1. Introduction
Oral cancer is one of the most common malignancies among head and neck squamous cell carcinomas (HNSCCs). In 2022, approximately 389,000 new cases and 188,000 deaths were estimated worldwide [
1,
2]. More than 90% of oral cancers are diagnosed as oral squamous cell carcinoma (OSCC) [
3,
4]. Although multimodal treatment strategies combining surgery, radiotherapy, and chemotherapy have improved disease management, the prognosis of patients with OSCC remains unsatisfactory [
5,
6]. Given the anatomical complexity and functional importance of the oral cavity, treatment often leads to impairment of critical oral functions such as speech and swallowing [
7,
8]. Consequently, there is an urgent unmet clinical need for novel therapeutic strategies that achieve durable tumor control while minimizing treatment-related complications and preserving oral function.
Immune checkpoint blockade has demonstrated clinical efficacy across a wide range of malignancies [
9]. In HNSCC, including OSCC, ICIs have improved overall survival in patients with recurrent or metastatic disease compared with conventional systemic therapies [
10,
11]. More recently, perioperative pembrolizumab, administered in the neoadjuvant and adjuvant settings in combination with standard therapy, has expanded the role of ICIs to resectable, locally advanced HNSCC [
12,
13]. Among other immunotherapeutic approaches, chimeric antigen receptor (CAR) T-cell therapy has been actively explored in HNSCC, as in other malignancies. However, the clinical application of CAR-T therapy in solid tumors remains limited due to multiple barriers, including antigen heterogeneity, insufficient trafficking and persistence within tumor sites, and functional impairment caused by the immunosuppressive tumor microenvironment (TME) [
14,
15]. Therefore, for CAR-T therapy to be effective in OSCC, it is essential to identify target molecules that are frequently expressed and localized on the tumor cell surface, and to develop delivery strategies that maximize intratumoral accessibility [
16,
17].
Ephrin type-B receptor 4 (EPHB4) is a receptor tyrosine kinase belonging to the Eph receptor family and has been implicated in tumor progression, invasion, and angiogenesis [
18,
19]. In OSCC, EPHB4 is frequently overexpressed, and immunohistochemical analyses have reported a high positivity rate in all examined cases (20/20) [
20]. Such a high prevalence of expression and membrane localization on tumor cells support EPHB4 as a promising target for antibody- or CAR-mediated recognition. Moreover, if its expression in normal tissues is limited, EPHB4 may provide a favorable therapeutic window by enhancing tumor selectivity while reducing on-target/off-tumor toxicity. Indeed, EphB4 staining has been observed in basal layer cells of the squamous epithelium of the pharynx and esophagus, as well as in mucosal epithelial cells of the stomach, gallbladder, and large intestine; however, the staining intensity is clearly weaker than that in cancerous areas [
21]. Collectively, these observations suggest that EPHB4 expression in normal tissues is relatively limited.
Yagyu and colleagues developed EPHB4-targeted CAR-T cells using ephrin-B2 (EFNB2), the physiological ligand of EPHB4. Using this Yagyu group–developed construct/platform, Ito and colleagues reported promising antitumor efficacy in OSCC cell lines and patient-derived xenograft (PDX) models [
22]. However, their preclinical evaluation was primarily based on subcutaneous tumor models, which may not fully capture the anatomical constraints of the oral cavity, including the unique vascular and lymphatic architecture, the local tumor microenvironment (TME), and factors that determine CAR-T cell accessibility depending on the route of administration.
In the present study, we established a new orthotopic tongue PDX model using the patient-derived PDX tumor OC-15 generated in our laboratory and evaluated the therapeutic efficacy of EPHB4-targeted CAR-T cells (131CAR) under conditions that more closely mimic the clinical setting. This study is distinguished by its use of an orthotopic tongue model to assess not only efficacy but also delivery strategy. Our findings may provide preclinical evidence supporting the development of function-preserving therapeutic approaches for OSCC.
2. Results
This section may be divided by subheadings. It should provide a concise and precise description of the experimental results, their interpretation, as well as the experimental conclusions that can be drawn.
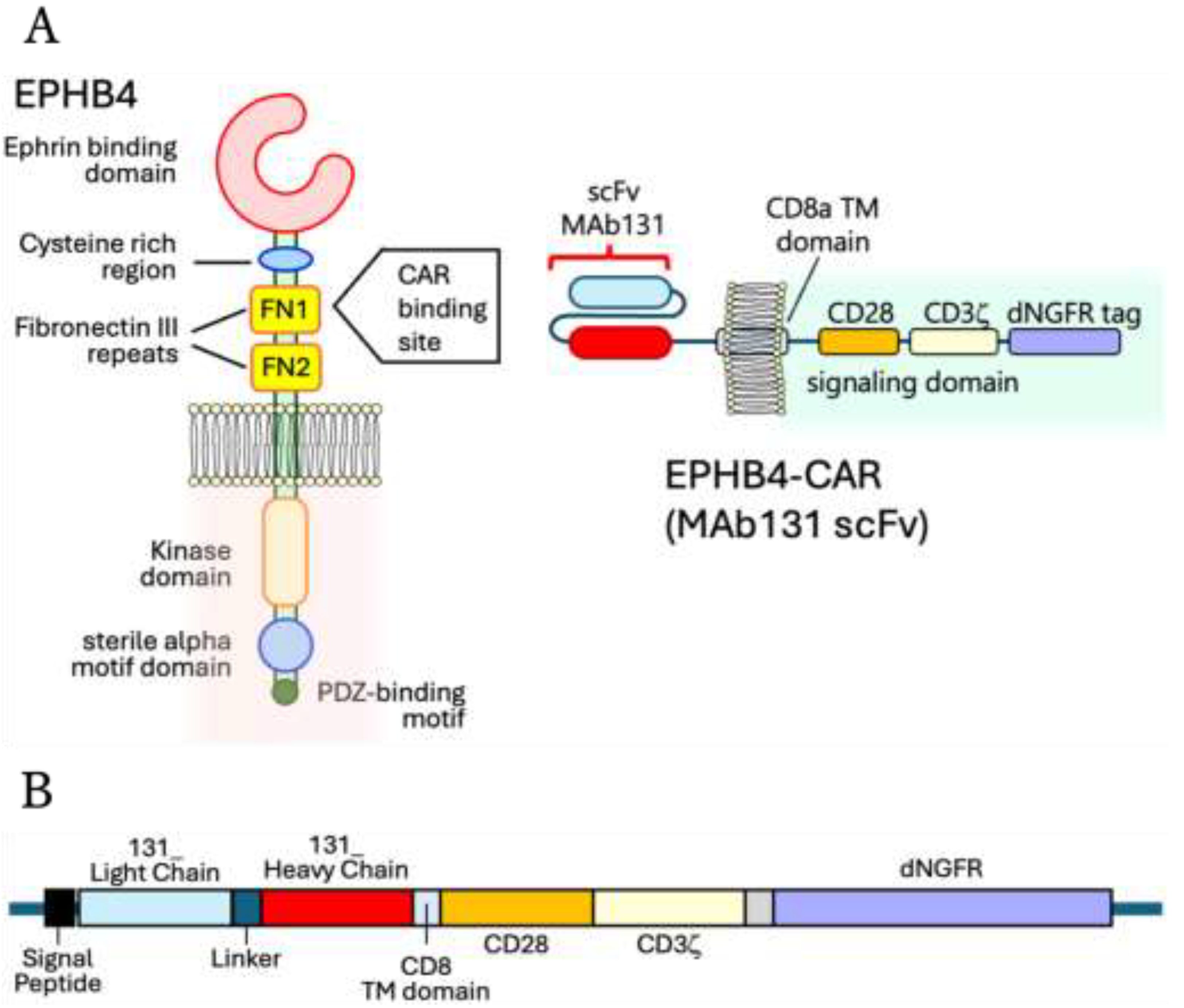
2.1. Molecular Structure of EPHB4 and Generation of Anti-EPHB4 CAR T Cells
EPHB4 contains an ephrin-binding domain, a cysteine-rich region, and two fibronectin type III–like domains (FN1 and FN2) in its extracellular region (
Figure 1A, left panel) [
23]. The anti-EPHB4 CAR used in this study was a second-generation construct incorporating an scFv derived from MAb131, which has been reported to bind the FN1 domain of EPHB4 (
Figure 1A, right panel;
Figure 1B) [
24]. The CAR T cells were generated by Optium Biotechnologies Inc. through our collaborative research. Hereafter, this anti-EPHB4 CAR T-cell product is referred to as “131CAR” throughout the study.
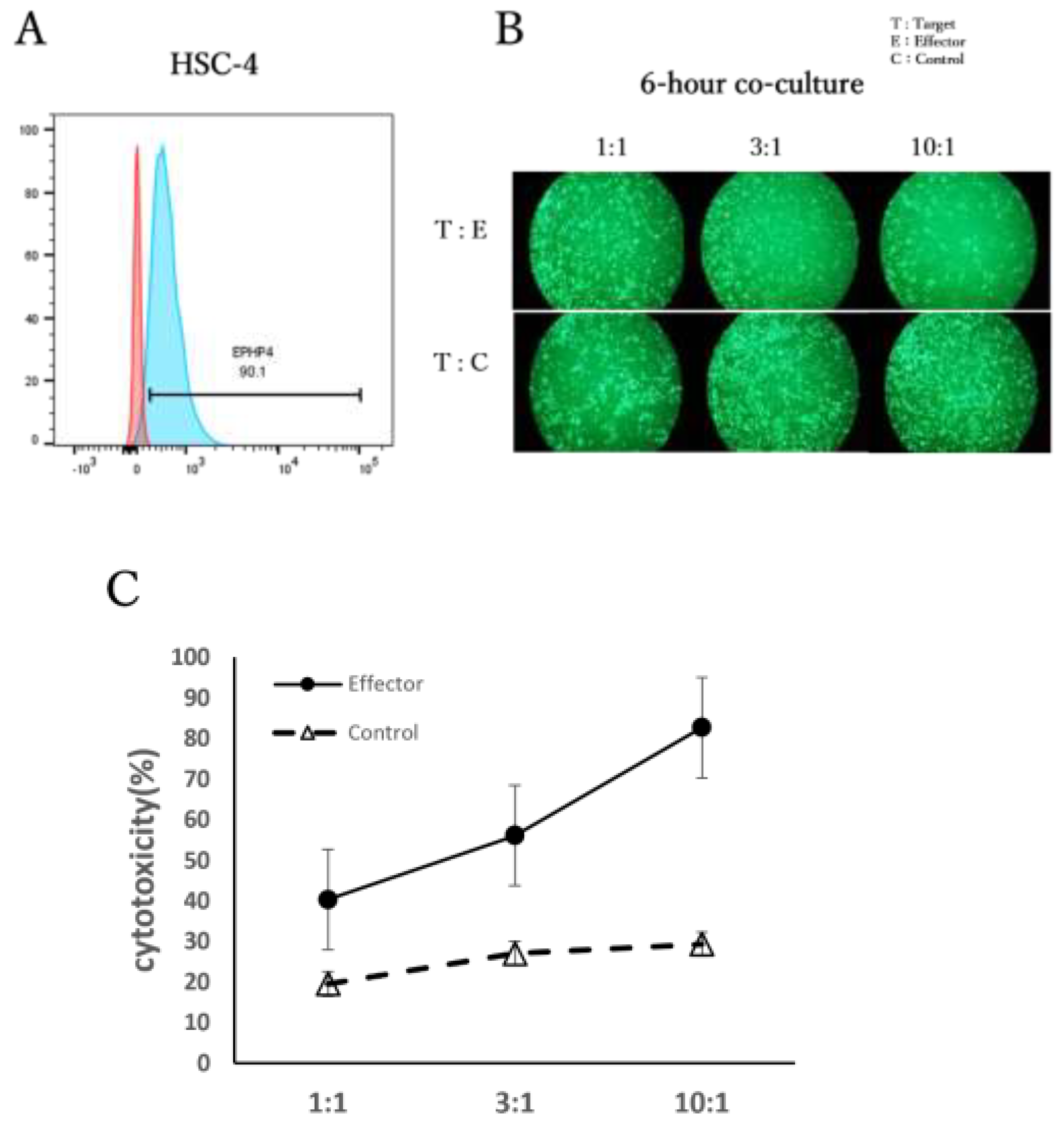
2.2. EPHB4 Expression in HSC-4 Cells and In Vitro Cytotoxic Activity of 131CAR T Cells
EPHB4 expression in HSC-4 cells and in vitro cytotoxic activity of 131CAR T cells. First, surface expression of EPHB4 on HSC-4 cells was evaluated by flow cytometry. Approximately 90% of HSC-4 cells were EPHB4-positive (
Figure 2A). Next, we assessed the cytotoxic activity of 131CAR T cells against HSC-4 target cells, using T cells expressing Δ nerve growth factor receptor (dNGFR) alone as a control. Target cells were co-cultured with effector cells for 6 h at the indicated effector-to-target (E:T) ratios. Fluorescence intensity of labeled target cells was measured before and after co-culture, and specific cytotoxicity was calculated. 131CAR T cells exhibited robust cytotoxicity compared with control T cells across all E:T ratios tested. Cytotoxicity increased in an effector-dose–dependent manner, and at an E:T ratio of 10:1, 131CAR T cells mediated approximately four-fold higher cytotoxicity than control T cells. These results indicate that 131CAR T cells exert potent, antigen-dependent cytotoxic activity against EPHB4-expressing OSCC cells in vitro (
Figure 2B,C).
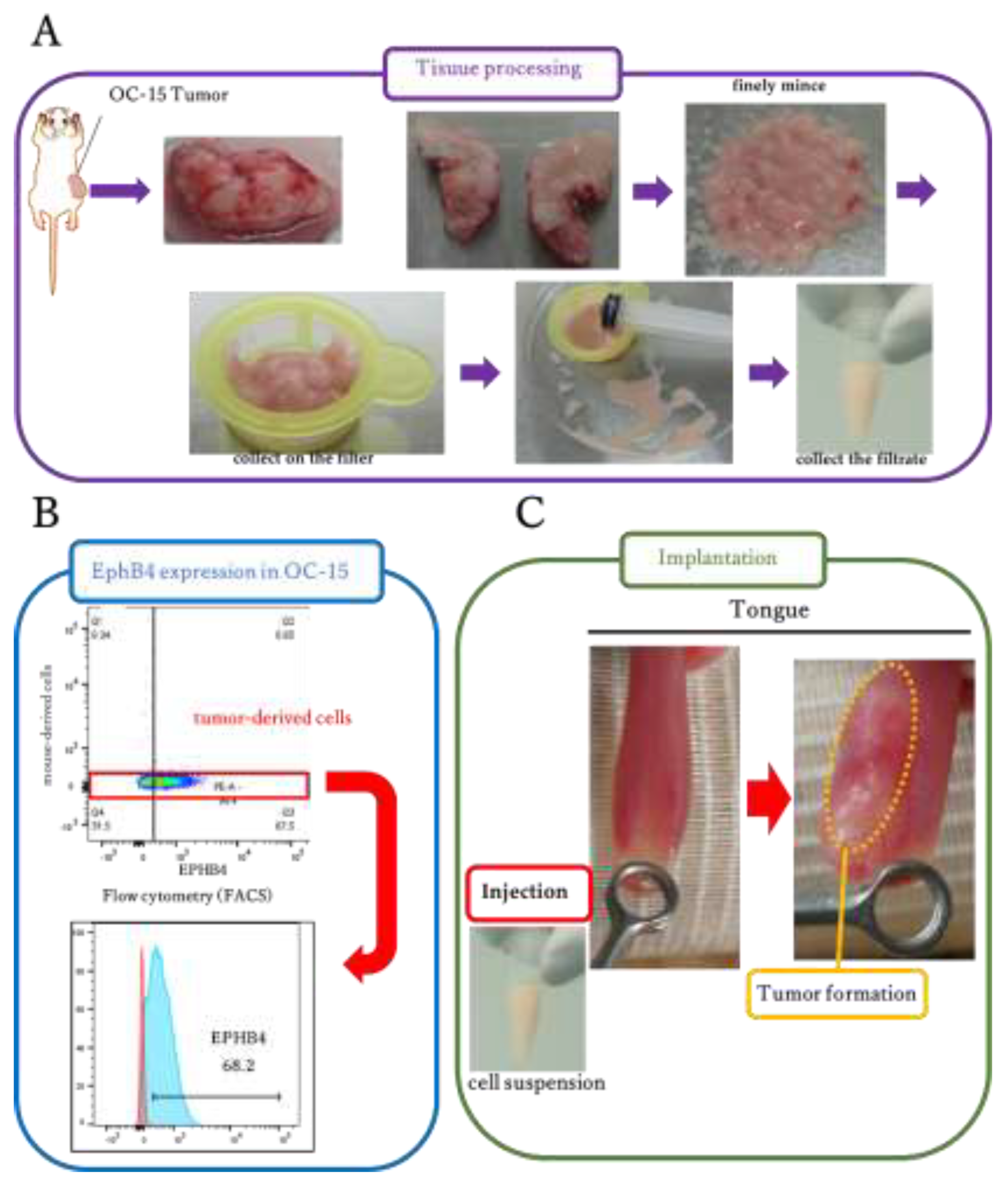
2.3. EPHB4 Expression in Patient-Derived Tongue Cancer Cells and Establishment of an Orthotopic Tongue PDX Model
To evaluate the efficacy of 131CAR in a clinically relevant preclinical setting, we established an orthotopic tongue PDX model using the OC-15 PDX (
Figure 3). Flow cytometry confirmed cell-surface EPHB4 expression in OC-15 tumor–derived cells (
Figure 3B). Because direct implantation of tumor fragments into the mouse tongue is technically challenging, we explored intralingual injection of tumor cell suspensions. Enzymatic dissociation (e.g., collagenase-based dispersion) resulted in a very low engraftment rate (data not shown). In contrast, intralingual injection of a mechanically dissociated tumor cell suspension prepared as shown in
Figure 3A reproducibly achieved tumor engraftment in 100% of mice (
Figure 3C). All subsequent therapeutic experiments were performed using this orthotopic tumor-bearing model.
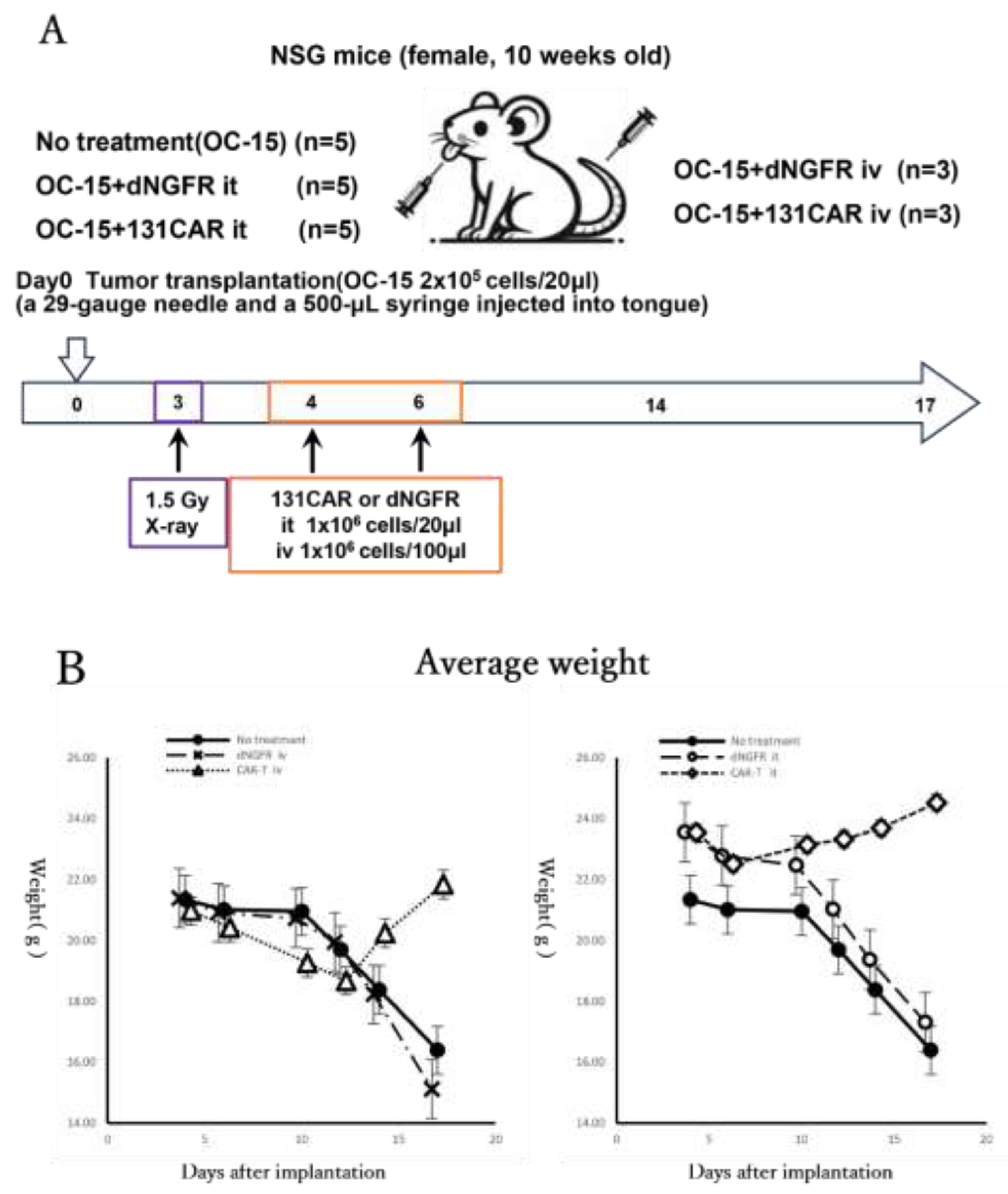
2.4. In Vivo Antitumor Activity of EPHB4-CAR T Cells in an Orthotopic Tongue PDX Model
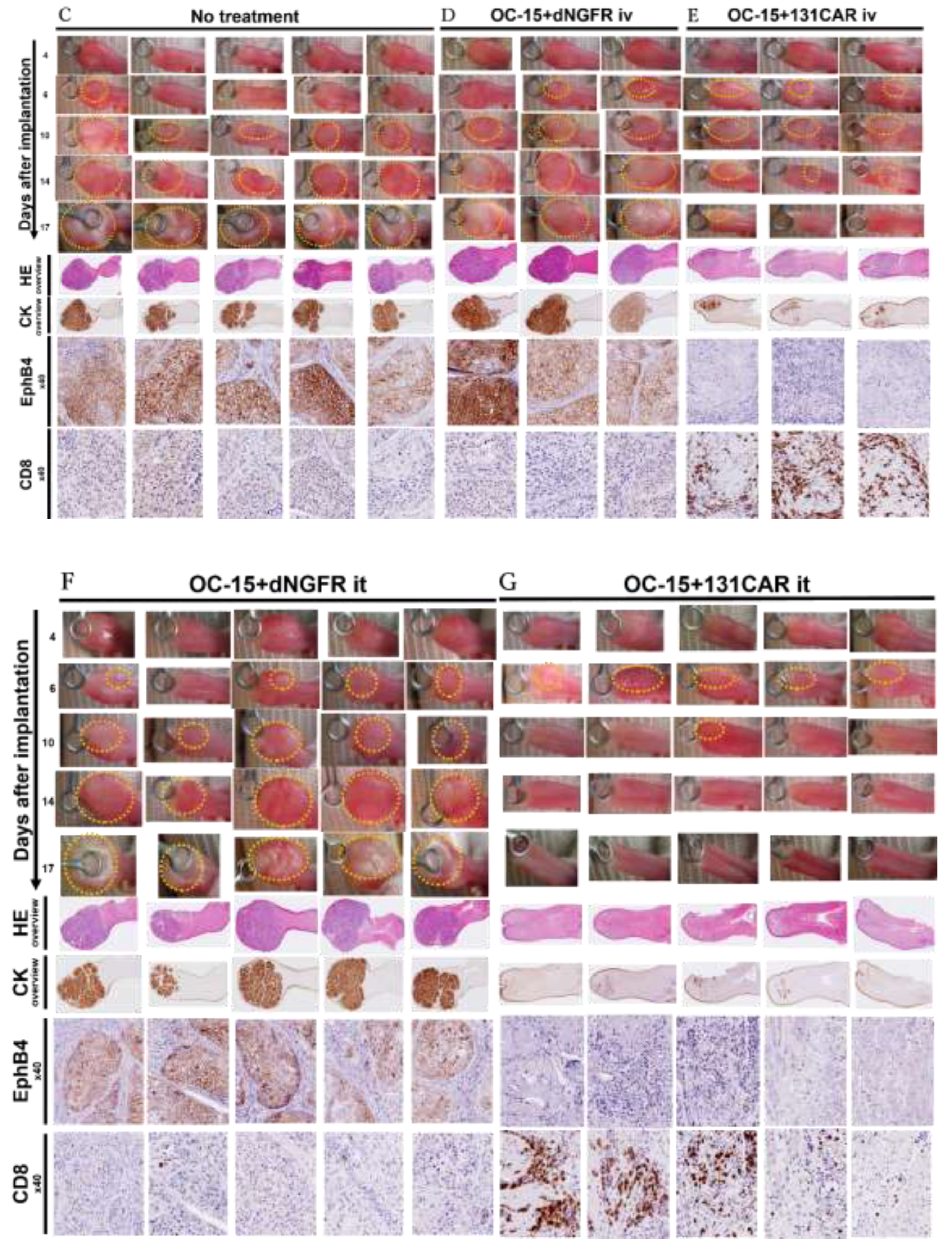
In the orthotopic tongue PDX model, mice received total body irradiation on day 3 after tumor implantation and were treated with dNGFR control T cells or 131CAR T cells on days 4 and 6, administered either intratumorally or intravenously according to the schedule shown in
Figure 4A. On day 17, because some mice in the no-treatment and control groups exhibited approximately 25% body weight loss (
Figure 4B), all animals were euthanized as a humane endpoint. No-treatment mice showed progressive macroscopic enlargement of tongue tumors over time (
Figure 4C). In the intravenous cohort, the control group likewise exhibited increasing tongue tumor growth similar to the no-treatment group (
Figure 4D). In contrast, in the 131CAR-treated group, a macroscopic tongue tumor was observed in only 1 of 3 mice at the predefined endpoint (day 17 after implantation), and no macroscopic tumor was evident in the remaining 2 mice (
Figure 4E). With respect to body weight, the no-treatment and control groups showed a sustained decrease in mean body weight throughout the observation period. By comparison, the 131CAR-treated group exhibited body weight loss until day 12 after implantation, followed by a recovery trend thereafter (
Figure 4B, left panel). In the intratumoral cohort, the control group showed progressive macroscopic tongue tumor growth similar to the no-treatment group (
Figure 4F). In contrast, in the 131CAR-treated group, a macroscopic tongue tumor was observed in only 1 of 5 mice on day 10 after implantation, whereas no macroscopic tumor was detected in the remaining 4 mice. Moreover, after day 14, no macroscopic tongue tumors were detectable in any of the mice in the 131CAR-treated group (
Figure 4G). With respect to body weight, the control group showed a downward trend in mean body weight from day 6 onward, whereas the 131CAR-treated group did not exhibit an apparent decrease in mean body weight after day 6 and instead showed a gradual increase (
Figure 4B, right panel). Intratumoral administration of 131CAR appeared to induce earlier macroscopic tumor regression and earlier body weight recovery than intravenous administration.
2.5. Pathological Assessment by Immunohistochemistry
At the humane endpoint, tongues were harvested from mice in each group and processed into formalin-fixed, paraffin-embedded (FFPE) specimens. Histopathological evaluation of FFPE sections by H&E staining and immunohistochemistry (IHC) showed that hematoxylinophilic tumor areas were expanded in the no-treatment and control groups regardless of the administration route (
Figure 4C,
4D, and
4F; upper low-magnification panels), whereas these tumor areas were markedly reduced in the 131CAR-treated groups (
Figure 4E and
4G; upper low-magnification panels).In addition, EPHB4 immunoreactivity was clearly detectable in the no-treatment and control groups but was diminished in the 131CAR-treated groups (
Figure 4C–G; upper high-magnification panels). In contrast to EPHB4 staining, cytokeratin staining intensity remained largely unchanged, although the extent of cytokeratin-positive areas was reduced in the 131CAR-treated groups (
Figure 4C–G; lower low-magnification panels). Moreover, clusters and infiltration of human CD8-positive (hCD8
+) cells were observed around the tumor regions [defined as cytokeratin-positive areas] in the 131CAR-treated groups, whereas such hCD8
+ cell accumulation was limited in the no-treatment and control groups (
Figure 4D and
4E; lower low-magnification panels).
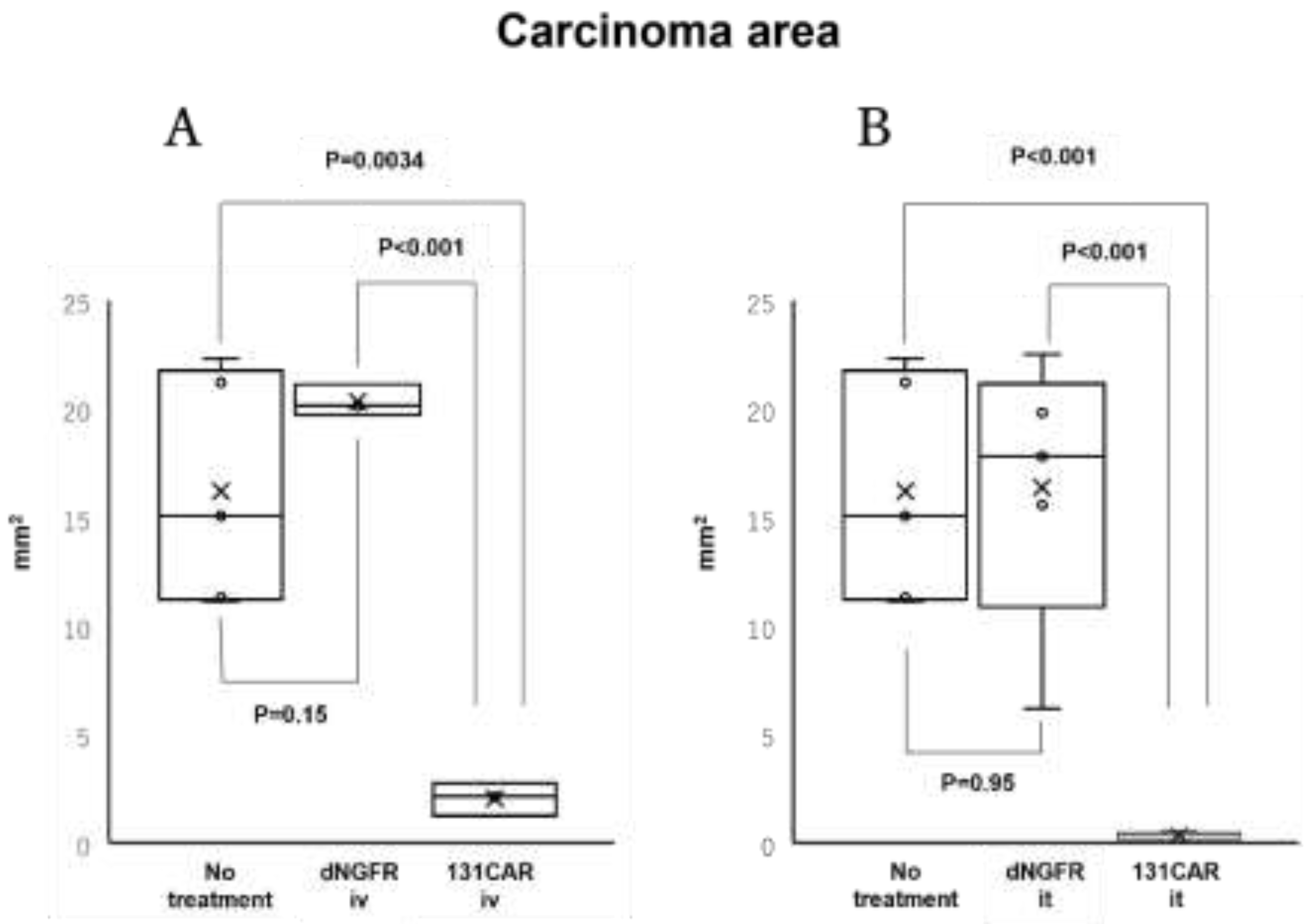
2.6. Digital Pathology–Based Quantification of Tumor Area
Using the acquired virtual slide images, we performed image analysis with Fiji to define cytokeratin-positive regions as tumor areas and quantified the tumor area (mm
2). In the intravenous cohort, tumor area was significantly lower in the 131CAR-treated group than in the no-treatment group (16.2 vs. 2.0 mm
2, p = 0.0034) and the dNGFR control group (20.3 vs. 2.0 mm
2, p < 0.001), whereas no significant difference was observed between the no-treatment and dNGFR control groups (16.2 vs. 20.3 mm
2, p = 0.15). Similarly, in the intratumoral cohort, tumor area was significantly reduced in the 131CAR-treated group compared with the no-treatment group (16.2 vs. 0.3 mm
2, p < 0.001) and the dNGFR control group (16.4 vs. 0.3 mm
2, p < 0.001), with no significant difference between the no-treatment and dNGFR control groups (16.2 vs. 16.4 mm
2, p = 0.95) (
Figure 5). Although no statistically significant difference in tumor area was observed between the intravenous and intratumoral 131CAR-treated groups, the intratumoral group tended to show a smaller tumor area (
Figure 5). Taken together, these findings indicate that 131CAR treatment markedly reduced tumor burden at the tissue level, supporting the gross observations and demonstrating effective elimination of EPHB4-expressing tumor cells.
3. Discussion
In this study, we evaluated the antitumor activity of EPHB4-targeted CAR T cells using both in vitro assays with the tongue-derived OSCC cell line HSC-4 and in vivo experiments in an orthotopic tongue PDX model established from a patient-derived tumor (OC-15). Previous work has reported that EPHB4-directed CAR T-cell approaches suppress tumor growth in OSCC cell line–based models and PDX models (Ito et al. [
20]). Our results extend these findings by demonstrating tumor control in an orthotopic tongue setting, which more closely reflects the clinical microenvironment than subcutaneous models. In addition, whereas therapeutic efficacy after intravenous administration was not observed in the previously reported subcutaneous tumor model, both intratumoral and intravenous delivery induced tumor regression in our orthotopic tongue model. Notably, the CAR construct used by Ito et al. employed EFNB2, the physiological ligand of EPHB4, thereby adopting a binding mode closer to the native receptor–ligand interaction; this design is consistent with an emphasis on mitigating off-tumor recognition. In contrast, the 131CAR used in the present study incorporates an scFv derived from MAb131 and is expected to exhibit stronger binding to EPHB4, which raises a theoretical concern regarding on-target/off-tumor toxicity. However, under our experimental conditions, systemic administration of 131CAR T cells in NSG mice did not result in overt injury to major normal organs, and the post-infusion distribution of 131CAR T cells was not associated with detectable tissue damage (data not shown). Taken together, within the scope of this study, 131CAR exhibited an acceptable in vivo safety profile.
In vitro, under conditions in which HSC-4 cells showed a high frequency of EPHB4 positivity, 131CAR exerted strong cytotoxic activity within a short time frame, and target-cell elimination increased in an E:T ratio–dependent manner (
Figure 2B,C). In contrast, the EPHB4-positive fraction of PDX-derived tumor cells was 68.2%, lower than that of HSC-4 (approximately 90%) (
Figure 3B). This difference is best interpreted as clinically relevant heterogeneity that reflects not only the proportion of antigen-positive cells but also variability in antigen density (expression levels). Moreover, while two-dimensional co-culture facilitates physical contact between effector and target cells, the orthotopic PDX setting introduces three-dimensional architecture and stromal components that restrict cell–cell interactions, such that CAR T-cell access and contact frequency can shape therapeutic efficacy.
In this study, we established an orthotopic tongue PDX model by preparing OC-15 tumors as a cell suspension via mechanical dissociation and filtration and injecting the suspension into the tongues of NSG mice. By omitting an ex vivo culture step, this workflow minimizes culture-associated phenotypic changes and clonal selection (e.g., enrichment of rapidly proliferating subclones and shifts in antigen expression), enabling therapeutic evaluation under conditions that better preserve intratumoral heterogeneity. In addition, the observation that enzymatic dissociation resulted in a low tumor take rate, whereas mechanical dissociation and filtration achieved robust engraftment, is consistent with rapid recovery of viable tumor cells while minimizing perturbation of cell-surface properties. Compared with subcutaneous models, the orthotopic tongue model better reflects site-specific microenvironmental and vascular/lymphatic features, thereby strengthening the relevance of evaluating CAR T-cell behavior and activity in a clinically contextual setting.
In the orthotopic tongue PDX model, gross examination confirmed tumor regression in the 131CAR-treated groups (
Figure 4). Histopathological analyses further showed diminished EPHB4 immunoreactivity after treatment, whereas cytokeratin-positive residual tumor regions remained detectable (
Figure 4), indicating that a fraction of tumor components persisted despite the marked macroscopic response. This pattern can be explained by preferential elimination of EPHB4-high tumor cells with relative persistence of EPHB4-low/negative clones, together with spatially heterogeneous intratumoral engagement at the tissue level even after intratumoral delivery. Accordingly, even when strong antitumor activity is achieved, complete eradication may be difficult with a single-antigen CAR strategy, underscoring the importance of developing CAR T cells that concurrently target multiple tumor-associated antigens (e.g., dual/tandem CAR designs) to improve the depth and uniformity of tumor control.
To quantify residual tumor burden, we performed digital pathology–based analysis of virtual slide images using Fiji/ImageJ, defining cytokeratin-positive regions as tumor areas. Tumor area was significantly reduced in the 131CAR-treated groups compared with the no-treatment and dNGFR control groups regardless of administration route (
Figure 5). Although no statistically significant difference in tumor area was observed between intravenous and intratumoral 131CAR delivery, the intratumoral group showed a numerically smaller tumor area than the intravenous group (i.v. vs. i.t., 2.0 mm
2 vs. 0.3 mm
2) (
Figure 5). This trend is consistent with more efficient local tumor engagement after intratumoral injection. Clinically, the oral cavity is anatomically accessible, and intratumoral administration can be considered in selected settings (e.g., perioperative or image-guided delivery) to achieve high local effector density while limiting systemic exposure. However, intratumoral delivery alone may be insufficient for regional nodal disease or occult disseminated lesions. In contrast, intravenous administration provides broader anatomical coverage, but in solid tumors its efficacy is often shaped by trafficking, penetration, and persistence within the tumor site. Therefore, clinical translation should consider route selection based on disease distribution and rational designs that combine local and systemic approaches in a complementary manner.
Provides broader anatomical coverage, but in solid tumors its efficacy is often shaped by trafficking, penetration, and persistence within the tumor site. Therefore, clinical translation should consider route selection based on disease distribution and rational designs that combine local and systemic approaches in a complementary manner.
4. Materials and Methods
4.1. EPHB4 Expression in the HSC-4 Cell Line
HSC-4 is a human oral squamous cell carcinoma (OSCC) cell line established from a tongue-derived tumor. Cell-surface expression of EPHB4 on HSC-4 cells (TKG 0489, Cell Resource Center for Biomedical Research, Institute of Development, Aging and Cancer, Tohoku University) was evaluated by flow cytometry. Cells were stained with a phycoerythrin (PE)-conjugated anti-EPHB4 monoclonal antibody (clone FAB3038-SP; R&D Systems) for 20 min at 4 °C. After washing with phosphate-buffered saline (PBS) containing 2% fetal bovine serum (FBS), samples were analyzed on a FACSCanto II flow cytometer (BD Biosciences). Data were analyzed using FlowJo software (version 10).
4.2. Preparation of EPHB4-CAR T Cells (131CAR) and Control dNGFR T Cells
EPHB4-targeted CAR T cells (131CAR) and control T cells expressing dNGFR alone were generated by Optium Biotechnologies. The 131CAR is a second-generation CAR incorporating an MAb131-derived scFv and intracellular signaling domains of CD28 and CD3ζ (a schematic of the construct is shown in
Figure 1). As a control, T cells expressing Δ nerve growth factor receptor (dNGFR; truncated NGFR) alone were used. For cell preparation, retroviral expression vectors (pMXs Retroviral Expression Vector; Cell Biolabs) encoding dNGFR-tagged 131CAR or dNGFR alone were constructed. Retroviral supernatants were produced using Plat-A packaging cells (Cell Biolabs) and amplified using PG13 cells (ATCC). Plat-A and PG13 cells were maintained in DMEM supplemented with 10% FBS and 50 μg/mL gentamicin at 37 °C in a humidified atmosphere with 5% CO
2. Human T cells were isolated from healthy donor PBMCs (Cellular Technology Limited) using a Pan T Cell Isolation Kit (Miltenyi Biotec) and activated with T Cell TransAct (Miltenyi Biotec) in the presence of human IL-2 (100 U/mL) for 2 days in AIM-V medium supplemented with 5% FBS. Activated T cells were transduced with retroviral supernatants by centrifugation-assisted transduction, and transduction was performed twice on consecutive days.
4.3. In Vitro Cytotoxicity Assay
The cytotoxic activity of 131CAR against target cells was evaluated using the TeraScan VPC system (Minerva Tech). The assay was performed according to a previously reported protocol [
25]. HSC-4 target cells were labeled with Calcein-AM (Dojindo Laboratories) and co-cultured with effector cells (131CAR or control T cells) at the indicated effector-to-target (E:T) ratios. Fluorescence intensity was measured before and after 6 h of co-culture, and specific cytotoxicity was calculated using the following equation:
where Fsample is the mean fluorescence intensity in the sample wells, Fmin is the mean fluorescence intensity in the minimum-release control wells, and Fmax is the mean fluorescence intensity in the maximum-release control wells.
4.4. Establishment of the Patient-Derived Xenograft (PDX) and Orthotopic Tongue Model
The PDX line (OC-15) was established using a tumor obtained from an OSCC patient who underwent surgical resection at National Cancer Center Hospital East. Tumor fragments were serially maintained by subcutaneous implantation in NOD scid gamma (NSG) mice (thigh region) and were harvested when the subcutaneous tumors reached approximately 2 cm in diameter. Excised tumors were bisected along the largest cross-section, and internal tumor tissue was collected while avoiding the capsule-adjacent area to minimize contamination with murine cells. The collected tissue was mechanically minced using scissors and a scalpel, and then forcibly passed through a 100-µm cell strainer (EASY strainer) to prepare a tumor cell suspension. EPHB4 expression in the resulting cells was assessed by flow cytometry. Murine cells were labeled with an anti-mouse MHC class I (H-2Kᵈ) antibody (clone SF1-1.1.1) and excluded during analysis. The cell suspension was adjusted to 2 × 10^6 cells/20 µL (containing 2.0 × 10^6 viable cells) and injected into the tongues of anesthetized NSG mice using a 29G syringe. Tumor formation in the tongue was subsequently monitored (
Figure 3).
4.5. EPHB4-CAR T-Cell Treatment Model
All animal experiments were approved by the Institutional Animal Care and Use Committee of the National Cancer Center (No. K24-009-M01). Ten-week-old NOD scid gamma (NSG) mice housed under specific pathogen-free (SPF) conditions were used. An orthotopic tongue tumor model was established by intralingual injection of OC-15–derived tumor cells (2 × 10^6 cells/20 µL). On day 3 after tumor implantation, mice received total body irradiation (TBI) at 1.5 Gy. On days 4 and 6, mice were treated by intratumoral (i.t.) injection with either control dNGFR T cells (1 × 10^6 cells/20 µL) or 131CAR T cells (1 × 10^6 cells/20 µL). To evaluate systemic administration, an intravenous (i.v.) cohort was treated on the same schedule via tail vein injection with dNGFR or 131CAR adjusted to 1 × 10^6 cells/100 µL. A no-treatment group (tumor implantation only) was included to assess the natural course of tumor growth. The experimental scheme is shown in
Figure 4A.
4.6. Immunohistochemistry (IHC)
After euthanasia, tumors and major organs were harvested, fixed in 10% neutral-buffered formalin, and paraffin-embedded. Sections (3.5 µm) were prepared and used for hematoxylin and eosin (H&E) staining and immunohistochemistry for EPHB4, cytokeratin, and human CD8 (hCD8).
4.7. Digital Pathology Image Analysis
IHC slides were digitized using a NanoZoomer S360 slide scanner (Hamamatsu Photonics) and used for visualization and analysis. Tumor regions were defined as cytokeratin-positive areas. Whole-slide overview images (equivalent to 1.25× magnification) were exported from the virtual slides in TIFF format. Images were imported into Fiji (ImageJ), and spatial calibration was performed based on the scale bar. When required, RGB images were separated into individual channels (“Split Channels”), and the blue-channel images were used for subsequent analyses. Thresholding (Image > Adjust > Threshold) was applied to the blue-channel images, followed by binarization (“Apply”) to extract positive areas. Tumor area was quantified as the positive area (mm2) in the binarized images. Thresholds were adjusted minimally for each specimen according to a consistent policy, and the analysis workflow was applied uniformly across all images.
4.8. Statistical Analysis
Quantitative data are presented as individual values, with summary statistics provided as appropriate. Group comparisons of tumor area (mm2) were performed using a two-sided Welch’s t-test. A p-value < 0.05 was considered statistically significant. Statistical analyses were conducted using Microsoft Excel.
5. Conclusions
In this study, EPHB4-targeted CAR T cells (131CAR) exhibited rapid and antigen-dependent cytotoxic activity against EPHB4-high OSCC cells in vitro. Furthermore, in an OC-15–derived orthotopic tongue PDX model, 131CAR induced tumor regression. Although diminished EPHB4 immunoreactivity and residual cytokeratin-positive regions were observed after treatment, the cytokeratin-defined tumor area was significantly reduced, supporting the antitumor efficacy of 131CAR. Collectively, these findings indicate that EPHB4-targeted CAR T cells may represent a promising therapeutic strategy for clinical translation.
Author Contributions
Conceptualization, M.K., K.O. and T.N.; Methodology, M.K., K.O. and T.N.; Software, M.K., K.O. and T.S.; Validation, M.K., K.O., K.T., T.S. and T.N.; Formal analysis, M.K., K.O., T.S. and T.N.; Investigation, M.K., K.O., K.T. and T.N.; Resources, M.K., K.O., A.I., L.S., D.F., K.N., K.M. and T.N.; Data curation, M.K., K.O. and K.T.; Writing—original draft preparation, M.K.; Writing—review and editing, M.K., K.O. and T.N.; Visualization, M.K., K.O., K.T. and T.N.; Supervision, K.O., M.S., K.M. and T.N.; Project administration, T.N.; Funding acquisition, T.N. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported in part by the National Cancer Center Research and Development Fund (2022-A-9), and AMED under Grant Number JP24bm1123042 and JP25bm1123042.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of the National Cancer Center Research Ethics Committee (protocol code 2020-436, approved Feb. 1, 2021).
Informed Consent Statement
Written informed consent has been obtained from the patients for publication of this paper.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
We thank Naoki Yagi, Masatake Taniguchi, and the Division of Cancer Immunotherapy, EPOC, National Cancer Center members for technical and experimental support.
Conflicts of Interest
Author Tetsuya Nakatsura (T.N.) has received research grants from Optieum Biotechnologies Inc., T.N. hold stock options from Optieum Biotechnologies Inc. The remaining authors declare that the research was conducted without commercial or financial relationships that could be construed as a potential conflict of interest.
Abbreviations
| AIM-V |
AIM-V medium; |
| CAR |
chimeric antigen receptor; |
| CAR-T |
chimeric antigen receptor T cells; |
| dNGFR |
truncated (Δ) nerve growth factor receptor; |
| DMEM |
Dulbecco’s modified Eagle’s medium; |
| E:T |
effector-to-target; |
| EFNB2 |
ephrin-B2; |
| EPHB4 |
Ephrin type-B receptor 4; |
| FACS |
fluorescence-activated cell sorting; |
| FBS |
fetal bovine serum; |
| FFPE |
formalin-fixed, paraffin-embedded; |
| FN1 |
fibronectin type III–like domain 1; |
| FN2 |
fibronectin type III–like domain 2; |
| FCS |
fetal calf serum; |
| H&E |
hematoxylin and eosin; |
| hCD8+
|
human CD8-positive; |
| HNSCC |
head and neck squamous cell carcinoma; |
| ICI |
immune checkpoint inhibitor; |
| IFN-γ |
interferon gamma; |
| IHC |
immunohistochemistry; |
| IL-2 |
interleukin-2; |
| i.t. |
intratumoral; |
| i.v. |
intravenous; |
| MAb |
monoclonal antibody; |
| MFI |
mean fluorescence intensity; |
| MHC |
major histocompatibility complex; |
| NGFR |
nerve growth factor receptor; |
| NSG |
NOD scid gamma; |
| OSCC |
oral squamous cell carcinoma; |
| PBMCs |
peripheral blood mononuclear cells; |
| PBS |
phosphate-buffered saline; |
| PDX |
patient-derived xenograft; |
| PE |
phycoerythrin; |
| RT |
radiotherapy; |
| scFv |
single-chain variable fragment; |
| SPF |
specific pathogen-free; |
| TBI |
total body irradiation; |
| TM |
transmembrane; |
| TME |
tumor microenvironment. |
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2024, 74, 229–263. [Google Scholar] [CrossRef] [PubMed]
- Filho, A.M.; Laversanne, M.; Ferlay, J.; Colombet, M.; Piñeros, M.; Znaor, A.; Parkin, D.M.; Soerjomataram, I.; Bray, F. The GLOBOCAN 2022 cancer estimates: Data sources, methods, and a snapshot of the cancer burden worldwide. Int. J. Cancer 2025, 156, 1336–1346. [Google Scholar] [CrossRef]
- Bagan, J.; Sarrion, G.; Jimenez, Y. Oral cancer: Clinical features. Oral Oncol. 2010, 46, 414–417. [Google Scholar] [CrossRef]
- Chamoli, A.; Gosavi, A.S.; Shirwadkar, U.P.; Wangdale, K.V.; Behera, S.K.; Kurrey, N.K.; Kalia, K.; Mandoli, A. Overview of oral cavity squamous cell carcinoma: Risk factors, mechanisms, and diagnostics. Oral Oncol. 2021, 121, 105451. [Google Scholar] [CrossRef]
- Rivera, C. Essentials of oral cancer. Int. J. Clin. Exp. Pathol. 2015, 8, 11884–11894. [Google Scholar] [PubMed]
- Sun, Z.; Sun, X.; Chen, Z.; Du, J.; Wu, Y. Head and neck squamous cell carcinoma: Risk factors, molecular alterations, immunology and peptide vaccines. Int. J. Pept. Res. Ther. 2022, 28, 19. [Google Scholar] [CrossRef] [PubMed]
- Schuster, M.; Stelzle, F. Outcome measurements after oral cancer treatment: Speech and speech-related aspects—An overview. Oral Maxillofac. Surg. 2012, 16, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Kreeft, A.M.; van der Molen, L.; Hilgers, F.J.M.; Balm, A.J.M. Speech and swallowing after surgical treatment of advanced oral and oropharyngeal carcinoma: A systematic review of the literature. Eur. Arch. Otorhinolaryngol. 2009, 266, 1687–1698. [Google Scholar] [CrossRef]
- Farkona, S.; Diamandis, E.P.; Blasutig, I.M. Cancer immunotherapy: The beginning of the end of cancer? BMC Med. 2016, 14, 73. [Google Scholar] [CrossRef]
- Ferris, R.L.; Blumenschein, G.; Fayette, J.; Guigay, J.; Colevas, A.D.; Licitra, L.; Harrington, K.; Kasper, S.; Vokes, E.E.; Even, C.; et al. Nivolumab for recurrent squamous-cell carcinoma of the head and neck. N. Engl. J. Med. 2016, 375, 1856–1867. [Google Scholar] [CrossRef] [PubMed]
- Burtness, B.; Harrington, K.J.; Greil, R.; Soulieres, D.; Tahara, M.; de Castro, G., Jr.; Psyrri, A.; Basté, N.; Neupane, P.; Bratland, Å.; et al. Pembrolizumab alone or with chemotherapy versus cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): A randomised, open-label, phase 3 study. Lancet 2019, 394, 1915–1928. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. FDA approves neoadjuvant and adjuvant pembrolizumab for resectable locally advanced head and neck squamous cell carcinoma. Available online: fda.gov/drugs/resources-information-approved-drugs/fda-approves-neoadjuvant-and-adjuvant-pembrolizumab-resectable-locally-advanced-head-and-neck-squamous-cell-carcinoma (accessed on 28 February 2026).
- Uppaluri, R.; et al. Neoadjuvant and adjuvant pembrolizumab in locally advanced head and neck squamous-cell carcinoma (KEYNOTE-689). N. Engl. J. Med. 2025, 393, 1969–1983. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Bu, L.-L. Advances in CAR-T cell therapy in head and neck squamous cell carcinoma. J. Clin. Med. 2023, 12, 2173. [Google Scholar] [CrossRef]
- Saeidpour Masouleh, S.; Nasiri, K.; Ostovar Ravari, A.; Saligheh Rad, M.; Kiani, K.; Sharifi Sultani, A.; Nejati, S.T.; Afjadi, M.N. Advances and challenges in CAR-T cell therapy for head and neck squamous cell carcinoma. Biomark. Res. 2025, 13, 69. [Google Scholar] [CrossRef]
- Escobar, G.; Berger, T.R.; Maus, M.V. CAR-T cells in solid tumors: Challenges and breakthroughs. Cell Rep. Med. 2025, 6, 102353. [Google Scholar] [CrossRef]
- Khan, S.H.; Choi, Y.; Veena, M.; Lee, J.K.; Shin, D.S. Advances in CAR T cell therapy: Antigen selection, modifications, and current trials for solid tumors. Front. Immunol. 2024, 15, 1489827. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, H.; Zhang, Y. Targeting receptor tyrosine kinase EphB4 in cancer therapy. Semin. Cancer Biol. 2019, 56, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Mosch, B.; Reissenweber, B.; Neuber, C.; Pietzsch, J. Eph receptors and ephrin ligands: Important players in angiogenesis and tumor angiogenesis. J. Oncol. 2010, 2010, 135285. [Google Scholar] [CrossRef]
- Nakatsura, T.; Ohnuki, K.; Takenouchi, K.; Suzuki, T.; Islam, A.; Shen, L.; Fujita, D.; Nakamaru, K.; Suzuki, M.; Matsuura, K. Expression Profiles of Five Common Cancer Membrane Protein Antigens Collected for the Development of Cocktail CAR-T Cell Therapies Applicable to Most Solid Cancer Patients. Int. J. Mol. Sci. 2025, 26, 2145. [Google Scholar] [CrossRef]
- Kinoshita, H.; et al. Identification of 68 HLA-A24- and -A2-restricted cytotoxic T lymphocyte-inducing peptides derived from 10 common cancer-specific antigens frequently expressed in various solid cancers. Neoplasia 2025, 61, 101135. [Google Scholar] [CrossRef]
- Ito, H.; et al. Feasibility of intratumoral administration with EPHB4-CAR-T cells for the treatment of oral squamous cell carcinoma. Cancer Sci. 2025, 116, 1227–1238. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, H.; Zhang, Y. Targeting receptor tyrosine kinase EphB4 in cancer therapy. Semin. Cancer Biol. 2019, 56, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Krasnoperov, V.; Kumar, S.R.; Ley, E.; et al. Novel EphB4 Monoclonal Antibodies Modulate Angiogenesis and Inhibit Tumor Growth. Am. J. Pathol. 2010, 176(4), 2029–2038. [Google Scholar] [CrossRef]
- Yoshikawa, T.; Nakatsugawa, M.; Suzuki, S.; Shirakawa, H.; Nobuoka, D.; Sakemura, N.; Motomura, Y.; Tanaka, Y.; Hayashi, S.-I.; Nakatsura, T. HLA-A2-restricted glypican-3 peptide-specific CTL clones induced by peptide vaccine show high avidity and antigen-specific killing activity against tumor cells. Cancer Sci. 2011, 102, 918–925. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).