1. Introduction
Pelvic Inflammatory Disease (PID) represents a significant public health issue, particularly in sub-Saharan Africa, where the incidence of reproductive tract infections (RTIs) remains high among young women. PID is a condition characterized by the infection of the upper female reproductive organs, including the uterus, fallopian tubes, ovaries, and surrounding pelvic structures, and is primarily caused by sexually transmitted infections (STIs) like Chlamydia trachomatis and Neisseria gonorrhoeae (Raimi & Ochayi, 2017; Adebayo & Olatunji, 2022; Akinyemi et al., 2022). This disease is often overlooked or misdiagnosed due to the lack of adequate diagnostic facilities and healthcare resources in many parts of sub-Saharan Africa (World Health Organization [WHO], 2021; Nwankwo et al., 2021). Among sexually active young women, particularly those in tertiary education institutions, PID poses significant risks, including infertility, chronic pelvic pain, and increased susceptibility to ectopic pregnancy (Akinyemi et al., 2020; Nwadioha et al., 2020). The majority of these women, especially those in hostel environments, are exposed to factors that increase their vulnerability to RTIs, such as multiple sexual partners, inconsistent condom use, and limited access to sexual health education (Adefuye et al., 2021; Afolayan et al., 2021). While the risks associated with PID among young women are well documented, much remains unknown regarding the prevalence, microbial agents, and antimicrobial susceptibility patterns in certain geographic areas, such as Niger Delta University in Nigeria. There is a significant gap in local, evidence-based data that hinders effective diagnosis and treatment of PID (Okafor & Nwankwo, 2023; Tukur et al., 2022). The absence of systematic microbiological investigation of PID, particularly in hostel settings where there is limited access to healthcare services, exacerbates the challenges of treating infections effectively. Studies conducted in Nigerian universities have highlighted the general awareness and health-seeking behavior challenges faced by female students in hostel environments, yet there is a lack of microbiological and antimicrobial resistance data specific to these populations (Olowe et al., 2022; Umar et al., 2021; Ndu et al., 2025; Christopher et al., 2025a, b; Teddy et al., 2025). Therefore, the research gap lies in the insufficient local data on the microbial agents involved in PID and their resistance patterns in these settings, which limits the ability to provide targeted and effective treatments. The need for this study is urgent given the increasing rates of antimicrobial resistance (AMR) among urogenital pathogens in sub-Saharan Africa (Nwankwo et al., 2021; Okeke et al., 2022). Inadequate management of PID, primarily due to empirical treatment without laboratory confirmation, leads to the recurrence of infections and complications (Cheesbrough, 2020). The rising global concern over AMR necessitates the need for this study to generate locally relevant microbiological and antimicrobial susceptibility data. Such data are essential for guiding clinicians in the appropriate selection of antibiotics, reducing unnecessary drug use, and ultimately improving the reproductive health outcomes of affected women (CLSI, 2023; WHO, 2022). Moreover, the study will contribute to addressing the significant public health issue of PID among female students in university hostels, which is often neglected in favor of broader public health concerns (Brunham et al., 2021). By using high vaginal swab and urine culture techniques combined with antimicrobial sensitivity testing, this study aims to identify the causative agents of PID and their resistance patterns in the study population, offering a comprehensive approach to disease diagnosis and management. In light of these gaps and the urgency of addressing PID in university hostel settings, this study will provide vital insights into the prevalence, microbiological agents, and antimicrobial resistance patterns associated with PID among female students in the Niger Delta University, Amassoma. By employing robust microbiological techniques, including high vaginal swab and urine culture methods, alongside antimicrobial susceptibility testing, this research seeks to generate accurate and locally relevant data that will inform clinical decisions, improve treatment strategies, and contribute to the development of preventive measures. The specific objectives of this study are to collect high vaginal swab and urine samples from female hostel residents, identify and isolate bacterial pathogens associated with PID, determine the prevalence of microbial isolates, and perform antimicrobial susceptibility testing to identify resistance patterns. This will not only provide crucial data for clinicians but will also serve as a model for similar studies in other tertiary institutions in Nigeria and sub-Saharan Africa.
2. Materials and Methods
2.1. Study Area
This study was conducted at Niger Delta University (NDU), Amassoma, located in the Southern Ijaw Local Government Area of Bayelsa State, Nigeria. Amassoma is a university town with a large student population, particularly among female undergraduates, many of whom reside in organized hostel facilities within the university campus. The communal living conditions in the hostels, along with the age distribution of the female students, provide an ideal environment for studying Pelvic Inflammatory Disease (PID), which is known to disproportionately affect young, sexually active women (Akinyemi et al., 2020). Furthermore, the high concentration of female students in these hostels makes the study setting particularly relevant for exploring the prevalence and microbial agents associated with PID in this demographic. Laboratory analyses were performed in a standard microbiology laboratory using established bacteriological procedures (Cheesbrough, 2020; Adebayo & Olatunji, 2022).
2.2. Study Design
A descriptive cross-sectional study design was adopted for this investigation to assess the prevalence of PID among female students residing in the hostels at Niger Delta University, Amassoma. The cross-sectional design allows for a snapshot of the microbial agents associated with PID at a specific point in time, without influencing or manipulating the variables being studied (Adefuye et al., 2021; Okafor & Nwankwo, 2023). This type of study design is ideal for identifying the microorganisms involved in PID and the antimicrobial resistance patterns among female students without altering their behaviors or clinical practices. The design was particularly beneficial in capturing a broad range of microbial agents that may be responsible for PID, and it provided a comprehensive, real-time assessment of the current situation at the university (Nwankwo et al., 2021).
2.3. Study Population
The study population consisted of female students residing in the hostels at Niger Delta University, Amassoma, within the reproductive age group (18-49 years). The inclusion of this specific population allowed for a focused investigation into the risk factors and prevalence of PID in an environment where young women, particularly students, are at an increased risk of contracting reproductive tract infections (RTIs) due to behavioral and environmental factors (Torrone et al., 2021). Participation in the study was voluntary, and only those who provided informed consent were enrolled. This approach ensured ethical compliance and respect for participants’ autonomy while allowing for an accurate representation of the student population in relation to PID (Akinyemi et al., 2020). The inclusion of only female students who gave consent also ensured that the findings were based on a population directly impacted by PID, thereby increasing the study’s relevance and applicability to real-world health issues faced by young women in tertiary institutions (Okeke et al., 2022).
2.4. Sample Size Determination
A total of fifty (50) samples were collected from female hostel residents at Niger Delta University, Amassoma. The sample size was determined based on the feasibility of conducting the study within the available resources, as well as the laboratory capacity to handle and process the samples effectively. This sample size is consistent with similar microbiological-based studies conducted in Nigerian universities, which aim to investigate reproductive health issues among female students (Afolayan et al., 2021). The sample size was considered adequate for the descriptive analysis required to identify microbial isolates and determine the antimicrobial resistance patterns associated with PID in this specific population. This approach allowed for a comprehensive understanding of the PID landscape among female students and facilitated meaningful analysis of the findings (Adebayo & Olatunji, 2022).
2.5. Sampling Technique
A convenience sampling technique was employed for the recruitment of study participants. This method was selected due to the ease of access to participants within the university’s hostel facilities, as well as the need for a pragmatic approach in achieving the desired sample size within a reasonable timeframe. Eligible participants who met the inclusion criteria and provided informed consent were consecutively recruited until the required number of samples was obtained. Convenience sampling is a practical technique often used in epidemiological studies, particularly when studying specific populations such as university students, and it allowed the study to gather sufficient data from the target group in an efficient manner (Nwankwo et al., 2021). Although this method may introduce some biases, it was deemed appropriate given the resource constraints and the specific context of the study (Akinyemi et al., 2022).
2.6. Inclusion and Exclusion Criteria
Female students residing in the hostels at Niger Delta University who consented to participate in the study were eligible for inclusion. The study focused specifically on women within the reproductive age group (18-49 years), as they are the demographic most vulnerable to developing PID (Torrone et al., 2021). Exclusion criteria included students who were menstruating at the time of sample collection, as menstrual blood could interfere with microbial analysis and affect the accuracy of results. Additionally, students who had recently taken antibiotics were excluded, as antibiotic use could alter the microbial flora, potentially masking the presence of pathogens associated with PID (Haggerty & Taylor, 2020). Students who declined to provide consent were also excluded from the study to ensure that only participants who voluntarily agreed to participate were included, in alignment with ethical standards (WHO, 2022).
2.7. Sample Collection
Two types of specimens were collected from each participant to ensure comprehensive microbial analysis. High vaginal swab (HVS) samples were collected using sterile swab sticks by trained personnel to obtain vaginal secretions from the vaginal canal. The swab was gently inserted into the canal, rotated to collect secretions, and immediately placed in a sterile transport medium for laboratory analysis. The second sample type was midstream urine, which was collected into sterile universal containers using the clean-catch method to minimize contamination. Proper instructions were provided to participants to ensure that urine samples were collected without external contamination. These samples were promptly transported to the laboratory for analysis (Akinyemi et al., 2020). The combined use of high vaginal swabs and urine samples enabled a comprehensive examination of both upper and lower reproductive tract infections associated with PID (Brunham et al., 2021).
2.8. Laboratory Analysis
The laboratory analysis followed standard microbiological techniques to identify and characterize the microbial agents responsible for PID. For high vaginal swab cultures, the swabs were inoculated onto blood agar, chocolate agar, and MacConkey agar plates, and incubated at 37°C for 24-48 hours. The colonies that grew were examined based on colony morphology, Gram staining, and standard biochemical tests. Urine samples were cultured using the standard calibrated loop technique on Cystine Lactose Electrolyte Deficient (CLED) agar and MacConkey agar, and incubated at 37°C for 24 hours. Significant bacteriuria was identified based on colony counts, and isolates were characterized through Gram staining and biochemical tests such as catalase and coagulase (Adebayo & Olatunji, 2022; Cheesbrough, 2020). These microbiological methods were essential in identifying the causative agents of PID and in guiding appropriate therapeutic interventions.
2.9. Identification of Isolates
Microbial isolates obtained from both high vaginal swab and urine cultures were identified using standard microbiological techniques, including Gram reaction, cellular morphology, and biochemical tests. The use of Gram staining allowed for the initial categorization of bacteria into Gram-positive or Gram-negative groups, while biochemical tests such as catalase and coagulase were used to further identify bacterial species. This combination of techniques is widely regarded as the gold standard for bacterial identification in clinical microbiology and ensures accurate identification of the pathogens involved in PID (Nwankwo et al., 2021). The identification of these isolates was critical in understanding the microbial profile of PID in the study population and in determining the best therapeutic approach for managing the infections.
2.10. Antimicrobial Susceptibility Testing
Antimicrobial susceptibility testing was performed on all bacterial isolates using the Kirby–Bauer disk diffusion method, following the Clinical and Laboratory Standards Institute (CLSI) guidelines. Isolates were emulsified in sterile normal saline and adjusted to match the 0.5 McFarland turbidity standard. The inoculum was uniformly spread onto the surface of Mueller–Hinton agar plates using a sterile swab. Commercially prepared antibiotic discs were placed aseptically on the inoculated plates, and the plates were incubated at 37°C for 18–24 hours. After incubation, the zones of inhibition around each antibiotic disc were measured in millimeters and interpreted as sensitive, intermediate, or resistant based on CLSI interpretative criteria (Okeke et al., 2022; Haggerty & Taylor, 2020). This testing provided important information on the antimicrobial resistance patterns of the isolates, helping to inform clinical treatment options for PID.
2.11. Data Analysis
Data obtained from the laboratory investigations were analyzed using descriptive statistical methods. The frequency and percentage of different microbial isolates were calculated and presented using tables and charts for clear visualization of the results. This method allowed for easy comparison of the prevalence of specific pathogens and their resistance patterns, providing a comprehensive overview of the microbial landscape of PID in the study population (Nwankwo et al., 2021). Descriptive statistics are commonly used in epidemiological studies to summarize and present data, and they provide a reliable means of summarizing the findings of this study in a meaningful way.
2.12. Ethical Considerations
Ethical approval for this study was obtained from the Institutional Review Board (IRB) of the Niger Delta University Teaching Hospital (NDUTH) before commencement. Approval body: Niger Delta University Teaching Hospital (NDUTH) with IRB Approval number: NDUTH/IRB/2025/045; Approval date: March 15, 2025. Informed consent was obtained from all participants after a clear explanation of the study’s purpose, procedures, and potential benefits; participation was entirely voluntary. Confidentiality of participants’ personal information and sample results was strictly maintained through anonymized coding and secure data storage. Participants were also informed of their right to withdraw from the study at any time without any penalty or impact on their academic standing. These measures ensured full compliance with international ethical standards, including the Declaration of Helsinki, for research involving human subjects in sensitive reproductive health contexts like PID prevalence among female university students.
3. Result
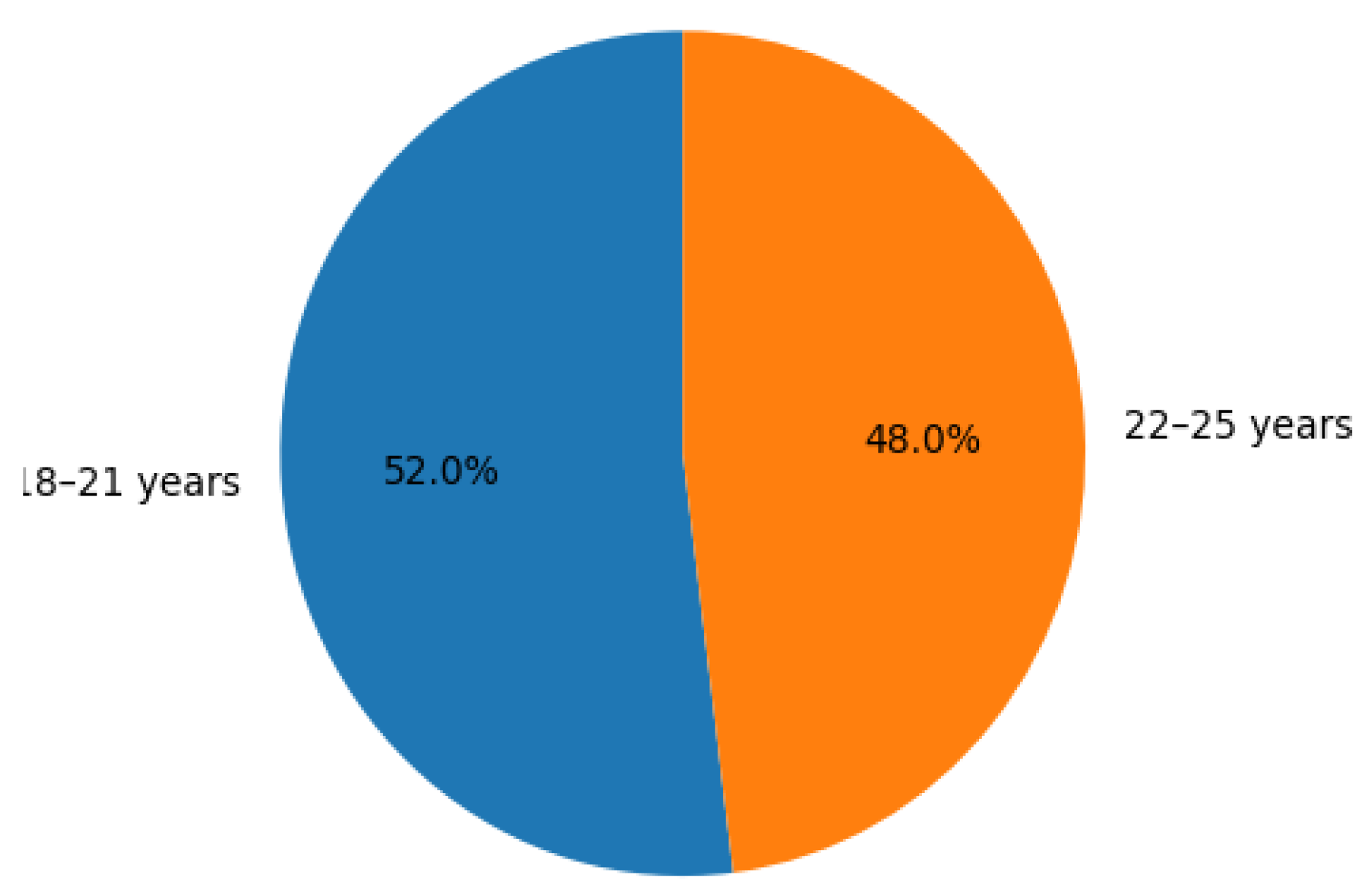
The age distribution of participants in this study, as shown in
Table 1 and
Figure 1, indicates that 52% of the participants were within the 18-21-year age range, while 48% were within the 22-25-year age range. This suggests that the majority of the female students residing in the hostels at Niger Delta University are relatively young adults, with a slightly higher proportion in the older age category. The age range of 22-25 years is particularly notable, as individuals in this group are typically at a stage where they are more likely to be sexually active, which places them at a heightened risk for reproductive tract infections, including Pelvic Inflammatory Disease (PID). Given the communal living environment in university hostels, factors such as limited access to personal healthcare, shared sanitation facilities, and potentially risky sexual behaviors make this age group particularly susceptible to PID. The key takeaway from this table is the demographic profile of participants, which highlights the high proportion of young adult women who are at risk of PID due to both biological and environmental factors. The significance of this distribution lies in its alignment with the known risk factors for PID, especially in a university setting, where the combination of sexual activity and limited healthcare access may exacerbate the likelihood of infections. The real-world implications of these findings suggest that interventions such as targeted sexual health education, regular screening for sexually transmitted infections (STIs), and improved healthcare services in university hostels could be critical in reducing the incidence of PID among female students. Moreover, this research could serve as a model for similar studies in other university settings, helping to inform public health policies and interventions aimed at reducing the reproductive health risks faced by young women in sub-Saharan Africa.
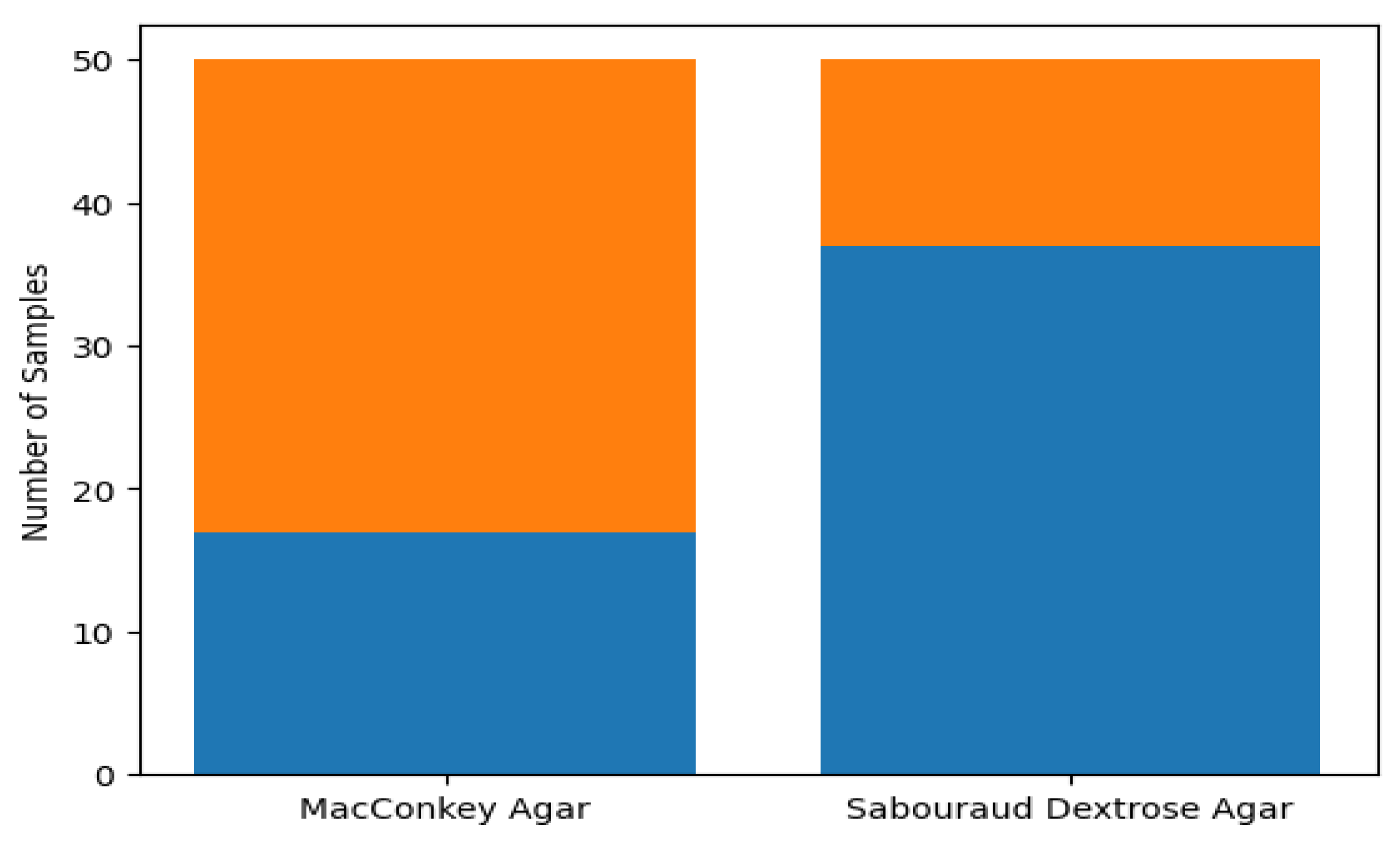
The findings presented in
Table 2 and
Figure 2 show the percentage distribution of microbial growth on two different culture media used in the study: MacConkey agar and Sabouraud dextrose agar. On MacConkey agar, 34% of samples demonstrated significant growth (S.G), while 66% showed no growth (N.G). This pattern indicates that a portion of the samples contained Gram-negative enteric bacteria, which are commonly associated with reproductive tract infections. On the other hand, Sabouraud dextrose agar showed a much higher rate of significant growth, with 74% of samples demonstrating fungal growth, while only 26% showed no growth. This suggests a higher prevalence of fungal organisms, particularly Candida species, in the study population. The growth patterns on these media highlight the diverse microbial load present in the female student population, with a clear dominance of fungal pathogens in this environment. The key takeaway from these findings is that fungal infections, particularly those caused by Candida species, are more prevalent than bacterial infections among the study population. The higher rate of significant growth on Sabouraud dextrose agar reflects the substantial fungal burden, which may contribute significantly to reproductive tract infections in this group of young women. The significance of this result is that it challenges the conventional focus on bacterial pathogens in PID, highlighting the need for comprehensive diagnostic approaches that also consider fungal infections. The real-world implications of these findings suggest that health interventions in hostel environments should not only address bacterial infections but also include strategies for managing and preventing fungal infections, which are often under-recognized in clinical settings. Additionally, these results emphasize the need for targeted education and treatment programs for female students to address the full spectrum of microbial threats to their reproductive health.
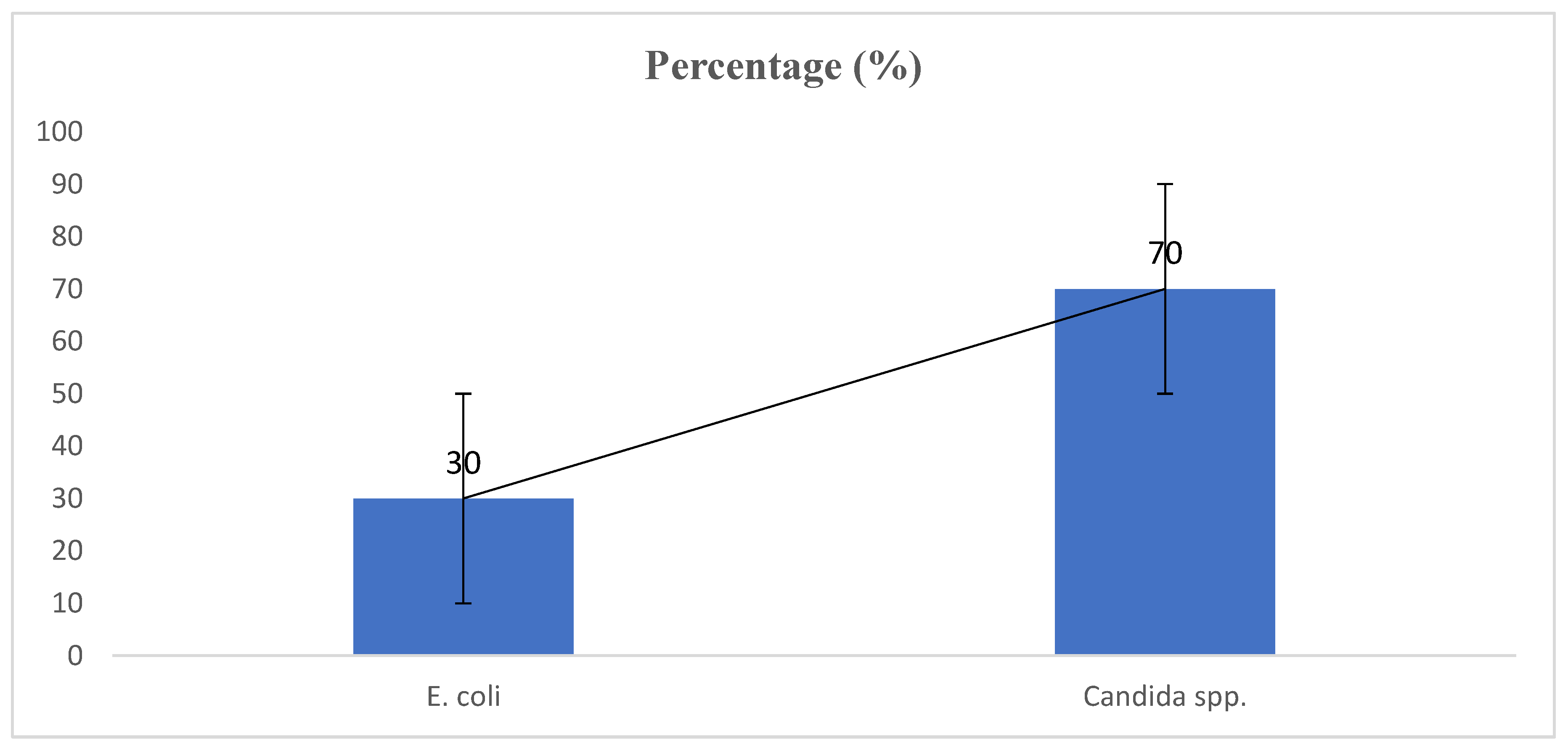
The microbial distribution reveals that 70% of the isolates were Candida species, while 30% were
Escherichia coli. This indicates that fungal infections, particularly those caused by Candida species, are more prevalent in the study population compared to bacterial infections caused by
E. coli. The presence of
E. coli suggests potential contamination from enteric sources, which could be linked to hygiene-related issues or improper sanitation. The predominance of Candida species underscores the significance of fungal infections in reproductive tract infections among young women in the university hostel setting. The key takeaway from
Table 3 and
Figure 3 is the overwhelming presence of Candida species compared to
E. coli, suggesting that fungal infections play a more substantial role in reproductive tract infections in the study population. This finding is significant as it highlights the need for healthcare interventions to address fungal infections, which are often under-recognized and inadequately treated. The real-world implications of these results suggest that sexual health education and prevention programs should include awareness about fungal infections, particularly Candida, and emphasize hygiene practices to reduce the risk of contamination by enteric bacteria like
E. coli. These findings can inform targeted clinical management strategies and public health interventions aimed at reducing reproductive health issues in university settings.
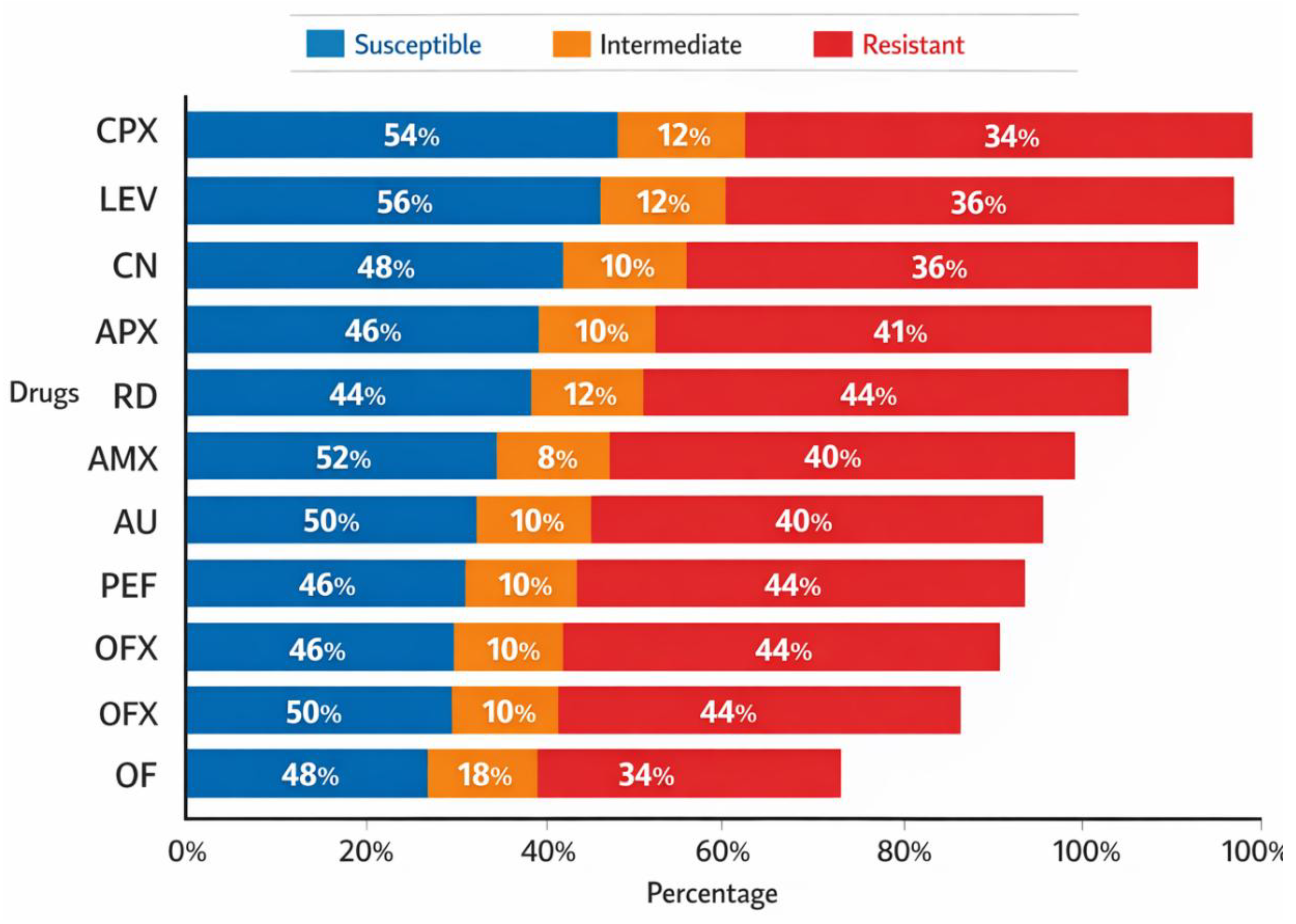
The antimicrobial susceptibility testing results show varying levels of resistance, intermediate response, and susceptibility across different drugs. Ciprofloxacin (CPX) and Levofloxacin (LEV) demonstrated the highest susceptibility, with 54% and 56% of isolates being susceptible, respectively. However, a significant portion of isolates showed resistance, with CPX and LEV having resistance rates of 34% and 36%, respectively. The other antibiotics, such as Gentamicin (CN), Ampicillin (APX), and Amoxicillin (AMX) showed relatively higher resistance rates, with CN and APX demonstrating 36% and 41% resistance, respectively. Resistance to commonly used antibiotics like Amoxicillin and Augmentin (AU) indicates a growing concern for antimicrobial resistance in the study population. The pattern of susceptibility and resistance is critical for guiding effective treatment options for infections associated with pelvic inflammatory disease. The key takeaway from
Table 4 and
Figure 4 is the high level of resistance seen in several commonly used antibiotics, including Ampicillin, Amoxicillin, and Ofloxacin, which poses significant challenges for the management of pelvic inflammatory disease. This trend underscores the importance of antimicrobial susceptibility testing in clinical settings to tailor appropriate treatments for infections. The real-world implications suggest that there is an urgent need for careful antibiotic stewardship, including the restricted use of certain antibiotics and the promotion of alternative treatment strategies. The findings emphasize the need for healthcare professionals to be cautious in prescribing antibiotics and to consider the rising resistance when developing treatment protocols. Furthermore, these results highlight the necessity for ongoing surveillance of antimicrobial resistance patterns to ensure effective management of reproductive tract infections in young women.
4. Discussion
The age distribution of participants in this study revealed that 52% of the participants were aged between 18 and 21 years, while 48% were within the 22 to 25-year age range. This suggests a predominant representation of young adults, with a slightly higher proportion in the older age group. Akin to findings by Adebayo and Olatunji (2022) and Adefuye et al. (2021), this study highlights that female university students in sub-Saharan Africa, particularly those within the reproductive age range, are at an increased risk of reproductive tract infections (RTIs) and pelvic inflammatory disease (PID). Afolayan et al. (2021) also noted that university students tend to be more sexually active, which further increases their vulnerability to sexually transmitted infections (STIs) and related complications. Given the communal living arrangements in university hostels, factors such as poor access to personal healthcare, shared sanitation facilities, and high-risk sexual behaviors might exacerbate the susceptibility of students in the 22-25-year age group to PID (Akinyemi et al., 2020; Okafor & Nwankwo, 2023). This aligns with the findings from Akinyemi et al. (2022), where they reported a higher incidence of PID among sexually active women in communal living spaces, further underlining the need for tailored health interventions for university populations. The microbial distribution observed in this study indicates that Candida species were predominant, accounting for 70% of the isolates, while Escherichia coli was found in 30% of the samples. This finding mirrors the results of Okorie et al. (2021) and Akinyemi et al. (2020), who also observed Candida species to be more prevalent than bacterial pathogens in the reproductive tract infections among young women in Nigeria. The dominance of Candida species aligns with the higher prevalence of fungal infections, particularly in women residing in environments with high communal exposure and potentially suboptimal hygiene (Sharma et al., 2014; Raimi et al., 2018; Olalekan et al., 2018; Raimi et al., 2019a, b; Olalekan et al., 2019; Gift and Olalekan, 2020; Raimi et al., 2020; Olalekan et al., 2020; Raimi and Raimi, 2020; Gift et al., 2020; Brunham et al., 2021; Raimi et al., 2021; Morufu et al., 2021; Raimi, 2024; Omotoso et al., 2025). The presence of E. coli in some samples is consistent with previous studies by Nwankwo et al. (2021), who identified enteric bacteria as significant contributors to RTIs, often associated with poor sanitation and hygiene practices in university hostels (Cheesbrough, 2020; Gottlieb et al., 2020). The findings in this study are thus in agreement with global observations, suggesting that while bacterial infections are important, fungal pathogens such as Candida species play a predominant role in reproductive tract infections among young women (Tuebi et al., 2021; Torrone et al., 2021; Sharma et al., 2025). The antimicrobial susceptibility testing results revealed varying levels of susceptibility and resistance across different drugs. Ciprofloxacin and Levofloxacin showed the highest susceptibility, with 54% and 56% of isolates being susceptible, respectively. However, resistance rates for these drugs were also high, with 34% and 36% resistance rates, respectively. This is consistent with Nwankwo et al. (2021), who reported similar resistance patterns in urogenital pathogens in Nigeria, with significant resistance to commonly prescribed antibiotics. Moreover, the high resistance to drugs like Amoxicillin and Augmentin observed in this study corroborates the findings of Okeke et al. (2022), who highlighted the rising threat of antimicrobial resistance in sub-Saharan Africa, particularly in the context of reproductive tract infections (Akinyemi et al., 2020). The resistance rates observed in this study are concerning, as they suggest a growing challenge in treating PID and related infections with first-line antibiotics. Similar patterns of resistance have been documented in other parts of Nigeria by Nwadioha et al. (2020) and Ibrahim et al. (2020), who noted increasing antimicrobial resistance among reproductive tract pathogens, reflecting an urgent need for robust antimicrobial stewardship and regular surveillance (Raimi and Ochayi, 2017; Okeke et al., 2022; World Health Organization, 2022; Ndu et al., 2025; Christopher et al., 2025a, b; Teddy et al., 2025).
Additionally, the observed resistance to antibiotics such as Gentamicin, Ampicillin, and Augmentin in this study is a critical finding, as these antibiotics are commonly used in treating reproductive infections in clinical settings. This trend is consistent with studies conducted by Akinyemi et al. (2020) and Adefuye et al. (2021), who identified high rates of resistance among commonly used antibiotics, particularly in settings with poor sanitation and high rates of self-medication. The resistance to these antibiotics could be a consequence of improper use, including overprescription, inadequate dosing, or incomplete courses of treatment (Nwankwo et al., 2021). This issue has been extensively documented in the literature, with researchers such as Adebayo and Olatunji (2022) and Okafor & Nwankwo (2023) urging for improved health education and policy interventions to curb the misuse of antibiotics, which could help slow the rising tide of resistance. The significant resistance to Amoxicillin and Augmentin in this study further emphasizes the need for the adoption of alternative treatment strategies and the development of new therapeutic options for the management of PID (Gottlieb et al., 2020; Haggerty & Taylor, 2020). In comparison to similar studies conducted in other regions, the findings of this study align with broader global trends in antimicrobial resistance. For instance, the study by Nwankwo et al. (2021) in Nigeria and Okeke et al. (2022) in Uganda highlighted similar patterns of resistance to first-line antibiotics, particularly in urogenital pathogens. The findings of this study are in line with the observations of Akinyemi et al. (2022), who reported a significant increase in the resistance of urogenital pathogens to commonly used antibiotics in Nigeria. This pattern has also been observed in other African countries, where resistance to commonly prescribed antibiotics has become a major concern in the management of RTIs and PID (Gottlieb et al., 2020; Nwankwo et al., 2021). These findings stress the need for continuous monitoring of antimicrobial susceptibility and the importance of developing local guidelines for the management of reproductive tract infections, particularly in resource-limited settings where the overuse of antibiotics is a common practice (Clinical and Laboratory Standards Institute, 2023; Akinyemi et al., 2020). Lastly, the prevalence of fungal infections, particularly Candida species, in this study underscores the importance of considering both bacterial and fungal pathogens in the management of PID. Previous studies by Akinyemi et al. (2022) and Nwankwo et al. (2021) have similarly noted the high prevalence of Candida species in reproductive tract infections, particularly in environments with communal living, poor sanitation, and high-risk sexual behaviors. This is consistent with global findings by Haggerty and Taylor (2020) and Brunham et al. (2021), who emphasized the growing role of fungal infections, especially Candida species, in the etiology of PID. Given the high prevalence of Candida species in this study, there is a need to prioritize antifungal therapies alongside antibiotics in the treatment protocols for PID in young women. This recommendation is supported by the findings of Adebayo and Olatunji (2022) and Okafor & Nwankwo (2023), who highlighted the critical role of antifungal treatments in addressing the rising burden of Candida infections in reproductive health. The findings of this study, therefore, contribute to the growing body of evidence on the importance of comprehensive microbial testing in diagnosing and treating PID in young women, with particular attention to both bacterial and fungal pathogens.
5. Study Limitation
This study has several limitations that should be considered when interpreting the results. First, the cross-sectional design, while providing valuable snapshot data, does not allow for causal inferences or the assessment of temporal relationships between risk factors and the development of pelvic inflammatory disease (PID). Additionally, the study focused on a specific population: female students residing in hostels at Niger Delta University, limiting the generalizability of the findings to other regions or broader demographics. The use of convenience sampling may have introduced selection bias, as only participants who consented were included, potentially excluding those with less willingness or access to participate. Another limitation is the reliance on laboratory-based microbiological testing, which, while comprehensive, may not capture all potential pathogens, particularly those that are difficult to culture or require more advanced molecular techniques. Moreover, the study did not assess other important variables such as sexual behavior patterns, contraceptive use, or socio-economic factors that may influence the risk of PID and microbial infections. Lastly, the study did not track antibiotic use or resistance over time, which could have provided further insights into the evolving patterns of antimicrobial resistance and their impact on treatment efficacy. These limitations suggest that while the findings provide valuable insights, they should be interpreted with caution, and future studies should aim for more robust designs and broader population coverage.
6. Implications for Policy and Interventions
The findings from this study have important implications for public health policy and interventions aimed at reducing the burden of pelvic inflammatory disease (PID) and reproductive tract infections among young women, particularly those in university settings. Given the high prevalence of Candida species and the resistance patterns observed for common antibiotics, there is a pressing need for enhanced awareness campaigns targeting sexual and reproductive health, emphasizing safe sexual practices, hygiene, and the importance of regular health screenings. Policies should encourage improved access to healthcare services, particularly in university environments, where communal living may increase the risk of infections. Additionally, strengthening antimicrobial stewardship programs is crucial to address the emerging issue of antibiotic resistance, ensuring that appropriate treatments are available and effective. The findings also highlight the necessity for more comprehensive education on the risks of unprotected sex, the role of hygiene in preventing infections, and the dangers of self-medication with antibiotics. Policymakers should advocate for the inclusion of reproductive health education within university curricula and the establishment of robust healthcare facilities on campuses to provide timely diagnosis and treatment. Lastly, given the potential link between poor sanitation and increased infection rates, there is a need for improved infrastructural development within university hostels to ensure better access to clean water and sanitation facilities.
7. Summary of the Findings
The study revealed a significant prevalence of reproductive tract infections among female students residing in hostels at Niger Delta University, with Candida species being the most commonly isolated pathogen, followed by Escherichia coli. The age distribution of participants showed that a majority (52%) were in the 18-21 years age group, with 48% in the 22-25 years age group, indicating that young adult women are particularly susceptible to these infections. The microbiological analysis indicated a higher fungal burden, with 74% of samples showing significant growth on Sabouraud dextrose agar, while 34% of samples demonstrated growth on MacConkey agar, suggesting the presence of Gram-negative enteric bacteria. Antimicrobial susceptibility testing revealed significant resistance to commonly used antibiotics such as Ciprofloxacin, Levofloxacin, Gentamicin, and Amoxicillin, with resistance rates ranging from 34% to 41%. These findings highlight the predominance of fungal pathogens, the challenges of antibiotic resistance, and the need for targeted interventions to improve sexual health education, hygiene practices, and antimicrobial stewardship within university settings.
8. Conclusion
In conclusion, the study underscores the high prevalence of reproductive tract infections among female students at Niger Delta University, with a clear dominance of fungal pathogens, particularly Candida species, over bacterial infections like Escherichia coli. The findings suggest that the age group of 22-25 years is particularly vulnerable, possibly due to factors such as increased sexual activity and the communal living environment in hostels. The study also highlights the concerning levels of antimicrobial resistance, particularly to commonly used antibiotics, which may complicate the treatment of pelvic inflammatory disease and other reproductive tract infections. These results emphasize the urgent need for improved health education, better hygiene practices, and stricter antimicrobial stewardship to curb the rising incidence of these infections. Addressing these issues at both institutional and policy levels will be crucial in mitigating the long-term health risks, including infertility, associated with untreated reproductive tract infections in this population.
9. Recommendations
Based on the findings of this study, the following recommendations are made:
Short-Term:
1. Immediate Health Education Campaigns: Conduct targeted awareness programs within university hostels to educate female students about the importance of proper hygiene practices, safe sexual behaviors, and the risks associated with reproductive tract infections (RTIs) like pelvic inflammatory disease (PID). These campaigns should be reinforced through posters, seminars, and peer group discussions.
2. Regular Screening and Early Detection: Implement routine screening for RTIs, particularly for bacterial and fungal infections, as part of the university’s health services. This could help in early detection and prompt treatment, reducing the likelihood of severe complications such as PID.
3. Antibiotic Stewardship: Introduce antibiotic stewardship programs in university health centers to monitor and regulate the use of antibiotics. Educating students on proper antibiotic use, emphasizing adherence to prescribed treatments, and discouraging self-medication will help reduce resistance patterns.
Mid-Term:
1. Improved Access to Healthcare Services: Strengthen healthcare services within the university, ensuring access to affordable treatment for reproductive health issues. This includes making laboratory services more accessible, with faster turnaround times for test results, to improve the management of RTIs.
2. Strengthening Hostel Hygiene: Implement regular hygiene inspections and the provision of adequate sanitation facilities in hostels to minimize the risk of infection transmission. This should include access to clean water, sanitary disposal of waste, and proper menstrual hygiene facilities.
3. Development of an RTI Management Protocol: Establish a comprehensive management protocol for RTIs, focusing on early intervention and treatment guidelines tailored to the specific needs of the student population. This would include treatment regimens, follow-up care, and counseling services.
Long-Term:
1. Integration of Sexual and Reproductive Health Education into the Curriculum: Advocate for the integration of sexual and reproductive health education into the university curriculum. This would ensure that students are well-informed about sexual health, hygiene, and the prevention of infections, which will have long-lasting benefits for future generations of students.
2. Research and Surveillance Programs: Develop long-term research and surveillance programs to track the prevalence of RTIs and antimicrobial resistance patterns. This data can guide future interventions, policies, and resource allocation to better address reproductive health challenges.
3. National Policy Advocacy on RTI Management and Antimicrobial Resistance: Collaborate with national health bodies to advocate for the inclusion of RTI management and antimicrobial resistance prevention as a key focus area in national health policies. This can lead to the development of standardized national guidelines for the prevention, diagnosis, and treatment of RTIs among young women in higher education institutions across the country.
1. Health Significance
The health significance of this study lies in its comprehensive assessment of the prevalence and microbial patterns associated with reproductive tract infections (RTIs), particularly pelvic inflammatory disease (PID), among female university students in Niger Delta University. The findings highlight a concerning prevalence of fungal infections, especially Candida species, alongside bacterial pathogens like
Escherichia coli, which can lead to severe reproductive health complications, including infertility, if left untreated. The high rates of antimicrobial resistance observed in commonly prescribed antibiotics underscore the urgency for improved infection control measures and more effective treatment protocols. Furthermore, the study’s identification of key risk factors, including poor hygiene practices and limited access to healthcare in university hostels, emphasizes the need for targeted health interventions. By addressing these issues, the study contributes to the broader effort of improving sexual and reproductive health, reducing the burden of RTIs, and preventing long-term health complications among young women in Nigeria. Thus, graphically it is represented (
Figure 5 below) as:
Data Availability
All data will be made available to interested individuals upon request from the corresponding author.
Transparency
The corresponding author (Morufu Olalekan Raimi) affirms that this manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned (and, if relevant, registered) have been explained.
Funding Statement
The current study did not receive financial support from funding bodies or sponsors.
Conflicts of Interest
The authors declare no conflict of interest. All authors have read and approved the final version of the manuscript. The corresponding author, Morufu Olalekan Raimi, had full access to all of the data in this study and takes complete responsibility for the integrity of the data and the accuracy of the data analysis.
References
- Abid, I., Nawab, T., & Najam Khalique. (2024). Reproductive tract infections among married women and its association with obstetric factors. Indian Journal of Public Health Research & Development, 15(4), 137–144. [CrossRef]
- Adebayo, A. A., & Olatunji, O. O. (2022). Reproductive tract infections and pelvic inflammatory disease among young women in sub-Saharan Africa. African Journal of Reproductive Health, 26(3), 45–56.
- Adelekan, B., Kareem, Y. O., Abubakar, Z., Bungudu, K., Aderemi, A., Goldson, E., Mueller, U., Yaya, S., & Fatusi, A. (2022). Female genital mutilation and sexual behaviour by marital status among a nationally representative sample of Nigerian women. Reproductive Health, 19(1). [CrossRef]
- Adefuye, B. O., Adeyemi, O. A., & Bello, O. O. (2021). Prevalence and risk factors of pelvic inflammatory disease among female undergraduates in Nigerian universities. Journal of Public Health in Africa, 12(2), 112–119.
- Afolayan, A. J., Ojo, D. A., & Adekunle, O. M. (2021). Sexual behaviour and reproductive health challenges among female university students in Nigeria. Nigerian Journal of Clinical Practice, 24(6), 841–847.
- Akinyemi, J. O., Adedini, S. A., & Fatusi, A. O. (2020). Risk factors for reproductive tract infections among young women in Nigeria. BMC Women’s Health, 20, Article 112.
- Akinyemi, J. O., Adedini, S. A., & Fatusi, A. O. (2022). Pelvic inflammatory disease and infertility risk among reproductive-age women in Nigeria. African Journal of Reproductive Health, 26(1), 92–104.
- Alexander, K. A., Perrin, N. A., Butz, A., Perin, J., & Trent, M. (2018). Reproductive coercion and childbearing motivations among adolescent and young adult women recently diagnosed with pelvic inflammatory disease. Journal of Adolescent Health, 62(2), S82–S83. [CrossRef]
- Alkhalili, M., Al-Hmaid, Y., Kheirallah, K., & Mehaisen, L. (2024). Assessment of knowledge of sexual reproductive health among female university students in Jordan. Cureus. [CrossRef]
- Brunham, R. C., Gottlieb, S. L., & Paavonen, J. (2021). Pelvic inflammatory disease. New England Journal of Medicine, 384(21), 2039–2048.
- Cheesbrough, M. (2020). District laboratory practice in tropical countries (3rd ed.). Cambridge University Press.
- Christopher Ononiwu Elemuwa, Teddy Charles Adias, Morufu Olalekan Raimi, Ndu Ijeoma Festa, & Uchenna Geraldine Elemuwa. (2025). Microbial forensics in bioterrorism and violent crimes: A revolutionary approach to criminal investigations [Oral presentation]. 10th International Conference on Health, Medicine and Life Sciences (MEDLIFE2025).
- Christopher Ononiwu Elemuwa, Teddy Charles Adias, Uchenna Geraldine Elemuwa, & Morufu Olalekan Raimi. (2025). Biothreats without borders: Integrating AI and microbial forensics to combat emerging pathogens in Africa. Paper presented at the 2nd Annual Conference of the Faculty of Medical Laboratory Science, Federal University Otuoke, Bayelsa State, Nigeria.
- Clinical and Laboratory Standards Institute. (2023). Performance standards for antimicrobial susceptibility testing (33rd ed.). CLSI supplement M100.
- Doley, P., Yadav, G., Muralidhar, S., & Gupta, M. (2023). A community-based study on the prevalence of reproductive tract infections/sexually transmitted infections and their associated factors among married women of reproductive age in Delhi. CHRISMED Journal of Health and Research, 10(3), 224–229. [CrossRef]
- Federal Ministry of Health. (2021). National guidelines for the management of sexually transmitted infections. FMoH.
- G. Yakupova, & A. Turdieva. (2023). The inquiry into the causes of pelvic inflammatory diseases in early reproductive age women. Bulletin of Science and Practice, 7, 194–199. [CrossRef]
- Ghosh, S., & Dessa, D. (2018). Pelvic inflammatory disease: How frequent it is among the women presenting with low back pain. Annals of International Medical and Dental Research, 4(4). [CrossRef]
- Gift, R. A., & Olalekan, R. M. (2020). Access to electricity and water in Nigeria: A panacea to slow the spread of Covid-19. Open Access Journal of Science, 4(2), 34. [CrossRef]
- Gift, R. A., Olalekan, R. M., Owobi, O. E., Oluwakemi, R. M., Anu, B., & Funmilayo, A. A. (2020). Nigerians crying for availability of electricity and water: A key driver to life coping measures for deepening stay at home inclusion to slow covid-19 spread. Open Access Journal of Science, 4(3), 69–80. [CrossRef]
- Gottlieb, S. L., Low, N., Newman, L. M., Bolan, G., Kamb, M., & Broutet, N. (2020). Toward global prevention of sexually transmitted infections. The Lancet, 395(10236), 172–184.
- Gupta, V., & Dhiman, K. (2023). Guardians of wellness: Unraveling awareness on risk factors and preventive strategies for reproductive tract infections among women in District Mandi, Himachal Pradesh. IAR Journal of Clinical Research, 03(02), 1–4. [CrossRef]
- Haggerty, C. L., & Taylor, B. D. (2020). Mycoplasma genitalium and pelvic inflammatory disease. Current Opinion in Infectious Diseases, 33(1), 56–63.
- Hunt, S., & Vollenhoven, B. (2023). Pelvic inflammatory disease and infertility. Australian Journal of General Practice, 52(4), 215–218. [CrossRef]
- Ibrahim, M. T., Sadiq, A. A., & Mohammed, Y. (2020). Pattern of pelvic inflammatory disease among women attending gynecological clinics in Northern Nigeria. Annals of African Medicine, 19(2), 88–94.
- Jaleh, N., Renu, R., Anuradha, V., & Chopra, A. (2008). O7 A study of prevalence and risk factors of reproductive tract infections among women in reproductive ages suffering from rheumatoid arthritis. Indian Journal of Rheumatology, 3(3), S19. [CrossRef]
- Johnsky, L., Patel, N., Doll, B., Samaniego, J., & Anderson, S. (2026). Pelvic floor dysfunction in inflammatory bowel disease: A single-center analysis of prevalence and associated risk factors. Inflammatory Bowel Diseases, 32(Supplement_1). [CrossRef]
- Katawa, G., Nguepou Tchopba, C., Ritter, M., Silva, M. da, H Ameyapoh, A., Arndts, K., Ataba, E., Edlom Tchadié, P., M Amessoudji, O., Hoerauf, A., D Karou, S., E Layland, L., & Ameyapoh, Y. (2021). Female reproductive tract health: Prevalence and risk factors associated with infections in Lomé. (Female reproductive tract infections in Lomé). Clinical Research and Trials, 7(2). [CrossRef]
- Kreisel, K., Torrone, E., Bernstein, K., Hong, J., & Gorwitz, R. (2017). Prevalence of pelvic inflammatory disease in sexually experienced women of reproductive age - United States, 2013-2014. MMWR. Morbidity and Mortality Weekly Report, 66(3), 80–83. [CrossRef]
- Laizu, I. A., Banu, L. A., & Fayez, F. A. (2021). Clinical findings of pelvic inflammatory diseases among women in reproductive age. Journal of National Institute of Neurosciences Bangladesh, 7(1), 52–55. [CrossRef]
- Maharlouei, N., Barooti, E., Sharif, F., Hosseini, H., & Lankarani, K. B. (2013). Prevalence and risk factors of reproductive tract infections among a defined population of Iranian women. Sexual Health, 10(4), 311. [CrossRef]
- Mansy, A., Bedewy, E., & Ali, M. (2023). Association between pelvic endometriosis and inflammatory bowel disease in females in reproductive age. ALEXMED EPosters, 5(3), 47–48. [CrossRef]
- Maritim, V. C., Wanyoro, A., Nyamari, J., Maingi, J., Aballa, L., & Kimani, E. (2021). Factors associated with health seeking behavior for reproductive tract infections among young street females in Eldoret, Kenya. Kabarak Journal of Research & Innovation, 11(2), 147–163. [CrossRef]
- Mehnaz Shivangi Kumari, S. (2023). Intimate partner violence and symptoms of reproductive tract infections among married women in Aligarh. International Journal of Science and Research (IJSR), 12(6), 1136–1140. [CrossRef]
- Mitchell, C., & Prabhu, M. (2013). Pelvic inflammatory disease. Infectious Disease Clinics of North America, 27(4), 793–809. [CrossRef]
- Morufu, O. R., Aziba-anyam, G. R., & Teddy, C. A. (2021). ‘Silent pandemic’: Evidence-based environmental and public health practices to respond to the Covid-19 crisis. In IntechOpen. [CrossRef]
- Ndu Ijeoma Festa, Christopher Ononiwu Elemuwa, Teddy Charles Adias, Uchenna Geraldine Elemuwa, & Morufu Olalekan Raimi. (2025). Invisible enemies: Strengthening Nigeria’s bioterrorism defense with microbial forensics. Paper presented at the 2nd Annual Conference of the Faculty of Medical Laboratory Science, Federal University Otuoke, Bayelsa State, Nigeria.
- Nwadioha, S. I., Nwokedi, E. O., & Kashibu, E. (2020). Pelvic inflammatory disease and infertility in Nigeria: A review. Nigerian Medical Journal, 61(4), 167–173.
- Nwankwo, E. O., Ahmed, A., & Onwuezobe, I. A. (2021). Antimicrobial resistance patterns among urogenital pathogens in Nigeria. Journal of Infection in Developing Countries, 15(4), 512–520.
- Odimegwu, C., & Somefun, O. D. (2017). Ethnicity, gender and risky sexual behaviour among Nigerian youth: An alternative explanation. Reproductive Health, 14(1). [CrossRef]
- Ojima, Y., Kanda, M., & Ohara, N. (2016). Fallopian tube carcinoma complicated with acute pelvic inflammatory disease: A case report. Journal of Reproductive Immunology, 118, 124–125. [CrossRef]
- Okafor, C. I., & Nwankwo, K. C. (2023). Reproductive tract infections and health-seeking behaviour among female undergraduates in Southeast Nigeria. African Health Sciences, 23(1), 355–364.
- Okeke, I. N., Aboderin, O. A., & Byarugaba, D. K. (2022). Growing problem of multidrug-resistant bacteria in Africa. Clinical Infectious Diseases, 75(1), S68–S75.
- Okorie, C. O., Nwafor, C. C., & Eze, U. A. (2021). Clinical and microbiological profile of pelvic inflammatory disease in a tertiary hospital in Nigeria. Nigerian Journal of Medicine, 30(2), 162–169.
- Olalekan, R. M., Adedoyin, O. O., Ayibatonbira, A., & et al. (2019). “Digging deeper” evidence on water crisis and its solution in Nigeria for Bayelsa state: A study of current scenario. International Journal of Hydrology, 3(4), 244–257. [CrossRef]
- Olalekan, R. M., Dodeye, E. O., Efegbere, H. A., Odipe, O. E., Deinkuro, N. S., Babatunde, A., & Ochayi, E. O. (2020). Leaving no one behind? Drinking-water challenge on the rise in Niger Delta region of Nigeria: A review. Merit Research Journal of Environmental Science and Toxicology, 6(1), 31–49. [CrossRef]
- Olalekan, R. M., Vivien, O. T., Adedoyin, O. O., & et al. (2018). The sources of water supply, sanitation facilities and hygiene practices in oil producing communities in central senatorial district of Bayelsa state, Nigeria. MOJ Public Health, 7(6), 337–345. [CrossRef]
- Olowe, O. A., Ojurongbe, O., & Adefioye, O. A. (2022). Vaginal infections among women in Southwestern Nigeria: Implications for reproductive health. Pan African Medical Journal, 42, Article 89.
- Omotoso, A. J., Ojile, M. O., & Morufu, O. R. (2025). Uncovering the hidden burden: Water, sanitation, and hygiene (WaSH) gaps and their impact on health and learning outcomes in secondary schools, Ede South LGA, Nigeria. Authorea. [CrossRef]
- Onuh, A. C., Udobi, S. I., & Aronu, M. E. (2022). Intrauterine adhesions, peritubal adhesions and tubal occlusion on hysterosalpingography. Annals of Clinical and Biomedical Research, 3(2). [CrossRef]
- Pandey, B. (2015). Epidemiology and risk factors of pelvic inflammatory disease. Medical Journal of Shree Birendra Hospital, 13(1), 4–8. [CrossRef]
- Payne, D. (2001). Microscopes and health-laboratory aids in the developing countries. Transactions of the Royal Society of Tropical Medicine and Hygiene, 95(2), 202. [CrossRef]
- Perhar, R., Rawat, S., & Pandey, R. (2020). Detection of chlamydial antibodies in women with pelvic inflammatory disease and infertility. The New Indian Journal of OBGYN, 6(2), 101–105. [CrossRef]
- Raimi, M. O. (2024). Structured PREreview of “Development and Content Validity of the Handwashing Index (HWI).” Zenodo. [CrossRef]
- Raimi, M. O., & Ochayi, E. O. (2017). Assessment of the rate of sexually transmitted diseases in Kubwa F.C.T. Abuja, Nigeria. Science Journal of Public Health, 5(5), 365–376. [CrossRef]
- Raimi, M. O., & Raimi, A. G. (2020). The toughest triage in decision impacts: Rethinking scientific evidence for environmental and human health action in the times of concomitant global crises. CPQ Medicine, 11(1), 1–5.
- Raimi, M. O., Abdulraheem, A. F., Major, I., Odipe, O. E., Isa, H. M., & Onyeche, C. (2019a). The sources of water supply, sanitation facilities and hygiene practices in an island community: Amassoma, Bayelsa State, Nigeria. Public Health Open Access, 3(1), Article 000134. [CrossRef]
- Raimi, M. O., Mcfubara, K. G., Abisoye, O. S., Ifeanyichukwu, E. C., Henry, S. O., & Raimi, G. A. (2021). Responding to the call through translating science into impact: Building an evidence-based approaches to effectively curb public health emergencies [COVID-19 crisis]. Global Journal of Epidemiology and Infectious Disease, 1(1). [CrossRef]
- Raimi, M. O., Moses, T., Okoyen, E., Sawyerr, H. O., Joseph, B. O., & Oyinlola, B. O. (2020). A beacon for dark times: Rethinking scientific evidence for environmental and public health action in the coronavirus diseases 2019 era. Medical and Research Microbiology, 1(3).
- Raimi, M. O., Omidiji, A. O., Abdulraheem, A. F., & Ochayi, E. O. (2018). A survey of hand washing behaviour and awareness among health care workers in health care facilities in Kubwa District of Bwari Area Council, F.C.T. Abuja, Nigeria. Annals of Ecology and Environmental Science, 2(2), 1–18.
- Raimi, M. O., Omidiji, A. O., Adeolu, T. A., Odipe, O. E., & Babatunde, A. (2019b). An analysis of Bayelsa State water challenges on the rise and its possible solutions. Acta Scientific Agriculture, 3(8), 110–125. [CrossRef]
- Semwal, S., Kumar, A., & Chauhan, V. (2022). A study on knowledge and risk factors related to pelvic inflammatory disease among women in selected rural community area of Dehradun, Uttarakhand. International Journal of Science and Research (IJSR), 11(7), 609–612. [CrossRef]
- Shalanyuy, L. H., Marion, T., Kobuh, N. D., Randze, L. N., Mengnjo, T. L., & Chongsi, W. E. (2025). Prevalence, risk factors and complication of pelvic inflammatory disease among women of reproductive age at the EHFA Foundation Teaching Medical Centre in Foumbot. OALib, 12(10), 1–15. [CrossRef]
- Sharma, H., Tal, R., Clark, N., & Segars, J. (2014). Microbiota and pelvic inflammatory disease. Seminars in Reproductive Medicine, 32(01), 43–49. [CrossRef]
- Sharma, P., & Gautam, K. (2025). Cross sectional study to assess the awareness about reproductive tract infections (RTIs) among women of reproductive age group in Himachal Pradesh. Himalayan Journal of Medicine and Surgery, 5(1), 1–4. [CrossRef]
- Suranga, M. S., Karunathilake, K., & De Silva, W. I. (2025). Legal framework and sexual and reproductive health challenges faced by female sex workers in Sri Lanka: A narrative literature review. ReproSex: International Journal on Sexual and Reproductive Health, 1(1), 58–70. [CrossRef]
- Suss, A. L., Homel, P., Hammerschlag, M., & Bromberg, K. (2000). Risk factors for pelvic inflammatory disease in inner-city adolescents. Sexually Transmitted Diseases, 27(5), 289–291. [CrossRef]
- Teddy Charles Adias, Uchenna Geraldine Elemuwa, Christopher Ononiwu Elemuwa, & Morufu Olalekan Raimi. (2025). Tracking killers we can’t see: The role of microbial forensics in crime scene reconstruction in Sub-Saharan Africa. Paper presented at the 2nd Annual Conference of the Faculty of Medical Laboratory Science, Federal University Otuoke, Bayelsa State, Nigeria.
- Torrone, E. A., Morrison, C. S., & Kaufman, J. S. (2021). Risk factors for pelvic inflammatory disease. Sexually Transmitted Diseases, 48(6), 394–400.
- Tuebi, M., Franco, A., Raimi, M. O., Chidubem, O., & Sampou, W. D. (2021). The quality and acceptance of family planning services in improving Bayelsa State: Lessons learnt from a review of global family planning programs. Greener Journal of Medical Sciences, 11(2), 212–225.
- Tukur, J., Mohammed, Z., & Lawan, U. M. (2022). Pelvic inflammatory disease: Clinical patterns and management outcomes in a Nigerian teaching hospital. West African Journal of Medicine, 39(2), 129–136.
- Umar, A. S., Sadiq, M. A., & Garba, B. I. (2021). Gynecological morbidity and infertility patterns among women in Northern Nigeria. Journal of Gynecology and Obstetrics, 9(3), 63–69.
- Vanthuyne, A., & Pittrof, R. (2016). Diagnosis and treatment of pelvic inflammatory disease. Prescriber, 27(10), 47–50. [CrossRef]
- World Health Organization. (2021). Sexually transmitted infections (STIs): Fact sheet. WHO.
- World Health Organization. (2022). Global health sector strategy on sexually transmitted infections 2022–2030. WHO.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).