Submitted:
27 February 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. LSTV Classification
2.2. Jenkins Classification of LSTV
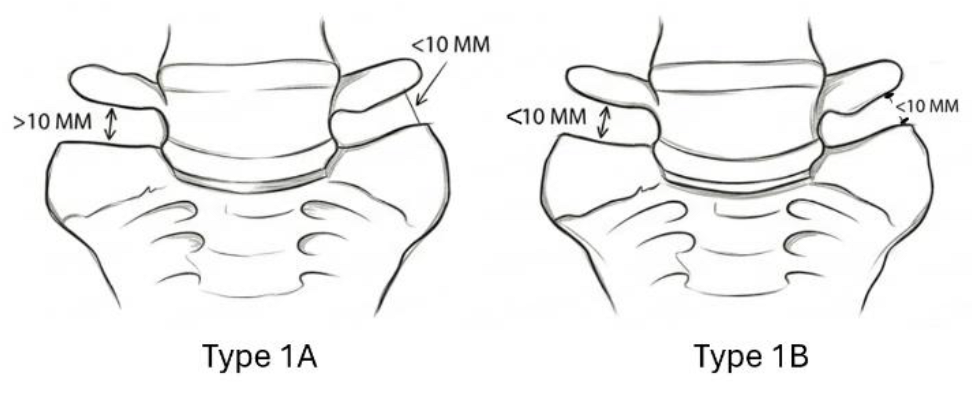
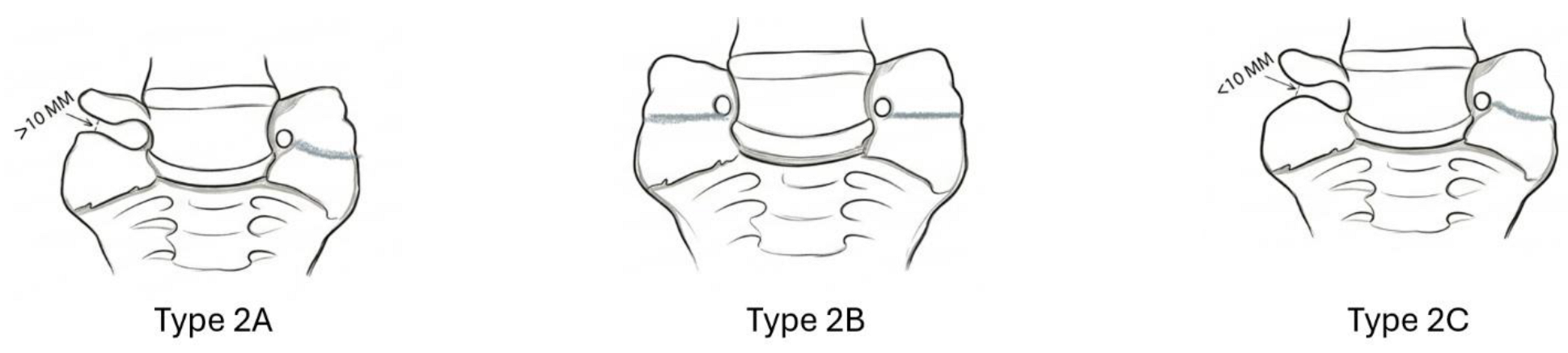
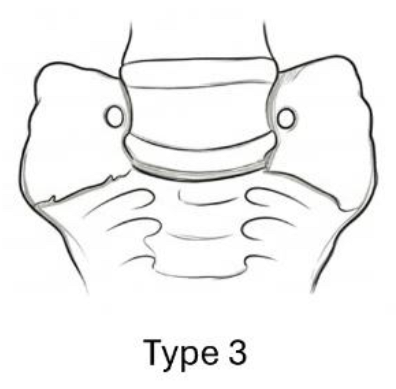
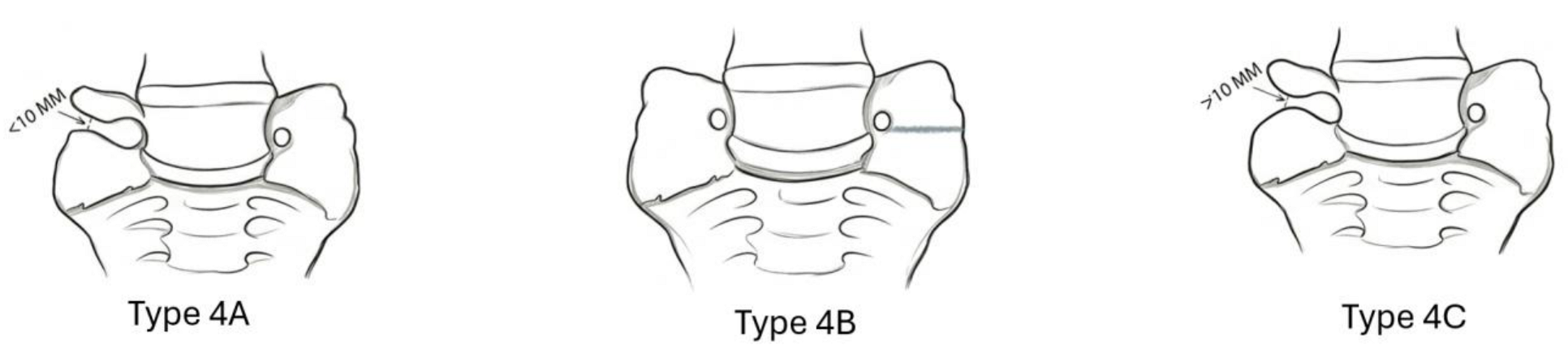
3. Symptoms Observed in Patients with Bertolotti’s Syndrome
4. Biomechanical Changes Due to Bertolotti’s Syndrome
4.1. Direct Effect of LSTV
- Arthritis of the pseudo-articulation – In LSTV types where the transverse process forms a “false joint” with the sacrum (i.e., type II & type IV), the bone-on-bone contact leads to mechanical grinding, inflammation, and the formation of osteophytes. This can lead to direct localized, aching pain (Poe, 2013).
- Radiculopathy – Radiculopathy (i.e., pain radiating down the leg) can occur through several mechanisms. This can be caused by direct compression of the exiting L5 nerve root due to the enlarged transverse process. Alternatively, the local inflammation caused by osteophyte formation due to pseudo-articulation can lead to irritation of the exiting L5 nerve root. Alternatively, the hypermobility at the L4-L5 level due to LSTV can cause disc herniation at that level (Jancuska et al., 2015).
4.2. Secondary Effects of LSTV
- Myofascial pain – The body’s attempt to stabilize the anomalous anatomy can cause increased strain on the musculature around the LSTV. This can lead to hypertonicity in the Quadratus Lumborum (QL) and the Iliopsoas that can cause formation of chronic strain and trigger points in these muscles (Jain et al., 2013).
- Contralateral facet joint stress – The asymmetrical motion caused by unilateral LSTV can place excessive load on the facet joint on the opposite side of the pseudo-articulation. This can lead to facet joint pain and development of arthritis (Jancuska et al., 2015).
- Adjacent segment disease – Compensatory hypermobility of the segment above the LSTV can lead to acceleration of degeneration of the disc. This, in turn, leads to higher incidence of disc herniation, annular tears (i.e., tear of the outer wall of the disc), and spinal stenosis compared to individuals with normal anatomy (Crane et al., 2021).
5. Genetic Basis for Bertolotti’s Syndrome
6. Congenital Conditions Associated with Bertolotti’s Syndrome
- Hypermobility Ehlers-Danlos Syndrome (hEDS) – hEDS is a rare heritable tissue disorder that causes general joint hypermobility. It is the most common type of EDS, accounting for about 90% of cases. Some patients with hEDS have also been shown to have Craniocervical Instability (CCI) and Thoracic Outlet Syndrome. The specific genes that cause hEDS have not been elucidated. As such, there is no laboratory test that can definitively identify patients with hEDS (Gensemer et al., 2021).
- Spina Bifida Occulta - Spina bifida is a rare congenital disorder where there is incomplete neural tube closure during fetal development. Spina bifida occulta is a mild version of this condition where a gap is present between the vertebrae (Imbard et al., 2013).
- Cervical stenosis - Cervical stenosis is caused by the narrowing of the spinal canal in the cervical spine. Cervical stenosis can be congenital or acquired due to degenerative changes after birth (Goodwin & Hsu, 2023).
- Extra L6 vertebra – An L6 vertebra is formed due to the complete lumbarization of the S1 vertebra (Matson et al., 2020). This likely stems from incorrect spine segmentation during development determined by the HOX gene family (Carapuço et al., 2005).
- Thoracic outlet syndrome (TOS) – TOS describes a set of symptoms in the neck and shoulder characterized by numbness, tingling, and pain. This is caused by the compression of the nerves and blood vessels traversing the narrow space between the clavicle and the first rib. Neurogenic TOS, caused by the compression of the C5-T1 brachial plexus nerve roots, accounts for over 90% of TOS cases. Furthermore, patients with a cervical rib (i.e., a transitional anatomy) are predisposed to developing neurogenic TOS (Jones et al., 2019).
7. Diagnosis of Bertolotti’s Syndrome
7.1. Diagnostic Protocol for Bertolotti’s Syndrome
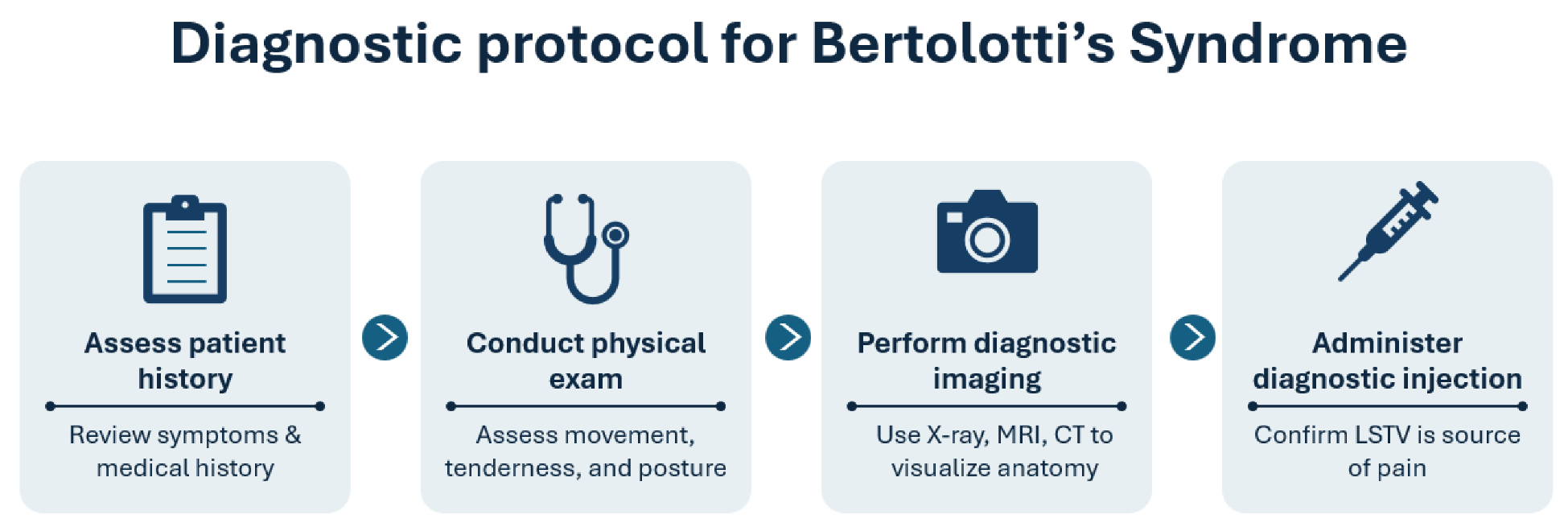
7.1.1. Assess Patient History
7.1.2. Conduct Physical Exam
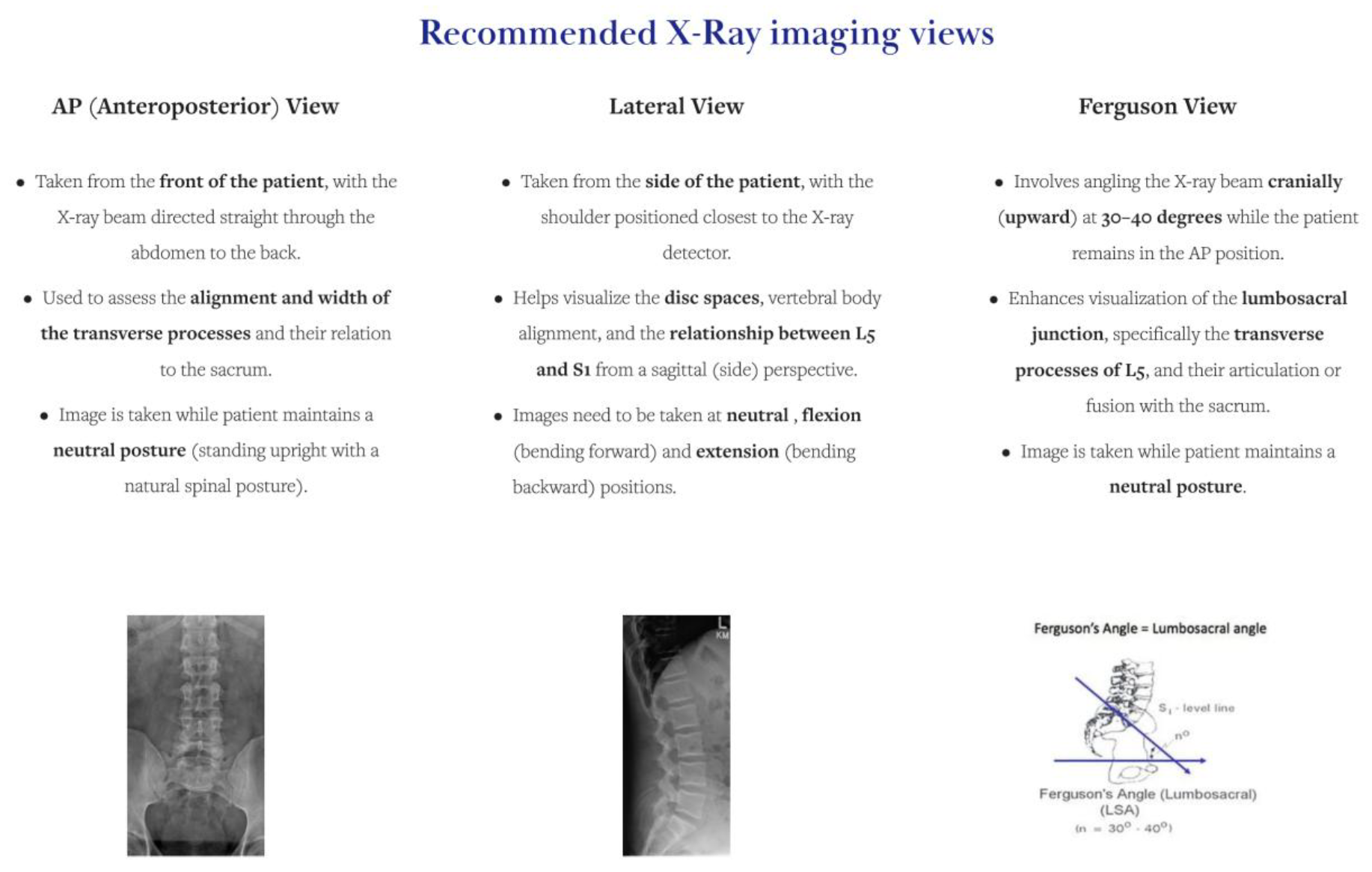
7.1.3. Diagnostic Imaging
7.1.4. Diagnostic Injection
8. Treatment Options for Bertolotti’s Syndrome
8.1. Step 1 – Conservative Therapy
- Avoiding provocative movements – Counsel patients to avoid or modify activities that require repetitive lumbar flexion, rotation and extension. Furthermore, high impact activities such as jumping exercises should be reduced or avoided.
- Proper lifting mechanics – Instruct patients proper lifting techniques (i.e., bending at the knees and keeping load close to the body) and to avoid excessive loading of the back. Patients should work closely with their physical therapist to determine safe ways to perform activities that induce strain on the lower back.
8.2. Step 2 – Interventional Therapy
8.3. Step 3 – Surgery
Resection vs Fusion
9. Conclusions and Areas for Further Investigation
9.1. What we Know so Far
9.2. Areas for Further Investigation
- Broad adoption of unified LSTV classification system – While the Jenkins classification offers a robust methodology that addresses the diagnostic limitations of the Castellvi classification, it has yet to be widely adopted across the medical field. Establishing a standardized nomenclature specific to Bertolotti’s Syndrome is critical for ensuring effective communication between diverse specialties, including radiology, neurology, and pain management. A unified language is the first step toward systematically documenting patient presentations, which will eventually allow researchers to aggregate data and correlate specific symptom profiles with distinct LSTV classifications.
- Standardization of conservative care protocols - While the principles of targeted physical therapy are broadly understood, there are no specific protocols to address the issues that arise in patients with Bertolotti’s Syndrome. Therefore, development and validation of standardized, evidence-based PT protocols could improve the efficacy of non-operative care, potentially reducing the number of patients who require surgery.
- Clarity on effective surgical techniques – There are limited studies currently available that investigate long-term outcomes of patients that have undergone surgery. There should be more studies aimed at developing a surgical protocol based on large-scale, long-term, evaluation of patient outcomes. Furthermore, there currently exists only one study (Jenkins, Chung, et al., 2023) that attempts to systematize the selection of a surgical approach based on the specific anatomy and symptoms of the patient. Although it is a great start, more studies are required to validate the findings before it can be widely adopted. The target state should be a clear rubric surgeons can utilize to determine the appropriate surgical approach for a patient.
- Establish global patient registry – The establishment of a global patient registry is critical for tracking the natural history of Bertolotti’s Syndrome outside of select surgical populations. Integrating Patient-Reported Outcome Measures (PROMs) into this registry will provide a quantifiable metric for patient burden that imaging alone cannot capture. This data is vital for enhancing diagnostic rigor, as it allows for the correlation of specific LSTV types with functional impairments. Ultimately, a robust dataset of PROMs will reveal the long-term efficacy of various interventions, guiding the development of more effective, patient-centered treatment protocols.
References
- Adams, R.; Herrera-Nicol, S.; Jenkins, A. L. Surgical Treatment of a Rare Presentation of Bertolotti’s Syndrome from Castellvi Type IV Lumbosacral Transitional Vertebra: Case Report and Review of the Literature. Journal of Neurological Surgery Reports 2018, 79(3), e70–e74. [Google Scholar] [CrossRef]
- Ahn, Y. H.; Seo, J. H.; Ju, C. I.; Kim, P. Surgical Options for Bertolotti Syndrome. Journal of Minimally Invasive Spine Surgery and Technique 2024, 9(2), 170–179. [Google Scholar] [CrossRef]
- Ali, M.; Enchev, Y. A rare cause of low back pain in adolescence – Bertolotti syndrome: A case report. Surgical Neurology International 2023, 14, 383. [Google Scholar] [CrossRef]
- Alonzo, F.; Cobar, A.; Cahueque, M.; Prieto, J. A. Bertolotti’s syndrome: An underdiagnosed cause for lower back pain. Journal of Surgical Case Reports 2018, 2018(10), rjy276. [Google Scholar] [CrossRef]
- Burnham, R. Radiofrequency sensory ablation as a treatment for symptomatic unilateral lumbosacral junction pseudarticulation (Bertolotti’s syndrome): A case report. Pain Medicine (Malden, Mass.) 2010, 11(6), 853–855. [Google Scholar] [CrossRef]
- Carapuço, M.; Nóvoa, A.; Bobola, N.; Mallo, M. Hox genes specify vertebral types in the presomitic mesoderm. Genes & Development 2005, 19(18), 2116–2121. [Google Scholar] [CrossRef] [PubMed]
- Castellvi, A. E.; Goldstein, L. A.; Chan, D. P. K. Lumbosacral Transitional Vertebrae and Their Relationship With Lumbar Extradural Defects. Spine 1984, 9(5), 493. [Google Scholar] [CrossRef]
- Crane, J.; Cragon, R.; O’Neill, J.; Berger, A. A.; Kassem, H.; Sherman, W. F.; Paladini, A.; Varrassi, G.; Odisho, A. S.; Miriyala, S.; Kaye, A. D. A Comprehensive Update of the Treatment and Management of Bertolotti’s Syndrome: A Best Practices Review. Orthopedic Reviews 2021, 13(2), 24980. [Google Scholar] [CrossRef] [PubMed]
- FABER Test. Physiopedia. n.d. Available online: https://www.physio-pedia.com/FABER_Test.
- Gensemer, C.; Burks, R.; Kautz, S.; Judge, D. P.; Lavallee, M.; Norris, R. A. Hypermobile Ehlers-Danlos syndromes: Complex phenotypes, challenging diagnoses, and poorly understood causes. Developmental Dynamics: An Official Publication of the American Association of Anatomists 2021, 250(3), 318–344. [Google Scholar] [CrossRef]
- Goodwin, A. M.; Hsu, W. K. Congenital Cervical Stenosis: A Review of the Current Literature. Current Reviews in Musculoskeletal Medicine 2023, 16(9), 438–445. [Google Scholar] [CrossRef] [PubMed]
- Holm, E. K.; Bünger, C.; Foldager, C. B. Symptomatic lumbosacral transitional vertebra: A review of the current literature and clinical outcomes following steroid injection or surgical intervention. SICOT-J 2017, 3, 71. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, S.; Yang, V.; Liew, D. F. Choosing a nonsteroidal anti-inflammatory drug for pain. Australian Prescriber 2025, 48(4), 139–144. [Google Scholar] [CrossRef]
- Hsieh, C.-Y. J.; Vanderford, J. D.; Moreau, S. R.; Prong, T. Lumbosacral transitional segments: Classification, prevalence, and effect on disk height. Journal of Manipulative & Physiological Therapeutics 2000, 23(7), 483–489. [Google Scholar] [CrossRef]
- Imbard, A.; Benoist, J.-F.; Blom, H. J. Neural Tube Defects, Folic Acid and Methylation. International Journal of Environmental Research and Public Health 2013, 10(9), 4352–4389. [Google Scholar] [CrossRef]
- Jain, A.; Agarwal, A.; Jain, S.; Shamshery, C. Bertolotti Syndrome: A Diagnostic and Management Dilemma for Pain Physicians. The Korean Journal of Pain 2013, 26(4), 368–373. [Google Scholar] [CrossRef]
- Jancuska, J. M.; Spivak, J. M.; Bendo, J. A. A Review of Symptomatic Lumbosacral Transitional Vertebrae: Bertolotti’s Syndrome. International Journal of Spine Surgery 2015, 9. [Google Scholar] [CrossRef]
- Jenkins, A. L.; Chung, R. J.; O’Donnell, J.; Hawks, C.; Jenkins, S.; Lazarus, D.; McCaffrey, T.; Terai, H.; Harvie, C.; Matsoukas, S. Redefining the Treatment of Lumbosacral Transitional Vertebrae for Bertolotti Syndrome: Long-Term Outcomes Utilizing the Jenkins Classification to Determine Treatment. World Neurosurgery 2023, 175, e21–e29. [Google Scholar] [CrossRef]
- Jenkins, A. L.; O’Donnell, J.; Chung, R. J.; Jenkins, S.; Hawks, C.; Lazarus, D.; McCaffrey, T.; Terai, H.; Harvie, C. Redefining the Classification for Bertolotti Syndrome: Anatomical Findings in Lumbosacral Transitional Vertebrae Guide Treatment Selection. World Neurosurgery 2023, 175, e303–e313. [Google Scholar] [CrossRef]
- Jones, M. R.; Prabhakar, A.; Viswanath, O.; Urits, I.; Green, J. B.; Kendrick, J. B.; Brunk, A. J.; Eng, M. R.; Orhurhu, V.; Cornett, E. M.; Kaye, A. D. Thoracic Outlet Syndrome: A Comprehensive Review of Pathophysiology, Diagnosis, and Treatment. Pain and Therapy 2019, 8(1), 5–18. [Google Scholar] [CrossRef] [PubMed]
- Konin, G. P.; Walz, D. M. Lumbosacral Transitional Vertebrae: Classification, Imaging Findings, and Clinical Relevance. AJNR: American Journal of Neuroradiology 2010, 31(10), 1778–1786. [Google Scholar] [CrossRef]
- Lawrence, J. E. G.; Roberts, K.; Tuck, E.; Li, T.; Mamanova, L.; Balogh, P.; Usher, I.; Piapi, A.; Mazin, P.; Anderson, N. D.; Bolt, L.; Richardson, L.; Prigmore, E.; He, X.; Barker, R. A.; Flanagan, A.; Young, M. D.; Teichmann, S. A.; Bayraktar, O.; Behjati, S. HOX gene expression in the developing human spine. Nature Communications 2024, 15(1), 10023. [Google Scholar] [CrossRef]
- Matson, D. M.; MACCORMICK, L. M.; SEMBRANO, J. N.; POLLY, D. W. Sacral Dysmorphism and Lumbosacral Transitional Vertebrae (LSTV) Review. International Journal of Spine Surgery 2020, 14 (Suppl 1), S14–S19. [Google Scholar] [CrossRef]
- McGrath, K. A.; Thompson, N. R.; Fisher, E.; Kanasz, J.; Golubovsky, J. L.; Steinmetz, M. P. Quality-of-life and postoperative satisfaction following pseudoarthrectomy in patients with Bertolotti syndrome. The Spine Journal: Official Journal of the North American Spine Society 2022, 22(8), 1292–1300. [Google Scholar] [CrossRef]
- Poe, L. B. Bertolotti’s Syndrome | Radsource. 27 December 2013. Available online: https://radsource.us/bertolottis-syndrome/.
- Riyami, K. S. A.; Alahmed, S. M.; Saad, Z. Z.; Bomanji, J. Evaluation and Identification of Lumbosacral Transitional Vertebra Causing Intractable Low Back Pain Utilizing Bone Single-Photon Emission Tomography with Computed Tomography. World Journal of Nuclear Medicine 2017, 16(4), 328–330. [Google Scholar] [CrossRef] [PubMed]
- Rux, D. R.; Wellik, D. M. Hox genes in the adult skeleton: Novel functions beyond embryonic development. In Developmental Dynamics; 2017. [Google Scholar] [CrossRef]
- Uçar, D.; Uçar, B. Y.; Coşar, Y.; Emrem, K.; Gümüşsuyu, G.; Mutlu, S.; Mutlu, B.; Çaçan, M. A.; Mertsoy, Y.; Gümüş, H. Retrospective Cohort Study of the Prevalence of Lumbosacral Transitional Vertebra in a Wide and Well-Represented Population. Arthritis 2013, 2013, 461425. [Google Scholar] [CrossRef] [PubMed]
- Wojtulewski, J. A.; Schattenkirchner, M.; Barceló, P.; Le Loët, X.; Bevis, P. J.; Bluhmki, E.; Distel, M. A six-month double-blind trial to compare the efficacy and safety of meloxicam 7.5 mg daily and naproxen 750 mg daily in patients with rheumatoid arthritis. British Journal of Rheumatology 1996, 35 Suppl 1, 22–28. [Google Scholar] [CrossRef]
- Yadav, R. K.; Thakur, J. P.; Amatya, S. P.; Sherpa, T. Radiofrequency Ablation for Treatment of Symptomatic Bertolotti’s Syndrome II B. Journal on Musculoskeletal Ultrasound and Pain Medicine 2024, 9(1), 17. [Google Scholar] [CrossRef]
- Zhu, W.; Ding, X.; Zheng, J.; Zeng, F.; Zhang, F.; Wu, X.; Sun, Y.; Ma, J.; Yin, M. A systematic review and bibliometric study of Bertolotti’s syndrome: Clinical characteristics and global trends. International Journal of Surgery 2023, 109(10), 3159. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



