Submitted:
02 March 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Subject and Methods
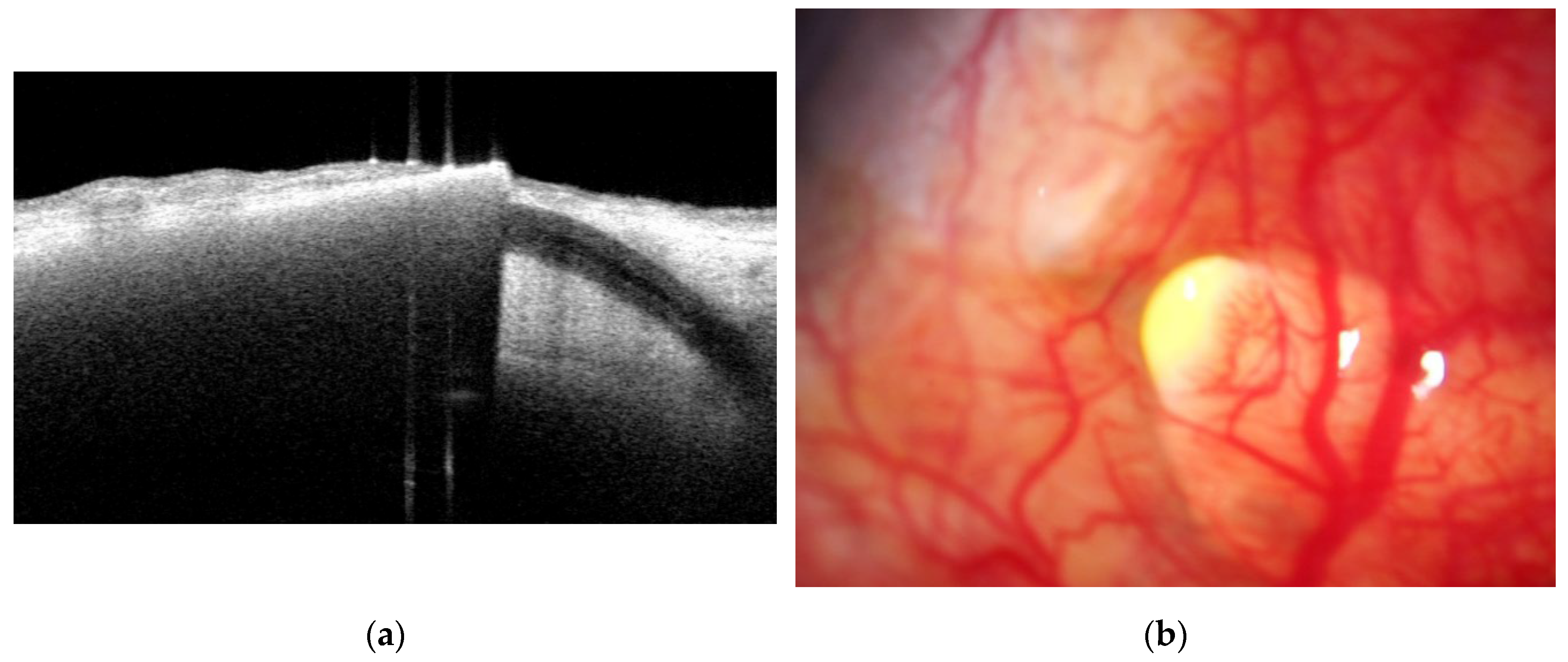
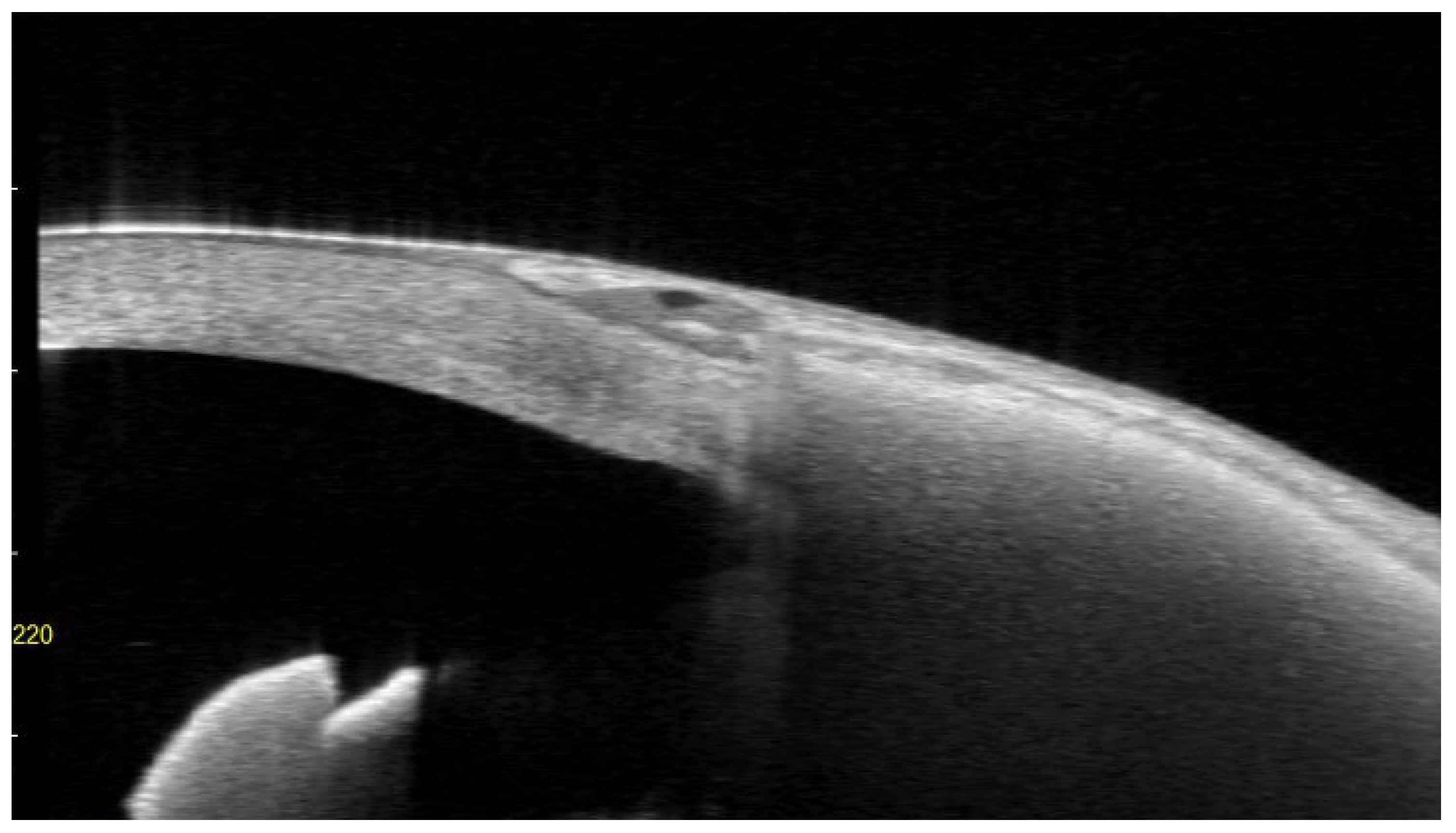
2.1. Anterior Segment Optical Coherence Tomography (AS-OCT) Evaluation
2.2. Statistical Analysis
2.3. Propensity Score & Weighting
2.4. Time-to-Event Outcome
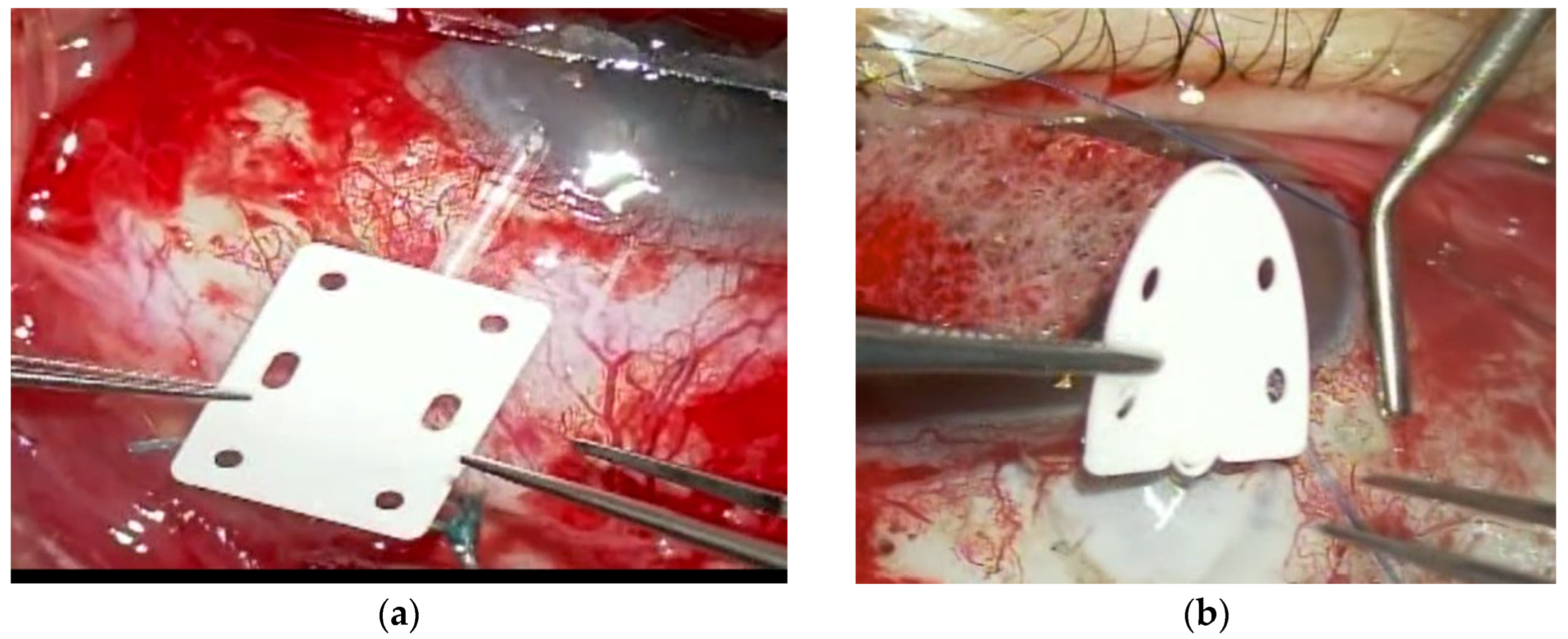
2.5. Surgical Techniques
3. Results
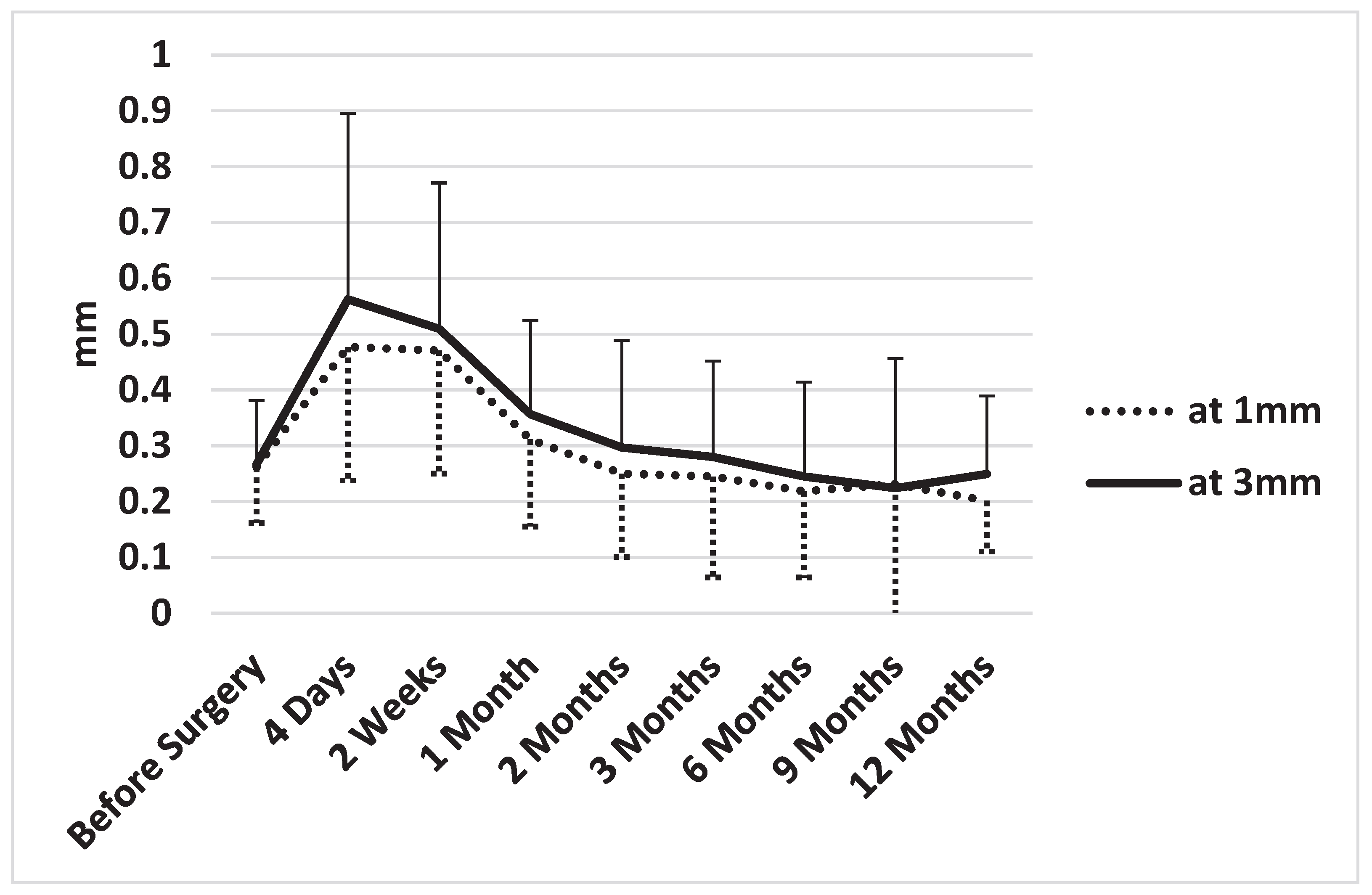
3.1. Clinical Course of Conjunctival Thickness
3.2. Propensity Score Overlap Weighting and Covariate Balance
| Covariate | SMD unweighted | SMD weighted |
|---|---|---|
| Age | 0.268543 | -0.23788 |
| NVG | -0.10863 | 0.306316 |
| prior surgery | -0.25719 | 0.026397 |
| Preop IOP | 0.15107 | -0.01721 |
| Preop meds | 0.205732 | -0.02854 |
| Quadrant | 0.782763 | 0.055892 |
| Insertion site | -1.04855 | -0.30277 |
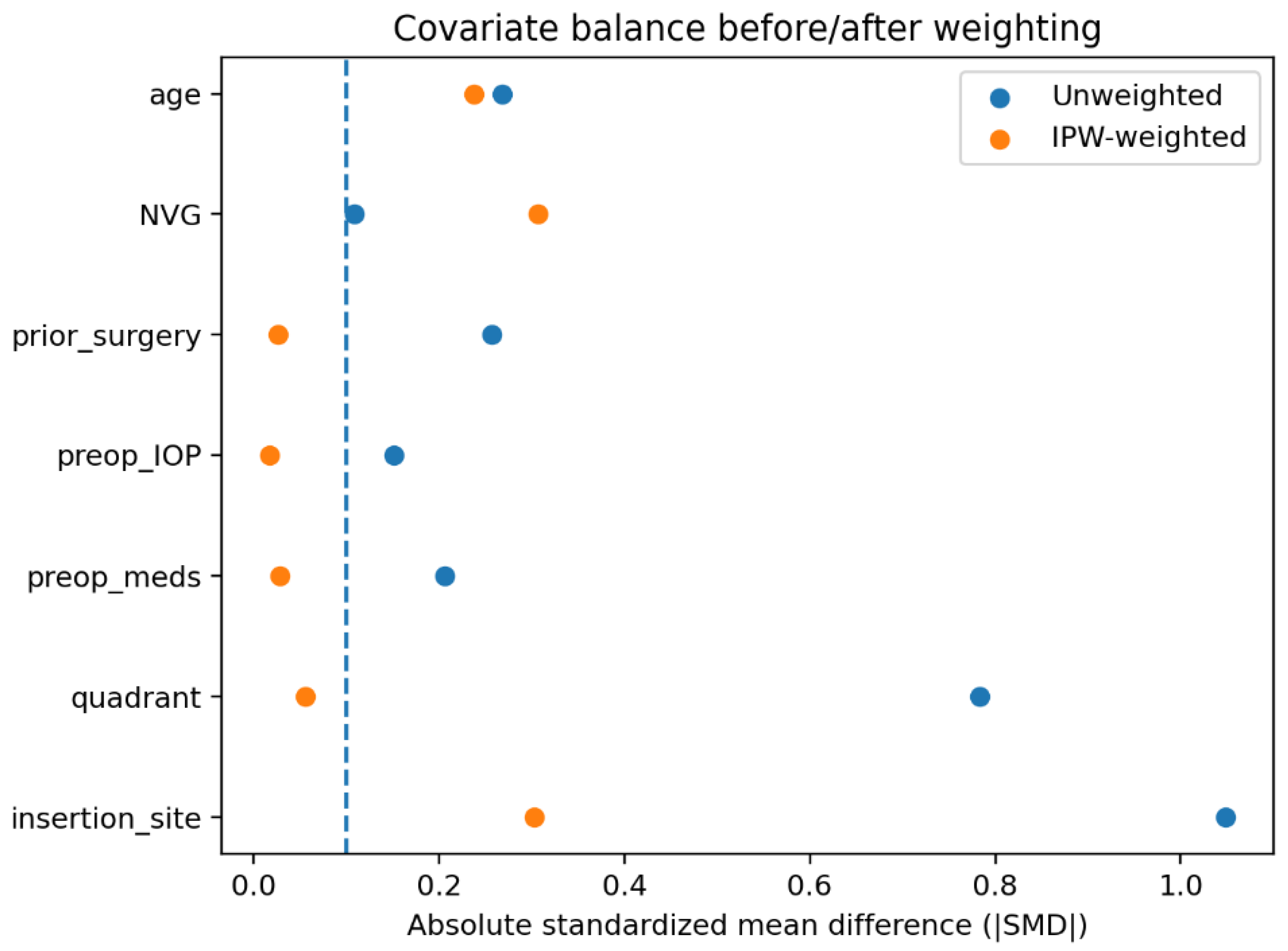
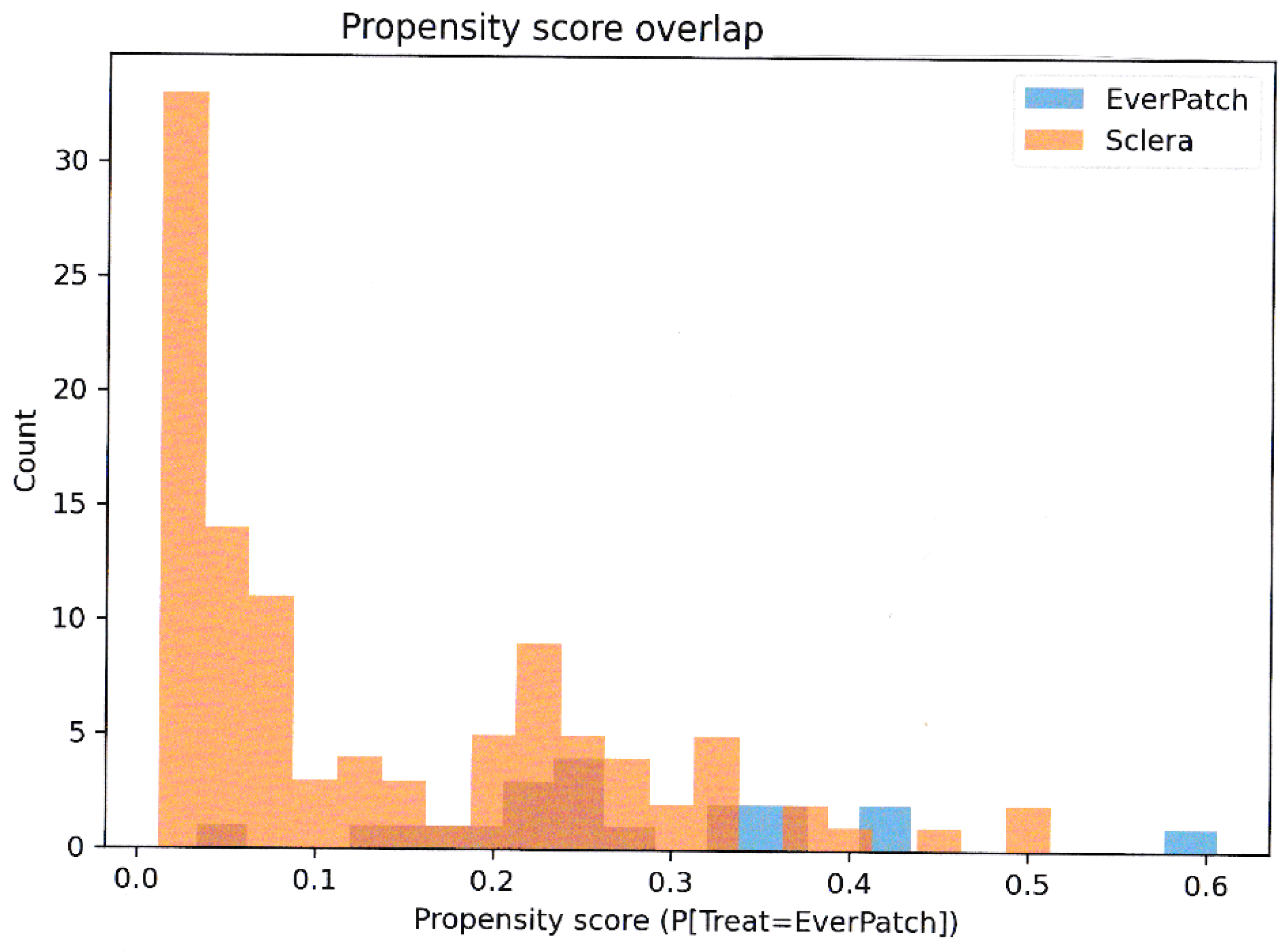
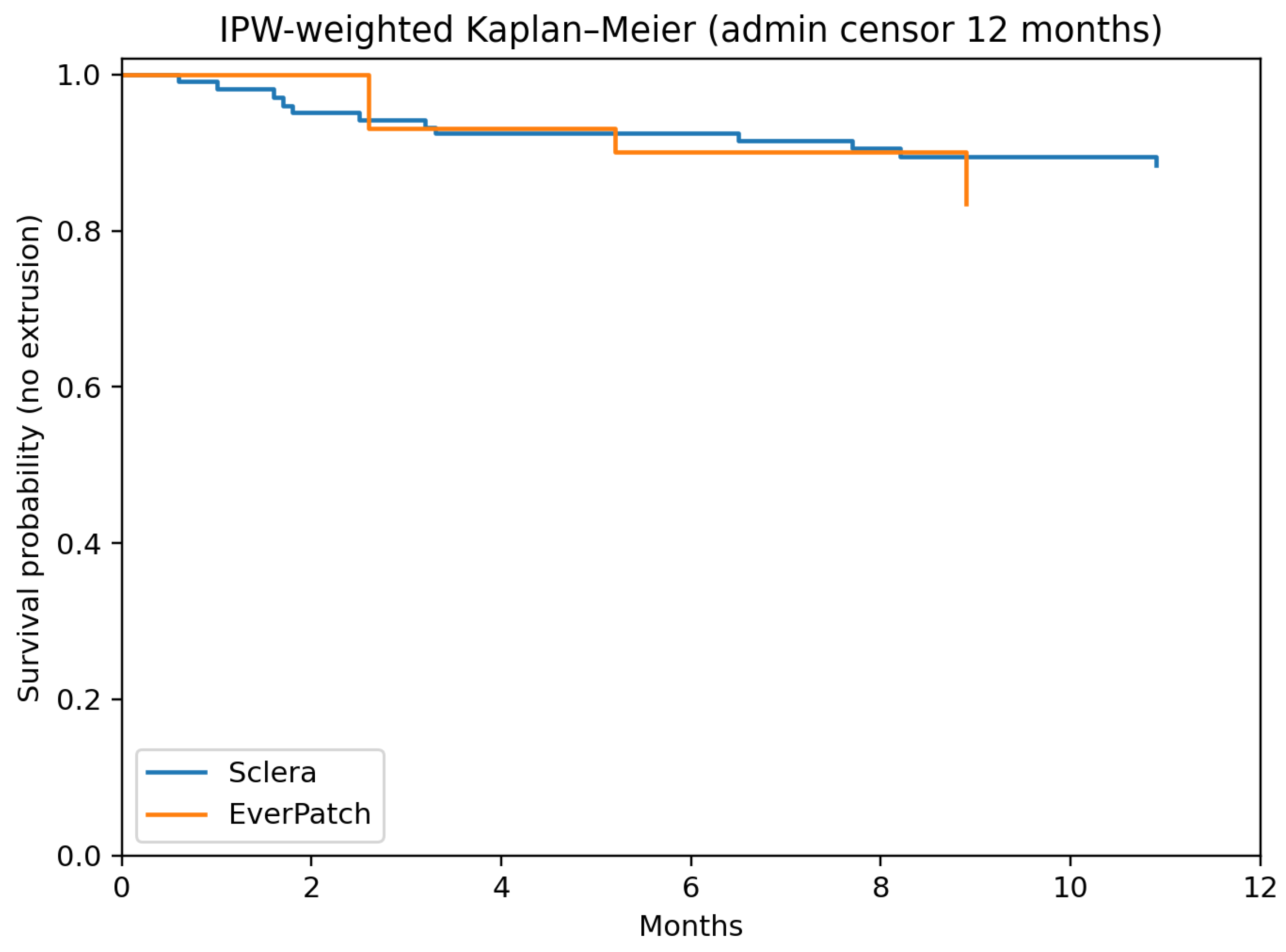
3.3. Propensity Score Weighting and Survival Analysis
3.4. IPW-Weighted Cox Proportional Hazards Model
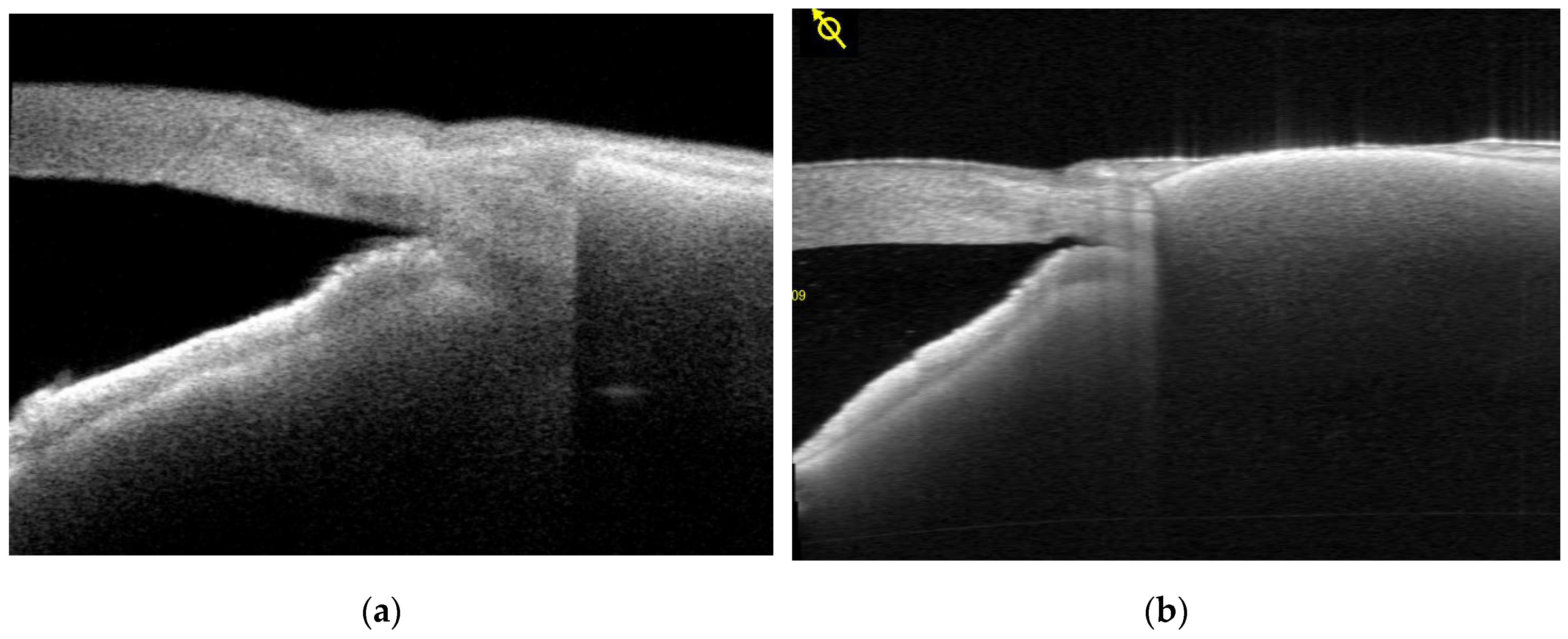
4. Discussion
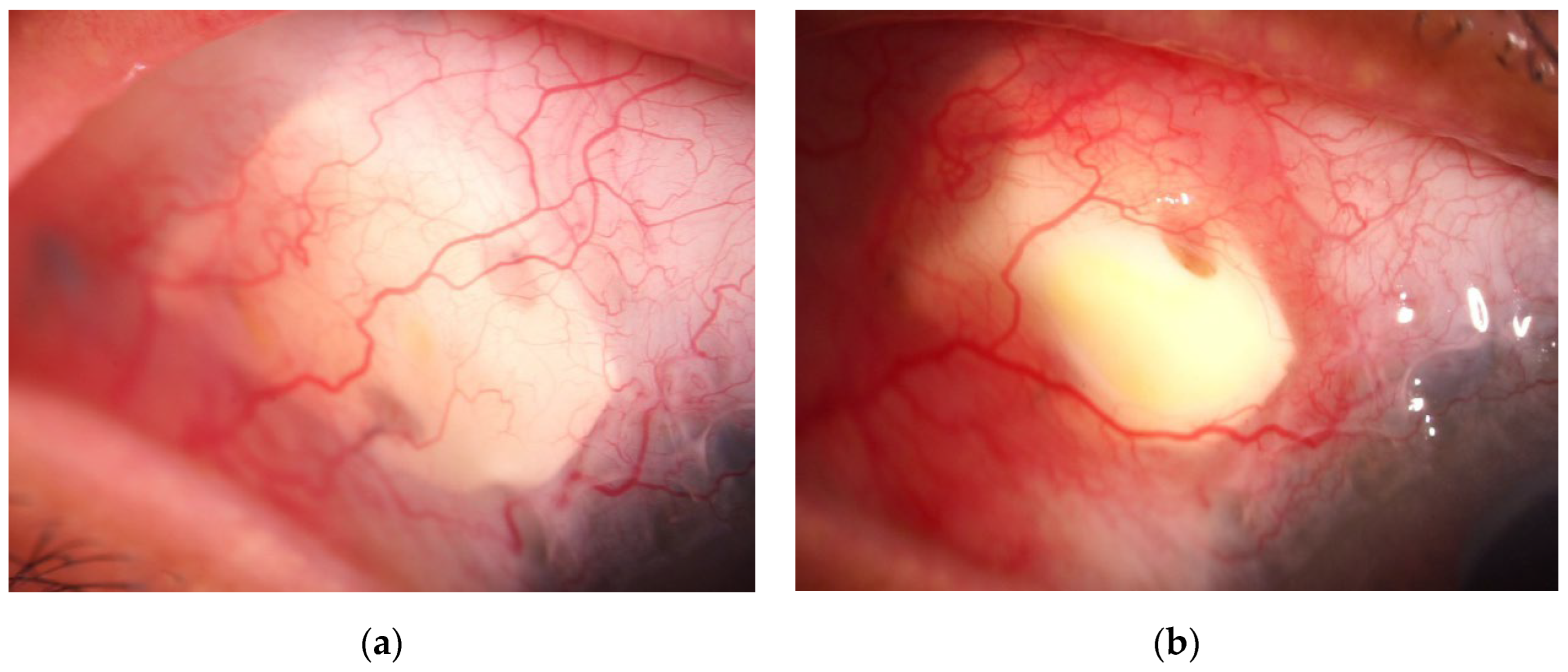
Tissue Reaction
5. Limitation:
6. Conclusion:
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EPP | EverPatch Plus® |
| GDD | Glaucoma drainage device |
| PCU | Polycarbonate urethane |
| FDA | Food and Drug Administration |
| AS-OCT | Anterior segment optical coherence tomograph |
| PS | propensity score |
| SMD | standardized mean difference |
| IPW | inverse probability of treatment weighting |
| HR | hazard ratio |
| CI | confidence interval |
| AGV | Ahmed glaucoma valve |
| PGI | Paul glaucoma implant |
| ANOVA | analysis of variance |
| IOP | Intraocular pressure |
| NVG | Neovascular glaucoma |
| POAG | Primary open angle glaucoma |
| PACG | Primary angle closure glaucoma |
| SG | Secondary glaucoma |
| Log MAR | Logarithmic minimal angular resolution |
| BCVA | Best corrected visual acuity |
| HFA | Humphrey visual field analyzer |
| MD | Mean deviation |
| dB | Decibel |
| SS | scleral spur |
References
- Akbas, Y.B.; Alagoz, N.; Sari, C.; Altan, C.; Yasar, T. Evaluation of pericardium patch graft thickness in patients with Ahmed glaucoma valve implantation: an anterior segment OCT study. Jpn J Ophthalmol. 2024, 68, 192–199. [Google Scholar] [CrossRef]
- de Luna, R.A.; Moledina, A.; Wang, J.; Jampel, H.D. Measurement of Gamma-Irradiated Corneal Patch Graft Thickness After Aqueous Drainage Device Surgery. JAMA Ophthalmol. 2017, 135, 941–946. [Google Scholar] [CrossRef]
- Smith, M.F.; Doyle, J.W.; Ticrney, J.W., Jr. A comparison of glaucoma drainage implant tube coverage. J Glaucoma. 2002, 11, 143–147. [Google Scholar] [CrossRef]
- Schipper, P.; Weber, C.; Lu, K.; Fan, S.; Prokosch, V.; Holz, F.G.; Mercieca, K. Anterior segment OCT for imaging PAUL(®) glaucoma implant patch grafts: a useful method for follow-up and risk management. Graefes Arch Clin Exp Ophthalmol. 2024. [Google Scholar] [CrossRef]
- Nagi, K.S.; Cumba, R.J.; Bell, N.P.; Blieden, L.S.; Chuang, A.Z.; Mankiewicz, K.A.; Nguyen, D.; Feldman, R.M. Short-Term Outcomes of KeraSys Patch Graft for Glaucoma Drainage Devices: A Case Series. J Ophthalmol. 2013, 2013, 784709. [Google Scholar] [CrossRef]
- Thakur, S.; Ichhpujani, P.; Kumar, S. Grafts in Glaucoma Surgery: A Review of the Literature. Asia Pac J Ophthalmol (Phila). 2017, 6, 469–476. [Google Scholar] [CrossRef]
- Zalta, A.H. Long-term experience of patch graft failure after Ahmed Glaucoma Valve® surgery using donor dura and sclera allografts. Ophthalmic Surg Lasers Imaging. 2012, 43, 408–415. [Google Scholar] [CrossRef]
- Jacob, T.; LaCour, O.J.; Burgoyne, C.F.; LaFleur, P.K.; Duzman, E. Expanded polytetrafluoroethylene reinforcement material in glaucoma drain surgery. J Glaucoma. 2001, 10, 115–120. [Google Scholar] [CrossRef]
- Yasuoka, K.; Tada, K.; Yasuoka, K. Ahmed Glaucoma Valve (AGV) implantation using Gore-Tex. Jpn J Ophthalmic Surg. 2024, 37, 541–545. [Google Scholar]
- Abramowitz, B.; Moster, M.R.; Pro, M.; Schmidt, C.; Dale, E.; Yung, E.; Williams, A. Pilot Study Assessing the Use of High-density Polyethylene (Su-Por) for Tube Shunt Patch Grafts. J Glaucoma. 2018, 27, e14–e16. [Google Scholar] [CrossRef]
- Leszczynski, R.; Gumula, T.; Stodolak-Zych, E.; Pawlicki, K.; Wieczorek, J.; Kajor, M.; Blazewicz, S. Histopathological Evaluation of a Hydrophobic Terpolymer (PTFE-PVD-PP) as an Implant Material for Nonpenetrating Very Deep Sclerectomy. Invest Ophthalmol Vis Sci. 2015, 56, 5203–5209. [Google Scholar] [CrossRef]
- Jawad, S.; Halenda, K. Late-onset Pseudomonas aeruginosa orbital cellulitis following glaucoma drainage device implantation. Am J Ophthalmol Case Rep. 2024, 34, 102054. [Google Scholar] [CrossRef]
- Nakamura, K.; Fujimoto, T.; Okada, M.; Maki, K.; Shimazaki, A.; Kato, M.; Inoue, T. Tissue Reactivity to, and Stability of, Glaucoma Drainage Device Materials Placed Under Rabbit Conjunctiva. Transl Vis Sci Technol. 2022, 11, 9. [Google Scholar] [CrossRef]
- Gu, Y.; Tian, C.; Qin, Y.; Sun, Y.; Liu, S.; Li, H.; Duan, X.; Shu, C.; Ouyang, C. The novel hybrid polycarbonate polyurethane / polyester three-layered large-diameter artificial blood vessel. J Biomater Appl. 2022, 36, 965–975. [Google Scholar] [CrossRef]
- Lazic, S.; Kellett, C.; Afzal, I.; Mohan, R.; Killampalli, V.; Field, R.E. Three-year results of a polycarbonate urethane acetabular bearing in total hip arthroplasty. Hip Int. 2020, 30, 303–308. [Google Scholar] [CrossRef]
- Sun, M.; Elkhodiry, M.; Shi, L.; Xue, Y.; Abyaneh, M.H.; Kossar, A.P.; Giuglaris, C.; Carter, S.L.; Li, R.L.; Bacha, E.; et al. A biomimetic multilayered polymeric material designed for heart valve repair and replacement. Biomaterials. 2022, 288, 121756. [Google Scholar] [CrossRef]
- The CorNeat EverPatch: Superior Alternative to Tissue Grafts. accessed on 2025/1/29. https://www.corneat.com/news/the-corneat-everpatch%3A-superior-alternative-to-tissue-grafts. (accessed on 2025/1/29).
- Kanter, J.; Garkal, A.; Cardakli, N.; Pitha, I.; Sabharwal, J.; Schein, O.D.; Ramulu, P.Y.; Parikh, K.S.; Johnson, T.V. Early Postoperative Conjunctival Complications Leading to Exposure of Surgically Implanted CorNeat EverPatch Devices. Ophthalmology. 2025, 132, 799–814. [Google Scholar] [CrossRef]
- Youn, S.; Yan, D.B. Five-Year Outcomes of Graft-Free Tube Shunts and Risk Factors for Tube Exposures in Glaucoma. J Glaucoma. 2024, 33, 139–147. [Google Scholar] [CrossRef]
- Al-Beishri, A.S.; Malik, R.; Freidi, A.; Ahmad, S. Risk Factors for Glaucoma Drainage Device Exposure in a Middle-Eastern Population. J Glaucoma. 2019, 28, 529–534. [Google Scholar] [CrossRef]
- Byun, Y.S.; Lee, N.Y.; Park, C.K. Risk factors of implant exposure outside the conjunctiva after Ahmed glaucoma valve implantation. Jpn J Ophthalmol. 2009, 53, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Jomar, D.E.; Al-Shahwan, S.; Al-Beishri, A.S.; Freidi, A.; Malik, R. Risk Factors for Glaucoma Drainage Device Exposure in Children: A Case-Control Study. Am J Ophthalmol. 2023, 245, 174–183. [Google Scholar] [CrossRef]
- Senthilkumar, V.A.; Akbar, U.S.; Rajendrababu, S. Incidence, etiology, and risk factors for tube explantation in patients undergoing patch-free glaucoma drainage device. Indian J Ophthalmol. 2025, 73, S267–s271. [Google Scholar] [CrossRef]
- Geffen, N.; Buys, Y.M.; Smith, M.; Anraku, A.; Alasbali, T.; Rachmiel, R.; Trope, G.E. Conjunctival complications related to Ahmed glaucoma valve insertion. J Glaucoma. 2014, 23, 109–114. [Google Scholar] [CrossRef]
- Chaku, M.; Netland, P.A.; Ishida, K.; Rhee, D.J. Risk factors for tube exposure as a late complication of glaucoma drainage implant surgery. Clin Ophthalmol. 2016, 10, 547–553. [Google Scholar] [CrossRef]
- Levinson, J.D.; Giangiacomo, A.L.; Beck, A.D.; Pruett, P.B.; Superak, H.M.; Lynn, M.J.; Costarides, A.P. Glaucoma drainage devices: risk of exposure and infection. Am J Ophthalmol. 2015, 160, 516–521.e512. [Google Scholar] [CrossRef]
- Edo, A.; Jian, K.; Kiuchi, Y. Risk factors for exposure of Baerveldt glaucoma drainage implants: a case-control study. BMC Ophthalmol. 2020, 20, 364. [Google Scholar] [CrossRef]
- Trubnik, V.; Zangalli, C.; Moster, M.R.; Chia, T.; Ali, M.; Martinez, P.; Richman, J.; Myers, J.S. Evaluation of Risk Factors for Glaucoma Drainage Device-related Erosions: A Retrospective Case-Control Study. J Glaucoma. 2015, 24, 498–502. [Google Scholar] [CrossRef]
- Muir, K.W.; Lim, A.; Stinnett, S.; Kuo, A.; Tseng, H.; Walsh, M.M. Risk factors for exposure of glaucoma drainage devices: a retrospective observational study. BMJ Open. 2014, 4, e004560. [Google Scholar] [CrossRef]
- Gizzi, C.; Rai, P.; Barton, K. Aqueous shunt exposure repair: outcomes and risk factors for recurrence. Eye (Lond). 2024, 38, 3065–3071. [Google Scholar] [CrossRef]
- Gedde, S.J.; Feuer, W.J.; Shi, W.; Lim, K.S.; Barton, K.; Goyal, S.; Ahmed, I.I.K.; Brandt, J. Treatment Outcomes in the Primary Tube Versus Trabeculectomy Study after 1 Year of Follow-up. Ophthalmology. 2018, 125, 650–663. [Google Scholar] [CrossRef]
- Singer, R.; Kapelushnik, N.; Rotenstreich, Y.; Leshno, A.; Barkana, Y.; Skaat, A. Surgical outcomes of Ahmed glaucoma valve implantation with plate fixation using vicryl sutures or no plate fixation. Eur J Ophthalmol. 2021, 11206721211012869. [Google Scholar] [CrossRef] [PubMed]
- Kapelushnik, N.; Singer, R.; Barkana, Y.; Rosenfeld, A.; Tucker, Y.; Jaber, W.; Gaton, D.; Leshno, A.; Skaat, A. Surgical Outcomes of Ahmed Glaucoma Valve Implantation Without Plate Sutures: A 10-Year Retrospective Study. J Glaucoma. 2021, 30, 502–507. [Google Scholar] [CrossRef] [PubMed]
| EverPatch | Scleral Patch | P Value (ANOVA) | |
|---|---|---|---|
| Age | 67.1±15.6 | 62.5±17.9 | 0.305 |
| Type of Glaucoma | NVG 5, POAG 8, PEG 2,SG 2,PACG 2 | NVG 27, POAG 25, PEG 10, SG 26, PACG 5 | 0.092 (residual analysis) |
| Sex: Male/Female | 12/7 | 57/48 | 0.617 |
| Side: R/L | 9/10 | 50/55 | 1.00 |
| Pre-op IOP (mmHg) | 31.5±12.8 | 29.4±9.4 | 0.521 |
| Pre-op BCVA (logMAR) | 0.980±0.960 | 0.708±0.857 | 0.213 |
| Pre-op meds | 4.4±1.2 | 4.1±1.2 | 0.409 |
| Number of prior surgeries | 2.4±1.7 | 2.7±1.9 | 0.444 |
| Visual field defects (HFA: MD) | -20.6±10.9* | -18.9±9.3* | 0.556 |
| Type of GDD used | AGV 6, PGI 13 | AGV 105 | NA |
| Endothelial cell density (cells/mm2) | 1981±773 | 1993±636 | 0.945 |
| Type of EverPatch | Shield type 16, Rectangular type 3 | NA | |
| Quadrant of tube insertion | Sup-temporal 16, Inf-nasal 3 |
Sup-temporal 52, Sup-nasal 12, Inf-temporal 30, Inf-nasal 11 |
0.005 (residual analysis) |
| Tube tip location | Anterior chamber 6, Ciliary sulcus 11, pars plana 2 | Anterior chamber 26, ciliary sulcus 21, pars plana 58 | P<0.001 (residual analysis) |
| Pre-op conjunctival thickness at 1 mm from SS | 0.262±0.100 | NA | |
| Pre-op conjunctival thickness at 3 mm from SS | 0.267±0.114 | NA |
| Pre-op | 1M | 3M | 6M | 12M | ||
|---|---|---|---|---|---|---|
| EverPatch | # of medications | 4.4±1.2 | NA | NA | NA | 2.6±1.8 |
| IOP | 31.5±12.8 | 16.7±4.3 | 15.2±6.0 | 14.8±4.3 | 15.4±4.8 | |
| Control | # of medications P by ANOVA |
4.1±1.2 P=0.409 | NA | NA | NA | 2.2±1.5 P=0.379 |
| IOP | 29.7±10.9 | 19.7±9.0 | 18.1±5.7 | 16.9±4.8 | 16.4±5.9 | |
| P by ANOVA | 0.521 | 0.17 | 0.041 | 0.075 | 0.549 |
| Time point | 1 mm thickness (mm) | % Change vs Baseline | P vs. Baseline | 3 mm Thickness (mm) | % Change vs Baseline | P vs Baseline |
|---|---|---|---|---|---|---|
| Baseline | 0.262±0.100 | 0 | - | 0.267±0.114 | 0 | - |
| 4 days N=19 | 0.477±0.239 | 81.9 | 0.002↑ | 0.562±0.333 | 110.8 | 0.001↑ |
| 2 weeks N=18 | 0.471±0.221 | 79.5 | 0.001↑ | 0.509±0.262 | 90.9 | 0.001↑ |
| 1 month N=19 | 0.311± 0.16 | 18.8 | 0.756(ns) | 0.357±0.170 | 33.7 | 0.077(ns) |
| 2 months N=19 | 0.250±0.149 | -4.6 | 0.779(ns) | 0.297±0.192 | 11.3 | 0.578(ns) |
| 3 months N=18 | 0.245±0.181 | -6.0 | 0.731(ns) | 0.280±0.172 | 4.9 | 0.793(ns) |
| 6 months N=19 | 0.218±0.154 | -16.7 | 0.731(ns) | 0.245±0.159 | -8.2 | 0.660(ns) |
| 9 months N=17 | 0.231±0.237 | -11.8 | 0.593(ns) | 0.224±0.232 | -16.0 | 0.465(ns) |
| 12 months N=9 | 0.201±0.091 | -23.2 | 0.278(ns) | 0.249±0.140 | -6.5 | 0.797(ns) |
| Time point | SS-EPP (mm) | P* |
|---|---|---|
| 4 days N=9 | 1.01±0.20 | |
| 2 weeks N=7 | 1.07±0.23 | 0.893 |
| 1 month N=14 | 1.13±0.34 | 0.263 |
| 2 months N=15 | 1.20±0.39 | 0.953 |
| 3 months N=14 | 1.21±0.40 | 0.484 |
| 6 months N=14 | 1.17±0.532 | 0.499 |
| 9 months N=17 | 1.23±0.542 | 0.214 |
| 12 months N=8 | 1.19±0.643 | 0.144 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




