Submitted:
28 February 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
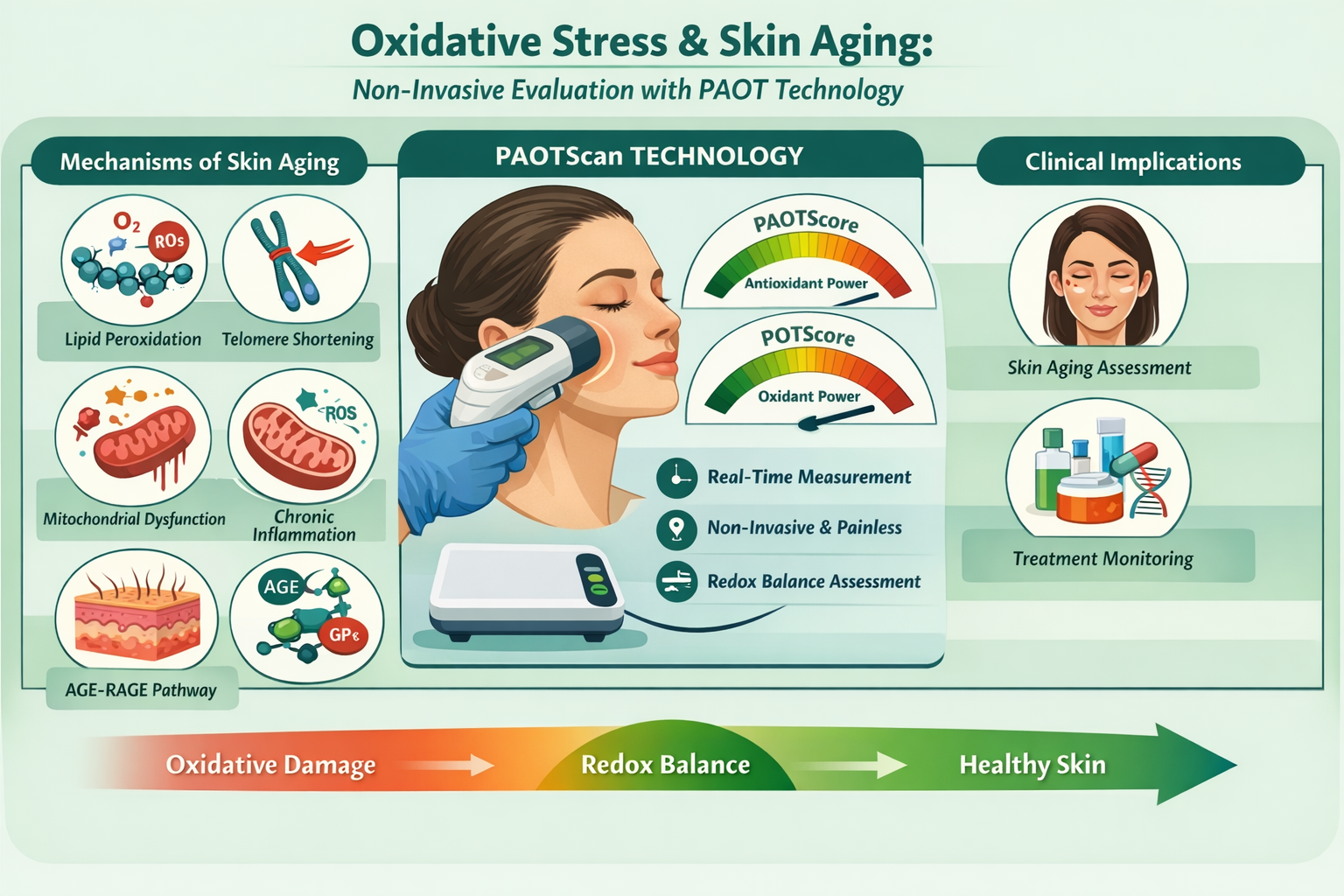
2. Biological Aging and Oxidative Stress
Radical Theory of Aging
- Cellular alterations linked to aging
- Damage to DNA and telomere function
- Lipid peroxidation
- The AGE-RAGE axis and aging
- Mitochondrial dysfunction and aging
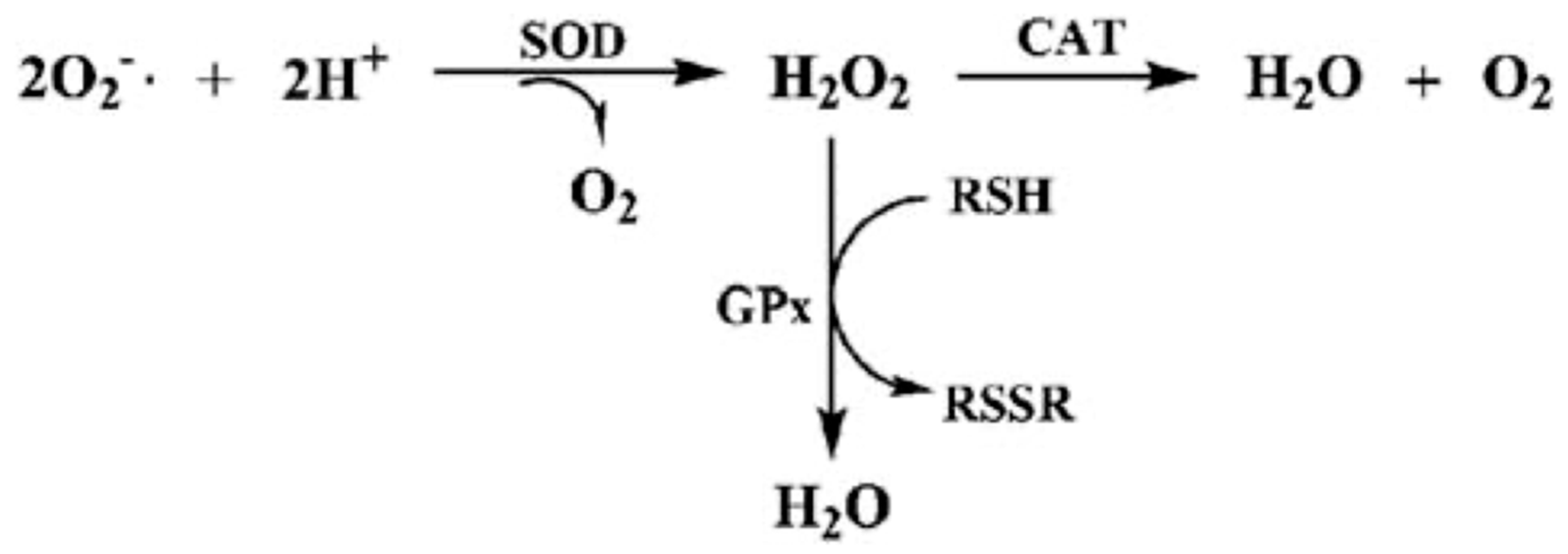
- Enzyme deregulation
- Oxidative stress promotes inflammation
- Dysbiosis and inflammation favor ROS generation
| Biomarkers | Impact | Tendance |
| 8-oxodG | DNA damages | Increase |
| MDA (lipid peroxidation) | Oxidative stress markers | Increase |
| Telomere length | Cellular senescence indicator | Decrease |
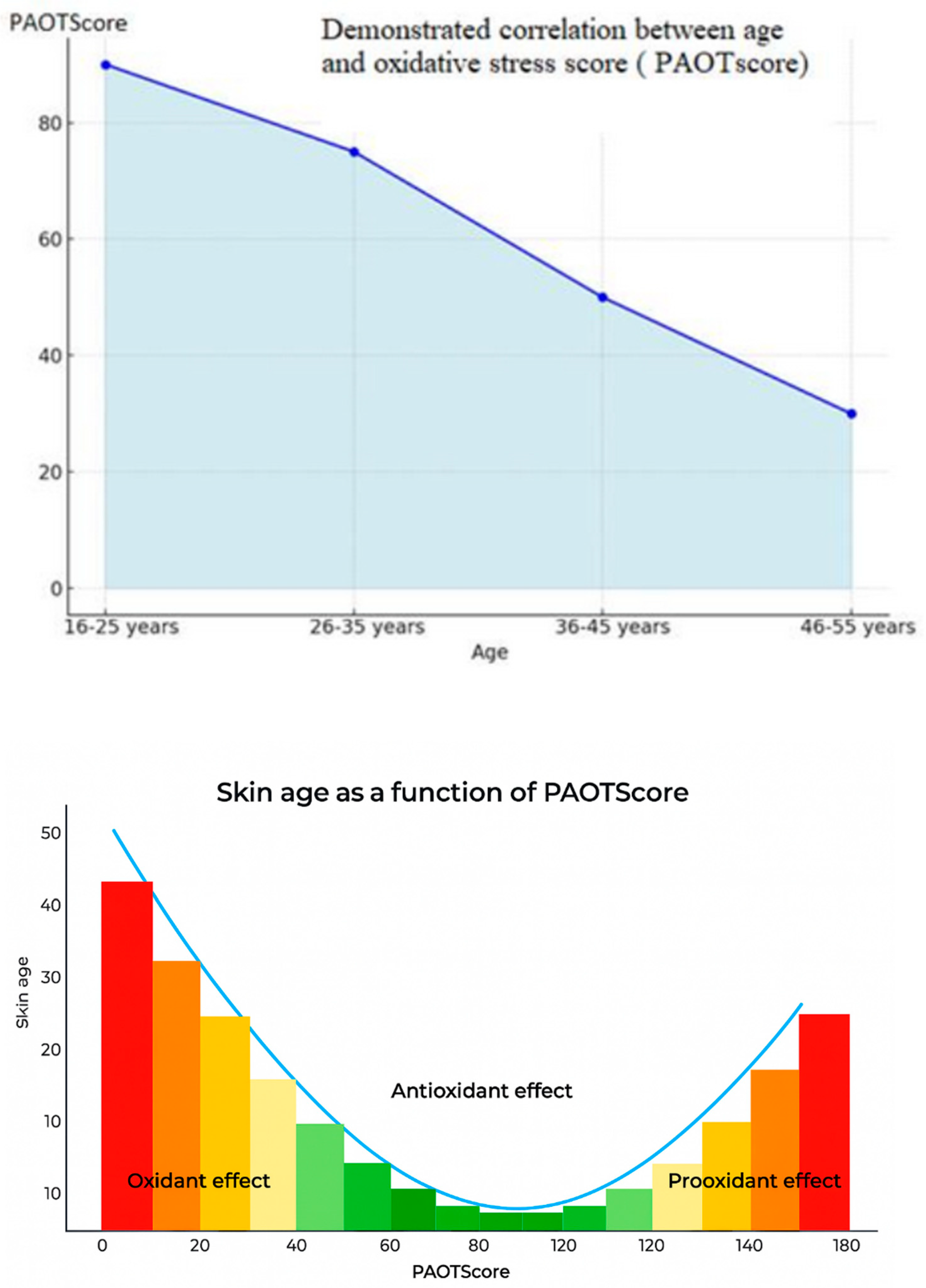
| PAOTScore | Total Antioxidant Power | Decrease |
| SOD | Antioxidant enzyme, ROS detoxification | Decrease |
| Catalase | Conversion of H2O2 into H2O and O2 | Decrease |
| GPx | Peroxide reduction, cell protection | Decrease |
| Pro-inflammatory cytokines (IL6, TNFa, etc.) | Pro-inflammatory cytokines | Increase |
| Bacteria of the intestinal microbiota | Modulation of inflammation and oxidative stress | Dysbiosis |
- Pro-oxidant effects of vitamins.
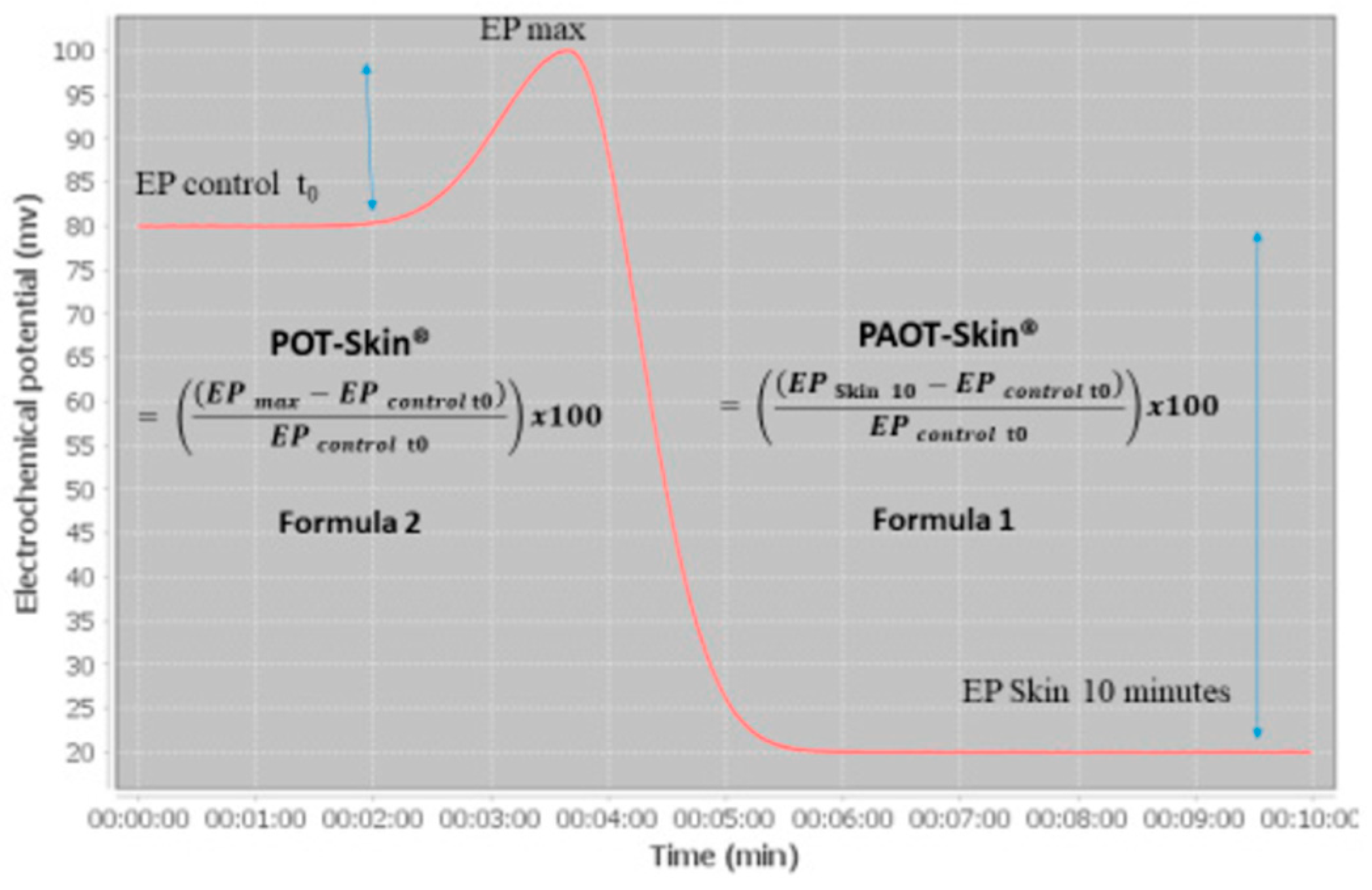
- PAOTScan to measure oxidative stress
- The PAOTscore
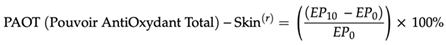
- The POTscore
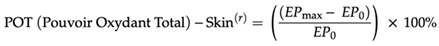
- Markers observed by PAOTScan
| Parameter | Human Reference interval |
| Antioxydants | |
| vitamin C (µg/mL) | 6.0 - 15 |
| vitamin E as a-tocopherol (µg/mL) | 8.6 - 19.2 |
| γ-tocopherol (µg/mL) | 0.39 - 2.42 |
| β-carotene (µg/mL) | 0.06 - 0.68 |
| thiol proteins (µM) | 314 - 516 |
| reduced glutathione (µM) | 717 - 1110 |
| oxidized glutathione (µM) | 0.96 - 10 |
| glutathione peroxidase (UI/g Hb) | 20 - 56 |
| Uric acid | >70 |
| Trace elements | |
| copper (mg/mL) | 0.70 - 1.1 |
| zinc (mg/mL) | 0.70 - 1.20 |
| selenium (µg/mL) | 73 - 110 |
| Biomarkers of lipid peroxidation | |
| lipid peroxides (µM) | 0 - 432 |
| oxidized LDL (ng/mL) | 28 - 70 |
| antibobies against oxidized LDL | 200 - 600 |
| Sources of ROS production | |
| copper/zinc ratio | 1 - 1.17 |
| myeloperoxidase | 27 - 72 |
| PAOT | |
| plasma | 1.42 - 36.78 |
| skin | 7.86 - 62.91 |
| saliva | 1.52 - 14.14 |
| urine | 42.85 - 104.63 |
| creatinine standardized urine | 6.23 - 121.96 |
- The aging speed

- -
- VitCmeasured, VitEmeasured and GSHmeasured represent the measured concentrations of vitamins C, E and glutathione in the skin.
- -
- VitCopt, VitEopt and GSHopt correspond to the optimal levels expected for these molecules.
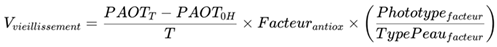
- -
- PAOTT and PAOT0H represent the values of total antioxidant power measured respectively after a certain time T and at the initial time.
- -
- T is the duration of the measurement.
- -
- Antiox factor is the previously defined coefficient, adjusting for the impact of vitamins and glutathione on antioxidant protection.
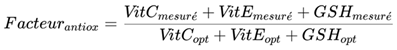
- -
- -Phototypefacteur / TypePeaufacteur introduces a correction taking into account the subject's skin type and phototype, in order to refine the assessment of skin aging (Krtmann et al., 2017).
- -
- Accuracy of skin ageing assessment
- Skin parameters influencing results
- Phototype
- Elasticity
- Texture
- Comparison of PAOTScan with other current techniques
| Method | Principle | Advantages | disadvantages |
| Raman Spectroscopy | Detection of oxidative biomarkers | Non-invasive | Limited sensitivity (Ruffien-Ciszak et al., 2008) |
| Biochemical assay | MDA and 8-oxodG assay | High precision | Requires sampling (Ayala et al., 2014) |
| PAOTScan | Real-time electrochemical analysis | Fast and non-invasive | No Specific species |
- Antioxidant protection strategies
| Strategy | Strategy | Efficiency |
|---|---|---|
| Vitamin C | Free radical neutralization, collagen stimulation | High |
| Vitamin C | Protection of lipid membranes | High |
| Glutathione | Regulation of intracellular oxidative stress | High |
| Polyphenols (resveratrol) | Activation of the Nrf2 pathway | Medium to High |
| Sun protection (SPF) | UV blocking | Essential |
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Chaudhary, P.; Janmeda, P.; Docea, A.O.; Yeskaliyeva, B.; Abdull Razis, A.F.; Modu, B.; et al. Oxidative stress, free radicals and antioxidants: potential crosstalk in the pathophysiology of human diseases. Front Chem. 2023, 11, 1158198. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.P. Oxidative Stress in Health and Disease. Biomedicines 2023, 11, 2925. [Google Scholar] [CrossRef]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; et al. Oxidative stress, aging, and diseases. Clin Interv Aging 2018, 13, 757-72. [Google Scholar] [CrossRef]
- Pole, A.; Dimri, M.; P Dimri G; Department of Biochemistry and Molecular Medicine; School of Medicine and Health Sciences; The George Washington University; Washington DC; USA. Oxidative stress, cellular senescence and ageing. AIMS Mol Sci. 2016, 3, 300-24. [Google Scholar] [CrossRef]
- Tan, B.L.; Norhaizan, M.E.; Liew, W.P.P.; Sulaiman Rahman, H. Antioxidant and Oxidative Stress: A Mutual Interplay in Age-Related Diseases. Front Pharmacol. 2018, 9, 1162. [Google Scholar] [CrossRef]
- Joël, P.; Mouna-Messaouda, K.; Jean-Paul, C.B.; Jean-Olivier, D.; Smail, M. Electrochemical Methodology for Evaluating Skin Oxidative Stress Status (SOSS). Diseases 2019, 7, 40. [Google Scholar] [CrossRef]
- Denham, H. Free-Radical Theory of Aging 1994, 717, 1-15.
- Ahmed, W.; Lingner, J. Impact of oxidative stress on telomere biology. Differentiation 2018, 99, 21-7. [Google Scholar] [CrossRef]
- Cooke, M.S.; Evans, M.D.; Dizdaroglu, M.; Lunec, J. Oxidative DNA damage: mechanisms, mutation, and disease. FASEB J. 2003, 17, 1195-214. [Google Scholar] [CrossRef]
- Lasch, J.; Schonfelder, U.; Walke, M.; Zellmer, S.; Beckert, D. Oxidative damage of human skin lipids Dependence of lipid peroxidation on sterol concentration.
- Villalón-García, I.; Álvarez-Córdoba, M.; Povea-Cabello, S.; Talaverón-Rey, M.; Villanueva-Paz, M.; Luzón-Hidalgo, R.; et al. Vitamin E prevents lipid peroxidation and iron accumulation in PLA2G6-Associated Neurodegeneration. Neurobiol Dis. 2022, 165, 105649. [Google Scholar] [CrossRef]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal. Oxid Med Cell Longev. 2014, 2014, 1-31. [Google Scholar] [CrossRef]
- Moldogazieva, N.T.; Zavadskiy, S.P.; Astakhov, D.V.; Terentiev, A.A. Lipid peroxidation: Reactive carbonyl species, protein/DNA adducts, and signaling switches in oxidative stress and cancer. Biochem Biophys Res Commun. 2023, 687, 149167. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.Q.; Zhang, D.D.; Wang, Y.N.; Tan, Y.Q.; Yu, X.Y.; Zhao, Y.Y. AGE/RAGE in diabetic kidney disease and ageing kidney. Free Radic Biol Med. 2021, 171, 260-71. [Google Scholar] [CrossRef] [PubMed]
- Senatus, L.M.; Schmidt, A.M. The AGE-RAGE Axis: Implications for Age-Associated Arterial Diseases. Front Genet. 2017, 8, 187. [Google Scholar] [CrossRef] [PubMed]
- Semba, R.D.; Nicklett, E.J.; Ferrucci, L. Does Accumulation of Advanced Glycation End Products Contribute to the Aging Phenotype? J Gerontol A Biol Sci Med Sci. 2010, 65A, 963-75. [Google Scholar] [CrossRef]
- Vitorakis, N.; Piperi, C. Pivotal role of AGE-RAGE axis in brain aging with current interventions. Ageing Res Rev. 2024, 100, 102429. [Google Scholar] [CrossRef]
- Kung, H.C.; Lin, K.J.; Kung, C.T.; Lin, T.K. Oxidative Stress, Mitochondrial Dysfunction, and Neuroprotection of Polyphenols with Respect to Resveratrol in Parkinson’s Disease. [CrossRef]
- Kudryavtseva, A.V.; Krasnov, G.S.; Dmitriev, A.A.; Alekseev, B.Y.; Kardymon, O.L.; Sadritdinova, A.F.; et al. Mitochondrial dysfunction and oxidative stress in aging and cancer. Oncotarget 2016, 7, 44879-905. [Google Scholar] [CrossRef]
- Peoples, J.N.; Saraf, A.; Ghazal, N.; Pham, T.T.; Kwong, J.Q. Mitochondrial dysfunction and oxidative stress in heart disease. Exp Mol Med. 2019, 51, 1-13. [Google Scholar] [CrossRef]
- Hayakawa, M.; Hattori, K.; Sugiyama, S.; Ozawa, T. Age-associated oxygen damage and mutations in mitochondrial DNA in human hearts. Biochem Biophys Res Commun. 1992, 189, 979-85. [Google Scholar] [CrossRef]
- Bafana, A.; Dutt, S.; Kumar, A.; Kumar, S.; Ahuja, P.S. The basic and applied aspects of superoxide dismutase. J Mol Catal B Enzym. 2011, 68, 129-38. [Google Scholar] [CrossRef]
- Doran, M.L.; Knee, J.M.; Wang, N.; Rzezniczak, T.Z.; Parkes, T.L.; Li, L.; et al. Metabolomic analysis of oxidative stress: Superoxide dismutase mutation and paraquat induced stress in Drosophila melanogaster. Free Radic Biol Med. 2017, 113, 323-34. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Yin, Y.; Zhu, J.; Huang, X.; Luo, Q.; Xu, J.; et al. A modulatory bifunctional artificial enzyme with both SOD and GPx activities based on a smart star-shaped pseudo-block copolymer. Soft Matter 2010, 6, 5342. [Google Scholar] [CrossRef]
- Buonocore, G.; Perrone, S.; Tataranno, M.L. Oxygen toxicity: chemistry and biology of reactive oxygen species. Semin Fetal Neonatal Med. 2010, 15, 186-90. [Google Scholar] [CrossRef]
- Bernard, K.E.; Parkes, T.L.; Merritt, T.J.S. A Model of Oxidative Stress Management: Moderation of Carbohydrate Metabolizing Enzymes in SOD1-Null Drosophila melanogaster. In PLoS ONE; Oliveira, PL, Ed.; 2011; Volume 6. [Google Scholar]
- Alfadda, A.A.; Sallam, R.M. Reactive Oxygen Species in Health and Disease. J Biomed Biotechnol. 2012, 2012, 1-14. [Google Scholar] [CrossRef]
- Terlecky, S.R.; Koepke, J.I.; Walton, P.A. Peroxisomes and aging. Biochim Biophys Acta BBA - Mol Cell Res. 2006, 1763, 1749-54. [Google Scholar] [CrossRef]
- Schrader, M.; Fahimi, H.D. Peroxisomes and oxidative stress. Biochim Biophys Acta BBA - Mol Cell Res. 2006, 1763, 1755-66. [Google Scholar] [CrossRef] [PubMed]
- Cui, Z.; He, F.; Li, X.; Li, Y.; Huo, C.; Wang, H.; et al. Response pathways of superoxide dismutase and catalase under the regulation of triclocarban-triggered oxidative stress in Eisenia foetida: Comprehensive mechanism analysis based on cytotoxicity and binding model. Sci Total Environ. 2023, 854, 158821. [Google Scholar] [CrossRef]
- Piao, X.; Liu, Z.; Li, Y.; Yao, D.; Sun, L.; Wang, B.; et al. Investigation of the effect for bisphenol A on oxidative stress in human hepatocytes and its interaction with catalase. Spectrochim Acta A Mol Biomol Spectrosc. 2019, 221, 117149. [Google Scholar] [CrossRef] [PubMed]
- Koepke, J.I.; Nakrieko, K.; Wood, C.S.; Boucher, K.K.; Terlecky, L.J.; Walton, P.A.; et al. Restoration of Peroxisomal Catalase Import in a Model of Human Cellular Aging. Traffic 2007, 8, 1590-600. [Google Scholar] [CrossRef] [PubMed]
- Selvaratnam, J.; Robaire, B. Overexpression of catalase in mice reduces age-related oxidative stress and maintains sperm production. Exp Gerontol. 2016, 84, 12-20. [Google Scholar] [CrossRef] [PubMed]
- Chu, F.F.; Esworthy, R.S.; Chu, P.G.; Longmate, J.A.; Huycke, M.M.; Wilczynski, S.; et al. Bacteria-Induced Intestinal Cancer in Mice with Disrupted Gpx1 and Gpx2 Genes. Cancer Res. 2004, 64, 962-8. [Google Scholar] [CrossRef]
- Flohé, L.; Toppo, S.; Orian, L. The glutathione peroxidase family: Discoveries and mechanism. Free Radic Biol Med. 2022, 187, 113-22. [Google Scholar] [CrossRef] [PubMed]
- Herbette, S.; Labrouhe, D.T.D.; Drevet, J.R.; Roeckel-Drevet, P. Transgenic tomatoes showing higher glutathione peroxydase antioxidant activity are more resistant to an abiotic stress but more susceptible to biotic stresses. Plant Sci. 2011, 180, 548-53. [Google Scholar] [CrossRef]
- Pei, J.; Pan, X.; Wei, G.; Hua, Y. Research progress of glutathione peroxidase family (GPX) in redoxidation. Front Pharmacol. 2023, 14, 1147414. [Google Scholar] [CrossRef]
- Bersuker, K.; Hendricks, J.M.; Li, Z.; Magtanong, L.; Ford, B.; Tang, P.H.; et al. The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis. Nature 2019, 575, 688-92. [Google Scholar] [CrossRef]
- Kipp, A.; Banning, A.; Brigelius-Flohé, R. Activation of the glutathione peroxidase 2 (GPx2) promoter by β-catenin. bchm 2007, 388, 1027-33. [Google Scholar] [CrossRef]
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Reactive Oxygen Species in Inflammation and Tissue Injury. Antioxid Redox Signal. 2014, 20, 1126-67. [Google Scholar] [CrossRef]
- Liu, K.L.; Sun, T.Z.; Yang, Y.; Gao, Q.X.; Tu, L.M.; Yu, J.Y.; et al. Blockade of PVN neuromedin B receptor alleviates inflammation via the RAS/ROS/NF-κB pathway in spontaneously hypertensive rats. Brain Res Bull. 2025, 220, 111180. [Google Scholar] [CrossRef]
- Chen, A.; Huang, H.; Fang, S.; Hang, Q. ROS: A “booster” for chronic inflammation and tumor metastasis. Biochim Biophys Acta BBA - Rev Cancer 2024, 1879, 189175. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhang, Y.; Jing, L.; Zhao, H. Microplastics induced inflammation in the spleen of developmental Japanese quail (Coturnix japonica) via ROS-mediated p38 MAPK and TNF signaling pathway activation1. Environ Pollut. 2024, 341, 122891. [Google Scholar] [CrossRef]
- Fortes, G.B.; Alves, L.S.; De Oliveira, R.; Dutra, F.F.; Rodrigues, D.; Fernandez, P.L.; et al. Heme induces programmed necrosis on macrophages through autocrine TNF and ROS production. Blood 2012, 119, 2368-75. [Google Scholar] [CrossRef]
- Bang, E.; Kim, D.H.; Chung, H.Y. Protease-activated receptor 2 induces ROS-mediated inflammation through Akt-mediated NF-κB and FoxO6 modulation during skin photoaging. Redox Biol. 2021, 44, 102022. [Google Scholar] [CrossRef]
- Fransen, F.; Van Beek, A.A.; Borghuis, T.; Aidy, S.E.; Hugenholtz, F.; Van Der Gaast – De Jongh, C.; et al. Aged Gut Microbiota Contributes to Systemical Inflammaging after Transfer to Germ-Free Mice. Front Immunol. 2017, 8, 1385. [Google Scholar] [CrossRef]
- Thevaranjan, N.; Puchta, A.; Schulz, C.; Naidoo, A.; Szamosi, J.C.; Verschoor, C.P.; et al. Age-Associated Microbial Dysbiosis Promotes Intestinal Permeability, Systemic Inflammation, and Macrophage Dysfunction. Cell Host Microbe 2017, 21, 455–466.e4. [Google Scholar] [CrossRef] [PubMed]
- Millman, J.F.; Kondrashina, A.; Walsh, C.; Busca, K.; Karawugodage, A.; Park, J.; et al. Biotics as novel therapeutics in targeting signs of skin ageing via the gut-skin axis. Ageing Res Rev. 2024, 102, 102518. [Google Scholar] [CrossRef]
- Gao, T.; Li, Y.; Wang, X.; Tao, R.; Ren, F. Bifidobacterium longum 68S mediated gut-skin axis homeostasis improved skin barrier damage in aging mice. Phytomedicine 2023, 120, 155051. [Google Scholar] [CrossRef]
- Krutmann, J.; Bouloc, A.; Sore, G.; Bernard, B.A.; Passeron, T. The skin aging exposome. J Dermatol Sci. 2017, 85, 152-61. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.;; Chen, Z.M.; Fan, X.Y.; Jin, Y.L.; Li, X.; Wu, S.R.; et al. Gut–Brain–Skin Axis in Psoriasis: A Review. Dermatol Ther. 2021, 11, 25-38. [Google Scholar] [CrossRef] [PubMed]
- Hodnick, W.F.; Milosavljevi, E.B. ELECTROCHEMISTRY OF FLAVONOIDS. [CrossRef] [PubMed]
- Carr, A.; Frei, B. Does vitamin C act as a pro-oxidant under physiological conditions? FASEB J 1999, 13, 1007-24. [Google Scholar] [CrossRef]
- Prasad, A.; Rathi, D.; Sedlářová, M.; Manoharan, R.R.; Průdková, E.; Pospíšil, P. Differential effects of ascorbic acid on monocytic cell morphology and protein modification: Shifting from pro-oxidative to antioxidant properties. Biochem Biophys Rep. 2024, 37, 101622. [Google Scholar] [CrossRef] [PubMed]
- León-González, A.J.; Auger, C.; Schini-Kerth, V.B. Pro-oxidant activity of polyphenols and its implication on cancer chemoprevention and chemotherapy. Biochem Pharmacol. 2015, 98, 371-80. [Google Scholar] [CrossRef]
- Tedesco, I.; Spagnuolo, C.; Russo, G.L.; Russo, M.; Cervellera, C.; Moccia, S. The Pro-Oxidant Activity of Red Wine Polyphenols Induces an Adaptive Antioxidant Response in Human Erythrocytes. Antioxidants 2021, 10, 800. [Google Scholar] [CrossRef]
- DePhillipo, N.N.; Aman, Z.S.; Kennedy, M.I.; Begley, J.P.; Moatshe, G.; LaPrade, R.F. Efficacy of Vitamin C Supplementation on Collagen Synthesis and Oxidative Stress After Musculoskeletal Injuries: A Systematic Review. Orthop J Sports Med. 2018, 6, 2325967118804544. [Google Scholar] [CrossRef]
- Darr, D.; Combs, S.; Dunston, S.; Manning, T.; Pinnell, S. Topical vitamin C protects porcine skin from ultraviolet radiation-induced damage. Br J Dermatol 1992, 127, 247-53. [Google Scholar] [CrossRef]
- Phillips, C.L.; Combs, S.B.; Pinnell, S.R. Effects of Ascorbic Acid on Proliferation and Collagen Synthesis in Relation to the Donor Age of Human Dermal Fibroblasts. J Invest Dermatol 1994, 103, 228-32. [Google Scholar] [CrossRef]
- Dulinska-Molak, I.; Pasikowska-Piwko, M.; Dębowska, R.; Święszkowski, W.; Rogiewicz, K.; Eris, I. Determining the effectiveness of vitamin C in skin care by atomic force microscope. Microsc Res Tech. 2019, 82, 1430-7. [Google Scholar] [CrossRef]
- Boo, Y.C. Ascorbic Acid (Vitamin C) as a Cosmeceutical to Increase Dermal Collagen for Skin Antiaging Purposes: Emerging Combination Therapies. Antioxidants 2022, 11, 1663. [Google Scholar] [CrossRef] [PubMed]
- Pullar, J.; Carr, A.; Vissers, M. The Roles of Vitamin C in Skin Health. Nutrients 2017, 9, 866. [Google Scholar] [CrossRef] [PubMed]
- Rychter, A.M.; Hryhorowicz, S.; Słomski, R.; Dobrowolska, A.; Krela-Kaźmierczak, I. Antioxidant effects of vitamin E and risk of cardiovascular disease in women with obesity – A narrative review. Clin Nutr. 2022, 41, 1557-65. [Google Scholar] [CrossRef]
- Higgins, M.; Izadi, A.; Kaviani, M. Antioxidants and Exercise Performance: With a Focus on Vitamin E and C Supplementation. Int J Environ Res Public Health 2020, 17, 8452. [Google Scholar] [CrossRef]
- Harris, I.S.; Treloar, A.E.; Inoue, S.; Sasaki, M.; Gorrini, C.; Lee, K.C.; et al. Glutathione and Thioredoxin Antioxidant Pathways Synergize to Drive Cancer Initiation and Progression. Cancer Cell. 2015, 27, 211-22. [Google Scholar] [CrossRef]
- Feng, Z.; Qin, Y.; Huo, F.; Jian, Z.; Li, X.; Geng, J.; et al. NMN recruits GSH to enhance GPX4-mediated ferroptosis defense in UV irradiation induced skin injury. Biochim Biophys Acta BBA - Mol Basis Dis. 2022, 1868, 166287. [Google Scholar] [CrossRef]
- Soeur, J.; Eilstein, J.; Léreaux, G.; Jones, C.; Marrot, L. Skin resistance to oxidative stress induced by resveratrol: From Nrf2 activation to GSH biosynthesis. Free Radic Biol Med. 2015, 78, 213-23. [Google Scholar] [CrossRef]
- Gunathilake, R.; Schurer, N.Y.; Shoo, B.A.; Celli, A.; Hachem, J.P.; Crumrine, D.; et al. pH-Regulated Mechanisms Account for Pigment-Type Differences in Epidermal Barrier Function. J Invest Dermatol 2009, 129, 1719-29. [Google Scholar] [CrossRef]
- Chikakane, K.; Takahashi, H. Measurement of skin pH and its significance in cutaneous diseases. Clin Dermatol 1995, 13, 299-306. [Google Scholar] [CrossRef]
- Ruffien-Ciszak, A.; Baur, J.; Gros, P.; Questel, E.; Comtat, M. Electrochemical microsensors for cutaneous surface analysis: Application to the determination of pH and the antioxidant properties of stratum corneum. IRBM 2008, 29(2-3), 162-70. [Google Scholar] [CrossRef]
- Agache, P. Physiologie de la peau et explorations fonctionnelles cutanées. 1ère; Tec & Doc Lavoisier, 2000. [Google Scholar]
- Proksch, E.; Brandner, J.M.; Jensen, J. The skin: an indispensable barrier. Exp Dermatol 2008, 17, 1063-72. [Google Scholar] [CrossRef] [PubMed]
- Boncheva, M.; De Sterke, J.; Caspers, P.J.; Puppels, G.J. Depth profiling of Stratum corneum hydration in vivo : a comparison between conductance and confocal Raman spectroscopic measurements. Exp Dermatol 2009, 18, 870-6. [Google Scholar] [CrossRef]
- Chicharro-Luna, E.; Zúnica-García, S.; Martinez-Algarra, C.; Gracia-Sánchez, A. Age-related variations in stratum corneum hydration in the foot. Maturitas 2024, 189, 108104. [Google Scholar] [CrossRef] [PubMed]
- Zouboulis, C.; Makrantonaki, E. Hormonal therapy of intrinsic aging. Rejuvenation Research 2012, 15, 302-12. [Google Scholar] [CrossRef] [PubMed]
- Hussen, N.H.A.; Abdulla, S.K.; Ali, N.M.; Ahmed, V.A.; Hasan, A.H.; Qadir, E.E. Role of antioxidants in skin aging and the molecular mechanism of ROS: A comprehensive review. Asp Mol Med. 2025, 5, 100063. [Google Scholar] [CrossRef]
- Schagen, S.K.; Zampeli, V.A.; Makrantonaki, E.; Zouboulis, C.C. Discovering the link between nutrition and skin aging. Dermatoendocrinol 2012, 4, 298-307. [Google Scholar] [CrossRef]
- Vierkötter, A.; Krutmann, J. Environmental influences on skin aging and ethnic-specific manifestations. Dermatoendocrinol 2012, 4, 227-31. [Google Scholar] [CrossRef]
- Solano, F. Photoprotection and Skin Pigmentation: Melanin-Related Molecules and Some Other New Agents Obtained from Natural Sources. Molecules 2020, 25, 1537. [Google Scholar] [CrossRef]
- Takeuchi, S.; Zhang, W.; Wakamatsu, K.; Ito, S.; Hearing, V.J.; Kraemer, K.H.; et al. Melanin acts as a potent UVB photosensitizer to cause an atypical mode of cell death in murine skin. Proc Natl Acad Sci. 2004, 101, 15076-81. [Google Scholar] [CrossRef]
- Tanaka, H.; Yamashita, Y.; Umezawa, K.; Hirobe, T.; Ito, S.; Wakamatsu, K. The Pro-Oxidant Activity of Pheomelanin is Significantly Enhanced by UVA Irradiation: Benzothiazole Moieties Are More Reactive than Benzothiazine Moieties. Int J Mol Sci. 2018, 19, 2889. [Google Scholar] [CrossRef]
- Pu, S.Y.; Huang, Y.L.; Pu, C.M.; Kang, Y.N.; Hoang, K.D.; Chen, K.H.; et al. Effects of Oral Collagen for Skin Anti-Aging: A Systematic Review and Meta-Analysis. Nutrients 2023, 15, 2080. [Google Scholar] [CrossRef]
- Nomoto, T.; Iizaka, S. Effect of an Oral Nutrition Supplement Containing Collagen Peptides on Stratum Corneum Hydration and Skin Elasticity in Hospitalized Older Adults: A Multicenter Open-label Randomized Controlled Study. Adv Skin Wound Care 2020, 33, 186-91. [Google Scholar] [CrossRef] [PubMed]
- Addor, F.A.S. Antioxidants in dermatology. An Bras Dermatol 2017, 92, 356-62. [Google Scholar] [CrossRef] [PubMed]
- Berneburg, M.; Plettenberg, H.; Krutmann, J. Photoaging of human skin. Photodermatol Photoimmunol Photomed. 2000, 16, 239-44. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




