Submitted:
02 March 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
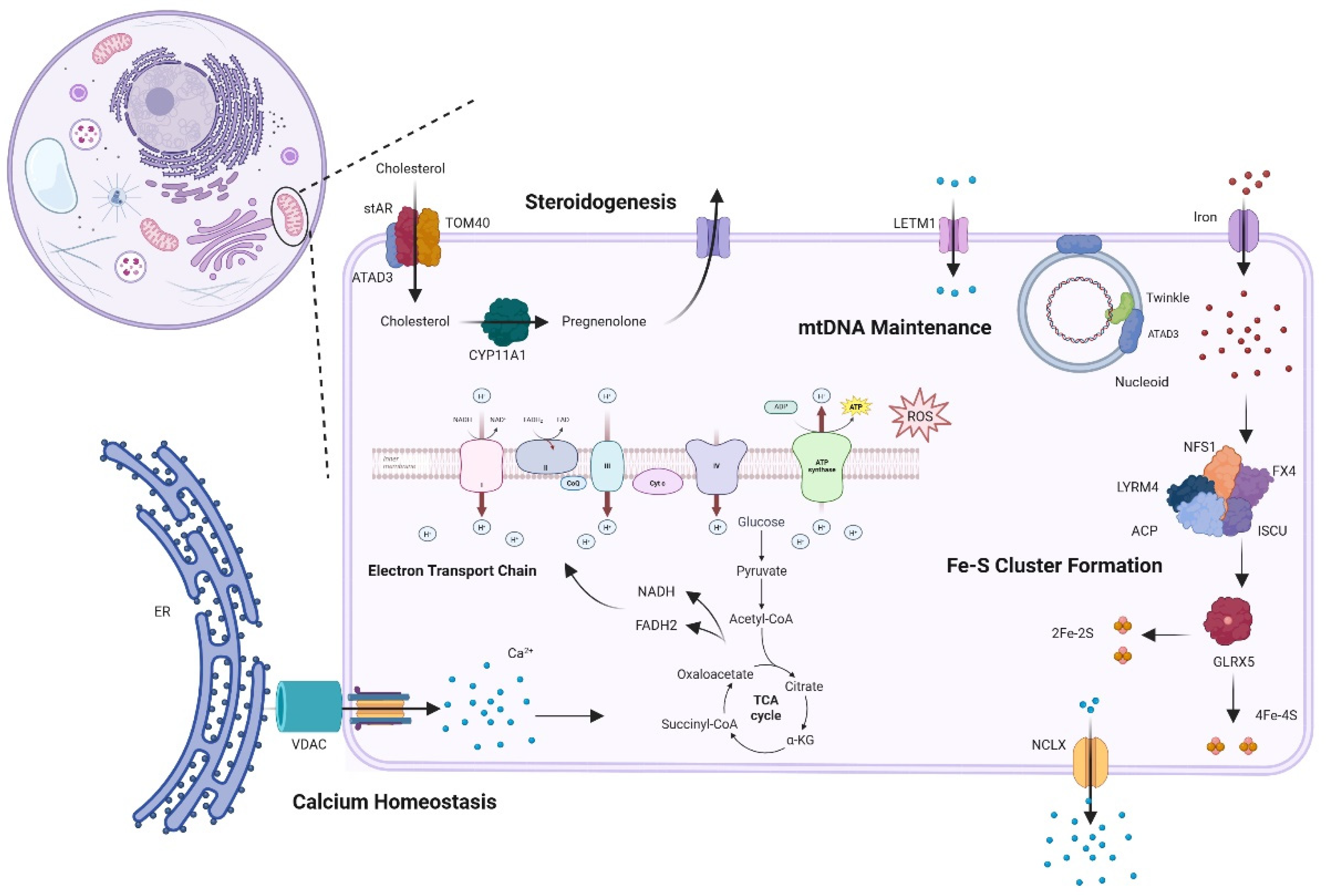
2. Molecular Pathology of Mitochondrial Disease
3. Evidence for Changes to Cholesterol Homeostasis in Mitochondrial Disease
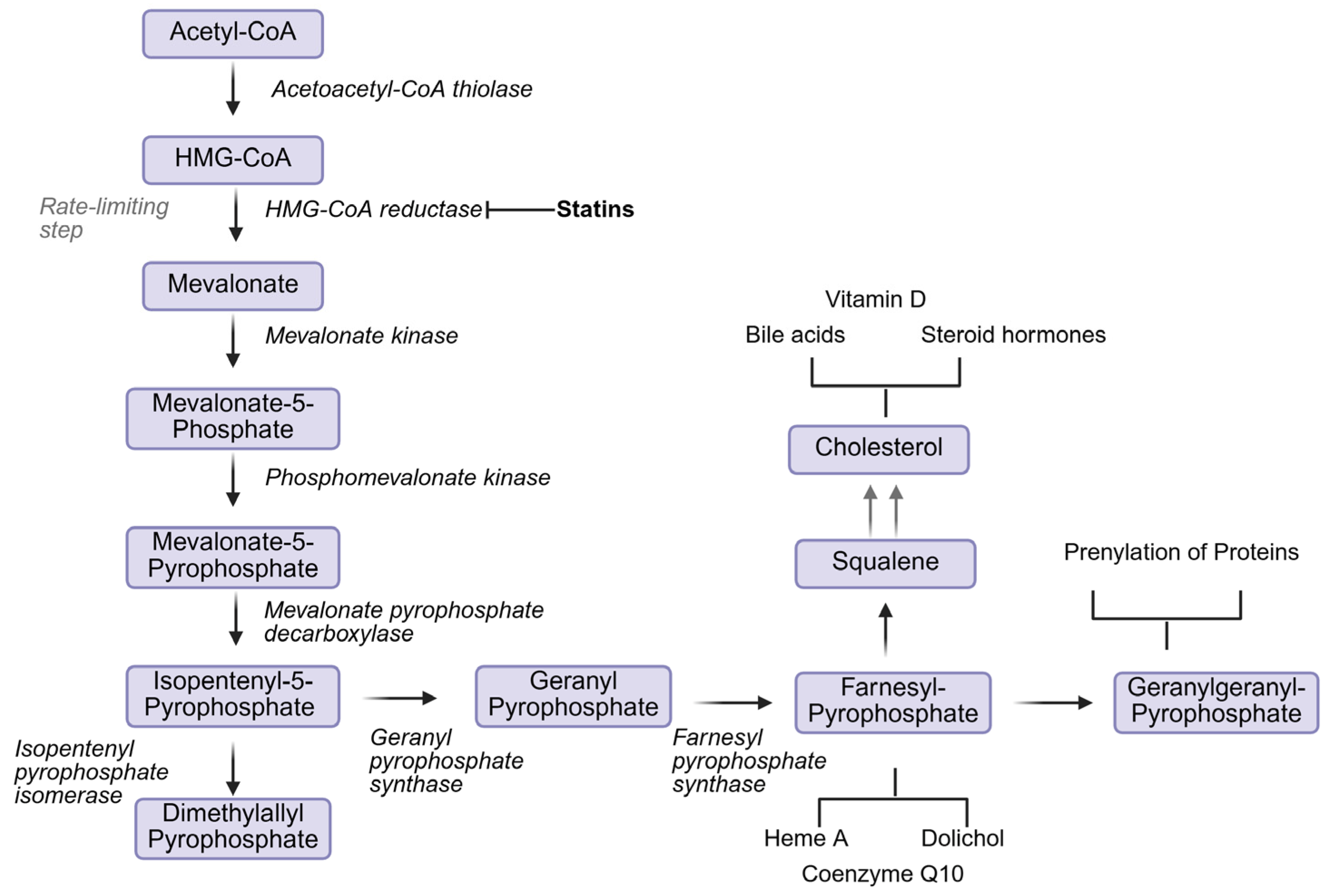
3.1. Role of Cholesterol in Health and Disease
3.2. Cholesterol in Mitochondrial Disease
3.2.1. Cholesterol in ATAD3-Related Pathologies
3.2.2. Cholesterol in Leigh Syndrome
3.2.3. Cholesterol in Other Mitochondrial Diseases
4. Adverse Effects of Statins in Mitochondrial Disease – Potential Role of Cholesterol?
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Gorman, G. S.; Schaefer, A. M.; Ng, Y.; Gomez, N.; Blakely, E. L.; Alston, C. L.; Feeney, C.; Horvath, R.; Yu-Wai-Man, P.; Chinnery, P. F.; Taylor, R. W.; Turnbull, D. M.; McFarland, R. Prevalence of nuclear and mitochondrial DNA mutations related to adult mitochondrial disease. Ann Neurol 2015, 77((5)), 753–9. [Google Scholar] [CrossRef] [PubMed]
- Gorman, G. S.; Chinnery, P. F.; DiMauro, S.; Hirano, M.; Koga, Y.; McFarland, R.; Suomalainen, A.; Thorburn, D. R.; Zeviani, M.; Turnbull, D. M. Mitochondrial diseases. Nat Rev Dis Primers 2016, 2, 16080. [Google Scholar] [CrossRef] [PubMed]
- Wen, H.; Deng, H.; Li, B.; Chen, J.; Zhu, J.; Zhang, X.; Yoshida, S.; Zhou, Y. Mitochondrial diseases: from molecular mechanisms to therapeutic advances. Signal Transduct Target Ther 2025, 10((1)), 9. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Oreja, M.; Sandoval, A.; Bruland, O.; Perez-Rodriguez, D.; Fernandez-Pelayo, U.; de Arbina, A. L.; Villar-Fernandez, M.; Hernandez-Eguiazu, H.; Hernandez, I.; Park, Y.; Goicoechea, L.; Pascual-Frias, N.; Garcia-Ruiz, C.; Fernandez-Checa, J.; Marti-Carrera, I.; Gil-Bea, F. J.; Hasan, M. T.; Gegg, M. E.; Bredrup, C.; Knappskog, P. M.; Gerenu-Lopetegui, G.; Varhaug, K. N.; Bindoff, L. A.; Spinazzola, A.; Yoon, W. H.; Holt, I. J. Elevated cholesterol in ATAD3 mutants is a compensatory mechanism that leads to membrane cholesterol aggregation. Brain 2024, 147((5)), 1899–1913. [Google Scholar] [CrossRef]
- Du, J.; Zhang, C.; Liu, F.; Liu, X.; Wang, D.; Zhao, D.; Shui, G.; Zhao, Y.; Yan, C. Distinctive metabolic remodeling in TYMP deficiency beyond mitochondrial dysfunction. J Mol Med (Berl) 2023, 101((10)), 1237–1253. [Google Scholar] [CrossRef]
- Toshima, T.; Yagi, M.; Do, Y.; Hirai, H.; Kunisaki, Y.; Kang, D.; Uchiumi, T. Mitochondrial translation failure represses cholesterol gene expression via Pyk2-Gsk3beta-Srebp2 axis. Life Sci Alliance 2024, 7((7)). [Google Scholar] [CrossRef]
- Schirris, T. J. J.; Rossell, S.; de Haas, R.; Frambach, S.; Hoogstraten, C. A.; Renkema, G. H.; Beyrath, J. D.; Willems, P.; Huynen, M. A.; Smeitink, J. A. M.; Russel, F. G. M.; Notebaart, R. A. Stimulation of cholesterol biosynthesis in mitochondrial complex I-deficiency lowers reductive stress and improves motor function and survival in mice. Biochim Biophys Acta Mol Basis Dis 2021, 1867((4)), 166062. [Google Scholar] [CrossRef]
- Argov, Z. Statins in hereditary myopathies: to give or not to give. Neuromuscul Disord 2024, 41, 35–39. [Google Scholar] [CrossRef]
- Golomb, B. A.; Evans, M. A. Statin adverse effects: a review of the literature and evidence for a mitochondrial mechanism. Am J Cardiovasc Drugs 2008, 8((6)), 373–418. [Google Scholar] [CrossRef]
- Azemawah, V.; Movahed, M. R.; Centuori, P.; Penaflor, R.; Riel, P. L.; Situ, S.; Shadmehr, M.; Hashemzadeh, M. State of the Art Comprehensive Review of Individual Statins, Their Differences, Pharmacology, and Clinical Implications. Cardiovasc Drugs Ther 2019, 33((5)), 625–639. [Google Scholar] [CrossRef]
- Pek, N. M. Q.; Phua, Q. H.; Ho, B. X.; Pang, J. K. S.; Hor, J. H.; An, O.; Yang, H. H.; Yu, Y.; Fan, Y.; Ng, S. Y.; Soh, B. S. Cell Death Dis 2019, 10((11)), 802. [CrossRef]
- Yang, L.; Lin, X.; Tang, H.; Fan, Y.; Zeng, S.; Jia, L.; Li, Y.; Shi, Y.; He, S.; Wang, H.; Hu, Z.; Gong, X.; Liang, X.; Yang, Y.; Liu, X. Mitochondrial DNA mutation exacerbates female reproductive aging via impairment of the NADH/NAD(+) redox. Aging Cell 2020, 19((9)), e13206. [Google Scholar] [CrossRef]
- Cotan, D.; Cordero, M. D.; Garrido-Maraver, J.; Oropesa-Avila, M.; Rodriguez-Hernandez, A.; Gomez Izquierdo, L.; De la Mata, M.; De Miguel, M.; Lorite, J. B.; Infante, E. R.; Jackson, S.; Navas, P.; Sanchez-Alcazar, J. A. Secondary coenzyme Q10 deficiency triggers mitochondria degradation by mitophagy in MELAS fibroblasts. FASEB J 2011, 25((8)), 2669–87. [Google Scholar] [CrossRef] [PubMed]
- Villanueva-Paz, M.; Povea-Cabello, S.; Villalon-Garcia, I.; Alvarez-Cordoba, M.; Suarez-Rivero, J. M.; Talaveron-Rey, M.; Jackson, S.; Falcon-Moya, R.; Rodriguez-Moreno, A.; Sanchez-Alcazar, J. A. Parkin-mediated mitophagy and autophagy flux disruption in cellular models of MERRF syndrome. Biochim Biophys Acta Mol Basis Dis 2020, 1866((6)), 165726. [Google Scholar] [CrossRef] [PubMed]
- Finsterer, J.; Jarius, C.; Eichberger, H. Phenotype variability in 130 adult patients with respiratory chain disorders. J Inherit Metab Dis 2001, 24((5)), 560–76. [Google Scholar] [CrossRef] [PubMed]
- Sezgin, E.; Levental, I.; Mayor, S.; Eggeling, C. The mystery of membrane organization: composition, regulation and roles of lipid rafts. Nat Rev Mol Cell Biol 2017, 18((6)), 361–374. [Google Scholar] [CrossRef]
- Buhaescu, I.; Izzedine, H. Mevalonate pathway: a review of clinical and therapeutical implications. Clin Biochem 2007, 40((9-10)), 575–84. [Google Scholar] [CrossRef]
- Duan, Y.; Gong, K.; Xu, S.; Zhang, F.; Meng, X.; Han, J. Regulation of cholesterol homeostasis in health and diseases: from mechanisms to targeted therapeutics. Signal Transduct Target Ther 2022, 7((1)), 265. [Google Scholar] [CrossRef]
- Rone, M. B.; Fan, J.; Papadopoulos, V. Cholesterol transport in steroid biosynthesis: role of protein-protein interactions and implications in disease states. Biochim Biophys Acta 2009, 1791((7)), 646–58. [Google Scholar] [CrossRef]
- Miller, W. L.; Auchus, R. J. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocr Rev 2011, 32((1)), 81–151. [Google Scholar] [CrossRef]
- Gerhold, J. M.; Cansiz-Arda, S.; Lohmus, M.; Engberg, O.; Reyes, A.; van Rennes, H.; Sanz, A.; Holt, I. J.; Cooper, H. M.; Spelbrink, J. N. Human Mitochondrial DNA-Protein Complexes Attach to a Cholesterol-Rich Membrane Structure. Sci Rep 2015, 5, 15292. [Google Scholar] [CrossRef] [PubMed]
- Goicoechea, L.; Conde de la Rosa, L.; Torres, S.; Garcia-Ruiz, C.; Fernandez-Checa, J. C. Mitochondrial cholesterol: Metabolism and impact on redox biology and disease. Redox Biol 2023, 61, 102643. [Google Scholar] [CrossRef] [PubMed]
- Wall, C. T. J.; Lefebvre, G.; Metairon, S.; Descombes, P.; Wiederkehr, A.; Santo-Domingo, J. Mitochondrial respiratory chain dysfunction alters ER sterol sensing and mevalonate pathway activity. J Biol Chem 2022, 298((3)), 101652. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Mao, C. C.; Reyes, A.; Sembongi, H.; Di Re, M.; Granycome, C.; Clippingdale, A. B.; Fearnley, I. M.; Harbour, M.; Robinson, A. J.; Reichelt, S.; Spelbrink, J. N.; Walker, J. E.; Holt, I. J. The AAA+ protein ATAD3 has displacement loop binding properties and is involved in mitochondrial nucleoid organization. J Cell Biol 2007, 176((2)), 141–6. [Google Scholar] [CrossRef]
- Chen, L.; Li, Y.; Zambidis, A.; Papadopoulos, V. ATAD3A: A Key Regulator of Mitochondria-Associated Diseases. Int J Mol Sci 2023, 24, 15. [Google Scholar] [CrossRef]
- Lee, S. R.; Han, J. Mitochondrial Nucleoid: Shield and Switch of the Mitochondrial Genome. Oxid Med Cell Longev 2017, 2017, 8060949. [Google Scholar] [CrossRef]
- Issop, L.; Fan, J.; Lee, S.; Rone, M. B.; Basu, K.; Mui, J.; Papadopoulos, V. Mitochondria-associated membrane formation in hormone-stimulated Leydig cell steroidogenesis: role of ATAD3. Endocrinology 2015, 156((1)), 334–45. [Google Scholar] [CrossRef]
- Goel, D.; Kumar, S. Advancements in unravelling the fundamental function of the ATAD3 protein in multicellular organisms. Adv Biol Regul 2024, 93, 101041. [Google Scholar] [CrossRef]
- Gunning, A. C.; Strucinska, K.; Munoz Oreja, M.; Parrish, A.; Caswell, R.; Stals, K. L.; Durigon, R.; Durlacher-Betzer, K.; Cunningham, M. H.; Grochowski, C. M.; Baptista, J.; Tysoe, C.; Baple, E.; Lahiri, N.; Homfray, T.; Scurr, I.; Armstrong, C.; Dean, J.; Fernandez Pelayo, U.; Jones, A. W. E.; Taylor, R. W.; Misra, V. K.; Yoon, W. H.; Wright, C. F.; Lupski, J. R.; Spinazzola, A.; Harel, T.; Holt, I. J.; Ellard, S. Recurrent De Novo NAHR Reciprocal Duplications in the ATAD3 Gene Cluster Cause a Neurogenetic Trait with Perturbed Cholesterol and Mitochondrial Metabolism. Am J Hum Genet 2020, 106((2)), 272–279. [Google Scholar] [CrossRef]
- Dorison, N.; Gaignard, P.; Bayot, A.; Gelot, A.; Becker, P. H.; Fourati, S.; Lebigot, E.; Charles, P.; Wai, T.; Therond, P.; Slama, A. Mitochondrial dysfunction caused by novel ATAD3A mutations. Mol Genet Metab 2020, 131((1-2)), 107–113. [Google Scholar] [CrossRef]
- Desai, R.; Frazier, A. E.; Durigon, R.; Patel, H.; Jones, A. W.; Dalla Rosa, I.; Lake, N. J.; Compton, A. G.; Mountford, H. S.; Tucker, E. J.; Mitchell, A. L. R.; Jackson, D.; Sesay, A.; Di Re, M.; van den Heuvel, L. P.; Burke, D.; Francis, D.; Lunke, S.; McGillivray, G.; Mandelstam, S.; Mochel, F.; Keren, B.; Jardel, C.; Turner, A. M.; Ian Andrews, P.; Smeitink, J.; Spelbrink, J. N.; Heales, S. J.; Kohda, M.; Ohtake, A.; Murayama, K.; Okazaki, Y.; Lombes, A.; Holt, I. J.; Thorburn, D. R.; Spinazzola, A. ATAD3 gene cluster deletions cause cerebellar dysfunction associated with altered mitochondrial DNA and cholesterol metabolism. Brain 2017, 140((6)), 1595–1610. [Google Scholar] [CrossRef] [PubMed]
- Peralta, S.; Goffart, S.; Williams, S. L.; Diaz, F.; Garcia, S.; Nissanka, N.; Area-Gomez, E.; Pohjoismaki, J.; Moraes, C. T. ATAD3 controls mitochondrial cristae structure in mouse muscle, influencing mtDNA replication and cholesterol levels. J Cell Sci 2018, 131, 13. [Google Scholar] [CrossRef] [PubMed]
- Kiesel, A. S.; Laugwitz, L.; Buchert, R.; Grimmel, M.; Baumann, S.; Sturm, M.; Reich, S.; Pauly, M. G.; Bruggemann, N.; Munchau, A.; Oleksiuk, O.; Synofzik, M.; Haack, T. B.; Peralta, S. Elevated cholesterol is a common phenotype for dominant and recessive ATAD3-associated disorders. Brain 2025, 148((4)), e24–e28. [Google Scholar] [CrossRef] [PubMed]
- Pesini, A.; Barriocanal-Casado, E.; Compagnoni, G. M.; Hidalgo-Gutierrez, A.; Yanez, G.; Bakkali, M.; Chhonker, Y. S.; Kleiner, G.; Larrea, D.; Tadesse, S.; Lopez, L. C.; Murry, D. J.; Di Fonzo, A.; Area-Gomez, E.; Quinzii, C. M. Coenzyme Q(10) deficiency disrupts lipid metabolism by altering cholesterol homeostasis in neurons. Free Radic Biol Med 2025, 229, 441–457. [Google Scholar] [CrossRef]
- Menacho, C.; Okawa, S.; Álvarez-Merz, I.; Wittich, A.; Muñoz-Oreja, M.; Lisowski, P.; Pentimalli, T. M.; Rybak-Wolf, A.; Inak, G.; Zakin, S.; Thevandavakkam, M.; Petersilie, L.; Zaliani, A.; Mlody, B.; Seibt, A.; Donnelly, J.; Woleben, K.; Fernandez-Checa, J.; Herebian, D.; Mayatepek, E.; Rajewsky, N.; Spinazzola, A.; Schuelke, M.; Perlstein, E.; Rossi, A.; Distelmaier, F.; Holt, I. J.; Pless, O.; Rose, O. R.; Del Sol, A.; Prigione, A. Deep learning-driven neuromorphogenesis screenings identify repurposable drugs for mitochondrial disease. Biorxiv preprint.
- van de Wal, M. A. E.; Doornbos, C.; Bibbe, J. M.; Homberg, J. R.; van Karnebeek, C.; Huynen, M. A.; Keijer, J.; van Schothorst, E. M.; t Hoen, P. A. C.; Janssen, M. C. H.; Adjobo-Hermans, M. J. W.; Wieckowski, M. R.; Koopman, W. J. H. Ndufs4 knockout mice with isolated complex I deficiency engage a futile adaptive brain response. Biochim Biophys Acta Proteins Proteom 2025, 1873((1)), 141055. [Google Scholar] [CrossRef]
- Ruiz, M.; Cuillerier, A.; Daneault, C.; Deschenes, S.; Frayne, I. R.; Bouchard, B.; Forest, A.; Legault, J. T.; Consortium, L.; Vaz, F. M.; Rioux, J. D.; Burelle, Y.; Des Rosiers, C. Lipidomics unveils lipid dyshomeostasis and low circulating plasmalogens as biomarkers in a monogenic mitochondrial disorder. JCI Insight 2019, 4((14)). [Google Scholar] [CrossRef]
- Thompson Legault, J.; Strittmatter, L.; Tardif, J.; Sharma, R.; Tremblay-Vaillancourt, V.; Aubut, C.; Boucher, G.; Clish, C. B.; Cyr, D.; Daneault, C.; Waters, P. J.; Consortium, L.; Vachon, L.; Morin, C.; Laprise, C.; Rioux, J. D.; Mootha, V. K.; Des Rosiers, C. A Metabolic Signature of Mitochondrial Dysfunction Revealed through a Monogenic Form of Leigh Syndrome. Cell Rep 2015, 13((5)), 981–9. [Google Scholar] [CrossRef]
- Cuillerier, A.; Honarmand, S.; Cadete, V. J. J.; Ruiz, M.; Forest, A.; Deschenes, S.; Beauchamp, C.; Consortium, L.; Charron, G.; Rioux, J. D.; Des Rosiers, C.; Shoubridge, E. A.; Burelle, Y. Loss of hepatic LRPPRC alters mitochondrial bioenergetics, regulation of permeability transition and trans-membrane ROS diffusion. Hum Mol Genet 2017, 26((16)), 3186–3201. [Google Scholar] [CrossRef]
- Bakare, A. B.; Lesnefsky, E. J.; Iyer, S. Leigh Syndrome: A Tale of Two Genomes. Front Physiol 2021, 12, 693734. [Google Scholar] [CrossRef]
- Lyu, J.; Zhao, Y.; Zhang, N.; Xu, X.; Zheng, R.; Yu, W.; Xin, W.; Yan, C.; Ji, K. Bezafibrate Rescues Mitochondrial Encephalopathy in Mice via Induction of Daily Torpor and Hypometabolic State. Neurotherapeutics 2022, 19((3)), 994–1006. [Google Scholar] [CrossRef]
- Frambach, S.; van de Wal, M. A. E.; van den Broek, P. H. H.; Smeitink, J. A. M.; Russel, F. G. M.; de Haas, R.; Schirris, T. J. J. Effects of clofibrate and KH176 on life span and motor function in mitochondrial complex I-deficient mice. Biochim Biophys Acta Mol Basis Dis 2020, 1866((6)), 165727. [Google Scholar] [CrossRef]
- Yatsuga, S.; Suomalainen, A. Effect of bezafibrate treatment on late-onset mitochondrial myopathy in mice. Hum Mol Genet 2012, 21((3)), 526–35. [Google Scholar] [CrossRef]
- Fournier, J.; Barret, L.; Khouri, C.; Naudet, F.; Boussageon, R.; Roustit, M. The evidence base of the 10 most prescribed drugs in England, France, and the United States: a scoping review. J Clin Epidemiol 2024, 174, 111478. [Google Scholar] [CrossRef]
| Mitochondrial disease | Gene | Model | Effect on Cholesterol | Citation |
| ATAD3-related pathologies |
ATAD3 | Patient-derived fibroblasts with a heterozygous ATAD3A variant and Drosophila carrying orthologous Atad3R472C mutation | ↑ free cholesterol ↓ Expression of proteins involved in cholesterol efflux (including ABCA1 and ABCG1) ↑ membrane-bound cholesterol levels ↑ reliance on dietary cholesterol |
[4] |
| Patient-derived fibroblasts, either with NAHR-mediated ATAD3A/C fusion gene lacking functional residues or bi-allelic ATAD3 cluster deletions | ↑ free cholesterol | [29] | ||
| Skeletal muscle-specific Atad3 cKO mice | ↓ cholesterol esters synthesized within ER ↑ dietary cholesterol esters ↓ cholesterol esters/free cholesterol ratio |
[32] | ||
| Patient-derived fibroblasts with biallelic deletions in ATAD3 gene cluster | ↑ free cholesterol ↑ expression of genes involved cholesterol biosynthesis pathway (such as SREBF2 and HMGCS1) and cholesterol efflux (ABCA1) • Decreasing cholesterol levels with cholesterol trafficking inhibitor or pravastatin increased mtDNA pathology |
[31] | ||
| Patient-derived fibroblasts with bi-allelic ATAD3A variants |
↑ free cholesterol ↑ expression of genes involved in cholesterol biosynthesis pathways |
[33] | ||
| Primary CoQ10 deficiency and Leigh Syndrome | PDSS2 | Patient-derived fibroblasts | ↓ cholesteryl esters (trend) ↓ expression of proteins involved in cholesterol biosynthesis (HMGCR) = Level of SREBP2 ↑ expression of proteins involved in cholesterol efflux (ABCA1) |
[34] |
| Leigh Syndrome |
SURF1 | Neural progenitor cells with homozygous SURF1 mutation | ↓ membrane cholesterol • Increasing membrane cholesterol associated with ameliorated disease phenotype |
[35] |
| NSDUF4 | Patient-derived fibroblasts and Nsduf4 KO mice | • Increasing cholesterol efflux and biosynthesis improved phenotype and increases lifespan of Nsduf4 KO mice | [7] | |
| Nsduf4 KO mice | ↑ Upregulated cholesterol biosynthesis pathways in cerebellum & hippocampus | [36] | ||
| Leigh Syndrome French Canadian |
LRPPRC | Plasma from patients | ↑ 1 cholesteryl ester | [37] |
| Plasma from patients | ↑ LDL cholesterol ↓ HDL cholesterol ↑ Total cholesterol/HDL cholesterol ratio |
[38] | ||
| Hepatocyte-specific Lrpprc cKO mice | ↓ mitochondrial membrane cholesterol | [39] | ||
| Ataxia with Oculomotor Apraxia Type 1 | APTX | Neurons derived from patient fibroblasts | ↓ cholesteryl esters ↑ expression of proteins involved in cholesterol biosynthesis (HMGCR, SREBP2) ↑ expression of proteins involved in cholesterol biosynthesis cholesterol efflux (ABCA1, ABCG1) |
[34] |
| Primary CoQ10 deficiency | COQ2 | ↓ expression of proteins involved in cholesterol biosynthesis (HMGCR) = Level of SREBP2 ↓ expression of proteins involved in cholesterol biosynthesis cholesterol efflux (ABCA1, ABCG1) |
||
| Mitochondrial Cardiomyopathy | C1QBP | Neuron-specific C1qbp cKO mice | ↓ brain expression of genes involved in cholesterol biosynthesis (including Srebf2, Hmgcr, Hmgcs1) | [6] |
| MNGIE | TYMP | Patient-derived fibroblasts | ↓ total cholesterol ↓ expression of protein involved in cholesterol biosynthesis (SREBP1, SREBP2) and efflux (ABCA1) |
[5] |
| MELAS | MT-TL1 (m.3243A > G) | = total cholesterol |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




