm2 [IQR] | 4.14 [3] |
| Final MSA, mm2 [IQR] | 4.97 [3.53] |
| Acute lumen gain, mm2 [IQR] | 1 [2.85] |
| Final Stent Expansion, % [IQR] | 90 [9] |
| Malapposition, n (%) | 3 (38) |
| Major malapposition, n (%) | 0 (0) |
| Medial edge dissection, n (%) | 0 (0) |
| N of patients = 16 | |
| Initial Troponin, ng/L [IQR] | 19 [15] |
| Initial CK, U/L [IQR] | 76 [43] |
| Initial CK-MB, U/L [IQR] | 16 [6] |
| Pre-discharge Troponin, ng/L [IQR] | 35 [201] |
| Pre-discharge CK, U/L [IQR] | 97.5 [41] |
| Pre-discharge CK-MB, U/L [IQR] | 15 [9] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




